Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Per- and polyfluoroalkyl substances (PFAS) and other contaminants of concern in tribal waters of Montana
Kelly L.
Smalling
 *a,
Paul M.
Bradley
*a,
Paul M.
Bradley
 b,
Kristin M.
Romanok
b,
Kristin M.
Romanok
 a,
John T.
Doyle
ce,
Margaret J.
Eggers†
a,
John T.
Doyle
ce,
Margaret J.
Eggers†
 de,
Christine
Martin‡
e,
Elliott P.
Barnhart
f,
Picabo
Binette
g,
Eric M.
Castro
eh,
Madisan
Chavez
eh,
Stephanie A.
Ewing
h,
Stephanie E.
Gordon
de,
Christine
Martin‡
e,
Elliott P.
Barnhart
f,
Picabo
Binette
g,
Eric M.
Castro
eh,
Madisan
Chavez
eh,
Stephanie A.
Ewing
h,
Stephanie E.
Gordon
 i,
Mathew W.
Fields
dj,
James L.
Gray
i,
Mathew W.
Fields
dj,
James L.
Gray
 k,
Ashley M.
Groshong
g,
Chiachi
Hwang
j,
Leslie K.
Kanagy
k,
JoRee WClay
LaFrance
le,
Keith A.
Loftin
k,
Ashley M.
Groshong
g,
Chiachi
Hwang
j,
Leslie K.
Kanagy
k,
JoRee WClay
LaFrance
le,
Keith A.
Loftin
 m,
Carrie Mae
Long
g,
R. Blaine
McCleskey
n,
Shannon M.
Meppelink
m,
Carrie Mae
Long
g,
R. Blaine
McCleskey
n,
Shannon M.
Meppelink
 o,
Crystal L.
Richards§
g,
Molly L.
Schreiner
o,
Crystal L.
Richards§
g,
Molly L.
Schreiner
 a,
Jonathan I.
Shikany
a,
Jonathan I.
Shikany
 gj and
Mahelat
Tesfamariam
g
gj and
Mahelat
Tesfamariam
g
aU.S. Geological Survey, New Jersey Water Science Center, Lawrenceville, New Jersey 08648, USA. E-mail: ksmall@usgs.gov
bU.S. Geological Survey, Columbia, South Carolina, USA
cLittle Big Horn College, Crow Agency, Montana, USA
dDepartment of Microbiology and Cell Biology, Montana State University, Bozeman, Montana, USA
eCrow Water Quality Project, Crow Agency, Montana, USA
fU.S. Geological Survey, Helena, Montana, USA
gLaboratory of Bacteriology, Rocky Mountain Laboratories, Division of Intramural Research, National Institute of Allergy and Infectious Disease, National Institutes of Health, Hamilton, Montana, USA
hDepartment of Land Resources & Environmental Sciences, Montana Water Center, Montana State University, Bozeman, Montana, USA
iU.S. Geological Survey, Kearneysville, West Virginia, USA
jCenter for Biofilm Engineering, Montana State University, Bozeman, Montana, USA
kU.S. Geological Survey, Lakewood, Colorado, USA
lUniversity of Arizona, Tucson, Arizona, USA
mU.S. Geological Survey, Lawrence, Kansas, USA
nU.S. Geological Survey, Boulder, Colorado, USA
oU.S. Geological Survey, Iowa City, Iowa, USA
First published on 23rd January 2026
Abstract
We assessed potential exposures to a broad suite of contaminants (inorganic, organic and microbial) in culturally important surface waters from three watersheds in a northern plains Native American community (Apsáalooke [Crow Tribe of Montana]) in south-central Montana, United States, with water insecurity concerns. Inorganic (37), organic (435) and microbial (3) constituents were assessed in 12 surface water sites from the Pryor Creek (n = 2), Bighorn River (n = 2) and Little Bighorn River (n = 8) valleys. Twenty-six organics, 33 inorganics and Escherichia coli were detected. Despite relatively low concentrations in surface waters within the Crow Reservation, mixture toxicity indicated prevalent chronic ecological effects and human-health secondary contact (recreation) effects at multiple sites. Further, to address Tribal concerns over the prevalence and corresponding risks of per- and polyfluoroalkyl substances (PFAS), we sampled water, sediment, biofilms and fish at a limited number of locations in the Little Bighorn River. Results indicated that PFAS were prevalent in fish tissues, including whole blood and filets, and to a lesser extent in biofilms, despite few detections in water and sediment samples. This is the first attempt to document environmental PFAS contamination within the reservation and the potential human-health concerns for the general population from consumption of recreational/subsistence fish. Overall, this effort provided preliminary information on the contaminant mixtures present and their potential health implications, which can support the protection of community health and culturally meaningful resources across the Crow Reservation.
Environmental significance“Water is life” (Bii'ishchíis) for the Crow Tribe of Montana. Water security and sustainability are issues affecting all aspects of their lives from drinking water to recreation and spiritual practices. Their connection to the rivers has been passed through generations but due to water quality concerns, use of the rivers for drinking, swimming, fishing, and subsistence hunting and gathering has decreased dramatically over time. Exposures to a range of naturally occurring (e.g., arsenic) and anthropogenic contaminants (e.g., PFAS) in culturally significant surface waters was prevalent and has been reported previously on a limited basis. This type of information can be used to define potential community risks and support Tribal prioritization of remediation/mitigation actions to protect their natural resources. |
Introduction
Water protection or water security on Indigenous lands is driven by the desire for equal access to clean water in support of public health/safety.1 Further, for many Tribal communities, water is considered culturally and spiritually sacred1 as part of deeply held Indigenous knowledge and practices regarding the “reciprocal relations” between Indigenous peoples and the waters that have sustained them for generations.1,2 Contrary to most cultures that consider water solely as a resource/commodity, Indigenous peoples maintain a spiritual connectedness with the land, water and non-human life. In many cases water is considered alive and the degradation of water quality on Tribal lands threatens their ability to maintain these relationships as an important part of their cultural identity.2–4 Degradation of water quality and questions about safety and consumption of traditional foods can have a direct effect on spiritual, mental, and physical health of Tribal communities.5–7In the United States (US), the Department of Interior and Department of Agriculture issued joint secretarial orders in 2021 that moved beyond consultation by mandating agencies to make “agreements with Indian Tribes to collaborate in the co-stewardship of Federal lands and waters under the Departments jurisdiction”, and calling for departments to incorporate Tribal expertise and Indigenous knowledge into land-resource assessments.8,9 Federal protection of Tribal surface-water quality generally falls under the Clean Water Act (CWA), with statutes allowing state or Tribe (through Tribes Approved for Treatment as a State, TAS) primacy if corresponding standards/regulations are equally or more protective than Federal standards. However, despite regulations and US trust responsibility, many Tribes consider federal protection inadequate, leading to impaired water quality and questions surrounding public safety.8,10 Although the CWA provides provisions for Tribes to address water-quality issues, obtaining the appropriate expertise and funding has made this option intractable for most.1,8 For example, of the 330 federally recognized Tribes that meet TAS requirements, only 54 have received TAS status of which only 44 have had their own water-quality standards approved by the U.S. Environmental Protection Agency (EPA).1
Studies have addressed inequities on Tribal lands with an emphasis on drinking-water quality and subsistence fishing/hunting. In the Dakotas and the western US, natural and anthropogenic (e.g., mining) arsenic and uranium have been documented in groundwater drinking-water supplies.11–13Escherichia coli (E. coli) and other potentially pathogenic bacteria have been observed in Tribal drinking water and surface waters used for recreation and ceremonies.14–17 Other emerging/unregulated contaminants including per- and polyfluoroalkyl substances (PFAS), pesticides, and pharmaceuticals have also been observed frequently in drinking water at the point of consumption18,19 including within Tribal communities20 and in surface waters.21,22 These precedents highlight the benefits of conducting studies on Tribal lands in collaboration with Tribal communities and public health agencies to increase the robustness of resource assessments.
PFAS, are a class consisting of thousands of substances that were first produced in the 1940s, are pervasive in the environment, and are major concerns for the health of humans, wildlife and ecosystems due to their environmental prevalence, persistence, and toxicity.23,24 PFAS accumulate in organisms through consumption of contaminated food and water and can be transferred maternally. In humans, PFAS have been documented widely in human plasma25 and have been linked to developmental, metabolic, and immune disorders as well as certain cancers.26–28 Limited information is currently available on PFAS occurrence, distribution, and accumulation in fish and other organisms consumed by Tribal communities.
For the northern plains Crow Tribe of Montana [Apsáalooke people (“Children of the Large Beaked Bird” mistranslated to Crow)],29 “water is life” (Bii'ishchíis). Water is inherent in Apsáalooke culture, sacred practices and beliefs and has always been held in high respect among Tribal members.7,30,31 River and spring waters have long been used in many ceremonies, for subsistence fishing and as a place to recreate and gather during hot summer months, and the local riparian ecosystems are home to medicinal plants and species vital to subsistence hunting and gathering, food security and cultural identity.7 The Crow Reservation encompasses the center of the Tribe's traditional territory and ancestral homeland for many centuries, and a significant amount of ecological knowledge has been retained by the community.7 For example, Tribal elders have noted the river remains ‘murky’ most of the year and no longer ‘clears up’ following spring runoff, potentially due to agriculture and ranching in the watersheds, causing rural families to switch from collecting river water for domestic use to shallow private wells drilled to first water and many within an existing floodplain.15 In 2005, a Crow environmental health assessment conducted by Tribal partners and a local academic partner concluded that water contamination was the most serious environmental health threat to the community15,32 which led to the formation of the Crow Environmental Health Steering Committee (CEHSC). This volunteer group of Tribal members and an academic partner (Little Bighorn College) initiated the Crow Water Quality Project (CWQP) which has resulted in a series of investigations documenting inorganic15 and microbial contamination16 of drinking water and high levels of fecal contamination, including pathogenic microorganisms, in the Little Bighorn River.33,34 A recent cumulative risk assessment indicated that more that 39% of drinking water wells on the Crow Reservation were unsafe due to mixtures of inorganics including uranium, manganese, nitrate, zinc, and arsenic.35 Point-of-use treatment is suitable for removing trace inorganics.36 However, for the Crow community, financial resources are limited and 95% of the participants in the study did not employ treatment even though 80% reported high dissolved solids and poor taste and odor.35 The CEHSC recognized the need to address possible exposures to other inorganics and a wide range of potential anthropogenic contaminants (e.g., PFAS) in drinking water and culturally significant surface waters, to define potential community risks and support Tribal community prioritization of remediation/mitigation actions.
In 2022, the CWQP at Little Big Horn College partnered with the U.S. Geological Survey (USGS) on a synoptic study designed to assess potential exposures to a broad suite of contaminants (inorganic, organic and microbial) in culturally significant surface water in the three major watersheds of the Crow Reservation. PFAS concentrations in fish, sediment and biofilms collected from the Little Bighorn River also were assessed to provide initial insight into the prevalence and corresponding risks of PFAS in Tribal trust resources. These assessments were part of a larger 2022–2023 study designed to assess the cumulative risk of mixed contaminant exposures to human health by collecting samples of tapwater of private wells and small community water supplies throughout the Crow community.37 This partnership follows the principles of Community Based Participatory Research,38,39 and is jointly guided by CEHSC, USGS, Montana State University (MSU) and National Institutes of Health (NIH) scientists.
