Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
How fluorine substituents strengthen aryl C–H bonds
Daniel A. Santos Oliveira ab,
Daniela Rodrigues Silva
ab,
Daniela Rodrigues Silva a,
Ataualpa A. C. Braga
a,
Ataualpa A. C. Braga b,
Célia Fonseca Guerra
b,
Célia Fonseca Guerra a,
Robin N. Perutz
a,
Robin N. Perutz *c,
Odile Eisenstein
*c,
Odile Eisenstein *de and
F. Matthias Bickelhaupt
*de and
F. Matthias Bickelhaupt *afg
*afg
aDepartment of Chemistry and Pharmaceutical Sciences, AIMMS, Vrije Universiteit Amsterdam, De Boelelaan 1108, Amsterdam, 1081 HZ, The Netherlands. E-mail: f.m.bickelhaupt@vu.nl; Web: https://www.theochem.nl
bDepartment of Fundamental Chemistry, Institute of Chemistry, University of São Paulo, Av. Prof. Lineu Prestes, 748, São Paulo, 055508-000, Brazil
cDepartment of Chemistry, University of York, York, YO10 5DD, UK. E-mail: robin.perutz@york.ac.uk
dICGM, Univ. Montpellier, CNRS, ENSCM, Montpellier, France. E-mail: odile.eisenstein@umontpellier.fr
eDepartment of Chemistry and Hylleraas Centre for Quantum Molecular Sciences, University of Oslo, PO Box 1033 Blindern, Oslo, 0315, Norway
fInstitute of Molecules and Materials, Radboud University, Heyendaalseweg 135, Nijmegen, 6525 AJ, The Netherlands
gDepartment of Chemical Sciences, University of Johannesburg, Auckland Park, Johannesburg, 2006, South Africa
First published on 27th April 2026
Abstract
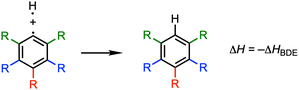
We have investigated the nature and bond dissociation energies (BDEs) of the aromatic C–H bonds in fluorinated benzenes C6R5H (each R can be H or F) using quantitative Kohn–Sham molecular orbital theory and a matching energy decomposition analysis (EDA). The C–H bond becomes stronger as the number of fluorine atoms in the benzene ring increases. This increase in the calculated BDE is additive and most pronounced for ortho-substituted C–H bonds. Our analyses of the C–H bond between 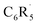 and H˙ reveal that a fluorine inductive effect is responsible for this. Fluorine polarizes the closed-shell molecular orbitals of
and H˙ reveal that a fluorine inductive effect is responsible for this. Fluorine polarizes the closed-shell molecular orbitals of 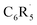 away from the carbon radical center and in this way reduces Pauli repulsion between [C˙] and the H˙ radical, leading to a stronger C–H bond. The ortho effect can be accurately modelled by a combination of Pauli repulsion (main contribution) and orbital interactions. We extend our analysis to other substituents, including ones with the opposite effect on C–H bond strength.
away from the carbon radical center and in this way reduces Pauli repulsion between [C˙] and the H˙ radical, leading to a stronger C–H bond. The ortho effect can be accurately modelled by a combination of Pauli repulsion (main contribution) and orbital interactions. We extend our analysis to other substituents, including ones with the opposite effect on C–H bond strength.
Introduction
Fluorine alters the physical properties of chemical species. For instance, it modifies the lipophilicity and acid–base properties, leading to the adoption of fluorine-containing organic compounds, including numerous F- or CF3-substituted aromatics, in pharmaceuticals, imaging, agrochemicals, and many other areas.1a–g In materials science, fluorinated conducting materials and fluorinated anions are important, especially in rechargeable batteries.1i–k Through its deep influence on the molecular dipole and quadrupole, it can serve as a conformational tool in organic and biological chemistry.1l The formation of the halogen bond is strongly associated with fluorinated organic molecules such as iodopentafluorobenzene.1m Through the modification of intermolecular interactions, it enhances charge mobilities in organic semiconductors.1n Zooming in on some fundamental properties of fluorinated benzenes and polyaromatics, fluorine has been found to decrease the aromaticity of aromatic rings while enhancing π-stacking properties. However, the decrease in aromaticity of the ring is accompanied by increased thermostability and resistance to chemical attack, a phenomenon known as fluoroaromaticity.1o–r The alternating stacking of benzene·hexafluorobenzene cocrystals and related cocrystals in a parallel but displaced geometry was originally attributed to interactions of the different quadrupoles of the two components. Although electrostatics make a major contribution to the interactions, the displaced geometry is suggested to result from Pauli repulsion and dispersion forces, illustrating the effectiveness of Energy Decomposition Analysis in these systems.1sAnother key effect of fluorine substitution is its impact on the strength of aryl C–H bonds, typically quantified by the homolytic bond dissociation energy (BDE), which can be modulated by both the number and the position of fluorine atoms.2 Several groups have investigated the correlations between C–H BDEs in (poly)fluorinated benzenes and C–metal bonds in corresponding metal aryl derivatives, demonstrating that the C–H bond strength increases upon fluorination.3 For instance, the calculated C–H bond dissociation energy (BDE) for pentafluorobenzene (C6F5H: 123.7 kcal mol−1) is about 6 kcal mol−1 higher than that for benzene (C6H6: 117.7 kcal mol−1). Perutz, Eisenstein, Jones and their co-workers have also found that the most significant increases in bond strength occur when the fluorine atom is in the ortho position relative to the C–H bond analyzed.4 For those reactions that are thermodynamically driven, particularly reversible reactions, this can result in ortho regioselectivity as demonstrated in numerous experimental studies.3 These findings are intriguing, since many examples in the literature have shown that neighboring bulky groups tend to weaken the C–H bond through steric repulsion,5 but it is unclear how this will apply to fluorine whose van der Waals radius is only slightly larger than that of hydrogen. Although the bond dissociation energies of (poly)fluorinated benzenes have been reported, a detailed explanation, firmly grounded in quantum mechanics, of why fluorine substituents strengthen C–H bonds in fluorinated benzenes is still lacking in the literature.
In this work, we unravel the physical mechanism underlying the strengthening of C–H bonds in (poly)fluorinated benzenes. We also address the origin of the variation in strengthening of the C–H bonds that follows the order ortho ≫ para > meta.4 To this end, we investigate the C–H bonding nature in the systems C6R5–H (each R can be H or F; see Fig. 1) using quantitative Kohn–Sham molecular orbital theory (KS-MO) combined with a matching energy decomposition analysis (EDA).6,7 Our results reveal that the main factor responsible for the strengthening of the C–H bonds upon fluorination is the inductive polarization by fluorine of the closed-shell molecular orbital density away from the aromatic ring. This lowers the Pauli repulsion in the bonding region and thus strengthens the C–H bond. The EDA analysis reveals that the regioselectivity of the C–H bond energies, especially the ortho/para ratio, can be reproduced in part by the Pauli repulsion but an accurate representation requires the sum of Pauli repulsion and orbital terms. We further include C6RH4–H systems (R = Cl, Br, I, Li) to assess the effect of different substituents beyond fluorine.
