Crucial breakthrough of second near-infrared biological window fluorophores: design and synthesis toward multimodal imaging and theranostics
Shuqing
He
 ab,
Jun
Song
ab,
Jun
Song
 *a,
Junle
Qu
*a,
Junle
Qu
 *a and
Zhen
Cheng
*a and
Zhen
Cheng
 *b
*b
aKey Laboratory of Optoelectronic Devices and Systems of Ministry of Education and Guangdong Province, College of Optoelectronic Engineering, Shenzhen University, Shenzhen 518060, P. R. China. E-mail: songjun@szu.edu.cn; jlqu@szu.edu.cn
bMolecular Imaging Program at Stanford (MIPS), Bio-X Program, and Department of Radiology, Canary Center at Stanford for Cancer Early Detection, Stanford University, Stanford, California 94305-5344, USA. E-mail: zcheng@stanford.edu
First published on 4th May 2018
Abstract
The development of fluorophores and molecular probes for the second near-infrared biological window (NIR-II, 1000–1700 nm) represents an important, newly emerging and dynamic field in molecular imaging, chemical biology and materials chemistry. Because of reduced scattering, minimal absorption and negligible autofluorescence, NIR-II imaging provides high resolution, a high signal-to-noise ratio, and deep tissue penetration capability. Among various state-of-the-art bioimaging modalities, one of the greatest challenges in developing novel probes is to achieve both high resolution and sensitivity. The chemical design and synthesis of NIR-II fluorophores suitable for multimodal imaging is thus emerging as a new and powerful strategy for obtaining high-definition images. NIR-II fluorophores may convert NIR-II photons into heat for photothermal therapy and be excited by NIR-II light to produce singlet oxygen for photodynamic therapy. The presence of simultaneous diagnostic and therapeutic capabilities in a single probe can be used for precise treatment. In this review, we have focused on recent advances in the chemical design and synthesis of NIR-II fluorophores from small organic molecules to organic and inorganic nanoparticles, and we have further discussed recent advances and key operational differences in reported NIR-II imaging systems and biomedical applications based on NIR-II imaging, such as multimodal imaging, photothermal and photodynamic therapy, guidance for intraoperative surgery, and drug delivery.
Key learning points(1) Recent advances in chemical strategies for designing and synthesizing NIR-II fluorophores(2) The power of chemical architecture in NIR-II probes for multimodal imaging techniques (3) Analysis at the cellular level using structure–function relationships between NIR-II probes and biological systems (4) Diversity-oriented NIR-II fluorophores as an alternative to conventional rational design in theranostics (5) Perspectives and challenges in applications of NIR-II fluorophores |
1. Introduction
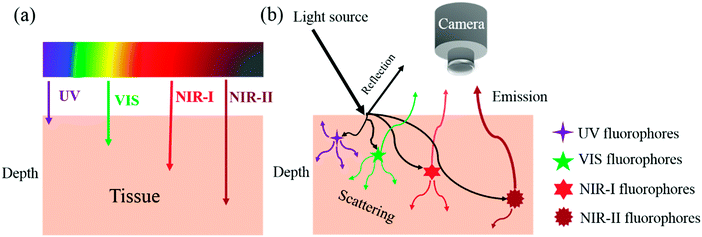
Fluorescence imaging does not require ionizing radiation and can be developed as a non-invasive or minimally invasive technique. It can be conveniently used for the investigation of biological phenomena and, importantly, enables the real-time acquisition of wide-field images with a high signal-to-noise ratio and excellent spatial and temporal resolution. Key issues in fluorescence imaging include autofluorescence, quenching, photobleaching and a low depth of tissue penetration. In comparison with fluorescence imaging in the visible region (400–700 nm), fluorescence imaging in the near-infrared (NIR) window (700–1700 nm) offers considerable advantages in terms of reduced photon scattering, lower absorption and minimal interference from autofluorescence (Scheme 1). NIR imaging enables high resolution and a high signal-to-noise ratio and has tremendous potential in molecular diagnostic and therapeutic applications.1Over the past decade, fluorescence imaging in the first NIR window (NIR-I, 700–900 nm) has been widely studied in fundamental research and preclinical and clinical applications, which is partly because of the ready availability of a wide range of fluorophores, such as indocyanine green (ICG, λex = 808 nm, λem = 822 nm) and methylene blue (MB, λex = 665 nm, λem = 686 nm), which have been approved by the Food and Drug Administration (FDA) for clinical use. The NIR-II biological window is broadly defined as wavelengths in the range of 1000–1700 nm. Smaller optical sub-windows such as NIR-IIa (1300–1400 nm) and NIR-IIb (1500–1700 nm) have provided further improvements in fluorescence imaging metrics. The 1400–1500 nm window is typically avoided owing to the presence of an absorbance peak due to a water overtone. NIR-II fluorescence imaging can provide an even higher signal-to-noise ratio (because of lower tissue autofluorescence) and deeper tissue penetration (because of lower signal attenuation)2 (Fig. 1). NIR-II imaging is also able to provide micron-scale resolution, which is very difficult to achieve with NIR-I and many other imaging modalities such as positron emission tomography (PET) and single-photon emission computed tomography (SPECT).
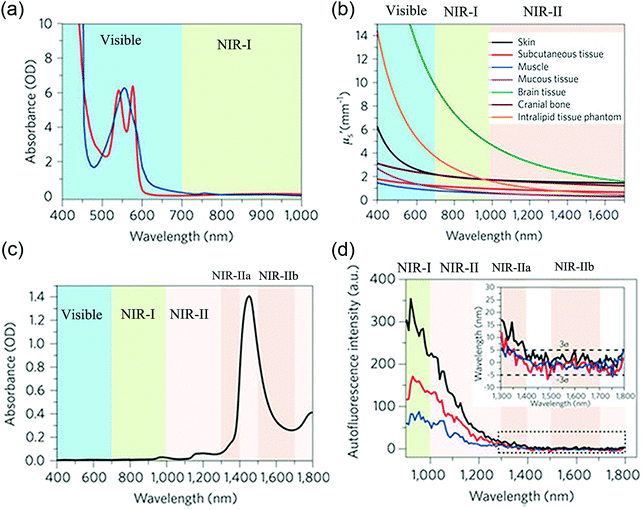 | ||
| Fig. 1 Motivation for NIR-II imaging: (a) absorption spectra of oxyhaemoglobin (red) and deoxyhaemoglobin (blue) through a 1 mm-long path in human blood. (b) Reduced scattering coefficients of different biological tissues and scattering intralipid tissue phantom as a function of the wavelength in the 400–1700 nm region. (c) Absorption spectrum of water through a 1 mm-long path. (d) Autofluorescence spectra of ex vivo mouse liver (black), spleen (red) and heart tissue (blue) under excitation with 808 nm light, showing the absence of autofluorescence in the >1500 nm NIR-IIb window. (a)–(d) Reprinted (adapted) with permission from ref. 2, Copyright 2017, Nature Publishing Group. | ||
Because NIR-II fluorescence imaging is a newly emerging field, only very recently have fluorophores with emissions at suitable wavelengths been studied. Poor water solubility, low photostability, low quantum yield, and the scarcity of molecules with suitable NIR-II bandgaps have further limited the applications and development of NIR-II imaging techniques. Interestingly, in the last couple of years a series of organic small molecules and organic and inorganic nanomaterials with precisely controlled structures and intrinsic near-infrared emissions in the NIR-II window have been developed to enable the acquisition of high-definition NIR-II images at wavelengths well in excess of 1000 nm.
One of the greatest challenges in bioimaging is to achieve both high resolution and high contrast. Despite huge advances in NIR-II fluorescence imaging for preclinical applications, comprehensive information at different spatial scales and resolutions is not likely to be obtained using a single imaging modality. Multimodal imaging, which combines two or more imaging modalities, has emerged as a strategy to address this challenge. Together with the advances in imaging techniques, it is critical to create complementary imaging contrast agents. These should have enhanced sensitivity to reveal physiological structures and specific molecular information on diseases. In diagnostic imaging, however, the selection of a modality is driven by the fact that high sensitivity and high resolution are very difficult to combine in one modality. The complementary abilities of different imaging modalities could be harnessed to achieve great positive effects. Imaging modalities that are used or investigated in clinical use include magnetic resonance imaging (MRI), computed tomography (CT), PET, SPECT and photoacoustic imaging (PAI). Among the above imaging techniques, MRI and CT generate high-resolution anatomical images, but the sensitivity of the contrast agent is very low. SPECT and PET have the advantages of high sensitivity, unlimited penetration depth and quantifiable results. These modalities are widely used for monitoring pharmacokinetics, biodistribution and accumulation at the target site but have limited spatial resolution. PAI is a relatively new imaging technique with high spatial resolution. It provides high contrast, although detection is limited when this technique is used in soft tissues.
Recently, various novel integrated NIR-II dual-modal imaging approaches have been developed, such as MRI/NIR-II, SPECT/NIR-II, PET/NIR-II and PAI/NIR-II, which exploit the advantages of NIR-II fluorophores. These approaches not only display high sensitivity but also have a great penetration depth. Although a series of desirable multimodal-imaging NIR-II fluorophores have been recently developed, the correlation between NIR-II and other imaging modalities is not clear, and few reports have focused on the critical mechanistic aspects of design. Most applications have specific mechanistic requirements for both fluorophores and devices to achieve the best performance in high-definition imaging.
To better understand and optimize both imaging and the delivery of therapy to a lesion site, it is crucial to monitor different aspects of the therapeutic process, including pharmacokinetics, biodistribution, accumulation at the target site, local distribution, kinetics of drug release and therapeutic efficacy. In recent years, NIR-II imaging techniques have been utilized to guide therapy to delineated targeted tissues, monitor the process of therapy (e.g. photothermal therapy, photocontrolled chemotherapy and photodynamic therapy) and visualize drug distribution. The visualization of drug delivery and drug release can be very useful in validating and enhancing drug delivery systems, as well as in predicting responses to therapy, especially if functional or molecular imaging information can be quantified.
This tutorial review is not only a comprehensive summary of advances in NIR-II fluorescence in in vivo imaging, but also a thorough investigation of multimodal NIR-II imaging probes and theranostics. In this review, we review chemical methods for preparing donor–acceptor (D–A) structures and the luminescence characteristics, size, morphology and surface characteristics of brightly emitting NIR-II fluorophores. The correlation between different imaging modalities using one NIR-II probe and specific applications is highlighted. The use of emerging applications of NIR-II fluorophores in the form of single nanoparticles or hybrid composites in biomedical imaging or to guide therapy is also reviewed. In addition, recent advances in reported NIR-II imaging systems are highlighted. Lastly, the future perspective and new challenges in the use of NIR-II fluorophores are discussed.
2. NIR-II imaging vs. NIR-I imaging
Photons emitted from NIR-I fluorophores have low energy, and these photons in the NIR-I region are not active for reacting with tissues, which leads to low light scattering and absorbance in the NIR-I window. In comparison with visible imaging in the region of 400–700 nm, NIR-I imaging in the region of 700–900 nm has the advantage of a greater tissue penetration depth to visualize anatomical structures and metabolic processes, as well as the intraoperative NIR-I-guided surgical removal of diseased tissues. However, beyond the NIR-I region, the longer-wavelength NIR-II regions of 1000–1400 nm and 1500–1700 nm will be more favourable windows owing to the lower scattering coefficients of tissues and autofluorescence, as shown by the declining trends of the scattering curves and slow decay of autofluorescence at longer wavelengths (Fig. 1b and d). In the NIR-II region beyond 1500 nm, autofluorescence almost completely disappears. This autofluorescence-free window beyond 1500 nm will further minimize interference during fluorescence biological imaging.In recent years, it has been demonstrated that the minimal scattering, low photon absorbance and reduced tissue autofluorescence in the NIR-II window can significantly increase signal-to-noise ratios and penetration depths. For example, in imaging of the hind limb vasculature, the signal-to-noise ratio achieved in NIR-II imaging was proved to be 4 times (4.8 vs. 1.2) higher than that in NIR-I imaging. In addition, we can distinguish blood vessels with a spatial resolution of as low as 220 μm in the NIR-II window, whereas it is very difficult to achieve such high spatial resolution of blood vessels in the NIR-I window.3 In imaging of lymphatic vasculature, the small-molecule NIR-II fluorophore CH1055-PEG provides sharper features at lymphatic dimensions (423 μm) than the blurred lymphatic features observed using the NIR-I fluorophore ICG, owing to both reduced photon scattering and the broadening of feature sizes in NIR-II imaging.4 In the through-skull non-invasive imaging of brain tumours at up to 4 mm, NIR-II imaging clearly demonstrates more advantages than NIR-I imaging. Both brain tumours and brain vasculature can be visualized in the NIR-II region with an obviously high degree of clarity, but no brain tumours or vasculature are observed using NIR-I imaging, even when the exposure time is 100 times longer than that in NIR-II imaging. In another instance, to accurately compare the penetration depth of NIR-II imaging to that of NIR-I imaging, the intensity of an NIR-I signal at 808 nm rapidly diminishes over increasing tissue depth with complete signal loss by 5 mm, whereas 1525 nm NIR-II light can be detected through 10 mm of phantom tissues.5
These results suggest that NIR-II imaging provides much higher resolution and more accurate information on deep tissues than NIR-I imaging, which has inspired scientists to investigate the synthesis of new NIR-II fluorophores and their biomedical applications.