Methodology
Study area and site selection
The Crow Reservation in south-central Montana, US is home to about 75% of the more than 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 enrolled tribal members (https://tribalnations.mt.gov/Directory/CrowNation) and encompasses about 2.2 million acres of traditional territories including three mountain ranges and three river valleys. Most of the communities including the Tribal capital, Crow Agency, are situated in the Little Bighorn River valley, with smaller communities in the Bighorn River and Pryor Creek valleys15 (Fig. 1). These waters have always been held in high respect by the Tribe and to this day, rivers and springs are used in many sacred ceremonies. Children play in these waters during the summer and local riparian areas are home to medicinal plants and food (e.g., berries, deer, etc.) vital to both food security and cultural identity.31
000 enrolled tribal members (https://tribalnations.mt.gov/Directory/CrowNation) and encompasses about 2.2 million acres of traditional territories including three mountain ranges and three river valleys. Most of the communities including the Tribal capital, Crow Agency, are situated in the Little Bighorn River valley, with smaller communities in the Bighorn River and Pryor Creek valleys15 (Fig. 1). These waters have always been held in high respect by the Tribe and to this day, rivers and springs are used in many sacred ceremonies. Children play in these waters during the summer and local riparian areas are home to medicinal plants and food (e.g., berries, deer, etc.) vital to both food security and cultural identity.31
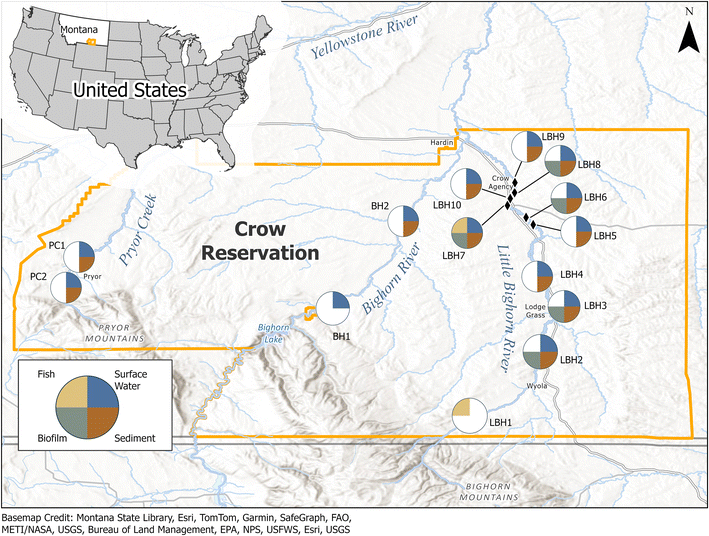 | ||
| Fig. 1 Map of Crow Reservation boundary (orange line) in Montana, United States, and locations of surface water, sediment, biofilm and fish sample locations within the (east to west) Little Bighorn River, Bighorn River, and Pryor Creek drainages. See Table 1 for the complete site list. | ||
The Bighorn River is an approximately 742 km long tributary of the Yellowstone River that runs through Wyoming and Montana in the western portion of the US. In Montana, the Bighorn River flows through the Bighorn Mountains on the Crow Reservation, where it is impounded by the Yellowtail Dam to form the Bighorn Lake Reservoir within the Bighorn Canyon National Recreation Area. In Apsáalooke, the Bighorn River is known as Iisaxpúatahcheeaashisee, which translates to English as Large Bighorn Sheep River. The Little Bighorn River is a 222 km long Bighorn River tributary that flows through the Crow Reservation, past the towns of Wyola and Lodge Grass, the Little Bighorn Battlefield National Monument, and Crow Agency to its confluence with the Bighorn River near the town of Hardin. In the Apsáalooke language, the Little Bighorn River is known as Iisaxpúatahcheeaashe Aliakáate. Finally, Pryor Creek is a Yellowstone River tributary that headwaters in the Pryor Mountains and flows through the town of Pryor, the site of Chief Plenty Coups State Park named for the last traditional chief of the Crow Nation (Aleek-chea-ahoosh, meaning “many achievements”).
In July of 2022 and 2023 samples were collected from sites along Pryor Creek (n = 2 sites, 2023 only), the Bighorn River (n = 2 sites; 2023 only) and the Little Bighorn River (n = 10; 2022 and 2023), to understand human and ecological exposures to contaminants in various media, including culturally significant surface waters, sediment, biofilms and fish tissues on the Crow Indian Reservation (Table 1 and Fig. 1). Sites were selected by Tribal partners based on cultural significance, community use (e.g., swimming holes, fishing) and potential susceptibility to contamination (e.g., downstream of landfills, wastewater effluent or agricultural drains). Following sample collection and analysis, all data were reported back to the Tribe through in-person community engagement events in the summer of 2024 organized by CEHSC.
| Site | Site number | Drainage | Year sampled | Surface water | Sediment | Biofilms | Fish tissue |
|---|---|---|---|---|---|---|---|
| a *, indicates site is not along the main stem of river. | |||||||
| Plenty Coups State Park Spring at Pryor | PC1* | Pryor Creek | 2023 | ✗ | ✗ | ||
| Pryor Creek at Sundance Bridge | PC2 | Pryor Creek | 2023 | ✗ | ✗ | ||
| Ft Smith WW Lagoon at Ft Smith, MT | BH1 | Bighorn River | 2023 | ✗ | |||
| Unnamed pond nr Co Rd 313, St Xavier, MT | BH2* | Bighorn River | 2023 | ✗ | ✗ | ||
| Little Bighorn R ab Sport Ck nr Wyola, MT | LBH1 | Little Bighorn River | 2023 | ✗ | |||
| Little Bighorn R nr Sloan Ditch nr Wyola MT | LBH2 | Little Bighorn River | 2022, 2023 | ✗ | ✗ | ✗ | |
| L Bighorn R bl Lodge Grass Cr nr Lodge Grass MT | LBH3 | Little Bighorn River | 2022, 2023 | ✗ | ✗ | ✗ | |
| Irr OF to L Bighorn R Oxbow nr Lodge Grass, MT | LBH4* | Little Bighorn River | 2023 | ✗ | ✗ | ||
| Irrigation ditch near Garryowen, MT | LBH5* | Little Bighorn River | 2023 | ✗ | ✗ | ||
| L Bighorn R bl Med Tail Coulee nr Crow Agency MT | LBH6 | Little Bighorn River | 2022, 2023 | ✗ | ✗ | ✗ | |
| Little Bighorn R ab Agency Canal nr Crow Agency MT | LBH7 | Little Bighorn River | 2022, 2023 | ✗ | ✗ | ✗ | ✗ |
| L Bighorn R bl Heritage Rd brdg at Crow Agency MT | LBH8 | Little Bighorn River | 2022, 2023 | ✗ | ✗ | ✗ | |
| Little Bighorn R. at Pitch's nr Crow Agency, MT | LBH9 | Little Bighorn River | 2022, 2023 | ✗ | ✗ | ||
| Little Bighorn R bl Custer Cr, at Crow Agency, MT | LBH10 | Little Bighorn River | 2023 | ✗ | ✗ | ||
Surface water sample collection and analysis
A total of 19 surface-water samples at 13 sites (Fig. 1) were collected by USGS in cooperation with the CEHSC and Crow college students in 2022 and 2023 (Tables 1 and S1) using standard methods. Surface water samples were analyzed by the USGS40 based on previously published methods for 435 unique organic compounds using six targeted methods41–47 and 46 inorganic constituents using four targeted methods.48–55 Three microbiological indicators were also analyzed (total coliforms, E. coli), and heterotrophic plate counts (general bacteria).16 Organic analytes included cyanotoxins, disinfection byproducts (DBPs), pesticides, PFAS, pharmaceuticals and volatile organic compounds (VOCs); additional method details are in the SI (Table S2). Water quality and contaminant data for all surface water samples as well as detailed information on analytes and detection limits for each of the methods are available in Romanok et al.37 and Tables S2, S4, S5.Sediment, biofilm and tissue sample collection and analysis
Sediment samples were collected from 12 sites in July 2023 (Table 1) using pre-cleaned, methanol-rinsed, stainless-steel scoops.56 Samples were collected from depositional zones in each stream reach from the top approximately 2–5 centimeters (cm) of sediment. Sediment from each stream reach was collected from approximately five different locations along the same side of the stream and composited in clean wide-mouth, high density polyethylene (HDPE) jars. Samples were not sieved. Samples were placed on ice in the field, shipped cold and stored frozen (−40 °C).Biofilms (an aggregate of microorganisms encased in exopolysaccharides (EPS)) are considered one of the most widely distributed producers in aquatic environments and have been used as a natural integrative sampling tool to assess anthropogenic contaminants in aquatic environments.57 In July 2023, biofilm samples were collected from multiple rock surfaces at five sites (Table 1), using methanol-rinsed, stainless-steel knives, and composited in a small Whirlpack or glass jar for PFAS and microbial-community analyses, respectively. Composited biofilms samples were transported on ice to the laboratory and stored frozen at −40 °C.
Fish were collected by hook and line from two sites on the Little Bighorn River in July 2023 by volunteers from the community (Table 1): a presumptive, low-impact location upstream of the town of Wyola and an impacted location in Crow Agency (Fig. 1). At the upstream site, we collected 10 Brown Trout (Salmo trutta), and at the site in Crow Agency we collected 10 catfish (Siluriformes), four goldeye (Hiodon alosoides) and six rock bass (Ambloplites rupestris). Fish were kept alive in aerated 5-gallon buckets prior to processing and were processed within 1-hour of collection. Fish were euthanized with Finquel (MS222; Argent Laboratories, Redmond, Washington) similar to Blazer et al.58 All procedures were conducted in accordance with the ARRIVE 2.0 (ref. 59) guidelines and institutional guidelines. The sampling and handling procedures outlined above were reviewed and approved by the U.S. Geological Survey Eastern Ecological Science Center's Institutional Animal Care and Use Committee and the Crow Environmental Health Steering Committee. Individual fish were measured to the nearest millimeter (mm) and whole blood samples were collected from the caudal-vein using heparinized 3-cm3 syringes with 23-gauge needles, transferred to 2-mL HDPE centrifuge tubes, and stored immediately on dry ice. All fish were inspected for visible abnormalities, and then, depending on size, field-dissected skin-off filets (two per fish) or whole fish, for laboratory dissection, were placed in 1-gallon plastic bags. All samples (whole fish, filets, and blood) were stored in the field on dry ice and shipped to the laboratory on dry ice, where they were stored at −40 °C prior to analysis.
Biofilm and tissue samples (whole blood and filets) were shipped frozen on dry ice within 90 days to RTI Laboratories (Livonia, Michigan, USA) for the extraction and analysis of 28 individual PFAS (Table S3). Sediment samples were shipped frozen within 90 days of collection on wet ice to SGS AXYS Analytical Services Ltd (British Columbia, Canada) for the extraction and analysis of 40 individual PFAS (Table S3). Prior to analysis, samples were air-dried, and percent moisture was determined. All samples were analyzed using EPA Method 1633 with slight laboratory-specific modifications.60 Briefly, all samples were spiked with isotopically labeled standards, extracted in potassium hydroxide and acetonitrile, followed by basic methanol and clean-up using a Waters Oasis WAX 150 mg, solid phase extraction (SPE) cartridge. Following extraction, clean-up and concentration, samples were analyzed by liquid chromatograph/tandem quadrupole mass spectrometry (LC-MS/MS). Method detection limits ranged from 0.036–0.17 ng g−1 wet weight (ww) for biofilms, 0.071–0.76 ng g−1 ww for filets, 0.048–5.4 ng mL−1 for whole blood and 0.027–1.8 ng g−1 dry weight (dw) for sediment (Table S3). PFAS concentrations measured in sediment, biofilms and fish tissue as well as detailed information on analytes and detection limits for each of the methods are available in Romanok et al.61
DNA from the five biofilm samples was extracted in duplicate with FastDNA™ SPIN Kit for Soil (MP Biomedicals, Santa Ana, CA), combined and concentrations measured with Qubit™ dsDNA HS Assay Kit (Thermo Fisher Scientific, Waltham, MA). Prior to the use in polymerase chain reaction (PCR), the DNA concentration in each sample was adjusted to 5–10 ng µL−1. PCR was carried out with primer pairs 341F-805R62 with universal Illumina overhang adapters. The 25 µL amplicon PCR components involving the use of PRIMATM Hot Start Taq Master Mix (MIDSCI, Fenton, MO) were prepared following manufacturer's instructions and PCR conditions were performed in duplicate for each sample as previously described.63 Subsequent PCR clean-up of 16S rRNA gene amplicons and library preparation were done following Illumina's 16S Metagenomic Sequencing Library Preparation sequencing protocol, followed by library sequencing with 2 × 300 bp Miseq Reagent kit on the Ilumina Miseq platform (Illumina, San Diego CA). The 16S rRNA gene amplicon sequences were imported into QIIME 2 pipeline and processed with demux, followed by DADA2 for sequence denoising, chimera check, and construction of paired-end sequences.64,65 Taxonomic identification was performed using the SILVA 138.1 taxonomy classifier.66 Raw DNA sequences were placed in the National Center for Biotechnology Information (NCBI) database under bioproject number: PRJNA1291266.
Quality assurance
Quantitative (≥limit of quantitation, ≥LOQ) and semi-quantitative (between LOQ and long-term method detection limit, MDL) results were treated as detections.67–69 Quality-assurance/quality-control included analyses of field blanks (surface water only; n = 2), laboratory blanks, laboratory spikes, and stable isotope surrogates. No inorganic or organic constituents were detected in blanks at concentrations in the range observed in surface water samples.37 For PFAS, one laboratory blank per batch for a total of five blanks were analyzed across all media (sediment, tissue and whole blood). PFBA (perfluorobutanoic acid) was detected in fish filet and biofilm, PFPeA (perfluoropentanoic acid) was detected in fish filet, and PFOSA (perfluorooctanesulfonamide) was detected in one sediment sample. PFOSA in sediment resulted in the removal of four values that were all at or below the level detected in the laboratory blank. Values for PFBA and PFPeA in fish tissue (filets and whole blood) and PFPeA in biofilm were removed owing to suspected interference and detections in blank samples. Both compounds have only one major ion transition and have been shown to suffer from interference from lipids.70–72 The median surrogate recovery for organic analytes in surface water was 95.5% (interquartile range (IQR): 82.5–104%). The median surrogate recoveries for PFAS in tissue, biofilms and sediment were 75.3% (IQR: 52.0–95.5%), 42.4% (IQR: 30.8–50.3%), and 96.0% (IQR: 88.1–102%), respectively. Detailed quality assurance information can be found in Romanok et al.61Statistical analysis and effects-based screening
Differences in centroids and dispersion in PFAS concentrations between fish species and between tissue type (whole blood vs. filets) were assessed by nonparametric one-way PERMANOVA (n = 9999 permutations) on Euclidean distance (Paleontological Statistics, PAST, vers. 4.03).73A screening-level assessment74,75 of potential cumulative effects of chemical mixtures in each surface-water sample was conducted using a Hazard Index Assessment as described previously.74,76,77 We considered four general classes of cumulative risk assessments focused on (1) ecological effects assuming acute exposure, (2) ecological effects assuming chronic exposure, (3) effects on human health due to recreational activities (e.g., incidental water consumption) and (4) effects on human health due to lifetime exposure (i.e., surface water as the primary drinking water source). In all four scenarios, we employed a cumulative benchmark-based toxicity quotient (TQ) approach18,77 where individual TQ (ratio of exposure concentration to corresponding benchmark) are summed using a non-interactive concentration addition model78 to predict cumulative TQ (ΣTQ).
