NMR studies of the interactions between AMB-1 Mms6 protein and magnetosome Fe3O4 nanoparticles†
Abstract
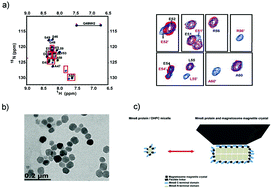
Mms6 protein from magnetotactic bacteria strain AMB-1 is responsible for controlling the formation of magnetite nanoparticles both in vitro and in vivo. High-resolution NMR studies showed the C-terminal DEEVE motif and the following residues undergoing conformation change upon magnetosome Fe3O4 crystal binding. The N-terminal hydrophobic packing of Mms6 protein is important for arranging the DEEVE motifs into a correct assembly and orientation that are crucial for magnetite crystal recognition.
- This article is part of the themed collection: International Year of the Periodic Table: Applications for magnetic materials


 Please wait while we load your content...
Please wait while we load your content...