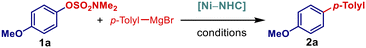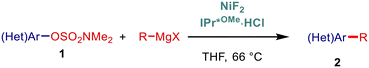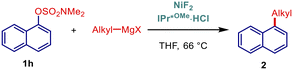C(sp2)–C(sp2) and C(sp2)–C(sp3) nickel-catalyzed Kumada–Corriu and Buchwald–Hartwig cross-coupling of aryl sulfamates enabled by sterically-demanding, electron-rich IPr*OMeN-heterocyclic carbenes
Marlena
Kardela
a,
Michal
Szostak
 *b and
Elwira
Bisz
*b and
Elwira
Bisz
 *a
*a
aDepartment of Chemistry and Pharmacy, Opole University, 48 Oleska Street, 45-052 Opole, Poland. E-mail: ebisz@uni.opole.pl
bDepartment of Chemistry, Rutgers University, 73 Warren Street, Newark, NJ 07102, USA. E-mail: michal.szostak@rutgers.edu
First published on 9th September 2025
Abstract
Nickel-catalyzed cross-coupling reactions are among the most powerful tools in organic synthesis owing to the low cost, natural abundance and high activity of versatile nickel catalysts. However, there is a scarcity of efficient Ni-catalyzed systems for the cross-coupling of bench-stable, readily accessible and orthogonal C–O electrophiles. Herein, we report the cross-coupling of aryl sulfamates enabled by sterically-demanding and electron-rich N-heterocyclic carbenes. The study identified sulfamates as the most reactive C–O electrophiles enabled by a combination of IPr*OMe bearing sterically-demanding ortho-diphenyl wingtip substitution and NiF2 exploiting the fluoride effect. The system enables efficient C(sp2)–C(sp2) and C(sp2)–C(sp3) cross-coupling avoiding hydrolysis, homocoupling and unselective C–O and S–N cleavages, providing a powerful platform for the orthogonal use of C–O electrophiles. The system is also applicable to Buchwald–Hartwig amination. The broad access to electron-rich, sterically-demanding N-heterocyclic carbenes combined with the high reactivity of aryl sulfamates furnishes a commanding avenue for the development of Ni-catalyzed processes in academic and industrial research.
As highlighted by the 2010 Nobel Prize,1 transition-metal-catalyzed cross-coupling reactions represent a critical method for the construction of carbon–carbon and carbon–heteroatom bonds in organic synthesis and catalysis with broad ranging applications in every aspect of chemical science, including pharmaceuticals, agrochemicals, and advanced materials.2 The importance of cross-coupling processes stems from the fact that, in particular, C(sp2)–C(sp2), C(sp2)–C(sp3) and C(sp2)–N disconnections are not possible using other established or alternative methods.3 In general, as emphasized by the 2010 Nobel Prize, palladium-based catalysts have dominated the field of cross-coupling research.1,2a,4 This has resulted in the development of a plethora of ligands and catalytic systems specific to palladium that enable broad generality and utility of the palladium catalytic platform. However, owing to the limited resources of palladium and impending supply shortage, the use of naturally abundant 3d nickel has received major attention.5 The pioneering studies by Dankwardt,6 Snieckus,7 Garg,8 Shi,9 Chatani10 and others11 have shown that Ni catalysts are effective for the traditionally-challenging cross-coupling of C–O electrophiles owing to the lower electronegativity and higher redox potential of nickel compared with palladium.12 However, in contrast to palladium, there is a scarcity of efficient Ni-catalyzed systems that would enable to expand the scope of cross-coupling partners and provide an efficient platform for the deployment of practical phenol-derived electrophiles as alternatives to aryl halides.5a
In this context, aryl sulfamates have emerged as highly valuable electrophiles for carbon–carbon bond formation.7b,8b,13 Aryl sulfamates are easily accessible by modular methods, are significantly stable to various reaction conditions, including traditional cross-couplings and feature an inherent directing group ability in both directed ortho-metallations and C–H functionalizations.7c Crucially, the C–O bond in aryl sulfamates is considerably more robust than in other related phenolic electrophiles, including triflates, tosylates and carbamates. While aryl sulfamates can be categorized as ideal and highly desirable electrophiles for cross-coupling, the strong C–OS(O)2NR2 bond presents a major challenge as a kinetic barrier to oxidative addition necessitating the development of highly tailored ligands.14
In our previous studies we have been focused on N-heterocyclic carbenes (N-heterocyclic carbene = NHC) as enabling ligands for transition-metal-catalysis.15N-Heterocyclic carbenes feature strong σ-donation16 of the carbenic center together with high steric demand and variable flexibility of N-aromatic wingtip,17 permitting for highly efficient catalysis of the elementary steps of cross-coupling cycle.18 Further, the large steric-demand of select N-heterocyclic carbene ligands is highly beneficial for suppressing β-hydride elimination and undesired homocoupling side processes.19 However, compared with palladium catalysis, where many classes of different NHC ligands have been secured, the establishment of efficient Ni–NHC systems has been significantly underdeveloped.3,5 Taking inspiration from the studies by Snieckus,7 Nicasio,11a Chatani10 and others6,8,9,11b–f on nickel catalysis and cognizant of the work by Nakamura20 on the utility of metal fluorides in the formation of high-valent metalate complexes preventing homocoupling,15a herein we report the nickel-catalyzed cross-coupling of aryl sulfamates enabled by sterically-demanding and electron-rich N-heterocyclic carbenes (Fig. 1). The study identified sulfamates as the most reactive C–O electrophiles enabled by a combination of IPr*OMe bearing sterically-demanding ortho-diphenyl wingtip substitution and NiF2 exploiting the fluoride effect. The system enables efficient C(sp2)–C(sp2) and C(sp2)–C(sp3) cross-coupling avoiding hydrolysis, homocoupling and unselective C–O and S–N cleavages, providing a powerful platform for the orthogonal use of C–O electrophiles. The system is also applicable to Buchwald–Hartwig amination.21 Overall, the broad access to electron-rich, sterically-demanding N-heterocyclic carbenes combined with the high reactivity of aryl sulfamates furnishes a commanding avenue for the development of Ni-catalyzed cross-coupling processes in academic and industrial research.
Our study commenced with the examination of the cross-coupling of electronically-deactivated 4-anisolyl electrophile (4-MeO-C6H4-OSO2NMe2) with 4-Tol-MgBr under a variety of conditions (Table 1). We established that a combination of NiF2 (10 mol%) and IPr*OMe·HCl (20 mol%) in THF at 66 °C permits for the cross-coupling in a quantitative yield in the absence of any further additives within 30 min reaction time (Table 1, entry 1). The reaction was fully selective with respect to the alternative C–OMe bond, the cleavage of the alternative S–O bond as well as deaminative S–N cleavage, all processes known to occur with Ni-catalyzed systems.14a This high selectivity was further confirmed by experiments with respect to the reaction time, temperature (see SI) and stoichiometry (Table 1, entries 2–5), demonstrating exquisite chemoselectivity under the developed conditions. Next, we comprehensively evaluated the effect of different NHC ligands (Table 1, entries 6–9).
| Entrya | Catalyst | Ligand | Ligand (equiv.) | Time | Yield 2a (%)b |
|---|---|---|---|---|---|
| a Conditions: 1a (0.25 mmol), [Ni] (10 mol%), THF (0.19 M), p-TolylMgBr (2.0 equiv., 1.0 M in THF), 66 °C, p-TolylMgBr added dropwise over 1–2 s. b Determined by 1H NMR. c [Ni] (5 mol%). d [Ni] (0.5 mol%). | |||||
| 1 | NiF2 | IPr*OMe·HCl | 0.2 | 30 min | 98 |
| 2c | NiF2 | IPr*OMe·HCl | 0.1 | 30 min | 86 |
| 3c | NiF2 | IPr*OMe·HCl | 0.05 | 30 min | 27 |
| 4c | NiF2 | IPr*OMe·HCl | 0.025 | 30 min | 34 |
| 5d | NiF2 | IPr*OMe·HCl | 0.2 | 30 min | 8 |
| 6 | NiF2 | IPr*·HCl | 0.2 | 30 min | 98 |
| 7 | NiF2 | IPaul·HCl | 0.2 | 30 min | 32 |
| 8 | NiF2 | IPr·HCl | 0.2 | 30 min | 0 |
| 9 | NiF2 | BIAN-IPr·HCl | 0.2 | 30 min | 0 |
| 10 | NiF2 | — | — | 30 min | 0 |
| 11 | [Ni(IPr*OMe)CpCl] | — | — | 30 min | 82 |
| 12 | [Ni(IPr*)CpCl] | — | — | 30 min | 77 |
| 13 | [Ni(IPaul)CpCl] | — | — | 30 min | 78 |
| 14 | [Ni(IPr*OMe)CpCl] | IPr*OMe·HCl | 0.2 | 30 min | 92 |
| 15 | Ni(acac)2 | IPr*OMe·HCl | 0.2 | 30 min | 85 |
| 16 | NiCl2·6H2O | IPr*OMe·HCl | 0.2 | 30 min | 14 |
| 17 | Ni(COD)2 | IPr*OMe·HCl | 0.2 | 30 min | 82 |
As such, sterically-demanding but less electron-rich IPr* is also effective in promoting the desired cross-coupling; however, it is less efficient requiring longer reaction time to reach the desired conversions (Table 1, entry 6 and SI). Its sterically-adjustable counterpart, IPaul, gives a noticeable decrease in yield (Table 1, entry 7), however, we note that this ligand is still effective under longer reaction time regimen (see SI). Most interestingly, the classic IPr and the more sterically-demanding BIAN–IPr featuring the buttressing acenaphthoimidazolylidene scaffold are ineffective (Table 1, entries 8–9), demonstrating the key role of steric ortho-diphenyl wingtip substitution on the reactivity. As expected, N-heterocyclic carbene ligand is absolutely required for the cross-coupling with no reaction observed in its absence (Table 1, entry 10). We also evaluated well-defined Ni(II)–NHC complexes based on the established cyclopentadienyl ancillary ligand (Table 1, entries 11–14). Interestingly, the well-defined [Ni(IPr*OMe)CpCl], [Ni(IPr*)CpCl], and [Ni(IPaul)CpCl] complexes can also be used for the cross-coupling with a yield slightly lower than the in situ prepared complexes. These results suggest the importance of activation to the catalytically active Ni(0)–NHC and excess of ligand for the desired cross-coupling. Finally, studies with different Ni sources, such as Ni(acac)2, NiCl2 and Ni(cod)2 (Table 1, entries 15–17), resulted in lower yields, consistent with the key role of NiF2 for this class of cross-couplings. It should also be noted that the use of bench-stable Ni(II)–NHC complexes is preferred, because in situ generation of Ni(0)–NHCs may reduce the effective concentration of Ni–NHC in the early stages of the reaction. Furthermore, Ni(0) sources such as Ni(cod)2 are known to exist in equilibrium with Grignard reagents with COD dissociation, leading to competitive reactions,22 whereas NiF2/IPr*OMe benefits from direct activation under Grignard conditions and consistently provides optimal reactivity.
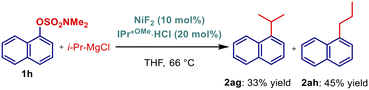
Next, kinetic studies were conducted to investigate the effect of the C–O leaving group (Fig. 2). Unexpectedly, in the series of OTf, OTs, OCONEt2, OSO2NMe2, the most challenging sulfamate featuring the strongest C–O bond was found the most reactive. Probably the main difference between sulfamates and the usually most reactive triflate electrophiles is the lower stability of triflates under the reaction conditions. Aryl sulfamates may also benefit from stabilizing nitrogen coordination to nickel, which lowers the barrier for oxidative addition.23 Undoubtedly, the high reactivity of bench-stable aryl sulfamates is very beneficial from the practical standpoint.
 | ||
| Fig. 2 Kinetic profiles. Conditions: 1 (0.25 mmol), p-TolylMgBr (2.0 equiv., 1.0 M in THF), [Ni] (10 mol%), ligand (20 mol%), THF (0.19 M), 66 °C. | ||
Kinetic studies were also performed to gain insight into the effect of NHC ligands (Fig. 3 and 4). In the cross-coupling of the electronically-deactivated 4-MeO-C6H4-OSO2NMe2 with 4-Tol-MgBr, IPr*MeO outperformed its electronically less σ-donating IPr* and less sterically-demanding IPaul, while the reactivity of IPr was negligible (Fig. 3). Further, in the reactivity of activated 1-naphthyl electrophile, IPr*MeO was still the most reactive catalyst, closely followed by IPr*, while IPr was again the least effective catalyst. These studies together with the optimization results clearly indicate the combined effect of strong σ-donation and high steric demand on the reactivity in this cross-coupling.
 | ||
| Fig. 3 Kinetic profiles. Conditions: 1a (0.25 mmol), p-TolylMgBr (2.0 equiv., 1.0 M in THF), [Ni] (10 mol%), ligand (20 mol%), THF (0.19 M), 66 °C. | ||
 | ||
| Fig. 4 Kinetic profiles. Conditions: 1h (0.25 mmol), 4-OMe-PhMgBr (2.0 equiv., 1.0 M in THF), [Ni] (10 mol%), ligand (20 mol%), THF (0.19 M), 66 °C. | ||
With the optimized conditions in hand, the scope of this C(sp2)–C(sp2) cross-coupling was next investigated (Table 2). In general, the scope of the reaction is very broad and encompasses various aryl sulfamates featuring electron donating (Table 2, entries 1–3), biaryl (Table 2, entries 4–5), electron-withdrawing (Table 2, entries 6–8), unsubstituted (Table 1, entries 9–10), sterically-hindered 1-naphthlyl (Table 2, entries 11–15), conjugated 2-naphthyl (Table 2, entries 16–18), heterocyclic 2- and 3-pyridyl (Table 2, entries 19–22) and quinolinyl (Table 2, entry 23) electrophiles using neutral (Table 2, entry 3), electron-deficient (Table 2, entry 2), electron-rich (Table 2, entry 7) and heterocyclic (Table 2, entry 15) organometallic reagents.
| Entrya | Substrate | R-MgX | Time | 2 | Product | Yield (%) |
|---|---|---|---|---|---|---|
| a Conditions: 1 (0.25 mmol), [Ni] (10 mol%), IPr*OMe·HCl (20 mol%); THF (0.19 M), RMgX (2.0 equiv., 1.0 M in THF), 66 °C, RMgX added dropwise over 1–2 s. b o-Tolyl-MgBr (2.0 M in Et2O). | ||||||
| 1 |

|
p-TolylMgBr | 30 min | 2a |
|
98 |
| 2 | 4-F-C6H4MgBr | 30 min | 2b |

|
98 | |
| 3 | PhMgBr | 30 min | 2c |
|
94 | |
| 4 |

|
p-TolylMgBr | 30 min | 2d |

|
86 |
| 5 | PhMgBr | 30 min | 2e |
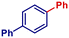
|
69 | |
| 6 |

|
p-TolylMgBr | 30 min | 2f |

|
90 |
| 7 | 4-MeO-C6H4MgBr | 30 min | 2b |

|
77 | |
| 8 | PhMgBr | 30 min | 2g |

|
77 | |
| 9 |

|
p-TolylMgBr | 30 min | 2h |

|
98 |
| 10 | 4-MeO-C6H4MgBr | 30 min | 2c |

|
98 | |
| 11 |
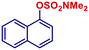
|
p-TolylMgBr | 30 min | 2i |
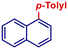
|
98 |
| 12 | 4-MeO-C6H4MgBr | 30 min | 2j |
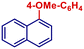
|
98 | |
| 13 | PhMgBr | 30 min | 2k |
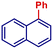
|
96 | |
| 14b | o-TolylMgBr | 30 min | 2l |
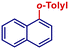
|
78 | |
| 15 | 2-ThienylMgBr | 21 h | 2m |
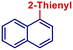
|
53 | |
| 16 |

|
p-TolylMgBr | 30 min | 2n |

|
91 |
| 17 | 4-MeO-C6H4MgBr | 30 min | 2o |

|
92 | |
| 18 | PhMgBr | 30 min | 2p |
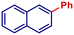
|
86 | |
| 19 |
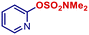
|
p-TolylMgBr | 3 h | 2q |
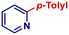
|
67 |
| 20 | 4-MeO-C6H4MgBr | 3 h | 2r |
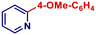
|
74 | |
| 21 | PhMgBr | 3 h | 2s |

|
98 | |
| 22 |
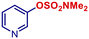
|
p-TolylMgBr | 3 h | 2t |

|
61 |
| 23 |

|
PhMgBr | 30 min | 2u |

|
75 |
In light of the unexpectedly high reactivity of the present NiF2/IPr*MeO system in the cross-coupling with aryl organometallics, we were also keen on investigating the significantly more challenging C(sp2)–C(sp3) cross-coupling (Table 3). In general, alkylative cross-couplings of C–O electrophiles are prone to β-hydride elimination, homocoupling and protodemetalation pathways. We were delighted to find that the present system is also highly efficient in the cross-coupling of alkyl organometallics (Table 3, entries 1–7). Interestingly, IPr*MeO again outperforms IPr* and IPr (Table 3, entries 2–4), indicating a generality of the catalyst system.
| Entrya | Ligand | R-MgX | Time | 2 | Product | Yield (%) |
|---|---|---|---|---|---|---|
| a Conditions: 1h (0.25 mmol), [Ni] (10 mol%), IPr*OMe·HCl (20 mol%); THF (0.19 M), RMgX (2.0 equiv., 1.0 M in THF), 66 °C, RMgX added dropwise over 1–2 s. b Me-MgCl (3.0 M in THF). c R-MgCl (2 M in THF). d c-HexMgBr (1 M in 2-MeTHF). | ||||||
| 1b | IPr*OMe·HCl | MeMgCl | 30 min | 2v |
|
96 |
| 2c | IPr*OMe·HCl | n-BuMgCl | 30 min | 2w |
|
96 |
| 3c | IPr*·HCl | 88 | ||||
| 4c | IPr·HCl | 34 | ||||
| 5c | IPr*OMe·HCl | n-HexMgCl | 30 min | 2x |
|
92 |
| 6d | IPr*OMe·HCl | c-HexMgCl | 30 min | 2y |

|
97 |
| 7 | IPr*OMe·HCl | C14H29MgCl | 30 min | 2z |
|
81 |
We also evaluated the highly challenging cross-coupling of bis-C–O electrophiles (Table 4). The NiF2/IPr*MeO catalytic system enables double C(sp2)–C(sp2) cross-coupling of variously substituted naphthyl electrophiles (Table 4, entries 1, 3–5) and is also feasible for double C(sp2)–C(sp3) cross-coupling (Table 4, entry 2) and double C–O cross-coupling of unactivated electrophiles (Table 4, entry 6), affording terphenyl and dialkylaryl products that have broad application in advanced materials and liquid crystals.
| Entrya | Substrate | R-MgX | Time | 2 | Product | Yield (%) |
|---|---|---|---|---|---|---|
| a Conditions: 1 (0.25 mmol), [Ni] (10 mol%), IPr*OMe·HCl (20 mol%), p-TolylMgBr (4 equiv., 1.0 M in THF), 66 °C, RMgX added dropwise over 1–2 s. | ||||||
| 1 |

|
p-TolylMgBr | 1 h | 2aa |

|
98 |
| 2 | n-BuMgCl | 1 h | 2ab |

|
59 | |
| 3 |
|
p-TolylMgBr | 1 h | 2ac |
|
88 |
| 4 |
|
p-TolylMgBr | 1 h | 2ad |
|
81 |
| 5 |
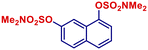
|
p-TolylMgBr | 1 h | 2ae |
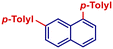
|
75 |
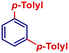
| 6 |
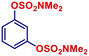
|
p-TolylMgBr | 1 h | 2af |
|
58 |
Preliminary mechanistic experiments were conducted to gain insight into the selectivity of the cross-coupling (Schemes 1 and 2). First, intermolecular competition experiments between aryl Grignard reagents established that electron-deficient nucleophiles are preferred in the C(sp2)–C(sp2) cross-coupling (p-Me![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) p-MeO = 54
p-MeO = 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46, Scheme 1A; p-F
46, Scheme 1A; p-F![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) p-Me = 52
p-Me = 52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 48, Scheme 1B; p-F
48, Scheme 1B; p-F![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) p-MeO = 55
p-MeO = 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45, Scheme 1C), consistent with reductive elimination as a kinetically relevant step. Second, further competition experiments indicated that alkyl nucleophiles cross-couple preferentially (n-Bu
45, Scheme 1C), consistent with reductive elimination as a kinetically relevant step. Second, further competition experiments indicated that alkyl nucleophiles cross-couple preferentially (n-Bu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) p-Tol = 88
p-Tol = 88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12) (Scheme 1D). Furthermore, competition experiments between unactivated aryl sulfamates and aryl carbamates indicated full selectivity for the sulfamate cross-coupling (OSO2NMe2
12) (Scheme 1D). Furthermore, competition experiments between unactivated aryl sulfamates and aryl carbamates indicated full selectivity for the sulfamate cross-coupling (OSO2NMe2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) OCONMe2 > 95
OCONMe2 > 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). This synthetically useful selectivity is consistent with the kinetic experiments (Fig. 2) and is characteristic to the Ni–IPr*MeO catalyst system, while other catalysts typically cannot distinguish between sulfamate and carbamate electrophiles. Finally, isomerization experiments using i-PrMgCl indicated that β-hydride elimination occurs at a rate similar to the cross-coupling (linear
5). This synthetically useful selectivity is consistent with the kinetic experiments (Fig. 2) and is characteristic to the Ni–IPr*MeO catalyst system, while other catalysts typically cannot distinguish between sulfamate and carbamate electrophiles. Finally, isomerization experiments using i-PrMgCl indicated that β-hydride elimination occurs at a rate similar to the cross-coupling (linear![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) branched = 58
branched = 58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 48, Scheme 2).
48, Scheme 2).
 | ||
| Scheme 1 Selectivity studies of Ni–NHC cross-coupling reactions. (A) p-Me vs. p-MeO, (B) p-F vs. p-Me, (C) p-F vs. p-MeO, (D) n-Bu vs. p-Tol, (E) OSO2NMe2vs. OCONMe2. | ||
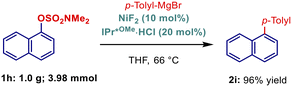
Importantly, scalability experiments demonstrated that the reaction can be readily performed on a gram scale without any modification of the reaction conditions, affording the desired product in 96% yield (Scheme 3).
Furthermore, this approach of C–O cross-coupling can be exploited in the sequential orthogonal cross-couplings using the same Ni–NHC reagent system (Scheme 4). Thus, the C–Br cross-coupling of 6-bromo-2-naphthol afforded the first cross-coupling in the presence of the unprotected hydroxyl group, which was followed by C–O protection with sulfamoyl chloride and C–O cross-coupling in 50% yield.
Finally, although our focus was on establishing an Ni–NHC system for C–O/C–C sulfamate cross-coupling, we are cognizant of the tremendous importance of Buchwald–Hartwig amination methods in pharmaceutical industry and the extreme scarcity of Ni-catalyzed amination methods of C–O electrophiles. Our preliminary studies indicate that the present Ni–IPr*MeO system is also capable of the C–O/C–N Buchwald–Hartwig amination of unactivated sulfamates. The order of reactivity (IPr*MeO > IPr* >> IPr) mirrors the findings from the C–O/C–C cross-coupling, indicating a generality of this catalyst system (Scheme 5A). Furthermore, the reaction with sulfamate 1a (Scheme 5B) unequivocally confirmed that under our conditions the reaction proceeds via C–O bond cleavage, without any C–B bond cleavage product.
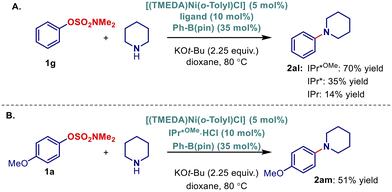 | ||
| Scheme 5 Ni–NHC catalyzed Buchald–Hartwig amination: (A) using Ph–OSO2NMe2, (B) using MeO–Ph–OSO2NMe2. | ||
In summary, although C–O electrophiles represent a powerful orthogonal class of substrates for cross-coupling reactions, there is a shortage of efficient Ni-catalyzed systems for the cross-coupling of bench-stable, readily accessible and orthogonal C–O electrophiles. In this manuscript, we have reported the C(sp2)–C(sp2) and C(sp2)–C(sp3) Kumada–Coriu and Buchwald–Hartwig cross-coupling of aryl sulfamates enabled by sterically-demanding and electron-rich N-heterocyclic carbenes. Crucially, this study identified sulfamates as the most reactive C–O electrophiles enabled by a combination of sterically-demanding IPr*OMe featuring ortho-diphenyl wingtip substitution. Comparative and kinetic studies demonstrate that electron-rich, sterically-demanding N-heterocyclic carbenes show high reactivity for the challenging C–O cross-coupling obviating common issues, such as hydrolysis, homocoupling and unselective C–O and S–N cleavages. The generality of the catalyst system has been demonstrated by the range of C(sp2)–C(sp2) and C(sp2)–C(sp3) C–O/C–C cross-couplings as well as by the C–O/C–N amination, where IPr*OMe outperforms less electron-donating and less sterically-demanding carbenes. The broad availability to NHC ligands with ortho-diphenyl wingtip substitution represents an attractive entryway for the development of Ni-catalyzed cross-coupling processes for academic and industrial research.
Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information: Experimental details and characterization data. See DOI: https://doi.org/10.1039/D5CY00685FAll experimental data supporting the findings of this study are available within the article and its SI files. Additional raw data are available from the corresponding author upon reasonable request.
Acknowledgements
We gratefully acknowledge Narodowe Centrum Nauki (grant no. 2019/35/D/ST4/00806), Rutgers University and the NIH (R35GM133326) for generous financial support.Notes and references
- C. C. C. Johansson Seechurn, M. O. Kitching, T. J. Colacot and V. Snieckus, Angew. Chem., Int. Ed., 2012, 51, 5062 CrossRef CAS PubMed.
- For application of cross-coupling see: (a) P. Ruiz-Castillo and S. L. Buchwald, Chem. Rev., 2016, 116, 12564 CrossRef CAS; (b) A. Piontek, E. Bisz and M. Szostak, Angew. Chem., Int. Ed., 2018, 57, 11116 CrossRef CAS; (c) L. Zani, A. Dessi, D. Franchi, M. Calamante, G. Reginato and A. Mordini, Coord. Chem. Rev., 2019, 392, 177 CrossRef CAS; (d) M. J. Buskes and M. J. Blanco, Molecules, 2020, 25, 3493 CrossRef CAS; (e) R. Emadi, A. B. Nekoo, F. Molaverdi, Z. Khorsandi, R. Sheibani and H. Sadeghi-Aliabadi, RSC Adv., 2023, 13, 18715 RSC; (f) V. Ravichandiran and A. Jana, Org. Chem. Front., 2023, 10, 267 RSC; (g) J. Magano and J. R. Dunetz, Chem. Rev., 2011, 3, 2177 CrossRef; (h) F. Izquierdo, S. Manzini and S. P. Nolan, Chem. Commun., 2014, 50, 14926 RSC.
- For general reviews, see: A. de Meijere, S. Bräse and M. Oestreich, Metal-Catalyzed Cross-Coupling Reactions and More, Wiley, New York, 2014 Search PubMed; G. A. Molander, J. P. Wolfe and M. Larhed, Science of Synthesis: Cross-Coupling and Heck-Type Reactions, Thieme, Stuttgart, 2013 Search PubMed; see ref. 1.
- For Pd-catalyzed cross-coupling see: (a) T. J. Colacot, New Trends in Cross-Coupling, The Royal Society of Chemistry, 2015 Search PubMed; (b) C. E. I. Knappke and A. J. von Wangelin, Chem. Soc. Rev., 2011, 40, 4948 RSC; (c) H. Yu, Y. Ji and M. Hanas, Synth. Commun., 2021, 51, 2732 CrossRef CAS; (d) P. Devendar, R. Y. Qu, W. M. Kang, B. He and G. Y. Yang, J. Agric. Food Chem., 2018, 66, 8914 CrossRef CAS; (e) C. Torborg and M. Beller, Adv. Synth. Catal., 2009, 351, 3027 CrossRef CAS.
- For Ni catalysis, see: (a) S. Z. Tasker, E. A. Standley and T. F. Jamison, Nature, 2014, 509, 299 CrossRef CAS PubMed; (b) V. P. Ananikov, ACS Catal., 2015, 5, 1964 CrossRef CAS; (c) J. B. Diccianni and T. Diao, Trends Chem., 2019, 1, 830 CrossRef CAS; (d) Y. Wang, Y. He and S. Zhu, Acc. Chem. Res., 2023, 56, 3475 CrossRef CAS; (e) R. Kranthikumar, Organometallics, 2022, 41, 667 CrossRef CAS.
- (a) J. W. Dankwardt, Angew. Chem., 2004, 116, 2482 CrossRef; (b) J. A. Miller and J. W. Dankward, Tetrahedron Lett., 2003, 44, 1907 CrossRef CAS.
- (a) S. Sengupta, M. Leite, D. S. Raslan, C. Quesnelle and V. Snieckus, J. Org. Chem., 1992, 57, 4066 CrossRef CAS; (b) R. R. Milburn and V. Snieckus, Angew. Chem., Int. Ed., 2004, 43, 888 CrossRef CAS PubMed; (c) T. K. Macklin and V. Snieckus, Org. Lett., 2005, 13, 2519 CrossRef.
- (a) A. L. Silberstein, S. D. Ramgren and N. K. Garg, Org. Lett., 2012, 14, 3796 CrossRef CAS PubMed; (b) T. Mesganaw and N. K. Garg, Org. Process Res. Dev., 2013, 17, 29 CrossRef CAS; (c) L. Hie, S. D. Ramgren, T. Mesganaw and N. K. Garg, Org. Lett., 2012, 14, 4182 CrossRef CAS.
- (a) B. T. Guan, X. Y. Lu, D. G. Yu, T. Wu, K. L. Li, B. J. Li and Z. J. Shi, Org. Lett., 2010, 12, 396 CrossRef CAS PubMed; (b) Z. C. Cao, Q. Y. Luo and Z. J. Shi, Org. Lett., 2016, 18, 5987 Search PubMed; (c) F. Zhao, Y. F. Zhang, J. Wen, D. G. Yu, J. B. Wei, Z. Xi and Z. J. Shi, Org. Lett., 2013, 15, 3230 CrossRef CAS PubMed.
- (a) M. Tobisu, T. Takahira, A. Ohtsuki and N. Chatani, Org. Lett., 2015, 17, 680 CrossRef CAS; (b) M. Tobisu, T. Takahira and N. Chatani, Org. Lett., 2015, 17, 4352 CrossRef; (c) A. Ohtsuki, K. Yanagisawa, T. Furukawa, M. Tobisu and N. Chatani, J. Org. Chem., 2016, 81, 9409 CrossRef; (d) K. Nakamura, M. Tobisu and N. Chatani, Org. Lett., 2015, 17, 6142 CrossRef.
- (a) M. J. Iglesias, A. Prieto and M. C. Nicasio, Org. Lett., 2012, 14, 4318 CrossRef; (b) J. Zhang, J. Xu, Y. Xu, H. Sun, Q. Shen and Y. Zhang, Organometallics, 2015, 34, 5792 CrossRef; (c) L. G. Xie and Z. X. Wang, Chem. – Eur. J., 2011, 17, 4972 CrossRef; (d) A. Piontek, W. Ochędzan-Siodłak, E. Bisz and M. Szostak, Adv. Synth. Catal., 2019, 361, 2329 CrossRef; (e) D. Ghorai, J. Loup, G. Zanoni and L. Ackrermann, Synlett, 2019, 30, 429 CrossRef; (f) R. Ambre, H. Yang, W. C. Chen, G. P. A. Yap, T. Jurca and T. G. Ong, Eur. J. Inorg. Chem., 2019, 30, 3511 CrossRef.
- J. Diccianni, Q. Lin and T. Diao, Acc. Chem. Res., 2020, 53, 906 CrossRef PubMed.
- (a) G. J. Chen and F. S. Han, Eur. J. Org. Chem., 2012, 3575 CrossRef; (b) H. H. Ke, X. F. Chen, Y. Y. Feng and G. Zou, Sci. China:Chem., 2014, 57, 1126 CrossRef; (c) P. M. McQueen and M. Stradiotto, Synlett, 2017, 28, 1652 CrossRef.
- (a) A. Monti, J. López-Serrano, A. Prieto and M. C. Nicasio, ACS Catal., 2023, 13, 10945 CrossRef PubMed; (b) P. R. Melvin, A. Nova, D. Balcells, N. Hazari and M. Tilset, Organometallics, 2017, 36, 3664 CrossRef PubMed.
- (a) M. Kardela, K. Halikowska-Tarasek, M. Szostak and E. Bisz, Catal. Sci. Technol., 2022, 12, 7275 RSC; (b) M. Kardela, B. Dziuk, R. Szostak, M. Szostak and E. Bisz, Catal. Sci. Technol., 2024, 14, 7002 RSC; (c) P. Podc'horodecka, B. Dziuk, R. Junga, R. Szostak, M. Szostak and E. Bisz, Dalton Trans., 2024, 53, 14975 RSC; (d) P. Podchorodecka, B. Dziuk, R. Szostak, M. Szostak and E. Bisz, Dalton Trans., 2023, 52, 13608 RSC.
- (a) D. J. Nelson and S. P. Nolan, Chem. Soc. Rev., 2013, 42, 6723 RSC; (b) S. Diez-Gonzalez and S. P. Nolan, Coord. Chem. Rev., 2007, 251, 874 CrossRef; (c) H. Jacobsen, A. Correa, A. Poater, C. Costabile and L. Cavallo, Coord. Chem. Rev., 2009, 253, 687 CrossRef; (d) T. Dröge and F. Glorius, Angew. Chem., Int. Ed., 2010, 49, 6940 CrossRef PubMed.
- (a) H. Clavier and S. P. Nolan, Chem. Commun., 2010, 46, 841 RSC; (b) A. Gomez-Suarez, D. J. Nelson and S. P. Nolan, Chem. Commun., 2017, 53, 2650 RSC; (c) G. Berthon-Gelloz, M. A. Siegler, A. L. Spek, B. Tinant, J. N. H. Reek and I. E. Markó, Dalton Trans., 2010, 39, 1444 RSC.
- (a) D. Martin, M. Melaimi, M. Soleilhavoup and G. Bertrand, Organometallics, 2011, 30, 5304 CrossRef PubMed; (b) M. Melaimi, M. Soleihavoup and G. Bertrand, Angew. Chem., Int. Ed., 2010, 49, 8810 CrossRef PubMed; (c) M. Soleilhavoup and G. Bertrand, Acc. Chem. Res., 2015, 48, 256 CrossRef PubMed; (d) J. Cheng, L. Wang and L. Deng, Chem. Rev., 2018, 118, 9930 CrossRef PubMed; (e) K. Matsubara, H. Yamamoto, S. Miyazaki, T. Inatomi, K. Nonaka, Y. Koga, Y. Yamada, L. F. Veiros and K. Kirchner, Organometallics, 2017, 36, 255 CrossRef.
- B. C. Lee, C. F. Liu, L. Q. H. Li, K. Z. Yap, N. Song, C. H. M. Ko, P. H. Chan and M. J. Koh, Chem. Soc. Rev., 2023, 52, 2946 RSC.
- (a) T. Hatakeyama, S. Hashimoto, K. Ishizuka and M. Nakamura, J. Am. Chem. Soc., 2009, 131, 11949 CrossRef PubMed; (b) T. Hatakeyama, K. Ishizuka and M. Nakamura, J. Synth. Org. Chem., Jpn., 2011, 69, 1282 CrossRef.
- A. R. Martin, Y. Makida, S. Meiries, A. M. Z. Slawin and S. P. Nolan, Organometallics, 2013, 32, 6265 CrossRef.
- (a) A. M. Borys and E. Hevia, Chem. Commun., 2024, 60, 11052 RSC; (b) N. Yoshikai, H. Mashima and E. Nakamura, J. Am. Chem. Soc., 2005, 127, 17978 CrossRef PubMed; (c) S. Lutz, L. Nattmann, N. Nöthing and J. Cornella, Organometallics, 2021, 40, 2220 CrossRef; (d) L. Nattamann, S. Lutz, P. Ortsack, R. Goddard and J. Cornella, J. Am. Chem. Soc., 2018, 140, 13628 CrossRef.
- C. Uthayopas and P. Surawatanawong, Dalton Trans., 2019, 48, 7817 RSC.
| This journal is © The Royal Society of Chemistry 2025 |