Next-generation perovskite-metal–organic framework (MOF) hybrids in photoelectrochemical water splitting: a path to green hydrogen solutions
Quan Yee
Tey
 a,
Joshua Zheyan
Soo
a,
Joshua Zheyan
Soo
 ab,
Wen Cai
Ng
ab,
Wen Cai
Ng
 a and
Meng Nan
Chong
a and
Meng Nan
Chong
 *ab
*ab
aDepartment of Chemical Engineering, School of Engineering, Monash University Malaysia, Jalan Lagoon Selatan, 47500 Bandar Sunway, Selangor, Malaysia. E-mail: chong.meng.nan@monash.edu
bCentre for Net-Zero Technology, Monash University Malaysia, Jalan Lagoon Selatan, 47500 Bandar Sunway, Selangor, Malaysia
First published on 17th February 2025
Abstract
Green hydrogen production through sustainable and clean methods, such as photoelectrochemical (PEC) water splitting, has garnered significant attention as a potential solution to the twin issues of fossil fuel depletion and excessive greenhouse gas emissions in energy production. Enabling efficient PEC reactions requires the discovery and utilisation of stable and efficient photoelectrode materials. Perovskite materials, renowned for their exceptional optoelectronic properties, have garnered attention due to their remarkable optoelectronic properties, including tuneable bandgaps, high absorption coefficients, and superior charge transport dynamics. However, their practical use is hindered by inherent stability issues and efficiency bottlenecks. To address these limitations, the integration of perovskites with secondary materials, particularly metal–organic frameworks (MOFs), has emerged as a promising strategy. MOFs, known for their high surface area, tuneable porosity, and catalytic activity, offer a unique opportunity to complement and enhance the performance of perovskites in PEC applications. This review begins by exploring the fundamental properties of perovskites and MOFs, highlighting recent advancements and key challenges in their individual and combined use as photoelectrodes for PEC water splitting. The synergistic interplay within MOF-perovskite hybrids is discussed, focusing on how this interaction improves charge separation while addressing the persistent issues of photocorrosion and operational stability. Finally, this review addresses critical issues that still limit the efficiency of MOF-perovskite photoelectrodes and offers some perspectives on potential solutions. By bridging the unique strengths of perovskites and MOFs, this work underscores the transformative potential of hybrid materials in advancing PEC water splitting toward sustainable and scalable green hydrogen production.
1 Introduction
The energy industry is at a pivotal moment, as the dominance of fossil fuels in energy production has become unsustainable due to emerging global challenges. Firstly, the pertinent issue of man-made climate change requires a drastic reduction in global greenhouse gas emissions, of which the fossil-fuel-reliant energy sector is a major contributor. At the same time, the possibility of fossil fuel depletion and its current concentration in selected geographical regions raises geopolitical concerns about its impact on global energy security. Therefore, exploring renewable and universally accessible alternatives to fossil fuels is essential to diversify the energy mix and mitigate these challenges, ensuring sustainable long-term energy production.Amid these considerations, solar energy stands out as an infinite energy resource, with a staggering 173![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 terawatts constantly reaching Earth, a magnitude capable of satisfying global energy needs over 10
000 terawatts constantly reaching Earth, a magnitude capable of satisfying global energy needs over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times.1 Nevertheless, the intermittent nature of solar energy, dictated by factors like time of day, weather, and geographic location, is a challenge that needs to be overcome to ensure a continuous on-demand supply of energy. A key solution to mitigate this inherent supply variability is converting solar energy into clean, storable fuels. In this regard, hydrogen (H2), which yields only water as a by-product of combustion, stands out as a prime choice.2 Its remarkable energy carrier attributes boast an impressive gravimetric energy density of 141.8 MJ kg−1 (higher heating value, HHV) and 120 MJ kg−1 (lower heating value, LHV) at 298 K, surpassing that of natural gas (54 MJ kg−1) and gasoline (47 MJ kg−1).3,4 As a result, a litre of H2 fuel has an energy capacity comparable to that of 0.25 litre of gasoline under ambient conditions.5 This high efficiency makes H2 a compelling fuel option. Beyond its use as a fuel, H2 is widely employed in industries like petroleum refining (hydrotreating), fertiliser production, food processing (fat and oil conversion), and drug manufacturing.6
000 times.1 Nevertheless, the intermittent nature of solar energy, dictated by factors like time of day, weather, and geographic location, is a challenge that needs to be overcome to ensure a continuous on-demand supply of energy. A key solution to mitigate this inherent supply variability is converting solar energy into clean, storable fuels. In this regard, hydrogen (H2), which yields only water as a by-product of combustion, stands out as a prime choice.2 Its remarkable energy carrier attributes boast an impressive gravimetric energy density of 141.8 MJ kg−1 (higher heating value, HHV) and 120 MJ kg−1 (lower heating value, LHV) at 298 K, surpassing that of natural gas (54 MJ kg−1) and gasoline (47 MJ kg−1).3,4 As a result, a litre of H2 fuel has an energy capacity comparable to that of 0.25 litre of gasoline under ambient conditions.5 This high efficiency makes H2 a compelling fuel option. Beyond its use as a fuel, H2 is widely employed in industries like petroleum refining (hydrotreating), fertiliser production, food processing (fat and oil conversion), and drug manufacturing.6
Currently, steam methane reforming remains the dominant method for industrial H2 production, which is a high-temperature process that relies on fossil fuels and steam (CH4 + 2H2O → 4H2 + CO2).7 However, this process undermines the low-carbon advantages of using H2 as a fuel, as it generates substantial amounts of carbon dioxide (CO2), with estimates ranging from 7.5 to 12 tons of CO2 produced per ton of H2.8,9 Harnessing renewable sources such as solar energy and converting it directly into H2, particularly through photoelectrochemical (PEC) water splitting, offers a far more sustainable pathway. This approach also addresses the intermittency predicament associated with direct solar energy use, positioning H2 as a key player in sustainable energy solutions. While photovoltaic-electrocatalysis (PV-EC) is another approach for solar-driven water splitting, the integrated design of PEC systems offers several advantages. PEC systems, which combine light harvesting and water electrolysis in a single component, have vast potential to reduce implementation costs by using fewer materials.10 Although PV-EC systems have achieved high technological readiness levels (TRL) and demonstrated higher solar-to-H2 (STH) efficiencies, their high solar cell costs remain a significant limitation.11 This makes PEC research, especially with cost-effective materials like metal oxides, a promising pathway for developing more efficient and affordable solar-driven water splitting systems.12
Over the years, a wealth of comprehensive investigations has been conducted to explore a wide range of semiconductor materials (e.g.: TiO2, Fe2O3, Cu2O, BiVO4, MoS2, and g-C3N4) for use as PEC photoelectrodes.13–17 Various iterations of these materials have been explored in these studies, focusing on tuning of properties such as crystallinity, surface behaviour, and heterojunction formation to enhance their intrinsic PEC performance. For example, the porous structure of the ZnO photoanode with oxygen vacancies was constructed which led to an increase in surface area and light harvesting, shortening the charge transport distance and promoting charge transfer and water oxidation kinetics at the electrode/electrolyte interface.18 However, the reported PEC efficiencies of these materials still fall short of meeting the key benchmarks for large-scale applications, including the 25% STH efficiency target, a 10-year operational lifetime, and H2 production costs of USD 1–10 per kg. This underscores the ongoing challenge of achieving the necessary efficiency targets to make PEC water splitting a truly transformative technology in the realm of green H2 production.
Recently, the scientific community has turned its attention to perovskite materials as a compelling alternative to traditional semiconductors, primarily due to their distinctive properties. Perovskites offer exceptional structural flexibility which provides a promising platform for the rational design and engineering of high-efficiency photoelectrodes. By precisely modulating the A and B site elements and composition, the crystal and electronic properties can be meticulously tailored to optimise photoelectrode performance. Additionally, certain perovskites exhibit ferroelectric or piezoelectric properties, which enhance charge carrier separation, reduce recombination and boost the catalytic activity of perovskite-based photoelectrodes for water splitting. Perovskites have already demonstrated remarkable light absorption efficiency in solar cells, reaching up to 26.1%, highlighting their potential as promising photoelectrode materials for PEC water splitting.19 Despite their remarkable optoelectronic characteristics, perovskites generally suffer from poor stability in water or aqueous solutions, leading to their rapid degradation upon exposure. This instability poses a significant bottleneck to their practical applications, particularly in PEC water splitting, where the photoelectrode must be immersed in an aqueous electrolyte solution. Consequently, researchers are actively exploring various strategies to improve perovskite stability and address this challenge. Among these approaches, encapsulating perovskites with materials such as polymers, metals, and TiO2 has emerged as the most promising and direct method to reinforce perovskite stability.20–23
Meanwhile, the versatile potential of metal–organic frameworks (MOFs) has garnered significant attention from researchers, particularly in applications such as gas adsorption, storage, molecular separation, and catalysis. The appeal of MOFs lies in their remarkable characteristics, including a highly porous structure, large specific surface area, and exceptional stability.24 These frameworks, comprising metal nodes intertwined with organic ligands, inherently exhibit tuneable photovoltaic properties such as bandgap, band edge positions, light absorption ability and porosity, making them easily tunable for specific applications. These advantages demonstrate MOFs to be a promising candidate for integration with perovskite-based photoelectrodes to form next-generation perovskite-MOF hybrids for PEC water splitting. In this review, we define next-generation perovskite-MOF hybrids as those that demonstrate significant advancements beyond traditional photoelectrode materials such as Cu2O, BiVO4, TiO2, and ZnO. By synergistically combining these two materials, perovskites can be protected from degradation while simultaneously elevating the efficiency of the photoelectrode. The inherent porosity and stability of MOFs create an environment that safeguards perovskite materials, thus extending their operational lifespan. Furthermore, the synergistic interaction between MOFs and perovskites holds promise for enhancing photoelectrode performance. The stability of MOFs can help mitigate the inherent instability of perovskites, while preserving their exceptional optoelectronic properties. This innovative amalgamation not only provides a solution to the challenges of perovskite stability but also paves the way for optimising the efficiency of energy conversion processes. Furthermore, next-generation perovskite-MOF hybrids often incorporate innovative design strategies, such as multifunctional components (e.g., integrated co-catalysts) and the utilisation of novel MOF structures and perovskite compositions. These materials aim to address the critical challenges faced by the current PEC water splitting technologies and pave the way for the development of highly efficient and sustainable solar-to-H2 conversion systems.
This review aims to comprehensively examine recent advancements in the strategic integration of MOFs with perovskite-based photoelectrodes for PEC water splitting applications. In this review, we systematically delve into the progress in this emerging field, shedding light on the tactical integration of MOFs and perovskites to improve the efficiency and stability of PEC water splitting processes. We first present the fundamental principles of PEC water splitting, specifically highlighting the role of photoelectrodes in this process. This is followed by a concise overview of the structure and properties of perovskite materials, setting the stage for an in-depth discussion of the recent advancements that have positioned perovskites as promising contenders for driving PEC water splitting reactions. Following this, we also address the stability challenges encountered during aqueous operations, offering insights into the ongoing efforts to overcome these obstacles. Building on this foundation, we then review recent studies of MOFs in the context of PEC water splitting, highlighting their potential as critical components in improving the efficiency of water splitting processes. The discussion culminates in a detailed exploration of the latest developments in MOF-perovskite hybrid photoelectrodes for PEC water splitting, as well as other relevant applications. Here, we emphasise the innovative and tactical design strategies aimed at enhancing stability and overall PEC and photoconversion performance. This comprehensive review provides valuable insights, charting pathways for further refinement and development of MOF-perovskite hybrids. We conclude this review by offering an outlook on the future of this material class, outlining strategies to address current challenges in PEC water splitting. By reviewing and analysing potential advancements, we envision the future trajectory of this dynamic field and highlight how these synergistic combinations could drive transformative progress in sustainable energy technologies.
2 Fundamental principles of PEC water splitting
The mechanism behind water splitting is relatively facile, and involves a redox reaction consisting of two distinct half-reactions: the reductive H2 evolution reaction (HER) transpiring at the cathode, and the oxidative oxygen evolution reaction (OER) occurring at the anode. The overall water splitting reaction, along with the individual half-reactions, is summarised in eqn (1) as follows:25| Overall 2H2O → 2H2 + O2 ΔE = 1.23 V at pH 0 | (1) |
The specific steps of both the HER and OER reactions vary depending on the type of electrolyte used in the water splitting cell. This is due to the different majority species (i.e. H+, OH−) present in the electrolyte, which participate in the water splitting reactions. Eqn (2) and (3) present the general equations for the OER and HER in acidic media, while eqn (4) and (5) present the corresponding half-reactions in basic media.26
OER (acidic medium):
| 2H2O → 4H+ + O2 + 4e− ΔE = +1.23 V at pH 0 | (2) |
HER (acidic medium):
| 4H+ + 4e− → 2H2 ΔE = 0 V at pH 0 | (3) |
OER (basic medium):
| 4OH− → O2 + 2H2O + 4e− ΔE = +0.401 V at pH 14 | (4) |
HER (basic medium):
| 4H2O + 4e− → 2H2 + 4OH− ΔE = −0.828 V at pH 14 | (5) |
Regardless of the electrolyte properties, water splitting is inherently an energy-consuming process. Under standard conditions (25 °C, 1 atm pressure, pH 0, 1 M concentration), it requires 285.8 kJ mol−1 of energy, which is divided into Gibbs free energy (237.2 kJ mol−1) and heat energy (48.6 kJ mol−1). The Gibbs free energy corresponds to 1.23 V per electron (eV), which represents the thermodynamic potential barrier needed to initiate the water splitting reaction. However, in practice, additional energy is typically required in reality to overcome kinetic losses in the HER and OER reactions at both electrodes of the PEC cell.
In a PEC process, the water splitting reaction is initiated by light illumination, either with or without the need for an external voltage bias. Hence, photoactivity is required at one or both electrodes in the electrolytic cell, which can be enabled by using photoactive semiconductors coupled with a cocatalyst. Additionally, to utilise solar energy to drive the water splitting reaction with a semiconductor photoelectrode, the bandgap of the semiconductor needs to be at least 1.23 eV. Moreover, the band positions of the semiconductor material need to align strategically with the water splitting half-reactions (HER and OER). Ideally, the Fermi level (EF – a measure of the semiconductor's electron filling) of the photoelectrode should be higher in energy than the H2 evolution potential for an n-type photoanode, where electrons are the majority carriers. On the other hand, the EF level of a p-type photocathode should have a lower energy than the O2 evolution potential, where holes are the majority carriers. Fig. 1 illustrates this process using a single photoanode and a counter cathode as an example. The PEC water splitting reaction inherently involves three fundamental stages. First, the semiconductor photoanode absorbs light with energy greater than its bandgap, leading to the generation of photoexcited charge carriers within the bulk of the photoanode. This photoexcitation creates electron–hole pairs, driving charge carrier separation: electrons (e−) are excited from the valence band (VB) to the conduction band (CB), while holes (h+) are left behind in the VB. It is worth noting that electron–hole recombination during charge separation can significantly impede efficiency. The final stage involves the participation of photogenerated charge carriers in the HER/OER reactions at the electrode surfaces. Specifically, in this photoanode-dark cathode example, photogenerated electrons travel through the circuit to the counter electrode, where they drive the cathodic HER, reducing H+ ions to produce H2. Simultaneously, holes generated at the photoanode migrate to the interface between the photoanode and electrolyte, catalysing the OER sequence at the interface and producing O2. In the process, the number of active sites and the extent of surface area play a pivotal role in facilitating efficient redox reactions. Ultimately, improving PEC performance relies on the continuous refinement and optimisation of these three fundamental processes. Enhancing charge separation efficiency and redox reaction effectiveness is crucial for advancing the overall efficiency of PEC water splitting systems.
2.1 Semiconductors as photoelectrode materials
Semiconductor photoelectrodes are crucial in PEC water splitting systems, as they generate charge carriers upon radiative illumination and ensure their separation before participating in surface HER or OER reactions. Therefore, the selection of suitable semiconductors as bulk materials for the photoelectrode is paramount for achieving high STH efficiencies in the overall PEC water splitting system.The efficiency of photoelectrodes in harnessing solar energy directly impacts the overall performance of the system. Since visible light accounts for approximately 45% of the solar spectrum, photoelectrode materials with strong visible light absorption are preferred.27 This characteristic is closely linked to the bandgap of semiconductor photoelectrodes, which determines both the range of absorbable light and the maximum achievable STH efficiency. While a narrower bandgap is generally advantageous, a practical balance must be struck between maximising light absorption and providing sufficient driving force for water splitting. While a theoretical minimum bandgap of 1.23 eV is sufficient to drive water splitting, practical considerations such as thermodynamic overpotentials due to kinetic HER/OER losses dictate a more suitable bandgap range of 2.00–2.25 eV.28,29 Additionally, the band edge positions must align with the redox potentials (HER and OER) of water for efficient charge transfer. For efficient HER at the photocathode, the CB edge must be more negative than the water reduction potential (H2/H2O), while effective OER at the photoanode requires a VB edge more positive than the water oxidation potential (O2/H2O).28,30,31
After photoexcitation, electrons and holes must remain separated enabling them to be transported to the semiconductor surface and participate in their respective redox reactions. The efficiency of charge separation is influenced by the inherent properties of the semiconductor, such as its dielectric constant, carrier mobility and concentration, as well as its diffusion length relative to its distance to the surface.28,32 These factors must be optimised to minimise recombination losses, which can hinder the carrier activity at the semiconductor surface. The recombination rate is also influenced by the type of semiconductor bandgap (i.e., direct or indirect) and the concentration of defects, which can serve as recombination sites. Efficient charge transfer across the semiconductor–electrolyte interface is crucial to prevent charge carrier depletion within the space charge region.33
To foster large-scale implementation of PEC water splitting, additional practical considerations must be factored into the selection of photoelectrode materials. Material stability is a key concern, particularly the resilience of semiconductors against corrosion when immersed in non-neutral electrolytes and exposed to radiative illumination. In many cases, (photo)corrosion reactions of certain semiconductors are more thermodynamically favourable than the HER/OER reactions themselves. Additionally, choosing non-toxic and cost-effective materials is also beneficial to support large-scale deployment.
3 Perovskite materials
Perovskites are defined as materials that inherently share the same crystalline unit cell structure as calcium titanate (CaTiO3), a discovery attributed to Lev Perovski in 1839.34 This commonality anchors perovskite materials following a structural framework represented by the chemical formula ABX3. In this representation, A and B represent cations of distinct sizes, where A (La3+, Sr2+, Ba2+) typically consists of larger ions compared to B (Bi2+, Sn2+, Ti4+). Accompanying these cations are anions (O2−, I−, Br−, Cl−) that bond with the two cations.35 As illustrated in Fig. 2a, this arrangement generates a BO6 octahedral configuration, with the B cation at the centre of the octahedron and surrounded by X anions occupying corner positions.37Fig. 2b visualises the formation of a three-dimensional extended structure, achieved by linking the corners of the octahedra. In this extended structure, the A cations occupy vacant sites between the octahedral units.38 | ||
| Fig. 2 (a) Cubic ABX3 perovskite unit cell, and (b) three-dimensional structure of perovskites formed via corner-linked octahedra. Adapted with permission from ref. 36. Copyright (2022), Elsevier. | ||
The Goldschmidt formulation provides a key criterion for determining the formation of a stable cubic perovskite structure. The stability of the ABX3 structure is predicted based on the calculation of the tolerance factor (t), as defined in eqn (6):39
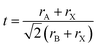 | (6) |
Perovskites, originally defined as CaTiO3, now encompass a broad class of materials that share the ABX3 crystal structure. This versatile structural framework offers significant compositional flexibility, leading to the development of diverse perovskite materials with unique physical and chemical properties. These include perovskite oxides, layered perovskites, and halide perovskites, each of which has distinct characteristics and applications.