Computational methods
Computational details
All calculations were carried out using the Amsterdam Density Functional (ADF) program (ADF2019.3 for Potential Energy Surface (PES) scans and ADF2024.1 for all other computations),8 which is part of the Amsterdam Modeling Suite (AMS2024.1).9 Geometries and energies were obtained with the BLYP level of the generalized gradient approximation (GGA).10 Dispersion interactions were accounted for using the DFT-D3(BJ) method developed by Grimme and coworkers,11 which includes the damping function proposed by Becke and Johnson.12 Scalar relativistic effects were treated using the zeroth-order regular approximation (ZORA).13 Molecular orbitals (MOs) were expanded in a large, uncontracted set of Slater-type orbitals (STOs), specifically the TZ2P basis set, which is of triple-ζ quality and includes two sets of polarization functions.14 All electrons were treated variationally. Radical fragments were treated using a spin-unrestricted formalism. The numerical accuracy15 was set to VERYGOOD. All optimized structures were confirmed as true minima by vibrational frequency analyses, showing no imaginary frequencies.16 The bond energies reported in this work correspond to electronic energies and do not include zero-point energy (ZPE) corrections. Multiple regression analyses were conducted using the statsmodels Python library,17 while graphical visualization of the results was performed with the Matplotlib library.18 The relative bond enthalpies computed at our final level of theory, ZORA-BLYP-D3(BJ)/TZ2P, are in good agreement with experimentally validated literature values,19 supporting the reliability of our methodological approach (see Table S1).Activation strain model and energy decomposition analysis
The overall homolytic C–H bond enthalpy ΔH, corresponding to −ΔHBDE, between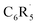 and H˙ in C6R5–H (R = H, F), is shown in Scheme 1. The corresponding value of ΔE is decomposed into two major components using the activation strain model (ASM, eqn (1)):20
and H˙ in C6R5–H (R = H, F), is shown in Scheme 1. The corresponding value of ΔE is decomposed into two major components using the activation strain model (ASM, eqn (1)):20| ΔE = ΔEstrain + ΔEint | (1) |
The interaction energy ΔEint is further analyzed within the framework of the quantitative Kohn–Sham molecular orbital (KS-MO)6 theory by partitioning it using our canonical energy decomposition analysis (EDA) scheme into electrostatic interactions, Pauli repulsion, (attractive) orbital interactions, dispersion corrections, and spin polarization (eqn (2)):7
| ΔEint = ΔVelstat + ΔEPauli + ΔEoi + ΔEdisp + ΔEspinpol | (2) |
The electrostatic energy ΔVelstat corresponds to the electrostatic interactions between the unperturbed charge distribution of the distorted fragments, which is usually attractive. The Pauli repulsion ΔEPauli comprises the destabilizing interactions between occupied orbitals (or, more precisely, same-spin electrons on either fragment) and is responsible for any steric repulsion. The orbital interactions ΔEoi term, accounts for stabilizing orbital interactions between the fragments including both polarization and overlap effects and can be further decomposed into the electron pair-bond energy ΔEpb and ΔErel (eqn (3)). ΔEpb is defined as the energy change associated with the formation of a doubly occupied bonding combination of the two SOMOs while all other virtual orbitals are deleted. The relaxation energy ΔErel, results from full relaxation after including all virtual orbitals. The ΔErel term includes both charge transfer interactions (donor–acceptor interaction between an occupied orbital of one fragment with an empty orbital of the other fragment) and polarization effects (empty/occupied) orbital mixing on one fragment due to the presence of another fragment.7
| ΔEoi = ΔEpb + ΔErel | (3) |
The dispersion energy ΔEdisp is added as a correction.11 Finally, the ΔEspinpol term refers to the spin polarization of the spin-α and spin-β electrons of the deformed unrestricted fragments and is destabilizing (i.e., the deformed unrestricted fragments without spin polarization lie consistently 2–4 kcal mol−1 higher in energy and therefore have a too stabilizing ΔEint).21 The open-source PyFrag2019 program was used to automate analyzing the bonding mechanism as a function of the C–H distance.22
Results and discussion
General trends in bond strength
The bond enthalpies ΔH (see Scheme 1) under standard conditions (298.15 K and 1 atm) and the C–H bond lengths (rC–H) of the C6R5–H model systems (R = H, F), obtained from our ZORA-BLYP-D3(BJ)/TZ2P computations, are collected in Table 1. The computed C–H bond strengths as represented by the homolytic bond enthalpies ΔH, are in excellent agreement with the previously reported results by Clot, Perutz, Eisenstein, and co-workers,3c,4 confirming that the introduction of fluorine atoms into the benzene ring strengthens the C–H bond for all substitution patterns. The calculated bond enthalpies relative to benzene (B) vary from ΔΔH = −0.4 kcal mol−1 for the meta-C–H bond in fluorobenzene (m) to ΔΔH = −6.4 kcal mol−1 for 1,3,5-trifluorobenzene (2o-p). Fluorination of the benzene ring also leads to a shortening of the C–H bonds. The variations are very small but are approximately proportional to the bond strengthening, with stronger bonds generally associated with shorter bond lengths (Fig. S1). The largest contraction of ΔrC–H = −0.004 Å occurs in 2o-p, i.e., the system with the strongest C–H bond.| System | rC–H | ΔH | ΔΔH | ΔE | ΔΔE | ΔEstrain | ΔΔEstrain | ΔEint | ΔΔEint |
|---|---|---|---|---|---|---|---|---|---|
| a Computed at ZORA-BLYP-D3(BJ)/TZ2P, enthalpies ΔH at 298.15 K and 1 atm. Energies relative to benzene (B) are given as ΔΔ. | |||||||||
| B | 1.088 | −109.3 | 0.0 | −115.9 | 0.0 | 1.8 | 0.0 | −117.7 | 0.0 |
| o | 1.086 | −111.8 | −2.5 | −118.4 | −2.5 | 1.8 | 0.0 | −120.2 | −2.5 |
| m | 1.087 | −109.7 | −0.4 | −116.3 | −0.4 | 1.8 | 0.0 | −118.1 | −0.4 |
| p | 1.087 | −110.6 | −1.3 | −117.2 | −1.3 | 1.7 | −0.1 | −118.9 | −1.2 |
| o-m | 1.087 | −111.8 | −2.5 | −118.3 | −2.4 | 1.9 | 0.1 | −120.2 | −2.5 |
| m-p | 1.087 | −110.7 | −1.4 | −117.3 | −1.4 | 1.8 | 0.0 | −119.1 | −1.4 |
| 2o | 1.085 | −114.8 | −5.5 | −121.1 | −5.2 | 1.8 | 0.0 | −122.9 | −5.2 |
| 2m | 1.087 | −110.2 | −0.9 | −116.8 | −0.9 | 1.9 | 0.1 | −118.7 | −1.0 |
| o-p | 1.086 | −113.0 | −3.7 | −119.5 | −3.6 | 1.7 | −0.1 | −121.2 | −3.5 |
| o-m′ | 1.086 | −112.2 | −2.9 | −118.7 | −2.8 | 1.8 | 0.0 | −120.5 | −2.8 |
| o-m-p | 1.086 | −112.7 | −3.4 | −119.2 | −3.3 | 1.9 | 0.1 | −121.1 | −3.4 |
| 2m-p | 1.086 | −110.9 | −1.6 | −117.5 | −1.6 | 1.9 | 0.1 | −119.4 | −1.7 |
| 2o-m | 1.085 | −114.7 | −5.4 | −121.1 | −5.2 | 1.8 | 0.0 | −122.9 | −5.2 |
| o-2m | 1.086 | −112.4 | −3.1 | −118.9 | −3.0 | 1.9 | 0.1 | −120.8 | −3.1 |
| o-m-p′ | 1.085 | −113.1 | −3.8 | −119.6 | −3.7 | 1.8 | 0.0 | −121.4 | −3.7 |
| 2o-p | 1.084 | −115.7 | −6.4 | −122.1 | −6.2 | 1.7 | −0.1 | −123.8 | −6.1 |
| o-2m-p | 1.086 | −113.0 | −3.7 | −119.5 | −3.6 | 1.9 | 0.1 | −121.4 | −3.7 |
| 2o-2m | 1.086 | −115.0 | −5.7 | −121.3 | −5.4 | 1.9 | 0.1 | −123.2 | −5.5 |
| 2o-m-p | 1.085 | −115.5 | −6.2 | −121.8 | −5.9 | 1.8 | 0.0 | −123.6 | −5.9 |
| 2o-2m-p | 1.085 | −115.5 | −6.2 | −121.8 | −5.9 | 1.9 | 0.1 | −123.7 | −6.0 |
As previously reported,4,23 and further confirmed here via multiple regression analysis, the ΔΔH values can be expressed as a linear function (r2 = 0.9890) of the number of ortho- (xortho), meta- (xmeta), and para-fluorine substituents (xpara), as shown in eqn (4) and Fig. 2a.
