Determination of sample stability for whole blood parameters using formal experimental design†
Abstract
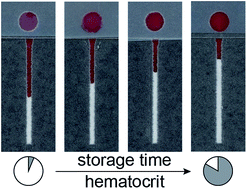
Biological samples have dynamic properties that can be affected by several preanalytical factors including collection, handling, and storage. When developing diagnostic assays that use biological samples as the input, these factors must be identified and managed to ensure that sample degradation is not jeopardizing the performance of the assay. In this work, we use formal experimental design to identify the factors that influence the stability and integrity of a sample of whole blood with respect to two parameters: the hematocrit and the extent of hemolysis. We found that storage time is the only preanalytical factor with a statistically significant impact on the hematocrit for both the reference centrifugation method and the response of a paper-based hematocrit device, with hematocrit increasing as a function of increased storage time. We attributed this trend to changes in red blood cell morphology that occur with an increased storage time. For this reason, we determined it to be critical to develop the paper-based device and hematocrit assay using whole blood within 48 hours of receipt to ensure that it is not introducing bias to the diagnostic output. Additionally, our results confirmed that our paper-based hematocrit device operates according to our established principle—that the transport distance is determined by the ratio of plasma volume to total volume of whole blood. The approach described in this manuscript can be applied to other parameters or biological samples to develop diagnostic assays with confidence that the selected parameters are consistent with the intended end-user input.
- This article is part of the themed collections: Analytical Methods Recent HOT articles and Analytical Methods Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...