3. Classification of NIR-II fluorophores
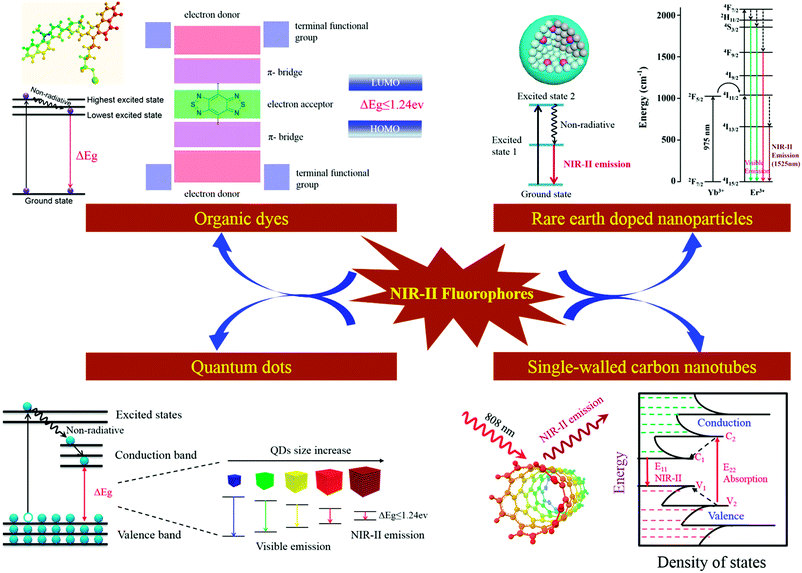
The first use of in vivo NIR-II fluorescence imaging in mice was reported in 2009.6 In this pioneering study, low-dose single-walled carbon nanotubes (SWCNTs) were used to detect individual vascular tumours noninvasively through the skin while monitored with an InGaAs camera, which is operated in the NIR-II spectral range. Since then, NIR-II fluorescence imaging has become an interesting and fast-growing research area. The development of novel NIR-II fluorophores for in vivo imaging applications is very important and is expected to have a great impact in many fields in medicine.An NIR-II fluorophore with broad utility is expected to have the following properties: low toxicity, bright emission (high quantum yield), and low susceptibility to photobleaching (high photostability and chemical stability), and be chemically modifiable to achieve specific targeting of disease lesions. To date, several types of organic and inorganic NIR-II fluorophores have been reported.3–6,9,17,19 Organic NIR-II fluorophores include small-molecule dyes (SMDs), small-molecule dye complexes (SMDCs), small-molecule dye-based organic nanoparticles (SMDNPs), and semiconducting polymer-based nanoparticles (SPNPs). Inorganic NIR-II fluorophores include quantum dots (QDs), rare earth-doped nanoparticles (RENPs) and SWCNTs. The photophysical pathways of NIR-II fluorophores in solution and the mechanism of NIR-II fluorescence emissions are summarized in Scheme 2. Among these NIR-II fluorophores, SMDs, SMDCs and SMDNPs are scarcely retained within the reticuloendothelial system, which shows high promise for future clinical translation. The bandgap of SPNPs is tunable to obtain various emission wavelengths in the NIR-II window. The size of QDs for NIR-II imaging is small (<10 nm), and they exhibit strong NIR-II emissions because of their high quantum yields of up to 10%. RENP probes act as NIR-II emitters at specific wavelengths depending on the lanthanide ions in single nanocrystals. SWCNTs have broad emission spectra at wavelengths between 1000 and 1800 nm, but with low quantum yields (<0.4%). A comparison of key aspects of different NIR-II fluorophores is provided in Table 1.
| Type | NIR-II fluorophore | Excitation (nm) | Emission (nm) | QY (%) | Application | Targeting | Ref. |
|---|---|---|---|---|---|---|---|
| SMDs | CH1055 | 808 | 900–1350 | 0.3 | Imaging of lymphatic vasculature, orthotopic glioblastoma and brain tumour and guiding surgery | Anti-EGFR affibody | 4 |
| Peak: 1050 | |||||||
| ICG | 782 | 880–1450 | 0.9 | Imaging of vasculature and hind limb | Non-specific | 7 and 8 | |
| CH-4T | 808 | 900–1400 | 0.1 | Video-rate and ultrafast imaging of hind limb vasculature and lymph node | Non-specific | 9 | |
| Peak: 1050 | |||||||
| Q4-1 | 808 | 900–1400 | 0.2 | Multiple conjugation sites for NIR-II imaging | Conjugated peptide GRPR targeting | 10 | |
| Peak: 1100 | |||||||
| CHS2 | 808 | 900–1400 | 0.2 | Guiding surgery and tumour delineation | Conjugated with RGD | 11 | |
| Peak: 1050 | |||||||
| IR-E1 | 808 | 900–1400 | 0.7 | Video-rate dynamic brain imaging | Non-specific | 12 | |
| Peak: 1071 | |||||||
| SMDCs | ICG–FBS | 808 | 880–1200 | — | — | — | 9 |
| CH-4T-FBS | 808 | 900–1400 | 5–11 | Video-rate and ultrafast imaging of hind limb vasculature and lymph node | Strong protein-binding affinity | 9 | |
| Peak: 1000 | |||||||
| IR-FGP | 808 | 900–1400 | 1.9 | 3D layer-by-layer molecular imaging | Bioconjugation with streptavidin, Erbitux, EGFR and IgG antibodies | 13 | |
| Peak: 1050 | |||||||
| FSH-CH | 808 | 900–1400 | — | NIR-II imaging of testicular tubules, bones, and ovarian follicles | Hormonally specific binding | 14 | |
| Peak: 1050 | |||||||
| SMDNPs | Q4NPs | 808 | 900–1400 | 0.2 | Imaging of blood vessels in tumour | Non-specific | 10 |
| Peak: 1100 | |||||||
| SXH and SDH NPs | 808 | 900–1500 | 2.0 | Imaging of whole-body blood vessels and guiding SLN surgery | RGD peptide | 15 | |
| Peak: 1100 | |||||||
| CQS1000 NPs | 808 | 900–1300 | — | Imaging for assessment of angiogenesis of tumour, precise tumour resection, and guiding SLN mapping and biopsy | Anti-EGFR affibody | 3 | |
| Peak: 1000 | |||||||
| IR1061-PEG NPs | 808 | 900–1300 | 1.8 | Whole-body imaging | Non-specific | 16 | |
| Peak: 1047 | |||||||
| SPNPs | pDA–PEG | 808 | 900–1400 | 1.7 | Ultrafast NIR-II imaging of arterial blood flow | EGFR antibody | 17 |
| Peak: 1047 | |||||||
| PDFT1032 | 808 | 900–1400 | — | Guiding surgery, vascular imaging, SLN biopsy mapping | Non-specific | 18 | |
| Peak: 1032 | |||||||
| QDs | Ag2S–6PEG | 808 | 1000–1400 | 15.5 | 4T1 tumour xenograft imaging | Non-specific | 19 |
| Peak: 1200 | |||||||
| Ag2S–PEG | 808 | 1000–1400 | 16 | Lymphatic drainage imaging, blood pool imaging, angiogenesis imaging | Non-specific | 20 | |
| Peak: 1200 | |||||||
| Ag2S–PEG | 808 | 1000–1500 | 15.5 | In vivo tracking of hMSCs | Tat peptide | 21 | |
| Peak: 1200 | |||||||
| Ag2S@SiO2 | 785 | 1000–1200 | 22.7 | NIR-II tumour imaging | cRGDfK-R6G peptide | 22 | |
| Peak: 1135 | |||||||
| RENPs | Y2O3:Yb, Er | 980 | 1400–1700 | — | Organ imaging | Non-specific | 23 |
| Peak: 1550 | |||||||
| NaYF4:Yb:Ln (Ln: Er, Ho, Tm or Pr) | 980 | 1000–1700 | — | Multispectral tumour imaging | Bioactive albumin | 5 | |
| Peak: 1525, 1185, 1475, 1310 | |||||||
| NaYF4:Yb:Er@NaYF4 | 980 | 1000–1700 | — | Cancer early detection | AMD3100 | 24 | |
| Peak: 1525 | |||||||
| NaYF4:Yb3+/X3+@NaYbF4@NaYF4:Nd3+-ICG (X = Er, Ho, Tm, or Pr) | 808/980 | 1000–1700 | 13 | ICG-enhanced NIR-II emission | Non-specific | 25 | |
| Peak: 1060 1165, 1310, 1460, 1530 | |||||||
| SWCNTs | SWCNTs-mPEG | 808 | 1100–1400 | 0.1 | 4T1 tumour xenograft imaging, video-rate imaging, and 3D reconstruction | Non-specific | 26 |
| SWCNTs-cholate | 808 | 900–1500 | 0.4 | Intravital tumour vessels imaging | RGD peptide | 6 | |
| SWCNTs-DSPE-mPEG | 808 | 1100–1700 | 0.1 | Video-rate in vivo imaging | Non-specific | 27 | |
| SWCNTs-IRDye800 | 785/808 | 1100–1400 | — | Video-rate venous vessels imaging, blood perfusion imaging and brain vasculature imaging | Non-specific | 28 and 29 | |
| Chiral SWCNTs | 725 | 850–1350 | — | Vascular imaging | Non-specific | 30 | |
| LV SWCNTs | 808 | 1200–1800 | 0.01 | High-magnification cerebrovascular imaging | Non-specific | 31 | |
| SWCNTs-DNA | 780 | 950–1050 | — | Implantable inflammation sensor | Non-specific | 32 | |
3.1 Organic NIR-II fluorophores: SMDs, SMDCs and SMDNPs
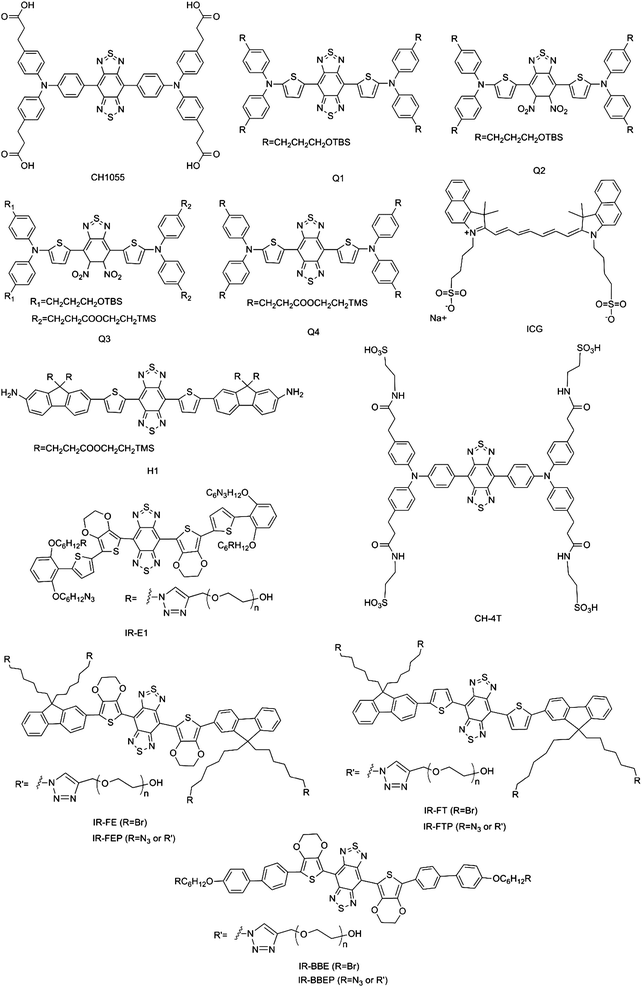
Thus far, several types of SMD NIR-II fluorophores with excellent performance have been developed, and representative chemical structures are shown in Fig. 2. SMDs have NIR-II emissions with different degrees of brightness and are rapidly excreted, and can be manufactured under Current Good Manufacturing Practice (cGMP) conditions. Because of these factors, SMDs are widely recognized as highly attractive and promising NIR-II fluorophores for future clinical applications. The first example of the use of an SMD NIR-II fluorophore, namely, CH1055, for in vivo imaging was reported in 2015 (Fig. 3a–d).4 The fluorophore units are composed of conjugated aromatic units with a donor–acceptor–donor (D–A–D) structure and a benzobisthiadiazole core. The energy gap could be reduced via π-spacers by attaching strong electron donors. The spatial configuration of strongly electron-donating groups flanking a central electron acceptor reduces the energy gap separating the hybridized highest occupied molecular orbital (HOMO)/lowest unoccupied molecular orbital (LUMO) levels and shifts the fluorescence emission into the NIR-II window. CH1055 has an emission peak at 1055 nm with a tail extending the emission into the NIR-IIa region (1300–1400 nm). The fluorophore enters the brain vasculature and enables the detection of tumours in the brain at depths of 4 mm by a non-invasive through-skull technique. In comparison with the FDA-approved fluorophore ICG for NIR-I imaging, CH1055-based NIR-II imaging provides higher resolution in the diagnosis of sentinel lymph nodes (SLNs) for surgical resection. Pharmacokinetic studies of ICG–PEG demonstrate rapid urinary excretion comparable to that of ICG, with 90% excretion via the kidneys in 24 h. An NIR-II fluorescent probe for tumour-specific targeting can also be obtained by conjugating CH1055 with an anti-epidermal growth factor receptor (EGFR) affibody molecule. The affibody-conjugated CH1055 specifically targets human squamous cell carcinoma xenograft tumours in immune-deficient mice and enables accurate image-guided tumour removal surgery. Molecular imaging in the NIR-II window gives a tumour-to-normal tissue ratio five times higher than that in the NIR-I window. In another work, Feng et al. conjugated CH1055 to follicle-stimulating hormone (FSH) and used the resulting NIR-II probe to specifically image ovaries in live mice. They were also able for the first time to specifically detect FSH receptor in bones. According to the expression of FSH receptors, earlier controversies regarding cultured osteoclasts could be resolved.14 Furthermore, the investigation of the structure-absorption/fluorescence properties of a small library of fluorescent compounds in which the core structure of CH1055 had been modified enabled the identification of another small-molecule NIR-II fluorophore, namely, Q4, owing to its attractive properties. The conjugation of Q4 with the GPCR-targeting peptide RM26 provided a new targeted NIR-II probe, namely, SCH1100, which was successfully used for the specific imaging of prostate cancer in vivo.10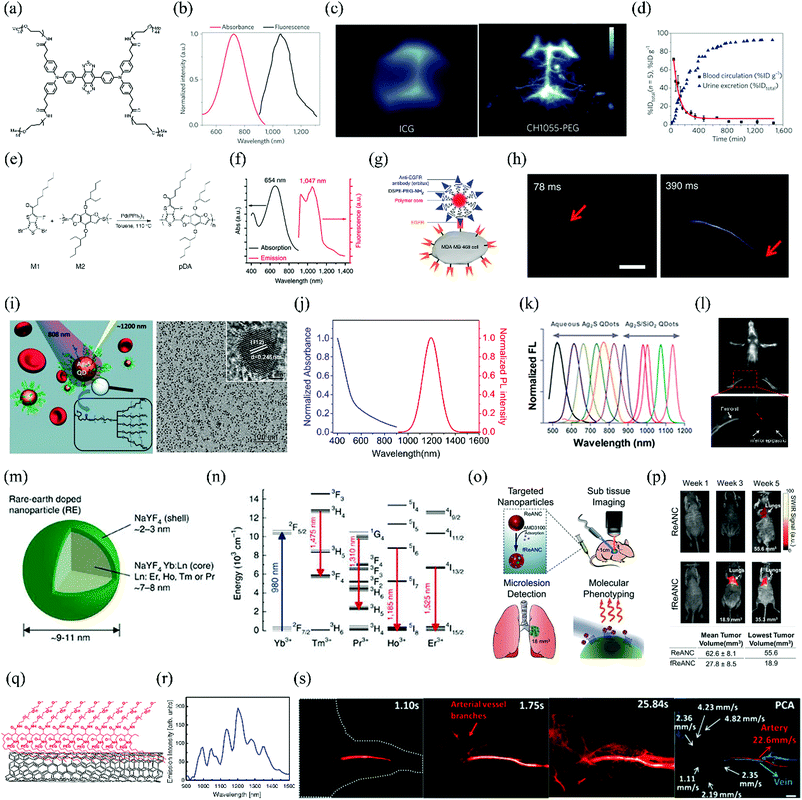 | ||