| ΔΔH = a + bxortho + cxmeta + dxpara | (4) |
 | ||
| Fig. 2 Multiple linear regression relating the number of fluorine substituents at the ortho, meta, and para positions to relative (a) C–H bond enthalpies ΔΔH, (b) electrostatic interactions ΔΔVelstat, (c) Pauli repulsions ΔΔEPauli, and (d) orbital interactions ΔΔEoi; (e) energy decomposition analysis for all EDA terms from regression coefficients for all 20 fluorinated benzenes relative to benzene; and (f) energy decomposition analysis terms in fluorobenzene relative to benzene at a consistent geometry with a C–H distance of 1.088 Å.24 Computed at ZORA-BLYP-D3(BJ)/TZ2P. | ||
The regression coefficients b, c, and d, represent the energy variation associated with the addition of a fluorine atom at the ortho, meta, and para positions, respectively. Accordingly, fluorination at the ortho position strengthens the C–H bond by approximately 2.5 ± 0.1 kcal mol−1, while meta- and para-fluorine substitution increase the bond strength by only 0.2 ± 0.1 and 0.9 ± 0.1 kcal mol−1, respectively. It is worth noting how closely the regression coefficients calculated here match those reported in the literature3c using the B3PW91 functional (a = −0.27 ± 0.12 vs. −0.17 ± 0.07, b = −2.49 ± 0.07 vs. −2.49 ± 0.05, c = −0.15 ± 0.07 vs. −0.07 ± 0.05, and d = −0.81 ± 0.07 vs. −0.87 ± 0.10 kcal mol−1), indicating the robustness of the results with respect to the level of theory. Since these values match so closely with two different functionals, there is no reason to expect significant differences in the EDA analysis. We therefore carried out further analysis with the ZORA-BLYP-D3(BJ)/TZ2P level of theory only.
As shown previously, the ΔΔH values cluster into three groups corresponding to a progressive increase in C–H bond strength as the number of ortho-F substituents increases (0 → 1 → 2). Because the para-coefficient (d = −0.87 kcal mol−1) is significant, a secondary subdivision is observed within each region, distinguishing systems that contain a para-fluorine from those without a para-fluorine. Overall, the linear correlation demonstrates that substituting a fluorine atom for hydrogen at a specific position has an additive effect on the C–H bond strength, with this effect being most pronounced at the ortho position, followed by the para and meta positions, respectively. Here we only analyze the changes in the C–H bond dissociation energy. In contrast,23 the C–F bond dissociation energy also changes but in the opposite direction, such that C–F BDEs decrease markedly with the number of ortho fluorine substituents, with smaller effects from meta- and para-fluorine substitution.
The overall trends in bond enthalpies ΔH are well reproduced by the electronic bond energies ΔE, as shown by the relative ΔΔ values in Table 1 and the multiple regression analyses shown in Fig. S2. Therefore, to elucidate the origin of the C–H bond strengthening in fluorinated benzenes, we analyzed the electronic bond energy ΔE using the Activation Strain Model (ASM).20 Within this framework, ΔE is decomposed into a strain energy ΔEstrain and an interaction energy ΔEint (eqn (1); see Computational methods). The corresponding ASM terms are also listed in Table 1. The only significant geometrical deformation observed upon bond formation is the in-plane bending of the neighboring substituents (H or F) in the aryl fragment. This deformation leads to a small strain energy ΔEstrain, which remains essentially constant across different substitution patterns. Thus, the trends in bond strength (i.e., in both ΔH and ΔE) are entirely governed by the interaction energy ΔEint. We further investigated the underlying C–H bonding mechanism using Kohn–Sham Molecular Orbital theory (KS-MO)6 and a matching Energy Decomposition Analysis (EDA).7 This method decomposes ΔEint into distinct physically meaningful components: electrostatic interactions ΔVelstat, Pauli repulsion ΔEPauli, orbital interactions ΔEoi, among others (see eqn (2) in the Computational methods section). The EDA terms for the C–H bonds in fluorinated benzenes, relative to benzene, can also be expressed as linear functions of the number of ortho- (xortho), meta- (xmeta), and para-fluorine substituents (xpara), as depicted in Fig. 2b–d. The corresponding absolute values are provided in Table S1. The graphs in Fig. 2a–c exhibit the characteristic patterns corresponding to the dominance of the ortho contribution (xortho = 0, 1, or 2) as shown by the rings around the points. Within each subset, we have all possible combinations of meta- and para-Fs (6 species for 0 and 2 ortho-Fs, 7 species for 1 ortho-F). The linear correlations indicate that each para substitution has a similar effect (see above). The contribution from meta substituents is negligible in Fig. 2a and c (ΔΔH and ΔΔEPauli) but is comparable to the para contribution for ΔΔVelstat. The spans of the electrostatic, Pauli, and orbital contributions are 8.5, 13.6, and 3.6 kcal mol−1, respectively (Fig. 2b–d).
The trends in the interaction energy, and therefore in the C–H bond enthalpies, are primarily dictated by the Pauli repulsion ΔEPauli, as shown in Fig. 2c and e and through linear correlation between ΔΔH and ΔΔEPauli (Fig. 3a). Relative to benzene, ΔEPauli is the most stabilizing contribution, becoming less repulsive as the number of ortho-F substituents increases and exhibiting the same clustering pattern observed for the bond enthalpies (Fig. 2c). Fluorination at the ortho position decreases the Pauli repulsion by approximately 4.8 ± 0.2 kcal mol−1, while para-F substitution reduces ΔEPauli by 2.5 ± 0.3 kcal mol−1. The stronger effect of ortho compared to para substitution is quantified by the ratio b/d. The value of b/d for ΔΔH is 2.86 ± 0.33. The corresponding value for ΔΔEPauli is 1.91 ± 0.26 thus accounting for approximately 2/3 of the ortho preference.25 Similar to the bond enthalpy, fluorination at the meta position has almost no effect on the Pauli repulsion.
In contrast to ΔEPauli, the electrostatic interaction term ΔVelstat follows the opposite trend to ΔH, becoming less attractive as the number of ortho-F substituents increases, as indicated by the positive regression coefficients (Fig. 2b). The magnitude of the electrostatic effects follows the pattern ortho ≫ meta > para, which may be ascribed to simple distance effects, 1/r, arising because of the 9 electrons of fluorine compared to 1 of hydrogen. Finally, unlike the other EDA terms, the orbital interaction term ΔEoi does not exhibit the same clustering pattern based on the number of ortho-F substituents and therefore follows a different trend from the bond strength (Fig. 2d). In fact, the stabilization upon fluorine substitution is larger for the meta position than for the ortho position (−0.9 ± 0.1 versus −0.5 ± 0.1 kcal mol−1, respectively). In contrast, para-F substitution leads to a destabilization of 0.8 ± 0.2 kcal mol−1 in the orbital interaction term. This destabilizing contribution at the para position accounts for the smaller, remaining difference between the ortho- and para-C–H bond strengths, as evidenced by the improved correlation coefficient and the closer match between the b/d ratios obtained from the multiple regressions of ΔΔEoi + ΔΔEPauli and of ΔΔH (2.99 ± 0.28 and 2.86 ± 0.33, respectively, Fig. 3b and c).
A systematic analysis of the ortho-, meta- and para-fluorine substituent effect on the EDA terms, using different systems as initial references, reveals trends similar to those obtained from the regression coefficients when all 20 C–H bonds are considered (Fig. 2e, f and S5).26 Additionally, the same conclusions obtained from the analysis at equilibrium and consistent geometries can be drawn from the analysis of the EDA terms as a function of the C–H bond distance (see Fig. S7 and S8).
Our results discussed so far demonstrate that the Pauli repulsion provides a unified explanation for both the overall C–H bond strengthening in (poly)fluorobenzenes and the major part of the selective increase in bond strength for the C–H bond ortho to fluorine, which is fully captured by the combined Pauli and orbital interaction terms. In this way, we address two important open questions of which the underlying causes had remained unknown until now.2b A comprehensive analysis of the origin of the Pauli repulsion trends is provided in the following section.
