Multifunctional PNN-NiII pincer catalyst for C–C and C–N bond formation via alkylation, cross-coupling, and hydroamination reactions†
Gazal
Sabharwal
 ,
Khilesh C.
Dwivedi
,
Khilesh C.
Dwivedi
 and
Maravanji S.
Balakrishna
and
Maravanji S.
Balakrishna
 *
*
Phosphorus Laboratory, Department of Chemistry, Indian Institute of Technology Bombay, Powai, Mumbai 400076, India. E-mail: krishna@chem.iitb.ac.in; msb_krishna@iitb.ac.in; Fax: +91 22 5172 3480/2576 7152
First published on 25th July 2025
Abstract
Ligand {(o-PPh2)C6H4C(O)N(H)CH2(C5H4N)} (1) upon reaction with [NiCl2(DME)] afforded [NiCl{(PNN)-κ3-P,N,N}] (2), while its oxide derivative {(o-(O)PPh2)C6H4C(O)N(H)CH2(C5H4N)} (3) yielded NiIII pincer [{NiCl(μ-Cl)2NiCl}{(P(O)NN)2-κ3-O,N,N}] (4). Complex 2 catalyzes diverse transformations, including α-alkylation of ketones, N-alkylation of anilines, and Suzuki–Miyaura cross-coupling of aryl chlorides. Notably, complex 2 promotes the hydroamination of styrene to selectively afford the anti-Markovnikov product, acting as an efficient and versatile catalyst, operating under mild conditions at low loadings (0.5–1 mol%) and delivering high yields (80–99%).
Introduction
Transition metal-catalyzed activation of inert bonds is central to modern catalysis.1–3 Pincer complexes, with their rigid tridentate ligands and meridional geometry, offer a versatile platform for such transformations.4–8 Their tunable steric and electronic properties, along with adaptive behavior upon metal coordination, enable broad catalytic applicability.9–12 While noble metal complexes have traditionally dominated this field,13 recent efforts have focused on developing catalysts based on earth-abundant, cost-effective alternatives.14 Nickel, in particular, stands out due to its accessibility, biocompatibility, and ability to access multiple oxidation states (0, +1, +2, +3 and +4), which supports diverse catalytic cycles.15–17 Its capacity for oxidative addition and its characteristically slow β-hydride elimination-attributable to the high rotational barrier of Ni–C bonds-further augment its catalytic utility.18–23 Herein, we present a PNN-Ni pincer complex 2 that functions as an efficient and versatile catalyst for key transformations, including α-alkylation, N-alkylation, Suzuki–Miyaura cross-coupling, and hydroamination reactions.Results and discussion
Ligand 2-(diphenylphosphaneyl)-N-(pyridine-2-ylmethyl) benzamide, {(o-PPh2)C6H4C(O)N(H)CH2(C5H4N)}(1), (hereafter referred to as “PN(H)N” and its anionic form as “PNN”), was synthesized according to the previously published procedure.24 Ligand 1 upon reacting with [NiCl2(DME)] in the presence of LiHMDS afforded the pincer complex [(NiCl){(PNN)-κ3-P,N,N}](2), which exhibited a singlet at 13.1 ppm in 31P{1H} NMR spectrum. Ligand 1 reacts with excess H2O2 to yield oxide derivative 3, which upon treatment with [NiCl2(DME)] in dichloroethane yielded the dimeric NiIII pincer complex [{NiCl(μ-Cl)2NiCl}{(P(O)NN)2-κ3-O,N,N}] (4) (Scheme 1).Complex 4 displays a νCO at 1706 cm−1, notably higher than that observed for the analogous NiII complex 2 (1615 cm−1), along with a νPO stretch at 1165 cm−1. The molecular structures of compounds 2–4 were confirmed by single-crystal X-ray analysis, as shown in Fig. 1. Selected bond lengths, bond angles, and crystallographic data are summarized in Table S1.† Complex 2 adopts a distorted square planar geometry around the NiII center, while complex 4 features a NiIII center in a distorted octahedral geometry, incorporating two seven- and two five-membered metallacycles, one terminal chloride, and one bridging chloride ligand.
 | ||
| Fig. 1 Molecular structure of compounds 2–4. All hydrogen atoms and solvent molecules have been omitted for clarity. Displacement ellipsoids are drawn at 50% probability level. | ||
Cyclic voltammetry (CV) studies were performed to investigate the redox behaviour of complexes 2 (NiII) and 4 (NiIII). Complex 2 exhibited two quasi-reversible reduction waves corresponding to sequential NiII/NiI and NiI/Ni0 redox couples, typical of square planar NiII complexes. In contrast, complex 4 displayed three distinct redox events, attributed to successive NiIII/NiII, NiII/NiI, and NiI/Ni0 (Fig. S121†). The additional redox wave supports the presence of a higher oxidation state (NiIII) and the possibility of electron delocalization or redox activity across the dimeric core. These results confirm the multivalent redox nature of the Ni centers and support the potential for multielectron redox processes relevant to catalysis.
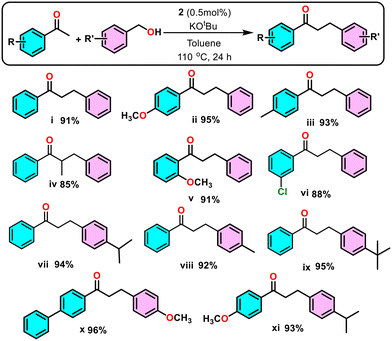
α-Alkylation reaction between various ketones and benzyl alcohol derivatives promoted by nickel pincer complex 2
Carbon–carbon bond formation is a fundamental goal in synthetic organic chemistry.25,26 α-Alkylation of ketone enolates with alkyl halides is a classical method but is often hindered by the toxicity of halide reagents and the generation of stoichiometric inorganic waste.27 In contrast, alcohols offer a more sustainable alternative due to their low toxicity, wide availability, and water as the sole byproduct.28–30 α-Alkylation via hydrogen-borrowing strategies has been widely explored using noble metals (Ir,31 Rh,32 Ru,33 Pd34) and more abundant metals (Mn,35 Fe,36 Co,37 and Cu38), though these often require harsh conditions and high catalyst loadings.39–41 Despite these developments, the application of earth-abundant, non-precious metals such as nickel remains limited.40,42–44 Compared to existing methods, complex 2 catalyzes the α-alkylation of ketones under mild conditions with low catalyst loadings, enabling efficient synthesis of both linear and branched ketones and providing a practical, sustainable alternative to traditional approaches.The α-alkylation of acetophenone with benzyl alcohol was used as a model reaction to optimize conditions (Table 1). Using catalyst 2 with NaOH in toluene gave 86% yield (entry 1). Changing the base to KOtBu improved the yield to 99% (entry 6), while other bases gave lower conversions (entries 2–5). Toluene was the optimal solvent, with other solvents being less effective (entries 7–9). No product formed with nickel salts (entries 10–11), and lower temperature or omission of the catalyst or base suppressed the reaction (entries 12–14). Under optimized conditions, various substituted acetophenones and benzyl alcohols gave the desired products in good yields (85–99%) (Scheme 2).
