Catalytic value addition of glycerol to lactic acid by chromium chloride and its corresponding pincer complex
Himani
Narjinari
a,
Sunil
Dhole
b and
Akshai
Kumar
 *ac
*ac
aDepartment of Chemistry, Indian Institute of Technology Guwahati, Guwahati, 781039, Assam, India. E-mail: akshaikumar@iitg.ac.in
bChemDist Group of Companies, Plot No 144 A, Sector 7, PCNTDA Bhosari, Pune – 411026, Maharashtra, India
cCentre for Nanotechnology, Indian Institute of Technology Guwahati, Guwahati, 781039, Assam, India
First published on 14th October 2025
Abstract
The 3d-metal salt chromium(III) chloride (CrCl3·6H2O) and its corresponding pincer-Cr complexes were utilized to bring about the catalytic transfer dehydrogenation of glycerol (GLY) to lactic acid (LA) in the presence of a sacrificial hydrogen acceptor, acetone. Among the catalysts that were screened, the (Cy2NNN)CrCl3 (0.5 mol%) catalyzed net transfer dehydrogenation of glycerol in the presence of 1.1 equivalents of NaOH at 160 °C afforded 94% lactic acid at 98% selectivity along with another value-added product isopropanol (IPA). Homogeneous molecular nature of the active catalyst in the reaction mixture was evident from the retention of catalytic activity in the presence of catalyst poisons such as PPh3, CS2 and Hg. These homogeneous molecular species are likely to be based on Cr(II) as inferred from detailed mechanistic investigations involving EPR analysis, HRMS studies and Evan's magnetic moment experiments. Isotope labelling experiments indicate a KIE of 1.3.
Introduction
The rapid depletion of fossil-fuel reserves has led to the increased utilization of biodiesel.1 Biodiesel production is accompanied by a significant amount of waste generation such as glycerol (GLY).2 This adds significantly to the global reservoirs already burdened by the glycerol contribution from a large number of industries involving cellulose hydrogenolysis3,4 and fermentation5 processes. Notably, GLY can serve as a starting material for numerous forms of specialty chemicals like lactic acid (LA), dihydroxyacetone and propanediol.6–8 GLY can also act as a potent hydrogen source as well as solvent in various metal aided catalysis owing to the presence of three hydroxyl groups.2,9,10 LA has a plethora of applications in diverse industries, which includes cosmetics, pharmaceuticals, food, green solvent production, biodegradable plastics, surfactants, chemical production and polylactic acid production (PLA).11 Traditionally, GLY to LA conversion is carried out in two pathways which includes bacterial fermentation (via bioconversion)12 and heterogeneous catalysis.3,8b,13 Special conditions required for bacterial survival apart from challenging product separation steps are the typical challenges associated with these classical routes to LA.13m Conversely, the use of homogeneous catalysts demands milder reaction conditions and hence provides better reactivity as well as selectivity.13d,n,14–22The Ir(I)–NHC catalyzed glycerol dehydrogenation in an acceptorless fashion was first demonstrated by Crabtree, where an impressive yield of LA (90%) was obtained at a very high selectivity (95%) with a high turnover number of 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100.14 Tu then utilized a copolymer based Ir–NHC complex to perform the glycerol dehydrogenation to LA where a very high yield of LA (98%) was observed with a turnover number of 3266.15 Williams obtained a very high turnover number of 1
100.14 Tu then utilized a copolymer based Ir–NHC complex to perform the glycerol dehydrogenation to LA where a very high yield of LA (98%) was observed with a turnover number of 3266.15 Williams obtained a very high turnover number of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 057
057![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 172 with a good yield of LA when glycerol was treated with an Ir(I) complex based on a bidentate (pyridylmethyl)imidazolium carbene ligand.16 Voutchkova-Kostal introduced highly charged sulfonate functionalization to Ir(I), Ir(III) and Ru(II) NHC complexes which rendered a turnover number of 45
172 with a good yield of LA when glycerol was treated with an Ir(I) complex based on a bidentate (pyridylmethyl)imidazolium carbene ligand.16 Voutchkova-Kostal introduced highly charged sulfonate functionalization to Ir(I), Ir(III) and Ru(II) NHC complexes which rendered a turnover number of 45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 592 under microwave conditions.17a Very recently, Li studied the conversion of glycerol to lactic acid in water in high yields (ca. 99%) and with excellent selectivity (ca. 99%) that was catalyzed by a water-soluble piano-stool iridium complex.17b Very recently, Kumar and Weller have reported PNP-Ir catalyzed glycerol to LA transformation, where 96% yield of LA was achieved at a high selectivity of 99% with a TON of 65
592 under microwave conditions.17a Very recently, Li studied the conversion of glycerol to lactic acid in water in high yields (ca. 99%) and with excellent selectivity (ca. 99%) that was catalyzed by a water-soluble piano-stool iridium complex.17b Very recently, Kumar and Weller have reported PNP-Ir catalyzed glycerol to LA transformation, where 96% yield of LA was achieved at a high selectivity of 99% with a TON of 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000.18a
000.18a
In the first report with catalytic systems based on Ru, Beller reported PNP pincer-Ru catalysed glycerol to LA transformation with an LA yield of 67% with 67% selectivity with a high turnover number of 270![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000.18b We have demonstrated the activity of 2,6-bis(benzimidazole-2-yl) pyridine based pincer-Ru towards dehydrogenating glycerol to LA. Under the optimized conditions, up to 90% yield of LA could be obtained with 98% selectivity.19a Recently, the research group of Daw have demonstrated that 78% yield of lactate was obtained from a mixture of glycerol, ethylene glycol and methanol (1
000.18b We have demonstrated the activity of 2,6-bis(benzimidazole-2-yl) pyridine based pincer-Ru towards dehydrogenating glycerol to LA. Under the optimized conditions, up to 90% yield of LA could be obtained with 98% selectivity.19a Recently, the research group of Daw have demonstrated that 78% yield of lactate was obtained from a mixture of glycerol, ethylene glycol and methanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) in the presence of NNN pincer-Ru (1 mol%) and KOH (3.5 equivalents) in t-BuOH (1 mL) at 120 °C in 72 h.19b Thus, it is evident that the catalytic systems based on Ir and Ru have enjoyed great success in accomplishing the dehydrogenative transformation of glycerol to lactic acid. However, in recent times, significant emphasis has been laid to accomplishing these transitions using earth-abundant first-row transition elements (Fig. 1).