Origin of the Pauli repulsion
As discussed above, our quantitative MO and EDA analyses reveal that Pauli repulsion ΔEPauli plays a key role in the strengthening of the C–H bonds upon fluorine substitution on the benzene ring. As will become clear in the following, the inductive effect of fluorine polarizes the closed-shell molecular orbitals of the aryl fragment towards the fluorine atom, which reduces their amplitude in the C–H bonding region of the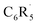 radical fragment. This, in turn, leads to a decrease in Pauli repulsion with the singly-occupied hydrogen 1s orbital, resulting in a stronger C–H bond.
radical fragment. This, in turn, leads to a decrease in Pauli repulsion with the singly-occupied hydrogen 1s orbital, resulting in a stronger C–H bond.
In the EDA framework, the ΔEPauli term arises from repulsive interactions between same-spin occupied orbitals on the respective fragments.7 Therefore, all ΔEPauli originates from occupied–occupied orbital interactions between the singly occupied hydrogen 1s orbital and the same-spin component of the occupied molecular orbitals in the σ-electron system of the aryl fragment (see Fig. 4a). The greater the orbital overlap between the hydrogen 1s and the aryl σ-MOs, the stronger the Pauli repulsion. Table S3 lists key overlaps between the hydrogen 1s AO and the σ-MOs of the aryl fragment. The occupied orbital most affected by the presence of a fluorine substituent is the σHOMO−6 of the aryl radical (Fig. 4b). The orbital overlap between the hydrogen 1s orbital and the aryl σHOMO−6 orbital amounts to 〈H1s|σHOMO−6〉 = 0.18 for benzene (Fig. 4c). This orbital overlap significantly decreases for the ortho- and para-C–H bonds, namely to 〈H1s|σHOMO−6〉 = 0.12 and 0.09, respectively. This leads to the lower Pauli repulsion for the latter two bonds. Note that for the meta-C–H bond, the overlap remains nearly unchanged: 〈H1s|σHOMO−6〉 = 0.19; hence the ΔEPauli value remains similar to that in benzene.
A similar, though less pronounced, effect is observed in the 〈H1s|σHOMO−2〉 orbital overlap. Fig. S9 presents the 〈H1s|σHOMO−6〉 and 〈H1s|σHOMO−2〉 overlaps as a function of the C–H bond distance. Interestingly, although the largest reduction in Pauli repulsion occurs for the ortho-C–H bond, the largest decrease in 〈H1s|σHOMO−6〉 overlap occurs at the para position. However, when considering the combined effect of both the 〈H1s|σHOMO−6〉 and 〈H1s|σHOMO−2〉, a more significant reduction is observed for the ortho-C–H bond, whereas the para position shows only a modest reduction in the second overlap. An approximate way to assess the overall contribution of all orbitals to the Pauli repulsion is by considering the sum of the squared overlaps (S2) between the H1s orbital and all same-spin occupied σ-orbitals of the aryl fragment.27 Accordingly, the ∑S2 values for the C–H bond in benzene and for the ortho-, meta-, and para-C–H bonds in fluorobenzene are 0.244, 0.227, 0.242, and 0.234, respectively. These values follow the same trend observed for both the Pauli repulsion and the C–H bond strength.
The essential question remaining is: why does fluorination reduce the Pauli-repulsive overlap between the occupied orbitals on the 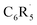 fragment and the radical electron of the hydrogen atom and thus strengthen the aryl C–H bond? To address this question, we analyze the formation of the aryl fragment
fragment and the radical electron of the hydrogen atom and thus strengthen the aryl C–H bond? To address this question, we analyze the formation of the aryl fragment 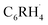 in benzene (R = H) and fluorobenzene (R = F) from the
in benzene (R = H) and fluorobenzene (R = F) from the 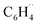 biradical and R˙ (see Fig. 5a). This approach enables us to understand how the relevant
biradical and R˙ (see Fig. 5a). This approach enables us to understand how the relevant 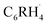 orbitals arise from the same
orbitals arise from the same 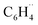 biradical, and how their nature becomes different in the case of a fluorine substituent R = F in fluorobenzene versus a hydrogen substituent R = H in benzene. The resulting MO diagram is shown qualitatively in Fig. 5b (for details, see Fig. S10–S12 and Tables S4–S6). From this point onward, we focus on the ortho position, where the reduction of Pauli repulsion is most pronounced and where the proximity of the substituent would intuitively suggest a repulsive effect.
biradical, and how their nature becomes different in the case of a fluorine substituent R = F in fluorobenzene versus a hydrogen substituent R = H in benzene. The resulting MO diagram is shown qualitatively in Fig. 5b (for details, see Fig. S10–S12 and Tables S4–S6). From this point onward, we focus on the ortho position, where the reduction of Pauli repulsion is most pronounced and where the proximity of the substituent would intuitively suggest a repulsive effect.
The σHOMO−6 orbitals of the  and
and  radicals arise from a σ-bonding interaction between a doubly occupied σ-orbital of the
radicals arise from a σ-bonding interaction between a doubly occupied σ-orbital of the  biradical and the singly occupied hydrogen 1s (H1s) or fluorine 2p (F2p) orbitals, respectively (see Fig. 5b). The F2p orbital lies lower in energy than the H1s orbital because fluorine is more electronegative than hydrogen. As a result, the F2p orbital is closer in energy to the doubly occupied σ-orbital of the
biradical and the singly occupied hydrogen 1s (H1s) or fluorine 2p (F2p) orbitals, respectively (see Fig. 5b). The F2p orbital lies lower in energy than the H1s orbital because fluorine is more electronegative than hydrogen. As a result, the F2p orbital is closer in energy to the doubly occupied σ-orbital of the  fragment than the H1s orbital, which leads to a larger fluorine contribution to the formation of the bonding combination, which is the σHOMO−6 orbital (see gross Mulliken contributions in Fig. 5b). This greater contribution of fluorine polarizes the σHOMO−6 orbital towards the fluorine atom and away from the carbon radical. Consequently, the overlap between σHOMO−6 and the hydrogen 1s orbital across the aromatic C–H bond between the carbon-radical center of
fragment than the H1s orbital, which leads to a larger fluorine contribution to the formation of the bonding combination, which is the σHOMO−6 orbital (see gross Mulliken contributions in Fig. 5b). This greater contribution of fluorine polarizes the σHOMO−6 orbital towards the fluorine atom and away from the carbon radical. Consequently, the overlap between σHOMO−6 and the hydrogen 1s orbital across the aromatic C–H bond between the carbon-radical center of 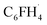 and H˙ is reduced. This is a clear manifestation of fluorine's inductive effect, which predominantly affects the σ-system in fluorobenzene, as previously discussed elsewhere.28 Furthermore, note that neither the F2p nor the H1s contributes significantly to the formation of the σSOMO in the aryl radical. As a result, the σSOMO is not polarized by the substituent, leading to similar 〈H1s|σSOMO〉 orbital overlaps and, consequently, similar electron-pair bond ΔEpb and orbital interaction ΔEoi energies for benzene and fluorobenzene (see Fig. S9 for overlaps and Table S7 for ΔEpb values).29
and H˙ is reduced. This is a clear manifestation of fluorine's inductive effect, which predominantly affects the σ-system in fluorobenzene, as previously discussed elsewhere.28 Furthermore, note that neither the F2p nor the H1s contributes significantly to the formation of the σSOMO in the aryl radical. As a result, the σSOMO is not polarized by the substituent, leading to similar 〈H1s|σSOMO〉 orbital overlaps and, consequently, similar electron-pair bond ΔEpb and orbital interaction ΔEoi energies for benzene and fluorobenzene (see Fig. S9 for overlaps and Table S7 for ΔEpb values).29
Predictive power of the model
Finally, we wish to extend our model to related systems. To this end, we have extended our MO and EDA analyses of the C–H bonds to a broader set of monosubstituted benzenes C6H5R with R still including H and F, but now also Cl, Br, I, and Li. Proceeding from the model we have established, one would expect that electronegative substituents R strengthen C–H bonds, unless, in cases of adjacent C–H bonds, R becomes so big that it destabilizes this C–H bond through direct R⋯H Pauli repulsion.5 On the other hand, electropositive substituents R are then expected to weaken the C–H bonds because they polarize orbital amplitude toward the pertinent carbon atoms. The results confirm this expectation.The bond dissociation enthalpies ΔH, bond dissociation energies ΔE, together with the ASM and EDA terms for this series of monosubstituted benzenes, are given in Table S7. The C–H bond dissociation enthalpy is larger than that in benzene for C6FH5 (−111.8 kcal mol−1) and C6ClH5 (−110.9 kcal mol−1) but very close to that in benzene (−109.3 kcal mol−1) for the other halogens (Br, −110.3; I, −109.4 kcal mol−1). However, the C–H bond dissociation enthalpy is significantly smaller in C6LiH5 (−91.9 kcal mol−1). A comparison with experimental BDE data would be valuable; however, such data are not listed in ref. 19 for the systems with R = Cl, Br, I, and Li. As expected, the ΔH values follow the same trends as the bond energies ΔE, which are in turn dominated by the interaction energy ΔEint. The relative EDA terms for the C–H bonds in the C6RH5 (R = F, Cl, Br, I, Li) systems are depicted in Fig. 6a. As shown in Fig. 6b, the inductive effect responsible for the reduction in Pauli repulsion in fluorobenzene is also observed in the other halobenzenes. In contrast to the case of fluorine, the orbital term is of the same order of magnitude as the Pauli repulsion term for Cl, Br, and I. Thus, the increase in ΔEoi compensates for the variation in the ΔEPauli and results in values of ΔEint for Cl, Br, and I within 1.4 kcal mol−1 of one another (Fig. 6a and Table S7).
