One-pot synthesis of confined structure Ru3Sn7 alloys on alumina for exceptionally rapid and selective hydrogenolysis of furfuryl alcohol to 1,5-pentanediol†
Rodiansono
 *ab,
Atina Sabila
Azzahra
*ab,
Atina Sabila
Azzahra
 a,
Edi
Mikrianto
ab,
Kiky Corneliasari
Sembiring
c,
Ahmad
Afandi
d,
Gagus Ketut
Sunnardianto
e and
Indri Badria
Adilina
a,
Edi
Mikrianto
ab,
Kiky Corneliasari
Sembiring
c,
Ahmad
Afandi
d,
Gagus Ketut
Sunnardianto
e and
Indri Badria
Adilina
 *c
*c
aCatalysis for Sustainable Energy & Environment (CATSuRe), Inorganic Materials & Catalysis (IMCat) Laboratory, Lambung Mangkurat University, Indonesia. E-mail: rodiansono@ulm.ac.id
bDepartment of Chemistry, Lambung Mangkurat University, Jl. A. Yani Km 36, Banjarbaru, 70714, Indonesia. Tel: +62 511 477 3112
cResearch Centre for Chemistry, BRIN, KST BJ Habibie, Serpong, Tangerang Selatan, Indonesia. E-mail: indr030@brin.go.id
dResearch Centre for Advanced Materials, BRIN, KST BJ Habibie, Serpong, Tangerang Selatan, Indonesia
eResearch Centre for Quantum Physics, BRIN, KST BJ Habibie, Serpong, Tangerang Selatan, Indonesia
First published on 24th July 2025
Abstract
A facile one-pot synthesis of confined structure Ruthenium–tin alloy catalysts on alumina ((op)Ru–(x)Sn@Al2O3) has been developed for highly efficient and selective synthesis of 1,5-pentanediol (1,5-PeD) from furfuryl alcohol (FFalc). A series of (op)Ru–(x)Sn@Al2O3 (x = Sn co-loading; 0.37–2.65 wt%) catalysts were synthesised via a one-pot coprecipitation-hydrothermal method of a solution containing ruthenium chloride and tin chloride and a hydrargillite-type of aluminium hydroxide (Al(OH)3) at 150 °C for 24 h, followed by reduction with H2 at 400 °C for 2 h. The (op)Ru–(1.30)Sn@Al2O3 catalyst obviously allowed the highest yield of 1,5-PeD (97%) with a productivity of 3.67 mmol 1,5-PeD per gcat.per min from FFalc in water at 140 °C and 10 bar H2, after 3 h. The presence of Ru3Sn7 or Ru–SnOx catalysed the partial hydrogenation of the furan ring of FFalc to form 4,5-dihydrofuranmethanol (4,5-DHFM), and the acidic species of SnOx or Snn+ or acidic Al2O3 were used for activating C2–O for ring opening processes, while Ru0 was used for the subsequent hydrogenation process, leading to high yield of the final 1,5-PeD product. This is the highest reported yield of 1,5-PeD from one-pot hydrogenolysis of FFalc under mild conditions. Catalytic performance of the recycled (op)Ru–(1.30)Sn@AL2O3 catalyst was restored after reactivation with H2 at 400 °C for 1 h.