Perovskite oxides are a subclass of perovskites in which O2− serves as the anion, with alkaline earth metals and transition metals occupying the A and B sites, respectively. Since the seminal work of Kato et al. in 2002 on their application in photocatalysis, perovskite oxides have drawn extensive interest.41 Their structural and compositional tunability makes them ideal candidates for optimizing catalytic reactions. In these materials, the CB is typically formed by the d and p orbitals of the B-site cations, while the VB consists of O 2p orbitals. A-site ions, such as alkali metals, alkaline earth metals, and certain lanthanides, generally stabilise the crystal structure without contributing to the bandgap. Advances in defect engineering and ion substitution have played pivotal roles in improving the PEC performance of perovskite oxides, particularly in water-splitting applications.42
Layered perovskites are a structural modification of perovskite oxides with the formula A2BX4, which introduces a more pliable structure while retaining excellent electronic and charge transfer properties. These properties make layered perovskites highly advantageous for photocatalytic H2 production compared to their bulk counterparts. A landmark study in 1997 by Takata et al. demonstrated the use of a hydrated layered perovskite structure for water splitting, highlighting the ability of these compounds to embed reactants within their interlayer spaces, thereby enhancing photocatalytic efficiency.43 Subsequent research has identified several subgroups of layered perovskites, including the Aurivillius phase ((Bi2O2)2+(Bn−1Mn)2−), Dion–Jacobson phase (A2BX6), and Ruddlesden–Popper (RP) phase (An+1BnO3n+1). Recent innovations, such as the fabrication of an S-scheme heterojunction, Fe2SnO4, coupled with g-C3N4 by Gupta et al., have further enhanced the catalytic performance of the material, achieving notable H2 production rates with excellent stability over multiple cycles.44
Halide perovskites have risen to prominence in the last decade due to their exceptional properties, including a tuneable direct bandgap, long carrier lifetime, high absorption coefficients, and defect tolerance. These characteristics have established halide perovskites as leading materials in solar cells, light-emitting diodes, and photodetectors. Their potential in photocatalytic and PEC applications is equally promising. Halide perovskites exhibit a relatively narrow bandgap compared to conventional photocatalysts, enabling efficient solar energy absorption at lower energies.45–47 Their conduction band position further supports overall water splitting, making them a compelling option for sustainable energy applications (Fig. 3).
 | ||
| Fig. 3 Energy band position of conventional photocatalysts and lead halide perovskites. Adapted with permission from ref. 48. Copyright (2020), American Chemical Society. | ||
The diversity and adaptability of perovskites—ranging from oxides and layered structures to halides—underscore their importance in advancing technologies across various fields, particularly in catalysis and renewable energy.
3.1 Synthesis strategy of perovskite materials
The synthesis methods of perovskites are fundamental to tailoring their structure and properties. These techniques enable precise control over crystallinity, morphology, and electronic characteristics, significantly influencing their functional performance, especially in photocatalysis and sustainable energy applications. Each method offers unique advantages and limitations, making the choice of synthesis route crucial.3.2 Recent progress of perovskite materials for water splitting
Perovskite materials have garnered significant attention as highly promising candidates for light harvesting in PEC cells and photovoltaic applications. This interest is driven by their exceptional attributes, including high charge carrier mobility, minimal defect density, extended diffusion lengths and lifetimes, and exceptional chemical and structural versatility.59–65 Impressively, solar cells based on lead halide perovskites have achieved power conversion efficiencies (PCE) of up to approximately 25%.66 Beyond their photovoltaic potential, perovskite materials have demonstrated the ability to simultaneously function as charge carrier transporters and generators – by efficiently creating electron–hole pairs under incident light irradiation. This dual functionality further bolsters their viability for PEC water splitting applications.59Fig. 3 highlights a key advantage of lead-based iodide perovskites: their narrower bandgaps (1.4–1.8 eV) compared to conventional semiconductors. This feature enhances their light absorption within the visible spectrum, resulting in a higher yield of electron–hole pairs generated from a given quantum of photons.67,68 As discussed earlier, the tunability of perovskite constituents allows for tailored properties suited to diverse applications. For instance, modifying the anion component can induce a shift in valence band energy with minimal effect on conduction band levels, enabling precise bandgap tuning of the perovskite material.69 Similarly, altering the ionic radii of cations introduces lattice distortions, as observed when methylammonium cations are replaced with slightly larger formamidinium cations, resulting in bandgap shifts.70Despite the popularity and promise of lead halide perovskites as high-performance semiconductors, their poor stability in humid environments remains one of the significant limitations for water splitting applications.71–73 This instability is due to the hygroscopic nature of amine salts, leading to the formation of perovskite hydrates.74 The decomposition of methylammonium lead iodide (CH3NH3PbI3), a widely studied perovskite material, in the presence of moisture is represented by the following equations:75
| CH3NH3PbI3 + H2O → CH3NH3PbI3·H2O | (7) |
| 4(CH3NH3PbI3) + 2H2O → (CH3NH3)4PbI6·2H2O + 3PbI2 | (8) |
Eqn (7) depicts a reversible reaction in which CH3NH3PbI3 can be reformed through a drying process after minimal exposure to moisture. However, with excessive water exposure, the perovskite undergoes structural degradation, resulting in an irreversible transformation to hydrates, as shown in eqn (8). This degradation alters its optical and electrical properties, which subsequently affects the overall efficiency of the PEC device. Moreover, Pb is both toxic and unstable at ambient temperature, prompting researchers to explore lead-free halide and oxide perovskites as alternatives, such as Sn-based, Bi-based, and Cu-based perovskites.76–79 For example, CsSnX3 (X = Cl, Br and I) were synthesized in 2016 by Chen et al. via a simple solvothermal process and demonstrated tuneable photoluminescence by substituting different halide ions of CsSnX3. Chung et al. reported that CsSnI3 exhibits strong photoluminescence properties and high electrical conductivity, likely due to defect centres in Sn rather than charge transfer between the VB and CB.80 This finding highlights the potential of Sn-based perovskites as viable alternatives to lead-based counterparts. Other promising lead-free perovskites include Cs2PtBr6, prepared by Peng and co-workers, which has a bandgap of 1.32 eV, excellent conductivity, and achieved a maximum photocurrent density of 335 μA cm−2 at −0.6 V vs. Ag/AgCl.81 This Pt-based Cs2PtBr6 nanocrystal outperformed CsPbBr3 and Cs2PdBr6-based PEC systems, as shown in Fig. 4a.81 Bi-based perovskites have also attracted much attention as viable alternatives due to their isoelectric valence shell with Pb,83 while Bi3+ ions, with an ionic radius of 1.03 Å, are analogous to those of Pb (1.19 Å) yet offer greater stability.84 Nandigana and colleagues synthesized Bi-based perovskites, specifically Cs2AgBiCl6 and Cs3Bi2Cl9, using a hydrothermal method to evaluate their properties towards water splitting.77 Their study showed that Cs2AgBiCl6 is a better photoelectrode material compared to Cs3Bi2Cl9 as the photocurrent densities of water oxidation of Cs2AgBiCl6 and Cs3Bi2Cl9 are 10 μA cm−2 and 6 μA cm−2 at 0.9 V vs. Ag/AgCl, respectively, with both materials displaying high stability over 10 h (Fig. 4b).77 This work suggests the potential of Bi-based perovskites for green H2 and O2 production through PEC water splitting. Despite this progress, their material stability remains a significant challenge for long-term PEC water splitting applications, and the power conversion efficiency of lead-free perovskites still falls short of their lead-based counterparts.
 | ||
| Fig. 4 (a) Current densities of different halide perovskite nanocrystal-based PEC systems. Adapted with permission from ref. 81. Copyright (2021), Wiley-VCH. (b) Stability studies of Cs2AgBiCl6 and Cs3Bi2Cl9 photoanodes. Adapted with permission from ref. 77. Copyright (2021), Wiley-VCH. (c) Projected density of states for Cs3Sb2I9 and Cs3Sb2I9 doped with (a) Ag, (b) In, (c) Mo, (d) Nb, (e) Sc, and (f) Bi, respectively. Adapted with permission from ref. 82. Copyright (2024), Elsevier. | ||
In order to improve the efficiency of lead-free perovskite materials, one effective strategy is doping with foreign elements, which helps to optimise band structure for water splitting and supports heterojunction formation to enhance charge separation. Another promising approach involves morphology engineering, where the nanostructure is engineered to reduce electron–hole recombination. These methods aim to refine perovskite materials by altering their elemental composition, enabling targeted adjustments to electronic band structures. Such modifications, including ion substitutions within the perovskite lattice, lead to energy band shifts that enhance responsiveness to visible light.85 In addition to compositional changes, encapsulation techniques are used to tackle the long-term stability challenges of lead-based perovskites while facilitating heterojunction formation to boost charge transfer efficiency.86–88 The following sub-sections will further elucidate these approaches.
Doping different sites within the perovskite structure with foreign elements can effectively tailor the material's energy band positions, further tuning the material's electronic properties for enhanced performance. This alteration stems from the interplay between the energy levels associated with the introduced dopant and those inherent to the host elements.82,89,90 This was demonstrated in a study performed by Exner et al. where the effects of doping on the optoelectronic properties of Cs3Sb2I9 were demonstrated using density functional theory (DFT).82 Doping Cs3Sb2I9 with In and Ag resulted in enhanced light absorption due to a reduced direct bandgap, while Mo and Nb doping increased light absorption by introducing mid-gap states, as illustrated in Fig. 4c. Consequently, doping stands as a powerful approach for tuning the band structure of perovskite materials, allowing them to achieve tailored electronic properties. Specifically, introducing dopants at the A-site impacts the bandgap by altering the distortion level of the octahedral units, with the degree of distortion influenced by the size of the introduced cation. Consequently, the modification of the A-site plays a significant role in enhancing the overall stability by tuning the tolerance factor. This effect was demonstrated in a study by Zhu et al., where Cs-doped FA0.85Cs0.15PbI3, exhibiting a smaller tolerance factor, showed improved stability compared to its FAPbI3 counterparts.91 Zou and co-workers have successfully performed water splitting with Zn-doped SrTiO3 and BaTiO3, which would otherwise be unattainable with pure SrTiO3 and BaTiO3 due to their high recombination rates and inappropriate band alignment for the reaction.92 Increasing the Zn concentration narrows the bandgap by contributing Zn 3d orbitals to the CB, which shifts the CB upwards, straddling it with the water splitting potential in BaTiO3. Additionally, the Zn doping enhances carrier mobility, which is essential to mitigate the high recombination observed in pure SrTiO3 and BaTiO3. However, no H2 production was observed under visible or UV light. In contrast, doping SrTiO3 with La and Fe at the A-site rendered it a visible light active catalyst.93,94 This is due to the interaction between the Ti 3d orbitals and Fe d orbitals, which introduces intra-bandgap states and narrows the bandgap, enabling a red-shift in the UV-vis spectra to allow visible light activity.
As for the well-known lead halide perovskites, substituting Pb2+ atoms at the B-site can potentially eliminate the material's toxicity. Moreover, phase stability can be enhanced through manipulating the B–O bond length, as substitution at the B-site influences the Goldschmidt's tolerance factor (eqn (6)), which predicts the structural stability of perovskites. Nag and colleagues doped Pb2+ (B-site) ions with metal cations such as Mn2+, Sn2+, Cd2+,Sr2+, Co2+ and Zn2+, observing an increase in phase stability as the bond length decreased in the ABX3 perovskite structure.95 However, B-site doping is generally known to be more challenging due to the higher binding energy compared to A- and X-site components.96 Furthermore, in the case of well-known lead halide perovskites, substituting Pb2+ atoms may compromise their exceptional optoelectronic characteristics. Since the metal cation in the B-site primarily influences these properties, B-site doping has become a preferred approach to achieve highly efficient perovskite solar cells. For instance, the OER performance of LaNiO3 was improved by incorporating Ni with Fe4+, which strengthened the hybridisation between Ni/Fe 3d and O 2p orbitals, thereby optimising the surface absorption energetics for reaction intermediates and improving OER activity.97 Additionally, doping the B-site of the LaFeO3 perovskite with a non-metal element such as phosphorus induced a partial valence state change in iron from Fe3+ to Fe4+. This doping modification approach optimised the bandgap electron filling and enhanced HO* intermediate adsorption for better catalytic performance.98
Lattice oxygen, present at the X-site of perovskite-type transition metal oxides, plays a crucial role in the OER through the lattice oxygen-mediated mechanism (LOM). This mechanism involves the participation of lattice oxygen and oxygen vacancies as active sites for OH* adsorption and O2 evolution. The formation of oxygen vacancies during lattice oxygen oxidation is critical for achieving high OER activity. Anion doping at the X-site of the ABX3 perovskite structure has emerged as an effective strategy to activate lattice oxygen and generate oxygen vacancies. For instance, substituting fluorine (F) for oxygen in ABX3 perovskites increases the concentration of reactive oxygen species (O22−/O−), thereby enhancing the redox activity of the BO6 octahedron and improving OER performance.99–101 Moreover, bandgap tuning, a widely used approach for optimising perovskite properties, can be achieved by varying the ratio of halogen atoms at the anion site.102 This is due to the influence of anion on the VB maximum causing the alterations in bandgap. For instance, modifying the halogen composition at the X-site of MAPbX3 perovskites from I to Br and Cl results in bandgap changes from 1.55 eV to 1.80 eV and 2.30 eV, respectively.103 Furthermore, bandgap tuning across the visible spectrum has been demonstrated by Sum et al. with MAPb(Br3−xClx) thin films.104 Notably, X-site doping has a more prominent impact on bandgap modulation compared to doping at the A- and B-sites. Oxynitrides which are known for their ability to absorb visible light for water splitting can be formed by doping the O atom with N3− to form perovskite oxynitrides such as LaTiO2N, LaTaON2, or CaNbO2N. The introduction of N creates a new VB from the N 2p orbital, which effectively reduces the bandgap. However, a narrower bandgap, which increases solar spectrum absorption, does not necessarily result in enhanced H2 evolution rates. For instance, LaNbON2 and BaNbO2N, with bandgaps of 1.6 and 1.7 eV, respectively, exhibit no water reduction and oxidation activity. This lack of redox activity is attributed to insufficient activation energy for the water splitting reaction to proceed.
B-site doping has been shown to enhance OER performance by facilitating interactions between transition metal ions and oxygen-containing adsorbates. Similarly, X-site doping, which introduces oxygen vacancies, has been explored as a strategy to improve OER activity. Moving beyond traditional single-site doping, dual-site doping has emerged as a highly promising approach for developing highly efficient perovskite oxide electrocatalysts for the OER. Using this approach, Liu et al. demonstrated significant enhancements in OER activity for perovskite oxides, including Ba0.5Sr0.5(Co0.8Fe0.2)0.9O3−δ (BSCF), SrCo0.9O3−δ (SCO) and SrNi0.9O3−δ (SNO), through the incorporation of iodine at both the B and X-sites.105 Notably, iodine dual-site doped BSCF exhibited superior OER performance with a reduced overpotential of 290 mV (compared to pristine BSCF) and a Tafel slope of 53 mV dec−1 at a current density of 10 mA cm−2.105 Fu et al. investigated dual-site doping in perovskite structures by incorporating strontium (Sr) at the A-site and iron (Fe) at the B-site of LaNiO3.97 While Fe doping alone maintained the Fe3+ oxidation state, the introduction of Sr induced a transformation to Fe4+, which was found to be critical for enhancing OER activity. This dual-site doping approach led to a 6-fold increase in current density, whereas Fe3+ alone had a negligible effect.97 Spectroscopic and theoretical studies attributed this improvement to the formation of Fe4+–O2–Ni3+ configurations, which facilitate electron transfer and accelerate the rate-determining deprotonation step of water oxidation by lowering the energy barrier.97
Heterostructures typically exhibit a band alignment that fall into one of three different configurations: type I (straddling gap), type II (staggered gap) and type III (broken gap). Most perovskite-based heterostructures are reported to exhibit type II band alignment, characterised by cascade-like transfer of photogenerated electrons and holes, which effectively addresses the challenge of charge carrier separation in perovskites.113 For instance, Jang et al. engineered a PbTiO3 perovskite@TiO2 heterojunction nanotube, with the PbTiO3 layer intricately formed along the inner wall of TiO2 nanotubes.114 This type II heterojunction facilitates efficient separation of light-induced electron–hole pairs. Beyond its role in promoting efficient charge separation, PbTiO3 functions as a photosensitiser, extending the absorption range of solar light. Consequently, this heterostructure significantly improves the PEC water splitting performance of the photoanode. To overcome the sluggish kinetics of water oxidation, Jayaraman et al. developed a dual-absorber heterojunction photoanode for PEC water oxidation, combining BiVO4 with the extremely stable Cs2PtI6 perovskite.115 This design increased the photocurrent density from 0.63 to 0.92 mA cm−2 at 1.23 V vs. RHE. The improvement is attributed to enhanced charge separation, as photogenerated holes transfer from the VB of BiVO4 to the VB of Cs2PtI6, while the panchromatic absorption of Cs2PtI6 broadens the light absorption range of the photoanode. Similarly, Kumar and Ganguli constructed a type II heterojunction using NaNbO3@CuS, as illustrated in Fig. 5a.79 The combination of n-type NaNbO3 and p-type CuS forms a p–n junction at the core–shell interface, which enhances charge separation and suppresses recombination. The successful creation of this p–n heterojunction leads to a 6-fold increase in photocurrent density, reaching 4.87 mA cm−2 at 0.45 V vs. RHE, compared to bare CuS.
 | ||
| Fig. 5 (a) Alignment of band structures of NaNbO3 and CuS. Adapted with permission from ref. 79. Copyright (2022), Elsevier; (b) Charge transfer mechanism of the LaFeO3/RGO S-scheme heterojunction. Adapted with permission from ref. 116. Copyright (2023), Elsevier; (c) FE-SEM images of (i) PMTS (nanospheres), (ii) PMTF (nanoflakes), (iii) PMTH (hierarchical flowers) and (iv) PMTT (thin microbelts) perovskites on the Ni foam respectively. Adapted with permission from ref. 117. Copyright (2017), Elsevier; (d) correlation between current densities of the Co(OH)x/BaNbO2N photoanode and BET surface areas and average particle size of the BaNbO2N powder. Adapted with permission from ref. 118. Copyright (2022), Elsevier. | ||
Recently, Ghosh and colleagues synthesized an S-scheme heterojunction of LaFeO3/reduced graphene oxide (RGO).116 Unlike the charge transfer mechanism in type II heterojunctions, S-scheme heterojunctions feature a staggered band structure similar to that of type II heterojunctions but with a distinct charge transfer pathway. As shown in Fig. 5b, it can be seen that RGO exhibits a higher CB and VB position compared to LaFeO3. When LaFeO3 and RGO come into contact, electrons from RGO spontaneously diffuse into LaFeO3, creating an electron depletion layer in RGO and an electron accumulation layer near the interface in LaFeO3. This electron transfer generates an internal electric field that accelerates the transfer of photogenerated electrons from LaFeO3 to RGO. Additionally, the contact between the materials induces a Fermi level shift, resulting in band bending. This prompts recombination of electrons from LaFeO3 and holes from RGO, driven by coulombic attraction. This selective recombination eliminates less effective charge carriers, while preserving the more potent photogenerated electrons and holes to actively participate in photocatalytic reactions.119 To address the rapid charge recombination of LaFeO3, RGO nanosheets have been employed as charge separators. Loading RGO nanosheets onto LaFeO3 resulted in an approximately 21-fold increase in current density compared to bare LaFeO3. This enhancement is attributed to the high electrical conductivity and specific surface area of RGO nanosheets, which provide efficient electron channels and facilitate charge carrier separation, hence further improving the photocatalytic performance of the composite catalyst.
In addition to modifying morphology during initial synthesis, the properties of perovskites can be altered through doping. For instance, the crystal structure of BiFeO3 can be tailored by the rational introduction of dopants such as La and Se.116 This doping induced a structural phase transformation from a rhombohedral to an orthorhombic crystal structure, corresponding to bandgap energies of 2.04 eV and 1.76 eV, respectively, thereby reducing the energy required to excite electrons under light irradiation. Moreover, the La and Se co-doping transformed the morphology of BiFeO3 from particles into nanosheets, significantly increasing the surface area available for catalytic reactions. These modifications resulted in La, Se co-doped BiFeO3 achieving superior photocatalytic activity under visible light. Therefore, the restructuring of the crystal and particle morphology of perovskites has the potential to enhance photoexcitation and improve their catalytic performance.
The crystallite size of a semiconductor plays a crucial role in determining its PEC water splitting efficiency, primarily by influencing the active surface area available for redox reactions. This relationship was exemplified in BaNbO2N, where increasing calcination temperatures resulted in larger crystallite size.118 Interestingly, higher temperatures led to decreased crystallinity due to the enlargement of oxide particles, which negatively affected the degree of nitridation and hindered the formation of porous BaNbO2N from Ba5Nb4O15. The variations in crystallite size and surface properties of BaNbO2N were found to significantly impacted PEC water splitting performance, as illustrated in Fig. 5d. Smaller oxynitride crystallites were correlated with a higher BET surface area, signifying more active sites for the water splitting reaction. Additionally, Kim and Choi highlighted a critical aspect of crystallite size in BiVO4 photoanodes. When the crystal size is smaller than the hole diffusion length, bulk carrier recombination is effectively suppressed, resulting in longer hole lifetimes and improved charge separation efficiency.120 These findings highlight the importance of optimising crystallite size to balance surface activity and charge dynamics for enhanced PEC water splitting performance.
Significant advancements have been made in enhancing the performance of perovskites through approaches such as heterojunction formation and morphology engineering, as summarised in Table 1. However, a key challenge persists: the recombination of photogenerated charge carriers, which continues to limit overall efficiency. This issue is particularly pronounced in lead-free perovskites which still fall short of their lead-based counterparts in terms of efficiency. This efficiency gap arises primarily from the inherent material properties and challenges in optimising charge carrier dynamics. Additionally, while lead-free perovskites are generally more stable than their lead-based alternatives, they remain vulnerable to degradation under certain conditions, including combined exposure to moisture, heat, and light. These factors can significantly reduce device lifetime and performance. To address these challenges, integrating hybrid materials such as MOFs with perovskites has emerged as a promising strategy. This integration can enhance perovskite crystallinity, improve charge separation, and boost PEC performance, making it a critical step toward ensuring the long-term stability and efficiency of perovskite-based PEC systems.
| Photoelectrode | Configuration/morphology | Synthesis method | Electrolyte | Photocurrent density (mA cm−2) (at 1.23 V vs. RHE) | Stability | Half-reaction | Ref. |
|---|---|---|---|---|---|---|---|
| PbTiO3@TiO2 | Type II heterojunction | Electron beam vapor deposition, anodic oxidation and spin coating | 0.1 M KOH | 0.30 | Steady-state behaviour throughout 300 s of operation | OER | 114 |
| BiVO4/Cs2PtI6 | Type II heterojunction | Spin coating, thermal oxidation and drop-casting | KOH solution (pH 13) | 0.92 | Tested for 2 h, sustained for 2 h | OER | 115 |
| Cs2PtI6 | — | Solvothermal method | KOH solution (pH 11) | 0.80 | Tested for 12 h, sustained for 12 h | OER | 121 |
| NaNbO3/CuS | Type II heterojunction | Hydrothermal method | 0.5 M Na2SO4 | 4.87 (at 0.45V vs. RHE) | Steady-state behaviour throughout 300 s of operation | HER | 79 |
| LaFeO3 | — | Hydrothermal method | 0.1 M Na2SO4 | 0.17 μA cm−2 (at 0.9 V vs. Ag/AgCl) | — | OER | 116 |
| LaFeO3/RGO | S-scheme heterojunction | 3.67 μA cm−2 (at 0.9 V vs. Ag/AgCl) | — | OER | 116 | ||
| PbTiO3 | — | In situ growth on Ni foam | 0.1 M Na2SO4 | 0.39 | Steady-state behaviour throughout 400 s of operation | OER | 117 |
| PMTT | Morphological (thin nanobelt) | 1.77 | Steady-state behaviour throughout 400 s of operation | OER | 117 | ||
| PMTS | Morphological (sphere) | 0.52 | Steady-state behaviour throughout 400 s of operation | OER | 117 | ||
| PMTF | Morphological (flake) | 0.51 | Steady-state behaviour throughout 400 s of operation | OER | 117 | ||
| PMTH | Morphological (hierarchical flower) | 1.18 | Steady-state behaviour throughout 400 s of operation | OER | 117 | ||
| BaNbO2N | — | Thermal annealing and nitridation | 0.5 M KBi | 5.2 | Tested for 1 h, 30% of photocurrent retained | OER | 122 |
| BaNbO2N | Morphological (particle size – 1.05 μm) | Polymerised complex method | 0.5 M KBi | 1.42 | — | OER | 118 |
| BaNbO2N | Morphological (particle size – 1.93 μm) | 2.20 | Tested for 1 h, 70% of photocurrent retained | OER | 118 | ||
| BaNbO2N | Morphological (particle size – 4.11 μm) | 1.17 | — | OER | 118 | ||
| BaNbO2N | Morphological (particle size – 6.92 μm) | 0.58 | — | OER | 118 |
4 MOFs
MOFs, also known as porous coordination polymers (PCPs), are constructed by linking metal nodes with organic ligands through coordination bonds, as illustrated in Fig. 6a. The diverse combinations of metal nodes and ligands give rise to remarkable structural variability, enabling their use in a wide range of applications, such as liquid and gas separation and storage, drug delivery, battery electrodes, and sensing systems.123–126 One of the main advantages of MOFs is their structural tunability, which allows flexible designs in terms of geometry and chemical functionality.127 MOFs also exhibit high porosity, resulting in a large surface area, while their tuneable pore size enhances selectivity for specific separation applications. For instance, MOF pores can be functionalised to target the removal of specific toxic gases. The pore size of MOFs can be tuned from microporous to mesoporous by manipulating the ligand lengths within the framework.128 In contrast to conventional semiconductors, MOFs offer superior electrical and optical properties owing to their photoactive organic ligands, which enhance light capture.129 Their bandgap can be readily regulated by modifying the building units, as shown in Fig. 6b.129,130 Additionally, the metal sites in MOFs were reported to act as additional active catalytic centers, which reduces charge diffusion lengths and improves catalytic performance.127,131 Certain MOFs also demonstrate exceptional stability in water and harsh chemical environments, making them recyclable for repeated use.129,130,132 When combined with perovskites, MOFs exhibit unique synergistic properties. They can effectively passivate defects on the perovskite surface or at grain boundaries, improving material stability and performance.133 The small apertures and large internal pores of MOFs allow for efficient encapsulation, preventing leaching of encapsulants while permitting reactant molecules to enter the framework, thus enhancing catalytic activity.128 | ||
| Fig. 6 (a) Structure of metal–organic frameworks (MOFs); (b) band gaps of some common MOFs; (c) schematic illustration of the ZnO@M@ZIF-67 photoanode for PEC water splitting. Adapted with permission from ref. 115. Copyright (2017), Royal Society of Chemistry. | ||
4.1 Classification of MOFs
MOFs are a versatile class of crystalline materials formed by connecting metal ions or clusters with multidentate organic linkers. Their modular structure enables the creation of a wide variety of frameworks with customizable properties. Key categories of MOFs include:Zeolitic Imidazolate Frameworks (ZIFs): ZIFs consist of tetrahedral metal ions, such as Zn2+ and Co2+, coordinated with imidazolate linkers, resulting in structures resembling zeolites. This similarity endows ZIFs with remarkable thermal and chemical stability. Their large pore sizes and robustness make them ideal for developing advanced MOF composites.
Isoreticular MOFs (IRMOFs): IRMOFs are a series of cubic MOFs featuring high porosity. They are typically constructed using zinc or other divalent metal ions linked with dicarboxylate ligands like terephthalate, making them highly adaptable for various applications.
Materials Institute Lavoisier (MIL) MOFs: the MIL series comprises MOFs based on trivalent metal ions such as Cr3+, Fe3+, or Al3+, combined with carboxylate linkers. These frameworks are known for their high stability, ultrahigh surface areas, uniform pores, and permanent porosity. Such features make them excellent candidates for biomedical and environmental applications, with a unique capacity for transfer between micropores and mesopores under external influences.
UiO Series: developed at the University of Oslo, the UiO series features MOFs based on zirconium (Zr4+) nodes linked by dicarboxylate ligands. These frameworks exhibit exceptional chemical and thermal stability. For example, UiO-66(Zr), synthesized via a solvothermal method using ZrCl4 and BDC, demonstrates excellent thermodynamic stability and can withstand highly alkaline conditions (pH 14).
Porous Coordination Networks (PCNs): PCNs are a diverse family of MOFs known for their intricate and complex structures. They exhibit high porosity and include variants like PCN-333, PCN-224, PCN-222, and PCN-57, each offering unique properties for specific applications.
This classification underscores the extensive variety of MOF architectures and compositions, enabling the design and synthesis of materials with tailored properties for targeted applications, including gas storage, drug delivery, and sensing.
4.2 Synthesis of MOFs
The synthesis of MOFs typically involves the self-assembly of metal ions or clusters with organic linkers under solvothermal or hydrothermal conditions. Key factors that influence MOF synthesis include the choice of metal ions, organic linkers, solvents, temperature, and reaction time.4.3 Application of MOFs in PEC water splitting
Recently, MOFs have garnered attention as promising photoelectrodes for PEC water splitting. Integrating MOFs with semiconductor photoanodes offers several advantages, including enhanced light utilisation, improved charge transport, and increased stability. The heterostructure formed by pairing MOFs with semiconductors significantly boosts light-harvesting efficiency. Photoactive MOFs possess broad absorption bands, allowing them to capture a wide spectrum of wavelengths effectively. Additionally, the high porosity of MOFs does not impede the light absorption path of the semiconductor, further optimising the overall water splitting efficiency.138,139 Furthermore, MOFs effectively mitigate charge carrier recombination through efficient charge transfer at the semiconductor/MOF interface. Their high porosity also provides a large number of active sites, facilitating catalytic activity and contributing to improved PEC performance.139 The stability of the device is significantly enhanced as the ordered structure of MOF could serve as a protective layer.138 For instance, a composite system was developed using visible light-active ZIF-67 to encapsulate a noble-metal sensitized semiconductor, ZnO@Au, as depicted in Fig. 6c.140 This synergistic design resulted in a remarkable improvement in photoconversion efficiency, reaching up to 0.80%, alongside an increased photocurrent density. The combination of the visible light-responsive ZIF-67 and the UV-responsive ZnO enabled efficient light utilisation across a broader spectrum. In terms of stability, the ZnO@Au@ZIF-67 composite demonstrated a consistent O2 evolution rate and stable current density over 5 cycles of operation. Structural analysis through XRD and SEM further confirmed the maintained integrity of the composite after the 5 cycles, in stark contrast to the decreased crystallinity observed in the unencapsulated ZIF-67 composite. This highlights that the encapsulation of ZnO@Au within the MOF shell not only improved its photoconversion performance but also significantly enhanced its structural integrity and durability.Ti-based MOFs, such as NH2-MIL-125, are commonly employed as photosensitisers, where their organic ligands serve as electron donors, transferring electrons to Ti-oxo clusters.138 In a study conducted by Yoon et al., a TiO2/NH2-MIL-125 heterostructure was developed, achieving a remarkable 2.7-fold increase in photocurrent density compared to bare TiO2 nanorods.141 NH2-MIL-125 was incorporated into the pores of TiO2 nanotube arrays (TNTAs) to overcome the inherent challenges of TNTAs, including their wide bandgap of 3.2 eV and low separation efficiency.142 This integration resulted in the formation of TNTAs@Ti-MOF, which demonstrated a substantial 14-fold increase in H2 generation compared to bare TNTAs. This improvement is primarily attributed to the heterostructure's enhanced light utilisation, which facilitated excellent charge separation and stability. Additionally, the study by Song et al. highlight the critical role of the MOF crystal structure in influencing H2 generation efficiency, further validating the potential of Ti-based MOFs in addressing the limitations of conventional TiO2 systems143 In their study, two distinct MOFs were synthesized (compound 1 – C42H46CuN6O21S4 and compound 2 – C21H19CuN3O9S2), both utilising the same copper (Cu) metal node and the ligand 4′-(2,4-disulfophenyl)-3,2′:6′,3′′-terpyridine (H2DSPTP).143 Despite sharing identical metal nodes and ligands, the resulting MOFs exhibited different crystal structures, leading to varied photocatalytic activities under visible and near-infrared-light. Furthermore, the π–π interactions present within the synthesized MOFs were found to facilitate the transfer of photogenerated holes, effectively reducing charge carrier recombination.143 Interestingly, this effect was more pronounced in compound 2 compared to compound 1, possibly due to shorter interatomic distances and stronger π–π interactions in compound 2.143 Consequently, when designing MOFs for PEC H2 production, it is crucial to pay careful attention to the crystal structure optimisation to maximise light absorption and charge carrier dynamics.
MOFs offer a unique advantage in designing microstructures for corresponding metal oxides, while the pyrolysis of their organic ligands induces a rich porous structure. This combination results in materials with high surface areas and abundant reaction sites, making them ideal for photoelectrode construction144–146 An increasing number of MOF-derived materials have been developed for the purpose of constructing photoelectrodes. For instance, a Co3O4 thin film derived from ZIF-67 was deposited onto a BiVO4 photoanode to address its suboptimal OER performance.147 The resulting Co3O4/BiVO4 photoanode exhibited a significant 3.2-fold improvement in PEC performance compared to pristine BiVO4, attributed to the increased surface area and additional active sites provided by the MOF-derived Co3O4. In another example, a controlled anatase-rutile TiO2 junction was created using NH2-MIL-125 as a precursor, producing a TiO2 photoanode sensitized with CdSe@CdS quantum dots (QDs).148 The TiO2-QDs displayed an octahedral morphology similar to the Ti-MOF precursor, resulting in a 47.6% increase in photocurrent density compared to anatase TiO2. Additionally, the MOF-derived TiO2 film exhibited improved electron transfer efficiency due to optimised band alignment within the photoelectrode structure. These examples underscore the role of MOFs as effective templates for controlling the morphology and performance of photoelectrode thin films, leading to significant enhancements in PEC efficiency. For a detailed overview of MOF and MOF-derived photoelectrodes and catalysts for water splitting and H2 generation, please refer to Table 2.
| Photoelectrode | MOF | Electrolyte | Synthesis method | Photocurrent density (mA cm−2) | H2 production rate (μmol h−1 g−1) | Stability | Ref. |
|---|---|---|---|---|---|---|---|
| TiO2/NH2-MIL-125 | NH2-MIL-125 | 0.5 M Na2SO4 | Solvothermal method | 0.03 at 0.20 V vs. Ag/AgCl | — | Steady-state behaviour throughout 200 s of operation | 149 |
| ZnO–C(ZIF-8) | ZIF-8 | 0.2 M Na2SO4 | Two-step calcination method | 0.08 at 1.23 V vs. RHE | — | No obvious deactivation after 5 degradation cycles | 150 |
| NH2-UiO-66 | NH2-UiO-66 | — | Solvothermal method | — | 1.72 | — | 151 |
| Pt/NH2-UiO-66 | — | — | 50.26 | 50% decay after the 1st catalytic run | |||
| Pt@NH2-UiO-66 | — | — | 257.38 | No obvious change throughout 4 catalytic runs for 10 h | |||
| ZnO@Au@ZIF-8 | ZIF-8 | 0.5 M Na2SO4 | Direct in situ growth of MOF on noble-metal-sensitized ZnO nanorods | 0.69 at 0.60 V vs. SCE | — | Steady-state behaviour throughout 3 h of operation | 140 |
| ZnO@Au@ZIF-67 | ZIF-67 | 1.93 at 0.60 V vs. SCE | — | Steady-state behaviour throughout 3 h of operation | |||
| Pd/UiO-66 | UiO-66 | 0.2 M Na2SO4 | Impregnation reduction method | 1.25 at 0.80 V vs. SCE | — | Continuous decrease in H2 production over 4 cycles for 20 h | 152 |
| MoS2 QDs/NH2-UiO-66/G | NH2-UiO-66 | 0.1 M Na2SO4 | Solvothermal method | — | 0.83 | Continuous decrease in H2 production over 4 cycles for 720 min | 153 |
| ∼30% decay in current density throughout 400 s of operation | |||||||
| Fe2O3:Ti/NH2-MIL-101(Fe) | NH2-MIL-101 | 1.0 M NaOH | Surfactant-assisted solvothermal method | 2.27 at 1.23 V vs. RHE | — | 13.3% decay in current density throughout 2500 s of operation | 154 |
| Ni(OH)2/ZIF-8/ZnO/NF | ZIF-8 | 0.1 M KOH | Seed-mediated hydrothermal growth process | 1.95 at 1.23 V vs. RHE | — | Steady state behaviour throughout 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s of operation 000 s of operation |
155 |
| ZnO/ZIF-8/67 | ZIF-8 and ZIF-67 | 0.5 M Na2SO4 | Solvothermal method | 0.11 at 1.23 V vs. RHE | — | 54% decay in current density throughout 2000 s of operation | 156 |
| Fe2O3/Co-MOF | Co-MOF | 1.0 M NaOH | Ion-exchange method | 2.0 at 1.23 V vs. RHE | — | — | 157 |
| PW12@NH2-UiO-66 | NH2-UiO-66 | 0.2 M Na2SO4 | One-step solvothermal method | — | 72.70 | No structural changes after 72 h of immersion in deionised water | 158 |
| Ni-MOF/ZnO | ZIF-8 | — | Chemical etching method | IPCE: about 11.00% at 355 nm at 0.5 V vs. Ag/AgCl | — | — | 159 |
| ZnNi MOF@ZnO | ZnNi MOF | 0.5 M Na2SO4 | Hydrothermal and ion exchange process | 1.40 at 1.23 V vs. RHE | — | Steady state behaviour throughout 100 s of operation | 160 |
| NH2-MIL-125/TiO2 | NH2-MIL-125 | 1.0 M NaOH | Hydrothermal method | 1.63 at 1.23 V vs. RHE | — | Steady state behaviour throughout 30 days of operation | 141 |
| TixFe1−xOy shell/Fe2O3 core | NH2-MIL-125 | 1.0 M NaOH | Solvothermal process, followed by a two-step calcination process | 0.72 at 1.23 V vs. RHE | — | 1.1% decay in current density throughout 5 h of operation | 161 |
| Co-MOF-BiVO4 | ZIF-67 | 0.5 M KH2PO4 | Electrodeposition and in situ growth method | 2.35 at 1.23 V vs. RHE | — | Slight decrease in current density throughout 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s of operation 000 s of operation |
147 |
| UiO-66-NH2@ Au@CdS | NH2-UiO-66 | 0.1 M Na2SO4 | Solvothermal and hydrothermal process | — | 39.50 μmol h−1 | Stable H2 generation throughout 24 h of operation and no structural changes observed after | 162 |
| TNTAs-Ti-MOF | NH2-MIL-125 | — | Hydrothermal method | — | 132.86 μmol h−1 cm−2 | Slight decline in H2 generation after 8 cycles of operation | 142 |
| Ag/NH2-MIL-125/TiO2 | NH2-MIL-125 | 0.5 M Na2SO4 | Hydrothermal and in situ reduction process | 1.06 at 1.23 V vs. RHE | — | Steady state behaviour throughout 3 h of operation | 163 |
| MOF-derived TiO2/CdSe@CdS | NH2-MIL-125 | 0.35 M Na2SO3 | MOF-template method | 10.72 at 0.9 V vs. RHE | — | 2.6% decay in current density throughout 2h of operation | 148 |
| CdSe/NH2-MIL-101(Cr) | NH2-MIL-101 | — | Hydrothermal method | — | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 664.00 μmol g−1 664.00 μmol g−1 |
7.3% decrease in H2 production after 4 cycles for 8 h | 164 |
4.4 Modification strategies of MOFs
MOFs have shown tremendous potential for PEC water splitting due to their customisable structures, extensive surface areas, and versatile functionalities. However, unlocking their full performance requires addressing key challenges and exploring strategies for enhancement. This section delves into various approaches to optimise MOFs for PEC applications, including bandgap engineering to enhance visible light absorption, the selection and optimisation of co-catalysts to boost catalytic activity, morphological engineering to improve material properties, and heterojunction formation to facilitate efficient charge transfer. | ||
| Fig. 7 (a) Schematic illustration of the HER over NH2-MIL-125 under visible light irradiation. Adapted with permission from ref. 168. Copyright (2013), Royal Society of Chemistry; (b) computational studies predicted the bandgaps of MIL-125 and its analogues containing functionalised ligands (inset: molecular structure of the terephthalate (bdc) ligand in MIL-125 with (left) monosubstituted linkers bdc-R and (right) diaminated linker bdc-(NH2)2). Adapted with permission from ref. 169. Copyright (2013), American Chemical Society; (c) molecular structure of the bdc ligand in MIL-125 and the N-substituted isoreticular MOFs and (d) its respective measured bandgap. Adapted with permission from ref. 166. Copyright (2017), Royal Society of Chemistry; (e) cycle runs for the photocatalytic H2 production of 1% MoS2/(50%) U6–CdS and pure CdS. Adapted with permission from ref. 170. Copyright (2015), Elsevier; (f) hypothetical mechanism of the NiFe-MOF/TiO2 photoanode for PEC water splitting. Adapted with permission from ref. 171. Copyright (2020), Elsevier. | ||
In addition to ligand modifications in MOFs, the light absorption edge can be extended by incorporating photosensitisers. For example, He and his group embedded CdS nanoparticles into MIL-100 via disproportionation in an aqueous solution.173 The introduction of CdS significantly enhanced light absorbance between 300 nm and 500 nm, accompanied by a noticeable red shift toward the absorption edge of bare CdS.173 Similarly, semiconductors with narrow bandgaps have been utilised as photosensitisers. For instance, Yuan and co-workers integrated In2S3, a semiconductor with a narrow bandgap, into MIL-125 as a core–shell composite, extending the light absorption edge to approximately 600 nm.174 Dye sensitisation has also gained traction since the first study of dye-sensitised TiO2 films in 1991, which demonstrated improved light-to-electric energy conversion efficiency.175 Strong van der Waals interactions and π–π stacking are expected to form between MOFs and dyes, as both contain benzene rings.176 These interactions are critical for facilitating efficient charge transfer from the dye to the MOF in photocatalytic applications. By constructing dye-sensitised MOF systems, researchers have successfully extended the optical response of MOFs into the visible-light region. For instance, He et al. sensitized Pt-loaded UiO-66 with Rhodamine B (RhB) dye, extending the absorption range to approximately 600 nm and significantly enhancing absorption intensity in the visible-light region.176 Upon absorbing light energy, RhB transforms into its excited state (RhB*), transferring photogenerated electrons to the VB of UiO-66, which then participates in the HER.176 However, RhB suffers from poor photostability, limiting its suitability as a long-term photosensitiser.177 To address this issue, Yuan and his group employed Erythrosin B (ErB) dye as a stabilising and cost-effective light-absorbing antenna.178 The ErB-sensitised Pt-UiO-66 system exhibited an extended optical response into the visible-light region, reaching approximately 549 nm due to the narrow bandgap of ErB dye.178 These studies highlight the potential of dye-sensitised MOF systems for H2 production, particularly given the cost-effectiveness and simplicity of dye sensitization. However, the stability of dyes remains a critical factor for consideration, as their degradation could lead to environmental pollution, emphasizing the quest for more robust and sustainable photosensitisers.
While MOFs can prevent nanoparticle aggregation, hydrothermal and solvothermal methods may sometimes impair photocatalytic activity. To overcome this limitation, encapsulating metal nanoparticles within MOFs using a core–shell structure has been explored. Han et al. developed a Pt/Au@Pd@MOF-74 composite, where Au nanoparticles acted as the core for Pd shells (Au@Pd), which were subsequently encapsulated in MOF-74 for applications in CO2 photoreduction and the reverse water gas shift reaction.186 In this system, the low concentration of Au@Pd nanoparticles in Au@Pd@MOF-74 limits photon absorption at the Au@Pd active sites.186 However, introducing Pt nanoparticles onto the surface reduces the composite's dependence on MOF-74 as an electron transfer medium. Furthermore, the higher amount of Pt nanoparticles compared to Au@Pd nanoparticles facilitates more efficient electron capture.186 As a result, Pt/MOF-74 demonstrates the ability to convert CO2 to CH4, whereas Au@Pd@MOF-74 primarily produces CO.186 The synergistic effect of combining Au, Pd, and Pt to form Pt/Au@Pd@MOF-74 enhances photon adsorption and improves CH4 selectivity, showcasing the potential of multi-metallic systems for advanced photocatalytic applications.186
The use of noble metals as co-catalysts in PEC systems poses challenges due to their high costs, limiting their large-scale applications. MoS2, with its layered hexagonal structure, has emerged as a promising, cost-effective alternative for the HER because of its high abundance compared to Pt. The S atoms at the Mo-edge sites can absorb H2, with each alternate S atom binding to a single H atom.187 Additionally, MoS2 has been shown to serve as active sites for H2 evolution, with the edges of the nanosized MoS2 promoting proton reduction.188 Shen and colleagues demonstrated that depositing MoS2 onto UiO-66/CdS photocatalysts resulted in a remarkable H2 production rate of 650 μmol h−1, which is 60 times higher than that of pure CdS.170 Remarkably, the MoS2/UiO-66/CdS composite also outperformed the Pt/UiO-66/CdS system under similar conditions (Fig. 7e) in terms of the H2 evolution rate.170 The strong interfacial contact between CdS, UiO-66, and MoS2 facilitated efficient electron transfer, enhancing proton reduction at the MoS2 nanosheet edges and significantly improving photocatalytic stability and performance.
Additionally, the IPCE (incident photon-to-current efficiency) at 390 nm increased to 42.10%, with charge injection and separation efficiencies reaching 92.8% and 37.4%, respectively.171 These enhancements are attributed to the NiFe-MOF structure, which shortens the photogenerated hole diffusion length and introduces additional active sites.199 Theoretical analysis indicates that the CB of NiFe-MOF is more negative than that of TiO2, allowing electron transfer from the CB of NiFe-MOF to TiO2.171 Simultaneously, holes in the VB of TiO2 migrate to the VB of NiFe-MOF, promoting efficient charge separation. As shown in Fig. 7f, Ni ions in NiFe-MOF are oxidised to Ni3+ and N4+via hole capture. The high-valence Ni species oxidise OH−/H2O into O2, while Ni4+ is reduced to Ni2+, preventing charge carrier recombination on the photoanode surface.171 Similarly, Li et al. developed a Z-scheme composite of Fe-MOF-derived Fe2O3 coupled with Ag–ZnO, which exhibited superior photocatalytic performance compared to its single-component counterparts.200 This improvement is attributed to the porous structure of Fe-MOF, which provides a large surface area and abundant active sites, as well as the Z-scheme structure that facilitates efficient separation of photogenerated electron–hole pairs.
A common limitation of most MOFs is their weak absorption of visible light, which reduces their photocatalytic efficiency. Integrating visible-light-responsive semiconductors with MOFs can effectively address this challenge. For example, g-C3N4, a visible-light-active semiconductor, is often used to modify MOFs to enhance both light absorption and charge separation efficiency. Zhao et al. developed a method for synthesizing dimension-matched heterojunctions containing MOFs, achieving excellent charge separation for efficient photocatalysis.201 The strong photoactivity of the Ni(II) MOF/g-C3N4 composite is due to the excellent charge transport properties of ultrathin g-C3N4, enhanced charge separation driven by the transfer of excited high-level electrons from g-C3N4 to Ni-MOF, and the catalytic functionality of the central Ni ion, which promotes CO2 activation.201 Doping g-C3N4/MOF composites with Na further improves their performance by introducing tunable light-harvesting capabilities and optimised band structures.202 For example, in Na-doped g-C3N4/Pt@UiO-66, Na doping enhances photocatalytic performance by creating electron transfer sites based on electron traps and a thermodynamic driving force. Moreover, MOFs have been modified with 2D carbon nitride nanosheets (CNNSs), which exhibit good conductivity, enabling rapid photocarrier transfer. Ye et al. constructed a UiO-66/CNNSs heterostructure photocatalyst using electrostatic self-assembly, leveraging the large surface area and strong CO2 capture ability of UiO-66.203 This heterostructure demonstrated significantly higher CO2 reduction activity compared to pristine CNNS.203 Safarifard and colleagues employed a facile in situ ultrasound synthesis to create a TMU-49/CNNS composite, which exhibited remarkable photocatalytic activity under visible light.204 The composite's lamellar morphology enhanced catalytic behaviour by providing greater access to internal active sites, as demonstrated in benzyl alcohol oxidation via a Z-scheme mechanism.204
Although MOFs hold great promise for PEC water splitting, their efficiency is often constrained by poor charge transport and separation.130,205 Furthermore, many MOFs suffer from limited stability in aqueous solutions, leading to component leaching and rendering them unsuitable for PEC applications. However, MOFs such as UiO-66, UiO-67, MIL-101, MIL-53, MIL-125, ZIF-8, and SIM-1 have demonstrated operational stability for up to 12 h.206,207 This stability is attributed to the high-valent metal ions (Zr4+, Cr3+, Ti4+, Fe3+) in these MOFs, which provide high charge density and form strong coordination bonds, contributing to their structural integrity and stability.208 To overcome performance limitations, researchers have employed strategies such as tailoring bandgaps, incorporating co-catalysts, optimising morphology, and forming heterojunctions. These approaches have significantly improved the light absorption, catalytic activity, and charge transfer properties of MOFs. For example, integrating MOFs as additives or interlayers in photoelectrodes has been shown to enhance charge transport and electrical conductivity.157,209–212 These advancements in MOF modifications highlight the potential of MOF-perovskite composites as next-generation photoelectrodes for solar-driven water splitting, which will be reviewed and discussed further in the next section.
5 MOF-perovskite hybrids
Perovskites have garnered significant attention in recent years as a cost-effective and efficient alternative to silicon in solar cells. However, their application in PEC water splitting remains relatively underexplored. On the other hand, MOFs are widely utilised across various fields, including gas storage, catalysis and drug delivery, due to their tuneable porosity, high surface area and structural versatility.123–126 These unique properties make MOFs highly promising for PEC water splitting, as they can enhance light absorption, facilitate charge transfer, and provide abundant active sites for efficient H2 production, particularly when integrated with perovskites.213 A significant milestone in MOF-perovskite integration was achieved in 2014 by Vinogradov et al., who synthesized a perovskite/TiO2-MIL-125 heterojunction for a perovskite solar cell (PSC).214 This development opened new avenues for leveraging MOFs in perovskite-based systems. This section will examine the advantages of integrating MOFs with perovskites and their current applications in PEC water splitting. Additionally, related applications such as PSC devices will be discussed, with an emphasis on key properties that are beneficial for PEC water splitting. Various architectures for integrating MOFs into perovskite-based PEC cells will also be explored, highlighting their potential to advance solar-driven water splitting technologies.While significant progress has been made in recent years, it is crucial to acknowledge that the literature on MOF-perovskite composites for PEC water splitting remains relatively limited. This presents a significant opportunity for further exploration and innovation in this field, particularly so given the known symbiotic benefits of this hybrid composite that are discussed in this section.
5.1 Key properties of MOF-perovskite hybrids
MOF-perovskite hybrids offer a unique combination of complementary properties that synergistically enhance their performance in PEC water splitting. These hybrids demonstrate notable improvements in light absorption and charge carrier transport, leading to higher catalytic activity.128,215 The interface between MOFs and perovskites plays a crucial role by facilitating efficient charge transfer and minimising recombination losses.216–218 The structural and functional versatility of MOFs allows for precise tailoring of their properties, enabling the design of hybrids optimised for PEC performance. Moreover, MOFs can act as protective layers around perovskites, significantly enhancing their stability and durability under operational conditions.216,219 This protective function can also address environmental concerns associated with lead-based perovskites by encapsulating them in non-toxic, eco-friendly MOF matrices.128 These unique attributes position MOF-perovskite hybrids as highly promising candidates for the development of advanced and next-generation photoelectrodes in solar-driven water splitting systems.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O active groups in MOF@COFs chemically fix uncoordinated Pb2+ ions in situ, thereby enhancing the internal stability of the perovskite film by modifying its local chemical environment.222 Furthermore, even if perovskite degradation occurs, the biomimetic nanoparticle structure of MOF@COF functions as an adsorbent, trapping Pb2+ ions via in situ chemical adsorption, similar to a spider web.222 This synergistic effect of chemical fixation and adsorption not only mitigates lead leakage but also underscores the potential of MOF@COF hybrids to optimise the electronic structure of perovskites and improve stability, as elucidated through DFT modelling and experimental approaches. MOFs have been utilised to form heterojunctions with perovskites in PSCs and as scaffolds for the growth of perovskite layers. In 2014, Vinogradov et al. first demonstrated that incorporating MOFs into PSCs improved the crystallinity of perovskite films.214 The primary motivation for integrating MOFs into PSCs is to produce high-quality perovskite films and enhance the overall stability of PSCs. For instance, Chang et al. incorporated a Zr-based porphyrin MOF (MOF-525) as an additive in the perovskite precursor solution to fabricate a MOF-525/CH3NH3PbI3-xClx heterojunction, resulting in a perovskite material with enhanced crystallinity.223 Since the crystallisation of perovskite begins at the top of the MOF/perovskite thin film, the ordered scaffold of the microporous MOF provides a regular arrangement of perovskite crystallites during the initial crystallisation stage. However, the device's stability against moisture did not improve due to the use of Spiro-OMeTAD as the hole transport layer. Further discussion of this hole transport layer and potential MOF-based modifications is provided in Section 5.3.1.2. Nevertheless, an average PCE of 12% was achieved with the MOF-perovskite hybrid layer, highlighting its potential for performance enhancement. Chang et al. further suggested that the pore size of MOFs plays a critical role in improving device performance.223 Incorporating UiO-67 into the perovskite layer resulted in improved performance compared to UiO-66, primarily due to the large pore size of UiO-67, which facilitated more efficient diffusion of the perovskite precursor solution. A similar study by Lee et al. involved blending a Zr-based MOF into the perovskite layer of a PSC.133 This integration created a “grain-locking” effect, where Zr atoms were distributed along the grain boundaries of the perovskite in the hybrid film, reinforcing the robustness of the perovskite layer.133 PL spectra confirmed the impact of MOFs on the perovskite layer (Fig. 8a), with hybrid films exhibiting higher PL intensity than pristine perovskite films.133 This increase indicates defect passivation induced by the hybrid MOFs.133 Moreover, the coordination between the lone pairs of oxygen in the MOF and Pb2+ ions in the perovskite provided additional protection against defects and moisture, enhancing the stability of the hybrid film. Both MOF-perovskite hybrids delivered higher PCE values compared to using singular UiO-66 and MOF-808 as interlayers, thereby underscoring their significant potential in PEC water splitting applications.
O active groups in MOF@COFs chemically fix uncoordinated Pb2+ ions in situ, thereby enhancing the internal stability of the perovskite film by modifying its local chemical environment.222 Furthermore, even if perovskite degradation occurs, the biomimetic nanoparticle structure of MOF@COF functions as an adsorbent, trapping Pb2+ ions via in situ chemical adsorption, similar to a spider web.222 This synergistic effect of chemical fixation and adsorption not only mitigates lead leakage but also underscores the potential of MOF@COF hybrids to optimise the electronic structure of perovskites and improve stability, as elucidated through DFT modelling and experimental approaches. MOFs have been utilised to form heterojunctions with perovskites in PSCs and as scaffolds for the growth of perovskite layers. In 2014, Vinogradov et al. first demonstrated that incorporating MOFs into PSCs improved the crystallinity of perovskite films.214 The primary motivation for integrating MOFs into PSCs is to produce high-quality perovskite films and enhance the overall stability of PSCs. For instance, Chang et al. incorporated a Zr-based porphyrin MOF (MOF-525) as an additive in the perovskite precursor solution to fabricate a MOF-525/CH3NH3PbI3-xClx heterojunction, resulting in a perovskite material with enhanced crystallinity.223 Since the crystallisation of perovskite begins at the top of the MOF/perovskite thin film, the ordered scaffold of the microporous MOF provides a regular arrangement of perovskite crystallites during the initial crystallisation stage. However, the device's stability against moisture did not improve due to the use of Spiro-OMeTAD as the hole transport layer. Further discussion of this hole transport layer and potential MOF-based modifications is provided in Section 5.3.1.2. Nevertheless, an average PCE of 12% was achieved with the MOF-perovskite hybrid layer, highlighting its potential for performance enhancement. Chang et al. further suggested that the pore size of MOFs plays a critical role in improving device performance.223 Incorporating UiO-67 into the perovskite layer resulted in improved performance compared to UiO-66, primarily due to the large pore size of UiO-67, which facilitated more efficient diffusion of the perovskite precursor solution. A similar study by Lee et al. involved blending a Zr-based MOF into the perovskite layer of a PSC.133 This integration created a “grain-locking” effect, where Zr atoms were distributed along the grain boundaries of the perovskite in the hybrid film, reinforcing the robustness of the perovskite layer.133 PL spectra confirmed the impact of MOFs on the perovskite layer (Fig. 8a), with hybrid films exhibiting higher PL intensity than pristine perovskite films.133 This increase indicates defect passivation induced by the hybrid MOFs.133 Moreover, the coordination between the lone pairs of oxygen in the MOF and Pb2+ ions in the perovskite provided additional protection against defects and moisture, enhancing the stability of the hybrid film. Both MOF-perovskite hybrids delivered higher PCE values compared to using singular UiO-66 and MOF-808 as interlayers, thereby underscoring their significant potential in PEC water splitting applications.
 | ||
| Fig. 8 (a) SEM-EDS images (red is I and green is Zr) and the PL spectra of MOF-808 and UiO-66 hybrid perovskite film. Adapted with permission from ref. 133. Copyright (2019), Wiley-VCH; (b) possible photocatalytic mechanism of the LaFeO3/MIL-125-NH2 composite. Adapted with permission from ref. 224. Copyright (2023), with permission from Elsevier; (c) schematic illustration of Co3O4/NaTaO3 composite photocatalysts with H2 production under simulated sunlight irradiation and production rates of H2 of pure NaTaO3 and Co3O4/NaTaO3 composite photocatalysts under simulated sunlight. Adapted with permission from ref. 225. Copyright (2019), Elsevier; (d) the charge density of Cs3Cu2I5@ZIF-9-III with an isosurface value of 0.25 e Å−3. Adapted with permission from ref. 226. Copyright (2024), Elsevier; (e) diagram of the light-harvesting and carrier separation mechanism in the MOF-derived TiO2(N–C)/CPB/TiO2 NR photoanode. Adapted with permission from ref. 227. Copyright (2021), Elsevier; (f) schematic synthesis process of CPB@Co3O4/N–C. Adapted with permission from ref. 228. Copyright (2020), Elsevier. | ||
Beyond the direct utilisation of MOFs, researchers have developed heterostructures combining MOF-derived metal oxides with other semiconductors to enhance water splitting activity. Metal oxides derived from MOFs are particularly advantageous because they retain the porous structure and large specific surface area of the parent MOF. For example, Xu et al. constructed Co3O4 from Co-MOF through thermal annealing and attached it to the surface of NaTaO3 through electrostatic attraction.225 The Co3O4 nanoparticles exhibited light absorption across a broad range of the light spectrum ranging from 200 to 700 nm, whereas NaTaO3 alone showed no visible light activity. As a p-type semiconductor, the MOF-derived Co3O4 formed a p–n junction with the n-type NaTaO3, resulting in effective carrier separation and an 8.2-fold enhancement in photocatalytic activity compared to bare NaTaO3 (Fig. 8c).225 Similarly, Zhu and colleagues developed a BiFeO3@Bi5O7I composite with an n–n heterojunction derived from a MOF for tetracycline degradation.233 The MOF derived composite exhibited a higher specific surface area than pure BiFeO3 and Bi5O7I, providing more catalytic centres on the surface for more efficient degradation. Moreover, the n–n junction between BiFeO3 and Bi5O7I improved charge transfer kinetics and reduced charge carriers' recombination through the internal electric field generated between the two compounds.
To further enhance our comprehension of the interaction between perovskites and the MOF hybrid, Sharma et al. performed DFT calculation on Cs3Cu2I5@ZIF-9-III hybrids and water concerning the pathway for the HER.226 The calculations revealed that water molecules preferentially bind to Cs sites on the Cs3Cu2I5 surface, with adsorption energies indicating favourable interactions. Under electron-rich conditions, the dissociation of water becomes energetically feasible, facilitating the HER.226 This process involves electron trapping at Cu sites, leading to structural rearrangements that promote water dissociation and H2 generation. Fig. 8d provides evidence of type II heterojunction formation between Cs3Cu2I5 and ZIF-9-III. Charge density analysis reveals electron accumulation near the phenyl rings of ZIF-9-III, while a concomitant decrease in electron density is observed around the iodine atoms of Cs3Cu2I5. This suggests potential electron transfer from the conduction band of the perovskite to the ZIF-9-III, facilitated by a weak interfacial interaction. This charge transfer mechanism is consistent with the formation of a type II heterojunction, effectively separating photogenerated electron–hole pairs and enhancing charge transport efficiency.226 Electrons excited under ultraviolet light transfer from the conduction band (CB) of Cs3Cu2I5 to ZIF-9-III, while holes move in the reverse direction, enabling photocatalytic reactions.226 These findings underscore the synergistic interactions within Cs3Cu2I5/ZIF-9-III composites, offering critical insights into their potential for sustainable energy applications.
5.2 Recent advancement of MOF-perovskite composites in PEC water splitting
Perovskites have shown tremendous potential in PEC water splitting, and their integration with MOFs represents a promising strategy to further enhance performance. However, the application of MOF-perovskite composites in PEC water splitting remains relatively underexplored, presenting significant opportunities for further research and innovation. Leveraging the broad light absorption range of inorganic perovskite materials, Tang et al. improved the performance of TiO2 through interface engineering (Fig. 8e).227 Specifically, they developed a MOF-derived TiO2/CsPbBr3 (CPB)/TiO2 composite, where the optical response range of the MOF-derived TiO2 (N–C)/CPB/TiO2 photoanode was extended from approximately 400 nm to 530 nm.227 This extension in light absorption, combined with enhanced charge separation efficiency, led to a substantial increase in photocurrent density, reaching 4.5 mA cm−2 at 1.2 V vs. RHE.227 The bonding interaction between CPB and the MOF-derived N–C framework further enhanced system stability by anchoring CPB nanoparticles to the TiO2 structure.227 In addition to coating perovskites with MOF-derived semiconductors to improve stability, Tang and colleagues utilised MOF-derived Co3O4 to encapsulate CPB (Fig. 8f), achieving a significantly boosted photocurrent density of 2.50 mA cm−2 and carrier migration of 41.40%—an improvement of 9.61 and 12.24 times, respectively, compared to pristine CPB.228 The exceptional PEC performance and stability of the CPB@Co3O4/N–C can be attributed to several factors: (1) the formation of N–Br halogen bonds at the interface between the Co3O4/N–C framework and CPB chemically immobilises the CPB, preventing its decomposition and significantly improving its stability in an aqueous environment. (2) The unique type-II heterojunction at the CPB/Co3O4/N–C interface enhances carrier separation performance. (3) The MOF-derived Co3O4/N–C serves as a co-catalyst, significantly improving the surface water oxidation kinetics of pristine CPB.228 These combined effects collectively contribute to the superior PEC performance and stability of the CPB@Co3O4/N–C system. Sharma et al. recently developed a novel Cu-based MOF integrated with Ba0.85Ca0.15Zr0.1Ti0.9O3 (BCZT) for PEC applications, demonstrating significant improvements in performance.234 Specifically, the photocurrent density of the Cu-MOF/BCZT composite increased fourfold, from 20 μA cm−2 to 80 μA cm−2, accompanied by a reduction in the bandgap from 2.8 eV to 2.2 eV.234 This enhancement in photoelectrochemical properties can be attributed to several factors: increased surface charge availability, faster separation and higher transfer rates of photogenerated charges, and improved optical absorption in the visible region. These improvements arise from the highly conductive Cu-MOF at the interface, which plays a crucial role in boosting overall PEC performance.234 In a recent study, Li and colleagues developed a plasmonic perovskite oxide-based Z-scheme catalyst, TiO2@NH2-MIL-125@reduced-SrTiO3 (R-STO), through in situ derivation of NH2-MIL-125 followed by oxygen-vacancy engineering.235 When applied as a photoanode for PEC water splitting, this catalyst achieved a remarkable photocurrent density of 4.4 mA cm−2 at 1.23 V vs. RHE—three times higher than that of pure TiO2, and achieved a H2 generation rate of 58.5 μmol h−1 cm−2.235 These enhancements are attributed to the synergistic effects of the MOF-based Z-scheme architecture and the surface plasmon resonance (SPR) properties of the non-noble R-STO.235To summarise, Tang et al. achieved a high photocurrent density of 2.5 mA cm−2 by leveraging a type-II heterojunction for efficient charge separation and utilising halogen bonding for stability in CPB@Co3O4/N–C.228 Sharma et al. demonstrated a fourfold increase in photocurrent density by integrating a highly conductive Cu-MOF with BCZT, thereby enhancing optical absorption and charge transfer.234 Li et al. developed a Z-scheme catalyst with oxygen-vacancy engineering and utilised the SPR properties of R-STO, achieving a remarkable photocurrent density of 4.4 mA cm−2 and a high H2 evolution rate.235 These studies collectively showcase the versatility of MOF-perovskite hybrids, with each approach offering unique advantages depending on the specific design and optimisation strategies employed.
Table 3 below summarises the MOF-perovskite composites for PEC water splitting.
| Composite | Role/application | Synthesis | Performance | Stability | Ref. |
|---|---|---|---|---|---|
| [MAPbBr3@MA-M(HCOO)3] (M = Mn and Co) | OER | Single-step, solid-state mechanochemical grinding process | N/A | The MOF template also imparts extra stability to the MAPbBr3 which is evident in the superior solvent stability over 15 days | 236 |
| No structural changes after 10 days of immersion in methanol | |||||
| MOF-derived TiO2(N–C)/CsPbBr3/TiO2 | OER + glycerol oxidation | Solvothermal method | 4.50 mA cm−2 photocurrent density at 1.2 V vs. RHE | Stable photocurrent density throughout 0.5 h of operation | 227 |
| CsPbBr3@MOF-derived Co3O4/N-doped C | OER | Co-precipitation-calcination method | 1.36 mA cm−2 photocurrent density at 1.23 V vs. RHE | Steady-state behaviour throughout 24 h of operation | 228 |
| TiO2@NH2-MIL-125@R-STO | OER | Solvothermal method | 4.4 mA cm−2 photocurrent density at 1.23 eV vs. RHE + H2 generation rate of 58.5 μmol h−1 cm−2 | Steady-state behaviour throughout 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s of operation and no structural changes observed after 5 h of operation 000 s of operation and no structural changes observed after 5 h of operation |
235 |
| Pt/Cs2Bi2I9@NH2-UiO-66 | HER | Ligand-assisted reprecipitation (LARP) method | H2 production of 141.87 μmol g−1 h−1 | No structural changes observed after 5 h of operation | 237 |
| La0.6Sr0.4Co0.8Fe0.2O3@Ni3(HITP)2 | OER | Electrospinning method | Low overpotential of 272 mV achieved at 10 mA cm−2 | Slight decay of 2.9% observed after 12 h of operation | 238 |
| Cu-MOF/Ba0.85Ca0.15Zr0.1Ti0.9O3 | OER | Solvothermal method | 80 μA cm−2 photocurrent density at 1 V vs. Ag/AgCl with an IPCE of ∼7.5% | N/A | 234 |
5.3 Architecture of perovskite-based cells with MOFs
While MOFs have shown significant advantages when combined with perovskites, the precise methods of integrating them into MOF-perovskite hybrids remain a topic of particular interest. Determining and optimising the architecture of this offers several potential benefits, including targeted modification of specific cell components for improved properties and performance, as well as addressing other pertinent issues relating to reducing manufacturing complexity. This subsection will explore various reported architecture designs in which MOFs have been incorporated into perovskite-based structures.5.3.1.1 MOFs in/as the electron transport layer. As a result, interface engineering, which focuses on optimising the properties of these interfaces, is essential for improving charge carrier extraction and transport. The main functions of interface engineering include: (a) ensuring optimal energy band alignment and charge transport, (b) passivating defects and minimising ion migration, and (c) enhancing device stability by forming protective layers. Thus, MOFs or MOF-derived materials can be strategically introduced into the perovskite-based photoelectrode forming a layered structure akin to stacking two sheets of paper to improve the functional layers, as illustrated in Fig. 9a. In this arrangement, the MOF and perovskite layers are physically distinct, with one layer positioned on top of the other. While in close contact, they maintain their individual structural integrity, forming a heterojunction. This layered structure enables interactions at the interface between the materials, which can facilitate improved charge transport and enhance the overall performance of the system.
 | ||
| Fig. 9 (a) Diagram of the possible locations of MOFs in perovskite-based cells; (b) schematic diagram of the Co-doped TiO2 ETL in PSCs and the Nyquist plot characteristic curves of films based on TiO2 and 1 wt% Co-doped TiO. Adapted with permission from ref. 239. Copyright (2020), American Chemical Society; (c) Schematic diagrams of the formation of MOF-derived ZnO polyhedra and their use as ETLs to enhance light harvesting and electron extraction. Adapted with permission from ref. 240. Copyright (2020), Elsevier; (d) comparison of the SEM images of the MAPbI3-based layer formed on the surface of mp-TiO2 with different ZIF-8 coating durations: (i) 0, (ii) 10, (iii) 20, and (iv) 40 min. Adapted with permission from ref. 241. Copyright (2018), Royal Society of Chemistry; (e) UV stability of perovskite film under continuous 365 nm UV illumination with the total dosage of 100 mW cm−2 for 120 min. Adapted with permission from ref. 242. Copyright (2024), Wiley. | ||
The primary role of the ETL is to transport electron charge carriers efficiently, making it essential for the material used to construct the ETL to possess high carrier mobility, a large surface area, and minimal defects.24 Non-toxic TiO2, known for its fast charge injection rate and excellent thermal stability, is one of the most commonly used ETL materials.243,244 Non-toxic TiO2, known for its fast charge injection rate and excellent thermal stability, is one of the most commonly used ETL materials.243,244 However, TiO2 suffers from low electron mobility, which increases the likelihood of charge carrier recombination. Similarly, ZnO, which shares comparable properties with TiO2, is another widely used ETL material. While ZnO offers higher charge mobility than TiO2, its chemical instability and poor compatibility with perovskite films pose significant challenges. Chemical residues from the manufacturing process can further exacerbate this issue by accelerating the decomposition of perovskite materials.240,243,245 Therefore, to achieve efficient and stable PSCs, modifications to TiO2 and ZnO ETLs or the development of alternative ETL materials is imperative. Moreover, utilising a Ti-based MOF template resulted in a highly porous structure, both internally and on the surface, due to the thermal decomposition of the MOF template. This structural improvement contributed to an increase in PCE from 12.32% to 15.75%.239 The performance enhancement was brought about by improved electron transfer of Co doping, which reduced charge carrier recombination. This was further confirmed by a significant decrease in charge transport resistance and charge recombination, as shown in Fig. 9b.239 Additionally, the high temperature (>450 °C) required for synthesizing TiO2 ETLs often limits their application in flexible perovskite solar cells. MOFs can overcome this limitation as demonstrated by Ryu et al.,246 where nanocrystalline formation of MIL-125(Ti) was employed at room temperature. This MOF-based ETL was successfully used in both rigid PSCs on glass substrates and flexible PSCs on polyethylene naphthalate (PEN) substrates, respectively.246 The electrical conductivity of Ti-MOF as an ETL was initially found to be lower than that of TiO2, primarily due to microcracks in the Ti-MOF structure. However, this issue was significantly mitigated with the incorporation of [6,6]-phenyl-C61-butyric acid methyl ester (PCBM).246 As a result, flexible PSCs displayed exceptional mechanical stability, maintaining an average PCE of 16.57% after 700 bending cycles.246 This enhanced performance was made possible by the additional H bonds provided by the organic linkers in the MOF framework.128 The small-sized nanocrystals of Ti-MOF were highly dispersed in organic solvents, which led to the simple fabrication of uniform and ultrathin films. Moreover, the electronic structure of Ti-MOF (CBM = −4.12 eV) is better suited for charge injection and transfer from the perovskite layer (CBM = −3.8 eV) to the ITO substrate compared to that of TiO2 (CBM = −3.98 eV). To overcome the instability issues caused by chemical residues, Zhang et al.240 proposed the use of porous MOF-derived ZnO as an ETL. The porous structure of the MOF-derived ZnO increased the contact area between the ETL and the perovskite layer, thereby suppressing charge carrier recombination and enhancing carrier extraction efficiency.240 Additionally, the poriferous structure of MOF-derived ZnO improved light utilisation by acting as light scattering sites, as illustrated in Fig. 9c.240 Zhang et al. also reported a reduction in trap states and photoluminescence decay, indicating a significant inhibition of charge carrier recombination in the presence of MOF-derived ZnO.240
5.3.1.2 MOFs in/as the hole transport layer. In addition to facilitating charge transport, the addition of a HTL in an n–i–p device configuration prevents direct contact between the anode (Au, Ag, or Al) and the perovskite layer. This minimises the transfer of electrons to the anode, thereby suppressing the recombination of photogenerated charge carriers.24 Common HTL materials include Spiro-OMeTAD, PEDOT: PSS, and CuI.243 However, despite being one of the best-performing HTLs, Spiro-OMeTAD raises concerns regarding its stability and its impact on the quality of the resulting perovskite layer.247 To remediate these issues, researchers have introduced multiple buffer layers to prevent the ingress of moisture and oxygen and have designed novel HTL materials.248,249 For instance, Zhang et al. introduced an interfacial carbon layer as an HTL using a simple and cost-effective glucose-derived carbonization process. This approach effectively suppressed undesirable charge recombination, thereby reducing charge losses and enhancing overall performance.250 While effective, this approach increases the complexity of the fabrication process, which is non-ideal for commercialisation.251 An alternative strategy is to directly incorporate MOFs into existing HTL materials. Li et al. demonstrated this approach by doping indium-based MOFs (In2) into Spiro-OMeTAD to improve the light responsiveness of PSCs and provide a buffer to block unwanted material penetration into the perovskite layer.251 The In2 MOF demonstrated strong UV light absorption and visible light emission. Furthermore, In2 with its cube-like structure acted as light scattering centres, extending the light path length and enhancing light utilisation.251 By sealing the gaps in Spiro-OMeTAD with In2, a denser HTL was formed, thus effectively preventing Au from the electrode and oxygen from penetrating the perovskite layer. Furthermore, the increased hydrophobicity imparted by the In2 protected the PSC device against moisture ingress significantly enhancing its stability.
In addition to improving HTL stability, the incorporation of MOFs has been reported to improve the conductivity of HTLs. As Spiro-OMeTAD suffers from low conductivity, lithium bis(trifluoromethane) sulfonimide (Li-TFSI) and 4-tert-butylpyridine (t-BP) were often added as standard bi-dopants to enhance the performance of Spiro-OMeTAD.252 The role of Li-TFSI is to act as an oxidising agent to enhance Spiro-OMeTAD conductivity, while t-BP is used to prevent charge carrier recombination. However, both dopants present significant drawbacks: Li-TFSI is hygroscopic and highly reactive to water, which results in the degradation of PSCs, while the low boiling point of t-BP causes it to volatilise during the fabrication process.253 Over time, t-BP forms salt aggregates on the surface, which reduces the mobility of charge carriers and accelerates the degradation of PSCs.254 MOFs have been proposed as alternative oxidisers to combat these drawbacks of Li-TFSI and t-BP. Wang et al. demonstrated the use of a three-dimensional (3D) MOF, {[Zn(Hcbob)]·(solvent)}n (Zn-CBOB), as a dopant for Spiro-OMeTAD. In their previous studies, Zn-CBOB was found to oxidise Spiro-OMeTAD effectively in inert environments while simultaneously addressing the uncoordinated Pb2+ ions in the perovskite layer. The carbonyl groups in Zn-CBOB with Lewis basicity interact with these uncoordinated Pb2+ ions, passivating surface traps and improving charge carrier extraction.253 Additionally, Wang et al. explored the possibility of combining Li-TFSI with MOFs to enhance both the electron transfer and oxidation processes of Spiro-OMeTAD (eqn (9) and (10)).
| Spiro-OMeTAD + Zn-CBOB ⇌ Spiro-OMeTADs˙+Zn-CBOB˙− | (9) |
| Spiro-OMeTADs˙+Zn-CBOB˙− + Li+TFSI− → Spiro-OMeTADs˙+Zn-CBOB˙− + Li+Zn-CBOB˙− | (10) |
Zn-CBOB not only facilitates the rapid oxidation of Spiro-OMeTAD but also enhances the overall hole mobility and conductivity of the PSC. The improved stability of the device was attributed to the hydrophobicity of Zn-CBOB, as revealed by SEM studies, which showed severe damage to the HTL in the absence of Zn-CBOB.253 Additionally, the porosity of MOFs contribute to uniform coverage of the HTL, preventing salt aggregation and blocking the penetration of undesired materials into the perovskite layer. Similarly, Zhou and co-workers developed an In(III)-based MOF (In-Pyia) as a substitute for t-BP in HTLs.255 The inherent porosity of the In-Pyia, along with its open pyridine nitrogen sites, allowed effective dispersion of Li-TFSI, resulting in a more uniform HTL film. Additionally, the open pyridine nitrogen atoms in In-Pyia coordinated with Li+ ions, creating a dense and crack-free HTL layer.255 In contrast, HTL films prepared with t-BP showed significant drawbacks, as the rapid volatilisation of the t-BP during fabrication led to the formation of numerous cracks on the film surface.255 The In-Pyia-modified HTL film also exhibited significantly reduced hygroscopicity, which is crucial for minimising hydration and permeation behaviours that could otherwise damage the perovskite layer.255 As a result, the In-Pyia modified HTL demonstrated significantly improved long-term stability, maintaining performance for 16 days in an ambient environment.255 Furthermore, the well-aligned energy levels and their positive impact on the oxidation of Spiro-OMeTAD make In-Pyia an excellent additive for HTLs, promoting more efficient hole extraction and conduction.255
5.3.1.3 MOFs as interlayers. MOFs have also been employed as interlayers in PSCs, where they enhance charge carrier mobility and to suppress charge recombination. Similar to blending MOFs into the perovskite layer, interlayering MOF beneath the perovskite layer can also improve the crystallinity of the perovskite film. For instance, Shen et al. observed significant improvement after introducing ZIF-8 as an interlayer between the perovskite and TiO2, which serves as the ETL, as illustrated in Fig. 9d.241 The introduction of the ZIF-8 interlayer eliminated pinholes in the perovskite films. Moreover, as the coating time of ZIF-8 increased, the grain size of the perovskite film grew while its surface roughness decreased. This suggests that the ZIF-8 interlayer not only preserved the morphology of the pristine film but also influenced the crystallisation dynamics, resulting in improved structural properties. The simultaneous increase in grain size and reduction in grain boundaries enhanced electron injection efficiency, reduced recombination rate, and ultimately improved device performance. While studies on MOFs as interlayers between the ETL and perovskite layer are more common, their use as interlayers between the HTL and perovskite layer is less prevalent. However, Lee's group provided an example of this application by utilising a MOF interlayer above an HTL, demonstrating its potential in further optimising PSC architectures. Specifically, UiO-66 and MOF-808 were specifically used to modify the surface of the NiOx HTL, effectively regulating the crystallisation of the perovskite film. The full width at half maximum (FWHM) values of the perovskite films grown on UiO-66- and MOF-808-modified NiOx films were 0.301° and 0.281°, respectively—both narrower than the 0.311° FWHM observed for films grown on unmodified NiOx.133 These results highlight the improved crystallinity of the perovskite films achieved through MOF modification. Additionally, the grain sizes of perovskite films grown on MOF-modified NiOx films were significantly larger, increasing from approximately 480 nm for unmodified samples to 720 nm and 640 nm for UiO-66- and MOF-808-modified samples, respectively.133 This demonstrates the beneficial role of the MOF scaffold in improving the crystallinity and grain size of the deposited perovskite film, which helps reduce defect density and grain boundaries, ultimately promoting charge transfer at the interface. Moreover, the perovskite films grown on MOF-modified layers exhibited a degree of PL quenching, which can be attributed to the 3D porous scaffold of the MOFs. This porous architecture facilitates the infiltration of perovskite precursors, leading to the formation of perovskite nanocrystals and improving interfacial compatibility. As a result, charge extraction efficiency is significantly enhanced, further contributing to improved device performance.214,251
6 Conclusion and future outlook
Perovskites have garnered significant attention for their exceptional optoelectronic properties, as evidenced by their record-breaking efficiencies in solar cells and LEDs. This remarkable performance has spurred interest in their application for PEC water splitting. However, conventional perovskite oxides are hindered by limited light absorption and poor stability, posing significant challenges for their practical use in PEC devices. To address these challenges, this review explores strategies to enhance PEC performance, focusing on the integration of perovskites with MOFs to form next-generation hybrid materials. Unlike perovskites, MOFs offer unique structural and functional advantages, such as tuneable porosity, high surface area, and a wide range of metal centres. By combining the strengths of these two materials, MOF-perovskite hybrids have demonstrated vast potential to overcome the individual shortcomings of their components. MOFs can enhance the stability of perovskites by shielding them from degradation while also providing additional active sites for catalytic reactions. Furthermore, the porous structure of MOFs facilitates efficient mass transport of reactants and products, thereby improving the overall system efficiency.Despite promising advancements, the field of MOF-perovskite hybrids for PEC water splitting remains relatively nascent. To date, integrating MOFs with perovskites has been shown to significantly enhance the stability and mechanical properties of perovskite-based materials. The inherent porosity of certain MOFs effectively prevents moisture infiltration, further improving perovskite stability. While the specific MOF structures employed may vary, their integration with perovskites consistently improves photoelectrode performance by enhancing perovskite film quality and crystallinity, facilitating efficient charge transfer, suppressing charge recombination, and boosting device stability. The synergy between perovskites, with their excellent light-harvesting capabilities and high charge carrier mobility, and MOFs, renowned for their tuneable porosity, large surface area, and catalytic versatility, provides a strong platform for developing next-generation photoelectrodes. These combined attributes position MOF-perovskite hybrids as highly promising candidates for high-efficiency and long-term stable PEC water-splitting systems.
The future of MOF-perovskite hybrid materials for PEC water splitting holds immense promise, but significant challenges remain. To fully unlock their potential, future research directions should prioritize the following.
6.1 Enhancing fundamental understanding
• Develop a robust theoretical framework: a comprehensive understanding of the optoelectronic properties of these hybrid materials is crucial. This necessitates the development of robust theoretical frameworks, including advanced computational models and simulations, to predict and understand their electronic structures, band alignments, charge carrier dynamics, and interfacial interactions.• Elucidate synergistic mechanisms: in-depth investigations with advanced technologies such as in situ characterization techniques are needed to elucidate the fundamental mechanisms underlying the synergistic interactions between MOFs and perovskites. This includes understanding how MOFs stabilize perovskites, influence charge carrier transport, and facilitate interfacial charge transfer.
6.2 Optimising material design and performance
• Fine-tune MOF properties: balancing MOF properties is crucial. While MOFs can enhance perovskite stability by providing a protective environment, their hydrophobic nature and narrow pore apertures can hinder mass transport. Future research should focus on fine-tuning MOF frameworks to achieve an optimal balance between perovskite protection and efficient reactant diffusion.• Explore novel hybrid architectures: drawing inspiration from successful designs in perovskite solar cells (PSCs), researchers can explore novel hybrid architectures, such as 3D hierarchical structures and the integration of co-catalysts within the MOF framework.
• Leverage AI-powered materials discovery: the emergence of artificial intelligence (AI) and machine learning offers exciting opportunities for accelerating materials discovery. AI-powered algorithms can be utilised to predict and design optimal MOF-perovskite combinations with enhanced performance.
6.3 Technological integration
• Develop scalable synthesis methods: unlike MOFs, the commercial use of perovskites is still restricted due to low STH efficiency and poor material stability. It is evident that solely focusing on the performance of the material itself is insufficient. As research progresses, we should consider the preparation process, equipment development, and the full life cycle costs associated with industrialization and commercial applications. Furthermore, current research has primarily focused on material composition and structure modifications, with a notable absence of innovative preparation methods.• Real-world operation: most of the reported perovskites have only been tested at a laboratory scale, and the potential challenges and implications of scaling up these materials for commercial application remain largely unexplored. Therefore, the large-scale preparation processes and operation that extend beyond laboratory conditions warrant further investigation.
Ultimately, the successful integration of MOFs and perovskites holds the potential to revolutionise PEC water-splitting technology, providing a sustainable and efficient solution for green H2 production. With ongoing advancements in this field, MOF-perovskite hybrids could lead to the development of high-efficiency PEC devices, making a significant contribution to the global renewable energy landscape.
Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We acknowledge financial support from the Fundamental Research Grant Scheme (FRGS) (Project Reference Code: FRGS/1/2020/STG05/MUSM/02/1) provided by the Ministry of Higher Education (MOHE), Malaysia, which supported the PhD stipend for Quan Yee Tey.Notes and references
- W. Wang, M. O. Tadé and Z. Shao, Chem. Soc. Rev., 2015, 44, 5371–5408 RSC.
- M. Ni, M. K. H. Leung, D. Y. C. Leung and K. Sumathy, Renewable Sustainable Energy Rev., 2007, 11, 401–425 CrossRef CAS.
- S. Satyapal, J. Petrovic, C. Read, G. Thomas and G. Ordaz, Catal. Today, 2007, 120, 246–256 CrossRef CAS.
- W. C. Ng, C. S. Yaw, S. N. A. Shaffee, N. A. A. Samad, Z. K. Koi and M. N. Chong, Sustainable Mater. Technol., 2024, 40, e00972 CrossRef.
- M. Aravindan, V. M. Kumar, V. S. Hariharan, T. Narahari, A. P. Kumar, K. Madhesh, G. P. Kumar and R. Prabakaran, Renewable Sustainable Energy Rev., 2023, 188, 113791 CrossRef.
- S. Lavate, H. P. S. Yalamati and R. Srivastava, Solar-Driven Green Hydrogen Generation and Storage, 2023, pp. 507–524 Search PubMed.
- J. Moore, J. Durham, A. Eijk, E. Karakas, R. Kurz, J. Lesak, M. McBain, P. McCalley, L. Moroz, Z. Mohamed, B. Pettinato, G. Phillippi, H. Watanabe and B. Williams, Machinery and Energy Systems for the Hydrogen Economy, 2022, pp. 333–424 Search PubMed.
- P. Sun, B. Young, A. Elgowainy, Z. Lu, M. Wang, B. Morelli and T. Hawkins, Environ. Sci. Technol., 2019, 53, 7103–7113 CrossRef CAS PubMed.
- M. Katebah, M. Al-Rawashdeh and P. Linke, Clean. Eng. Technol., 2022, 10, 100552 CrossRef.
- Z. Wang, Y. Gu and L. Wang, Front. Energy, 2021, 15, 596–599 CrossRef.
- P. Zhai, S. Haussener, J. Ager, R. Sathre, K. Walczak, J. Greenblatt and T. McKone, Energy Environ. Sci., 2013, 6, 2380–2389 RSC.
- Y. Chen, Y. Liu, F. Wang, X. Guan and L. Guo, J. Energy Chem., 2021, 61, 469–488 CrossRef CAS.
- S. Balachandran, N. Prakash, K. Thirumalai, M. Muruganandham, M. Sillanpää and M. Swaminathan, Ind. Eng. Chem. Res., 2014, 53, 8346–8356 CrossRef CAS.
- J. Wang, T. van Ree, Y. Wu, P. Zhang and L. Gao, Metal Oxide Semiconductors for Solar Water Splitting, Elsevier Inc., 2018, pp. 205–249 Search PubMed.
- C. S. Yaw, Q. Ruan, J. Tang, A. K. Soh and M. N. Chong, Chem. Eng. J., 2019, 364, 177–185 CrossRef CAS.
- D. Chen and Z. Liu, ChemSusChem, 2018, 11, 3438–3448 CAS.
- W. Ni, D. Yan, S. Fu, Y. Wei, H. Yang, X. Zhang, F. Zhao, K. You and H. Luo, Chem. Eng. J., 2023, 465, 142905 CAS.
- C. Zhong, T. Duan, D. Yan, S. Zhang, H. Yang, Y. Pei, J. Yan, K. You, Y. Li and H. Luo, Sep. Purif. Technol., 2024, 331, 125728 CrossRef CAS.
- NREL, Best Research Cell-Efficiencies, https://www.nrel.gov/pv/cell-efficiency.html.
- Y.-F. Xu, X.-D. Wang, J.-F. Liao, B.-X. Chen, H.-Y. Chen, D.-B. Kuang, Y.-F. Xu, X.-D. Wang, J.-F. Liao, B.-X. Chen, H.-Y. D. Chen and B. Kuang, Adv. Mater. Interfaces, 2018, 5, 1801015 CrossRef.
- K. Ma, X. Y. Du, Y. W. Zhang and S. Chen, J. Mater. Chem. C, 2017, 5, 9398–9404 RSC.
- M. Crespo-Quesada, L. M. Pazos-Outón, J. Warnan, M. F. Kuehnel, R. H. Friend and E. Reisner, Nat. Commun., 2016,(7), 1–7 Search PubMed.
- K. Rokesh, M. Sakar and T. O. Do, Mater. Lett., 2019, 242, 99–102 CrossRef CAS.
- D. Y. Heo, H. H. Do, S. H. Ahn and S. Y. Kim, Polymers, 2020, 12, 2061 CrossRef CAS PubMed.
- T. Himanshu, A. K. Agarwal, P. R. Chakraborty and S. Powar, Advances in Solar Energy Research, Springer US, 2018 Search PubMed.
- A. Raveendran, M. Chandran and R. Dhanusuraman, RSC Adv., 2023, 13, 3843–3876 RSC.
- Y. Sang, H. Liu and A. Umar, ChemCatChem, 2015, 7, 559–573 CrossRef CAS.
- C. Jiang, S. J. A. Moniz, A. Wang, T. Zhang and J. Tang, Chem. Soc. Rev., 2017, 46, 4645–4660 RSC.
- O. K. Varghese and C. A. Grimes, Sol. Energy Mater. Sol. Cells, 2008, 92, 374–384 CrossRef CAS.
- Z. Chen, T. F. Jaramillo, T. G. Deutsch, A. Kleiman-Shwarsctein, A. J. Forman, N. Gaillard, R. Garland, K. Takanabe, C. Heske, M. Sunkara, E. W. McFarland, K. Domen, E. L. Milled and H. N. Dinh, J. Mater. Res., 2010, 25, 3–16 CrossRef CAS.
- T. Hisatomi, J. Kubota and K. Domen, Chem. Soc. Rev., 2014, 43, 7520–7535 RSC.
- J. W. Jang, D. Friedrich, S. Müller, M. Lamers, H. Hempel, S. Lardhi, Z. Cao, M. Harb, L. Cavallo, R. Heller, R. Eichberger, R. van de Krol and F. F. Abdi, Adv. Energy Mater., 2017, 7, 1701536 CrossRef.
- Y. Lu, Y. Yang, X. Fan, Y. Li, D. Zhou, B. Cai, L. Wang, K. Fan and K. Zhang, Adv. Mater., 2022, 34, 2108178 CrossRef CAS PubMed.
- G. Rose, in De Novis Quibusdam Fossilibus Quae in Montibus Uraliis Inveniuntur, 1839 Search PubMed.
- K. Bienkowski, R. Solarska, L. Trinh, J. Widera-Kalinowska, B. Al-Anesi, M. Liu, G. K. Grandhi, P. Vivo, B. Oral, B. Yılmaz and R. Yıldırım, ACS Catal., 2024, 14, 6603–6622 CrossRef CAS PubMed.
- D. Saikia, A. Betal, J. Bera and S. Sahu, Mater. Sci. Semicond. Process., 2022, 150, 106953 CrossRef CAS.
- Y. Huang, J. Liu, Y. Deng, Y. Qian, X. Jia, M. Ma, C. Yang, K. Liu, Z. Wang, S. Qu and Z. Wang, J. Semicond., 2020, 41, 011701 CrossRef CAS.
- J. Zhou and J. Huang, Adv. Sci., 2018, 5, 1700256 CrossRef PubMed.
- V. M. Goldschmidt, Naturwissenschaften, 1926, 14, 477–485 CrossRef CAS.
- M. Moniruddin, B. Ilyassov, X. Zhao, E. Smith, T. Serikov, N. Ibrayev, R. Asmatulu and N. Nuraje, Mater. Today Energy, 2018, 7, 246–259 CrossRef.
- H. Kato, H. Kobayashi and A. Kudo, J. Phys. Chem. B, 2002, 106, 12441–12447 CrossRef CAS.
- B. Weng, C. R. Grice, J. Ge, T. Poudel, X. Deng and Y. Yan, Adv. Energy Mater., 2017, 8, 1701655 CrossRef.
- T. Takata, K. Shinohara, A. Tanaka, M. Hara, J. N. Kondo and K. Domen, J. Photochem. Photobiol., A, 1997, 106, 45–49 CrossRef CAS.
- A. Gupta, U. Alam and N. Verma, Int. J. Hydrogen Energy, 2024, 58, 69–78 CrossRef CAS.
- B. Du, W. Yang, Q. Jiang, H. Shan, D. Luo, B. Li, W. Tang, F. Lin, B. Shen, Q. Gong, X. Zhu, R. Zhu and Z. Fang, Adv. Opt. Mater., 2018, 6, 1701271 CrossRef.
- A. Fakharuddin, M. K. Gangishetty, M. Abdi-Jalebi, S. H. Chin, A. R. bin Mohd Yusoff, D. N. Congreve, W. Tress, F. Deschler, M. Vasilopoulou and H. J. Bolink, Nat. Electron., 2022,(5), 203–216 CrossRef CAS.
- K. Shum and A. Tsatskina, Nat. Energy, 2016,(1), 1–2 Search PubMed.
- J. A. Gómez-Cuaspud, E. Vera-López, J. B. Carda-Castelló and E. Barrachina-Albert, React. Kinet. Mech. Catal., 2017, 120, 167–179 CrossRef.
- X. Yang, G. Xu, Z. Ren, X. Wei, C. Chao, S. Gong, G. Shen and G. Han, CrystEngComm, 2014, 16, 4176–4182 RSC.
- E. Grabowska, Appl. Catal., B, 2016, 186, 97–126 CrossRef CAS.
- L. Yang, S. Fu, K. Leng, Q. Tang, Z. Wu, K. Yi and X. Zhu, Open Ceram., 2022, 9, 100238 CrossRef CAS.
- H. Li, C. Lai, Z. Wei, X. Zhou, S. Liu, L. Qin, H. Yi, Y. Fu, L. Li, M. Zhang, F. Xu, H. Yan, M. Xu, D. Ma and Y. Li, Chemosphere, 2023, 344, 140395 CrossRef CAS PubMed.
- M. A. I. Islam, T. Sato, F. Ara and M. A. Basith, J. Alloys Compd., 2023, 944, 169066 CrossRef CAS.
- S. Boumaza, R. Brahimi, L. Boudjellal, A. Belhadi and M. Trari, Synthesis and Characterization of the Double Perovskite La2NiO4-Application for Hydrogen Production, ed. A. Khellaf, Springer Nature Singapore, Singapore, 2021, pp. 339–346 Search PubMed.
- J. Kirchnerova and D. Klvana, Int. J. Hydrogen Energy, 1994, 19, 501–506 CrossRef CAS.
- S. Feng, Y. Zhang, P. Zhang, J. Cao, J. Wang, Y. Zheng, L. Shi, J. Pan and C. Li, ACS Appl. Nano Mater., 2023, 6, 14469–14477 CrossRef CAS.
- T. Soto-Montero and M. Morales-Masis, ACS Energy Lett., 2024, 9, 4199–4208 CrossRef CAS PubMed.
- S. Sharma, D. Kumar, A. Manori Dhyani and C. Bhardwaj, Mater. Today Proc., 2023, 73, 118–121 CrossRef CAS.
- B. M. Bresolin, Y. Park and D. W. Bahnemann, Catalysts, 2020, 10, 709 CrossRef CAS.
- T. Bu, J. Li, Q. Lin, D. P. McMeekin, J. Sun, M. Wang, W. Chen, X. Wen, W. Mao, C. R. McNeill, W. Huang, X. L. Zhang, J. Zhong, Y. B. Cheng, U. Bach and F. Huang, Nano Energy, 2020, 75, 104917 CrossRef CAS.
- Q. Dong, Y. Fang, Y. Shao, P. Mulligan, J. Qiu, L. Cao and J. Huang, Science, 2015, 347, 967–970 CrossRef CAS PubMed.
- D. W. DeQuilettes, S. M. Vorpahl, S. D. Stranks, H. Nagaoka, G. E. Eperon, M. E. Ziffer, H. J. Snaith and D. S. Ginger, Science, 2015, 348, 683–686 CrossRef CAS PubMed.
- C. C. Stoumpos, C. D. Malliakas and M. G. Kanatzidis, Inorg. Chem., 2013, 52, 9019–9038 CrossRef CAS PubMed.
- Y. Gao, K. Wang, M. Wang, J. I. Khan, A. H. Balawi, W. Liu, S. De Wolf and F. Laquai, Sol. RRL, 2020, 4, 2000072 CrossRef CAS.
- X. Wan, Z. Yu, W. Tian, F. Huang, S. Jin, X. Yang, Y. B. Cheng, A. Hagfeldt and L. Sun, J. Energy Chem., 2020, 46, 8–15 CrossRef.
- National Renewable Energy Laboratory, Research Cell Efficiency Record, 2019 Search PubMed.
- Q. Jiang, Y. Zhao, X. Zhang, X. Yang, Y. Chen, Z. Chu, Q. Ye, X. Li, Z. Yin and J. You, Nat. Photonics, 2019, 12, 460–466 CrossRef.
- H. Huang, B. Pradhan, J. Hofkens, M. B. J. Roeffaers and J. A. Steele, ACS Energy Lett., 2020, 5, 1107–1123 Search PubMed.
- P. Schulz, E. Edri, S. Kirmayer, G. Hodes, D. Cahen and A. Kahn, Energy Environ. Sci., 2014, 7, 1377–1381 Search PubMed.
- G. E. Eperon, S. D. Stranks, C. Menelaou, M. B. Johnston, L. M. Herz and H. J. Snaith, Energy Environ. Sci., 2014, 7, 982–988 Search PubMed.
- S. Wang, Y. Jiang, E. J. Juarez-Perez, L. K. Ono and Y. Qi, Nat. Energy, 2017, 2, 16195 CrossRef CAS.
- Y. Xie, Y. Yu, J. Gong, C. Yang, P. Zeng, Y. Dong, B. Yang, R. Liang, Q. Ou and S. Zhang, Opt. Mater. Express, 2018, 8, 3494–3505 CrossRef CAS.
- L. Gao, S. Huang, L. Chen, X. Li, B. Ding, S. Huang and G. Yang, Sol. RRL, 2018, 2, 1800088 CrossRef.
- M. Xie and J. Tian, J. Phys. Chem. Lett., 2022, 13, 1962–1971 CrossRef CAS PubMed.
- M. Shirayama, M. Kato, T. Miyadera, T. Sugita, T. Fujiseki, S. Hara, H. Kadowaki, D. Murata, M. Chikamatsu and H. Fujiwara, J. Appl. Phys., 2016, 119, 115501 CrossRef.
- S. J. A. Moniz, C. S. Blackman, P. Southern, P. M. Weaver, J. Tang and C. J. Carmalt, Nanoscale, 2015, 7, 16343–16353 Search PubMed.
- P. Nandigana, S. Pari, D. Sujatha, M. Shidhin, C. Jeyabharathi and S. K. Panda, ChemistrySelect, 2023, 8, e202204731 CrossRef CAS.
- J. Li, Z. Tan, M. Hu, C. Chen, J. Luo, S. Li, L. Gao, Z. Xiao, G. Niu and J. Tang, Front. Optoelectron., 2019, 12, 352–364 CrossRef.
- S. Kumar and A. K. Ganguli, Appl. Surf. Sci. Adv., 2022, 9, 100239 CrossRef.
- I. Chung, J. H. Song, J. Im, J. Androulakis, C. D. Malliakas, H. Li, A. J. Freeman, J. T. Kenney and G. Kanatzidis, J. Am. Chem. Soc., 2012, 134, 8579–8587 Search PubMed.
- H. Peng, L. Xu, Y. Sheng, W. Sun, Y. Yang, H. Deng, W. Chen and J. Liu, Small, 2021, 17, 2102149 Search PubMed.
- A. Exner, R. A. Gouvêa, A. Köche, S. Khan, J. F. L. Santos and M. José Leite Santos, J. Sci.:Adv. Mater. Devices, 2024, 9, 100700 CAS.
- A. M. Ganose, C. N. Savory and D. O. Scanlon, Chem. Commun., 2016, 53, 20–44 RSC.
- O. A. Lozhkina, A. A. Murashkina, V. V. Shilovskikh, Y. V. Kapitonov, V. K. Ryabchuk, A. V. Emeline and T. Miyasaka, J. Phys. Chem. Lett., 2018, 9, 5408–5411 CrossRef CAS PubMed.
- T. Kako, N. Kikugawa and J. Ye, Catal. Today, 2008, 131, 197–202 Search PubMed.
- F. Matteocci, L. Cinà, E. Lamanna, S. Cacovich, G. Divitini, P. A. Midgley, C. Ducati and A. Di Carlo, Nano Energy, 2016, 30, 162–172 Search PubMed.
- R. Cheacharoen, C. C. Boyd, G. F. Burkhard, T. Leijtens, J. A. Raiford, K. A. Bush, S. F. Bent and M. D. McGehee, Sustainable Energy Fuels, 2018, 2, 2398–2406 RSC.
- F. Corsini and G. Griffini, JPhys Energy, 2020, 2, 031002 Search PubMed.
- R. Konta, T. Ishii, H. Kato and A. Kudo, J. Phys. Chem. B, 2004, 108, 8992–8995 CrossRef CAS.
- T. Ohno, T. Tsubota, Y. Nakamura and K. Sayama, Appl. Catal., A, 2005, 288, 74–79 CrossRef CAS.
- Z. Li, M. Yang, J. S. Park, S. H. Wei, J. J. Berry and K. Zhu, Chem. Mater., 2016, 28, 284–292 CrossRef CAS.
- J. P. Zou, L. Z. Zhang, S. L. Luo, L. H. Leng, X. B. Luo, M. J. Zhang, Y. Luo and G. C. Guo, Int. J. Hydrogen Energy, 2012, 37, 17068–17077 CrossRef CAS.
- M. Abdi, V. Mahdikhah and S. Sheibani, Opt. Mater., 2020, 102, 109803 CrossRef CAS.
- T. Oshikiri, X. Shi, K. Ueno, A. -, T. Yu, X. Chen, Z. Zhao, X. Zhang, Y. Lei, P. Yang, J. Fan, B. Zhang and S. Yin, Mater. Res. Express, 2020, 7, 15034 CrossRef.
- A. Swarnkar, W. J. Mir and A. Nag, ACS Energy Lett., 2018, 3, 286–289 CrossRef CAS.
- G. E. Eperon and D. S. Ginger, ACS Energy Lett., 2017, 2, 1190–1196 CrossRef CAS.
- G. Fu, W. Li, J.-Y. Zhang, M. Li, C. Li, N. Li, Q. He, S. Xi, D. Qi, J. L. MacManus-Driscoll, J. Cheng, K. Hongliang Zhang, G. Fu, J. Zhang, J. Cheng, K. H. Zhang, W. Li, J. L. MacManus-Driscoll, M. Li, C. Li, N. Li, Q. He, S. Xi and D. Qi, Small, 2021, 17, 2006930 CAS.
- Z. Li, L. Lv, J. Wang, X. Ao, Y. Ruan, D. Zha, G. Hong, Q. Wu, Y. Lan, C. Wang, J. Jiang and M. Liu, Nano Energy, 2018, 47, 199–209 CAS.
- B. Hua, M. Li, W. Pang, W. Tang, S. Zhao, Z. Jin, Y. Zeng, B. Shalchi Amirkhiz and J. L. Luo, Chem, 2018, 4, 2902–2916 CAS.
- J. Xiong, H. Zhong, J. Li, X. Zhang, J. Shi, W. Cai, K. Qu, C. Zhu, Z. Yang, S. P. Beckman and H. Cheng, Appl. Catal., B, 2019, 256, 117817 CAS.
- W. Wang, Y. Yang, D. Huan, L. Wang, N. Shi, Y. Xie, C. Xia, R. Peng and Y. Lu, J. Mater. Chem. A, 2019, 7, 12538–12546 CAS.
- C.-H. Lu, G. V Biesold-Mcgee, Y. Liu, Z. Kang and Z. Lin, Chem. Soc. Rev., 2020, 49, 4953 CAS.
- A. Buin, R. Comin, J. Xu, A. H. Ip and E. H. Sargent, Chem. Mater., 2015, 27, 4405–4412 CAS.
- G. Xing, N. Mathews, S. Sien Lim, N. Yantara, X. Liu, D. Sabba, M. Grätzel, S. Mhaisalkar and T. Chien Sum, Nat. Mater., 2014, 13, 476–480 CAS.
- Y. Liu, C. Ye, S. N. Zhao, Y. Wu, C. Liu, J. Huang, L. Xue, J. Sun, W. Zhang, X. Wang, P. Xiong and J. Zhu, Nano Energy, 2022, 99, 107344 CrossRef CAS.
- J. K. Cooper, S. Gul, F. M. Toma, L. Chen, P. A. Glans, J. Guo, J. W. Ager, J. Yano and I. D. Sharp, Chem. Mater., 2014, 26, 5365–5373 CAS.
- J. H. Kim, Y. Jo, J. H. Kim, J. W. Jang, H. J. Kang, Y. H. Lee, D. S. Kim, Y. Jun and J. S. Lee, ACS Nano, 2015, 9, 11820–11829 CAS.
- Y. Pihosh, I. Turkevych, K. Mawatari, T. Asai, T. Hisatomi, J. Uemura, M. Tosa, K. Shimamura, J. Kubota, K. Domen and T. Kitamori, Small, 2014, 10, 3692–3699 CrossRef CAS.
- L. Han, F. F. Abdi, R. Van De Krol, R. Liu, Z. Huang, H. J. Lewerenz, B. Dam, M. Zeman and A. H. M. Smets, ChemSusChem, 2014, 7, 2832–2838 CrossRef CAS PubMed.
- Y. Yang, D. Xu, Q. Wu and P. Diao, Sci. Rep., 2016, 6, 35158 CrossRef CAS PubMed.
- J. Wen, Y. Zhao, P. Wu, Y. Liu, X. Zheng, R. Lin, S. Wan, K. Li, H. Luo, Y. Tian, L. Li and H. Tan, Nat. Commun., 2023, 14, 1–10 Search PubMed.
- H. Li, Y. Zhou, W. Tu, J. Ye and Z. Zou, Adv. Funct. Mater., 2015, 25, 998–1013 CrossRef CAS.
- B. Y. Cheng, J. S. Yang, H. W. Cho and J. J. Wu, ACS Appl. Mater. Interfaces, 2016, 8, 20032–20039 CrossRef CAS PubMed.
- J. S. Jang, C. W. Ahn, S. S. Won, J. H. Kim, W. Choi, B.-S. Lee, J.-H. Yoon, H. G. Kim and J. S. Lee, J. Phys. Chem. C, 2017, 121, 15063–15070 CrossRef CAS.
- J. P. Jayaraman, M. Hamdan, M. Velpula, N. S. Kaisare and A. K. Chandiran, ACS Appl. Mater. Interfaces, 2021, 13, 16267–16278 CrossRef CAS PubMed.
- S. Samajdar, S. Bera, P. S. Das, H. Finch, V. R. Dhanak, S. Chakraborty, T. Maiyalagan, K. Annapurna and S. Ghosh, Int. J. Hydrogen Energy, 2023, 48, 17838–17851 CrossRef CAS.
- S. Chandrasekaran, E. J. Kim, J. S. Chung, I. K. Yoo, V. Senthilkumar, Y. S. Kim, C. R. Bowen, V. Adamaki and S. Hyun Hur, Chem. Eng. J., 2017, 309, 682–690 CrossRef CAS.
- T. T. T. Tran, S. Kim and J. Seo, J. Catal., 2022, 406, 157–164 CrossRef CAS.
- Q. Xu, L. Zhang, B. Cheng, J. Fan and J. Yu, Chem, 2020, 6, 1543–1559 CAS.
- T. W. Kim and K. S. Choi, Science, 2014, 343, 990–994 CrossRef CAS PubMed.
- M. Hamdan and A. K. Chandiran, Angew. Chem. Int. Ed., 2020, 59, 16033–16038 CrossRef CAS PubMed.
- J. Seo, T. Hisatomi, M. Nakabayashi, N. Shibata, T. Minegishi, M. Katayama, K. Domen, J. Seo, T. Hisatomi, T. Minegishi, M. Katayama, K. Domen, M. Nakabayashi and N. Shibata, Adv. Energy Mater., 2018, 8, 1800094 CrossRef.
- J. R. Long and O. M. Yaghi, Chem. Soc. Rev., 2009, 38, 1213–1214 RSC.
- R. Kaur, K. H. Kim, A. K. Paul and A. Deep, J. Mater. Chem. A, 2016, 4, 3991–4002 RSC.
- S. Lin, P. M. Usov and A. J. Morris, Chem. Commun., 2018, 54, 6965–6974 RSC.
- A. Crake, K. C. Christoforidis, A. Gregg, B. Moss, A. Kafizas and C. Petit, Small, 2019, 15, 1805473 CrossRef PubMed.
- X. Feng, Y. Pi, Y. Song, C. Brzezinski, Z. Xu, Z. Li and W. Lin, J. Am. Chem. Soc., 2020, 142, 690–695 CrossRef CAS PubMed.
- J. Hou, Z. Wang, P. Chen, V. Chen, A. K. Cheetham and L. Wang, Angew. Chem., Int. Ed., 2020, 59, 19434–19449 CrossRef CAS PubMed.
- Y. Shi, A. F. Yang, C. S. Cao and B. Zhao, Coord. Chem. Rev., 2019, 390, 50–75 CrossRef CAS.
- Y. Xiao, X. Guo, N. Yang and F. Zhang, J. Energy Chem., 2021, 58, 508–522 CrossRef CAS.
- Y. Pi, X. Feng, Y. Song, Z. Xu, Z. Li and W. Lin, J. Am. Chem. Soc., 2020, 142, 10302–10307 CAS.
- S. Yuan, L. Feng, K. Wang, J. Pang, M. Bosch, C. Lollar, Y. Sun, J. Qin, X. Yang, P. Zhang, Q. Wang, L. Zou, Y. Zhang, L. Zhang, Y. Fang, J. Li and H. C. Zhou, Adv. Mater., 2018, 30, 1704303 Search PubMed.
- C. C. Lee, C. I. Chen, Y. Te Liao, K. C. W. Wu and C. C. Chueh, Adv. Sci., 2019, 6, 1801715 CrossRef PubMed.
- J. Tian, J. Yu, Q. Tang, L. Zhang, F. Liand and L. Luo, IOP Conf. Ser. Earth Environ. Sci., 2018, 108, 042104 Search PubMed.
- Z. Ni and R. I. Masel, J. Am. Chem. Soc., 2006, 128, 12394–12395 CAS.
- Y. K. Seo, G. Hundal, I. T. Jang, Y. K. Hwang, C. H. Jun and J. S. Chang, Microporous Mesoporous Mater., 2009, 119, 331–337 CAS.
- T. Tsuzuki, Commun. Chem., 2021,(4), 1–11 Search PubMed.
- M. Ali, E. Pervaiz, T. Noor, O. Rabi, R. Zahra and M. Yang, Int. J. Energy Res., 2021, 45, 1190–1226 CAS.
- Z. Zhuang and D. Liu, Nano-Micro Lett., 2020, 12, 132 CAS.
- Y. Dou, J. Zhou, A. Zhou, J. R. Li and Z. Nie, J. Mater. Chem. A, 2017, 5, 19491–19498 CAS.
- J. W. Yoon, D. H. Kim, J. H. Kim, H. W. Jang and J. H. Lee, Appl. Catal., B, 2019, 244, 511–518 CAS.
- H. Song, Z. Sun, Y. Xu, Y. Han, J. Xu, J. Wu, T. Sun, H. Meng and X. Zhang, Sep. Purif. Technol., 2019, 228, 115764 CAS.
- T. Song, L. Zhang, P. Zhang, J. Zeng, T. Wang, A. Ali and H. Zeng, J. Mater. Chem. A, 2017, 5, 6013–6018 CAS.
- X. Cao, C. Tan, M. Sindoro and H. Zhang, Chem. Soc. Rev., 2018, 47, 5997 CAS.
- S. Dang, Q. L. Zhu and Q. Xu, Nat. Rev. Mater., 2018, 3, 17075 CAS.
- C. Guan, X. Liu, W. Ren, X. Li, C. Cheng, J. Wang, C. Guan, X. M. Liu, X. Li, J. Wang, W. N. Ren and C. W. Cheng, Adv. Energy Mater., 2017, 7, 1602391 CrossRef.
- D. Xu, T. Xia, W. Fan, H. Bai, J. Ding, B. Mao and W. Shi, Appl. Surf. Sci., 2019, 491, 497–504 CrossRef CAS.
- L. Shi, D. Benetti, F. Li, Q. Wei and F. Rosei, Appl. Catal., B, 2020, 263, 118317 CrossRef CAS.
- L. Zhang, P. Cui, H. Yang, J. Chen, F. Xiao, Y. Guo, Y. Liu, W. Zhang, F. Huo and B. Liu, Adv. Sci., 2015, 3, 1500243 CrossRef PubMed.
- L. Pan, T. Muhammad, L. Ma, Z. F. Huang, S. Wang, L. Wang, J. J. Zou and X. Zhang, Appl. Catal., B, 2016, 189, 181–191 CrossRef CAS.
- J. D. Xiao, Q. Shang, Y. Xiong, Q. Zhang, Y. Luo, S. H. Yu and H. L. Jiang, Angew. Chem. Int. Ed., 2016, 55, 9389–9393 CrossRef CAS PubMed.
- Z. Jin and H. Yang, Nanoscale Res. Lett., 2017, 12, 539 CrossRef PubMed.
- X. Hao, Z. Jin, H. Yang, G. Lu and Y. Bi, Appl. Catal., B, 2017, 210, 45–56 CrossRef CAS.
- Y. J. Dong, J. F. Liao, Z. C. Kong, Y. F. Xu, Z. J. Chen, H. Y. Chen, D. Bin Kuang, D. Fenske and C. Y. Su, Appl. Catal., B, 2018, 237, 9–17 CrossRef CAS.
- X. Li, S. Liu, K. Fan, Z. Liu, B. Song and J. Yu, Adv. Energy Mater., 2018, 8, 1800101 CrossRef.
- G. Jia, L. Liu, L. Zhang, D. Zhang, Y. Wang, X. Cui and W. Zheng, Appl. Surf. Sci., 2018, 448, 254–260 CrossRef CAS.
- Q. Zhang, H. Wang, Y. Dong, J. Yan, X. Ke, Q. Wu and S. Xue, Sol. Energy, 2018, 171, 388–396 CAS.
- P. Tian, X. He, W. Li, L. Zhao, W. Fang, H. Chen, F. Zhang, W. Zhang and W. Wang, J. Mater. Sci., 2018, 53, 12016–12029 CrossRef CAS.
- Z. Q. Liu, K. Qiu, H. Bai, F. Wang, Y. Ge, W. Cui, G. Zheng, J. Cui and W. Fan, Funct. Mater. Lett., 2018, 11, 1850085 CrossRef CAS.
- Z. Peng, S. C. Abbas, J. Lv, R. Yang, M. Wu and Y. Wang, Int. J. Hydrogen Energy, 2019, 44, 2446–2453 CrossRef CAS.
- C. H. Li, C. L. Huang, X. F. Chuah, D. Senthil Raja, C. T. Hsieh and S. Y. Lu, Chem. Eng. J., 2019, 361, 660–670 CrossRef CAS.
- X. Hou, L. Wu, L. Gu, G. Xu, H. Du and Y. Yuan, J. Mater. Sci. Mater. Electron., 2019, 30, 5203–5211 CrossRef CAS.
- W. Cui, J. Shang, H. Bai, J. Hu, D. Xu, J. Ding, W. Fan and W. Shi, Chem. Eng. J., 2020, 388, 124206 CrossRef CAS.