| Entry | Catalyst | Solvent | Base | Yielda (%) |
|---|---|---|---|---|
| a Yield determined by GC-MS. Acetophenone (0.50 mmol), benzyl alcohol (0.55 mmol), base (0.75 mmol), 2 (0.5 mol%) and solvent (2 mL), 110 °C. b Reaction temp = 80 °C. | ||||
| 1 | 2 | Toluene | NaOH | 86 |
| 2 | 2 | Toluene | KOH | 78 |
| 3 | 2 | Toluene | Cs2CO3 | 67 |
| 4 | 2 | Toluene | K3PO4 | 24 |
| 5 | 2 | Toluene | LiOH | 64 |
| 6 | 2 | Toluene | KOtBu | 99 |
| 7 | 2 | THF | KOtBu | 84 |
| 8 | 2 | Benzene | KOtBu | 87 |
| 9 | 2 | Dioxane | KOtBu | 92 |
| 10 | NiCl2(DME) | Toluene | KOtBu | 0 |
| 11 | Ni(OAc)2·4H2O | Toluene | KOtBu | 0 |
| 12b | 2 | Toluene | KOtBu | 74 |
| 13 | 2 | Toluene | No base | 0 |
| 14 | No catalyst | Toluene | KOtBu | 0 |
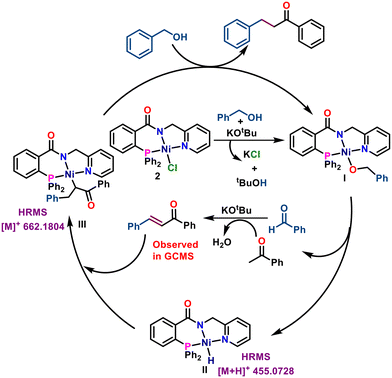
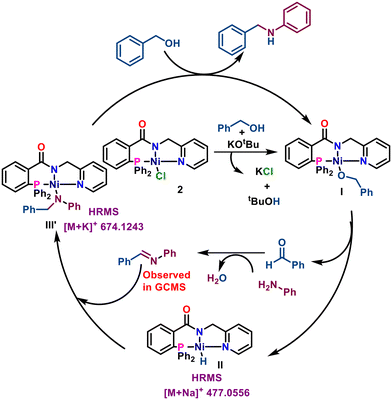
Based on literature precedents,45,46 a plausible mechanism is proposed and outlined in Scheme 3. The reaction begins with the formation of Ni-alkoxide species I from complex 2 and benzyl alcohol in the presence of base. β-Hydride elimination yields Ni–H species II and an aldehyde, which undergoes base-promoted aldol condensation with a ketone to form an α,β-unsaturated ketone (detected by GC-MS). Insertion of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond into Ni–H species II gives intermediate III, which undergoes alcoholysis with benzyl alcohol to regenerate species I and release the α-alkylated ketone.
C bond into Ni–H species II gives intermediate III, which undergoes alcoholysis with benzyl alcohol to regenerate species I and release the α-alkylated ketone.
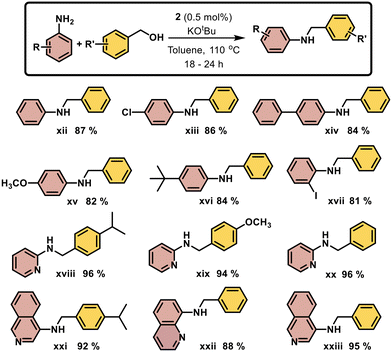
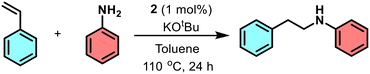
N-Alkylation reaction between various anilines and benzyl alcohol derivatives promoted by nickel pincer complex 2
N-Alkylation of amines is a key transformation in pharmaceuticals, materials science, and agrochemicals. Traditional methods such as nucleophilic substitution, Buchwald–Hartwig coupling, ullmann reactions, and hydroamination, often face issues like low selectivity, over-alkylation, and poor yields, highlighting the need for more efficient and sustainable approaches.47 Transition metal-catalyzed N-alkylation via the borrowing hydrogen strategy has emerged as a highly atom-economical and environmentally benign approach, utilizing alcohols as both alkylating agents and hydrogen source.48,49 While noble metal catalysts (e.g., Ru,50 Ir,51 Rh,52 and Pd53) have demonstrated high efficacy, nickel-based catalysts present an earth-abundant and cost-effective alternative. In heterogeneous catalysis, Yus has demonstrated effective hydrogen transfer using nickel nanoparticles,54 while Barta and co-workers developed a heterogeneous Ni(COD)2-derived nickel oxide cluster for N-alkylation. However, homogeneous nickel catalysts remain less explored.55,56Recent advances in N-alkylation reactions typically rely on protocols that require high catalyst loadings and elevated temperatures, affording moderate to high yields (50–90%) (Scheme 4).57–59 Given these limitations, we report catalyst 2 that facilitates efficient N-alkylation across a broad range of alcohols, operates under mild conditions with minimal catalyst loading, and achieves high yields (85–96%) of the desired N-alkylated products.
The N-alkylation of benzyl alcohol with aniline using 0.5 mol% of catalyst 2 was selected as a benchmark reaction to optimize C–N bond formation. The optimized reaction conditions are mentioned in Table 2. A high yield of 87% in the initial test reaction prompted further investigation of key catalytic parameters, including base, solvent, and temperature. Among the bases tested (KOtBu, NaOH, KOH, Cs2CO3), KOtBu proved the most effective in activating the catalyst (entries 1–4). Lower reaction temperatures resulted in moderate yields (entry 10), while nonpolar solvents significantly enhanced the alkylation process (entries 4–7). Control experiments confirmed that both the catalyst and base were essential, as the reaction did not proceed in their absence (entries 12–13). Ultimately, toluene and KOtBu at 110 °C were identified as the optimal reaction conditions (entry 4), and were subsequently applied to assess the substrate scope (Scheme 5).
| Entry | Catalyst | Solvent | Base | Yielda (%) |
|---|---|---|---|---|
| a Yield determined by GC-MS. Aniline (0.50 mmol), benzyl alcohol (0.55 mmol), base (0.75 mmol), catalyst 2 (0.5 mol%) and solvent (2 mL), 110 °C. b Reaction temp = 80 °C. c 2 (0.25 mol%). | ||||
| 1 | 2 | Toluene | NaOH | 73 |
| 2 | 2 | Toluene | KOH | 69 |
| 3 | 2 | Toluene | Cs2CO3 | 54 |
| 4 | 2 | Toluene | KOtBu | 87 |
| 5 | 2 | n-Hexane | KOtBu | 71 |
| 6 | 2 | Benzene | KOtBu | 83 |
| 7 | 2 | Dioxane | KOtBu | 73 |
| 8 | NiCl2(DME) | Toluene | KOtBu | 0 |
| 9 | Ni(OAc)2·4H2O | Toluene | KOtBu | 0 |
| 10b | 2 | Toluene | KOtBu | 67 |
| 11c | 2 | Toluene | KOtBu | 58 |
| 12 | 2 | Toluene | No base | 0 |
| 13 | No catalyst | Toluene | KOtBu | 0 |
Interestingly, aniline derivatives bearing electron-donating substituents (4-methoxy and 4-tbutyl) and electron-withdrawing substituents (4-chloro, 2-iodo, and 4-biphenyl) proved to be effective coupling partners, affording the corresponding products in good yields 81–86% (xiii–xvii). Notably, 2-aminopyridine exhibited excellent conversion when coupled with different benzyl alcohol derivatives, affording products in 94–96% yield (xviii–xx). Similarly, isoquinolin-4-amine and quinoline-8-amine displayed high reactivity, providing yields of 88–95% (xxi–xxiii).
A plausible catalytic mechanism for C–N bond formation is proposed based on experimental evidence and supported by literature precedents (Scheme 6).60,61 The reaction begins with alcohol coordination to nickel in the presence of KOtBu, forming Ni-alkoxide I. β-Hydride elimination produces an aldehyde and Ni-hydride intermediate II. The aldehyde condenses with an amine to form an imine (confirmed by GCMS), which inserts into the Ni–H bond and is reduced to intermediate III′. Reaction of III′ with another alcohol regenerates Ni-alkoxide I, completing the catalytic cycle. Consistent with literature, attempts to isolate the unstable Ni-hydride intermediate were unsuccessful.58
Suzuki–Miyaura cross coupling reaction between aryl chlorides and boronic acids promoted by nickel pincer complex 2
The Suzuki–Miyaura cross-coupling reaction is a widely utilized method for synthesizing substituted diphenyls, valued for its mild conditions and broad functional group tolerance.62 Its popularity stems from the use of boronic acid nucleophiles, which are non-toxic, stable to air and moisture, and readily available.63,64 Kirchner et al. demonstrated the use of a NiII PNP complex65 while Hu et al. employed [(MeN2N)NiCl] complex, for alkyl–alkyl and alkyl–aryl couplings,66 both methods required high catalyst loadings and additives (Scheme 7).While aryl bromides and iodides are commonly employed under basic conditions with metal catalysts,67–69 aryl chlorides are increasingly attractive due to their lower cost, commercial availability, and structural diversity.70,71 However, their activation remains challenging, typically necessitating harsh conditions. To overcome these limitations, we sought to develop a highly efficient PNN-ligated Ni catalyst 2 capable of promoting Suzuki–Miyaura cross-coupling of aryl chlorides under mild conditions and with low catalyst loading.
The PNN Ni pincer complex 2 efficiently catalyzed the Suzuki–Miyaura cross-coupling reaction, affording the desired products in yields ranging from 85% to 99% under optimized conditions reported in Table 3. An initial assessment of the coupling reaction between chlorobenzene and phenylboronic acid in the absence of catalyst 2 showed no product formation (entry 1). Upon the addition of 0.25 mol% of the catalyst, a yield of 78% was observed (entry 2). Increasing the catalyst loading to 0.5 mol% significantly enhanced the yield to 96% (entry 3). A variety of bases, including K3PO4, KOH, NaOtBu, and KOtBu, were screened, with K3PO4 demonstrating superior performance (entries 3–6). Similarly, the effect of different solvents-toluene, 1,4-dioxane, THF, and water, was investigated, with toluene proving to be the most effective medium for the reaction (entries 3, 7–9). Lowering the reaction temperature to 80 °C resulted in a reduced yield of 69% (entry 10), while decreasing the reaction time also led to diminished product formation, with an optimized reaction time of 24 h (entry 11). Notably, no product formation was observed in the absence of a base (entry 12).
| Entry | Catalyst [mol%] | Base | Solvent | Yielda [%] |
|---|---|---|---|---|
| a Aryl chloride (0.50 mmol), phenyl boronic acid (0.75 mmol), base (1 mmol), 2 (0.5 mol%) and solvent (2 mL), 110 °C, yield was determined by GCMS. b Temp, 80 °C. c Reaction time, 18 h. | ||||
| 1 | No cat | K3PO4 | Toluene | 0 |
| 2 | 0.25 | K3PO4 | Toluene | 78 |
| 3 | 0.5 | K3PO4 | Toluene | 96 |
| 4 | 0.5 | KOH | Toluene | 62 |
| 5 | 0.5 | NaOtBu | Toluene | 76 |
| 6 | 0.5 | KOtBu | Toluene | 81 |
| 7 | 0.5 | K3PO4 | 1,4-Dioxane | 80 |
| 8 | 0.5 | K3PO4 | THF | 56 |
| 9 | 0.5 | K3PO4 | H2O | 0 |
| 10b | 0.5 | K3PO4 | Toluene | 69 |
| 11c | 0.5 | K3PO4 | Toluene | 77 |
| 12 | 0.5 | No base | Toluene | 0 |
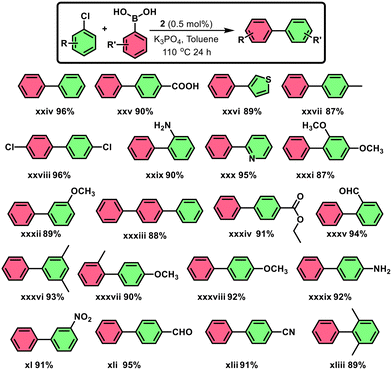
Using the optimized conditions (0.5 mol% catalyst 2, K3PO4, toluene, 110 °C, 24 h) (Scheme 8), a broad range of aryl chlorides and phenylboronic acids underwent smooth coupling to afford biphenyl derivatives in excellent yields. The method showed broad functional group tolerance and outperformed or matched existing systems, highlighting the efficiency of catalyst.
Aryl chlorides are typically challenging substrates due to the high bond dissociation energy of the C–Cl bond and their reluctance to undergo oxidative addition. The high catalytic efficiency observed with complex 2, even with aryl chlorides, can be attributed to the strong electron-donating and rigid tridentate nature of the PNN ligand, which stabilizes the NiII center and enhances its nucleophilicity. The square planar geometry enforced by the ligand framework is also conducive to oxidative addition. Additionally, the reaction conditions, including the presence of base and elevated temperature, likely facilitate this transformation. Literature reports support the formation of NiIV species from NiIIvia oxidative addition, particularly in pincer-ligated systems, suggesting that the transformation may proceed through a NiII/NiIV redox pathway under the employed conditions.17,72–75
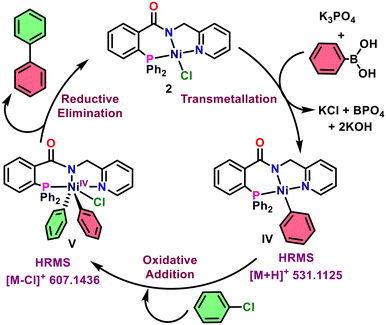
A plausible catalytic mechanism for the reaction, supported by experimental data and literature (Scheme 9), involves an initial transmetallation step.58,76,77 The PNN-NiII complex 2 reacts with a boronic acid and base to form intermediate IV. This is followed by oxidative addition of an aryl halide, yielding the PNN-NiIV complex (V). Subsequent reductive elimination affords the biphenyl product and regenerates the active NiII catalyst, thereby completing the catalytic cycle.
Hydroamination reaction between various anilines and styrene derivatives promoted by nickel pincer complex 2
Hydroamination is the atom-economical addition of an N–H bond across C–C multiple bonds, forming C–N and C–H bonds. It enables efficient amine synthesis from alkenes (e.g., vinyl arenes, dienes, allenes, strained alkenes) and alkynes.78,79 Hydroamination of alkenes is generally more challenging than that of alkynes due to their lower reactivity. The reaction can proceed via Markovnikov or anti-Markovnikov pathways, but achieving anti-Markovnikov selectivity, especially in intermolecular reactions remains a major challenge.80Previous studies have relied on expensive metals such as palladium and ruthenium, often under harsh conditions involving high temperatures,81 acidic media, and high catalyst loadings (Scheme 10).82 In contrast, our approach employs nickel, a much more economical alternative, under mild conditions with low catalyst loading, while still achieving high selectivity for the anti-Markovnikov product.
Styrene and aniline were selected as model substrates for this study. The optimized reaction conditions are provided in Table 4. In the presence of 0.5 mol% of the catalyst 2 at 110 °C in toluene with KOtBu as the base, the reaction yielded 76% of the selective anti-Markovnikov product (entry 1). Increasing the catalyst loading to 1 mol% improved the selectivity to 88% (entry 2). Screening various bases identified KOtBu as the most effective (entries 2–5). Lowering the reaction temperature to 80 °C reduced the yield to 64% (entry 8). Among the solvents tested, toluene provided the highest yield (entries 2, 6–7).
| Entry | Catalyst | Solvent | Base | Yield (%) |
|---|---|---|---|---|
| a Yield determined by GC-MS. b Styrene (0.50 mmol), aniline (0.55 mmol), base (0.75 mmol), toluene 2 mL, 110 °C and catalyst 2 (1 mol%). c Ni cat 2 (0.5 mol%). d Reaction temp = 80 °C. | ||||
| 1c | 2 | Toluene | KOtBu | 76 |
| 2 | 2 | Toluene | KOtBu | 88 |
| 3 | 2 | Toluene | NaOH | 55 |
| 4 | 2 | Toluene | KOH | 69 |
| 5 | 2 | Toluene | Cs2CO3 | 24 |
| 6 | 2 | Benzene | KOtBu | 72 |
| 7 | 2 | Dioxane | KOtBu | 60 |
| 8d | 2 | Toluene | KOtBu | 64 |
| 9 | 2 | Toluene | No base | 0 |
| 10 | No catalyst | Toluene | KOtBu | 0 |
Optimized conditions (2 (1 mol%), KOtBu, toluene, 110 °C) enabled efficient anti-Markovnikov hydroamination. Substrate scope studies (Scheme 11) showed that both electron-donating and electron-withdrawing groups on styrene or aniline were well tolerated, delivering excellent yields (88–94%, xlv–l).
The catalytic cycle for the base-catalyzed hydroamination of alkenes begins with the deprotonation of the amine by a base, generating a metal amide species (VI).83 This metal amide then acts as a nucleophile, facilitating the addition to the alkene. In our system, the nucleophilic attack preferentially occurs at the terminal carbon of the alkene, leading to exclusive formation of the anti-Markovnikov product. This regioselectivity can be attributed to the steric and electronic characteristics of the PNN-NiII complex, which likely favour an outer-sphere nucleophilic pathway and disfavours attack at the more hindered internal carbon. The resulting highly reactive 2-aminoalkyl complex (VII) subsequently undergoes proton transfer with a fresh amine molecule, regenerating the active metal amide catalyst and yielding the hydroamination product (Scheme 12).
While a comprehensive kinetic investigation was beyond the scope of this study, preliminary mechanistic insights suggest that the rate-determining step varies depending on the transformation. In the α-alkylation and N-alkylation reactions, the β-hydride elimination step, which generates the aldehyde and Ni–H intermediate, is likely the rate-determining step, consistent with established borrowing hydrogen mechanisms.59,84,85 In the case of Suzuki–Miyaura cross-coupling, transmetallation is the rate determining step.86,87 For the hydroamination reaction, nucleophilic attack of the metal-amido intermediate on the alkene likely constitutes the slowest step in the catalytic cycle.79,83,88 To further support these mechanistic proposals, key intermediates were detected and characterized by high-resolution mass spectrometry.
Mechanistic studies, including mercury drop tests, confirmed the homogeneous nature of all the reactions. Gram-scale reactions under optimized conditions demonstrated the robustness of the system, delivering excellent yields across various transformations: 94% (α-alkylation), 84% (N-alkylation), 91% (Suzuki–Miyaura), and 80% (hydroamination).
Conclusions
In summary, the PNN Ni pincer complex 2 was synthesized and shown to catalyse diverse transformations, including α-alkylation of ketones, N-alkylation of anilines, and Suzuki–Miyaura cross-coupling of aryl chlorides. It also enables selective anti-Markovnikov hydroamination of styrene under mild conditions with low loadings (0.5–1 mol%) and high yields (80–99%) across a broad substrate scope, underscoring its versatility and efficiency as a catalyst.Data availability
The data supporting this article have been included as part of the ESI† contents. Crystal structure determination, NMR and HRMS spectra of complexes, controlled experiments of reaction mechanism, NMR and mass spectra of catalytic products.Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Acknowledgements
MSB thank Indian Institute of Technology Bombay for supporting this work through Research Development Fund (RDF). We are thankful to the Department of Chemistry, IIT Bombay, for instrumentation facilities, as well as spectral and analytical data. GS acknowledges the financial support from IITB and KCD thanks UGC for the fellowship.Notes and references
- L. González-Sebastián, A. Reyes-Sanchez and D. Morales-Morales, Hydrogenation and Cross-Coupling Reactions Catalyzed by Mn, Fe, and Co Aromatic Pincer Complexes, Organometallics, 2023, 42, 2426–2446 CrossRef.
- K. K. Manar and P. Ren, in Ad. Organomet. Chem., ed. P. J. Pérez, Academic Press, 2021, vol. 76, pp. 185–259 Search PubMed.
- Z.-Q. Tian, Y. Zhang and M.-B. Li, Pincer Ligand Facilitated Metal-Catalyzed Multicomponent Reactions, Eur. J. Org. Chem., 2025, 28, e202401469 CrossRef.
- V. Arora, H. Narjinari and A. Kumar, Pincer-Nickel Catalyzed Selective Guerbet-Type Reactions, Organometallics, 2021, 40, 2870–2880 CrossRef.
- H. Valdés, M. A. García-Eleno, D. Canseco-Gonzalez and D. Morales-Morales, Recent Advances in Catalysis with Transition-Metal Pincer Compounds, ChemCatChem, 2018, 10, 3136–3172 CrossRef.
- J. A. Cruz-Navarro, A. Sánchez-Mora, J. S. Serrano-García, A. Amaya-Flórez, R. Colorado-Peralta, V. Reyes-Márquez and D. Morales-Morales, Advances in Cross-Coupling Reactions Catalyzed by Aromatic Pincer Complexes Based on Earth-Abundant 3d Metals (Mn, Fe, Co, Ni, Cu), Catalysts, 2024, 14, 69 CrossRef.
- A. Kasera, J. P. Biswas, A. Ali Alshehri, S. Ahmed Al-Thabaiti, M. Mokhtar and D. Maiti, Transition metal pincer complexes: A series of potential catalysts in CH activation reactions, Coord. Chem. Rev., 2023, 475, 214915 CrossRef.
- H. Valdés and D. Morales-Morales, in Catalysis for a Sustainable Environment, 2024, pp. 389–408 Search PubMed.
- N. Selander and K. J. Szabó, Catalysis by Palladium Pincer Complexes, Chem. Rev., 2011, 111, 2048–2076 CrossRef PubMed.
- L. Piccirilli, D. L. J. Pinheiro and M. Nielsen, Recent Progress with Pincer Transition Metal Catalysts for Sustainability, Catalysts, 2020, 10, 773 CrossRef.
- H. A. Younus, W. Su, N. Ahmad, S. Chen and F. Verpoort, Ruthenium Pincer Complexes: Synthesis and Catalytic Applications, Adv. Synth. Catal., 2015, 357, 283–330 CrossRef.
- J. S. Serrano-García, A. Amaya-Flórez, J.-R. Galindo, L. González-Sebastián, L. H. Delgado-Rangel and D. Morales-Morales, C–H Activation via Group 8–10 Pincer Complexes: A Mechanistic Approach, Inorganics, 2024, 12, 221 CrossRef.
- X. Sun, X. Dong, H. Liu and Y. Liu, Recent Progress in Palladium-Catalyzed Radical Reactions, Adv. Synth. Catal., 2021, 363, 1527–1558 CrossRef.
- D. Baidilov, D. Hayrapetyan and A. Y. Khalimon, Recent advances in homogeneous base-metal-catalyzed transfer hydrogenation reactions, Tetrahedron, 2021, 98, 132435 CrossRef CAS.
- V. M. Chernyshev and V. P. Ananikov, Nickel and Palladium Catalysis: Stronger Demand than Ever, ACS Catal., 2022, 12, 1180–1200 CrossRef.
- J. Bae and E. J. Cho, P,N Ligand in Ni-Catalyzed Cross-Coupling Reactions: A Promising Tool for π-Functionalization, ACS Catal., 2023, 13, 13540–13560 CrossRef.
- S. Z. Tasker, E. A. Standley and T. F. Jamison, Recent advances in homogeneous nickel catalysis, Nat., 2014, 509, 299–309 CrossRef.
- V. Arora, H. Narjinari, P. G. Nandi and A. Kumar, Recent advances in pincer–nickel catalyzed reactions, Dalton Trans., 2021, 50, 3394–3428 RSC.
- Z. N. Gafurov, A. A. Kagilev, A. O. Kantyukov, A. A. Balabaev, O. G. Sinyashin and D. G. Yakhvarov, Classification and synthesis of nickel pincer complexes, Russ. Chem. Bull., 2018, 67, 385–394 CrossRef.
- V. P. Ananikov, Nickel: The “Spirited Horse” of Transition Metal Catalysis, ACS Catal., 2015, 5, 1964–1971 CrossRef.
- V. B. Phapale and D. J. Cárdenas, Nickel-catalysed Negishi cross-coupling reactions: scope and mechanisms, Chem. Soc. Rev., 2009, 38, 1598–1607 RSC.
- J. E. Dander and N. K. Garg, Breaking Amides using Nickel Catalysis, ACS Catal., 2017, 7, 1413–1423 CrossRef.
- N. A. Weires, E. L. Baker and N. K. Garg, Nickel-catalysed Suzuki–Miyaura coupling of amides, Nat. Chem., 2016, 8, 75–79 CrossRef CAS.
- G. Sabharwal, K. C. Dwivedi, C. Das, T. R. K. Rana, A. Dutta, G. Rajaraman and M. S. Balakrishna, Detailed mechanistic studies on PNN-palladium pincer complex catalyzed Suzuki-Miyaura cross-coupling reaction proceeding through a PdII/PdIII/PdIV catalytic cycle, J. Catal., 2024, 440, 115825 CrossRef CAS.
- F. Song, B. Wang and Z.-J. Shi, Transition-Metal-Catalyzed C–C Bond Formation from C–C Activation, Acc. Chem. Res., 2023, 56, 2867–2886 CrossRef CAS PubMed.
- N. Lal Verma, M. Kumar, D. Sharma, S. Jaipal, N. Rathore Kumar and G. Jaiswar, Transition-metal-mediated C-C bond activation: Recent advances and its applications in organic synthesis, Results Chem., 2023, 6, 101130 CrossRef CAS.
- X. Wu, Y. Zhao and H. Ge, Nickel-Catalyzed Site-Selective Alkylation of Unactivated C(sp3)–H Bonds, J. Am. Chem. Soc., 2014, 136, 1789–1792 CrossRef CAS.
- R. Kumar and E. V. Van der Eycken, Recent approaches for C–C bond formation via direct dehydrative coupling strategies, Chem. Soc. Rev., 2013, 42, 1121–1146 RSC.
- M. H. S. A. Hamid, P. A. Slatford and J. M. J. Williams, Borrowing Hydrogen in the Activation of Alcohols, Adv. Synth. Catal., 2007, 349, 1555–1575 CrossRef CAS.
- C. Gunanathan and D. Milstein, Applications of Acceptorless Dehydrogenation and Related Transformations in Chemical Synthesis, Science, 2013, 341, 1229712 CrossRef.
- S. Genç, S. Günnaz, B. Çetinkaya, S. Gülcemal and D. Gülcemal, Iridium(I)-Catalyzed Alkylation Reactions To Form α-Alkylated Ketones, J. Org. Chem., 2018, 83, 2875–2881 CrossRef PubMed.
- W.-D. Li, J.-S. Zhang, L.-Y. Zhang, Z.-W. Liu, J. Fan and X.-Y. Shi, Rhodium-Catalyzed Alkylation of Aromatic Ketones with Allylic Alcohols and α,β-Unsaturated Ketones, Catalysts, 2023, 13, 1157 CrossRef CAS.
- X.-N. Cao, X.-M. Wan, F.-L. Yang, K. Li, X.-Q. Hao, T. Shao, X. Zhu and M.-P. Song, NNN Pincer Ru(II)-Complex-Catalyzed α-Alkylation of Ketones with Alcohols, J. Org. Chem., 2018, 83, 3657–3668 CrossRef CAS PubMed.
- C. Wang and G. Dong, Direct β-Alkylation of Ketones and Aldehydes via Pd-Catalyzed Redox Cascade, J. Am. Chem. Soc., 2018, 140, 6057–6061 CrossRef CAS PubMed.
- M. Peña-López, P. Piehl, S. Elangovan, H. Neumann and M. Beller, Manganese-Catalyzed Hydrogen-Autotransfer C−C Bond Formation: α-Alkylation of Ketones with Primary Alcohols, Angew. Chem., Int. Ed., 2016, 55, 14967–14971 CrossRef PubMed.
- M.-S. Abdallah, N. Joly, S. Gaillard, A. Poater and J.-L. Renaud, Blue-Light-Induced Iron-Catalyzed α-Alkylation of Ketones, Org. Lett., 2022, 24, 5584–5589 CrossRef CAS.
- G. Zhang, J. Wu, H. Zeng, S. Zhang, Z. Yin and S. Zheng, Cobalt-Catalyzed α-Alkylation of Ketones with Primary Alcohols, Org. Lett., 2017, 19, 1080–1083 CrossRef CAS PubMed.
- D.-Y. Yang, H. Wang and C.-R. Chang, Recent Advances for Alkylation of Ketones and Secondary Alcohols Using Alcohols in Homogeneous Catalysis, Adv. Synth. Catal., 2022, 364, 3100–3121 CrossRef CAS.
- J. Das, K. Singh, M. Vellakkaran and D. Banerjee, Nickel-Catalyzed Hydrogen-Borrowing Strategy for α-Alkylation of Ketones with Alcohols: A New Route to Branched gem-Bis(alkyl) Ketones, Org. Lett., 2018, 20, 5587–5591 CrossRef CAS.
- J. Das, M. Vellakkaran and D. Banerjee, Nickel-Catalyzed Alkylation of Ketone Enolates: Synthesis of Monoselective Linear Ketones, J. Org. Chem., 2019, 84, 769–779 CrossRef CAS.
- L. Zhang, G. Raffa, D. H. Nguyen, Y. Swesi, L. Corbel-Demailly, F. Capet, X. Trivelli, S. Desset, S. Paul, J.-F. Paul, P. Fongarland, F. Dumeignil and R. M. Gauvin, Acceptorless dehydrogenative coupling of alcohols catalysed by ruthenium PNP complexes: Influence of catalyst structure and of hydrogen mass transfer, J. Catal., 2016, 340, 331–343 CrossRef CAS.