As in the case of fluorine, strong contributions to the decrease of the Pauli repulsion are found in σHOMO−6. The halogen substituent polarizes the σHOMO−6 orbital toward itself and away from the C–H bonding region, leading to a progressive decrease in 〈H1s|σHOMO−6〉 overlap as the electronegativity of the substituent increases from I to F. However, the EDA results in Fig. 6a (the complete dataset is provided in Table S7) reveal that inductive effects alone cannot fully account for the observed trends in Pauli repulsion across the halobenzene series. Although bromine and iodine are more electronegative than hydrogen and thus exert a higher inductive effect, the Pauli repulsion is actually higher in bromobenzene and iodobenzene compared to benzene. This apparent discrepancy arises from the increase in size of the substituent atom. As the size of R increases, the spatial extension of the valence AOs and the number of subvalence shells increase, causing a stronger Pauli repulsion. Thus, the observed trends in Pauli repulsion reflect a balance between two opposing effects, namely, the electron-withdrawing (inductive) effect, which reduces Pauli repulsion, and the steric (size-related) effect, which enhances Pauli repulsion.
For fluorine and chlorine, the most electronegative atoms in the series, the atom size effect is offset by the electron-withdrawing (inductive) effect, resulting in reduced 〈H1s|σHOMO−6〉 overlap (see Fig. 6b), negative ΔΔEPauli, and stronger C–H bonds compared to benzene. In the case of bromine, the atom size effect begins to outweigh the inductive effect, leading to a modest increase in Pauli repulsion (ΔΔEPauli = +0.9 kcal mol−1). Nevertheless, this destabilization is compensated by an enhanced orbital interaction,30 still resulting in a stronger ortho-C–H bond compared to benzene. For iodobenzene, the atom size effect becomes dominant, resulting in greater Pauli repulsion. However, even in this case, the gain in attractive orbital interactions counterbalances the increase in ΔEPauli, yielding a C–H bond with nearly the same strength as in benzene.
Finally, we have also investigated phenyllithium (C6H5Li), a system in which the substituent R is less electronegative than hydrogen. The 2s orbital of lithium lies significantly higher in energy than the np orbitals of the halogens and does not contribute meaningfully to the formation of the σHOMO−6 orbital in the aryl radical. As a result, the 〈H1s|σHOMO−6〉 orbital overlap in phenyllithium is nearly identical to that in benzene (see Fig. 7a). However, lithium does affect higher-energy orbitals of the aryl fragment. Notably, Fig. 7b shows a substantial increase in the 〈H1s|σHOMO−1〉 orbital overlap in C6H4Li–H relative to both benzene and fluorobenzene. Unlike the halogens, lithium raises the orbital amplitude in the C–H bonding region rather than depleting it, effectively donating electron density into the carbon scaffold of C–C σ bonds through an effect opposite to that of fluorine. This electron-donating character leads to increased occupied–occupied overlap and thus increased Pauli repulsion across the aromatic C–H bond. This is reminiscent of the lone-pair shielded radical effect induced by the substituent on the C–C bond strength.31 This effect outweighs all stabilizing contributions, resulting in a substantially weaker C–H bond.
To disentangle size and inductive effects, we also performed EDA analyses for the same substituents at the para position. In this configuration, direct R⋯H repulsion is eliminated, allowing the intrinsic electronic nature of the substituent (electron-withdrawing or electron-donating) to dominate the Pauli repulsion trends. As shown in Fig. S13, all substituents more electron-withdrawing than hydrogen lead to a decrease in Pauli repulsion, following the electronegativity trend. In contrast, for phenyllithium, the only electron-donating substituent, Pauli repulsion remains significantly higher than in benzene, even in the absence of direct steric interactions.
Conclusion
Fluorine substitution strengthens the C–H bond in fluorinated benzenes C6R5H (R = H, F), as indicated by the increase in the homolytic bond dissociation energies (BDE). Our quantum chemical bonding analyses, based on dispersion-corrected, (scalar) relativistic density functional theory, uncover that fluorination strengthens the C–H bond in large part by reducing steric (Pauli) repulsion between filled orbitals. As demonstrated previously, the effects of multiple fluorine substitution are additive and markedly greater for ortho than for para substitution while the effect of meta substitution is barely significant. The ortho effect on BDE is calculated to be ca. 2.9 times the effect of para substitution. The EDA analysis shows that the Pauli repulsion is an important contributor to the bond strengthening but that this ortho/para ratio can be reproduced by the sum of the Pauli repulsion and orbital terms.Our quantitative MO and energy decomposition analyses reveal how the strong inductive effect of the fluorine atom withdraws electron density, especially from the ipso carbon of the aryl fragment and polarizes its closed-shell molecular orbitals towards the fluorine atom. This polarization reduces the spatial extension of the occupied orbitals on the aryl fragment towards the hydrogen 1s orbital. Therefore, the same-spin orbital overlap across the C–H bond shrinks, and Pauli repulsion becomes weaker, resulting in stronger C–H bonds.
Extension of the analysis to other monosubstituted benzenes C6RH5 (R = Cl, Br, I, Li) shows that substituents R modulate the strength of the adjacent C–H bond by reducing or enhancing Pauli repulsion, either via an indirect, inductive effect or via direct steric repulsion between C–R and C–H bonds. For R = F, the C–H BDE is notably bigger than for other halogens which, in turn, are slightly bigger than that of benzene. For R = F, the strong inductive effects decrease the overall Pauli repulsion, resulting in a stronger C–H bond. For R = Cl, the overall Pauli repulsion remains smaller than for benzene. In contrast, for R = Br and I, weaker inductive effects are offset by larger atomic size, which increases direct R⋯H steric repulsion. For all halogens except fluorine, this repulsion is balanced by enhanced orbital attraction, yielding C–H bonds of similar strength for R = Cl, Br, I, and H. Thus, fluorine remains as a special case among electron-withdrawing substituents that we have studied so far. Finally, for R = Li, the inductive effect is inverted: lithium pushes electron density to the aryl fragment, increasing Pauli repulsion and leading to the weakest C–H bond in the series.