To inform potential aggregated acute (scenario 1) and chronic (scenario 2) ecological effects broadly, we used EPA's freshwater aquatic life quality criteria (water quality standards [WQS]) for priority pollutants)79 and Montana Department of Environmental Quality (MTDEQ) aquatic life standards.80 Benchmark-based assessments of aggregate organic contaminant (e.g., pesticide) risk were conducted for fish, aquatic invertebrates and vascular/nonvascular plants using EPA Office of Pesticide Programs (OPP) aquatic life benchmark(s) (ALB) for acute (scenario 1) and chronic (scenario 2) effects of individual pesticides.81 To assess short-term exposure to humans through recreational activities (scenario 3; Table S9b), we included a separate assessment of aggregated risk based on the most protective criteria for E. coli (e.g. primary contact through recreation; not to exceed 126 cfu/100 mL)82 and cyanotoxins.83 Lastly, to provide insight into potential risks of use of surface water as primary drinking-/cooking-water source (scenario 4), the most protective human-health benchmarks (i.e., lowest benchmark concentration) among maximum contaminant level goal(s) (MCLG),84,85 World Health Organization (WHO) guideline values (GV) and provisional GV (pGV),86 and other available state benchmarks were employed (Table S9b), assuming life-time exposure conditions, as described previously.18 MCLG values of zero (i.e., no identified safe-exposure level for sensitive sub-populations, including infants, children, the elderly, and those with compromised immune systems and chronic diseases)84,87 were set to 0.1 µg L−1 for arsenic, lead, uranium and 0.0001 µg L−1 for PFOA.18 Due to the inclusion of a margin of safety in health benchmarks, for all assessments a ΣTQ = 1 indicates a high probability of risk whereas a ΣTQ < 0.1 indicates negligible risk. ∑TQ results and respective health-based benchmarks are summarized in Tables S10a–d. The toxEval version 1.4.0 package88 and the open source statistical software R89 were used for all screening assessments.
Results and discussion
Water security and sustainability are issues affecting all aspects of the northern plains Apsáalooke from drinking water to recreation and spiritual practices.7,15,35 This study and its companion effort37 were designed to inform the potential ecological and human-health risks from exposures to organic and inorganic contaminant mixtures present on the Crow Reservation at Tribe-selected sites. Surface-water sites were selected based on their intended uses (e.g., summer swimming holes and sacred ceremonies) as well as their potential vulnerability to both point and nonpoint sources (e.g., agricultural activities, unlined landfills, wastewater inputs). The Apsáalooke connection to the rivers has been passed through generations. However, due to water-quality concerns, use of the rivers for drinking, swimming and fishing has decreased over time.7 Information on the contaminant mixtures present and their potential health implications can support protection of the community's environmental health and water resources across the Crow Reservation.Surface-water quality: mixtures of contaminants and pathogens
Mixtures of organic and inorganic contaminants as well as E. coli were observed in surface water collected from the three valleys on the Crow Reservation (Tables S4–S6 (ref. 37)). Of the 435 organic contaminants measured in the 19 surface-water samples, 31 (7%) were detected at least once including eight pesticides, eight VOCs, eight pharmaceuticals, five PFAS and two cyanotoxins (Table S4), with detections per sample ranging from 0 to 10 (median: 3). In contrast to previous studies in rivers and streams across the US,21,22 individual concentrations and the number of organic contaminants detected in the summer of 2022 and 2023 were low across all three rivers (median: 25 ng L−1; IQR: 1–7 ng L−1; Fig. S1 and Table S4), with no exceedances of available acute or chronic aquatic-life or human-health benchmarks. The most frequently detected compounds were the VOCs butanal (57%; 11 of 19 samples) and carbon disulfide (52%; 10 of 19 samples), the pharmaceutical metformin (26% 5 of 19 samples) and the herbicide glyphosate (21%; 4 of 19 samples). PFAS were observed infrequently in surface waters at concentrations ranging from 0.1–2 ng L−1 (Table S4), consistent with prior findings in areas without direct point sources.90 The greatest number of compounds were observed at two agriculturally impacted sites (BH2, LBH4), one site below a wastewater lagoon near Ft. Smith, MT (BH1) and a spring impacted by a leaking septic tank (PC1; Table S4), indicating a higher potential risk to aquatic organisms and people in areas influenced by point sources. Point and non-point sources, including agricultural activities (e.g., pesticide and biosolid applications91) and municipal/industrial wastewater,21,92 have been long been identified as primary drivers of contaminant exposure to aquatic organisms.Of the 37 inorganic constituents measured, 33 were observed at least once in surface-water samples. Reporting limits for eight constituents (antimony, arsenic, cadmium, cobalt, lead, molybdenum, nickel and selenium) were higher in 2022 compared to 2023 due to method differences (Table S2) which resulted in non-detects in samples collected in 2022 compared to 2023 (Table S5). Trace elements including aluminum, barium, boron, iron, lithium, manganese, strontium, thallium, uranium and zinc were observed in 100% (19/19) of the samples collected (Table S5 and Fig. S2).37 Only two exceedances of available aquatic-life benchmarks were observed (Table S5). Copper was detected in less than 50% of the samples with one exceedance (9.0 µg L−1 site PC1) of the aquatic life benchmark for non-vascular plants (ALB: 3.1 µg L−1), whereas iron was observed in 100% of the samples with one exceedance (1240 µg L−1 site LBH4) of EPA's chronic freshwater aquatic life criteria (WQS: 1000 µg L−1; Table S5). Trace elements observed herein are naturally occurring, but elevated concentrations can result from anthropogenic activities93 and have the potential to negatively affect human and ecological health particularly when exposures occur as mixtures.
General heterotrophic bacteria (heterotrophic plate counts; HPC), total coliforms and E. coli were observed frequently in surface waters from the three basins (Fig. 2 and Table S6). Heterotrophic bacteria which are ubiquitous in the environment94 were detected in 89% (17/19) of surface waters [median: 3700 CFU/100 mL (IQR: 680–11,000 CFU/100 mL)] with no detections at either the agricultural pond in the Bighorn Valley (BH2) or in the Little Bighorn River in Crow Agency (LBH8) in 2023. Total coliforms were observed in 95% (18/19) of surfaces waters [median: 850 CFU/100 mL (IQR: 402–1650 CFU/100 mL) with no detections at site LBH6 in 2023. E. coli was observed less frequently (68% [13/19] of samples collected in 2022 and 2023 [median: 110 CFU/100 mL; IQR: 50–400 CFU/100 mL]). In rural settings, E. coli and other fecal indicator bacteria are widespread, occur widely in wildlife and domesticated animals and may enter waterways at elevated concentrations through domestic sewage and runoff from ranching and agricultural practices.33 With the abundance of homes and ranching operations proximal to streams across the reservation, and aging public wastewater infrastructure, leaking septic tanks, livestock and inadequate municipal treatment may be potential sources that could negatively affect tribal water quality. More frequent sampling across time and space as well as detailed microbial source tracking studies,34,95 could identify specific sources and drivers (e.g., seasonality) of E. coli abundance in the study area.
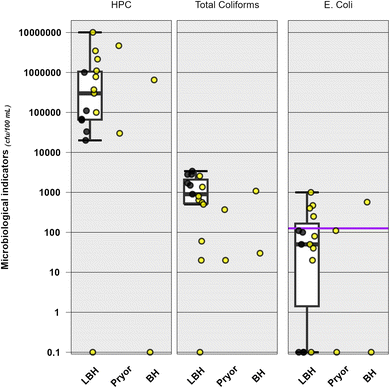 | ||
Fig. 2 Concentrations (colony forming units per 100 mL) of heterotrophic plate counts (HPC, left plot), total coliforms (center plot), and Escherichia coli (E. coli, right plot) detected during 2022 (black circles, ●) and 2023 (yellow circles,  ) in surface water samples collected from up to nine sites in the Little Bighorn River (LBH), two sites in the Bighorn River drainage (BH) and two sites along Pryor Creek (PC) within the Crow Reservation, Montana. Boxes, centerlines, and whiskers indicate interquartile range, median, and 5th and 95th percentiles, respectively. Purple solid line represents Montana Department of Environmental Protection's recreational criteria for E. coli.82 ) in surface water samples collected from up to nine sites in the Little Bighorn River (LBH), two sites in the Bighorn River drainage (BH) and two sites along Pryor Creek (PC) within the Crow Reservation, Montana. Boxes, centerlines, and whiskers indicate interquartile range, median, and 5th and 95th percentiles, respectively. Purple solid line represents Montana Department of Environmental Protection's recreational criteria for E. coli.82 | ||
Montana DEQ has issued E. coli criteria for class A and B waters during different times of the year based on recreational use. Further, Montana's E. coli criteria for class A use waters are associated with drinking water and are more stringent because these waters are to be maintained suitable for drinking-water use after simple disinfection rather than conventional treatment.82 The year-round criterion for drinking water is 32 CFU/100 mL which was exceeded in all but one surface-water site with detections (Fig. 2 and Table S6). Recreational E. coli criteria for class A and B waters in the summer (April 1–October 31) are 126 CFU/100 mL which was exceeded at five of the 13 sites where E. coli was observed (Fig. 2). Exceedances of the recreational E. coli criteria were not observed in the swimming hole upstream near Wyola (LBH2; 2022: 110 CFU/100 mL and 2023: 20 CFU/100 mL)) but were observed at the Crow Agency swimming hole (LBH10; 1000 CFU/100 mL), indicating a potential recreation risk to the community particularly children during the summer months. The Little Bighorn River in Crow Agency (LBH7) serves as a drinking-water source for the Crow Reservation; previous data have reported E. coli concentrations >7000 most probably number (MPN) per 100 mL at the drinking-water intake during spring run-off conditions,33 which resulted in several studies designed to assess E. coli contamination throughout the reservation with an emphasis on private-well drinking water.16 In the current study, E. coli concentrations collected just above the drinking-water intake in 2022 and 2023 were 100 and 250 CFU/100 mL, respectively (Table S6) but despite detections in surface water, E. coli was not observed in the Crow public water supply prior to distribution,37 indicating effective removal during treatment. Chlorine-based disinfection (e.g., chlorination, chloramination, or chlorine dioxide) is common in US public-supply drinking water96 and designed to reduce or eliminate exposures to harmful microorganisms, including viruses and pathogenic bacteria.97
Potential ecological and human-health effects
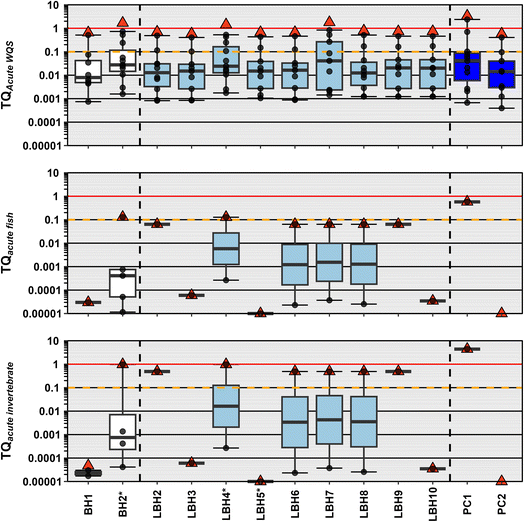
Several benchmark-based ΣTQ screening approaches were used to estimate both the ecological and human-health risks of exposure to contaminant mixtures in surface waters collected from the Crow Reservation. Unlike available human-health benchmarks (Table S9b), the available aquatic life benchmarks for contaminants observed in this study are limited primarily to pesticides and trace metals (Table S9a). We assessed the acute and chronic risk to aquatic life using the most conservative freshwater aquatic life standards (WQS) based on information from EPA79 and MTDEQ80 (Table S9a). As an additional line of evidence for potential risks to vertebrates and to inform the potential risks to lower aquatic trophic levels, analogous EPA OPP ALB-based ΣTQ risk approaches22 were employed, wherein the potential risks associated with pesticide exposures were assessed based on acute and chronic benchmarks for fish, invertebrates and vascular/nonvascular plants81 (Table S9a).Based on freshwater aquatic life WQS, all samples exceeded a ΣTQWQS of 1 (Fig. 3 and Table S10a) indicating a high probability of adverse effects from co-occurring exposures to multiple trace metals, if measured concentrations are indicative of persistent, chronic exposures, with risk driven primarily by lead, aluminum, copper and zinc depending on sample (Fig. 3). Assuming measured concentrations are indicative of transient, acute-exposure conditions, four samples (PC1, BH2, LBH4, LBH7) exceeded a ΣTQWQS of 1 indicating a high probability of aggregated risk, and the remaining 15 samples exceeded a ΣTQWQS of 0.1 indicating a potential risk to aquatic life (Fig. S3), with copper and zinc as primary risk drivers (Table S10a).