- A. K. Bains, A. Biswas, A. Kundu and D. Adhikari, Nickel-Catalysis Enabling α-Alkylation of Ketones by Secondary Alcohols, Adv. Synth. Catal., 2022, 364, 2815–2821 CrossRef CAS.
- X. Zhang, B. Tutkowski, A. Oliver, P. Helquist and O. Wiest, Mechanistic Study of the Nickel-Catalyzed α,β-Coupling of Saturated Ketones, ACS Catal., 2018, 8, 1740–1747 CrossRef CAS.
- S. Genç, B. Arslan, D. Gülcemal, S. Gülcemal and S. Günnaz, Nickel-catalyzed alkylation of ketones and nitriles with primary alcohols, Org. Biomol. Chem., 2022, 20, 9753–9762 RSC.
- G. Chakraborty, S. Pal, S. Chakraborty and N. D. Paul, Nickel-Catalyzed Sustainable and Selective Alkylation of Alcohols to α-Alkylated Ketones via Borrowing Hydrogen Approach, Synlett, 2022, 34, 635–644 Search PubMed.
- S. Chakraborty, P. Daw, Y. Ben David and D. Milstein, Manganese-Catalyzed α-Alkylation of Ketones, Esters, and Amides Using Alcohols, ACS Catal., 2018, 8, 10300–10305 CrossRef CAS PubMed.
- X. Ma, C. Su and Q. Xu, in Hydrogen Transfer Reactions: Reductions and Beyond, ed. G. Guillena and D. J. Ramón, Springer International Publishing, Cham, 2016, pp. 291–364, DOI:10.1007/978-3-319-43051-5_7.
- A. R. Bayguzina and R. I. Khusnutdinov, Catalytic N-Alkylation of Anilines, Russ. J. Gen. Chem., 2021, 91, 305–347 CrossRef CAS.
- J. Mahato, R. Das and T. K. Saha, 3d transition metal complexes as homogeneous catalysts in N-alkylation reactions using alcohols: A recent update, Tetrahedron, 2024, 165, 134192 CrossRef CAS.
- B. Patel, R. Ranjan, N. R. Chauhan, S. Mukhopadhyay, A. R. Choudhury and K. M. Vyas, N-coordinated Ru(II) catalyzed solvent free N-alkylation of primary amines with alcohols through borrowing hydrogen strategy, New J. Chem., 2023, 47, 8305–8317 RSC.
- K.-i. Fujita, Y. Enoki and R. Yamaguchi, Cp*Ir-catalyzed N-alkylation of amines with alcohols. A versatile and atom economical method for the synthesis of amines, Tetrahedron, 2008, 64, 1943–1954 CrossRef CAS.
- L. Huang, Z. Wang, L. Geng, R. Chen, W. Xing, Y. Wang and J. Huang, Selective and recyclable rhodium nanocatalysts for the reductive N-alkylation of nitrobenzenes and amines with aldehydes, RSC Adv., 2015, 5, 56936–56941 RSC.
- S. Sabu, P. V. Saranya and G. Anilkumar, An overview of palladium-catalyzed N-alkylation reactions, Org. Biomol. Chem., 2025, 23, 1533–1551 RSC.
- F. Alonso, P. Riente and M. Yus, Nickel Nanoparticles in Hydrogen Transfer Reactions, Acc. Chem. Res., 2011, 44, 379–391 CrossRef CAS.
- A. Afanasenko, S. Elangovan, M. C. A. Stuart, G. Bonura, F. Frusteri and K. Barta, Efficient nickel-catalysed N-alkylation of amines with alcohols, Catal. Sci. Technol., 2018, 8, 5498–5505 RSC.
- T. Yan, B. L. Feringa and K. Barta, Direct Catalytic N-Alkylation of α-Amino Acid Esters and Amides Using Alcohols with High Retention of Stereochemistry, ChemSusChem, 2021, 14, 2303–2307 CrossRef CAS.
- G. Balamurugan, R. Ramesh and J. G. Malecki, Nickel(II)–NΛNΛO Pincer Type Complex-Catalyzed N-alkylation of Amines with Alcohols via the Hydrogen Autotransfer Reaction, J. Org. Chem., 2020, 85, 7125–7135 CrossRef CAS.
- A. K. Bains, A. Kundu, S. Yadav and D. Adhikari, Borrowing Hydrogen-Mediated N-Alkylation Reactions by a Well-Defined Homogeneous Nickel Catalyst, ACS Catal., 2019, 9, 9051–9059 CrossRef CAS.
- M. Vellakkaran, K. Singh and D. Banerjee, An Efficient and Selective Nickel-Catalyzed Direct N-Alkylation of Anilines with Alcohols, ACS Catal., 2017, 7, 8152–8158 CrossRef CAS.
- X. Yang, X. Tian, N. Sun, B. Hu, Z. Shen, X. Hu and L. Jin, Geometry-Constrained N,N,O-Nickel Catalyzed α-Alkylation of Unactivated Amides via a Borrowing Hydrogen Strategy, Organometallics, 2023, 42, 38–47 CrossRef CAS.
- M. Subaramanian, G. Sivakumar and E. Balaraman, Recent advances in nickel-catalyzed C–C and C–N bond formation via HA and ADC reactions, Org. Biomol. Chem., 2021, 19, 4213–4227 RSC.
- B. S. Kadu, Suzuki–Miyaura cross coupling reaction: recent advancements in catalysis and organic synthesis, Catal. Sci. Technol., 2021, 11, 1186–1221 RSC.
- S. Akkoç, Importance of some factors on the Suzuki-Miyaura cross-coupling reaction, J. Chin. Chem. Soc., 2021, 68, 942–951 CrossRef.
- L.-C. Campeau and N. Hazari, Cross-Coupling and Related Reactions: Connecting Past Success to the Development of New Reactions for the Future, Organometallics, 2019, 38, 3–35 CrossRef CAS.
- D. Benito-Garagorri and K. Kirchner, Modularly Designed Transition Metal PNP and PCP Pincer Complexes based on Aminophosphines: Synthesis and Catalytic Applications, Acc. Chem. Res., 2008, 41, 201–213 CrossRef CAS PubMed.
- X. Hu, Nickel-catalyzed cross coupling of non-activated alkyl halides: a mechanistic perspective, Chem. Sci., 2011, 2, 1867–1886 RSC.