5) in the presence of NNN pincer-Ru (1 mol%) and KOH (3.5 equivalents) in t-BuOH (1 mL) at 120 °C in 72 h.19b Thus, it is evident that the catalytic systems based on Ir and Ru have enjoyed great success in accomplishing the dehydrogenative transformation of glycerol to lactic acid. However, in recent times, significant emphasis has been laid to accomplishing these transitions using earth-abundant first-row transition elements (Fig. 1).
 | ||
| Fig. 1 Homogeneous 3d-metal based molecular catalytic systems reported for the transformation of GLY to LA. | ||
The first report on the use of a 3d metal for the GLY to LA transformation again came from Crabtree who along with Hazari performed this reaction utilizing a PNP pincer-Fe complex which afforded a turnover number of 880 at 20% conversion of glycerol and 80% selectivity towards LA.20a Fu have reported a highly reactive pincer-Mn complex, which afforded high yield of LA (96%) with high selectivity (96%).20b Very recently, we have reported a catalytic system based on a simple base metal salt CoCl2 which could accomplish the glycerol dehydrogenation to LA at 93% yield with a selectivity of 96% in the presence of acetone as a hydrogen acceptor.22a The CoCl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22a based system was one of the very few examples of catalysts derived from earth-abundant, readily available 3d metals apart from the report on catalytic systems based on pincer-Mn by Fu20b and pincer-Fe by Crabtree and Hazari.20a In our recent report, we have demonstrated (Ph2NNN)MnCl2 (0.5 mol%) catalyzed transfer dehydrogenation of glycerol in the presence of acetone, where we could achieve 92% yield of LA with 99% selectivity in the presence of 1 equivalent NaOH at 140 °C.21a We have also accomplished this reaction using an Fe complex based on the same bis(imino)pyridine ligand where excellent yield of LA (91%) at 99% selectivity was afforded in the presence of 1.2 equivalents NaOH at 140 °C.21b To the best of our knowledge, there are no other reports on homogeneous 3d-metal GLY dehydrogenation catalytic systems reported for the production of LA.
22a based system was one of the very few examples of catalysts derived from earth-abundant, readily available 3d metals apart from the report on catalytic systems based on pincer-Mn by Fu20b and pincer-Fe by Crabtree and Hazari.20a In our recent report, we have demonstrated (Ph2NNN)MnCl2 (0.5 mol%) catalyzed transfer dehydrogenation of glycerol in the presence of acetone, where we could achieve 92% yield of LA with 99% selectivity in the presence of 1 equivalent NaOH at 140 °C.21a We have also accomplished this reaction using an Fe complex based on the same bis(imino)pyridine ligand where excellent yield of LA (91%) at 99% selectivity was afforded in the presence of 1.2 equivalents NaOH at 140 °C.21b To the best of our knowledge, there are no other reports on homogeneous 3d-metal GLY dehydrogenation catalytic systems reported for the production of LA.
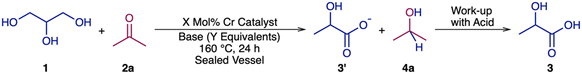
Taking a cue from the previous success on achieving alcohol dehydrogenation using commercially available chromium(III) chloride22b and its corresponding pincer complexes,22b,j we attempted the conversion of GLY to LA using CrCl3·6H2O and its corresponding pincer-Cr complexes based on bis(imino)pyridine ligands.22b,j To our delight, in the presence of acetone, we could achieve LA in 94% yield at a selectivity of 98% along with the formation of isopropanol as a net transfer dehydrogenation product in the (Cy2NNN)CrCl3 catalyzed reaction of glycerol with acetone at 160 °C after 24 h.
Results and discussion
Studies on the pincer-Cr catalyzed net transfer dehydrogenation of glycerol to lactic acid using acetone
For the proposed studies on the net transfer dehydrogenation of GLY 1 to LA 3 using acetone 2a, CrCl3·6H2O and its corresponding NNN pincer-Cr complexes based on bis(imino)pyridine ligands22b,j of the type (R2NNN)CrCl3 (5a; R = Ph, 5b; R = Cy, 5c; R = iPr and 5d; R = tBu) were used as potential catalysts. During the course of the synthesis of the considered pincer-Cr complexes using our previously reported protocol, we were able to obtain crystals of 5a which were suitable for single-crystal XRD analysis (Table S1). Green crystal was obtained for complex 5a in a solution of MeOH and H2O via slow evaporation. SC-XRD analysis of 5a revealed that Cr is in an octahedral environment, to which the pincer ligand is bound in meridional geometry with one chloride trans to pyridyl-N and the other two chlorides are trans to each other (Fig. 2b). Complex 5a has crystallized in the P21/n space group. The Cr–N(pyridyl) bond distance is 1.994(8) Å (Table S1). Both the Cr–N(imine) bonds are comparable (2.108(9) Å and 2.099(8) Å) in length with the pyridyl(N)–Cr–N(imine) bond angles of 77.0(4) and 77.4(4), respectively. | ||
| Fig. 2 (a) The considered catalysts 5a–d. (b) The molecular structure of 5a is provided as ORTEP drawn at 50% probability. All of the hydrogen atoms on 5a are omitted for the sake of clarity. | ||
The catalytic investigations were initiated using 5 mmol of glycerol 1, 5 mmol of acetone 2a, 0.5 mol% CrCl3·6H2O and KOH (0.5 equivalents with respect to glycerol) where 25% glycerol conversion was observed that resulted in 23% lactate 3′ with 92% selectivity at 160 °C in 24 hours (entry 1, Table 1). A higher yield of 3′ (40%) was observed at a higher KOH loading of 0.75 equivalents (entry 2, Table 1). In the presence of one equivalent of KOH, 50% yield of lactate 3′ was obtained at 98% selectivity (entry 3, Table 1).
| Entry | Cr catalyst (X mol%) | Base (Y equivalents) | % conversionb1 | % yieldb3′ | % selectivityc3′ | % yieldb4a | |
|---|---|---|---|---|---|---|---|
| a Reaction conditions: 5 mmol glycerol, X mol% Cr catalyst, base (Y equivalents with respect to glycerol), 5 mmol 2a, at 160 °C, in a 15 mL pressure tube, under air. b Determined from 1H NMR analysis using sodium acetate as the standard. c Selectivity of lactic acid = (yield of lactic acid/conversion of glycerol) × 100. d Average of two runs. e Reaction was done for 96 h. f 0.555 mL acetone was used (1.5 equivalents with respect to glycerol). g Reaction was done at 140 °C. h Reaction was done at 120 °C. i Reaction was done at 100 °C. | |||||||
| 1 | CrCl3·6H2O (0.5) | KOH | 0.5 | 25% | 23% | 92% | 19% |
| 2 | CrCl3·6H2O (0.5) | KOH | 0.75 | 40% | 40% | 100% | 38% |
| 3 | CrCl3·6H2O (0.5) | KOH | 1 | 51% | 50% | 98% | 42% |
| 4d | CrCl3·6H2O (0.5) | KOH | 1.1 | 62 ± 2% | 62 ± 1% | >99% | 55 ± 1% |
| 5d | CrCl3·6H2O (0.5) | KOH | 1.2 | 62 ± 2% | 61 ± 3% | 98% | 57 ± 1% |
| 6d | CrCl3·6H2O (0.5) | KOH | 1.3 | 67 ± 1% | 66 ± 1% | 98% | 60 ± 2% |
| 7 | CrCl3·6H2O (0.75) | KOH | 1.1 | 70% | 68% | 97% | 68% |
| 8 | CrCl3·6H2O (0.25) | KOH | 1.1 | 63% | 62% | 98% | 54% |
| 9 | CrCl3·6H2O (0.1) | KOH | 1.1 | 58% | 58% | 100% | 51% |
| 10 | CrCl3·6H2O (0.075) | KOH | 1.1 | 55% | 53% | 96% | 49% |
| 11 | CrCl3·6H2O (0.05) | KOH | 1.1 | 55% | 54% | 98% | 40% |
| 12 | CrCl3·6H2O (0.025) | KOH | 1.1 | 60% | 58% | 97% | 48% |
| 13 | CrCl3·6H2O (0.01) | KOH | 1.1 | 55% | 54% | 98% | 54% |
| 14 | CrCl3·6H2O (0.0075) | KOH | 1.1 | 55% | 55% | 100% | 45% |
| 15 | CrCl3·6H2O (0.005) | KOH | 1.1 | 51% | 50% | 98% | 38% |
| 16 | CrCl3·6H2O (0.0025) | KOH | 1.1 | 50% | 49% | 98% | 31% |
| 17e | CrCl3·6H2O (0.0025) | KOH | 1.1 | 60% | 59% | 98% | 53% |
| 18d | CrCl3·6H2O (0.5) | NaOH | 1.1 | 88 ± 2% | 84 ± 1% | 95% | 67 ± 1% |
| 19f | CrCl3·6H2O (0.5) | NaOH | 1.1 | 82% | 81% | 99% | 52% |
| 20d | CrCl3·6H2O (0.5) | NaOtBu | 1.1 | 40 ± 1% | 39 ± 1% | 99% | 5 ± 1% |
| 21d | CrCl3·6H2O (0.5) | KOtBu | 1.1 | 27 ± 1% | 26 ± 1% | 99% | 21 ± 1% |
| 22d | CrCl3·6H2O (0.5) | NaOEt | 1.1 | 26 ± 1% | 26 ± 1% | >99% | 24 ± 1% |
| 23 | CrCl3·6H2O (0.5) | Na2CO3 | 1.1 | Trace | Trace | Trace | Trace |
| 24 | CrCl3·6H2O (0.5) | K2CO3 | 1.1 | Trace | Trace | Trace | Trace |
| 25 | CrCl3·6H2O (0.5) | NaHCO3 | 1.1 | 0 | 0 | 0 | 0 |
| 26 | CrCl3·6H2O (0.5) | KHCO3 | 1.1 | 0 | 0 | 0 | 0 |
| 27 | 5a (0.5) | NaOH | 1.1 | 92% | 91% | 99% | 67% |
| 28 | 5b (0.5) | NaOH | 1.1 | 96 ± 1% | 94 ± 1% | 98% | 79 ± 1% |
| 29 | 5c (0.5) | NaOH | 1.1 | 86% | 84% | 98% | 53% |
| 30 | 5d (0.5) | NaOH | 1.1 | 78% | 75% | 96% | 43% |
| 31 | 5b (0.75) | NaOH | 1.1 | 87% | 86% | 99% | 76% |
| 32 | 5b (0.25) | NaOH | 1.1 | 75% | 73% | 95% | 38% |
| 33 | 5b (0.05) | NaOH | 1.1 | 68% | 67% | 98% | 28% |
| 34 | 5b (0.005) | NaOH | 1.1 | 61% | 57% | 91% | 26% |
| 35 | 5b (0.00125) | NaOH | 1.1 | 56% | 53% | 95% | 13% |
| 36g | 5b (0.5) | NaOH | 1.1 | 73% | 72% | 99% | 47% |
| 37h | 5b (0.5) | NaOH | 1.1 | 63% | 61% | 97% | 48% |
| 38i | 5b (0.5) | NaOH | 1.1 | 26% | 24% | 92% | 20% |
| 39 | No Catalyst | NaOH | 1.1 | 21% | 19% | 90% | Trace |
The yield of lactate further increased to 62 ± 1% when 1.1 equivalents of KOH was used (entry 4, Table 1). On further increasing the equivalents of the base, comparable yield of lactate 3′ was observed (entries 5 and 6, Table 1). Further optimization was hence carried out using 1.1 equivalents of KOH. While increasing the loading of CrCl3·6H2O to 0.75 mol% led to comparable yield of lactate 3′ (68%, entry 7, Table 1), further lowering the catalyst loading resulted in lactate yields that were in the range of 60–50% after 24 hours of reaction (entries 8–16, Table 1). Notably, with a very low loading of 0.0025 mol% CrCl3·6H2O, 59% yield of lactate 3′ which corresponds to an unprecedented turnover number of 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 TON was obtained in 96 hours (entry 17, Table 1).
600 TON was obtained in 96 hours (entry 17, Table 1).
To investigate the effect of various bases, the GLY to LA transformation reaction was done using 5 mmol glycerol, 5 mmol acetone, 0.5 mol% CrCl3·6H2O, and 1.1 equivalents of base at 160 °C for 24 hours (entries 18–26, Table 1). A very good yield of lactate 3′ (ca. 84 ± 1%, entry 18, Table 1) was obtained in the presence of NaOH. Use of NaOtBu gave lower yield of lactate 3′ (39 ± 1%, entry 20, Table 1). Both KOtBu and NaOEt have shown poor efficiency giving rise to similar yield of 3′ (26 ± 1%, entries 21 and 22, Table 1). The carbonate and bicarbonate salts of neither sodium nor potassium were active (entries 23–26, Table 1). Increasing the amount of acetone hardly improved the productivity of 3′ (entry 18 vs. 19, Table 1).
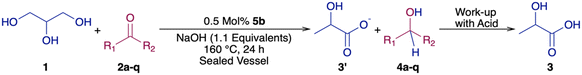
Under the optimized conditions (entry 18, Table 1), the transfer dehydrogenation of glycerol with acetone was repeated using pincer-Cr complexes based on bis(imino)pyridine ligands22b,j of the type (R2NNN)CrCl3 (5a; R = Ph, 5b; R = Cy, 5c; R = iPr and 5d; R = tBu) which were previously reported by our group.22b,j To our delight, complexes 5a and 5b have shown better reactivity than the precursor CrCl3·6H2O in terms of productivity of lactate 3′ (entry 18 vs. entries 27 and 28, Table 1). In particular, 91% lactate 3′ was obtained with 99% selectivity, when glycerol (5 mmol) was treated with 0.5 mol% 5a in the presence of acetone (5 mmol) and NaOH (1.1 equivalents with respect to glycerol) (entry 27, Table 1). Under similar conditions, slightly better reactivity was demonstrated by 5b compared to 5a (entry 27 vs. entry 28, Table 1) where 94 ± 1% lactate 3′ yield was obtained with 98% selectivity along with 79 ± 1% of isopropanol. Meanwhile, slightly lower yield of lactate (84%, entry 29, Table 1) but still at high selectivity (98%) was observed upon the use of catalyst 5c under identical conditions.
The optimization of the loading of 5b was then investigated. On increasing the loading of 5b to 0.75 mol%, 86% yield of lactate 3′ with 99% selectivity was obtained, which is slightly lower compared to that of 0.5 mol% 5b (compared entry 31 with 27, Table 1) which may be attributed to the lower solubility of the higher amount of the catalyst. On lowering the loading of 5b to 0.25 mol%, a decrement in the yield of lactate 3′ was observed (73% with 95% selectivity, entry 32, Table 1). Further sequential lowering of 5b loading led to gradual decrement in lactate 3′ yield however with good turnover numbers (entries 33–35, Table 1).
To investigate the effect of the temperature, the reaction was performed at lower temperatures also. On lowering the temperature from 160 °C to 140 °C, a lower yield of lactate 3′ (72%, entry 36, Table 1) was obtained which drops to 61% at 120 °C (entry 37, Table 1). The yield of lactate 3′ further dropped to 24% at 100 °C (entry 38, Table 1). One would observe that in all entries in Table 1, the yield of IPA 4a is slightly lower than 3′, which apparently can be attributed to the secondary dehydrogenation of 4a back to 2a along with the generation of hydrogen. Accordingly, hydrogen was detected in GC measurements for representative examples (see Fig. S1a–f for H2 detected from entries 4, 28, and 31–34, Table 1). Alternatively, the possibility of a competing aldol reaction of 2a with the corresponding aldol products acting as hydrogen acceptors can also explain the lower yield of 4a. A similar argument could explain the lower yields of 4a–4q in Table 2 relative to 3′.
| Entry | Acceptor | % conversionb1 | % yieldb3′ | % selectivityc3′ | % yieldb4a–4q |
|---|---|---|---|---|---|
| a Reaction conditions: 5 mmol glycerol, 0.5 mol% 5b, NaOH (1.1 equivalents with respect to glycerol), 5 mmol acetone (1 equivalent with respect to glycerol), at 160 °C, in a 15 mL pressure tube, under air. b Determined from 1H NMR analysis using sodium acetate as the standard. c Selectivity of lactic acid = (yield of lactic acid/conversion of glycerol) × 100. d Average of two runs. e Reaction was done in the absence of a base. f Reaction was done in the absence of 5b. g Reaction was done in the absence of any acceptor. | |||||
| 1d | Acetone (2a) | 96 ± 1% | 94 ± 1% | 98% | 67 ± 1% |
| 2e | Acetone (2a) | 0% | 0% | 0% | 0% |
| 3f | Acetone (2a) | 18% | 18% | 100% | 0% |
| 4 | Acetophenone (2b) | 76% | 74% | 97% | 72% |
| 5 | Benzaldehyde (2c) | 31% | 30% | 97% | 49% |
| 6 | 4′-Chloroacetophenone (2d) | 78% | 76% | 97% | 32% |
| 7 | 4′-Methylacetophenone (2e) | 90% | 89% | 93% | 84% |
| 8 | Cyclopentanone (2f) | 74% | 72% | 97% | 45% |
| 9 | 4′-Nitroacetophenone (2g) | 32% | 18% | 56% | Trace |
| 10 | Benzophenone (2h) | 54% | 50% | 93% | 47% |
| 11 | 3′-Methoxyacetophenone (2i) | 75% | 73% | 97% | 56% |
| 12 | 4-Acetylpyridine (2j) | 48% | 45% | 94% | 6% |
| 13 | 4′-Bromoacetophenone (2k) | 87% | 84% | 96% | 22% |
| 14 | 4′-Fluoroacetophenone (2l) | 7% | 6% | 86% | 0% |
| 15 | 4′-CF3-Acetophenone (2m) | 47% | 45% | 96% | 15% |
| 16 | 4′-Methoxyacetophenone (2n) | 74% | 73% | 98% | 51% |
| 17 | 4′-tert-Butylacetophenone (2o) | 25% | 23% | 92% | 0% |
| 18 | 2-Acetylthiophene (2p) | 65% | 62% | 95% | 49% |
| 19 | 4′-Hydroxyacetophenone (2q) | 15% | 12% | 50% | Trace |
| 20g | No acceptor | 10% | 8% | 80% | 0% |
Under the optimized conditions but in the absence of a base, no reactivity was observed (entry 2, Table 2), while in the absence of 5b, only 18% yield of 3′ was observed with 100% selectivity towards 3′ (entry 3, Table 2). The GLY to LA transformation was then performed in the presence of various acceptors under the optimized reaction conditions (entry 28, Table 1), to understand the effect of acceptors on the productivity of lactate 3′ (Table 2).
In the presence of acetophenone 2b as an acceptor, moderate yield of lactate 3′ (74%) was observed (entry 4, Table 2). In the presence of benzaldehyde 2c, poorer yield of lactate 3′ was obtained (30%, entry 5, Table 2). Hence, further studies were continued with various derivatives of acetophenone.
A very poor yield of lactate 3′ (6%) was obtained when 2l with a strong electron withdrawing p-fluoro group was employed as an acceptor (entry 14, Table 2). The yield of 3′ improved to 45% when the p-fluoro group was replaced by a p-trifluoromethyl group in 2m (entry 15, Table 2). In the presence of 2d containing a poor electron-withdrawing chloro group, comparable yield of lactate 3′ (76% at 97% selectivity) was observed in comparison to 2b that results in 70% yield of 3′ (entry 4 vs. entry 6, Table 2). Similarly, 2i containing an poor electron withdrawing methoxy group in the meta position shows comparable reactivity yielding 73% of lactate to 2b (entry 4 vs. entry 11, Table 2). Acceptor 2k with a relatively less electron withdrawing p-bromo substituent resulted in 84% yield of lactate 3′ (entry 13, Table 2). Based on the observation, it is evident that the yield of lactate 3′ systematically decreased on increasing the electron deficiency and followed the trend p-Br > p-Cl ∼ m-OMe > p-CF3 > p-F. When the reaction was performed in the presence of 2e, which contains an electron-donating methyl group, a much higher yield of 89% lactate 3′ with 93% selectivity was obtained (entry 7, Table 2).
When cyclopentanone 2f was used as an acceptor, a lower reactivity was observed when compared to the corresponding activity with another aliphatic acceptor acetone (compared entry 8 with entry 1, Table 2). It might be attributed to the fact that cyclopentanone is more sterically crowded than acetone. Poor yield of lactate 3′ (18%) was observed in the presence of a nitro derivative of acetophenone 2g (entry 9, Table 2) along with the formation of ethylene glycol EG (3%) and formic acid FA (11%), ultimately resulting in very low selectivity towards LA (56%). When benzophenone 2h was used, 50% yield of lactate 3′ was obtained (entry 10, Table 2).
Mechanistic investigation on the pincer-Cr catalyzed net transfer dehydrogenation of glycerol to lactic acid using acetone
To check the homogeneity of the reaction, three well-established catalyst poisons23 involving PPh3, CS2 and mercury were used. Negligible lowering of reactivity towards production of 3′ was observed when the transfer dehydrogenation of GLY catalyzed by CrCl3·6H2O and 5b (compared eqn (1) with eqn (3) and eqn (2) with eqn (4), Scheme 1) was performed in the presence of PPh3. Similar observation was made in the presence of CS2 (compared eqn (1) vs. eqn (5), Scheme 1 and eqn (1) vs. eqn (6), Scheme 1). Considering the fact that PPh3 or CS2 can poison both homogeneous and heterogeneous catalysts,23d a confirmatory mercury poisoning test was also performed, where slight lowering of yield was observed (compared eqn (2) vs. eqn (7), Scheme 1). These observations draw evidence of the reaction being mostly homogeneous. Interestingly, the high reactivity under open vessel conditions (compared entry 4, Table 2vs. eqn (8), Scheme 1) in the presence of acetophenone (the boiling point is 202 °C) under the optimized reaction conditions gives an indication of the involvement of both hydrogenolysis and alcoholysis.From the EPR analysis of the reaction mixture (entry 28, Table 1) at 0 h and 1 h, it is evident that at 0 h Cr(III) species (g = 2.0006)24 is involved, whereas at 1 h Cr(II) species (g = 1.9843)24 is involved (Fig. 3). HRMS analysis (Fig. 4) of the reaction mixture demonstrated the presence of several pincer-Cr mono-glyceroxide and pincer-Cr bis-glyceroxide species. Magnetic moment measurement (via Evan's method)24b of 5b in the absence and presence of a base in 2% tBuOH in D2O (Fig. 5) bodes well with the observation from EPR (Fig. 3) of the reaction mixture. A magnetic moment of 3.42 BM of the reaction mixture before the addition of a base corresponds to Cr(III) species,24b which converts to a Cr(II) species with a magnetic moment of 4.5 BM in the presence of a base.24b Subjecting a mixture of glycerol (2.5 mmol) and glycerol-d8 (2.5 mmol) to the optimized reaction conditions (eqn (10), Scheme 1) resulted in a modest KIE of 1.3 along with 46% lactate 3′. This KIE gives an indication of the involvement of C–H activation as a part of the catalytic cycle, but it might not be a part of the rate determining step. Kinetic studies indicate that the reaction demonstrates a first-order dependence of rate on the concentration of 5b and NaOH (Fig. 6). Thus, from the control experiments, it may be inferred that well-defined molecular pincer-Cr(II) complexes are involved in the catalytic cycle with a likely contribution from the alcoholysis and/or a Meerwein–Ponndorf–Verley type pathway (Scheme 2).
 | ||
| Fig. 3 EPR analysis of the reaction mixture obtained from the optimized transfer dehydrogenation of glycerol (entry 28, Table 1 and eqn (2), Scheme 1) (a) at 0 h in the absence of NaOH and (b) at 1 h in the presence of NaOH. The X-band EPR spectra were recorded in methanol on a JESFA200 ESR spectrometer at 78 K with a microwave power of 0.998 mW and a microwave frequency of 9.14 GHz. | ||
 | ||
| Fig. 4 HRMS analysis of the reaction mixture obtained from the optimized transfer dehydrogenation of glycerol (entry 2, Table 2 and eqn (2), Scheme 1) at (a) t = 0 h at room temperature and (b) t = 1 h at 160 °C (also see Fig. S2a–c). | ||
 | ||
| Fig. 5 Magnetic moment determination via Evan's method of a) 5b before addition of NaOH and b) 5b after addition of NaOH at room temperature. | ||
 | ||
| Fig. 6 (a) Time-profile of transfer dehydrogenation of glycerol in an open vessel using glycerol (0.460 g, 5 mmol, 3.3 M), NaOH (0.2198 g, 5.4956 mmol, 3.6 M), acetophenone (1.2 g, 10 mmol, 6.6 M), at 160 °C, in the presence of 5b (0.0013–0.0114 g, 0.003–0.025 mmol, 0.002–0.016 M). (b) Dependence of the initial rate of transfer dehydrogenation of the glycerol reaction on the concentration of 5b (extracted from the data at 15 minutes in Fig. 6a). (c) Dependence of the initial rate of transfer dehydrogenation of the glycerol reaction on the concentration of NaOH (extracted from the data at 15 minutes in Fig. S59. Reaction conditions: glycerol (0.460 g, 5 mmol, 3.3 M), 5b (0.0114 g, 0.025 mmol, 0.016 M), acetophenone (1.2 g, 10 mmol, 6.6 M), at 160 °C, in the presence of NaOH (0.05–0.22 g, 1.25–5.5 mmol, 0.8–3.6 M)). | ||
 | ||
| Scheme 2 Plausible mechanism involved in the 5b catalyzed net transfer dehydrogenation of glycerol to lactic acid. | ||
The first step in the pincer-Cr catalyzed net transfer dehydrogenation of glycerol to lactic acid using acetone involves the generation of the active Cr(II) species 6 from the Cr(III) precursor (5b) in the presence of a base (Scheme 2). Subsequent salt metathesis of 6 with NaOH in the presence of glycerol 1 will generate the alkoxide species 7 along with loss of water. This will be followed by a β-hydride elimination at 7 which will give rise to the hydride species 9viaTS-8 along with the extrusion of a molecule of glyceraldehyde 1′. Glyceraldehyde 1′ will follow a series of base mediated reactions which involves dehydration of 1′ to 10 and a subsequent tautomerization of 10 to 11. Ultimately, a base mediated intramolecular Cannizzaro reaction in 11 leads to the final lactate 3′.
The pincer-Cr-hydride species 9 has three paths (path A–C) at its disposal (Scheme 2). Along path A, the Cr–H bond in 9 would undergo a σ-bond metathesis with the O–H bond of glycerol 1 leading to the regeneration of alkoxide species 7viaTS-12 along with the extrusion of H2. Notably, hydrogen was detected from the headspace of the reaction mixture (eqn (1) and (2), Scheme 1 and Fig. S1a and b). However, entry 20 in Table 2 indicates that the contribution (ca. 8% yield of 3′) from acceptorless path A is very minor.
Along path B, 2a would insert into the Cr–H bond in 9 to give the pincer-Cr isopropoxide 14 while going through TS-13. Species 14 can undergo a σ-bond metathesis (alcoholysis) with the O–H bond of glycerol 1 leading to the regeneration of alkoxide species 7viaTS-15 while generating isopropanol 4a (Scheme 2). Reactivity under open-vessel conditions (eqn (8), Scheme 1) is suggestive of the involvement of the alcoholysis along path B. Alternatively, if the pincer-Cr isopropoxide species 14 undergoes a σ-bond metathesis (hydrogenolysis) with hydrogen, the pincer-Cr hydride 9 would be regenerated along with the formation of 4a. Notably, though hydrogen is detected from the headspace of the reaction mixture (eqn (1) and (2), Scheme 1 and Fig. S1a and b), the hydrogenolysis through path C is unlikely to contribute to the observed catalysis, as independent experiments with molecular hydrogen under the optimized reaction conditions yielded no product (eqn (9), Scheme 1).
In alternative path D, the pincer-Cr glyceroxide species 7 would interact with 2a to initiate a direct transfer of the β-hydrogen via Meerwein–Ponndorf–Verley type TS-17 leading to the extrusion of glyceraldehyde 1′ and formation of pincer-Cr isopropoxide species 14 (Scheme 2). Species 14 upon alcoholysis would regenerate 7 and produce 4a. Notably, while path B has a sequence of β-hydride elimination and insertion steps, path D has a concerted β-hydride transfer step prior to the alcoholysis step that is common to both the paths.
Experimental
Materials and methods
All the complexes were synthesized in anhydrous THF under argon using the Schlenk technique at room temperature. All the reactions were performed in a sealed vessel under air. Kinetic experiments were performed in an open vessel under air. Glycerol, NaOH, KOH, Na2CO3, K2CO3, NaHCO3, and KHCO3 were purchased from Himedia. Deuterated glycerol-d8, D2O, NaOtBu, KOtBu and NaOEt were purchased from Sigma-Aldrich. The ketones were purchased from MERCK or Sigma-Aldrich or SRL or Alfa Aesar or TCI and used as such.Physical measurements
1H, 13C {1H} and 2H NMR were performed either on a Bruker ASCEND 600 operating at 600 MHz for 1H and 2H and 151 MHz for 13C or on a Bruker AVANCE 400 operating at 400 MHz for 1H. Chemical shifts (δ) are reported in ppm, spin–spin coupling constants (J) are expressed in Hz, and other data are reported as follows: s = singlet, d = doublet and q = quartet. HRMS measurements were recorded using an Agilent Accurate-Mass QTOF ESI-MS 6520. The X-band EPR spectra were recorded on a JESFA200 ESR spectrometer.General procedure for transfer dehydrogenation of glycerol to lactic acid using 5b
In a 15 mL sealed tube, glycerol (0.460 g, 5 mmol, 6.8 M) followed by sodium acetate (0.05 g, 0.6095 mmol, 0.8 M), NaOH (0.2198 g, 5.4956 mmol, 7.5 M), 5b (0.0114 g, 0.025 mmol, 0.02 M) and acetone (0.37 mL, 5 mmol, 6.8 M) were added. The reaction mixture was heated in a preheated oil bath at 160 °C for 24 hours. After 24 hours, the reaction mixture was brought to room temperature. Subsequently, 0.5 mL H2O was added to the reaction mixture and was allowed to homogenize under continuous stirring for 10 minutes. An aliquot was drawn and submitted for NMR analysis in D2O as the NMR solvent. The yield of lactate (3′) was determined as 94% from 1H NMR using sodium acetate as an internal standard.General procedure for kinetic studies
Conclusions
Commercially available first-row transition metal salt CrCl3·6H2O and its corresponding pincer complexes in the presence of a sacrificial hydrogen acceptor, acetone, is found to accomplish the transfer dehydrogenation of glycerol (GLY) to lactic acid (LA). In particular, the pincer-Cr complex, (Cy2NNN)CrCl3 (0.5 mol%) was found to efficiently bring about the catalytic net transfer dehydrogenation of glycerol in the presence of 1.1 equivalents of NaOH, at 160 °C to afford 94% lactic acid at 98% selectivity along with another value-added product isopropanol (IPA). The scope of the studies on the GLY to LA conversion under transfer dehydrogenation conditions has been extended to include various carbonyl compounds (16 examples) as acceptors. The acceptor 4′-methylacetophenone with an electron donating methyl group has shown the best result with 89% yield of lactate.While reactions with catalyst poisons such as PPh3, CS2 and elemental mercury revealed the homogeneous molecular nature of the catalyst, EPR studies and magnetic moment measurements using Evan's method revealed that these homogeneous molecular species are likely to be based on Cr(II). Detailed mechanistic studies are indicative of the sole contribution of the alcoholysis path towards the observed reactivity with a KIE of 1.3.
Author contributions
H. N. performed all the experiments. S. D. provided intellectual inputs. A. K. conceptualized the project, analyzed the data and wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI).Supplementary information: detailed experimental procedure, NMR of the reaction mixtures, GC and HRMS analysis (PDF). See DOI: https://doi.org/10.1039/d5cy01041a.
CCDC 2310415 contains the supplementary crystallographic data for this paper.25
Acknowledgements
A. K. is grateful for the grants received from the Science and Engineering Research Board (Grant No. DST-SERB CRG/2022/002354). A. K. is grateful for the funds received from the Ministry of Electronics and Information Technology via the INUP-I2I program (5(1)/2021-NANO) and the SWASTHA COE (5(1)/2022-NANO). A. K. acknowledges the financial support from the Indian Council of Medical Research, New Delhi (5/3/8/20/2019-ITR). The DST-FIST program, NECBH-IITG, the Department of Chemistry at IITG, the Centre for Nanotechnology at IITG and CIF-IITG are acknowledged for various instrumental facilities.Notes and references
- F. Cherubini, Energy Convers. Manage., 2010, 51, 1412–1421 CrossRef CAS.
- R. H. Crabtree, ACS Sustainable Chem. Eng., 2019, 7, 15845–15853 CrossRef CAS.
- P. D. Vaidya and A. E. Rodrigues, Chem. Eng. Technol., 2009, 32, 1463–1469 CrossRef CAS.