Based on OPP ALB, no sites exceeded ΣTQfish-acute of 1 for fish, but eight sites (PC1, BH2, LBH2, LBH4, LBH6, LBH7, LBH8, LBH9) exceeded a ΣTQfish-chronic of 0.1 for chronic exposure conditions and four of those (PC1, BH2, LBH4, LBH7) also exceeded a ΣTQfish-acute of 0.1 for acute exposure conditions (Fig. 3, S3 and Table S10b). One sample (PC1) exceeded the OPP ALB-based ΣTQinvertebrate-acute of 1 whereas seven sites (BH2, LBH2, LBH4, LBH6, LBH7, LBH8, LBH9 exceeded the ΣTQinvertebrate-acute of 0.1 for acute exposures to invertebrates (Fig. S3), with copper driving the exposure risk. Similarly, chronic exposure risk to invertebrates was also driven by copper with four sites (PC1, BH2, LBH4, LBH7) exceeding a ΣTQinvertebrate-chronic of 1 and four (LBH2, LBH6, LBH8, LBH9) exceeding a ΣTQinvertebrate-chronic of 0.1 (Fig. 3). No OPP ALB-based ΣTQplant > 1 were observed for vascular or nonvascular plants (Table S10b). Copper is an essential mineral that at low concentrations is vital to organismal health but has also been used as a pesticide (bactericide, algaecide, fungicide) for centuries.98 Exposure to copper has the potential to affect ecosystem processes by reducing biodiversity at the lower trophic levels, including phototrophic and heterotrophic microbial communities and terrestrial and aquatic invertebrates.98 For vertebrates, prolonged exposure to copper can result in oxidative stress leading to tissue damage and effects on bioenergetics and endocrine function.99
To inform potential short-term exposure risk to the Crow community through recreational activities, we assessed aggregated risk based on the most protective criteria for E. coli82 and cyanotoxins.83 Five sites, one in the Bighorn (BH2) and four in the Little Bighorn (LBH3, LBH7, LBH8, LBH10) including a local swimming hole (LBH10) exceeded a ΣTQrecreation of 1 indicating a high probability of aggregated risk from short-term recreational activities (Fig. S4 and Table S10c) driven predominantly by E. coli. Subsequent recreational risks from cyanotoxins were low across our study area as anatoxin-a was only detected at site BH2, a local farm pond. During this study, only two sites had a ΣTQrecreation < 0.1 indicating no risk (Fig. S4). Although only two locations (LBH2 and LBH10) have been identified as community swimming holes, the water and its surrounding shoreline is utilized for other traditional practices and ceremonies. Understanding potential short-term risk through recreation or contact will help the community determine if preventative measures or mitigation activities should be taken at various locations to improve water quality and reduce potential exposures.
Lastly, to inform the risks of systematic consumption of untreated surface-water for cooking and drinking, historically commonplace but increasingly rare practice, we assumed lifetime exposure and employed the most protective human-health benchmarks. Every site exceeded a ΣTQHH > 1 indicating high aggregated risk to human-health (Fig. S4) with arsenic, uranium and PFOA, all with MCLG of zero, driving the exposure risk (Table S10d). Similarly, frequent ∑TQHH > 1 exceedances were observed in residential private wells collected throughout the community in tandem with the current study with exposure risk being driven predominantly by uranium and arsenic37 illustrating the hydrologic connectivity between shallow groundwater wells and surface waters. Historically, drinking water was sourced from springs and rivers within the three watersheds sampled as part of this study.7 Today public drinking water supply for Crow Agency is sourced from the Little Bighorn15 (downstream of site LBH7), and most residential private wells are proximal to the rivers, drilled to first water and thus hydrologically connected to and influenced by adjacent surface-water quality.15,100
Synoptic studies like this one are only the first step in addressing surface water quality concerns expressed by the Crow community. More observations across more sites/seasons could enhance understanding of human and ecological exposure risk and support future mitigation, conservation or remediation plans.
Occurrence, accumulation and potential human-health effects of per- and polyfluoroalkyl substances in sediment, biofilms and biota
As noted previously, PFAS are widespread in surface water, groundwater, public drinking water supplies prior to distribution and private-well tapwater,19,101,102 so this study also sought to address potential PFAS contamination in Tribal waters and aquatic resources including sediment, biofilms and fish filets and whole blood. PFAS concentrations in fish filets informed a secondary route of human exposure through consumption, while whole blood concentrations provided insight into potential ecological exposure and accumulation. Ecologically, sediment and biofilms could be important sources of PFAS to lower trophic-level aquatic organisms including invertebrates, larval fish and larval frogs and toads. Further, microbial consortia have demonstrated the ability to utilize PFAS as a carbon source while facilitating defluorination.103,104Similar to surface water, the number and concentrations of individual PFAS in sediment and biofilms were low (<5 ng g−1) across all sites sampled with no overlap between sediment and water. In sediment, only two PFAS were observed including a single detection of 6:2 FTS (1H,1H,2H,2H-perfluorooctanesulfonate; C8) from an irrigation ditch in the Little Bighorn River basin (LBH5) and N-MeFOSA (N-methylperfluorooctanesulfonamide; C8), which was detected in 83% (10/12) of the sites with a median concentration of 0.43 ng g−1 dw (IQR: 0.42–0.45 ng g−1 dw; Table S7). In biofilms, three PFAS of varying chain length (C4–C13) were observed infrequently at sub ng g−1 levels, with no overlap between biofilms and water or sediment. It is important to note, method surrogate recoveries were low (median: 42.4%; IQR: 30.8–50.3%) and some were outside the acceptable range for method performance, indicating values should be considered estimated. We observed single detections of PFHxA (perfluorohexanoic acid; C6), PFTrDA (perfluorotridecanoic acid; C13) and PFBS (perfluorobutanesulfonic acid; C4) (Table S7;61). With the exception of PFTrDA, individual PFAS observed in biofilms in our study included shorter chain compounds (C4–C7), reported frequently in surface waters105 globally. Although detection was infrequent, our results contrast with previous reports of higher accumulation of longer chain PFOS (perfluorooctanesulfonic acid) and perfluoroalkyl carboxylic acids (PFCAs; C8–C14) and noticeable differences in chain length between biofilms and water.57,106 Members of the bacterial order Pseudomonadales (class: Gammaproteobacteria) —including Pseudomonas spp.—were detected (0.8–3.1% relative abundance) as part of a diverse biofilm community at all five sites sampled (Fig. S5). Gammaproteobacteria are a diverse class of bacteria with certain Pseudomonas strains having the potential to degrade PFAS107 and their presence in biofilms suggests the potential for in situ degradation. This possibility is further supported by the observation that the PFAS detected in the biofilms (PFHxA and PFBS) were shorter-chain compared to those found in the surrounding sediment (6:2 FTS and N-MeFOSA), which may indicate microbial transformation or breakdown of longer-chain PFAS within the biofilm matrix to terminal products (e.g. PFBA, PFHxA) that can also persist in the environment.108 Supporting this interpretation, previous studies have shown that 6:2 FTS can rapidly transform into PFPeA, PFHxA, and 5:3 acid under aerobic conditions, with limited transformation occurring under anaerobic conditions.109,110 Although limited, these findings underscore the potential importance of oxygen-rich microenvironments—such as those found in riverine biofilms—in potentially facilitating degradation of some longer-chain PFAS.
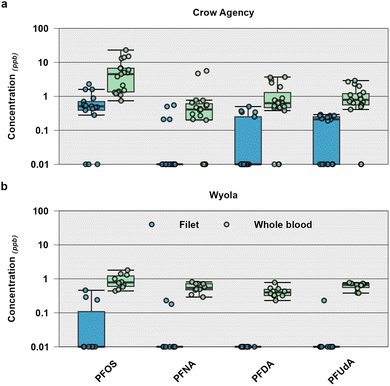
To provide initial insight into potential PFAS occurrence, bioaccumulation and human-health implications of consumption of fish caught within the Crow Reservation, whole blood and filets were collected from 29 individual fish in 2023 at two sites on Little Bighorn River and analyzed for 28 individual PFAS (Tables 1 and S8;61). Of the 28 PFAS measured in fish tissues, 13 (46%) were detected at least once. Similar to other studies,90,111,112 PFOS (C8; 82%; 46 of 56 samples) and several other long chain PFCAs including PFUdA (perfluoroundecanoic acid (C11); 68%; 38/56), PFDA (perfluorodecanoic acid (C10); 55%; 31 of 56 samples) and PFNA (perfluorononanoic acid (C9); 55%; 31/56) were detected frequently in fish (Fig. 4). The median PFAS concentration across sites and species was 0.59 ng g−1 wet weight (ww) (IQR: 0.35–1.1 ng g−1 ww) with cumulative PFAS concentrations ranging from non-detect-34.9 ng g−1 ww (median: 2.83 ng g−1 ww; Fig. 5 and Table S8). These results were within the low end of the range of concentrations observed in a variety of sportfish species collected across the US (median: 11.8; range 0.43–286 ng g−1 ww).111 Cumulative PFAS concentrations observed in this study are similar to those found in commercial fish and shellfish collected from across the US (0.45–17.80 ng g−1 ww).113 Similarly, PFOS was the most frequently detected compound in commercial fish in the US and globally.113 Further, the low fish PFAS concentrations (<40 ng g−1 ww) observed in our study, are consistent with prior reports of lower fish tissue concentrations in rural/remote areas with limited PFAS sources, compared to urban areas.114
Concentrations and the numbers/types of individual PFAS varied among species (Crow Agency only, data not shown) and tissue type (filet vs. whole blood), irrespective of site (Fig. 5). For example, in brown trout collected upstream of Wyola, seven PFAS were detected in filets and nine PFAS were detected in whole blood samples (Table S8) whereas in fish collected from Crow Agency six PFAS were observed in filets and 10 were observed in whole blood samples (Table S8). Median individual PFAS concentrations in brown trout filet and whole blood collected from above Wyola were 0.290 ng g−1 ww (IQR: 0.230–0.900 ng g−1 ww) and 0.540 ng mL−1 (IQR: 0.370–0.730 ng mL−1), respectively.
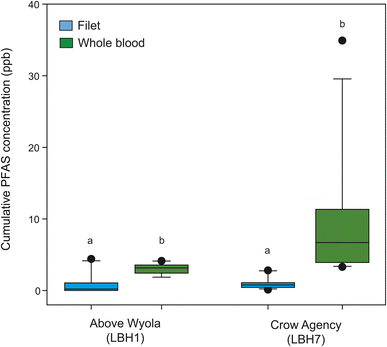
In fish from above Wyola, median cumulative PFAS were higher (p = 0.0001) in whole blood (3.16 ng g−1 ww) than in filets (0.350 ng g−1 ww; Fig. 5), and whole blood had a greater number (median: 5.5) of PFAS detected compared to filets (median: 1; p = 0.0033). Similarly, fish from Crow Agency higher median cumulative PFAS concentrations (p = 0.0002; Fig. 5) and greater number of detected compounds (p = 0.001) in whole blood (concentration: 6.70 ng mL−1; number: 6) compared to filets (concentration: 0.780 ng g−1 ww; number: 2). PFAS typically do not behave like neutral hydrophobic contaminants and instead prefer proteins due to their anionic nature,101 resulting in generally higher concentrations in blood/plasma and other organs compared to filets.115
Despite the limited number of individual fish collected per species, we did note several species level differences in filets from Crow Agency where we were able to collect multiple species. A greater number of individual PFAS in goldeye (median: 4) filets compared to catfish (median: 2) filets (p = 0.015) were observed, as well as higher median cumulative concentrations in goldeye (2.73 ng g−1 ww) filets compared to catfish (0.470 ng g−1 ww; p = 0.03). No other species differences were observed for filets or whole blood and due to potential site level differences, we were not able to compare all species collected. Catfish, particularly the channel catfish, which is native to Montana and prevalent in the Yellowstone and Missouri River drainages including the Little Bighorn River, are omnivorous benthic feeders whereas goldeye and rock bass are carnivorous pelagic feeders. Differences in concentrations and number of individual PFAS detected could be a result of feeding guild (e.g., benthic, pelagic, omnivore, piscivore, etc.), habitat, body size and collection location.116 Despite some apparent differences among species at one location, more information across a larger number of species, life histories and habitats could help determine which species are most at risk of PFAS accumulation in the study area. Fish consumption advisories are issued by federal, state, Tribal, or local entities in the US to inform communities on safe consumption of locally caught fish and national advisories have been developed for ubiquitous contaminants such as mercury and polychlorinated biphenyls. Significant variations in PFAS bioaccumulation and tissue concentrations among species and waterbodies117 pose unique challenges to the development of national fish consumption advisories for PFAS. As a result, several states have derived their own PFAS-driven fish consumption guidelines, many of which focus only on PFOS, thus likely underestimating potential exposure risk. Further, differences in the PFOS trigger concentrations, which underlie various state PFOS fish consumption advisories, create challenges for risk communication.118 In the absence of PFAS fish consumption advisories nationally or for Montana, we compared observed fish tissue concentrations to both Colorado's newly adopted (May 2025) advisories for PFOS and New Jersey's advisories for PFOS, PFOA, PFNA and PFUdA (Table 2). In New Jersey, advisories are established for the general population and for high risk individuals including pregnant women, young children and women of childbearing age.90 In Colorado, advisories are established for the general population (anyone over 18 years of age), women of childbearing age (18–40) and children (ages 0–6).119 In Crow Agency, PFOS concentrations in filets corresponded to a Colorado advisory of no more than 4 meals per month (>1.30–2.70) in three individual fish (one rock bass and two goldeye) for the general population and at least one individual among all species sampled (3/9 catfish, 4/5 rock bass and 2/3 goldeye) corresponded to no more than 4 meals per month or less for children ages 0–6 years old. Similarly, at least one individual among all species sampled (1/9 catfish, 3/5 rock bass and 2/3 goldeye) corresponded to the more protective New Jersey advisory consumption frequency of “no more than weekly” for both the general population and high-risk individuals (Table S8). Concentrations of PFNA were also high enough to trigger “no more than weekly” consumption advisories in all three goldeye (0.23–1.6 ng g−1 ww) collected, and one brown trout filet triggered a “no more than weekly” advisory for PFOA (0.62–4.3 ng g−1 ww; Table S8) despite infrequent detections. Although we had a limited number of samples, this preliminary assessment documents, for the first time, fish-tissue PFAS concentrations of human health concern in the Little Bighorn River and highlights that further understanding of PFAS exposures risks of important recreational/subsistence fish species on the Crow Reservation could support risk communication and management of their trust resources.
| CO-consumption frequency | PFOS | NJ – consumption frequency | PFOS | PFNA | PFOA | PFUdA | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Children | Generalb | High risk | General | High risk | General | High risk | High risk | General | High risk | ||
| a NA, not applicable; ng g−1, nanograms per gram; ppb; parts per billion. b Adults including women of childbearing age. | |||||||||||
| 24 Meals per month | 0–0.15 | 0–0.40 | Unlimited (daily) | ≤0.56 | ≤0.56 | ≤0.23 | ≤0.23 | ≤0.62 | ≤0.62 | ≤0.40 | ≤0.40 |
| 4 Meals per month | >0.50–0.90 | >1.30–2.70 | Weekly | 0.56–3.9 | 0.56–3.9 | 0.23–1.6 | 0.23–1.6 | 0.62–4.3 | 0.62–4.3 | 0.40–2.8 | 0.40–2.8 |
| 1 Meal per month | >1.80–3.60 | >5.30–10.70 | Monthly | >3.9–17.0 | >3.9–17.0 | >1.6–6.9 | >1.6–6.9 | >4.3–19.0 | >4.3–19.0 | >2.8–12.0 | >2.8–12.0 |
| 6 Meals per year | >3.60–7.10 | >10.70–21.40 | Once every 3 months | >17.0–51.0 | NA | >6.9–21.0 | NA | >19.0–57.0 | NA | >12.0–36.5 | NA |
| 3 Meals per year | >7.10–14.30 | >21.40–42.80 | Yearly | >17.0–204 | NA | >21.0–84.0 | NA | >19.0–226 | NA | >36.5–146 | NA |
| Consumption not advised | >14.30 | >42.80 | Do not eat | >204 | >17.0 | >84.0 | >6.9 | >226 | >19.0 | >146 | >12.0 |
Limitations
The current study was designed to broadly assess contaminant exposures/accumulation in three basins within the Crow Reservation and thus several notable limitations warrant discussion. First, the temporal (only two snapshots in time) and spatial (primarily Little Bighorn River, with fewer samples in Pryor Creek and the Bighorn River) coverage provides important initial insight and illustrates the importance of more in-depth sampling to more accurately assess surface-water quality within the Crow Reservation. Second, the target analytical scope, while extensive, represents only a fraction of the estimated chemicals in production120 and potentially present in ambient surface waters;21 thus human and ecological exposure risk may be underestimated. Third, ΣTQ approaches utilized herein to assess potential human and ecological health implications are limited by available ecological-endpoint and species-specific benchmarks. Fourth, fish tissues were not analyzed for other bioaccumulative contaminants with consumption advisories (e.g., mercury observed at low concentrations in surface waters), thus likely underestimating potential consumption risk. Finally, a small number of individual fish were collected across sites (n = 2) and species (4 species with 10 or less individuals) limiting our ability to address variability in PFAS concentrations among individual, species, and site. The small sample sizes and limited spatial extent reduced our ability to broadly address potential risk to human-health from consumption at this time. However, despite these notable limitations, the information presented provides the Crow community with information on surface-water quality and corresponding human and ecological health implications.Conclusions
Water is sacred to the Apsáalooke people and over the last few decades, the quality of that water has come into question due to increased human activity, point and non-point source discharges into the rivers/streams, and notable detections of pathogenic bacteria and trace metals in surface waters and drinking water. Currently, there is a paucity of information on low-level exposures to complex mixtures of organic, inorganic and microbial contaminants in Tribal surface waters and even less information on PFAS accumulation in the region. Despite relatively low concentrations in surface waters within the Crow Reservation, mixture toxicity indicated prevalent chronic ecological effects and human-health secondary contact (recreation) effects. Although drinking of untreated surface water is no longer required and is presumed minimal, conservative risk estimates in the event of lifetime drinking-water exposure indicate multiple exceedances of human-health benchmarks, notably for arsenic and uranium, in line with those for concurrently sampled tapwater sourced from stream-adjacent private wells throughout the reservation.37PFAS is another emerging issue for the Crow community with limited information currently available on PFAS sources that could potentially affect water resources on the reservation. This initial snapshot (two sites on the Little Bighorn) indicated PFAS was prevalent in fish tissues and to a lesser extent in biofilms, despite few detections in water and sediment. The results document environmental PFAS contamination within the reservation and the potential for human exposures from consumption of recreational/subsistence fish. Multiple exceedances of the State of New Jersey's “no more than weekly” fish consumption advisories for PFOA, PFOS and PFNA indicated potential PFAS human-health concerns for the general population and high-risk individuals. Studies designed to address potential risk from fish consumption could potentially support the development of consumption advisories by focusing on species of Tribal importance across a greater spatial extent based on a detailed understanding of Tribal-consumption patterns (e.g., meal size and frequency).
Studies like these involving a Tribal-USGS-academic partnership and designed around the principles of community-based participatory research help to ensure that (1) the research addresses questions that are of importance to the Tribal community as well as to science, (2) the community understands and values the results and is more empowered to act on them to reduce perceived risks and (3) the research partnership provides opportunities for local students to learn and advance their careers in science in ways their community values. The results of the current study emphasize the importance of continued characterization of contaminant exposures in surface waters throughout the Crow Reservation with an emphasis on PFAS accumulation and human-health risk associated with consumption of fish. Although only a snapshot in time and space, this preliminary reconnaissance provided information on the quality of surface waters to support Tribal conservation and risk mitigation decision-making.
Author contributions
Apsáalooke culture, language, ecological knowledge: JD (Tribal Elder), CM, EMC, JWL, MC. Conceptualization: JD, KLS, KMR, MJE, PMB. Methodology: JD, CM, EB, KLS, KMR, MJE, PMB. Investigation: AMG, CM, CLR, JD, JLG, KAL, KLS, KMR, LKK, JS, MJE, MTPMB, PB RBM, SMM. Data curation: JD, KLS, KMR, MLS, PMB. Formal analysis: KLS, KMR, MLS. Visualization: KLS, KMR, SEG. Community engagement & education: JD (Tribal Elder), CM, EB, JWL, KLS, MC, MJE, PMB. Writing – original draft: KLS. Writing – review & editing: AMG, CLR, CM, CML, EMC, EPB, JD, JLG, JS, JWL, KAL, KLS, KMR, LKK, MC, MJE, MLS, MT, PMB, PB, RBM, SAE, SEG, SJW, SMM. Supervision (student interns): JD, MJE. Project administration: JD, KLS, MJE, PMB. Funding: AMG, CML, JD, KAL, KLS, MJE, PMB.Conflicts of interest
The authors declare that they have no competing interests that could have appeared to influence the work reported in this paper.Data availability
Data are available in two USGS ScienceBase data releases. Water quality and contaminant data for all surface water samples as well as detailed information on analytes and detection limits for each of the methods are available at https://doi.org/10.5066/P9R7MV6I. PFAS concentrations measured in sediment, biofilms and fish tissue as well as detailed information on analytes and detection limits for each of the methods are available at https://doi.org/10.5066/P1DAMSMX. Data related to raw DNA sequences were placed in the National Center for Biotechnology Information (NCBI) database under bioproject number: PRJNA1291266.Supplementary information (SI): Tables S1–S10. See DOI: https://doi.org/10.1039/d5em00565e.
Acknowledgements
The USGS, the Crow Water Quality Project, and Montana State University thank the Apsáalooke people for their support of this research. We would also like to thank Myra Lefthand and Sara Young of the Crow Environmental Health Steering Committee and its Crow Water Quality Project for their support as well as the Guardians of the Living Water program for help with fish collection. Funding for this research was provided by the USGS Ecosystems Mission Area, Environmental Health Program with additional funding provided by RII Track-1 Consortium for Research on Environmental Water Systems, Award # 1757351, Montana EPSCoR Program, National Science Foundation; Center for Native Environmental Health Equity Research, Award # 9P50MD015706-06 to University of New Mexico Health Sciences Center, NIH Center of Excellence on Environmental Health Disparities Research, National Institute of Environmental Health Sciences & NIMHD, National Institutes of Health (NIH), subaward 3RJN7 to Montana State University Bozeman; USDA National Institute for Food and Agriculture, New Beginnings for Tribal Students, Award # 2021-70411-35304 to Montana State University Bozeman and subaward to Little Bighorn College. The microbial indicator assessments were supported in part by the Intramural Research Program of the National Institutes of Health (NIH). The contributions of the NIH authors were made as part of their official duties as NIH federal staff, are in compliance with agency policy requirements, and are considered works of the United States Government. The findings and conclusions presented in this paper do not necessarily reflect the views of the NIH or the Department of Health and Human Services. We thank Dr Brent Peyton (MSU) as a collaborator and supervisor of John Shikany. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. This report contains CAS Registry Numbers®, which is a registered trademark of the American Chemical Society. CAS recommends the verification of the CASRNs through CAS Client ServicesSM.References
- S. Diver, Native Water Protection Flows Through Self-Determination: Understanding Tribal Water Quality Standards and “Treatment as a State”, J. Contemp. Water Res. Educ., 2018, 163, 6–30, DOI:10.1111/j.1936-704X.2018.03267.x.
- R. Arsenault, S. Diver, D. McGregor, A. Witham and C. Bourassa, Shifting the Framework of Canadian Water Governance through Indigenous Research Methods: Acknowledging the Past with an Eye on the Future, Water, 2018, 10, 49 CrossRef , https://www.mdpi.com/2073-4441/10/1/49.
- M. Blackstock, Water: A First Nations' Spiritual and Ecological Perspective, J. Ecosyst. Manage., 2001, 1, 1–14, DOI:10.22230/jem.2001v1n1a216.
- J. Reid and M. Rout, Getting to know your food: the insights of indigenous thinking in food provenance, Agric. Hum. Val., 2016, 33, 427–438, DOI:10.1007/s10460-015-9617-8.
- K. Cozzetto, K. Chief, K. Dittmer, M. Brubaker, R. Gough, K. Souza, F. Ettawageshik, S. Wotkyns, S. Opitz-Stapleton, S. Duren and P. Chavan, Climate change impacts on the water resources of American Indians and Alaska Natives in the U.S, Clim. Change, 2013, 120, 569–584, DOI:10.1007/s10584-013-0852-y.
- J. L. Donatuto, T. A. Satterfield and R. Gregory, Poisoning the body to nourish the soul: Prioritising health risks and impacts in a Native American community, Health Risk Soc., 2011, 13, 103–127, DOI:10.1080/13698575.2011.556186.
- C. Martin, J. Doyle, J. LaFrance, M. J. Lefthand, S. L. Young, E. Three Irons and M. J. Eggers, Change Rippling through Our Waters and Culture, J. Contemp. Water Res. Educ., 2020, 169, 61–78, DOI:10.1111/j.1936-704X.2020.03332.x.
- S. M. P. Sulliván, D. Hedden-Nicely and G. Bulltail, Enhancing water protection on Tribal lands, Front. Ecol. Environ., 2024, 22, e2751, DOI:10.1002/fee.2751.
- U.S. Department of the Interior and U.S. Department of Agriculture, Order No 3403: Joint Secretarial Order on Fulfilling the Trust Responsibility to Indian Tribes in the Stewardship of Federal Lands and Waters, Washington DC, 2021 Search PubMed.
- H. Tanana, J. Combs and A. Hoss, Water Is Life: Law, Systemic Racism, and Water Security in Indian Country, Health Secur., 2021, 19, S78–s82 CrossRef PubMed.
- M. Sobel, T. R. Sanchez, T. Zacher, B. Mailloux, M. Powers, J. Yracheta, D. Harvey, L. G. Best, A. B. Bear, K. Hasan, E. Thomas, C. Morgan, D. Aurand, S. Ristau, P. Olmedo, R. Chen, A. Rule, M. O'Leary, A. Navas-Acien, C. M. George and B. Bostick, Spatial relationship between well water arsenic and uranium in Northern Plains native lands, Environ. Pollut., 2021, 287, 117655 CrossRef CAS PubMed.
- M. Powers, T. R. Sanchez, M. Grau-Perez, F. Yeh, K. A. Francesconi, W. Goessler, C. M. George, C. Heaney, L. G. Best, J. G. Umans, R. H. Brown and A. Navas-Acien, Low-moderate arsenic exposure and respiratory health in American Indian communities in the Strong Heart Study, Environ. Health, 2019, 18, 104 CrossRef CAS.
- J. C. Ingram, L. Jones, J. Credo and T. Rock, Uranium and arsenic unregulated water issues on Navajo lands, J. Vac. Sci. Technol. A, 2020, 38 DOI:10.1116/1.5142283.
- G. E. A. Amarawansha, F. Zvomuya and A. Farenhorst, Water delivery system effects on coliform bacteria in tap water in First Nations reserves in Manitoba, Canada, Environ. Monit. Assess., 2021, 193, 339, DOI:10.1007/s10661-021-09114-x.
- J. T. Doyle, L. Kindness, J. Realbird, M. J. Eggers and A. K. Camper, Challenges and Opportunities for Tribal Waters: Addressing Disparities in Safe Public Drinking Water on the Crow Reservation in Montana, USA, Int. J. Environ. Res. Publ. Health, 2018, 15, 567 CrossRef PubMed , https://www.mdpi.com/1660-4601/15/4/567.
- C. L. Richards, S. C. Broadaway, M. J. Eggers, J. Doyle, B. H. Pyle, A. K. Camper and T. E. Ford, Detection of Pathogenic and Non-pathogenic Bacteria in Drinking Water and Associated Biofilms on the Crow Reservation, Montana, USA, Microb. Ecol., 2018, 76, 52–63, DOI:10.1007/s00248-015-0595-6.
- L. S. Rowles, A. I. Hossain, S. Aggarwal, M. J. Kirisits and N. B. Saleh, Water quality and associated microbial ecology in selected Alaska Native communities: Challenges in off-the-grid water supplies, Sci. Total Environ., 2020, 711, 134450 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0048969719344419.
- P. M. Bradley, K. M. Romanok, K. L. Smalling, S. E. Gordon, B. J. Huffman, K. Paul Friedman, D. L. Villeneuve, B. R. Blackwell, S. C. Fitzpatrick, M. J. Focazio, E. Medlock-Kakaley, S. M. Meppelink, A. Navas-Acien, A. E. Nigra and M. L. Schreiner, Private, public, and bottled drinking water: Shared contaminant-mixture exposures and effects challenge, Environ. Int., 2024, 195, 109220 CrossRef PubMed.
- K. L. Smalling, K. M. Romanok, P. M. Bradley, M. C. Morriss, J. L. Gray, L. K. Kanagy, S. E. Gordon, B. M. Williams, S. E. Breitmeyer, D. K. Jones, L. A. DeCicco, C. A. Eagles-Smith and T. Wagner, Per- and polyfluoroalkyl substances (PFAS) in United States tapwater: Comparison of underserved private-well and public-supply exposures and associated health implications, Environ. Int., 2023, 178, 108033 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0160412023003069.
- P. M. Bradley, K. M. Romanok, K. L. Smalling, M. J. Focazio, R. Charboneau, C. M. George, A. Navas-Acien, M. O’’Leary, R. Red Cloud, T. Zacher, M. C. Cardon, C. Cuny, G. Ducheneaux, K. Enright, N. Evans, J. L. Gray, D. E. Harvey, M. L. Hladik, K. A. Loftin, R. B. McCleskey, E. K. Medlock Kakaley, S. M. Meppelink, J. F. Valder and C. P. Weis, Tapwater exposures, effects potential, and residential risk management in northern plains nations, Environ. Sci. Technol. Water, 2022, 2, 1772–1788 CAS.
- P. M. Bradley, C. A. Journey, K. M. Romanok, L. B. Barber, H. T. Buxton, W. T. Foreman, E. T. Furlong, S. T. Glassmeyer, M. L. Hladik, L. R. Iwanowicz, D. K. Jones, D. W. Kolpin, K. M. Kuivila, K. A. Loftin, M. A. Mills, M. T. Meyer, J. L. Orlando, T. J. Reilly, K. L. Smalling and D. L. Villeneuve, Expanded target-chemical analysis reveals extensive mixed-organic-contaminant exposure in USA streams, Environ. Sci. Technol., 2017, 51, 4792–4802 CrossRef CAS PubMed , http://pubs.er.usgs.gov/publication/70186864.
- P. M. Bradley, C. A. Journey, K. Romanok, S. Breitmeyer, D. T. Button, D. Carlisle, B. J. Huffman, B. Mahler, L. H. Nowell, S. L. Qi, K. Smalling, I. R. Waite and P. C. VanMetre, Multi-region assessment of chemical mixture exposures and predicted cumulative effects in USA wadeable urban/agriculture-gradient streams, Sci. Total Environ., 2021, 773, 145062 CrossRef CAS PubMed.
- J. Glüge, M. Scheringer, I. T. Cousins, J. C. DeWitt, G. Goldenman, D. Herzke, R. Lohmann, C. A. Ng, X. Trier and Z. Wang, An overview of the uses of per- and polyfluoroalkyl substances (PFAS), Environ. Sci.: Processes Impacts, 2020, 22, 2345–2373 RSC.
- A. K. Tokranov, P. M. Bradley, M. J. Focazio, D. B. Kent, D. R. LeBlanc, J. W. McCoy, K. L. Smalling, J. A. Steevens and P. L. Toccalino, Integrated science for the study of perfluoroalkyl and polyfluoroalkyl substances (PFAS) in the environment—A strategic science vision for the U.S. Geological Survey, Report U. S. Geological Survey Circular 1490, Reston, VA, 2021 Search PubMed.
- X. C. Hu, A. K. Tokranov, J. Liddie, X. Zhang, P. Grandjean, J. E. Hart, F. Laden, Q. Sun, L. W. Y. Yeung and E. M. Sunderland, Tap Water Contributions to Plasma Concentrations of Poly- and Perfluoroalkyl Substances (PFAS) in a Nationwide Prospective Cohort of U.S. Women, Environ. Health Perspect., 2019, 127, 067006, DOI:10.1289/EHP4093.
- G. Liu, K. Dhana, J. D. Furtado, J. Rood, G. Zong, L. Liang, L. Qi, G. A. Bray, L. Dejonge, B. Coull, P. Grandjean and Q. Sun, Perfluoroalkyl substances and changes in body weight and resting metabolic rate in response to weight-loss diets: A prospective study, PLoS Med., 2018, 15, e1002502, DOI:10.1371/journal.pmed.1002502.
- P. Grandjean and E. Budtz-Jørgensen, Immunotoxicity of perfluorinated alkylates: calculation of benchmark doses based on serum concentrations in children, Environ. Health, 2013, 12, 35 CrossRef CAS PubMed.
- V. Barry, A. Winquist and K. Steenland, Perfluorooctanoic Acid (PFOA) Exposures and Incident Cancers among Adults Living Near a Chemical Plant, Environ. Health Perspect., 2013, 121, 1313–1318, DOI:10.1289/ehp.1306615.
- D. E. Yarlott Jr, Historical Uses of Natural Resources: Transference of Knowledge in the Crow Indian Environment, Montana State University, Montana State University, Bozeman, MT, 1999 Search PubMed.
- J. T. Doyle, M. H. Redsteer and M. J. Eggers, in Climate Change and Indigenous Peoples in the United States: Impacts, Experiences and Actions, ed. J. K. Maldonado, B. Colombi and R. Pandya, Springer International Publishing, Cham, 2014, pp. 135–147, DOI:10.1007/978-3-319-05266-3_11.
- C. Martin, V. W. Simonds, S. L. Young, J. Doyle, M. Lefthand and M. J. Eggers, Our Relationship to Water and Experience of Water Insecurity among Apsáalooke (Crow Indian) People, Montana, Int. J. Environ. Res. Publ. Health, 2021, 18, 582 Search PubMed , https://www.mdpi.com/1660-4601/18/2/582.
- C. Cummins, J. Doyle, L. Kindness, M. J. Lefthand, U. J. Bear Don’t Walk, A. Bends, S. C. Broadaway, A. K. Camper, R. Fitch, T. E. Ford, S. Hamner, A. R. Morrison, C. L. Richards, S. L. Young and M. J. Eggers, Community-Based Participatory Research in Indian Country: Improving Health Through Water Quality Research and Awareness, Fam. Community Health, 2010, 33, 9 Search PubMed.
- S. Hamner, S. C. Broadaway, E. Berg, S. Stettner, B. H. Pyle, N. Big Man, J. Old Elk, M. J. Eggers, J. Doyle, L. Kindness, B. Good Luck, T. E. Ford and A. C. Camper, Detection and source tracking of Escherichia coli, harboring intimin and Shiga toxin genes, isolated from the Little Bighorn River, Montana, Int. J. Environ. Health Res., 2014, 24, 341–362, DOI:10.1080/09603123.2013.835030.
- S. Hamner, B. L. Brown, N. A. Hasan, M. J. Franklin, J. Doyle, M. J. Eggers, R. R. Colwell and T. E. Ford, Metagenomic Profiling of Microbial Pathogens in the Little Bighorn River, Montana, Int. J. Environ. Res. Publ. Health, 2019, 16, 1097 Search PubMed , https://www.mdpi.com/1660-4601/16/7/1097.
- M. J. Eggers, J. T. Doyle, M. J. Lefthand, S. L. Young, A. L. Moore-Nall, L. Kindness, R. Other Medicine, T. E. Ford, E. Dietrich, A. E. Parker, J. H. Hoover and A. K. Camper, Community Engaged Cumulative Risk Assessment of Exposure to Inorganic Well Water Contaminants, Crow Reservation, Montana, Int. J. Environ. Res. Publ. Health, 2018, 15, 76 CrossRef PubMed , https://www.mdpi.com/1660-4601/15/1/76.
- K. W. Brown, B. Gessesse, L. J. Butler and D. L. MacIntosh, Potential effectiveness of point-of-use filtration to address risks to drinking water in the United States, Environ. Health Insights, 2017, 11, 1178630217746997 Search PubMed.
- K. M. Romanok, P. M. Bradley, K. Smalling, J. T. Doyle, M. J. Eggers, J. L. Gray, M. L. Hladik, L. K. Kanagy, R. B. McCleskey, S. M. Meppelink, C. L. Richards and D. A. Roth, Target-chemical concentrations and microbiological results in surface water and tapwater, Montana, 2022-23, Report U.S. Geological Survey Data Release, 2023, DOI:10.5066/P9R7MV6I.
- N. Wallerstein, B. Duran, J. G. Oetzel and M. Minkler, Community-Based Participatory Research for Health: Advancing Social and Health Equity, Wiley, 2017 Search PubMed.
- B. A. Israel, A. J. Schulz, E. A. Parker and A. B. Becker, Community-based participatory research: policy recommendations for promoting a partnership approach in health research, Educ. Health, 2001, 14, 182–197 CrossRef CAS.
- K. M. Romanok, D. W. Kolpin, S. M. Meppelink, M. Argos, J. Brown, M. DeVito, J. E. Dietz, C. E. Givens, J. Gray, C. P. Higgins, M. L. Hladik, L. R. Iwanowicz, B. R. McCleskey, C. McDonough, M. T. Meyers, M. Strynar, C. P. Weis, V. Wilson and P. M. Bradley, Methods used for the collection and analysis of chemical and biological data for the Tapwater Exposure Study, United States, 2016–17, Report U.S. Geological Survey Open-File Report 2018-1098, Reston, VA, 2018 Search PubMed.
- K. A. Loftin, J. L. Graham, E. D. Hilborn, S. C. Lehmann, M. T. Meyer, J. E. Dietze and C. B. Griffith, Cyanotoxins in inland lakes of the United States: Occurrence and potential recreational health risks in the EPA National Lakes Assessment 2007, Harmful Algae, 2016, 56, 77–90 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S1568988315300883.
- J. L. Graham, K. A. Loftin, M. T. Meyer and A. C. Ziegler, Cyanotoxin Mixtures and Taste-and-Odor Compounds in Cyanobacterial Blooms from the Midwestern United States, Environ. Sci. Technol., 2010, 44, 7361–7368, DOI:10.1021/es1008938.
- E. T. Furlong, M. C. Noriega, C. J. Kanagy, L. K. Kanagy, L. J. Coffey and M. R. Burkhardt, Determination of Human-Use Pharmaceuticals in Filtered Water by Direct Aqueous Injection: High-Performance Liquid Chromatography/tandem Mass Spectrometry, Report 5-B10, Reston, VA, 2014 Search PubMed.
- M. S. Gross, C. J. Sanders, M. D. De Parsia and M. L. Hladik, Methods of Analysis—Determination of Pesticides in Filtered Water and Suspended Sediment Using Liquid Chromatography- and Gas Chromatography-Tandem Mass Spectrometry, Report 5-A12, Reston, VA, 2024 Search PubMed.
- M. L. Hladik, M. J. Focazio and M. Engle, Discharges of produced waters from oil and gas extraction via wastewater treatment plants are sources of disinfection by-products to receiving streams, Sci. Total Environ., 2014, 466–467, 1085–1093 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0048969713009224.
- D. L. Rose, M. W. Sandstrom and L. K. Murtagh, Determination of Heat Purgeable and Ambient Purgeable Volatile Organic Compounds in Water by Gas Chromatography/mass Spectrometry, Report 5-B12, Reston, VA, 2016 Search PubMed.
- J. L. Gray, L. K. Kanagy, C. A. Anderson and C. J. Kanagy, Determination of per- and polyfluoroalkyl substances in water by direct injection of matrix-modified centrifuge supernatant and liquid chromatography-tandem mass spectrometry with isotope dilution, Report Techniques and Methods 5-B13, Reston, VA, 2025 Search PubMed.
- J. W. Ball and R. B. McCleskey, A new cation-exchange method for accurate field speciation of hexavalent chromium, Talanta, 2003, 61, 305–313 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0039914003002820.
- M. J. Fishman and L. C. Friedman, Methods for Determination of Inorganic Substances in Water and Fluvial Sediments, Report 05-A1, 1989 Search PubMed.
- R. L. Hergenreder, Trace Metals in Waters by GFAAS, in Accordance with U.S. EPA and Health Canada Requirements, Perkin Elmer, Inc., Waltham, MA, 2011 Search PubMed.
- R. B. McCleskey, D. K. Nordstrom and J. W. Ball, Metal interferences and their removal prior to the determination of As(T) and As(III) in acid mine waters by hydride generation atomic absorption spectrometry, Report U.S. Geological Survey Water-Resources Investigations, Report 2003-4117, 2003 Search PubMed.
- D. A. Roth, M. O. Johnson, B. R. McCleskey, M. L. Riskin and P. A. Bliznik, Evaluation of preservation techniques for trace metals and major cations for surface waters collected from the U.S. Geological Survey's National Water Quality Network Sites, Report U.S. Geological Survey Data Release, 2022, DOI:10.5066/P9SMPZ3M.
- U.S. Environmental Protection Agency, Method 6010D Inductively Coupled Plasma-Optical Emission Spectrometry (Revision 5), U.S. Environmental Protection Agency, Washington D.C, 2018 Search PubMed.
- T. I. Brinton, J. R. Barbarino, D. B. Peartt, H. E. Taylor and R. C. Antweiler, Concentration and Transport Data for Dissolved Inorganic Constituents in Water Collected during Seven Cruises on the Mississippi River and Some of its Tributaries, July 1987–June 1990, Report 94-524, Reston, VA, 1995 Search PubMed.
- J. D. Pfaff, Method 300.0, Determination of Inorganic Anions by Ion Chromatography. Revision 2.1, Report EPA/600/R-93/100, U.S. Environmental Protection Agency, 1993 Search PubMed.
- S. Fisher, T. Reilly, D. Jones, W. Benzel, D. Griffin, K. Loftin, L. R. Iwanowicz and J. A. Cohl, Standard Operating Procedures for Collection of Soil and Sediment Samples for the Sediment-Bound Contaminant Resiliency and Response (SCoRR) Strategy Pilot Study, Report 2015-1188B, Reston, VA, 2015 Search PubMed.
- Y. Zhang, Z. Qv, J. Wang, Y. Yang, X. Chen, J. Wang, Y. Zhang and L. Zhu, Natural biofilm as a potential integrative sample for evaluating the contamination and impacts of PFAS on aquatic ecosystems, Water Res., 2022, 215, 118233 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0043135422001968.
- V. S. Blazer, S. E. Gordon, H. L. Walsh and C. R. Smith, Perfluoroalkyl Substances in Plasma of Smallmouth Bass from the Chesapeake Bay Watershed, Int. J. Environ. Res. Publ. Health, 2021, 18, 5881 CrossRef CAS PubMed.
- N. Percie du Sert, A. Ahluwalia, S. Alam, M. T. Avey, M. Baker, W. J. Browne, A. Clark, I. C. Cuthill, U. Dirnagl, M. Emerson, P. Garner, S. T. Holgate, D. W. Howells, V. Hurst, N. A. Karp, S. E. Lazic, K. Lidster, C. J. MacCallum, M. Macleod, E. J. Pearl, O. H. Petersen, F. Rawle, P. Reynolds, K. Rooney, E. S. Sena, S. D. Silberberg, T. Steckler and H. Würbel, Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 2.0, PLoS Biol., 2020, 18, e3000411, DOI:10.1371/journal.pbio.3000411.
- U.S. Environmental Protection Agency, Method 1633 Revision A: Analysis of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous, Solid, Biosolids, and Tissue Samples by LC-MS/MS, Report EPA 820-R-24-007, U.S. Environmental Protection Agency, Washington, D.C., 2024 Search PubMed.
- K. M. Romanok, K. L. Smalling, P. M. Bradley, E. P. Barnhart, M. J. Eggers, J. T. Doyle, S. M. Meppelink and M. L. Schreiner, Concentration results for per- and polyfluoroalkyl substances (PFAS) analyzed in fish tissue, bed sediment and biofilm samples, Montana, 2023, U.S. Geological Survey Data Release, 2025, DOI:10.5066/P1DAMSMX.
- D. P. R. Herlemann, M. Labrenz, K. Jürgens, S. Bertilsson, J. J. Waniek and A. F. Andersson, Transitions in bacterial communities along the 2000 km salinity gradient of the Baltic Sea, ISME J., 2011, 5, 1571–1579, DOI:10.1038/ismej.2011.41.
- K. M. Han, M. N. Choi, J. H. Pi, S. H. Kim, Y. S. Kim and C. S. Na, Microbial Diversity Analysis of Bupleurum latissimum Nakai from Ulleung-Do of South Korea Using Next-Generation Sequencing, Microbiol. Resour. Announce., 2022, 11, e0102821 CrossRef PubMed.
- E. Bolyen, J. R. Rideout, M. R. Dillon, N. A. Bokulich, C. C. Abnet, G. A. Al-Ghalith, H. Alexander, E. J. Alm, M. Arumugam, F. Asnicar, Y. Bai, J. E. Bisanz, K. Bittinger, A. Brejnrod, C. J. Brislawn, C. T. Brown, B. J. Callahan, A. M. Caraballo-Rodríguez, J. Chase, E. K. Cope, R. Da Silva, C. Diener, P. C. Dorrestein, G. M. Douglas, D. M. Durall, C. Duvallet, C. F. Edwardson, M. Ernst, M. Estaki, J. Fouquier, J. M. Gauglitz, S. M. Gibbons, D. L. Gibson, A. Gonzalez, K. Gorlick, J. Guo, B. Hillmann, S. Holmes, H. Holste, C. Huttenhower, G. A. Huttley, S. Janssen, A. K. Jarmusch, L. Jiang, B. D. Kaehler, K. B. Kang, C. R. Keefe, P. Keim, S. T. Kelley, D. Knights, I. Koester, T. Kosciolek, J. Kreps, M. G. I. Langille, J. Lee, R. Ley, Y. X. Liu, E. Loftfield, C. Lozupone, M. Maher, C. Marotz, B. D. Martin, D. McDonald, L. J. McIver, A. V. Melnik, J. L. Metcalf, S. C. Morgan, J. T. Morton, A. T. Naimey, J. A. Navas-Molina, L. F. Nothias, S. B. Orchanian, T. Pearson, S. L. Peoples, D. Petras, M. L. Preuss, E. Pruesse, L. B. Rasmussen, A. Rivers, M. S. Robeson II, P. Rosenthal, N. Segata, M. Shaffer, A. Shiffer, R. Sinha, S. J. Song, J. R. Spear, A. D. Swafford, L. R. Thompson, P. J. Torres, P. Trinh, A. Tripathi, P. J. Turnbaugh, S. Ul-Hasan, J. J. J. van der Hooft, F. Vargas, Y. Vázquez-Baeza, E. Vogtmann, M. von Hippel, W. Walters, Y. Wan, M. Wang, J. Warren, K. C. Weber, C. H. D. Williamson, A. D. Willis, Z. Z. Xu, J. R. Zaneveld, Y. Zhang, Q. Zhu, R. Knight and J. G. Caporaso, Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2, Nat. Biotechnol., 2019, 37, 852–857 CrossRef CAS PubMed.
- B. J. Callahan, P. J. McMurdie, M. J. Rosen, A. W. Han, A. J. A. Johnson and S. P. Holmes, DADA2: High-resolution sample inference from Illumina amplicon data, Nat. Methods, 2016, 13, 581–583, DOI:10.1038/nmeth.3869.
- C. Quast, E. Pruesse, P. Yilmaz, J. Gerken, T. Schweer, P. Yarza, J. Peplies and F. O. Glöckner, The SILVA ribosomal RNA gene database project: improved data processing and web-based tools, Nucleic Acids Res., 2013, 41, D590–D596 CrossRef CAS PubMed.
- C. Childress, W. Foreman, B. Conner and T. Maloney, New reporting procedures based on long-term method detection levels and some considerations for interpretations of water-quality data provided by the U.S. Geological Survey National Water Quality Laboratory, Report U.S. Geological Survey Open-File Report 99-193, 1999 Search PubMed.
- W. T. Foreman, T. L. Williams, E. T. Furlong, D. M. Hemmerle, S. J. Stetson, V. K. Jha, M. C. Noriega, J. A. Decess, C. Reed-Parker and M. W. Sandstrom, Comparison of detection limits estimated using single- and multi-concentration spike-based and blank-based procedures, Talanta, 2021, 228, 122139 CrossRef CAS PubMed.
- D. K. Mueller, T. L. Schertz, J. D. Martin and M. W. Sandstrom, Design, analysis, and interpretation of field quality-control data for water-sampling projects, Report U.S. Geological Survey Techniques and Methods Book 4 Chapter C4, 2015 Search PubMed.
- J. Bangma, K. M. Barry, C. M. Fisher, S. Genualdi, T. C. Guillette, C. A. Huset, J. McCord, B. Ng, B. J. Place, J. L. Reiner, A. Robuck and A. E. Rodowa, PFAS ghosts: how to identify, evaluate, and exorcise new and existing analytical interference, Anal. Bioanal. Chem., 2024, 416, 1777–1785, DOI:10.1007/s00216-024-05125-y.
- J. Bangma, J. McCord, N. Giffard, K. Buckman, J. Petali, C. Chen, D. Amparo, B. Turpin, G. Morrison and M. Strynar, Analytical method interferences for perfluoropentanoic acid (PFPeA) and perfluorobutanoic acid (PFBA) in biological and environmental samples, Chemosphere, 2023, 315, 137722 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0045653522042163.
- J. T. Bangma, J. Reiner, R. C. Fry, T. Manuck, J. McCord and M. J. Strynar, Identification of an Analytical Method Interference for Perfluorobutanoic Acid in Biological Samples, Environ. Sci. Technol. Lett., 2021, 8, 1085–1090, DOI:10.1021/acs.estlett.1c00901.
- Ø. Hammer, D. A. Harper and P. D. Ryan, PAST: paleontological statistics software package for education and data analysis, Palaeontol. Electron., 2001, 4, 9 Search PubMed , http://palaeo-electronica.org/2001_1/past/issue1_01.htm.
- M. Goumenou and A. Tsatsakis, Proposing new approaches for the risk characterisation of single chemicals and chemical mixtures: The source related Hazard Quotient (HQS) and Hazard Index (HIS) and the adversity specific Hazard Index (HIA), Toxicol Rep, 2019, 6, 632–636 Search PubMed , https://www.sciencedirect.com/science/article/pii/S2214750019303117.
- U.S. Environmental Protection Agency, EPA's National-scale Air Toxics Assessment, An Overview of Methods for EPA's National-Scale Air Toxics Assessment, 2011 Search PubMed.
- B. R. Blackwell, G. T. Ankley, S. R. Corsi, L. A. De Cicco, K. A. Houck, R. S. Judson, S. Li, M. T. Martin, E. Murphy and A. Schroeder, An “EAR” on environmental surveillance and monitoring: A case study on the use of exposure-activity ratios (EARs) to prioritize sites, chemicals, and bioactivities of concern in Great Lakes waters, Environ. Sci. Technol., 2017, 51, 8713–8724 CrossRef CAS PubMed.
- P. M. Bradley, K. M. Romanok, K. L. Smalling, J. R. Masoner, D. W. Kolpin and S. E. Gordon, Predicted aquatic exposure effects from a national urban stormwater study, Environ. Sci.: Water Res. Technol., 2023, 9, 3191, 10.1039/D2EW00933A.
- N. Cedergreen, Quantifying synergy: a systematic review of mixture toxicity studies within environmental toxicology, PLoS One, 2014, 9, e96580 Search PubMed.
- U.S. Environmental Protection Agency, Office of Pesticide Programs-Aquatic Life Benchmarks and Ecological Risk Assessments for Registered Pesticides, 2025, https://www.epa.gov/pesticide-science-and-assessing-pesticide-risks/aquatic-lifebenchmarks-and-ecological-risk and-ecological-risk, accessed March 18, 2025 Search PubMed.
- Montana DEQ, DEQ-7 Montana Numeric Water Quality Standards, Montana DEQ, Water Quality Division, Water Quality Planning Bureau, Water Quality Standards and Modeling Section, Helena, MT, 2019 Search PubMed.