1. Introduction
Alpha–omega (α,ω)-C5-diols (e.g., 1,2-, 1,4- or 1,5-pentanediol (PeD)) have gained significant attention as versatile precursors for the production of various materials, including polyesters, polyurethanes, plasticizers, cosmetics, printing inks, and fungicides.1,2 Among these (α,ω)-C5-diols, 1,5-PeD is particularly important and can be synthesised from C5-furans (e.g., furfuraldehyde (FFald), furfuryl alcohol (FFalc), and tetrahydrofurfuryl alcohol (THFalc). However, achieving a high selectivity and high yield synthesis of 1,5-PeD from either FFald or FFalc is challenging due to the reactivity of the furan ring and the complex interactions on the catalyst surface.3–5 Tomishige et al. highlighted that increasing the selectivity of 1,5-PeD or 1,2-PeD from FFalc is associated with metal active sites and the basicity or acidity of catalysts used.6 Nevertheless, metal–acid catalysts with strong acidity led to the hydrogenative rearrangement of the furan ring to C5-cyclic compounds such as cyclopentanone (CPO) or cyclopentanol (CPL).7 Consequently, designing and controlling catalyst behaviour to achieve high selectivity for 1,2-PeD or 1,5-PeD remains a significant challenge.Catalysts based on supported precious metals such as Pt, Ir, Rh, Pd, or Ru, either in monometallic or bimetallic form, have been extensively investigated for the synthesis of 1,2-PeD and 1,5-PeD.8–12 For instance, a Pt/CeO2-nanocube catalyst with exposed (100) ceria-terminal facets produced a significantly higher yield of 1,2-PeD (77%) compared to Pt/CeO-nanorods and Pt/CeO-octahedron.8 However, controlling the crystal plane of CeO2 and the chemical state of Pt poses challenges for further fabrication and industrial application. Additionally, a spinel-type Pt/CoAl2O4 catalyst, obtained through the coprecipitation method, was employed for the direct conversion of FFald. This catalyst yielded a mixture of 1,2-PeD (16%) and 1,5-PeD (35%) at 140 °C and 10 bar H2 for 24 h.13 Various Ir-, Rh-, Pd-, and Ru-based catalysts have also been developed by Tomishige and co-workers for the hydrogenolysis of FFald and its derivatives (e.g., FFalc and THFalc) with a preference for higher selectivity of 1,5-PeD over 1,2-PeD.11,14,15 They hypothesised that 1,5-PeD or 1,2-PeD was produced through the ring-cleavage of the saturated C–O bond in THFalc depending on the acidity or basicity of the catalyst systems.16,17 Dumesic and co-worker proposed multi-step reaction pathways for 1,5-PeD synthesis from FFald, which involve total hydrogenation of FFald to THFalc by using Ni- or Ru-based catalysts, dehydration of THFalc to dihydropyran (DHP) on γ-Al2O3, hydration–tautomerisation–ring opening of DHP to 5-hydroxy valeraldehyde (5-HY-Val), and finally the hydrogenation of 5-HY-Val to 1,5-PeD on a Ru/C catalyst.18 Since there are two competitive reaction pathways: the saturation of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond to form THFalc and the cleavage of the C–O bond leading to 1,2-PeD and 1,5-PeD, the exploration of heterogeneous catalysts that allow the one-step reaction of FFald or FFalc towards high yield of 1,5-PeD is still greatly challenging.
C bond to form THFalc and the cleavage of the C–O bond leading to 1,2-PeD and 1,5-PeD, the exploration of heterogeneous catalysts that allow the one-step reaction of FFald or FFalc towards high yield of 1,5-PeD is still greatly challenging.
Several non-precious heterogeneous metal-based catalysts, including layered-double hydroxides or hydrotalcite structures have been intensively developed for the synthesis of 1,5-PeD from FFald or FFalc.19–21 They proposed that controlling the basicity and increasing the density of low-coordination oxygen anions on the support led to higher basicity which facilitated the activation of the furan ring. For example, layered La-doped Cu–Co–Al hydrotalcites (LDHs),19 the hydrotalcite-derived Ni–Co–Al,20 and the Ni–CoAlOx (ref. 21) system with metallic Ni and CoAlOx demonstrated a synergistic action between Cu0, CoOx, and the basicity of metal oxides towards partial hydrogenation of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C furan ring, followed by the cleavage of the C2–O bond to selectively produce 1,5-PeD. The Cu–Mg3AlO4.5 catalyst can function as a bifunctional catalyst for the hydrogenolysis of FFalc, achieving an 80% yield of the 1,2-PeD and 1,5-PeD mixture.22 Bimetallic oxides of perovskites or spinel structure catalysts such as CoOx–PtOx23 and Co–CoOx systems derived from MOFs,24 and La-doped CuCoAl25 systems, along with interfacial metal oxides and metals have preferentially interacted with the –CH2OH group of FFalc. This interaction induced the C2–O oblique adsorption during the cleavage of the C2–O bond, leading to yield 1,5-PeD (40–60%) that was higher than 1,2-PeD. Specifically, the presence of the Cu–La interface in La-doped CuCoAl can simultaneously activate the furan ring and the –OH group in FFalc with an intermediate six-membered ring transition state, leading to high selectivity of 1,5-PeD with a yield up to 60% at 160 °C and 40 bar H2 for 2 h.25 Furthermore, Dai et al. claimed that the combination of balanced Cu0–Cu+ and Brønsted acid sites in the Cu/MFI catalyst afforded a 69% yield of 1,5-PeD at 160 °C and 25 bar H2.26 Wang et al. introduced a K promoter to form xK–Cu/MgO (x = 0–2 wt%), raising the yield of 1,5-PeD from 21% to 36% at 150 °C and 30 bar H2 for 4 h.27 Despite the development of several heterogeneous catalysts, the catalytic reactions inevitably produced moderate yield of 1,5-PeD (60%) with the yields of 1,2-PeD and THFalc still remaining noticeable.
C furan ring, followed by the cleavage of the C2–O bond to selectively produce 1,5-PeD. The Cu–Mg3AlO4.5 catalyst can function as a bifunctional catalyst for the hydrogenolysis of FFalc, achieving an 80% yield of the 1,2-PeD and 1,5-PeD mixture.22 Bimetallic oxides of perovskites or spinel structure catalysts such as CoOx–PtOx23 and Co–CoOx systems derived from MOFs,24 and La-doped CuCoAl25 systems, along with interfacial metal oxides and metals have preferentially interacted with the –CH2OH group of FFalc. This interaction induced the C2–O oblique adsorption during the cleavage of the C2–O bond, leading to yield 1,5-PeD (40–60%) that was higher than 1,2-PeD. Specifically, the presence of the Cu–La interface in La-doped CuCoAl can simultaneously activate the furan ring and the –OH group in FFalc with an intermediate six-membered ring transition state, leading to high selectivity of 1,5-PeD with a yield up to 60% at 160 °C and 40 bar H2 for 2 h.25 Furthermore, Dai et al. claimed that the combination of balanced Cu0–Cu+ and Brønsted acid sites in the Cu/MFI catalyst afforded a 69% yield of 1,5-PeD at 160 °C and 25 bar H2.26 Wang et al. introduced a K promoter to form xK–Cu/MgO (x = 0–2 wt%), raising the yield of 1,5-PeD from 21% to 36% at 150 °C and 30 bar H2 for 4 h.27 Despite the development of several heterogeneous catalysts, the catalytic reactions inevitably produced moderate yield of 1,5-PeD (60%) with the yields of 1,2-PeD and THFalc still remaining noticeable.
Recently, we reported on bimetallic ruthenium–tin nanoparticles supported on gamma-alumina catalysts synthesised by a coprecipitation-hydrothermal method (Ru–(y)SnOx/γ-Al2O3, where y represents the Sn co-loading; 0.65–2.15 wt%) for the aqueous phase hydrogenolysis of FFalc to 1,5-PeD. The Ru–(1.30)SnOx/γ-Al2O3 catalyst, with a precise Sn co-loading, revealed the presence of Ru0, Ru3Sn7 and SnOx species that yielded exclusively 1,5-PeD (up to 94%) at 180 °C and 30 bar H2 in H2O after 7 h.28 Attenuated total reflectance-infrared (ATR-IR) spectra of the reaction mixture under controlled reaction conditions exhibited a sharp absorption peak at 1637 cm−1, which is assigned to trisubstituted C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C in the 4,5-dihydrofuranmethanol (4,5-DHFM) intermediate. This intermediate was consistently observed during the kinetic studies of reactions as shown in Scheme 1.29 Furthermore, it was noted that THFalc was completely diminished alongside the evaluation of reaction parameters, suggesting a new reaction pathway for the formation of 1,5-PeD from FFald or FFalc that differs from the previously established pathways.11,30
C in the 4,5-dihydrofuranmethanol (4,5-DHFM) intermediate. This intermediate was consistently observed during the kinetic studies of reactions as shown in Scheme 1.29 Furthermore, it was noted that THFalc was completely diminished alongside the evaluation of reaction parameters, suggesting a new reaction pathway for the formation of 1,5-PeD from FFald or FFalc that differs from the previously established pathways.11,30
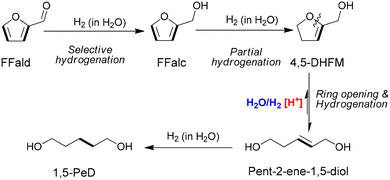 | ||
| Scheme 1 Proposed reaction pathways of furfural into 1,5-PeD over bimetallic Ru–Sn alloy catalysts.28 | ||