Author contributions
RNP, OE, and FMB conceived the project, which was supervised by DRS, AACB, and CFG. DASO carried out the quantum-chemical computations and bonding analyses and drafted the manuscript. All authors discussed the results and reviewed the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
All data of this study are available in the main text and supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc01846g.Acknowledgements
The authors thank the Dutch Research Council (NWO) for financial support. This work was carried out on the ADA (VU) Cluster and the Dutch national e-infrastructure with the support of SURF Cooperative. Additionally, D. A. S. O. acknowledges the São Paulo Research Foundation (FAPESP) (Grants #2022/01685-6 and #2024/03646-3) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Grant #162366/2021-3) for funding their doctoral scholarship. A. A. C. B. thanks the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil) for financial support (Grant #313720/2023-1). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES), Finance Code 001, Program 33002010191P0. OE acknowledges the support of the Research Council of Norway through the Centre of Excellence Hylleraas Centre for Quantum Molecular Sciences (No. 262695).References
- (a) D. O'Hagan, Understanding organofluorine chemistry. An Introduction to the C−F Bond, Chem. Soc. Rev., 2008, 37, 308–319 RSC; (b) M. Inoue, Y. Sumii and N. Shibata, Contribution of Organofluorine Compounds to Pharmaceuticals”, ACS Omega, 2020, 5, 10633–10640 Search PubMed; (c) Y. Zhou, J. Wang, Z. Gu, S. Wang, W. Zhu, J. L. Aceña, V. A. Soloshonok, K. Izawa and H. Liu, Next Generation of Fluorine-Containing Pharmaceuticals: Compounds Currently in Phase II–III Clinical Trials of Major Pharmaceutical Companies: New Structural Trends and Therapeutic Areas, Chem. Rev., 2016, 116, 422–518 Search PubMed; (d) M. Bernús, G. D. Núñez, W. C. Hartley, M. Guasch, J. Mestre, M. Besora, J. J. Carbó and O. Boutureira, Impact of Fluorine Pattern on Lipophilicity and Acid–Base Properties of 2-(Thiofluoroalkyl)pyridines: Insights from Experiments and Statistical Modeling, J. Med. Chem., 2025, 68, 4787–4800 CrossRef PubMed; (e) Y. Du, Y. Bian, D. Baecker, G. Dhawan, A. Semghouli, L. Kiss, W. Zhang, A. E. Sorochinsky, V. A. Soloshonok and J. Han, Fluorine in the Pharmaceutical Industry: FDA-Approved Fluorine-Containing Drugs in 2024, Chem.–Eur. J., 2025, 31, e202500662 Search PubMed; (f) E. Henary, S. Casa, T. L. Dost, J. C. Sloop and M. Henary, The Role of Small Molecules Containing Fluorine Atoms in Medicine and Imaging Applications, Pharmaceuticals, 2024, 17, 281 Search PubMed; (g) S. Preshlock, M. Tredwell and V. Gouverneur, 18F-Labeling of Arenes and Heteroarenes for Applications in Positron Emission Tomography, Chem. Rev., 2016, 116, 719–766 CrossRef CAS PubMed; (h) P. Jeschke, Recent Developments in Fluorine-Containing Pesticides, Pest Manage. Sci., 2024, 80, 3065–3087 CrossRef CAS PubMed; (i) T. Zhang, Z. Chen, W. Zhang, L. Wang and G. Yu, Recent Progress of Fluorinated Conjugated Polymers, Adv. Mater., 2024, 36, 2403961 CrossRef CAS PubMed; (j) H. Hyrondelle, A. Terry, J. Lhoste, S. Tencé, K. Lemoine, J. Olchowka, D. Dambournet, C. Tassel, J. Gamon and A. Demourgues, Fluorine as a Key Element in Solid-State Chemistry of Mixed Anions 3d Transition Metal-Based Materials for Electronic Properties and Energy, Chem. Rev., 2025, 125, 4287–4358 CrossRef CAS PubMed; (k) Y. Wang, X. Yang, Y. Meng, Z. Wen, R. Han, X. Hu, B. Sun, F. Kang, B. Li, C. Wang and G. Wang, Fluorine Chemistry in Rechargeable Batteries: Challenges, Progress, and Perspectives, Chem. Rev., 2024, 124, 3494–3589 CrossRef CAS PubMed; (l) L. Hunter, The C–F Bond as a Conformational Tool in Organic and Biological Chemistry, Beilstein J. Org. Chem., 2010, 6, 38 Search PubMed; (m) G. Cavallo, P. Metrangolo, R. Milani, T. Pilati, A. Priimagi, G. Resnati and G. Terraneo, The halogen bond, Chem. Rev., 2016, 116, 2478–2601 CrossRef CAS PubMed; (n) B. Maiti, A. Schubert, S. Sarkar, S. Bhandari, K. Wang, Z. Li, E. Geva, R. J. Twieg and B. D. Dunietz, Enhancing Charge Mobilities in Organic Semiconductors by Selective Fluorination: A Design Approach Based on a Quantum Mechanical Perspective, Chem. Sci., 2017, 8, 6947–6953 RSC; (o) N. M. P. Rosa, M. Máximo-Canadas and I. Borges Jr., Assessing the Aromaticity of Fluorinated Benzene Derivatives Using New Descriptors Based on the Distributed Multipole Analysis (DMA) Partition of the Electron Density, J. Phys. Org. Chem., 2025, 38, e70024 CrossRef CAS; (p) J. J. Torres-Vega, A. Vásquez-Espinal, L. Ruiz, M. A. Fernández-Herrera, L. Alvarez-Thon, G. Merino and W. Tiznado, ChemistryOpen, 2015, 4, 302–307 Search PubMed; (q) S. Balser and O. V. Boltalina, Electron Deficient Fluorous Polycyclic Aromatic and Heteroaromatic Hydrocarbons, Coord. Chem. Rev., 2026, 554, 217592 CrossRef CAS; (r) T. J. Fuhrer, M. Houck and S. T. Iacono, Fluoromaticity: The Molecular Orbital Contributions of Fluorine Substituents to the π-Systems of Aromatic Rings, ACS Omega, 2021, 6, 32607–32617 CrossRef CAS PubMed; (s) K. Carter-Fenk and J. M. Herbert, Electrostatics does not dictate the slip-stacked arrangement of aromatic π–π interactions, Chem. Sci., 2020, 11, 6758–6765 RSC.
- (a) O. Eisenstein, J. Milani and R. N. Perutz, Selectivity of C−H activation and competition between C−H and C−F bond activation at fluorocarbons, Chem. Rev., 2017, 117, 8710–8753 CrossRef CAS PubMed; (b) Y. P. Budiman, R. N. Perutz, P. G. Steel, U. Radius and T. B. Marder, Applications of Transition Metal-Catalyzed ortho-Fluorine-Directed C−H Functionalization of (Poly)fluoroarenes in Organic Synthesis, Chem. Rev., 2024, 124, 4822–4862 CrossRef CAS PubMed; (c) T. Rogge, N. Kaplaneris, N. Chatani, J. Kim, S. Chang, B. Punji, L. L. Schafer, D. G. Musaev, J. Wencel-Delord, C. A. Roberts, R. Sarpong, Z. E. Wilson, M. A. Brimble, M. J. Johansson and L. Ackermann, C–H activation, Nat. Rev. Methods Primers, 2021, 1, 43 CrossRef CAS; (d) T. Dalton, T. Faber and F. Glorius, C−H Activation: Toward Sustainability and Applications, ACS Cent. Sci., 2021, 7, 245–261 CrossRef CAS PubMed; (e) T. P. Bast, J. V. Obligacion, E. Rochette, I. Pappas and P. J. Chirik, Cobalt-Catalyzed Borylation of Fluorinated Arenes: Thermodynamic Control of C(sp2)−H Oxidative Addition Results in ortho-to-Fluorine Selectivity, J. Am. Chem. Soc., 2019, 141, 15378–15389 CrossRef PubMed.