- U.S. Environmental Protection Agency, Office of Pesticide Programs-Aquatic Life Benchmarks and Ecological Risk Assessments for Registered Pesticides, (accessed March 18, 2025, DOI: https://www.epa.gov/pesticide-science-and-assessing-pesticide-risks/aquatic-life-benchmarks-and-ecological-risk).
- K. Makarowski, Escherichia coli (E. coli) Assessment Method for State Surface Waters, Report Document WQDWQPBWQA-01, Version 1.0, Montana Department of Environmental Quality, Helena, MT, 2020 Search PubMed.
- U.S. Environmental Protection Agency, Recommended Human Health Recreational Ambient Water Quality Criteria or Swimming Advisories for Microcystins and Cylindrospermopsin, Report EPA 822-F-19-001, Office of Water, 2019 Search PubMed.
- U.S. Environmental Protection Agency, National Primary Drinking Water Regulations, (accessed February 10, 2025, DOI: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations).
- U.S. Environmental Protection Agency, 40 C.F.R. § 141: National Primary Drinking Water Regulations, 2025, https://www.ecfr.gov/cgi-bin/retrieveECFR?gp=1%26SID=339dba02478821f8412c44292656bd34%26ty=HTML%26h=L%26mc=true%26n=pt40.25.141%26r=PART#_top Search PubMed.
- World Health Organization (WHO), Guidelines for Drinking-Water Quality, Fourth Edition Incorporating the First and Second Addenda, 2022 Search PubMed.
- U.S. Environmental Protection Agency, How EPA Regulates Drinking Water Contaminants, (accessed February 10, 2025, DOI: https://www.epa.gov/dwregdev/how-epa-regulates-drinking-water-contaminants).
- L. De Cicco, S. R. Corsi, D. Villeneuve, B. R. Blackwell and G. T. Ankley, toxEval: Exploring Biological Relevance of Environmental Chemistry Observations. R package version 1.4.0, (accessed February 10, 2025, DOI: https://github.com/DOI-USGS/toxEval).
- R Development Core Team, R: A Language and Environment for Statistical Computing, Version 4.5.0, R Foundation for Statistical Computing, Vienna, Austria, 2025 Search PubMed.
- S. M. Goodrow, B. Ruppel, R. L. Lippincott, G. B. Post and N. A. Procopio, Investigation of levels of perfluoroalkyl substances in surface water, sediment and fish tissue in New Jersey, USA, Sci. Total Environ., 2020, 729, 138839 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0048969720323561.
- K. L. Smalling, O. H. Devereux, S. E. Gordon, P. J. Phillips, V. S. Blazer, M. L. Hladik, D. W. Kolpin, M. T. Meyer, A. J. Sperry and T. Wagner, Environmental and anthropogenic drivers of contaminants in agricultural watersheds with implications for land management, Sci. Total Environ., 2021, 774, 145687 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0048969721007555.
- E. Nilsen, K. L. Smalling, L. Ahrens, M. Gros, K. S. B. Miglioranza, Y. Pico and H. L. Schoenfuss, Critical review: Grand challenges in assessing the adverse effects of contaminants of emerging concern on aquatic food webs, Environ. Toxicol. Chem., 2019, 38, 46–60 CrossRef CAS PubMed , http://pubs.er.usgs.gov/publication/70201863.
- N. Kolarova and P. Napiórkowski, Trace elements in aquatic environment. Origin, distribution, assessment and toxicity effect for the aquatic biota, Ecohydrol. Hydrobiol., 2021, 21, 655–668 Search PubMed , https://www.sciencedirect.com/science/article/pii/S164235932100015X.
- S. Langenheder and H. Ragnarsson, The role of environmental and spatial factors for the composition of aquatic bacterial communities, Ecology, 2007, 88, 2154–2161, DOI:10.1890/06-2098.1.
- K. L. Smalling, J. C. Rowe, C. A. Pearl, L. R. Iwanowicz, C. E. Givens, C. W. Anderson, B. McCreary and M. J. Adams, Monitoring wetland water quality related to livestock grazing in amphibian habitats, Environ. Monit. Assess., 2021, 193, 58, DOI:10.1007/s10661-020-08838-6.
- S. D. Richardson, M. J. Plewa, E. D. Wagner, R. Schoeny and D. M. DeMarini, Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: a review and roadmap for research, Mutat. Res., Rev. Mutat. Res., 2007, 636, 178–242 Search PubMed.
- D. Schoenen, Role of disinfection in suppressing the spread of pathogens with drinking water: possibilities and limitations, Water Res., 2002, 36, 3874–3888 CrossRef CAS PubMed.
- S. Pesce, L. Mamy, W. Sanchez, J. Artigas, A. Bérard, S. Betoulle, A. Chaumot, M.-A. Coutellec, O. Crouzet, J. Faburé, M. Hedde, C. Leboulanger, C. Margoum, F. Martin-Laurent, S. Morin, C. Mougin, D. Munaron, S. Nélieu, C. Pelosi and S. Leenhardt, The use of copper as plant protection product contributes to environmental contamination and resulting impacts on terrestrial and aquatic biodiversity and ecosystem functions, Environ. Sci. Pollut. Res., 2025, 32, 2830–2846, DOI:10.1007/s11356-024-32145-z.
- K. V. Brix, G. De Boeck, S. Baken and D. J. Fort, Adverse Outcome Pathways for Chronic Copper Toxicity to Fish and Amphibians, Environ. Toxicol. Chem., 2022, 41, 2911–2927, DOI:10.1002/etc.5483.
- L. K. Tuck, Ground-water resources along the Little Bighorn River, Crow Indian Reservation, Report U.S. Geological Survey Water-Resources Investigations, Report 2003-4052, Montana, 2003 Search PubMed.
- M. G. Evich, M. J. B. Davis, J. P. McCord, B. Acrey, J. A. Awkerman, D. R. U. Knappe, A. B. Lindstrom, T. F. Speth, C. Tebes-Stevens, M. J. Strynar, Z. Wang, E. J. Weber, W. M. Henderson and J. W. Washington, Per- and polyfluoroalkyl substances in the environment, Science, 2022, 375, eabg9065, DOI:10.1126/science.abg9065.
- A. K. Tokranov, K. M. Ransom, L. M. Bexfield, B. D. Lindsey, E. Watson, D. I. Dupuy, P. E. Stackelberg, M. S. Fram, S. A. Voss, J. A. Kingsbury, B. C. Jurgens, K. L. Smalling and P. M. Bradley, Predictions of groundwater PFAS occurrence at drinking water supply depths in the United States, Science, 2024, eado6638 Search PubMed.
- A. L. Mokale Kognou, R. A. Ngono Ngane, Z.-H. Jiang, C. C. Xu, W. Qin and H. Inui, Harnessing the power of microbial consortia for the biodegradation of per- and polyfluoroalkyl substances: Challenges and opportunities, Chemosphere, 2025, 374, 144221 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0045653525001638.
- B. Ji and Y. Zhao, Interactions between biofilms and PFASs in aquatic ecosystems: Literature exploration, Sci. Total Environ., 2024, 906, 167469 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0048969723060965.
- J. L. Sims, K. M. Stroski, S. Kim, G. Killeen, R. Ehalt, M. F. Simcik and B. W. Brooks, Global occurrence and probabilistic environmental health hazard assessment of per- and polyfluoroalkyl substances (PFASs) in groundwater and surface waters, Sci. Total Environ., 2022, 816, 151535 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0048969721066134.
- T. N. Penland, W. G. Cope, T. J. Kwak, M. J. Strynar, C. A. Grieshaber, R. J. Heise and F. W. Sessions, Trophodynamics of Per- and Polyfluoroalkyl Substances in the Food Web of a Large Atlantic Slope River, Environ. Sci. Technol., 2020, 54, 6800–6811, DOI:10.1021/acs.est.9b05007.
- F. L. Chiriac, C. Stoica, C. Iftode, F. Pirvu, V. A. Petre, I. Paun, L. F. Pascu, G. G. Vasile and M. Nita-Lazar, Bacterial Biodegradation of Perfluorooctanoic Acid (PFOA) and Perfluorosulfonic Acid (PFOS) Using Pure Pseudomonas Strains, Sustainability, 2023, 15, 14000 Search PubMed , https://www.mdpi.com/2071-1050/15/18/14000.
- Q. Niu, X. Lin, X. Zheng, Y. Wu, M. Long and Y. Chen, Aerobic or anaerobic? Microbial degradation of per- and polyfluoroalkyl substances: A review, J. Hazard. Mater., 2024, 480, 136173 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0304389424027523.
- S. Zhang, X. Lu, N. Wang and R. C. Buck, Biotransformation potential of 6:2 fluorotelomer sulfonate (6:2 FTSA) in aerobic and anaerobic sediment, Chemosphere, 2016, 154, 224–230 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0045653516303770.
- T. Yin, N. H. Tran, C. Huiting, Y. He and K. Y.-H. Gin, Biotransformation of polyfluoroalkyl substances by microbial consortia from constructed wetlands under aerobic and anoxic conditions, Chemosphere, 2019, 233, 101–109 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0045653519311488.
- N. Barbo, T. Stoiber, O. V. Naidenko and D. Q. Andrews, Locally caught freshwater fish across the United States are likely a significant source of exposure to PFOS and other perfluorinated compounds, Environ. Res., 2023, 220, 115165 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0013935122024926.
- E. Ivantsova, A. Lu and C. J. Martyniuk, Occurrence and toxicity mechanisms of perfluorobutanoic acid (PFBA) and perfluorobutane sulfonic acid (PFBS) in fish, Chemosphere, 2024, 349, 140815 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0045653523030850.
- B. Ruffle, U. Vedagiri, D. Bogdan, M. Maier, C. Schwach and C. Murphy-Hagan, Perfluoroalkyl Substances in U.S. market basket fish and shellfish, Environ. Res., 2020, 190, 109932 Search PubMed , https://www.sciencedirect.com/science/article/pii/S0013935120308276.
- A. D. Delinsky, M. J. Strynar, P. J. McCann, J. L. Varns, L. McMillan, S. F. Nakayama and A. B. Lindstrom, Geographical Distribution of Perfluorinated Compounds in Fish from Minnesota Lakes and Rivers, Environ. Sci. Technol., 2010, 44, 2549–2554, DOI:10.1021/es903777s.
- V. S. Blazer, H. L. Walsh, C. R. Smith, S. E. Gordon, B. J. Keplinger and T. A. Wertz, Tissue distribution and temporal and spatial assessment of per- and polyfluoroalkyl substances (PFAS) in smallmouth bass (Micropterus dolomieu) in the mid-Atlantic United States, Environ. Sci. Pollut. Res., 2024, 31, 59302–59319, DOI:10.1007/s11356-024-35097-6.
- M. L. Hedgespeth, D. L. Taylor, S. Balint, M. Schwartz and M. G. Cantwell, Ecological characteristics impact PFAS concentrations in a U.S. North Atlantic food web, Sci. Total Environ., 2023, 880, 163302 CrossRef CAS PubMed , https://www.sciencedirect.com/science/article/pii/S0048969723019216.
- G. T. Ankley, P. Cureton, R. A. Hoke, M. Houde, A. Kumar, J. Kurias, R. Lanno, C. McCarthy, J. Newsted, C. J. Salice, B. E. Sample, M. S. Sepúlveda, J. Steevens and S. Valsecchi, Assessing the Ecological Risks of Per- and Polyfluoroalkyl Substances: Current State-of-the Science and a Proposed Path Forward, Environ. Toxicol. Chem., 2020, 40, 564–605, DOI:10.1002/etc.4869.
- J. M. Petali, E. L. Pulster, C. McCarthy, H. M. Pickard, E. M. Sunderland, J. Bangma, C. C. Carignan, A. Robuck, K. A. Crawford, M. E. Romano, R. Lohmann and K. von Stackelburg, Considerations and challenges in support of science and communication of fish consumption advisories for per- and polyfluoroalkyl substances, Integrated Environ. Assess. Manag., 2024, 20, 1839–1858, DOI:10.1002/ieam.4947.
- Colorado Department of Public Health & Environment, Colorado's Fish Consumption Advisory Program: Technical Guidance Document, https://cdphe.colorado.gov/water-quality/clean-water/rivers-lakes-and-streams/fish-consumption-advisories-in-colorado, accessed June 4, 2025 Search PubMed.
- Z. Wang, G. W. Walker, D. C. G. Muir and K. Nagatani-Yoshida, Toward a global understanding of chemical pollution: a first comprehensive analysis of national and regional chemical inventories, Environ. Sci. Technol., 2020, 54, 2575–2584, DOI:10.1021/acs.est.9b06379.
Footnotes |
| † Current affiliation: Environmental Health Program, Land Resources and Environmental Sciences, Montana State University, Bozeman, Montana, USA. |
| ‡ Current affiliation: Human Development and Community Health, Montana State University, Bozeman, Montana, USA. |
| § Current affiliation: Health Research and Development Branch, Division of Research Capacity Building, National Institute of General Medical Sciences, National Institutes of Health, Bethesda, Maryland, USA. |
| This journal is © The Royal Society of Chemistry 2026 |