| Fig. 3 Advances in NIR-II fluorophores and imaging in vivo. (a) Chemical structure of CH1055. (b) Absorbance and fluorescence emission of CH1055-PEG (808 nm excitation). (c) Imaging of brain vasculature through the scalp and skull in mice using either ICG or CH1055-PEG. (d) PEGylated CH1055 (8.9 kDa) exhibits 90% renal clearance in the first 24 h post-injection. (e) Scheme of the synthesis of the SPNP NIR-II-emitting polymer pDA. (f) Absorption and emission spectra of pDA–PEG. (g) Schematic showing the structure of a pDA–PEG–Erbitux bioconjugate. (h) Ultrafast NIR-II imaging of arterial blood flow. (i) Schematic of 6PEG–Ag2S QDs that emit at 1200 nm upon excitation at 808 nm and TEM images of Ag2S QDs. (j) Absorbance and photoluminescence spectra of PEGylated Ag2S QDs. (k) Tunable NIR-II emission of Ag2S QDs. (l) Intravital NIR-II fluorescence image of nude mouse in the supine position. The PL signal from the Ag2S QDs is easily distinguishable from the endogenous autofluorescence without any image processing. (m) Rare earth nanoprobes consisting of an NaYF4:Yb:Ln-doped core (Ln: Er, Ho, Tm or Pr) surrounded by an undoped shell of NaYF4. (n) Probes consisting of an NaYF4 host doped with Yb, Ho, Pr, Tm and Er enable emissions at 1185, 1310, 1475 and 1525 nm, respectively. The emissions of the Ho-, Pr-, Tm- and Er-doped samples are attributed to the 5I6 → 5I8, 1G4 → 3H5, 3H4 → 3F4, and 4I13/2 → 4I15/2 transitions, respectively. (o) AMD3100 was adsorbed onto the surface of fully formed ReANCs to generate AMD3100-functionalized ReANCs (fReANCs). Athymic nude mice were inoculated with human breast cancer cells through the tail vein. Animals were treated with either ReANCs or fReANCs and NIR-IIB imaging was performed to determine the localization of the nanoprobe in the tumour. (p) Longitudinal tracking of internal lesions via NIR-IIB imaging. The mean and minimum volumes of tumours detectable via NIR-IIB imaging were calculated to determine the smallest tumours capable of resolution using an InGaAs camera. (q) Schematic of water-soluble SWCNTs conjugated with C18-PMH-mPEG (90 kDa). (r) Fluorescence spectrum of SWCNTs excited at 808 nm, showing several emission peaks spanning the NIR-II region. (s) Video-rate NIR-IIb fluorescence imaging of mouse hind limb vessels (n = 3) and dynamic contrast-based differentiation of the vessel type. PCA overlaid image showing the differentiation of arterial (red) and venous (blue) vessels. Scale bar: 2 μm. (a–d) Reprinted (adapted) with permission from ref. 4, Copyright 2015, Nature Publishing Group. (e–h) Reprinted (adapted) with permission from ref. 17, Copyright 2014, Nature Publishing Group. (i) Reprinted (adapted) with permission from ref. 19, Copyright 2012, Wiley-VCH Verlag GmbH & Co. KGaA. (j and l) Reprinted (adapted) with permission from ref. 20, Copyright 2014, Elsevier B.V. (k) Reprinted (adapted) with permission from ref. 22, Copyright 2012, American Chemical Society. (m and n) Reprinted (adapted) with permission from ref. 5, Copyright 2013, Nature Publishing Group. (o and p) Reprinted (adapted) with permission from ref. 24, Copyright 2015, Wiley-VCH Verlag GmbH & Co. KGaA. (q) Reprinted (adapted) with permission from ref. 26, Copyright 2012, American Chemical Society. (r) Reprinted (adapted) with permission from ref. 27, Copyright 2011, National Academy of Sciences. (s) Reprinted (adapted) with permission from ref. 31, Copyright 2015, Wiley-VCH Verlag GmbH & Co. KGaA. | ||
Another interesting small-molecule dye, namely, IR-E1, was reported recently (Fig. 2). This novel NIR-II fluorophore exhibits a higher quantum yield (0.7%) than CH1055 (QY = 0.3%), as well as fast renal excretion (83%) within 24 h post-injection. In addition, it achieves high-resolution dynamic imaging of the vascular changes particular to a model of traumatic brain injury, including initial transient hypoperfusion due to leakage and accumulation of the NIR-II fluorophore caused by cerebrovascular damage.12
Interestingly, in 2017 the small-molecule dyes ICG and IR800 were reported for the first time to exhibit NIR-II fluorescence with enhanced brightness (tail up to 1150 nm) via assembly with FBS.9 Following this research work, by switching the detection from conventional silicon-based NIR cameras to emerging high-performance InGaAs SWIR cameras, Bruns et al. found that ICG exhibited a higher quantum yield (0.9%) in an aqueous solution than IR-E1050 (quantum yield of 0.2%) in the NIR-II emission region (detectable up to 1575 nm).8 Moreover, Starosolski et al. demonstrated that ICG displayed a significantly enhanced NIR-II (1000–1250 nm) emission in plasma and low-polar solvents such as ethanol.7In vivo imaging results have demonstrated that the signal-to-noise ratio when ICG was used in the NIR-II window was twice that in the NIR-I window.7 These findings open a quick route for the translation of NIR-II imaging techniques into clinical applications. In general, the following categories of dye–solvent and dye–dye interactions in non-polar or polar solvents may play a vital role in deactivating excited-state fluorophores. Several important factors are (i) electrostatic interactions, (ii) weak associations with a defined stoichiometry and structure, (iii) dye–solvent and dye–dye hydrogen bonding.
Besides, for such low-energy-gap NIR-II fluorophores, a great variety of complicated and uncertain processes are involved in the deactivation of the excited state. The following factors may play a vital role in the deactivation of excited states: (i) intermolecular interactions between NIR-II fluorophores; (ii) the formation of strong complexes in the excited state and ground state. Excitation can cause structural reorganization, and the deactivation process of excited states may be fast; (iii) dye diffusion processes in solvents may deactivate excited states; and (iv) dielectric NIR-II fluorophores may affect proton translocation and electron transfer, leading to the deactivation of excited states.
However, most SMDs are hydrophobic and need to be encapsulated into a surfactant (e.g. PEG or liposomes) or conjugated with a protein to form NIR-II SMD complexes or SMD nanoparticles, which are named as SMDCs and SMDNPs, respectively. Although CH1055 has excellent pharmacokinetic properties, factors such as the low quantum yield (0.3%), fluorophore absorbance, emission wavelength and complexity of synthesis could be improved. Low quantum yields of NIR-II fluorophores limit penetration depths and cause low temporal resolution. Diversified absorbances and emissions of fluorophores in the NIR-II region would offer more options. By replacing the carboxylic acid groups of CH1055 with more negatively charged sulfonic acid groups and using supramolecular assemblies with serum proteins, Antaris et al. produced a water-soluble SMDC NIR-II dye in FBS (CH-4T-FBS), which exhibited a 110-fold enhancement in fluorescence.9 CH-4T-FBS enables fast video-rate imaging at 50 frames per second in the NIR-II window and reduces the exposure time more than 50-fold in comparison with ICG. CH-4T-FBS clearly demonstrates the benefits of the NIR-II window for imaging deep anatomical features.
Novel SMDNP NIR-II fluorophores, namely, SXH and SDH, were recently prepared and used for integrin αVβ3-targeted imaging of glioma.15 These NIR-II organic nanoparticles enable the differentiation of tumours from surrounding normal tissue and can also be employed for image-guided surgery. It is worth noting that the SXH and SDH NIR-II probes are based on the small molecule H1. In the chemical structure of H1, 2-amino-9,9-dialkyl-substituted fluorene moieties are introduced into the CH1055 structure. They can distort the benzobisthiadiazole core and effectively tune the distribution of electrostatic potential and the bandgap to a desired range. The fluorene moieties act not only as electron donors but also as protecting groups that prevent the backbone from aggregating. In another work, a multifunctional NIR-II probe, namely, CQS1000 SMDNPs, was prepared by encapsulating CH1055 into phospholipid vesicles.3 CQS1000 can be used to noninvasively and dynamically visualize and monitor many physiological and pathological conditions of circulatory systems. Processes that can be monitored include lymphatic drainage and routing, tumour angiogenesis and vascular diseases. In comparison with traditional NIR-I imaging, CQS1000 offers the promise of improved guidance for surgery by mapping sentinel lymph nodes. This new NIR-II fluorophore enabled the complete and precise resection of tumours by distinguishing the blood supply and lymphatic drainage, which may lead to higher survival rates and reduced relapse rates. Another novel water-soluble NIR-II SMDC, namely, IR-FGP, was synthesized using click chemistry, as recently reported by Zhu et al.13 IR-FGP not only provides a bright NIR-II emission via the systematic tuning of a D–A–D architecture but also offers many targeting channels. In addition, the IR-FGP NIR-II SMDC nanoprobe enables 2D and 3D tomographic imaging in deep tissue and reduces autofluorescence, which strongly benefits a broader spectral imaging window in the NIR-II region.
It is worth noting that SMD, SMDC and SMDNP NIR-II fluorophores still need further improvement. The versatility of SMD, SMDC and SMDNP NIR-II fluorophores for the precise visualization and evaluation of biological processes also needs to be thoroughly investigated. Importantly, many unmet clinical needs could be evaluated and potentially addressed by the use of SMD, SMDC and SMDNP NIR-II fluorophores.
3.2 Organic NIR-II fluorophores: SPNPs
SPNPs are semiconducting organic macromolecules that are characterized by a backbone chain of alternating double and single bonds. They have been widely used in organic solar cells, diodes and electronics. Fluorescence imaging with SPNPs has been limited to emission wavelengths of <900 nm. Shifting their emissions, specifically into the NIR-II window, is challenging but essential for broadening their biomedical applications. Increasing the π-conjugation length by designing quinoid-stabilized D–A polymers is an effective strategy for reducing the bandgap and bringing about a bathochromic shift into the NIR-II window. Hong et al. have described a facile method for synthesizing SPNPs emitting in the NIR-II region with a tunable bandgap energy by D–A alternating copolymerization. One of these copolymers, namely, poly(benzo[1,2-b:3,4-b′]difuran-alt-fluorothieno[3,4-b′]thiophene) (pDA), exhibits an NIR-II emission peak at 1050 nm and a high quantum yield of up to 1.7%, which is much higher than that of typical SWCNTs (∼0.4%) used previously (Fig. 3e–h).17 Because of the unique charge transfer structure between the donor and acceptor, the pDA NIR-II fluorophore exhibits red shifts in absorption and emission in comparison with the corresponding homopolymers. This leads to a series of SPNPs with tunable excitation and emission wavelengths for NIR-II imaging. The conjugation of PEGylated pDA with cetuximab (Erbitux), which is an antibody against EGFR, using standard crosslinking reactions between NH2 groups and SH groups gave pDA–PEG–Erbitux. When used for NIR-II fluorescence imaging, the antibody-conjugated probe selectively targets EGFR-positive cells in preference to EGFR-negative cells. This NIR-II fluorophore enables the imaging of dynamic changes in blood flow at a frame rate of over 25 frames per second, which extends the limits of temporal resolution to a previously unattainable level. In addition, the pDA SPNP NIR-II fluorophore can track blood flow in capillaries with diameters of as little as 10 μm, which is even below the spatial resolutions of ultrasound and optical coherence tomography (OCT). The combination of the pDA NIR-II fluorophore and the sensitivity of the detector gives a dynamic measurement range for blood velocity of 0 to 640 mm s−1. The pDA–PEG NIR-II fluorophore can also track regional blood flow and redistribution and thus provides a diagnostic tool for directly visualizing metabolic differences in tissues. This study provides the possibility of NIR-II fluorescence probes that can enable haemodynamic imaging with high spatial and high temporal resolution by minimizing scattering with high quantum yields and ultrashort imaging exposure times. However, one limitation of pDA SPNPs that should be considered is that photodegradation upon excitation at 808 nm for 1 h results in a decline in NIR-II fluorescence intensity of up to 20%. Another major drawback of pDA SPNPs is the potential risk of high toxicity.Very recently, a highly photostable SPNP, namely, PDFT1032, was developed.18 PDFT1032 was synthesized from the highly planar electron acceptor diketopyrrolopyrrole (DPP), which is able to couple with many electron donors easily, and the bandgap can be tuned to obtain the expected fluorescence emission. PDFT1032 not only exhibits an excellent maximum absorption wavelength of 809 nm, but also a large Stokes shift of 223 nm to 1032 nm under excitation at 808 nm. In addition, PDFT1032 demonstrates excellent performance in real-time imaging, including tumour diagnosis and SLN mapping. Moreover, in NIR-II image-guided tumour surgery and embolization therapy, PDFT1032 displays high spatial resolution (μm range) and high temporal resolution (>25 frames per second). These excellent properties highlight PDFT1032 NIR-II SPNPs as a promising and widely applicable NIR-II probe for preclinical and, potentially, clinical imaging.
3.3 Inorganic NIR-II fluorophores: QDs
Amongst the seven classes of NIR-II-emitting probes reported thus far, QDs exhibit the highest fluorescence quantum yields (up to 15.5%).19 The currently most desirable brightly fluorescent NIR-II QDs, such as PbS and Ag2S, act as versatile NIR-II QD fluorophores because of their high resistance to photobleaching, tunable bandgaps and large multiphoton absorption cross-sections.The first and most promising NIR-II fluorescent QDs to be used in preclinical research comprise Ag2S, which does not contain a toxic heavy metal and exhibits high uptake in tumours (Fig. 3i).19 Ag2S QDs have been used in the imaging of blood vasculature with micrometre-level resolution (down to 40 μm) of the peripheral vasculature and tumour angiogenesis during cancer growth and metastasis. It is critical to monitor the formation of new blood vessels, both around and within tumours, for surgical treatment, assessment of the blood supply in tissues and screening of anti-angiogenic drugs. Ag2S QD-based NIR-II imaging provides greater clarity and penetration depth for visualizing lymphatic vessels and lymph nodes during resection of sentinel lymph nodes than NIR-I imaging based on the FDA-approved clinical standard contrast agent ICG (Fig. 3j and l).20 In addition, Ag2S QDs are able to track and visualize populations of mesenchymal stem cells (MSCs) during intricate biological processes.21 They provide a promising method of monitoring the transplantation of MSCs and acquiring important information about cell dynamics and interactions with the central nervous system of the host. Furthermore, Ag2S QDs offer the chance of enhancing both the sensitivity of early disease diagnosis and the identification of lesions by targeting functional molecules (e.g. proteins, peptides, folic acid and antibodies). For example, by conjugating 9 nm Ag2S QDs with a cyclic pentapeptide that binds to the αVβ3 integrin receptor, which is upregulated on the surface of tumour cells, the uptake efficiency has been improved 8- to 10-fold. Imaging sensitivity is significantly enhanced by creating a brightly NIR-II-emitting fluorophore for targeting specific sites (Fig. 3k).22
Although different strategies have been investigated for using QDs for high-quality NIR-II bioimaging, several points should be noted. Firstly, the leakage of encapsulated NIR-II QDs from surfactants in a physiological microenvironment causes toxicity in vivo. Secondly, protein or polymer coatings increase the particle size, and the pharmacokinetics may be changed. Thirdly, quenching of NIR-II emissions can be facilitated by functional groups (e.g. hydroxyl groups and hydrocarbons) attached to the surface of QDs. Fourthly, the functionalization process may cause the leaching of elements or etching, which leads to a shift in the NIR-II wavelength (i.e. a blue shift).
3.4 Inorganic NIR-II fluorophores: RENPs
RENPs that are composed of lanthanide ions (from La to Lu, together with Sc and Y) embedded within an inorganic crystalline host matrix (e.g. NaYF4, GdO2S2 and CaF2) have attracted considerable attention in the fields of materials science and optical imaging. Because of their large Stokes shifts, narrow and multi-peak emission profiles, long lifetimes and excellent photostability, RENPs have been considered as promising alternatives to conventional organic fluorophores and QDs for NIR-II imaging. The NIR-II fluorescence of RENPs occurs because of resonant energy transfer from a sensitizer to an RE activator dopant, such as erbium (Er), thulium (Tm), holmium (Ho), neodymium (Nd) or praseodymium (Pr), of which the relaxation generates a unique activator-dependent downshift. Yb/Er-co-doped ceramic phosphors with an emission peak at 1550 nm have been modified with PEG-b-poly(4-vinylbenzylphosphonate) (PEG-b-PVBP) and PEG possessing a monophosphonate end group (PEG–PO3H2), and the resulting RENPs have been successfully used for the NIR-II imaging of organs in mice.23Highly monodisperse RENPs are typically formed in high-boiling organic solvents via thermal decomposition. The poor aqueous solubility of the surfactant on the surface of the nanoparticles prevents subsequent functionalization for biomedical applications. Surface modification is thus crucial for improving the dispersion of RENPs in water. For example, by encapsulating RENPs with FDA-approved albumin, Naczynski et al. successfully demonstrated real-time multispectral in vivo NIR-II imaging, which provided anatomical resolution using a library of tunable RENPs (Fig. 3m and n).5 This work introduced a new generation of versatile NIR-II nanoprobes that facilitate disease monitoring using minimally invasive NIR-II imaging. Interestingly, RENPs have also been excited by X-rays instead of a laser to produce emission light for NIR-II imaging, which highlights their potential use for obtaining both anatomical and molecular information via CT and NIR-II imaging.33 Improvements in sensitivity to enable the detection of much smaller cancerous lesions can be made by the further surface functionalization of RENPs with specific targeting moieties. For example, targeting NIR-II fluorophores have been prepared by encapsulating RENPs with AMD3100, which is a hydrophobic small-molecule antagonist of the chemokine receptor CXCR4. These targeting RENPs are preferentially localized in receptor-positive tumours in mice, which enables the detection of CXCR4-positive tumours that are 4 times smaller than receptor-negative tumours (Fig. 3o and p).24 These targeted NIR-II fluorophores enable the imaging of microlesions in the lungs at a depth of ∼1 cm and are able to simultaneously identify the phenotype of the tumour population. This study is significant for the functionalization of NIR-II RENPs to detect sub-tissue microlesions at an early stage.
The optical efficiency of these RENPs is, however, quite low (<0.5%), and there is ample scope for this to be improved to maximize the full potential benefits of RENP-based bioimaging. For example, hybrid organic–inorganic nanocrystals that incorporate the contrast agent ICG exhibit a large Stokes shift (>200 nm) with multiple narrow-band emissions in the NIR-II range. ICG not only enhances the brightness of the NIR-II emission by a factor of ∼4 but also produces a broad excitation band at 700–860 nm. ICG-sensitized nanocrystals enable the clear observation of a sharp NIR-II image through chicken breast tissue, with signal detection at a depth of 23 mm.25
3.5 Inorganic NIR-II fluorophores: SWCNTs
SWCNTs have unique intrinsic physical and chemical properties (e.g. Raman scattering cross-sections, UV/visible/NIR absorption). Their biological and biomedical applications have been widely investigated over the past two decades. Because of van Hove transitions across bandgaps, semiconducting SWCNTs exhibit intrinsic fluorescence in the NIR-II window (Fig. 3q and r).26 In general, SWCNTs are hydrophobic and require surface functionalization with water-soluble polymers or proteins that render them non-toxic.6,27 In 2009, NIR-II probes were prepared for the first time by coating SWCNTs with a phospholipid–polyethylene glycol complex using an exchange procedure with sodium cholate. This marks the beginning of the use of SWCNTs for NIR-II imaging.6 Since then, SWCNTs have been extensively used for dynamic contrast-enhanced NIR-II imaging using principal component analysis (PCA). Applications include tumour-specific imaging,26 registration of mouse organs,27 and haemodynamic imaging of mouse peripheral vessels with higher spatial resolution than that afforded by in vivo micro-CT.28,30Non-invasive brain imaging is still largely reliant on CT and MRI, but these techniques are being challenged by fluorescence imaging. For the first time, SWCNTs have enabled NIR-II imaging of mouse cerebral vasculature with high spatial (<10 μm) and temporal (<200 ms per frame) resolution at a depth of >2 mm in an epifluorescence imaging mode.29 This is a truly non-invasive procedure that does not require invasive surgical procedures. The study enables real-time dynamic cerebrovascular imaging with high spatial and temporal resolution at a molecular level. Although longer-wavelength fluorescence emissions would further reduce scattering, the region between 1400 and 1500 nm is typically avoided because of the presence of an absorbance peak due to a water overtone.1,31 The long-wavelength NIR-II region (1500–1700 nm) provides a balance between photon scattering and water absorption effects and is able to achieve greater penetration depth and resolution. For example, SWCNTs with smaller bandgaps and larger average diameters (up to 1.24 nm) were recently synthesized by laser vaporization. They enable in vivo NIR-II vascular imaging at a depth of up to 3 mm in the mouse hind limb and brain with an intact skull and scalp with high spatial resolution (∼4 mm). Single-vessel-resolved mapping of blood flow speed in multiple hind limb arterial vessels has also been achieved simultaneously by video-rate NIR-II fluorescence imaging (Fig. 3s).31
The early detection of cancer ultimately improves patient survival. Detection sensitivity can be improved by creating targeted brightly emitting SWCNTs as NIR-II-emitting probes. For example, a genetically engineered multifunctional M13 phage has been assembled with SWCNTs via π–π interactions for use as a ligand for the targeted NIR-II imaging of tumours. In comparison with control non-targeted SWCNTs, targeted M13-SWCNTs displayed an up to 4-fold improvement in uptake in prostate-specific membrane antigen-positive prostate tumours. The detection and excision of deep, small metastatic tumour nodules by a gynaecological surgeon is also improved with M13-SWCNTs, which enable the identification of sub-mm tumours that are not easily identified by either visible or NIR-I fluorophores.34 Other suitable targeting strategies, such as analyte-specific receptors, which can transform SWCNTs into an NIR-II biosensor for detecting traces of biomolecules, have also been developed recently. For example, SWCNTs wrapped in A-DNA exhibit selective changes in NIR-II signal output in response to different NO concentrations and are capable of detecting changes in NO levels in response to wound-induced tissue inflammation.32 Careful engineering of the surfaces of SWCNTs to achieve high chemical stability and resistance to photobleaching will enable SWCNTs to have many promising applications. It should be noted, however, that SWCNTs display high infrared absorbance, which may cause NIR-II losses. Local increases in temperature could also result in thermal injury. Achieving excitation with lower power and higher quantum yields will thus be a major focus of future research into SWCNTs.
4. NIR-II fluorophore-based multimodal imaging
NIR-II is an emerging imaging technique that provides images of deep tissue with high spatial and temporal resolution, together with the inherent advantages of real-time imaging capability and high sensitivity. It can be used to visualize many dynamic processes in real time, such as cancer development and metastasis, cell trafficking, cellular events in immune reactions, etc. Despite this, there are still many limitations that need to be addressed because of the nature of optical imaging techniques. Firstly, tissue penetration by NIR-II light is still limited in comparison with many other modalities such as MRI, CT, PET, SPECT, and US, which can be used for whole-body imaging. Secondly, it is still very difficult to evaluate NIR-II images quantitatively in vivo. Thirdly, NIR-II imaging can provide molecular and physiological, but not anatomical, information on diseases. Therefore, it is highly desirable to develop NIR-II-based multimodal imaging techniques to overcome the limitations of NIR-II alone.(1) Single-component-based multimodality imaging
In the design of a multimodality imaging probe, sometimes a single component of the probe can be used to generate signals for different imaging modalities. For example, many fluorescent dyes can be used for both PAI and NIR imaging. PAI is an emerging optical imaging modality in which absorbed photon energy is converted into acoustic waves that are detected using ultrasound. PAI provides higher spatial resolution and better contrast than conventional optical imaging techniques, because ultrasound scattering in tissues is lower by ∼2–3 orders of magnitude than optical scattering. PAI also has high sensitivity and enables quantitative 3D visualization, which makes it particularly suitable for the diagnosis and assessment of a variety of diseases, including tumours. Recently, donor–acceptor chromophore-based nanoparticles (DAPs) have been synthesized by a nanoprecipitation process to encapsulate molecules of the NIR-II chromophore CH1000 within amphiphilic phospholipids. Moreover, to enhance the tumour targeting efficiency, an EGFR affibody was successfully conjugated with DAP. This affibody-DAP organic nanoprobe displayed the ability to selectively target EGFR-positive tumours in an FTC-133 subcutaneous mouse model with enormous enhancements in both PAI and NIR-II fluorescence contrast both in vitro and in vivo (Fig. 4a).35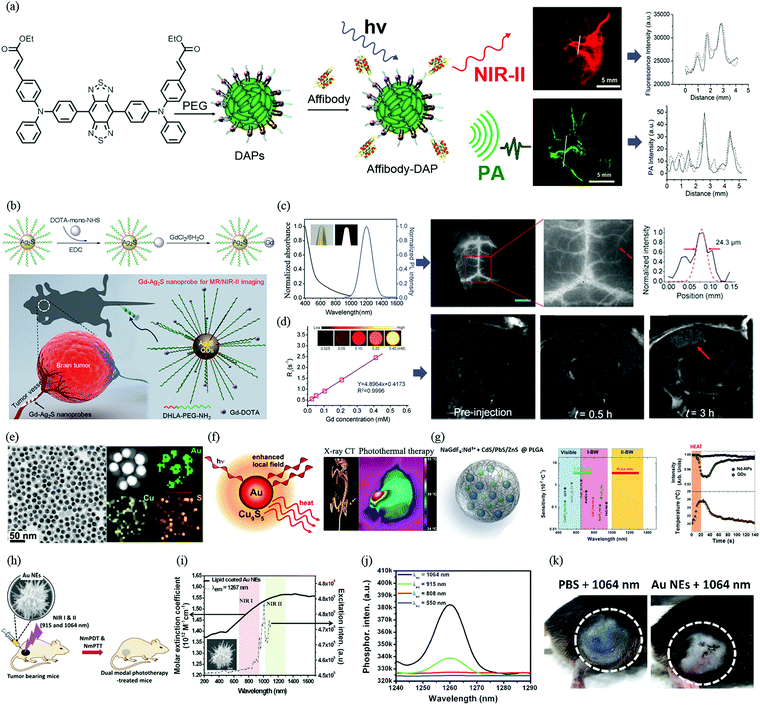 | ||
| Fig. 4 Progress of NIR-II fluorophore-based dual-modal imaging and phototherapy in vivo. (a) Schematic illustration of preparation of affibody-DAPs and dual-modal PA imaging and NIR-II imaging. Fluorescence image of an FTC-133 thyroid tumour in the NIR-II region and cross-sectional fluorescence intensity profiles along the white line across the FTC-133 tumour. 3D volume rendering of photoacoustic images of a thyroid tumour in a mouse before and after the injection of DAPs. Scale bar = 5 mm. Cross-sectional PA intensity profiles along the white line across the FTC-133 tumour. (b) Schematic illustration of the procedure for preparing a Gd–Ag2S nanoprobe and diagram of targeting of a brain tumour with the Gd–Ag2S nanoprobe. (c) Absorption and emission spectra of Gd–Ag2S nanoprobe. (d) Intravital NIR-II fluorescence image of brain vessels in a nude mouse (left). Amplified fluorescence image of vasculature in the nude mouse (middle). Cross-sectional intensity profile recorded along the red dashed line in (middle) with its peak fitted to a Gaussian function (right). The scale bar represents 3 mm. Relaxation rate R1; the inset shows a T1 map of an MR imaging phantom containing the Gd–Ag2S nanoprobe with different concentrations. In vivo progressive T1-weighted MR images of a U87MG brain tumour at different time points from a transverse view and immunohistochemical staining of vessels in brain tissues and tumour tissues with an anti-VEGF antibody. The red arrow indicates the tumour. (e) TEM and HRTEM images of synthesized Au–Cu9S5 NPs. (f) Schematic of a dual plasmonic hybrid nanosystem based on Au–Cu9S5 for enhanced near-field photothermal transduction due to LSPR and application for CT imaging and robust photothermal therapy. (g) Schematic diagram of PLGA nanostructures encapsulating both NaGdF4:Nd3+ nanoparticles and PbS/CdS/ZnS quantum dots. Thermal sensitivities and spectral operating ranges of materials in the visible, NIR-I and NIR-II regions. Evolution of the emission intensity of hybrid nanostructures injected into chicken breast tissue during a heating/cooling cycle in the tissue. Evolution of the temperature of the tissue during the heating/cooling cycle. The temperatures were calculated on the basis of the ratio of the emission intensities of the NaGdF4:Nd3+ nanoparticles and PbS/CdS/ZnS quantum dots. (h) Schematic illustration of multi-branched gold nanoechinus for NIR light-activated dual-modal photodynamic and photothermal therapy in the second biological window. (i and j) UV/VIS/NIR absorption spectrum (black solid line) and excitation spectrum (black dashed line) for singlet oxygen phosphorescence of lipid-coated Au NEs (λem = 1267 nm). (k) Relative volumes of tumours after different treatments and representative images of mice showing the sizes of tumours (highlighted by the dashed white circles) at day 14 under NIR II radiation. (a) Reprinted (adapted) with permission from ref. 35, Copyright 2017, American Chemical Society. (b–d) Reprinted (adapted) with permission from ref. 38, Copyright 2015, Wiley-VCH Verlag GmbH & Co. KGaA. (e and f) Reprinted (adapted) with permission from ref. 42, Copyright 2014, American Chemical Society. (g) Reprinted (adapted) with permission from ref. 46, Copyright 2015, Wiley-VCH Verlag GmbH & Co. KGaA. (h)–(k) Reprinted (adapted) with permission from ref. 47, Copyright 2014, Wiley-VCH Verlag GmbH & Co. KGaA. | ||
RENPs have been established as NIR-II fluorescent probes with sharp absorption and emission peaks. By changing the RE dopants, the NIR-II emission peak of RENPs can be precisely tuned. Part of the absorbed light could be converted into radiative fluorescence emissions, while other parts are converted into non-radiative forms of energy that can be used for PAI. Consequently, lost non-radiative energy that was previously considered to be a disadvantage of fluorescent probes can now be harnessed to contribute to the enhancement of the PA signal. Typically, fluorescence and PA signals are complementary forms of signal. Materials that exhibit bright fluorescence emissions would have weak PA signals, whereas weak fluorescence emissions would be accompanied by strong PA signals. A recent study indicates that both NIR-II fluorescence emissions and the amplitude of PA signals can be increased by controlling the size and manipulating the thickness of the inorganic shell coating.36 Size-dependent Ag2S nanodots (Ag2S-NDs) for PAI/NIR-II imaging and simultaneous photothermal therapy have recently been synthesized via the precisely controlled growth of Ag2S in albumin nanocages.37 Ag2S-NDs produce size-dependent NIR-II fluorescence and high photothermal conversion in the blood circulation. Ag2S-NDs exhibit high resistance to photobleaching, good cellular uptake, preferential accumulation in tumours and in vivo elimination. All these advantages enable Ag2S-NDs to be utilized in ultrasensitive fluorescence imaging in the NIR-II region, PAI with microscopic spatial resolution and photothermal ablation of tumours.
(2) Two-component-based multimodality imaging
The typical design of a multimodality imaging probe involves the assembly of multiple reporters and their integration into one entity. Recently, Li et al. reported a Gd–Ag2S nanoprobe that combines the deep tissue penetration capability of MRI with the high spatial and temporal resolution of NIR-II fluorescence imaging (Fig. 4b–d).38 The location of a brain tumour can be clearly identified preoperatively using an MRI signal generated from the Gd component of the nanoprobe, and then precise resection of the tumour can be guided by intraoperative NIR-II fluorescence imaging enabled by Ag2S. The integration of MRI and NIR-II fluorescence imaging techniques for tumour detection and resection provides opportunities for future clinical applications. In comparison with Gd, iron oxide nanoparticles (IONPs) have much higher relaxivities and are often used for T2-weighted imaging. An MRI/NIR-II imaging probe comprising IONPs and DNA-encapsulated SWCNT complexes has been successfully prepared to exhibit NIR-II emissions, a high magnetic relaxation time scale ratio (T1/T2 = 12) and a long spin–spin relaxation time (T2 ≈ 164 ms), which is better than that of typical ferromagnetic nanoparticles.39 A novel PET/NIR-II dual-modal imaging platform using 68Ga-SCH2 was recently developed using a highly efficient base-catalyzed selective assembly method.11 This small-molecule-based PET/NIR-II probe can be successfully used for αVβ3-targeted tumor imaging. The excellent properties of NIR-II/PET dual-modal imaging, such as a high signal-to-background ratio and specificity, enable tumour-free resection in small animal models. These inspiring results show high potential for tumor surgery and translation into clinical use.A new PAI/NIR-II nanoprobe, namely, ICG@PEG–Ag2S, has recently been fabricated by the simple self-assembly of dot-based theranostic Ag2S (DT-Ag2S) QDs, amphipathic C18-PEG and ICG.40 This novel PAI/NIR-II nanoprobe is retained in the blood for up to 6.88 h. The probe selectively accumulated in the region of atherosclerotic plaques because of the attraction of the lipophilic C18 chain to the atherosclerosis microenvironment and provided a contrast-enhanced PA signal. The use of Ag2S QDs enabled fluorescence imaging with high spatial resolution in the NIR-II window in real time. The combination of dyes with high NIR absorption and NIR-II imaging should enable the development of novel strategies for the design of multimodal probes for the imaging, targeting and treatment of disease in clinical situations.
5. NIR-II-based treatment and theranostics
Because many types of imaging techniques can be combined with NIR-II imaging, as described above, numerous developments that focus on the integration of diagnosis and therapy have also been pursued. By incorporating a variety of techniques, theranostics has shown promise for providing effective targeted therapies and enhancing the efficacy of treatment. As a newly emerging optical imaging technique, NIR-II imaging demonstrates great promise for diagnosing diseases, monitoring the development of disease, guiding surgery, and evaluating the efficacy of treatment. Furthermore, NIR-II imaging has also been investigated for combination with treatment modalities to achieve disease theranostics.5.1 Photothermal therapy guided by NIR-II imaging (PTT/NIR-II)
PTT employs photoabsorbers to generate heat, which results in limitation of the growth of cancer cells upon NIR laser irradiation. In comparison with traditional therapeutic modalities, PTT displays excellent performance in terms of not only high specificity but also precise spatial and temporal selectivity. The efficacy of PTT largely depends on the ability of the photothermal agent to transform light into sufficient heat. Single NIR-II imaging or multimodal imaging based on NIR-II can provide real-time guidance for PTT for better planning of therapy before and during PTT. In addition, targeting NIR-II fluorophores also treat disease with high specificity and high sensitivity. Imaging can minimize side effects by enabling specific irradiation of a tumour and improve therapeutic efficacy by enabling the irradiation time to be optimized.SPNPs are promising candidates for use as NIR-II-absorbing materials. Increasing the π-conjugation length by designing quinoid-stabilized D–A polymers is an effective strategy for reducing the bandgap and provides a beneficial bathochromic shift in the absorption to the NIR-II window. Recently, a narrow-bandgap D–A conjugated polymer with thiophene-fused benzodifurandione-based oligo(p-phenylenevinylene) as the acceptor and 2,2′-bithiophene as the donor (TBDOPV-DT) has been synthesized and exploited as a solution-processable NIR-II (peak at 1102 nm) photothermal material.41 TBDOPV-DT has a high extinction coefficient of up to 62.57 L g−1 cm−1 at a wavelength of 1102 nm, which enables high photon utilization efficiency in the NIR-II window. This novel NIR-II-absorbing theranostics probe also exhibits photothermal conversion efficiency of as high as 50.5% with a low power of 0.45 W under excitation at 1102 nm. The high-performance photothermal conversion can be extended to photothermal/electrical and photothermal/mechanical conversions for more convenient use in biological systems or for remote manipulation.
Stoichiometric semiconducting metal sulphide QDs, such as Ag2S nanocrystals, are not only used for NIR-II fluorescence imaging but are also promising in PTT because of their excellent NIR absorbance and high photothermal conversion efficiency. For example, hollow nanocage multifunctional Ag2S nanodots were synthesized by precisely controlled crystal growth in the presence of human serum albumin.37 Ag2S nanodots exhibit size-dependent increases in temperature and have high photothermal conversion efficiencies of 33.7–35.0%, which are comparable to those of most other photothermal nanoparticles, such as Au nanorods (up to 22.8%) and ICG-loaded micelles (up to 25.2%). Ag2S nanodots also produce remarkable fluorescence in the NIR-II region and distinct photoacoustic signals under irradiation with single-wavelength NIR light and thereby generate sufficient in vivo NIR-II fluorescence and PA signals, as well as hyperthermia at tumours.
To improve NIR-II absorbance and photothermal transduction efficiency, a dual plasmonic hybrid system, namely, Au–Cu9S5 NP, has been developed and investigated (Fig. 4e and f).42 The localized surface plasmon resonance (LSPR) of Cu9S5 contributes to 50% of the NIR-II absorption cross-section, and the photothermal transduction efficiency is as high as 37%. This study can improve the future chemical design and optimization of NIR-II absorbers and photothermal heaters by utilizing SPR enhancement phenomena for a broad range of applications. However, high doses of Cu and Ag may not be biotoxic in in vivo applications. Besides these Cu-based nanomaterials with inherent LSPR, there are, however, very few other types of inorganic semiconducting nanomaterials that exhibit strong LSPR. Therefore, it is a challenge to design and develop heavy-metal-free semiconducting photothermal nanomaterials.
Titanium is a transition metal element that has been used as a key constituent of metal alloys in implantable medical devices. When exposed to UV light or radioluminescence, TiO2 nanoparticles can produce cytotoxic hydroxyl and superoxide radicals for eradicating cancer cells via photodynamic therapy (PDT). Many efforts have been made to extend the photoabsorption range of TiO2 from the UV to the visible-NIR region. Strategies have included the preparation of TiO2 nanoparticles with defects, doping and/or surface modification by hydrogenation, arc-melting or pulsed laser irradiation. Recently, NIR-II-active Nb-doped TiO2 nanocrystals have been synthesized by methanol-assisted thermal decomposition.43 In comparison with white TiO2, blue Nb-doped TiO2 nanocrystals exhibit an intense NIR-II absorption band (1064 nm), high photothermal conversion efficiency (up to 40.6%) and excellent stability. Cancer cells can be thermally ablated under irradiation with a 1064 nm laser, which results in the elimination of tumours and longer lifespans in mice. This new finding provides insights into the tuning of other wide-bandgap semiconductors without NIR absorption to provide a strong photothermal effect.
Two-dimensional (2D) nanomaterials have attracted much attention in the past decade because of their ultrathin structure and fascinating physicochemical properties. However, current studies of 2D materials mainly focus on PTT in the NIR-I window, and there are very few relevant reports regarding the photothermal effect of 2D nanomaterials in the NIR-II window. With the inspiration of the great potential of 2D nanomaterials for biomedical applications, a novel ultrathin 2D niobium carbide (Nb2C) has been developed as a novel phototherapeutic agent for the highly efficient in vivo photothermal ablation of mouse tumour xenografts in both the NIR-I and the NIR-II window.44 Ultrathin laterally nanosized Nb2C NSs exhibit extraordinarily high photothermal conversion efficiency (36.4% in the NIR-I window and 45.65% in the NIR-II window), as well as high photothermal stability. In addition, another 2D nanoplatform based on Cu2MnS2 nanoplates (NPs) for MRI/multispectral optoacoustic tomography (MSOT) dual-modal imaging-guided PTT of cancer in the NIR-II window has recently been developed using a facile and effective solvothermal method.45 The Cu2MnS2 NPs exhibit low cytotoxicity, as well as high photothermal conversion efficiency (∼49.38%) and outstanding photostability. Together with their good T1-shortening effect and strong absorbance in the NIR-I and NIR-II regions, the Cu2MnS2 NPs display high-contrast imaging performance in both MRI and MSOT (900 nm laser source). Moreover, the Cu2MnS2 NPs possess excellent PTT efficiency with a low power density (0.6 W cm−2) under irradiation with a 1064 nm laser. By rationally designing their compositions and investigating related physicochemical properties, these studies provide new perspectives for broadening the applications of 2D materials in phototherapy of cancer.
The process of hyperthermia at a single-cell level is very difficult to control. Intracellular thermal sensing provides an innovative way to investigate nanostructured systems capable of high resolution. A new thermal sensing structure, which is a combination of Nd(III)-doped NaGdF4 dielectric RENPs and semiconducting PbS/CdS/ZnS QDs stabilized by poly(lactic-co-glycolic acid) (PLGA), has recently been reported (Fig. 4g).46 The NIR-II emission intensity of these hybrid nanostructures from 1000 to 1400 nm is strongly temperature-dependent at 1220 nm but not at 1060 nm. The coexistence of these two NIR-II luminescence bands makes these structures excellent ratiometric thermal sensors. The unique NIR-II thermal sensitivity (∼2.5 × 10−2 °C−1) of the hybrid nanostructures is an order of magnitude higher than that of nanothermometers utilized in the NIR-I region.
5.2 NIR-II-based photodynamic therapy (PDT/NIR-II)
PDT is a phototherapy that uses light and a photosensitizing chemical substance to treat abnormalities. Light, a photosensitizer, and oxygen are the three key components in a typical PDT process. The photosensitizer can produce reactive oxygen species (ROS) under excitation with the appropriate light wavelength, such as singlet oxygen, free radicals or peroxides, which leads to irreversible antioxidative treatment of cancer cells or inflammation. PDT treatment is carried out only under light irradiation, which is generally localized in a small area. Clinical treatment using PDT is currently limited to surface tumours because most organic photosensitizers are only activated by UV or visible light, which has high photon energy but very short tissue penetration depths. Thus far, very few nanomaterials have been able to be sensitized for the formation of singlet oxygen or ROS and to exert PDT effects in the second biological window (1000–1700 nm), because most NIR-II excited states do not have enough energy to produce singlet oxygen. However, a recent study reported that the morphology of the sensitizers and plasmonic nanomaterials may play a significant role in the formation of singlet oxygen and ROS. A schematic illustration of the PDT process using NIR-II light is shown in Scheme 3a.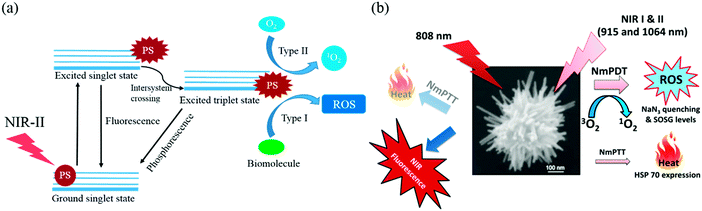 | ||
| Scheme 3 Schematic illustration of PDT process using NIR-II light. (a) Type I and Type II mechanism reactions in PDT using NIR-II light. After absorption of NIR-II light, the PS reaches an excited singlet state. After intersystem crossing, the PS, now in an excited triplet state, can react in two ways: with biomolecules via the transfer of a hydrogen atom (electron) to form radicals, which react with molecular oxygen to generate ROS (Type I reaction); or the PS in its triplet state can react directly with oxygen via energy transfer to generate singlet oxygen (Type II reaction). PS: photosensitizer; ROS: reactive oxygen species; 1O2: singlet oxygen. (b) Schematic representation of the working mechanisms of NIR light-induced fluorescence emission, as well as phototherapeutic effects exerted by a gold nanoechinus.47 | ||
For example, a novel gold nanoechinus was developed with a high extinction coefficient (12 M−1 cm−1) in the NIR region (800–1700 nm), which is about 7–9 times higher than those of traditional organic dyes and 3–4 times higher than those of traditional gold nanoparticles or nanorods. This novel nanoagent could also be used as a PTT/PDT dual-modal agent for the simultaneous treatment of tumours using NIR-I (915 nm) and NIR-II (1064 nm) radiation, as shown in Scheme 3b. Moreover, as an effective NIR-II photosensitizer, it has great potential to monitor and destroy tumours in deep tissue in vivo (up to 1 cm) (Fig. 4h–k).47 With the inspiration of this result, transition metal oxide semiconducting nanomaterials comprising oxygen-deficient molybdenum oxide nanoparticles with localized surface plasmon resonance were synthesized. Exposure to irradiation at 1064 nm can not only effectively convert light into heat but also sensitize the formation of ROS, which enable dramatic cancer cell death and suppression in vivo owing to the synergistic effect of PTT and PDT.48 In general, the mechanisms of the production of ROS by plasmonic nanomaterials under NIR irradiation are very complicated. It has been reported that one of the plasmon-activated pathways proceeds mainly via an indirect photothermal process in plasmonic nanomaterials under NIR laser irradiation, which generates extreme amounts of heat that lead to particle fragmentation and an increase in the emission of thermionic electrons. The higher temperature of the NP solution under irradiation at 1064 nm may be the main reason for the clear production of 1O2. The detailed mechanism of such production of ROS that depends on the NIR-II excitation wavelength still requires further study in the future.
5.3 Intraoperative surgical guidance by NIR-II imaging
In clinical surgical oncology, the delineation of the tumour margin is very important for precise resection of tumours. MRI and CT have a considerable role in guiding intraoperative resection plans. However, these systems are very expensive and complex and cannot be easily used during the surgical procedure. Currently, optesthesia and touching remain the dominant ways to decide which parts should be resected or preserved. Optical molecular imaging is a promising technique with high sensitivity in the identification of tumour margins. In comparison with other imaging modalities, fluorescence imaging offers real-time imaging, superior resolution and high sensitivity and specificity for very small tumours. However, current fluorophores that are widely used for intraoperative surgery are excited by UV or visible light, which causes photobleaching and poor resolution. These fluorophores also have a low penetration depth and are not able to locate deeper tumours. NIR-II imaging enables not only increased spatial resolution but also tissue penetration depth of the order of cm and rapid identification of diseases in real time for surgical guidance. Furthermore, NIR-II light minimizes the scope for misjudgement by doctors because it is invisible to the human eye and only the area excited by NIR-II light is visible and displayed on the monitor.In one study, SWCNTs were employed in NIR-II imaging guidance for cancer resection.34 Many sub-millimeter-sized tumour nodules at a maximum depth of 1.8 cm have been discovered using an NIR-II image-guided system in comparison with a non-image-guided system. However, for large tumours (>3 mm in diameter) there is no difference regardless of whether an image guided system is used, which indicates that SWCNTs are superior for detecting small tumours at earlier stages. In addition, NIR-II imaging involves low tissue autofluorescence, which enhances target-to-background ratios and enables the detection of small tumour nodules in confined anatomical regions. The NIR-II small-molecule-based probes H1 and CQS1000 also demonstrate great potential for NIR-II image-guided surgery for the intraoperative resection of tumours in deep tissue.3,15 Under NIR-II imaging, SLNs can be successfully detected in deep soft tissue. Moreover, the edge of an SLN is easily distinguished, which avoids unnecessary damage to nerves, vessels and tendons.
5.4 NIR-II-based drug delivery
In comparison with traditional therapeutic agents, controllable drug delivery systems, including polymeric nanoparticles, liposome particles, nanoemulsions and mesoporous silica particles, offer many advantages, such as high loading capacities, efficient protection of bioactive drugs and high therapeutic indices. A novel type of RENP-based NIR-II fluorescent mesoporous nanocarrier for the oral delivery of protein drugs has recently been reported.49 The fate of the nanocarrier can be tracked and the amount of the drug released in different organs can be monitored semiquantitatively in real time by measuring NIR-II signals using an absorption competition-induced emission bioimaging system. The results of NIR-II bioimaging demonstrate that protein drugs display little release in neutral and acidic organs (e.g. the stomach and duodenum) but sustained release in alkaline organs (e.g. the intestine). The nanocarriers have a prolonged residence time of up to three days in the intestine, with little deposition (<0.1%) in other organs such as the liver, spleen and kidney. The gastrointestinal effective percentage of protein released from the nanocarriers reaches 62% after three days of release, whereas the activity of the enzymes released from the orally delivered nanocarriers is largely preserved. For NIR-II-based orally administered drug delivery systems, this provides new design strategies.Ag2S QDs possess appealing features for in vivo NIR-II imaging, with a greater tissue penetration depth (up to 1.2 cm), high temporal resolution (down to 50 ms) and high spatial resolution (down to 25 μm). Ag2S QDs loaded with drugs also exhibit perfect drug release under excitation with NIR-II light. For example, a smart Ag2S QD nanoplatform (DOX@PEG–Ag2S) has been obtained by loading the anticancer drug doxorubicin (DOX) into polyethylene glycol-functionalized Ag2S QDs (PEG–Ag2S QDs). The Ag2S QDs had a drug loading capability of up to 93 wt%, a long circulating half-life in blood (10.3 h) and a passive tumour-targeting efficiency of greater than 8.9% ID per g in living mice.50 After accumulating in the tumour tissue, drug cargoes from PEG–Ag2S are rapidly delivered into cancer cells, which causes significant tumour inhibition.
NIR-II-based agents are among promising approaches for photocontrolled drug delivery. During drug delivery, transient or intermittent NIR-II exposure strategies may be more appropriate and could minimize or avoid possible deleterious thermal effects.
6. Advances in NIR-II imaging systems
An NIR-II imaging system is an image capture setup that acquires NIR-II emission signals from NIR-II fluorophores. The development of NIR-II fluorescence imaging systems has to be in sync with the development of NIR-II fluorophores to fully realize the power of NIR-II imaging, and such systems promote wide-ranging applications of NIR-II fluorophores in preclinical research and for future clinical translation. Unlike conventional imaging modalities such as MRI and CT, NIR-II fluorescence imaging systems are more versatile, possess advantages such as real-time imaging capability (rapid video-frame-rate visual feedback at up to 30 frames per second) and the absence of high-energy and ionizing radiation, and can even be potentially constructed as portable imaging systems.The emergence of NIR-II detectors (e.g. indium gallium arsenide (InGaAs) detectors) that are suitable for the region of 1000–1700 nm makes it possible to develop NIR-II fluorescence imaging systems. Current NIR-II imaging systems are mostly constructed in-house using original equipment manufacturer components such as a continuous-wave NIR laser and an NIR camera together with suitable filters, focusing lenses and collimators that depend on the optical characteristics of the complementary NIR-II fluorophores. In brief, an InGaAs camera is placed at a fixed distance away from a platform; lenses and filters are placed between the camera and platform, whereby the image magnification is adjusted by changing the lens sets and filters; a xenon flashlight is used to resolve the location of the animal; and the excitation source is collimated to provide a uniform and constant radius of the excitation beam.
Currently, two basic types of NIR-II imaging systems have been reported (see Fig. 5a and b). The main differences between these two types of system lie in the different approaches adopted to minimize the thermal influence of the NIR excitation source. In the system reported by Moghe et al.,5 the collimated excitation source is mounted on a translation stage to enable a slow scan across the whole body of the animal. The moving excitation source prevents “overheating” of the animal, because its exposure to the source is limited by setting a rapid scan rate (∼10 to 20 mm s−1). In another design reported by Dai et al.,29 the NIR excitation source is kept in one fixed position. With a suitable selection of appropriate lenses, full-body imaging is achieved at the desired magnification. In this setup, a computer-controlled shutter is incorporated to reduce thermal effects. Lenses and objectives between the camera and animal enable fine adjustment of the position and further magnification of the region of interest. In a further development, an in-house stereotactic animal platform fixed on a motorized translation stage enables digital adjustments of the position of a mouse. Using small-animal NIR-II imaging systems, high spatial (micrometer resolution) and high temporal (<200 ms per frame) resolution can be achieved at great depths.
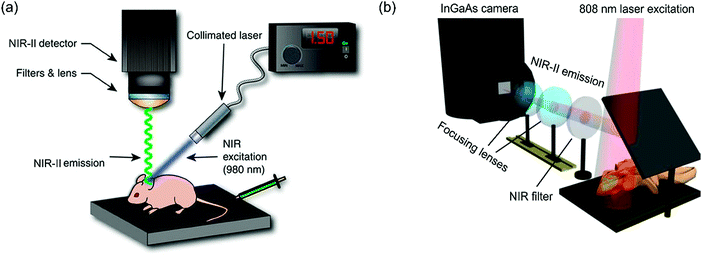 | ||
| Fig. 5 Schematics of NIR-II imaging systems. (a) The NIR-II imaging system reported by Moghe et al. mainly consists of a fiber-coupled NIR laser photodiode and a thermoelectrically cooled InGaAs camera with appropriate filter sets. (b) The NIR-II imaging system reported by Dai et al. mainly consists of a liquid-nitrogen-cooled InGaAs camera, a diode laser coupled to a collimator with a focal length of 4.5 mm with accompanying appropriate filter sets and objectives according to the preferred magnification. (a) Reprinted (adapted) with permission from ref. 5, Copyright 2013, Nature Publishing Group. (b) Reprinted (adapted) with permission from ref. 29, Copyright 2014, Nature Publishing Group. | ||
Interestingly, the NIR-II imaging setup reported by Moghe et al. can acquire images of a whole animal body rapidly, but this largely depends on brightly emitting NIR-II fluorophores because of the shorter acquisition time. The NIR-II setup reported by Dai et al. enables the averaging of multiple images over a smaller area and longer durations, which provides higher-resolution images with a reduced dependence on brightly emitting NIR-II fluorophores.
Moreover, there are also different approaches for reducing the thermal influence on the NIR-II detector. Moghe et al. used a thermoelectrically cooled InGaAs camera detector (Sensors Unlimited, Goodrich, Princeton, NJ), whereas Dai et al. used a liquid-nitrogen-cooled InGaAs camera unit (Princeton Instruments, Princeton, NJ), which is typically more stable with a lower dark count and higher signal-to-noise ratio. Using a liquid-nitrogen-cooled camera unit also enables the detection of relatively weaker NIR-II emission signals.
Although recent advances in NIR-II fluorescence systems have been achieved, a multimodal imaging approach would enable more accurate diagnosis with a potential reduction in false positives or negatives. However, the integration of two or more imaging systems into one device platform remains a challenge. The difficulties in designing multimodal imaging systems arise from hardware incompatibilities and the distinctly different imaging principles of diverse imaging modalities, which lead to a difference in image acquisition timescales between each imaging modality.
7. Perspectives
Thus far, various NIR-II fluorophores ranging from small-molecule dyes to small-molecule protein complexes, small-molecule-encapsulated organic nanoparticles, D–A copolymers, and inorganic particles (QDs, RENPs, SWCNTs) have been designed and synthesized. During the development of NIR-II fluorophores and NIR-II-based multimodal imaging and theranostics, several key points should be noted.(1) Clinical translation of NIR-II imaging: besides applications in preclinical research and in vitro assays, it is paramount to translate NIR-II imaging techniques into clinical use. Importantly, the conventional NIR-I dye ICG has been demonstrated to be suitable for NIR-II imaging, which makes the rapid clinical translation of NIR-II imaging highly feasible. However, ICG may suffer from some limitations. Firstly, the tail portion of the emission spectrum of ICG is used for NIR-II imaging, and thus the efficiency of photon utilization is not high. Secondly, ICG has no targeting specificity, and the clinical indications approved by the FDA are rather limited. Thirdly, the photostability of ICG is not high. Fourthly, the in vivo pharmacokinetics of ICG is not ideal for many applications. All these problems demand the continued synthesis of other novel small-molecule NIR-II dyes for a broad range of clinical uses.
(2) Aim at synthesis of new NIR-II fluorophores: a high quantum yield and photostability, non-biotoxicity, and functionalizability are the principal important criteria to be considered in designing a new generation of NIR-II fluorophores. In addition, the direct chemical design of NIR-II fluorophores with targeting functions, rather than post-synthetic surface-targeting modifications, is more effective in improving therapeutic efficacy. The chemical design and synthesis of biodegradable NIR-II fluorophores with rationally controlled degradation rates should also be encouraged. Among recently developed NIR-II fluorophores, small-molecule dye-based organic nanoparticles and complexes have promising potential for translation from small-animal studies into clinical use. However, the biological use of these NIR-II fluorophores was only recently reported, and further improvements are required, including improving their quantum yields and molar attenuation coefficient, as well as their synthesis process according to the FDA's current GMP regulations.
(3) Aim at NIR-II-based multimodal imaging: considering that PET can be used in preoperative imaging and NIR-II can be used in intraoperative real-time imaging with high resolution, PET/NIR-II imaging will be likely to have a great impact on patient imaging and image-guided therapy. It would be important to test the advantages of PET/NIR-II dual-modal probes in clinical use. In addition, it is expected that PAI/NIR-II dual-modal imaging will make rapid progress, because many dyes can serve as reporters for both PAI and optical imaging. However, the integration of several imaging modalities into one system is a great challenge owing to the differences in hardware and imaging mechanisms. At present, there are no reports of NIR-II-based imaging systems that combine more than two modalities (e.g. NIR-II/MRI/PET or NIR-II/PET/PAI), but such multimodal imaging systems hold great promise for acquiring complementary information. For example, with an NIR-II/MRI/PET trimodal imaging system, the high sensitivity and unlimited penetration depth of PET (10−11–10−12 M) could be used for whole-body screening to identify regions of interest roughly and then reduce the volume of tissue that needs to be scanned. MRI involves no radiation, and high-resolution images could be obtained. NIR-II imaging would provide real-time imaging and molecular and functional information on diseases.
(4) Aim at NIR-II-based treatment and theranostics: it is important to note that most NIR-II fluorophores exhibit low quantum yields. One important reason is that absorbed photon energy is likely to be converted into thermal energy. Thus, NIR-II fluorophores are potential sources for enabling photothermal therapy. Few NIR-II fluorophores have higher triplet electronic energy levels than the singlet energy level of an oxygen molecule, and it may be a great challenge to achieve photodynamic therapy using NIR-II fluorophores. Moreover, it would be important to keep a rational balance between NIR-II emissions and non-radiative emissions according to different requirements. The doses of NIR-II fluorophores should receive careful consideration in both imaging and therapy. For example, a large amount of an NIR-II fluorophore may result in fluorescence quenching but may be useful in enhancing thermal conversion efficiency, in particular in PTT and PAI applications. In intraoperative NIR-II image-guided surgery, it will be helpful to integrate other clinical imaging modalities, such as PET or MRI, which will provide more information beyond that available via single NIR-II imaging.
(5) Aim at NIR-II imaging systems: benefiting from an NIR-II imaging platform is very reliant on an understanding of the requirements of each imaging platform and the physical characteristics of the NIR-II fluorophores. Besides, the hardware platforms (scan rate, lenses, collimators, working distance, exposure duration) and software algorithms for signal processing and image acquisition need to be designed to reduce undesired thermal noise often associated with NIR-II imaging and to increase the signal-to-noise ratio.
On the way to constructing novel clinically translatable NIR-II fluorophores, many challenges remain but will be overcome on the basis of previous success in the development of NIR-II probes and the use of an FDA-approved NIR-I dye for NIR-II imaging. It is hoped that promising multifunctional NIR-II fluorophores that will enable multimodal imaging and theranostics will make a significant contribution to human healthcare.
8. Conclusions
In summary, NIR-II imaging, as a newly emerging modality, has several advantages over conventional NIR-I imaging and other current medical imaging modalities. Significant improvements in imaging resolution and depth can be achieved with NIR-II imaging because of reduced photon scattering and low autofluorescence background at longer wavelengths. NIR-II imaging enables the visualization of deeper anatomical structures with much higher clarity, which is not feasible with NIR-I imaging. Numerous NIR-II fluorophores from small organic molecules to organic and inorganic nanoparticles have been successfully developed, and their wide-ranging biomedical applications such as multimodal imaging, photothermal and photodynamic therapy, guidance for intraoperative surgery, and drug delivery have been demonstrated. The crucial breakthrough in research on NIR-II fluorophores and imaging systems over the past decade highlights the great impact of this technique on basic research and, potentially, clinical theranostics of diseases.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was partially supported by the Office of Science (BER), US Department of Energy (DE-SC0008397); the National Natural Science Foundation of China (61775145, 31771584, 61605124, 61620106016, 61525503, 81727804, 61605130 and 51602201); Guangdong Natural Science Foundation Innovation Team (2014A030312008); the National Basic Research Program of China (Grant 2015CB352005); Hong Kong, Macao, and Taiwan cooperation innovation platform & major projects of international cooperation in Colleges and Universities in Guangdong Province (2015KGJHZ002); and Shenzhen Basic Research Project (JCYJ20170412110212234, JCYJ20160308093035903, JCYJ20150930104948169, JCYJ20160328144746940, GJHZ20160226202139185).References
- A. B. Eva Hemmer, F. Légaréa and F. Vetrone, Nanoscale Horiz., 2016, 1, 168–184 RSC.
- G. Hong, A. L. Antaris and H. Dai, Nat. Biomed. Eng., 2017, 1, 0010 CrossRef.
- K. Shou, C. Qu, Y. Sun, H. Chen, S. Chen, L. Zhang, H. Xu, X. Hong, A. Yu and Z. Cheng, Adv. Funct. Mater., 2017, 27, 1700995 CrossRef PubMed.
- A. L. Antaris, H. Chen, K. Cheng, Y. Sun, G. Hong, C. Qu, S. Diao, Z. Deng, X. Hu, B. Zhang, X. Zhang, O. K. Yaghi, Z. R. Alamparambil, X. Hong, Z. Cheng and H. Dai, Nat. Mater., 2015, 15, 235–242 CrossRef PubMed.
- D. J. Naczynski, M. C. Tan, M. Zevon, B. Wall, J. Kohl, A. Kulesa, S. Chen, C. M. Roth, R. E. Riman and P. V. Moghe, Nat. Commun., 2013, 4, 1–10 Search PubMed.
- K. Welsher, Z. Liu, S. P. Sherlock, J. T. Robinson, Z. Chen, D. Daranciang and H. Dai, Nat. Nanotechnol., 2009, 4, 773–780 CrossRef CAS PubMed.
- Z. Starosolski, R. Bhavane, K. B. Ghaghada, S. A. Vasudevan, A. Kaay and A. Annapragada, PLoS One, 2017, 12, e0187563 Search PubMed.
- J. A. Carr, D. Franke, J. R. Caram, C. F. Perkinson, M. Saif, V. Askoxylakis, M. Datta, D. Fukumura, R. K. Jain, M. G. Bawendi and O. T. Bruns, Proc. Natl. Acad. Sci. U. S. A., 2018, 115, 4465–4470 CrossRef PubMed.
- A. L. Antaris, H. Chen, S. Diao, Z. Ma, Z. Zhang, S. Zhu, J. Wang, A. X. Lozano, Q. Fan, L. Chew, M. Zhu, K. Cheng, X. Hong, H. Dai and Z. Cheng, Nat. Commun., 2017, 8, 15269 CrossRef CAS PubMed.
- Y. Sun, C. Qu, H. Chen, M. He, C. Tang, K. Shou, S. Hong, M. Yang, Y. Jiang, B. Ding, Y. Xiao, L. Xing, X. Hong and Z. Cheng, Chem. Sci., 2016, 7, 6203–6207 RSC.
- Y. Sun, X. Zeng, Y. Xiao, C. Liu, H. Zhu, H. Zhou, Z. Chen, F. Xu, J. Wang, M. Zhu, J. Wu, M. Tian, H. Zhang, Z. Deng, Z. Cheng and X. Hong, Chem. Sci., 2018, 9, 2092–2097 RSC.
- X.-D. Zhang, H. Wang, A. L. Antaris, L. Li, S. Diao, R. Ma, A. Nguyen, G. Hong, Z. Ma, J. Wang, S. Zhu, J. M. Castellano, T. Wyss-Coray, Y. Liang, J. Luo and H. Dai, Adv. Mater., 2016, 28, 6872–6879 CrossRef CAS PubMed.
- S. Zhu, Q. Yang, A. L. Antaris, J. Yue, Z. Ma, H. Wang, W. Huang, H. Wan, J. Wang, S. Diao, B. Zhang, X. Li, Y. Zhong, K. Yu, G. Hong, J. Luo, Y. Liang and H. Dai, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 962–967 CrossRef CAS PubMed.
- Y. Feng, S. Zhu, A. L. Antaris, H. Chen, Y. Xiao, X. Lu, L. Jiang, S. Diao, K. Yu, Y. Wang, S. Herraiz, J. Yue, X. Hong, G. Hong, Z. Cheng, H. Dai and A. J. Hsueh, Chem. Sci., 2017, 8, 3703–3711 RSC.
- Y. Sun, M. Ding, X. Zeng, Y. Xiao, H. Wu, H. Zhou, B. Ding, C. Qu, W. Hou, A. G. A. Er-bu, Y. Zhang, Z. Cheng and X. Hong, Chem. Sci., 2017, 8, 3489–3493 RSC.
- Z. Tao, G. Hong, C. Shinji, C. Chen, S. Diao, A. L. Antaris, B. Zhang, Y. Zou and H. Dai, Angew. Chem., Int. Ed., 2013, 52, 13002–13006 CrossRef CAS PubMed.
- G. Hong, Y. Zou, A. L. Antaris, S. Diao, D. Wu, K. Cheng, X. Zhang, C. Chen, B. Liu, Y. He, J. Z. Wu, J. Yuan, B. Zhang, Z. Tao, C. Fukunaga and H. Dai, Nat. Commun., 2014, 5, 4206 CAS.
- K. Shou, C. Qu, Y. Sun, H. Chen, S. Chen, L. Zhang, H. Xu, X. Hong, A. Yu and Z. Cheng, Adv. Funct. Mater., 2017, 27, 1700995 CrossRef PubMed.
- G. Hong, J. T. Robinson, Y. Zhang, S. Diao, A. L. Antaris, Q. Wang and H. Dai, Angew. Chem., Int. Ed., 2012, 124, 9956–9959 CrossRef.
- C. Li, Y. Zhang, M. Wang, Y. Zhang, G. Chen, L. Li, D. Wu and Q. Wang, Biomaterials, 2014, 35, 393–400 CrossRef CAS PubMed.
- G. Chen, F. Tian, Y. Zhang, Y. Zhang, C. Li and Q. Wang, Adv. Funct. Mater., 2014, 24, 2481–2488 CrossRef CAS.
- R. Tang, J. Xue, B. Xu, D. Shen, G. P. Sudlow and S. Achilefu, ACS Nano, 2015, 9, 220–230 CrossRef CAS PubMed.
- M. Kamimura, N. Kanayama, K. Tokuzen, K. Soga and Y. Nagasaki, Nanoscale, 2011, 3, 3705–3713 RSC.
- M. Zevon, V. Ganapathy, H. Kantamneni, M. Mingozzi, P. Kim, D. Adler, Y. Sheng, M. C. Tan, M. Pierce, R. E. Riman, C. M. Roth and P. V. Moghe, Small, 2015, 11, 6347–6357 CrossRef CAS PubMed.
- W. Shao, G. Chen, A. Kuzmin, H. L. Kutscher, A. Pliss, T. Y. Ohulchanskyy and P. N. Prasad, J. Am. Chem. Soc., 2016, 138, 16192–16195 CrossRef CAS PubMed.
- J. T. Robinson, G. Hong, Y. Liang, B. Zhang, O. K. Yaghi and H. Dai, J. Am. Chem. Soc., 2012, 134, 10664–10669 CrossRef CAS PubMed.
- K. Welsher, S. P. Sherlock and H. Dai, Proc. Natl. Acad. Sci. U. S. A., 2011, 108, 8943–8948 CrossRef CAS PubMed.
- G. Hong, J. C. Lee, J. T. Robinson, U. Raaz, L. Xie, N. F. Huang, J. P. Cooke and H. Dai, Nat. Med., 2012, 18, 1841–1846 CrossRef CAS PubMed.
- G. Hong, S. Diao, J. Chang, A. L. Antaris, C. Chen, B. Zhang, S. Zhao, D. N. Atochin, P. L. Huang, K. I. Andreasson, C. J. Kuo and H. Dai, Nat. Photonics, 2014, 8, 723–730 CrossRef CAS PubMed.
- Y. Yomogida, T. Tanaka, M. Zhang, M. Yudasaka, X. Wei and H. Kataura, Nat. Commun., 2016, 7, 12056 CrossRef CAS PubMed.
- S. Diao, J. L. Blackburn, G. Hong, A. L. Antaris, J. Chang, J. Z. Wu, B. Zhang, K. Cheng, C. J. Kuo and H. Dai, Angew. Chem., Int. Ed., 2015, 54, 14758–14762 CrossRef CAS PubMed.
- N. M. Iverson, P. W. Barone, M. Shandell, L. J. Trudel, S. Sen, F. Sen, V. Ivanov, E. Atolia, E. Farias, T. P. McNicholas, N. Reuel, N. M. A. Parry, G. N. Wogan and M. S. Strano, Nat. Nanotechnol., 2013, 8, 873–880 CrossRef CAS PubMed.
- D. J. Naczynski, C. Sun, S. Türkcan, C. Jenkins, A. L. Koh, D. Ikeda, G. Pratx and L. Xing, Nano Lett., 2015, 15, 96–102 CrossRef CAS PubMed.
- D. Ghosh, A. F. Bagley, Y. J. Na, M. J. Birrer, S. N. Bhatia and A. M. Belcher, Proc. Natl. Acad. Sci. U. S. A., 2014, 111, 13948–13953 CrossRef CAS PubMed.
- K. Cheng, H. Chen, C. H. Jenkins, G. Zhang, W. Zhao, Z. Zhang, F. Han, J. Fung, M. Yang, Y. Jiang, L. Xing and Z. Cheng, ACS Nano, 2017, 11, 12276–12291 CrossRef CAS PubMed.
- Y. Sheng, L.-D. Liao, A. Bandla, Y.-H. Liu, N. Thakor and M. C. Tan, ACS Biomater. Sci. Eng., 2016, 2, 809–817 CrossRef CAS.
- T. Yang, Y. a. Tang, L. Liu, X. Lv, Q. Wang, H. Ke, Y. Deng, H. Yang, X. Yang, G. Liu, Y. Zhao and H. Chen, ACS Nano, 2017, 11, 1848–1857 CrossRef CAS PubMed.
- C. Li, L. Cao, Y. Zhang, P. Yi, M. Wang, B. Tan, Z. Deng, D. Wu and Q. Wang, Small, 2015, 11, 4517–4525 CrossRef CAS PubMed.
- J. H. Choi, F. T. Nguyen, P. W. Barone, D. A. Heller, A. E. Moll, D. Patel, S. A. Boppart and M. S. Strano, Nano Lett., 2007, 7, 861–867 CrossRef CAS PubMed.
- C. Wu, Y. Zhang, Z. Li, C. Li and Q. Wang, Nanoscale, 2016, 8, 12531–12539 RSC.
- Y. Cao, J.-H. Dou, N.-J. Zhao, S. Zhang, Y.-Q. Zheng, J.-P. Zhang, J.-Y. Wang, J. Pei and Y. Wang, Chem. Mater., 2017, 29, 718–725 CrossRef CAS.
- X. Ding, C. H. Liow, M. Zhang, R. Huang, C. Li, H. Shen, M. Liu, Y. Zou, N. Gao, Z. Zhang, Y. Li, Q. Wang, S. Li and J. Jiang, J. Am. Chem. Soc., 2014, 136, 15684–15693 CrossRef CAS PubMed.
- N. Yu, Y. Hu, X. Wang, G. Liu, Z. Wang, Z. Liu, Q. Tian, M. Zhu, X. Shi and Z. Chen, Nanoscale, 2017, 9, 9148–9159 RSC.
- H. Lin, S. Gao, C. Dai, Y. Chen and J. Shi, J. Am. Chem. Soc., 2017, 139, 16235–16247 CrossRef CAS PubMed.
- K. Ke, W. Yang, X. Xie, R. Liu, L.-L. Wang, W.-W. Lin, G. Huang, C.-H. Lu and H.-H. Yang, Theranostics, 2017, 7, 4763–4776 CrossRef PubMed.
- E. N. Cerón, D. H. Ortgies, B. del Rosal, F. Ren, A. Benayas, F. Vetrone, D. Ma, F. Sanz-Rodríguez, J. G. Solé, D. Jaque and E. M. Rodríguez, Adv. Mater., 2015, 27, 4781–4787 CrossRef PubMed.
- P. Vijayaraghavan, C.-H. Liu, R. Vankayala, C.-S. Chiang and K. C. Hwang, Adv. Mater., 2014, 26, 6689–6695 CrossRef CAS PubMed.
- W. Yin, T. Bao, X. Zhang, Q. Gao, J. Yu, X. Dong, L. Yan, Z. Gu and Y. Zhao, Nanoscale, 2018, 10, 1517–1531 RSC.
- R. Wang, L. Zhou, W. Wang, X. Li and F. Zhang, Nat. Commun., 2017, 8, 14702 CrossRef PubMed.
- F. Hu, C. Li, Y. Zhang, M. Wang, D. Wu and Q. Wang, Nano Res., 2015, 8, 1637–1647 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2018 |