- (a) W. D. Jones and E. T. Hessel, Photolysis of Tp’Rh(CN-neopentyl)(η2-PhN=C=N-neopentyl) in Alkanes and Arenes: Kinetic and Thermodynamic Selectivity of [Tp’Rh(CN-neopentyl)] for Various Types of C−H Bonds, J. Am. Chem. Soc., 1993, 115, 554–562 CrossRef CAS; (b) B. A. Vanchura, S. M. Preshlock, P. C. Roosen, V. A. Kallepalli, R. J. Staples, R. E. Maleczka Jr., D. A. Singleton and M. R. Smith, Electronic Effects in Iridium C−H Borylations: Insights from Unencumbered Substrates and Variation of Boryl Ligand Substituents, Chem. Commun., 2010, 46, 7724–7726 RSC; (c) E. Clot, C. Mégret, O. Eisenstein and R. N. Perutz, Validation of the M−C/H−C Bond Enthalpy Relationship through Application of Density Functional Theory, J. Am. Chem. Soc., 2006, 128, 8350–8357 CrossRef CAS PubMed; (d) P. J. Chirik and T. P. Pabst, A Tutorial on Selectivity Determination in C(sp2)−H Oxidative Addition of Arenes by Transition Metal Complexes, Organometallics, 2021, 40, 813–831 CrossRef PubMed.
- (a) E. Clot, M. Besora, F. Maseras, C. Mégret, O. Eisenstein, B. Oelckers and R. N. Perutz, Bond energy M–C/H–C correlations: dual theoretical and experimental approach to the sensitivity of M–C bond strength to substituents, Chem. Commun., 2003, 490–491 RSC; (b) E. Clot, C. Mégret, O. Eisenstein and R. N. Perutz, Exceptional Sensitivity of Metal-Aryl Bond Energies to ortho-Fluorine Substituents: Influence of the Metal, the Coordination Sphere, and the Spectator Ligands on M−C/H−C Bond Energy Correlations, J. Am. Chem. Soc., 2009, 131, 7817–7827 CrossRef CAS PubMed; (c) M. E. Evans, C. L. Burke, S. Yaibuathes, E. Clot, O. Eisenstein and W. D. Jones, Energetics of C−H Bond Activation of Fluorinated Aromatic Hydrocarbons Using a [Tp’Rh(CNneopentyl)] Complex, J. Am. Chem. Soc., 2009, 131, 13464–13473 CrossRef CAS PubMed; (d) T. Tanabe, W. W. Brennessel, E. Clot, O. Eisenstein and W. D. Jones, Synthesis, structure, and reductive elimination in the series Tp’Rh(PR3)(ArF)H; Determination of rhodium–carbon bond energies of fluoroaryl substituents, Dalton Trans., 2010, 39, 10495–10509 RSC; (e) E. Clot, B. Oelckers, A. H. Klahn, O. Eisenstein and R. N. Perutz, cis–trans Isomerisation of CpRe(CO)2(H)(ArF) (ArF = C6FnH5-n; n = 0–5) is the rate determining step in C–H activation of fluoroarenes: a DFT study, Dalton Trans., 2003, 4065–4072 RSC.
- (a) E. Blokker, W.-J. van Zeist, X. Sun, J. Poater, J. M. van der Schuur, T. A. Hamlin and F. M. Bickelhaupt, Methyl Substitution Destabilizes Alkyl Radicals, Angew. Chem. Int. Ed., 2022, 61, e202207477 (Angew. Chem., 2022, 134, e202207477) CrossRef CAS PubMed; (b) P. Vermeeren, W.-J. van Zeist, T. A. Hamlin, C. Fonseca Guerra and F. M. Bickelhaupt, Not Carbon s–p Hybridization, but Coordination Number Determines C–H and C–C Bond Length, Chem.–Eur. J., 2021, 27, 7074–7079 CrossRef CAS PubMed; (c) J. Poater, R. Visser, M. Solà and F. M. Bickelhaupt, Polycyclic Benzenoids: Why Kinked is More Stable than Straight, J. Org. Chem., 2007, 72, 1134–1142 CrossRef CAS PubMed; (d) J. Poater, J. Paauwe, S. Pand, G. Merino, C. Fonseca Guerra and F. M. Bickelhaupt, Kekulene: Structure, stability and nature of H•••H interactions in large PAHs, Mol. Astrophys., 2017, 8, 19–26 CrossRef; (e) D. Rodrigues Silva, P. Vermeeren, J. M. van der Schuur and F. M. Bickelhaupt, Radical Stability Paradox: Substituent Effects versus Heats of Formation, Chem.–Eur. J., 2025, e03600 Search PubMed.
- (a) R. van Meer, O. V. Gritsenko and E. J. Baerends, Physical Meaning of Virtual Kohn−Sham Orbitals and Orbital Energies: An Ideal Basis for the Description of Molecular Excitations, J. Chem. Theory Comput., 2014, 10, 4432–4441 CrossRef CAS PubMed; (b) T. A. Albright, J. K. Burdett and W. H. Whangbo, Orbital Interactions in Chemistry, Wiley, New York, 2nd edn, 2013, ISBN 978-0-471-08039-8 CrossRef.
- F. M. Bickelhaupt and E. J. Baerends, in Reviews in Computational Chemistry, ed. K. B. Lipkowitz and D. B. Boyd, Wiley–VCH, Hoboken, 2000, pp. 1–86 Search PubMed.
- (a) ADF2019.3/2024.1, SCM: Theoretical Chemistry, Vrije Universiteit Amsterdam, The Netherlands, 2019, http://www.scm.com Search PubMed; (b) ADF2024.1, SCM, Theoretical Chemistry, Vrije Universiteit Amsterdam, The Netherlands, 2024, http://www.scm.com Search PubMed; (c) G. te Velde, F. M. Bickelhaupt, E. J. Baerends, C. Fonseca Guerra, S. J. A. van Gisbergen, J. G. Snijders and T. Ziegler, Chemistry with ADF, J. Comput. Chem., 2001, 22, 931–967 CrossRef CAS; (d) C. Fonseca Guerra, J. G. Snijders, G. te Velde and E. J. Baerends, Towards an order-N DFT method, Theor. Chem. Acc., 1998, 99, 391–403 Search PubMed.
- (a) E. J. Baerends, N. F. Aguirre, N. D. Austin, J. Autschbach, F. M. Bickelhaupt, R. Bulo, C. Cappelli, A. C. T. van Duin, F. Egidi, C. Fonseca Guerra, A. Förster, M. Franchini, T. P. M. Goumans, T. Heine, M. Hellström, C. R. Jacob, L. Jensen, M. Krykunov, E. van Lenthe, A. Michalak, M. M. Mitoraj, J. Neugebauer, V. P. Nicu, P. Philipsen, H. Ramanantoanina, R. Rüger, G. Schreckenbach, M. Stener, M. Swart, J. M. Thijssen, T. Trnka, L. Visscher, A. Yakovlev and S. van Gisbergen, The Amsterdam Modeling Suite, J. Chem. Phys., 2025, 162, 162501 CrossRef CAS PubMed; (b) AMS 2024.1, SCM: Theoretical Chemistry, Vrije Universiteit Amsterdam, The Netherlands, 2024, http://www.scm.com Search PubMed.
- (a) A. D. Becke, Density-functional exchange-energy approximation with correct asymptotic behavior, Phys. Rev. A, 1988, 38, 3098–3100 CrossRef CAS PubMed; (b) C. T. Lee, W. T. Yang and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B, 1988, 37, 785–789 CrossRef CAS PubMed.
- (a) S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed; (b) S. Grimme, S. Ehrlich and L. Goerigk, Effect of the damping function in dispersion corrected density functional theory, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- E. R. Johnson and A. D. Becke, A post-Hartree–Fock model of intermolecular interactions, J. Chem. Phys., 2005, 123, 024101 CrossRef PubMed.
- E. van Lenthe, E. J. Baerends and J. G. Snijders, Relativistic total energy using regular approximations, J. Chem. Phys., 1994, 101, 9783–9792 CrossRef CAS.