- C.-S. Yan, Y. Peng, X.-B. Xu and Y.-W. Wang, Nickel-Mediated Inter- and Intramolecular Reductive Cross-Coupling of Unactivated Alkyl Bromides and Aryl Iodides at Room Temperature, Chem. – Eur. J., 2012, 18, 6039–6048 CrossRef CAS PubMed.
- S. K. Gurung, S. Thapa, B. Shrestha and R. Giri, Copper-catalysed cross-couplings of arylboronate esters with aryl and heteroaryl iodides and bromides, Org. Chem. Front., 2015, 2, 649–653 RSC.
- N. Kambe, T. Iwasaki and J. Terao, Pd-catalyzed cross-coupling reactions of alkyl halides, Chem. Soc. Rev., 2011, 40, 4937–4947 RSC.
- T. E. Barder and S. L. Buchwald, Efficient Catalyst for the Suzuki−Miyaura Coupling of Potassium Aryl Trifluoroborates with Aryl Chlorides, Org. Lett., 2004, 6, 2649–2652 CrossRef CAS.
- J. P. Simeone and J. R. Sowa, Palladium on carbon as a precatalyst for the Suzuki–Miyuara cross-coupling of aryl chlorides, Tetrahedron, 2007, 63, 12646–12654 CrossRef CAS.
- J. R. Bour, N. M. Camasso and M. S. Sanford, Oxidation of Ni(II) to Ni(IV) with Aryl Electrophiles Enables Ni-Mediated Aryl–CF3 Coupling, J. Am. Chem. Soc., 2015, 137, 8034–8037 CrossRef CAS PubMed.
- N. M. Camasso and M. S. Sanford, Design, synthesis, and carbon-heteroatom coupling reactions of organometallic nickel(IV) complexes, Science, 2015, 347, 1218–1220 CrossRef CAS.
- C. C. Roberts, E. Chong, J. W. Kampf, A. J. Canty, A. Ariafard and M. S. Sanford, Nickel(II/IV) Manifold Enables Room-Temperature C(sp3)–H Functionalization, J. Am. Chem. Soc., 2019, 141, 19513–19520 CrossRef CAS PubMed.
- E. A. Meucci, A. Ariafard, A. J. Canty, J. W. Kampf and M. S. Sanford, Aryl–Fluoride Bond-Forming Reductive Elimination from Nickel(IV) Centers, J. Am. Chem. Soc., 2019, 141, 13261–13267 CrossRef CAS PubMed.
- Z. Li and L. Liu, Recent advances in mechanistic studies on Ni catalyzed cross-coupling reactions, Chin. J. Catal., 2015, 36, 3–14 CrossRef CAS.
- J. M. Neely, M. J. Bezdek and P. J. Chirik, Insight into Transmetalation Enables Cobalt-Catalyzed Suzuki–Miyaura Cross Coupling, ACS Cent. Sci., 2016, 2, 935–942 CrossRef CAS.
- M. Ciobanu, A. Tirsoaga, P. Amoros, D. Beltran, S. M. Coman and V. I. Parvulescu, Comparative hydroamination of aniline and substituted anilines with styrene on different zeolites, triflate based catalysts and their physical mixtures, Appl. Catal., A, 2014, 474, 230–235 CrossRef CAS.
- M. Kawatsura and J. F. Hartwig, Palladium-Catalyzed Intermolecular Hydroamination of Vinylarenes Using Arylamines, J. Am. Chem. Soc., 2000, 122, 9546–9547 CrossRef CAS.
- P. Yamuna, R. M. Philip and G. Anilkumar, Nickel catalyzed hydroamination reactions: An overview, Tetrahedron, 2022, 122, 132936 CrossRef CAS.
- Y. Gök, B. Yiğit, Ö. Özeroğlu Çelikal and M. Yiğit, Anti-Markovnikov hydroaminations of styrene catalyzed by palladium(II) N-heterocyclic carbene complexes under conventional and microwave heating, Transition Met. Chem., 2018, 43, 591–596 CrossRef.
- M. Utsunomiya and J. F. Hartwig, Ruthenium-Catalyzed Anti-Markovnikov Hydroamination of Vinylarenes, J. Am. Chem. Soc., 2004, 126, 2702–2703 CrossRef CAS PubMed.
- J. Escorihuela, A. Lledós and G. Ujaque, Anti-Markovnikov Intermolecular Hydroamination of Alkenes and Alkynes: A Mechanistic View, Chem. Rev., 2023, 123, 9139–9203 CrossRef CAS.
- E. Podyacheva, O. I. Afanasyev, D. V. Vasilyev and D. Chusov, Borrowing Hydrogen Amination Reactions: A Complex Analysis of Trends and Correlations of the Various Reaction Parameters, ACS Catal., 2022, 12, 7142–7198 CrossRef CAS.
- T. Irrgang and R. Kempe, 3d-Metal Catalyzed N- and C-Alkylation Reactions via Borrowing Hydrogen or Hydrogen Autotransfer, Chem. Rev., 2019, 119, 2524–2549 CrossRef CAS.
- L. Jin, J. Xin, Z. Huang, J. He and A. Lei, Transmetalation is the Rate-Limiting Step: Quantitative Kinetic Investigation of Nickel-Catalyzed Oxidative Coupling of Arylzinc Reagents, J. Am. Chem. Soc., 2010, 132, 9607–9609 CrossRef CAS.
- Y. Li, Y. Luo, L. Peng, Y. Li, B. Zhao, W. Wang, H. Pang, Y. Deng, R. Bai, Y. Lan and G. Yin, Reaction scope and mechanistic insights of nickel-catalyzed migratory Suzuki–Miyaura cross-coupling, Nat. Commun., 2020, 11, 417 CrossRef CAS PubMed.
- M. Beller, H. Trauthwein, M. Eichberger, C. Breindl, J. Herwig, T. E. Müller and O. R. Thiel, The First Rhodium-Catalyzed Anti-Markovnikov Hydroamination: Studies on Hydroamination and Oxidative Amination of Aromatic Olefins, Chem. – Eur. J., 1999, 5, 1306–1319 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 2359370, 2359374 and 2359375. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d5cy00718f |
| This journal is © The Royal Society of Chemistry 2025 |