- J. Han, Y. Wang, Y. Liu, J. Bai, R. Zhao and L. Wang, Catal. Lett., 2021, 151, 2560 CrossRef CAS.
- C. G. Silva, I. Luz, F. X. Llabrés, I. Xamena, A. Corma and H. García, Chem.–Eur. J., 2010, 16, 11133–11138 CrossRef CAS PubMed.
- M. W. Logan, S. Ayad, J. D. Adamson, T. Dilbeck, K. Hanson and F. J. Uribe-Romo, J. Mater. Chem. A, 2017, 5, 11854–11863 RSC.
- Y. Li, Y. Fu, B. Ni, K. Ding, W. Chen, K. Wu, X. Huang and Y. Zhang, AIP Adv., 2018, 8, 035012 CrossRef.
- T. Toyao, M. Saito, Y. Horiuchi, K. Mochizuki, M. Iwata, H. Higashimura and M. Matsuoka, Catal. Sci. Technol., 2013, 3, 2092–2097 RSC.
- C. H. Hendon, D. Tiana, M. Fontecave, C. Sanchez, L. D'Arras, C. Sassoye, L. Rozes, C. Mellot-Draznieks and A. Walsh, J. Am. Chem. Soc., 2013, 135, 10942–10945 CrossRef CAS PubMed.
- L. Shen, M. Luo, Y. Liu, R. Liang, F. Jing and L. Wu, Appl. Catal., B, 2015, 166–167, 445–453 CrossRef CAS.
- W. Cui, H. Bai, J. Shang, F. Wang, D. Xu, J. Ding, W. Fan and W. Shi, Electrochim. Acta, 2020, 349, 136383 CrossRef CAS.
- M. A. Nasalevich, M. Van Der Veen, F. Kapteijn and J. Gascon, CrystEngComm, 2014, 16, 4919–4926 RSC.
- J. He, H. Yang, Y. Chen, Z. Yan, Y. Zeng, Z. Luo, W. Gao and J. Wang, Water Air Soil Pollut., 2015, 226, 197 CrossRef.
- H. Wang, X. Yuan, Y. Wu, G. Zeng, H. Dong, X. Chen, L. Leng, Z. Wu and L. Peng, Appl. Catal., B, 2016, 186, 19–29 CrossRef CAS.
- B. O'Regan and M. Grätzel, Nature, 1991, 353, 737–740 CrossRef.
- J. He, J. Wang, Y. Chen, J. Zhang, D. Duan, Y. Wanga and Z. Yan, Chem. Commun., 2014, 50, 7063–7066 RSC.
- H. Tributsch, Coord. Chem. Rev., 2004, 248, 1511–1530 CrossRef CAS.
- Y. P. Yuan, L. S. Yin, S. W. Cao, G. S. Xu, C. H. Li and C. Xue, Appl. Catal., B, 2015, 168–169, 572–576 CrossRef CAS.
- B. Ma, F. Wen, H. Jiang, J. Yang, P. Ying and C. Li, Catal. Lett., 2010, 134, 78–86 CrossRef CAS.
- Y. Fu, D. Sun, Y. Chen, R. Huang, Z. Ding, X. Fu and Z. Li, Angew. Chem. Int. Ed., 2012, 51, 3364–3367 CrossRef CAS PubMed.
- M. Sun, Q. Q. Wang, C. Qin, C. Y. Sun, X. L. Wang and Z. M. Su, Chem.–Eur. J., 2018, 25, 2824–2830 Search PubMed.
- S.-Y. Han, D.-L. Pan, H. Chen, X.-B. Bu, Y.-X. Gao, H. Gao, Y. Tian, G.-S. Li, G. Wang, S.-L. Cao, C.-Q. Wan and G.-C. Guo, Angew. Chem., 2018, 31, 10012–10017 Search PubMed.
- S. M. Liu, Z. Zhang, X. Li, H. Jia, M. Ren and S. Liu, Adv. Mater. Interfaces, 2018, 5, 1801062 CrossRef.
- Q. Yang, Q. Xu and H. L. Jiang, Chem. Soc. Rev., 2017, 46, 4774–4808 RSC.
- T. He, S. Chen, B. Ni, Y. Gong, Z. Wu, L. Song, L. Gu, W. Hu and X. Wang, Angew. Chem. Int. Ed., 2018, 57, 3493–3498 CrossRef CAS PubMed.
- Y. Han, H. Xu, Y. Su, Z. liang Xu, K. Wang and W. Wang, J. Catal., 2019, 370, 70–78 CrossRef CAS.
- B. Hinnemann, P. G. Moses, J. Bonde, K. P. Jørgensen, J. H. Nielsen, S. Horch, I. Chorkendorff and J. K. Nørskov, J. Am. Chem. Soc., 2005, 127, 5308–5309 CrossRef CAS PubMed.
- Y. Hou, B. L. Abrams, P. C. K. Vesborg, M. E. Björketun, K. Herbst, L. Bech, A. M. Setti, C. D. Damsgaard, T. Pedersen, O. Hansen, J. Rossmeisl, S. Dahl, J. K. Nørskov and I. Chorkendorff, Nat. Mater., 2011, 10, 434–438 CrossRef CAS PubMed.
- K. Arifin, R. M. Yunus, L. J. Minggu and M. B. Kassim, Int. J. Hydrogen Energy, 2021, 46, 4998–5024 CrossRef CAS.
- T. F. Hou, M. A. Johar, R. Boppella, M. A. Hassan, S. J. Patil, S. W. Ryu and D. W. Lee, J. Energy Chem., 2020, 49, 262–274 CrossRef.
- T. N. Q. Trang, L. T. N. Tu, T. V. Man, M. Mathesh, N. D. Nam and V. T. H. Thu, Composites, Part B, 2019, 174, 106969 CrossRef CAS.
- S. Li, W. Xu, L. Meng, W. Tian and L. Li, Small Sci., 2022, 2, 2100112 CrossRef CAS.
- C. V. Reddy, K. R. Reddy, V. V. N. Harish, J. Shim, M. V. Shankar, N. P. Shetti and T. M. Aminabhavi, Int. J. Hydrogen Energy, 2020, 45, 7656–7679 CrossRef CAS.
- G. E. M. Schukraft, B. Moss, A. G. Kafizas and C. Petit, ACS Appl. Mater. Interfaces, 2022, 14, 19342–19352 CrossRef CAS PubMed.
- S. Guo, L. Chi, T. Zhao, Y. Nan, X. Sun, Y. Huang, B. Hou and X. Wang, J. Electroanal. Chem., 2021, 880, 114915 CrossRef CAS.
- W. W. Dong, J. Jia, Y. Wang, J. R. An, O. Y. Yang, X. J. Gao, Y. L. Liu, J. Zhao and D. S. Li, Chem. Eng. J., 2022, 438, 135622 CrossRef CAS.
- T. Zeng, D. Shi, Q. Cheng, G. Liao, H. Zhou and Z. Pan, Environ. Sci.:Nano, 2020, 7, 861–879 RSC.
- R. Peng, C. M. Wu, J. Baltrusaitis, N. M. Dimitrijevic, T. Rajh and R. T. Koodali, Chem. Commun., 2013, 49, 3221–3223 RSC.
- Y. Horiuchi, T. Toyao, K. Miyahara, L. Zakary, D. Do Van, Y. Kamata, T. H. Kim, S. W. Lee and M. Matsuoka, Chem. Commun., 2016, 52, 5190–5193 RSC.
- Y. Li, Y. Xia, K. Liu, K. Ye, Q. Wang, S. Zhang, Y. Huang and H. Liu, ACS Appl. Mater. Interfaces, 2020, 12, 25494–25502 CrossRef PubMed.
- L. Zhao, Z. Zhao, Y. Li, X. Chu, Z. Li, Y. Qu, L. Bai and L. Jing, Nanoscale, 2020, 12, 10010–10018 RSC.
- Y. Pan, D. Li and H. L. Jiang, Chem.–Eur. J., 2018, 24, 18403–18407 CrossRef CAS PubMed.
- L. Shi, T. Wang, H. Zhang, K. Chang and J. Ye, Adv. Funct. Mater., 2015, 25, 5360–5367 CrossRef CAS.
- S. M. Hosseini, M. Karimi and V. Safarifard, New J. Chem., 2021, 45, 17674–17682 RSC.
- V. R. Bakuru, M. E. DMello and S. B. Kalidindi, ChemPhysChem, 2019, 20, 1177–1215 CrossRef CAS PubMed.
- K. Meyer, M. Ranocchiari and J. A. Van Bokhoven, Energy Environ. Sci., 2015, 8, 1923–1937 RSC.
- S. Wang and X. Wang, Small, 2015, 11, 3097–3112 CrossRef CAS PubMed.
- C. Zhang, W. Li and L. Li, Angew. Chem. Int. Ed., 2020, 60, 7488–7501 CrossRef PubMed.
- L. Li, H. Zhang, C. Liu, P. Liang, N. Mitsuzaki and Z. Chen, Energy Technol., 2019, 7, 1801069 CrossRef.
- Z. Jiao, J. Zheng, C. Feng, Z. Wang, X. Wang, G. Lu and Y. Bi, ChemSusChem, 2016, 9, 2824–2831 CrossRef CAS PubMed.
- W. Zhang, R. Li, X. Zhao, Z. Chen, A. W. K. Law and K. Zhou, ChemSusChem, 2018, 11, 2710–2716 CrossRef CAS PubMed.
- H. Yang, J. Bright, S. Kasani, P. Zheng, T. Musho, B. Chen, L. Huang and N. Wu, Nano Res., 2019, 12, 643–650 CrossRef CAS.
- W. Wang, X. Xu, W. Zhou and Z. Shao, Adv. Sci., 2017, 4, 1600371 CrossRef PubMed.
- A. V. Vinogradov, H. Zaake-Hertling, E. Hey-Hawkins, A. V. Agafonov, G. A. Seisenbaeva, V. G. Kessler and V. V. Vinogradov, Chem. Commun., 2014, 50, 10210–10213 RSC.
- R. Dutta, R. Shrivastav, M. Srivastava, A. Verma, S. Saxena, N. K. Biswas, V. R. Satsangi and S. Dass, Int. J. Hydrogen Energy, 2022, 47, 5192–5210 CrossRef CAS.
- S. K. Yadav, G. K. Grandhi, D. P. Dubal, J. C. de Mello, M. Otyepka, R. Zbořil, R. A. Fischer and K. Jayaramulu, Small, 2020, 16, 1–32 Search PubMed.
- J. Lee, N. Tsvetkov, S. R. Shin and J. K. Kang, ACS Appl. Mater. Interfaces, 2022, 14, 35495–35503 CrossRef CAS PubMed.
- R. Stanton and D. J. Trivedi, Nano Lett., 2023, 23, 11932–11939 CrossRef CAS PubMed.
- Y. Ye, Y. Yin, Y. Chen, S. Li, L. Li and Y. Yamauchi, Small, 2023, 19, 2208119 CrossRef CAS PubMed.
- S. Mollick, T. N. Mandal, A. Jana, S. Fajal, A. V. Desai and S. K. Ghosh, ACS Appl. Nano Mater., 2019, 2, 1333–1340 CrossRef CAS.
- L. Y. Wu, Y. F. Mu, X. X. Guo, W. Zhang, Z. M. Zhang, M. Zhang and T. B. Lu, Angew. Chem. Int. Ed., 2019, 58, 9491–9495 CrossRef CAS PubMed.
- Y. Dong, J. Zhang, H. Zhang, W. Wang, B. Hu, D. Xia, K. Lin, L. Geng and Y. Yang, Nano-Micro Lett., 2024, 16, 1–14 CrossRef PubMed.
- T. H. Chang, C. W. Kung, H. W. Chen, T. Y. Huang, S. Y. Kao, H. C. Lu, M. H. Lee, K. M. Boopathi, C. W. Chu and K. C. Ho, Adv. Mater., 2015, 27, 7229–7235 CrossRef CAS PubMed.
- H. A. Younes, M. Taha, R. Khaled, H. M. Mahmoud and R. M. Abdelhameed, J. Alloys Compd., 2023, 930, 167322 CrossRef CAS.
- D. Xu, L. Li, H. Xu, J. Zhu, W. Fan, J. Ding and W. Shi, J. Solid State Chem., 2019, 280, 120986 CrossRef CAS.
- S. Sharma, N. Jacob, G. K. Grandhi, M. B. Choudhary, S. Ippili, V. R. Hathwar, P. Vivo, R. Lo, M. Motapothula and K. Jayaramulu, iScience, 2024, 27, 110924 CrossRef CAS PubMed.
- R. Tang, L. Wang, Z. Zhang, W. Yang, H. Xu, A. Kheradmand, Y. Jiang, R. Zheng and J. Huang, Appl. Catal., B, 2021, 296, 120382 CrossRef CAS.
- R. Tang, S. Zhou, H. Li, R. Chen, L. Zhang and L. Yin, Appl. Catal., B, 2020, 265, 118583 CrossRef CAS.
- H. Ramezanalizadeh and F. Manteghi, RSC Adv., 2016, 6, 99096–99104 RSC.
- J. Swain, A. Priyadarshini, S. Hajra, S. Panda, J. Panda, R. Samantaray, Y. Yamauchi, M. Han, H. J. Kim and R. Sahu, J. Alloys Compd., 2023, 965, 171438 CrossRef CAS.
- A. Afzalinia and M. Mirzaee, J. Mol. Struct., 2023, 1283, 135322 CrossRef CAS.
- X. Kang, K. Lyu, L. Li, J. Li, L. Kimberley, B. Wang, L. Liu, Y. Cheng, M. D. Frogley, S. Rudić, A. J. Ramirez-Cuesta, R. A. W. Dryfe, B. Han, S. Yang and M. Schröder, Nat. Commun., 2019, 10, 1–9 CrossRef CAS PubMed.
- L. Zhang, Z. Shen, D. Li, J. Lin, Y. Chen, C. Ma, R. Pu, L. Wu, Y. Zhu, C. Li, D. Hou, G. Gao, W. Luo, Z. Duan, T. Zhang, Q. Xv, Y. Wang and J. Tang, Nanotechnology, 2022, 33, 205601 CrossRef PubMed.
- D. Sharma, Sungjemmenla, D. Kumar, D. Takhar, B. Birajdar, V. Kumar and N. Khare, Mater. Sci. Eng., B, 2023, 297, 116769 CrossRef CAS.
- C. Li, T. Hang, H. Zhou, P. Ge, S. Li and X. D. Zhu, Inorg. Chem. Front., 2023, 10, 3375–3382 RSC.
- D. Rambabu, S. Bhattacharyya, T. Singh, M. L. C and T. K. Maji, Inorg. Chem., 2020, 59, 1436–1443 CrossRef CAS PubMed.
- Y. Y. Wang, X. Y. Ji, M. Yu and J. Tao, Mater. Chem. Front., 2021, 5, 7796–7807 RSC.
- Z. Li, J. G. Li, X. Ao, H. Sun, H. Wang, M. F. Yuen and C. Wang, Electrochim. Acta, 2020, 334, 135638 CrossRef CAS.
- T. M. H. Nguyen and C. W. Bark, ACS Omega, 2020, 5, 2280–2286 CrossRef CAS PubMed.
- Y. N. Zhang, B. Li, L. Fu, Q. Li and L. W. Yin, Electrochim. Acta, 2020, 330, 135280 CrossRef CAS.
- D. Shen, A. Pang, Y. Li, J. Dou and M. Wei, Chem. Commun., 2018, 54, 1253–1256 RSC.
- J. Wu, X. Liang, J. Liu, L. yu, F. Wang, T. Wang, Y. Sun, X. Zhou, X. yuan Liu, H. Lin, Q. Zhu, H. Wang, G. Li and H. Hu, Adv. Funct. Mater., 2024, 34, 2407381 CrossRef CAS.
- D. Kim, D. K. Lee, S. M. Kim, W. Park and U. Sim, Materials, 2020, 13, 210 CrossRef CAS PubMed.
- A. Nikolskaia, M. Vildanova, S. Kozlov, N. Tsvetkov, L. Larina and O. Shevaleevskiy, Sustainability, 2020, 12, 788 Search PubMed.
- J. Ma, Z. Lin, X. Guo, L. Zhou, J. Su, C. Zhang, Z. Yang, J. Chang, S. Liu and Y. Hao, Sol. RRL, 2019, 3, 1900096 CrossRef.
- U. J. Ryu, S. Jee, J. S. Park, I. K. Han, J. H. Lee, M. Park and K. M. Choi, ACS Nano, 2018, 12, 4968–4975 CrossRef CAS PubMed.
- L. K. Ono, S. R. Raga, M. Remeika, A. J. Winchester, A. Gabe and Y. Qi, J. Mater. Chem. A, 2015, 3, 15451–15456 RSC.
- T. Shi, J. Chen, J. Zheng, X. Li, B. Zhou, H. Cao and Y. Wang, Sci. Rep., 2016, 6, 39132 CrossRef CAS PubMed.
- S. Guarnera, A. Abate, W. Zhang, J. M. Foster, G. Richardson, A. Petrozza and H. J. Snaith, J. Phys. Chem. Lett., 2015, 6, 432–437 CrossRef CAS PubMed.
- S. Zhang, T. Duan, D. Yan, H. Yang, L. Cheng, Y. Pei, K. You and H. Luo, Chem. Eng. J., 2024, 486, 150176 CrossRef CAS.
- M. Li, D. Xia, Y. Yang, X. Du, G. Dong, A. Jiang and R. Fan, Adv. Energy Mater., 2018, 8, 1702052 CrossRef.
- S. Wang, W. Yuan and Y. S. Meng, ACS Appl. Mater. Interfaces, 2015, 7, 24791–24798 CrossRef CAS PubMed.
- J. Wang, J. Zhang, Y. Yang, S. Gai, Y. Dong, L. Qiu, D. Xia, X. Fan, W. Wang, B. Hu, W. Cao and R. Fan, ACS Appl. Mater. Interfaces, 2021, 13, 5235–5244 CrossRef CAS.
- B. Xu, Z. Zhu, J. Zhang, H. Liu, C. C. Chueh, X. Li and A. K. Y. Jen, Adv. Energy Mater., 2017, 7, 1700683 CrossRef.
- X. Zhou, L. Qiu, R. Fan, H. Ye, C. Tian, S. Hao and Y. Yang, J. Power Sources, 2020, 473, 228556 CrossRef CAS.
- L. Valenzano, B. Civalleri, S. Chavan, S. Bordiga, M. H. Nilsen, S. Jakobsen, K. P. Lillerud and C. Lamberti, Chem. Mater., 2011, 23, 1700–1718 CrossRef CAS.
- S. Yuan, J. S. Qin, C. T. Lollar and H. C. Zhou, ACS Cent. Sci., 2018, 4, 440–450 CrossRef CAS PubMed.
- Y. Bai, Y. Dou, L. H. Xie, W. Rutledge, J. R. Li and H. C. Zhou, Chem. Soc. Rev., 2016, 45, 2327–2367 RSC.
- M. Li, D. Xia, A. Jiang, X. Du, X. Fan, L. Qiu, P. Wang, R. Fan and Y. Yang, Energy Technol., 2019, 7, 1900027 CrossRef.
- X. Zhou, L. Qiu, R. Fan, J. Zhang, S. Hao and Y. Yang, Nano-Micro Lett., 2020, 12, 80 CrossRef CAS PubMed.
- S. Yoon, S. J. Kim, H. S. Kim, J. S. Park, I. K. Han, J. W. Jung and M. Park, Nanoscale, 2017, 9, 16305–16312 RSC.
| This journal is © The Royal Society of Chemistry 2025 |