- E. van Lenthe and E. J. Baerends, Optimized Slater-type basis sets for the elements 1–118, J. Comput. Chem., 2003, 24, 1142–1156 CrossRef CAS PubMed.
- (a) M. Franchini, P. H. T. Philipsen, E. van Lenthe and L. Visscher, Accurate Coulomb Potentials for Periodic and Molecular Systems through Density Fitting, J. Chem. Theory Comput., 2014, 10, 1994–2004 CrossRef CAS PubMed; (b) M. Franchini, P. H. T. Philipsen and L. Visscher, The Becke Fuzzy Cells Integration Scheme in the Amsterdam Density Functional Program Suite, J. Comput. Chem., 2013, 34, 1819–1827 CrossRef CAS PubMed.
- (a) A. Bérces, R. M. Dickson, L. Fan, H. Jacobsen, D. Swerhone and T. Ziegler, An implementation of the coupled perturbed Kohn-Sham equations: perturbation due to nuclear displacements, Comput. Phys. Commun., 1997, 100, 247–262 CrossRef; (b) H. Jacobsen, A. Bérces, D. P. Swerhone and T. Ziegler, Analytic second derivatives of molecular energies: a density functional implementation, Comput. Phys. Commun., 1997, 100, 263–276 CrossRef CAS; (c) S. K. Wolff, Analytical second derivatives in the Amsterdam density functional package, Int. J. Quantum Chem., 2005, 104, 645–659 CrossRef CAS.
- S. Seabold and J. Perktold, Statsmodels: Econometric and statistical modeling with Python, Proceedings of the 9th Python in Science Conference, 2010 Search PubMed.
- J. D. Hunter, Matplotlib: A 2D Graphics Environment, Computing in Science & Engineering, 2007, 3, 90–95 Search PubMed.
- Y. R. Luo, Comprehensive handbook of chemical bond energies. CRC press, Boca Raton, 2007 Search PubMed.
- (a) P. Vermeeren, T. A. Hamlin and F. M. Bickelhaupt, Chemical reactivity from an activation strain perspective, Chem. Commun., 2021, 57, 5880–5896 RSC; (b) P. Vermeeren, S. C. C. van der Lubbe, C. Fonseca Guerra, F. M. Bickelhaupt and T. A. Hamlin, Understanding chemical reactivity using the activation strain model, Nat. Protoc., 2020, 15, 649–667 CrossRef CAS PubMed; (c) F. M. Bickelhaupt and K. N. Houk, Analyzing Reaction Rates with the Distortion/Interaction-Activation Strain Model, Angew. Chem. Int. Ed., 2017, 56, 10070–10086 (Angew. Chem., 2017, 129, 10204–10221) CrossRef CAS PubMed; (d) L. P. Wolters and F. M. Bickelhaupt, The activation strain model and molecular orbital theory, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2015, 5, 324–343 CAS; (e) I. Fernández and F. M. Bickelhaupt, The activation strain model and molecular orbital theory: understanding and designing chemical reactions, Chem. Soc. Rev., 2014, 43, 4953–4967 RSC; (f) W.-J. van Zeist and F. M. Bickelhaupt, The activation strain model of chemical reactivity, Org. Biomol. Chem., 2010, 8, 3118–3127 RSC; (g) F. M. Bickelhaupt, Understanding reactivity with Kohn–Sham molecular orbital theory: E2–SN2 mechanistic spectrum and other concepts, J. Comput. Chem., 1999, 20, 114–128 CrossRef CAS.
- (a) F. M. Bickelhaupt, M. Solà and C. Fonseca Guerra, Highly polar bonds and the meaning of covalency and ionicity—structure and bonding of alkali metal hydride oligomers, Faraday Discuss., 2007, 135, 451–468 RSC; (b) D. Rodrigues Silva, E. Blokker, J. M. van der Schuur, T. A. Hamlin and F. M. Bickelhaupt, Nature and strength of group-14 A–A’ bonds, Chem. Sci., 2024, 15, 1648–1656 RSC; (c) E. Blokker, X. Sun, J. Poater, J. M. van der Schuur, T. A. Hamlin and F. M. Bickelhaupt, The Chemical Bond: When Atom Size Instead of Electronegativity Difference Determines Trend in Bond Strength, Chem.–Eur. J., 2021, 27, 15616–15622 CrossRef CAS PubMed.
- (a) W. J. van Zeist, C. Fonseca Guerra and F. M. Bickelhaupt, PyFrag—Streamlining Your Reaction Path Analysis, J. Comput. Chem., 2008, 29, 312–315 CrossRef CAS PubMed; (b) X. Sun, T. M. Soini, J. Poater, T. A. Hamlin and F. M. Bickelhaupt, PyFrag 2019—Automating the Exploration and Analysis of Reaction Mechanisms, J. Comput. Chem., 2019, 40, 2227–2233 CrossRef CAS PubMed; (c) X. Sun, T. Soini, L. P. Wolters, W.-J. van Zeist, C. Fonseca Guerra, T. A. Hamlin and F. M. Bickelhaupt, PyFrag 2019, Vrije Universiteit Amsterdam, The Netherlands, 2019 Search PubMed.
- Concomitant changes are also observed in the C–F bond dissociation energies. S. A. Macgregor, D. McKay, J. A. Panetier and M. K. Whittlesey, Computational study of the hydrodefluorination of fluoroarenes at [Ru(NHC)(PR3)2(CO)(H)2]: predicted scope and regioselectivities, Dalton Trans., 2013, 42, 7386–7396 RSC.
- To ensure a consistent comparison across systems, all analyses for the monosubstituted system were carried out at a fixed C–H bond length of 1.088 Å, which corresponds to the equilibrium bond length in benzene. In this way, the original variations in bonding capabilities along the various model systems are separated from the resulting geometrical relaxations. Table S2 and Fig. S4 report the EDA terms at this consistent geometry for a representative subset of systems (Fig. S3). Comparison of the data in Tables S1 and S4 shows that the same conclusions are obtained for both equilibrium and fixed geometries.
- The b/d ratio can be interpreted as a measure of how much stronger the ortho-C–H bond becomes for each 1 kcal mol−1 increase in the para-C–H bond strength. As such, it provides a meaningful quantitative descriptor for analyzing the regioselectivity of C–H bond strength between the ortho and para positions.
- An approximately additive effect for the polyfluorinated benzene is also observed for both the bond enthalpies and Pauli repulsion when the changes in their values are compared with those of the monosubstituted system (Fig. S6).
- The use of the sum of squared overlaps (∑S2) provides an approximate, although representative, measure of the overall Pauli repulsive interactions. A more rigorous descriptor would require weighting each S2 contribution by the corresponding kinetic energy of the interacting orbitals, which more accurately reflects their Pauli repulsive character. However, such an approach would substantially increase the complexity of the analysis and obscure the clear trends discussed here. For theoretical details see reference 7.
- C. Nieuwland, P. Vermeeren, F. M. Bickelhaupt and C. Fonseca Guerra, Understanding chemistry with the symmetry-decomposed Voronoi deformation density charge analysis, J. Comput. Chem., 2023, 44, 2108–2119 CrossRef CAS PubMed.
- As the electron pair-bond energy is approximately proportional to Δε + c • S2/Δε (c is a constant) it is not possible to deduce the variation of this term from the variation in Δε (see also Ref. 6b).
- The increase in atomic size contributes not only to greater Pauli repulsion but also to enhanced orbital interaction stabilization of the C–H bond. In the case of halogens, this stabilization arises primarily from an increase in the C–H electron-pair bond (ΔEpb), which becomes progressively more significant from fluorine to iodine (see values in Table S7). For R = Li, however, the observed increase in orbital attraction cannot be attributed solely to ΔEpb. Instead, other orbital interaction components, such as, donor–acceptor interactions and polarization, also play a substantial role in the overall stabilization.
- E. Blokker, M. ten Brink, J. M. van der Schuur, T. A. Hamlin and F. M. Bickelhaupt, Origin of the Captodative Effect: The Lone-Pair Shielded Radical, ChemistryEurope, 2023, 1, e202300006 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |