Multifunctional ammonium salts synergizing etching and passivation capability enable efficient deep-blue CsPbBr3 nanoplates†
Yanyin
Wang
ab,
Shirong
Wang
 ab,
Zhanpeng
Qin
ab,
Jing
Zhou
ab,
Sixiao
Gu
ab,
Xianggao
Li
ab,
Zhanpeng
Qin
ab,
Jing
Zhou
ab,
Sixiao
Gu
ab,
Xianggao
Li
 ab and
Hongli
Liu
ab and
Hongli
Liu
 *ab
*ab
aTianjin University, School of Chemical Engineering and Technology, Tianjin 300072, China. E-mail: liuhongli@tju.edu.cn
bCollaborative Innovation Center of Chemical Science and Engineering (Tianjin), Tianjin 300072, China
First published on 17th May 2024
Abstract
Two-dimensional CsPbBr3 nanoplates (NPLs) are highly promising blue emitters due to their precisely tunable emission wavelength by adjusting the thickness. However, synthesizing high-performance deep-blue emissive CsPbBr3 NPLs with fewer monolayers remains challenging. Here, multifunctional ligands of 2,6-diaminopyridinium hydrobromide (DaPyBr) with multiple coordination sites were designed and synthesized to attenuate the thickness of blue CsPbBr3 NPLs and suppress surface defects. DaPyBr serving both as a Lewis acid and a Lewis base possesses the dual functions of etching and passivation. It can detach the outer defect layers of CsPbBr3 NPLs reducing the thickness to 3 monolayers and passivate inner [PbBr6]4− layers suppressing the surface defects. Ultimately, CsPbBr3 NPLs treated by DaPyBr achieved a near-unity photoluminescence quantum yield at 456 nm. The target NPLs gained long-term storage stability and outstanding resistance to polar solvents. A bright deep-blue pattern display with commendable luminescence stability is achieved via aerosol printing technology.
Introduction
Metal halide perovskites have gained considerable attention as an emerging class of emitter materials for next-generation lighting and display technologies.1–4 Typically, two-dimensional CsPbBr3 nanoplates (NPLs) bearing a narrow photoluminescence full-width at half-maximum (FWHM), large exciton binding energies, and thickness-tunable emission merits exhibit high potential for spectral-stable emitters.5–12 Blue-emitting CsPbBr3 NPLs often require more organic ligands to stabilize the nanocrystals (NCs) due to their large surface area and high surface energy.11,13 However, because of the weak absorption ability of traditional ligands of oleic acid and oleyl amine (OAm and OA), most NPLs often suffer from numerous halides and Pb2+ vacancies, resulting in poor stability and photoluminescence quantum yield (PLQY).7,12,14–16To address these issues, Zeng's group introduced HBr in the preparation of CsPbBr3 NPLs, which could reduce bromine vacancies and suppress nonradiative recombination, achieving a high PLQY for pure blue-emitting NPLs.17 Chen et al. utilized a novel dual-site ligand 4-aminobenzenesulfonic acid to reduce the trap state density of NPLs and obtained pure-blue (460 nm) NPLs with 99.8% PLQY.18 Besides, Rogach et al. employed a multidentate ligand polyethyleneimine as a soft template to effectively hinder the merging of NPLs and lower the vacancy defect density.19 Based on this, high PLQY of 85% for blue emission in solution was achieved and the stability of the NPLs was enhanced. Most passivation strategies are typically viable in blue light emission, while replacing the original ligands with short ligands is not suitable for deep-blue NPLs (<460 nm). This is because emission red-shift or aggregation will occur since the one dimension of deep-blue NPLs is just a few single monolayers, which arises even larger surface area and higher surface energy.19,20
In this work, multifunctional ligands of ammonium salts (DaPyBr) with multiple coordination sites were designed and synthesized, which enables the attenuation of the thickness of blue CsPbBr3 NPLs to a few molecular layers and suppresses the surface defects. DaPyBr features polyfunctional groups that could serve both as a Lewis acid and a Lewis base.21 They can detach the outer defect layers of NPLs and the thickness was reduced from 5 monolayers (MLs) to 3 MLs, exhibiting a blue shift in the PL spectrum. Meanwhile, inner [PbBr6]4− layers were completely passivated by DaPyBr. During this process, NPLs underwent crystal regrowth, leading to a more organized arrangement and improved crystallinity. Ultimately, surface defects were reduced and a near-unity PLQY of NPLs with CIE coordinates of (0.1452, 0.0394) was achieved. Due to the strong affinity between DaPyBr and the surface, the treated NPLs exhibited satisfactory long-term storage stability in ambient air, retaining 80% of the initial PL intensity for 45 days. Moreover, the treated NPLs exhibited favorable resistance to ethyl alcohol. These outcomes facilitate the realization of a bright and clear deep-blue pattern display that depends on aerosol printing technology, thereby advancing the development of high-quality deep-blue perovskites in display applications.
Results and discussion
Mechanism explanation
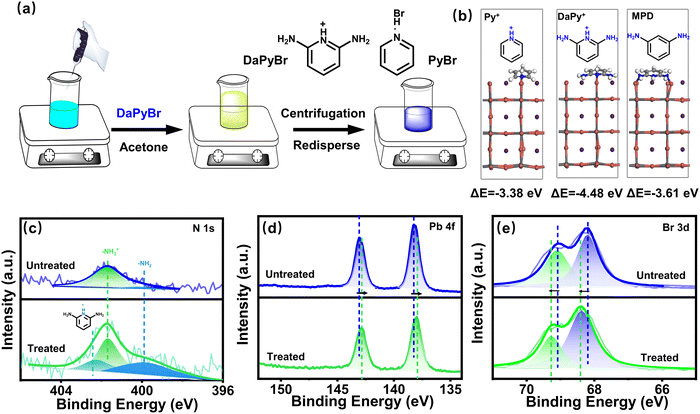
CsPbBr3 NPLs are synthesized using a room temperature method reported by Bohn.22 Herein, 2,6-diaminopyridinium hydrobromide (DaPyBr) was selected as a ligand to treat NPLs, which are denoted as DaPyBr-NPLs. DaPyBr was synthesized and the proton nuclear magnetic resonance (1H NMR) spectrum was conducted to elucidate its chemical structure. As shown in Fig. S1 (ESI†), a chemical shift at 12.19 ppm corresponding to the protonated NH of the pyridine ring was observed, indicating that the DaPyBr was prepared successfully. DaPyBr was introduced during the process of the addition of acetone into the CsPbBr3 NPL solution. The treated NPLs were obtained through centrifugation and dispersion after stirring for 30 minutes, as illustrated in Fig. 1(a). Density functional theory (DFT) was conducted to evaluate the interaction force between DaPyBr and the NPL surface (Fig. 1(b)). The binding energies (ΔE) between the N of pyridine (Py+) and ortho ammonium group (MPD) with NPLs are −3.38 and −3.61 eV, respectively. While the collaborative binding energy between DaPy+ and NPLs is −4.48 eV, which is much larger than that of OA-NPLs or OAm-NPLs (−0.75 and −0.86 eV, respectively).23X-ray photoelectron spectroscopy (XPS), including the full spectrum (Fig. S2 and S3, ESI†) and high-resolution spectrum was conducted to investigate the surface element composition, binding energy, and chemical state of NPLs with and without DaPyBr treated. As displayed in Fig. 1(c), the N 1s spectrum of the untreated CsPbBr3 NPL can be fitted with two components at 399.98 and 401.74 eV, corresponding to the terminal amine groups (–NH2) and protonated amine groups (–NH3+), respectively. In contrast, a new peak at 402.3 eV corresponding to the N of the pyridine ring appeared in DaPyBr-NPLs, confirming the existence of DaPyBr. Moreover, Fig. S4 (ESI†) shows the integrated areas of the C and N peaks of the control, PyBr-NPL and DaPyBr-NPL. According to the value of the integrated areas of the C and N peaks, the atomic concentration of N (nN) was calculated to be 0.47 of the control, 0.60 of PyBr-NPL, and 0.65 of DaPyBr-NPL, respectively by eqn (S1) (ESI†). The increased nN value from 0.47 to 0.65 indicated that DaPyBr could effectively interact with the surface and adsorb on it. Fig. 1(d) reveals that the Pb 4f spectrum of DaPyBr-NPLs with Pb 4f5/2 (142.84 eV) and 4f7/2 (138.17 eV) shifted towards a lower energy field. The shift of binding energy indicated a more stable Pb–DaPy+ bond formation compared to Pb–Br or Pb-oleate.18 Furthermore, the Br 3d spectrum of DaPyBr-NPLs (Fig. 1(e)) exhibited a peak shift toward a higher energy field, which announces that the electron cloud of Br transferred more to the ligand on the surface, indicative of strong interaction between DaPyBr and NPLs. The introduction of DaPyBr could strongly coordinate with Pb on the surface of NPLs and even remove some incomplete coordinated Pb to achieve surface etching as discussed in the following.
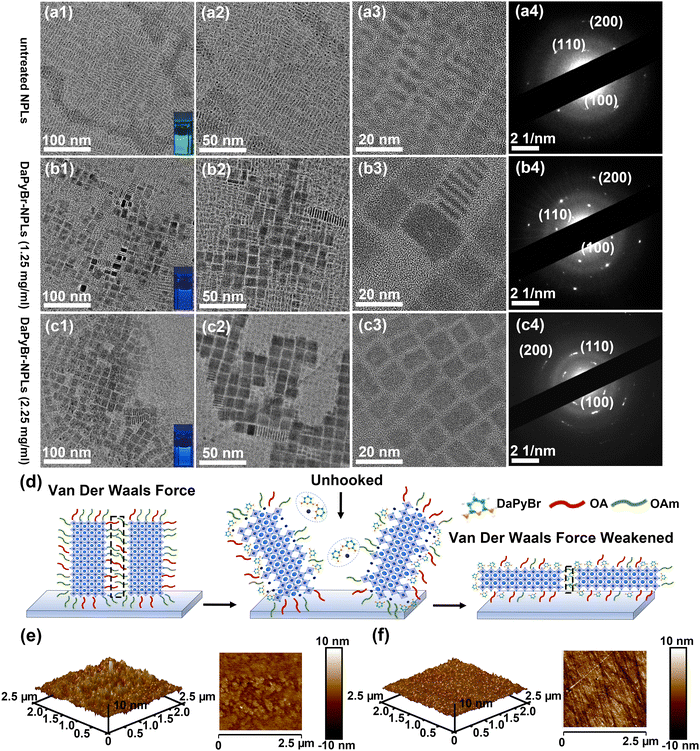
Transmission electron microscopy (TEM) was conducted to characterize the morphology and crystal structure of CsPbBr3 NPL. The NPL morphology with different resolution is demonstrated in Fig. 2(a1)–(a3)–(c1)–(c3) respectively for pristine NPL, and DaPyBr-NPLs with varied concentration. The thickness of the NPLs (Fig. S5, ESI†) was calculated by particle size statistical analysis from the random selection of 30 samples in selected areas. The untreated NPLs showed a homogeneous thickness of ≈2.76 nm, which corresponds to 5 MLs. With the addition of DaPyBr, the thickness of the DaPyBr-NPLs gradually decreased to 4 MLs (≈2.29 nm) and ultimately stabilized at 3 MLs (≈1.65 nm), suggesting an etching effect of DaPyBr to NPLs. It can also be observed that the pristine NPLs stacked face to face, standing upright on the carbon film. For DaPyBr-NPLs, partial NPLs transformed into a flat-lying arrangement. The majority of DaPyBr-NPLs displayed a uniform and flat arrangement with consistent orientation when the concentration of DaPyBr was 2.25 mg ml−1. The transformation was attributed to the weakened van der Waals interaction forces between the NPLs, stemming from the original long-chain ligands (OAm and OA).23 As a result, the face-to-face stacked configuration could not be maintained, suggesting that some original ligands were partially replaced by DaPyBr. Meanwhile, the length of the NPLs (Fig. S6, ESI†) was calculated by particle size statistical analysis from a random selection of 25 samples in Fig. 2(a2)–(c2), respectively. The untreated NPLs showed a homogeneous length of ≈12.55 nm, while the lengths of DaPyBr-NPLs with concentrations of 1.25 and 2.25 mg ml−1 were measured to be 16.02 nm and 18.21 nm, respectively. The increased length of the DaPyBr-NPLs suggested a crystal growth process, promoting improved crystallinity and a more uniform size distribution.
The morphological transformation would inevitably impact other properties of the thin film, such as crystallinity and roughness. The selected-area electron diffraction (SADE) patterns were employed to observe the crystal structure of the NPLs. As shown in Fig. 2(a4)–(c4), three resolved diffraction rings correspond to the (100), (110), and (200) crystal planes of CsPbBr3, respectively. Meanwhile, X-ray diffraction (XRD) patterns of the NPLs and DaPyBr-NPLs with different additions of NPLs were displayed in Fig. S7 (ESI†). The diffraction peaks that appeared at around 15, 21, and 30° correspond to (100), (110), and (200) planes of orthorhombic CsPbBr3 (JCPDF #01-072-7929), respectively. These phenomena confirmed that the crystal structure can be retained with DaPyBr treatment. In addition, the intensity of the diffraction ring and the relative intensity of the (110) plane increased obviously for DaPyBr-NPLs, signifying improved crystallinity with a more exposed surface. Meanwhile, atomic force microscopy (AFM) characterization was conducted to further verify the morphological changes with DaPyBr treatment. As illustrated in Fig. 2(e) and (f), the rough mean square (RMS) of DaPyBr-NPLs was just 0.626 nm, which was much lower than the untreated NPLs (1.44 nm). This was attributed to the transformation of the morphology from the face-to-face stacked configuration to uniformly flat-lying NPLs. The enhanced crystallinity and flatness of the film resulting from crystal regrowth were beneficial for the improvement of the optical properties24 and offered the possibility of forming a luminous layer of high quality in optical display.
Optical performance
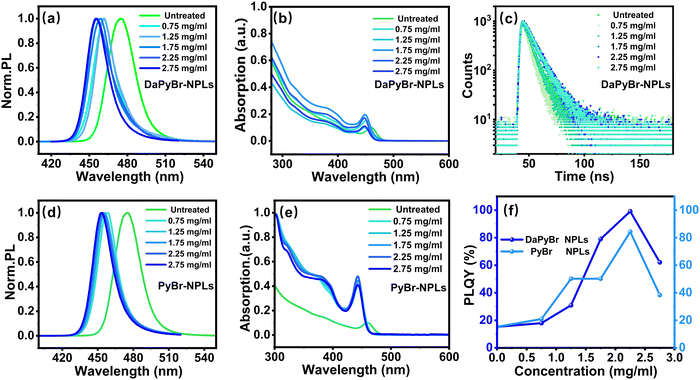
The optical properties were further explored to investigate the mechanism of DaPyBr on the NPL surface. PL and UV-vis absorption spectra of NPLs with different DaPyBr concentrations are plotted in Fig. 3(a) and (b). The untreated NPLs exhibited an emission peak at 474 nm and the central peak position blue-shifted from 461 to 455 nm when DaPyBr increased from 0.75 to 2.75 mg ml−1. The treatment of DaPyBr facilitated precise tuning of the spectral range of NPLs from blue to deep-blue emission (Fig. S8, ESI†). Additionally, the peak of the UV-vis absorption spectra shifted from 462 to 447 nm with the increase of the DaPyBr concentration. The blue shift in the PL and UV-vis absorption spectra can be ascribed to the thickness attenuation of DaPyBr-NPLs as verified in TEM characterization.Moreover, the PLQY of the DaPyBr-NPLs exhibited a trend of first increasing and then decreasing with the increase of DaPyBr (Fig. 3(c)). Notably, deep-blue emitting CsPbBr3 NPLs achieved a near-unity PLQY at 456 nm as DaPyBr was 2.25 mg ml−1. As DaPyBr further increased to 2.75 mg ml−1, the PLQY of the DaPyBr-NPLs suffered a decrease (Table S1, ESI†). This can be attributed to the fact that the outer layers were completely peeled off and the internal structure of the NPLs would subject to further adverse etching. Time-resolved photoluminescence (TRPL) decay curves with different concentrations of DaPyBr were fitted to gain insight into the carrier dynamics of the DaPyBr-NPLs (Fig. 3(c)). The decay curves showed good bi-exponential fitted results.25 The untreated NPLs had an average PL lifetime of 3.68 ns, while the lifetime of the DaPyBr-NPLs decreased at low concentrations, but gradually increased with concentration, reaching the maximum at 2.25 mg ml−1 (7.62 ns). This phenomenon indicated that at low concentrations, DaPyBr primarily etched the surface of the NPLs, leading to more defective layers and non-radiative recombination centers. When the concentration of DaPyBr gradually increased, the excess DaPyBr can bind with NPLs and passivate the surface, reducing defect-related non-radiative trap centers. However, with a further increase in concentration, the internal structure of the NPLs would be subject to further adverse etching, resulting in a reduction in lifetime, consistent with the PLQY results. The effective passivation of the NPL surface was achieved at a DaPyBr concentration of 2.25 mg ml−1, indicating that 2.25 mg ml−1 was the optimal concentration.
To deeply confirm the passivation effect of DaPyBr on the NPL surface, pyridinium hydrobromide (PyBr) was employed to treat CsPbBr3 NPLs with the same method (denoted as PyBr-NPLs). As depicted in Fig. 3(d) and (e), both the PL and UV-vis absorption peaks of PyBr-NPLs exhibited a blue shift with the addition of PyBr, which is similar to DaPyBr-NPLs. Notably, in the case of the same PyBr and DaPyBr, PyBr-NPLs displayed a greater extent of blue shift than that of DaPyBr-NPLs, announcing a stronger etching ability of PyBr.
Similar to the etching effect observed with DaPyBr, the thickness of the PyBr-NPLs decreased from 2.76 nm to 1.60 nm and further to 1.42 nm (Fig. S9, ESI†). The change of length with different concentrations (Fig. S10, ESI†) indicated a process of crystal growth, leading to a regular size distribution and higher crystallinity. When thickness was reduced to the same, the XRD pattern (Fig. S11, ESI†) of both DaPyBr-NPL and PyBr-NPL exhibited (100), (110) and (200) crystal planes, and the corresponding peak intensities were similar. This indicated that DaPyBr-NPL and PyBr-NPL underwent analogous etching and regrowth process. However, the PLQY and TRPL results of PyBr-NPL and DaPyBr-NPL were different, as shown in Tables S1 and S2 (ESI†). For example, the PLQY of DaPyBr-NPL at 456 nm is 99.16%, significantly higher than that of PyBr-NPLs at 456 nm (50.25%). The enhancement of PLQY and interaction between DaPyBr and NPLs (verified by XPS mentioned above) suggested that the –NH2 group in DaPyBr can effectively passivate defects. Moreover, the TRPL test of DaPyBr-NPLs and PyBr-NPLs (Fig. S12, ESI†) further proved this result. With the same emission wavelength (456 nm), the average PL lifetime of DaPyBr-NPL (7.62 ns) is longer than that of the PyBr-NPLs (5.62 ns), indicating that defect-related non-radiative trap centers of DaPyBr-NPL were effectively passivated.
Stability of DaPyBr-NPLs
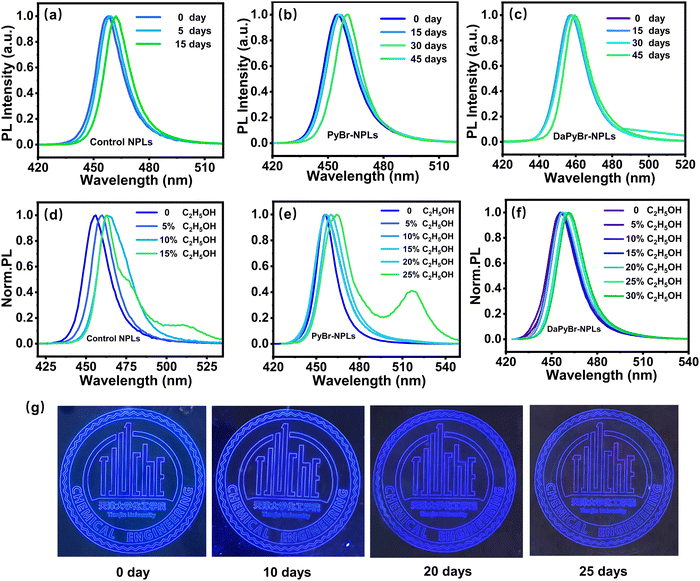
The long-term stability of control NPLs, PyBr-NPLs and DaPyBr-NPLs with the same emission peak was measured by storing them in ambient air conditions (30% relative humidity). Fig. 4(a) displays that control NPLs suffered an apparent PL redshift of 6 nm only after 15 days. PyBr-NPLs experienced the same redshift at 45 days later, as revealed in Fig. 4(b). In contrast, even after 45 days had passed, the DaPyBr-NPLs only showed a redshift of 4 nm (Fig. 4(c)), indicative of preferable stability. Moreover, DaPyBr-NPLs could retain 80% of the initial PL intensity even after 45 days, as supported in Fig. S13 (ESI†). The long-term stability of the DaPyBr-NPLs could be attributed to the fact that DaPyBr with multi binding sites possesses high ΔE with NPLs and is thus hard to detach.Furthermore, the stability against polar solvents of NPLs was also tested by treating the samples with absolute ethanol. As revealed in Fig. 4(d), the PL peak of the control NPLs exhibited a red shift of 9 nm and three peaks appeared at 464, 477 and 511 nm, respectively, with just a tiny amount of alcohol (15%). This was probably caused by the OH− group in the alcohol, which can destroy the integrity of the OA and OAm ligands. And then, OA and OAm can easily detach from the surface upon polar solvent treatment, leading to the aggregation and decomposition of the NPLs.26–28 Relatively, PyBr-NPLs underwent a red shift of 9 nm and exhibited two peaks at 465 nm and 517 nm until the addition of alcohol was 25% (Fig. 4(e)). This is because the binding energy between PyBr and NPLs (−3.38 eV) was higher than that of OA (−0.75 eV) and OAm (−0.86 eV). Hence, more alcohols are needed to destroy the integrity of the ligands. On the contrary, DaPyBr-NPLs still maintained a single peak at 461 nm and only showed a red shift of 5 nm even with 30% alcohol (Fig. 4(f)). Subsequently, NPLs were purified and then treated with absolute ethanol to further elucidate their stability. As Fig. S14 (ESI†) shows that the PL of the control NPLs underwent a notable red shift of 18 nm, indicating that the crystals were damaged and aggregated. In contrast, both PyBr-NPL and DaPyBr-NPL exhibited a minimal red shift of only 3 nm to 459 nm. Particularly for DaPyBr-NPL, the purified NPLs maintained a single peak at 459 nm, even when subjected to 25% alcohol. These results confirmed that the multiple functional groups of DaPyBr (Py+ and ortho-ammonium group) were capable of binding with NPLs, resulting in stronger interactions and making them less prone to detachment from the surface in highly polar environments. And thus, alcohols have poor ability to compel DaPyBr to detach from NPL, which maintains the structural integrity of the PQDs.
Based on the superior optical properties and satisfactory stability, DaPyBr-NPLs demonstrated their potential for display applications. The pattern display device with DaPyBr-NPLs was fabricated by a novel aerosol spray printing technology, demonstrating a bright and clear deep-blue display under ultraviolet light, as exhibited in Fig. 4(g). The pattern also demonstrated excellent optical stability at room temperature in an atmospheric environment, maintaining a notably deep-blue luminance even after 25 days. This pattering involved a simple and low-cost process, which was an innovative step to incorporate NPLs into display, anti-counterfeiting, and optical chip applications.
Conclusion
Herein, ammonium salts (DaPyBr) with multiple coordination sites were designed and synthesized to enable efficient deep-blue CsPbBr3 NPLs by the merits of their dual functions of etching and passivation. With the treatment of DaPyBr, the outer defect layers of NPLs would be peeled off and simultaneously, the inner [PbBr6]4− layer could be passivated entirely. Consequently, the thickness of the NPLs was attenuated to 3 MLs, and surface defects were effectively suppressed. The process is accompanied by crystal growth, which improved the arrangement and crystallinity of the NPLs, further reducing defects. Resultantly, a near-unity PLQY of deep-blue CsPbBr3 DaPyBr-NPLs with CIE coordinates of (0.1452, 0.0394) was obtained. Moreover, DaPyBr-NPLs gained satisfactory long-term storage stability and polar solvent resistance capability. Ultimately, a bright and clear deep-blue pattern display was fabricated and maintained brightness for 25 days. Our work highlights the critical role of the dual effects of ligand etching and passivation on the high luminescence stability NPLs and their display applications.Author contributions
Y. Y. Wang performed investigation, executed experiments, performed data analysis and interpretation, wrote, and revised the original manuscript. Z. P. Qin revised and edited the manuscript. J. Zhou and S. X. Gu performed investigation, designed the experimental methodology. H. L. Liu performed conceptualization, supervision, investigation, revised and edited the manuscript. S. R. Wang and X. G. Li performed conceptualization, funding acquisition, and supervision.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the National Key Research and Development Program of China (2022YFB3603003).References
- X. M. Li, Y. Wu, S. L. Zhang, B. Cai, Y. Gu, J. Z. Song and H. B. Zeng, Adv. Funct. Mater., 2016, 26, 2435–2445 CrossRef CAS.
- F. Zhang, H. Z. Zhong, C. Chen, X. G. Wu, X. M. Hu, H. L. Huang, J. B. Han, B. S. Zou and Y. P. Dong, ACS Nano, 2015, 9, 4533–4542 CrossRef CAS PubMed.
- J. Z. Song, J. H. Li, X. M. Li, L. M. Xu, Y. H. Dong and H. B. Zeng, Adv. Mater., 2015, 27, 7162 CrossRef CAS PubMed.