- D. M. Alonso, S. G. Wettstein and J. A. Dumesic, Chem. Soc. Rev., 2012, 41, 8075–8098 RSC.
- P. T. Anastas and J. B. Zimmerman, Innovations in Green Chemistry and Green Engineering: Selected Entries from the Encyclopedia of Sustainability Science and Technology, Springer Science & Business Media, 2012 Search PubMed.
- (a) D. B. Lao, A. C. E. Owens, D. M. Heinekey and K. I. Goldberg, ACS Catal., 2013, 3, 2391–2396 CrossRef CAS; (b) D. D. Falcone, J. H. Hack, A. Y. Klyushin, A. Knop-Gericke, R. Schlögl and R. J. Davis, ACS Catal., 2015, 5, 5679–5695 CrossRef CAS; (c) X. Zhao, J. Wang, M. Yang, N. Lei, L. Li, B. Hou, S. Miao, X. Pan, A. Wang and T. Zhang, ChemSusChem, 2017, 10, 819–824 CrossRef CAS PubMed; (d) Q. Sun, S. Wang and H. Liu, ACS Catal., 2017, 7, 4265–4275 CrossRef CAS; (e) M. Sherbi, A. Wesner, V. K. Wisniewski, A. Bukowski, H. Velichkova, B. Fiedler and J. Albert, Catal. Sci. Technol., 2021, 11, 6649–6653 RSC.
- (a) R. M. Painter, D. M. Pearson and R. M. Waymouth, Angew. Chem., Int. Ed., 2010, 49, 9456–9459 CrossRef CAS PubMed; (b) C. Crotti, J. Kašpar and E. Farnetti, Green Chem., 2010, 12, 1295–1300 RSC; (c) K. Chung, S. M. Banik, A. G. De Crisci, D. M. Pearson, T. R. Blake, J. V. Olsson, A. J. Ingram, R. N. Zare and R. M. Waymouth, J. Am. Chem. Soc., 2013, 135, 7593–7602 CrossRef CAS PubMed; (d) S.-S. Liu, K.-Q. Sun and B.-Q. Xu, ACS Catal., 2014, 4, 2226–2230 CrossRef CAS; (e) N. Gupta, O. Khavryuchenko, A. Villa and D. Su, ChemSusChem, 2017, 10, 3030–3034 CrossRef CAS PubMed.
- (a) C. H. Lam, A. J. Bloomfield and P. T. Anastas, ChemSusChem, 2017, 19, 1958–1968 CAS; (b) Z. Jiang, Z. Zhang, T. Wu, P. Zhang, J. Song, C. Xie and B. Han, Chem. – Asian J., 2017, 12, 1598–1604 CrossRef CAS PubMed; (c) D. Ainembabazi, K. Wang, M. Finn, J. Ridenour and A. Voutchkova-Kostal, Green Chem., 2020, 22, 6093–6104 RSC.
- (a) M. Pagliaro, R. Ciriminna, H. Kimura, M. Rossi and C. Della Pina, Angew. Chem., Int. Ed., 2007, 46, 4434–4440 CrossRef CAS PubMed; (b) F. Jérôme, Y. Pouilloux and J. J. C. C. Barrault, ChemSusChem, 2008, 1, 586–613 CrossRef PubMed; (c) B. Katryniok, H. Kimura, E. Skrzyńska, J.-S. Girardon, P. Fongarland, M. Capron, R. Ducoulombier, N. Mimura, S. Paul and F. Dumeignil, Green Chem., 2011, 13, 1960–1979 RSC; (d) A. Behr, J. Eilting, K. Irawadi, J. Leschinski and F. Lindner, Green Chem., 2008, 10, 13–30 RSC; (e) P. Gallezot, Catal. Today, 2007, 121, 76–91 CrossRef CAS.
- A. Wolfson, C. Dlugy, Y. Shotland and D. Tavor, Tetrahedron Lett., 2009, 50, 5951–5953 CrossRef CAS.
- (a) M. Dusselier, P. Van Wouwe, A. Dewaele, E. Makshina and B. F. Sels, Energy Environ. Sci., 2013, 6, 1415–1442 RSC; (b) A. Komesu, J. A. R. de Oliveira, L. H. da Silva Martins, M. R. W. Maciel and R. Maciel Filho, Bioresour., 2017, 12, 4364–4383 CAS.
- (a) S. J. Sarma, S. K. Brar, E. B. Sydney, Y. Le Bihan, G. Buelna and C. R. Soccol, Int. J. Hydrogen Energy, 2012, 37, 6473–6490 CrossRef CAS; (b) J. Tang, Z. Hu, Y. Pu, X. C. Wang and A. Abomohra, J. Environ. Manage., 2024, 369, 122372 CrossRef CAS PubMed.
- (a) A. C. C. De Souza and J. L. Silveira, Renewable Sustainable Energy Rev., 2011, 15, 1835–1850 CrossRef; (b) G. W. Huber, J. W. Shabaker and J. A. Dumesic, Science, 2003, 300, 2075–2077 CrossRef CAS PubMed; (c) D. L. King, L. Zhang, G. Xia, A. M. Karim, D. J. Heldebrant, X. Wang, T. Peterson and Y. Wang, Appl. Catal., B, 2010, 99, 206–213 CrossRef CAS; (d) S. H. Cho and D. J. J. Moon, Nanosci. Nanotechnol., 2011, 11, 7311–7314 CAS; (e) J. J. Wang, Z. J. Li, X. B. Li, X. B. Fan, Q. Y. Meng, S. Yu, C. B. Li, J. X. Li, C. H. Tung and L. Z. Wu, ChemSusChem, 2014, 7, 1468–1475 CrossRef CAS PubMed; (f) S. H. Siddiki, A. S. Touchy, K. Kon, T. Toyao and K. I. Shimizu, ChemCatChem, 2017, 9, 2816–2821 CrossRef CAS; (g) G. Dodekatos, S. Schünemann and H. Tüysüz, ACS Catal., 2018, 8, 6301–6333 CrossRef CAS; (h) W. Oberhauser, C. Evangelisti, A. Liscio, A. Kovtun, Y. Cao and F. J. Vizza, Catal., 2018, 368, 298–305 CrossRef CAS; (i) S. Feng, K. Takahashi, H. Miura and T. Shishido, Fuel Process. Technol., 2020, 197, 106202 CrossRef CAS; (j) R. Palacio, D. López and D. J. Hernández, J. Nanopart. Res., 2019, 21, 1–13 CrossRef CAS; (k) C. S. Lee, M. K. Aroua, W. A. Wan Daud, P. Cognet, Y. Pérès and M. A. Ajeel, Front. Chem., 2019, 7, 110 CrossRef CAS PubMed; (l) S. A. Zavrazhnov, A. L. Esipovich, S. Y. Zlobin, A. S. Belousov and A. V. Vorotyntsev, Catalysts, 2019, 9, 231 CrossRef; (m) M. A. Abdel-Rahman and K. J. Sonomoto, Biotechnol., 2016, 236, 176–192 CAS; (n) A. Bisarya, S. Karim, H. Narjinari, A. Banerjee, V. Arora, S. Dhole, A. Dutta and A. Kumar, Chem. Commun., 2024, 60, 4148–4169 RSC.
- L. S. Sharninghausen, J. Campos, M. G. Manas and R. H. Crabtree, Nat. Commun., 2014, 5, 5084 CrossRef CAS PubMed.
- Z. Sun, Y. Liu, J. Chen, C. Huang and T. Tu, ACS Catal., 2015, 5, 6573–6578 CrossRef CAS.
- Z. Lu, I. Demianets, R. Hamze, N. J. Terrile and T. J. A. Williams, ACS Catal., 2016, 6, 2014–2017 CrossRef CAS.
- (a) M. Finn, J. A. Ridenour, J. Heltzel, C. Cahill and A. Voutchkova-Kostal, Organometallics, 2018, 37, 1400–1409 CrossRef CAS; (b) X. Xu, H. You, B. Dong, Y. He and F. Li, Inorg. Chem., 2024, 63, 12929–12934 CrossRef CAS PubMed; (c) K. Wang, J. Horlyck, N. An and A. Voutchkova-Kostal, Green Chem., 2024, 26, 3546 RSC; (d) B. Dong, Y. Ji, F. Zhou, N. T. Tung, Y. Fan and F. Li, J. Catal., 2026, 453, 116442 CrossRef CAS.
- (a) Vikas, L. Kathuria, C. N. Brodie, M. J. Cross, F. A. Pasha, A. S. Weller and A. Kumar, Inorg. Chem., 2025, 64, 3760–3770 CrossRef CAS PubMed; (b) Y. Li, M. Nielsen, B. Li, P. H. Dixneuf, H. Junge and M. Beller, Green Chem., 2015, 17, 193–198 RSC.
- (a) M. Dutta, K. Das, S. J. Prathapa, H. K. Srivastava and A. Kumar, Chem. Commun., 2020, 56, 9886–9889 RSC; (b) S. T. Sahoo, A. Sinku and P. Daw, RSC Adv., 2024, 14, 37082–37086 RSC.
- (a) L. S. Sharninghausen, B. Q. Mercado, R. H. Crabtree and N. Hazari, Chem. Commun., 2015, 51, 16201–16204 RSC; (b) C.-Q. Deng, J. Deng and Y. Fu, Green Chem., 2022, 24, 8477–8483 RSC.
- (a) A. Bisarya, S. Dhole and A. Kumar, Dalton Trans., 2024, 53, 12698–12709 RSC; (b) B. Venkateshappa, A. Bisarya, P. G. Nandi, S. Dhole and A. Kumar, Inorg. Chem., 2024, 63, 15294–15310 CrossRef CAS PubMed.
- (a) H. Narjinari, S. Dhole and A. Kumar, Chem. – Eur. J., 2024, 30, e202302686 CrossRef CAS PubMed; (b) H. Narjinari, N. Tanwar, L. Kathuria, R. V. Jasra and A. Kumar, Catal. Sci. Technol., 2022, 12, 4753–4762 RSC; (c) P. G. Nandi, P. Thombare, S. J. Prathapa and A. Kumar, Organometallics, 2022, 41, 3387–3398 CrossRef CAS; (d) P. G. Nandi, P. Kumar and A. Kumar, Catal. Sci. Technol., 2022, 12, 1100–1108 RSC; (e) K. Islam, Vikas, B. Nag and A. Kumar, ChemCatChem, 2022, e202200440 CrossRef CAS; (f) M. Dutta, H. K. Srivastava and A. Kumar, Comput. Chem., 2021, 42, 1728–1735 CrossRef CAS PubMed; (g) V. Arora, H. Narjinari, P. G. Nandi and A. Kumar, Dalton Trans., 2021, 50, 3394–3428 RSC; (h) V. Arora, H. Narjinari and A. Kumar, Organometallics, 2021, 40, 2870–2880 CrossRef CAS; (i) V. Arora, M. Dutta, K. Das, B. Das, H. K. Srivastava and A. Kumar, Organometallics, 2020, 39, 2162–2176 CrossRef CAS; (j) N. Tanwar, H. Narjinari, H. Sharma, S. Dhole, R. V. Jasra and A. Kumar, Inorg. Chem., 2024, 63, 3005–3018 CrossRef CAS PubMed.
- (a) S. Werkmeister, J. Neumann, K. Junge and M. Beller, Chem. – Eur. J., 2015, 21, 12226–12250 CrossRef CAS PubMed; (b) D. H. Nguyen, Y. Morin, L. Zhang, X. Trivelli, F. Capet, S. Paul, S. Desset, F. Dumeignil and R. M. Gauvin, ChemCatChem, 2017, 9, 2652–2660 CrossRef CAS; (c) L. Chen, S. Ren and X. P. Ye, React. Kinet., Mech. Catal., 2015, 114, 93–108 CrossRef CAS; (d) V. Artero and M. Fontecave, Chem. Soc. Rev., 2013, 42, 2338–2356 RSC.
- (a) D. V. Azamat, A. Dejneka, J. Lančok, L. Jastrabik, V. A. Trepakov, Z. Bryknar, E. V. Neverova and A. G. Badalyan, J. Appl. Phys., 2013, 113, 174106 CrossRef; (b) J. E. Huheey, E. A. Keiter, R. L. Keiter and O. K. Medhi, Inorganic Chemistry: Principles of Structure and Reactivity, Pearson Education, 2006 Search PubMed; (c) N. Y. Garces, N. C. Giles, L. E. Halliburton, K. Nagashio, R. S. Feigelson and P. G. Schunemann, J. Appl. Phys., 2003, 94, 75677570 Search PubMed; (d) A. G. Avanesov, V. V. Badikov and G. S. Shakurov, Solid State Phys., 2003, 45, 1451–1454 CrossRef CAS; (e) J. Telser, L. A. Pardi, J. Krzystek and L.-C. Brunel, Inorg. Chem., 1998, 37, 5769–5775 CrossRef CAS; (f) D. Himmelbauer, B. Stöger, L. F. Veiros, M. Pignitter and K. Kirchner, Organometallics, 2019, 38, 4669–4678 CrossRef CAS.
- CCDC 2310415: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2hk5js.
| This journal is © The Royal Society of Chemistry 2025 |