- L. Protesescu, S. Yakunin, M. I. Bodnarchuk, F. Krieg, R. Caputo, C. H. Hendon, R. X. Yang, A. Walsh and M. V. Kovalenko, Nano Lett., 2015, 15, 3692–3696 CrossRef CAS PubMed.
- Y. Bekenstein, B. A. Koscher, S. W. Eaton, P. D. Yang and A. P. Alivisatos, J. Am. Chem. Soc., 2015, 137, 16008–16011 CrossRef CAS PubMed.
- J. A. Sichert, Y. Tong, N. Mutz, M. Vollmer, S. Fischer, K. Z. Milowska, R. G. Cortadella, B. Nickel, C. Cardenas-Daw, J. K. Stolarczyk, A. S. Urban and J. Feldmann, Nano Lett., 2015, 15, 6521–6527 CrossRef CAS PubMed.
- M. C. Weidman, M. Seitz, S. D. Stranks and W. A. Tisdale, ACS Nano, 2016, 10, 7830–7839 CrossRef CAS PubMed.
- J. Shamsi, Z. Y. Dang, P. Bianchini, C. Canale, F. Di Stasio, R. Brescia, M. Prato and L. Manna, J. Am. Chem. Soc., 2016, 138, 7240–7243 CrossRef CAS PubMed.
- M. C. Weidman, A. J. Goodman and W. A. Tisdale, Chem. Mater., 2017, 29, 5019–5030 CrossRef CAS.
- J. Liu, Y. Xue, Z. Wang, Z.-Q. Xu, C. Zheng, B. Weber, J. Song, Y. Wang, Y. Lu and Y. Zhang, ACS Nano, 2016, 10, 3536–3542 CrossRef CAS PubMed.
- L. T. Dou, A. B. Wong, Y. Yu, M. L. Lai, N. Kornienko, S. W. Eaton, A. Fu, C. G. Bischak, J. Ma, T. N. Ding, N. S. Ginsberg, L. W. Wang, A. P. Alivisatos and P. D. Yang, Science, 2015, 349, 1518–1521 CrossRef CAS PubMed.
- V. A. Hintermayr, A. F. Richter, F. Ehrat, M. Döblinger, W. Vanderlinden, J. A. Sichert, Y. Tong, L. Polavarapu, J. Feldmann and A. S. Urban, Adv. Mater., 2016, 28, 9478 CrossRef CAS PubMed.
- C. Otero-Martínez, J. Z. Ye, J. Sung, I. Pastoriza-Santos, J. Pérez-Juste, Z. G. Xia, A. Rao, R. L. Z. Hoye and L. Polavarapu, Adv. Mater., 2022, 34, 2107105 CrossRef PubMed.
- P. Tyagi, S. M. Arveson and W. A. Tisdale, J. Phys. Chem. Lett., 2015, 6, 1911–1916 CrossRef CAS PubMed.
- Q. A. Akkerman, S. G. Motti, A. R. S. Kandada, E. Mosconi, V. D’Innocenzo, G. Bertoni, S. Marras, B. A. Kamino, L. Miranda, F. De Angelis, A. Petrozza, M. Prato and L. Manna, J. Am. Chem. Soc., 2016, 138, 1010–1016 CrossRef CAS PubMed.
- V. K. Ravi, A. Swarnkar, R. Chakraborty and A. Nag, Nanotechnology, 2016, 27, 325708 CrossRef PubMed.
- Y. Wu, C. Wei, X. Li, Y. Li, S. Qiu, W. Shen, B. Cai, Z. Sun, D. Yang and Z. Deng, ACS Energy Lett., 2018, 3, 2030–2037 CrossRef CAS.
- W. Shen, Y. Yu, W. Z. Zhang, Y. F. Chen, J. B. Zhang, L. Yang, J. T. Feng, G. Cheng, L. H. Liu and S. F. Chen, ACS Appl. Mater. Interfaces, 2022, 14, 5682–5691 CrossRef CAS PubMed.
- W. Yin, M. Li, W. Dong, Z. Luo, Y. Li, J. Qian, J. Zhang, W. Zhang, Y. Zhang and S. V. Kershaw, ACS Energy Lett., 2021, 6, 477–484 CrossRef CAS.
- D. Yang, Y. T. Zou, P. L. Li, Q. P. Liu, L. Z. Wu, H. C. Hu, Y. Xu, B. Q. Sun, Q. Zhang and S. T. Lee, Nano Energy, 2018, 47, 235–242 CrossRef CAS.
- T. Xia, B. Jiang, W. T. Liu, X. Y. Li, H. Y. Dong, N. Tian, G. Y. Zheng, Y. Peng, D. S. Yao and F. Long, Appl. Surf. Sci., 2023, 608, 155042 CrossRef CAS.
- B. J. Bohn, Y. Tong, M. Gramlich, M. L. Lai, M. Döblinger, K. Wang, R. L. Z. Hoye, P. Müller-Buschbaum, S. D. Stranks, A. S. Urban, L. Polavarapu and J. Feldmann, Nano Lett., 2018, 18, 5231–5238 CrossRef CAS PubMed.
- J. K. Liu, K. P. Song, Y. Shin, X. Liu, J. Chen, K. X. Yao, J. Pan, C. Yang, J. Yin, L. J. Xu, H. Z. Yang, A. M. El-Zohry, B. Xin, S. Mitra, M. N. Hedhi, I. S. Roqan, O. F. Mohammed, Y. Han and O. M. Bakr, Chem. Mater., 2019, 31, 6642–6649 CrossRef CAS.
- M. Q. Zhang, C. H. Bi, Y. X. Xia, X. J. Sun, X. Y. Wang, A. Q. Liu, S. Y. Tian, X. F. Liu, N. H. de Leeuw and J. J. Tian, Angew. Chem., Int. Ed., 2023, 62, e202300149 CrossRef CAS PubMed.
- K. B. Zheng, K. Zídek, M. Abdellah, M. E. Messing, M. J. Al-Marri and T. Pullerits, J. Phys. Chem. C, 2016, 120, 3077–3084 CrossRef CAS.
- Y. Kim, E. Yassitepe, O. Voznyy, R. Comin, G. Walters, X. W. Gong, P. Kanjanaboos, A. F. Nogueira and E. H. Sargent, ACS Appl. Mater. Interfaces, 2015, 7, 25007–25013 CrossRef CAS PubMed.
- S. Q. Huang, Z. C. Li, B. Wang, N. W. Zhu, C. Y. Zhang, L. Kong, Q. Zhang, A. D. Shan and L. Li, ACS Appl. Mater. Interfaces, 2017, 9, 7249–7258 CrossRef CAS PubMed.
- Y. F. Sun, H. D. Zhang, K. Zhu, W. G. Ye, L. S. She, X. M. Gao, W. Y. Ji and Q. H. Zeng, RSC Adv., 2021, 11, 27333–27337 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Fig. S1: NMR (nuclear magnetic resonance) hydrogen spectrum of DaPyBr; Fig. S2: XPS spectrum of the untreated NPLs; Fig. S3: XPS spectrum of DaPyBr-NPLs; Fig. S4: (a), (c), (e) high-resolution XPS spectra of C 1 of control, PyBr-NPL and DaPyBr-NPL: (b), (d), (f) high-resolution XPS spectra of N 1 of control, PyBr-NPL and DaPyBr-NPL; Fig. S5: the selected regions for particle size statistical analysis and their corresponding data results for untreated NPLs and DaPyBr-NPLs with different concentrations; Fig. S6: distribution diagram of the length of the untreated NPLs and DaPyBr-NPLs with different concentrations; Fig. S7: XRD patterns of the untreated CsPbBr3 NPLs and different concentrations of DaPyBr; Fig. S8: NPLs of different DaPyBr concentration emission comparison under UV light; Fig. S9: TEM images and the selected-area particle size statistical analysis of the untreated NPLs (a), 1.25 mg ml−1 PyBr-NPLs (b) and 2.25 mg ml−1 PyBr-NPLs (c) with different resolution (100 nm, 50 nm and 20 nm); Fig. S10: distribution diagram of the length of the untreated NPLs and PyBr-NPLs with different concentrations; Fig. S11: comparison of XRD spectra of DaPyBr-NPL and PyBr-NPL with the same thickness (3MLs); Fig. S12: time-resolved photoluminescence of PyBr-NPLs with various concentrations; Fig. S13: the luminescence comparison after 45 days under UV light; Fig. S14: changes in fluorescence emission spectra with varying ethanol concentrations of control NPLs (a) PyBr-NPLs (b) DaPyBr-NPLs (c) after purification. See DOI: https://doi.org/10.1039/d4tc01261e |
| This journal is © The Royal Society of Chemistry 2024 |