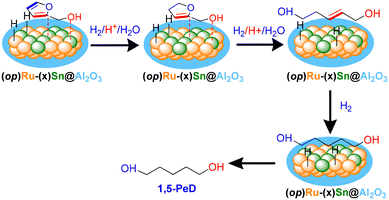
Herein, we describe our extended investigation into the development of confined structure bimetallic Ru3Sn7 alloy catalysts supported on alumina for an exceptionally selective synthesis of 1,5-PeD from FFalc under mild conditions (140–160 °C, 10–20 bar H2, and 3–6 h). The confined structure Ru3Sn7 catalysts on the alumina structure were synthesised using a one-pot coprecipitation-hydrothermal method (op), followed by reduction with H2 at 400 °C for 2 h and produced the (op)Ru–(x)Sn@Al2O3 (x = Sn co-loading; 0.37–2.65 wt%) as shown in the schematic diagram (Fig. 1). Detailed experimental procedures of catalyst preparation, characterisation, and catalytic reactions are available in the ESI.† The reference Ru–(x)Sn/Al2O3 catalysts were synthesised using a coprecipitation-hydrothermal method (cop) producing (cop)Ru–(x)Sn/Al2O3-500 or (cop)Ru–(x)Sn/γ-Al2O328 and a wet-impregnation method (imp) producing (imp)Ru–(x)Sn/Al2O3-500. The (cop)Ru–(1.30)Sn@γ-Al2O3-500 catalyst achieved a yield of 81% for 1,5-PeD, whilst the (imp)Ru–(1.30)Sn@Al2O3-500 catalyst produced only 13%. Surprisingly, the (op)Ru–(1.30)Sn@Al2O3 catalyst gave the highest yield of 1,5-PeD at 97%. Although the catalytic performance of the recycled (op)Ru–(1.30)Sn@AL2O3 catalyst can be restored after the reactivation with H2 at 400 °C for 1 h, the recyclability of the catalyst remains low. Therefore, systematic investigations to improve the recyclability of the catalyst after repeated reactions are essential for future investigation.
 | ||
| Fig. 1 Schematic diagram for the one-pot synthesis of confined structure Ru3Sn7 on alumina (Ru–(x)Sn@Al2O3; x = Sn co-loading (wt%)). | ||
2. Results and discussion
2.1. Catalyst characterisation
A series of (op)Ru–(x)Sn@Al2O3 catalysts with different Sn co-loadings (x = 0.37–2.65 wt%) were synthesised by using a one-pot coprecipitation-hydrothermal method (op), followed by reduction with H2 at 400 °C for 2 h, and the XRD patterns are shown in Fig. 2.The sole structure of γ-Al2O3 or Al2O3 with a high intensity diffraction peak at 2θ = 67° is clearly observed in all samples (JCPDS #10-0425). This indicates that the Al(OH)3 hydrargillite structure was perfectly transformed into γ-Al2O3 or Al2O3 during the catalyst preparation (see Fig. S1 in the ESI†). A series of sharp diffraction peaks at 2θ = 39.8, 43.5, 57.4, 72.1, and 85.7°, which can be attributed to Ru3Sn7(411), Ru3Sn7(420), Ru3Sn7(530), Ru3Sn7(721), and Ru3Sn7(653), respectively, were obtained (JCPDS #26-0504). The diffraction peaks of Ru3Sn7 alloy phases were intensified with an increase in Sn co-loading (Fig. 2(c and d)). The crystallite sizes of Ru3Sn7(411) at 2θ = 39.8° were determined to be 30.5 nm and 33.1 nm for (op)Ru–(1.30)Sn@Al2O3 and (op)Ru–(2.65)Sn@Al2O3 samples, respectively. The XRD patterns of the original Al(OH)3 hydrargillite structure, along with its transformation into amorphous Al2O3 (γ-Al2O3) after calcination at 500 °C under N2 for 2 h, are observed in Fig. S1(A), in the ESI.† The XRD patterns of monometallic (op)Ru@Al2O3 and (op)Sn@Al2O3 are shown in Fig. S1(B) in the ESI.† Additionally, the (cop)Ru–(1.30)Sn@Al2O3 sample exhibited a diffraction peak at 2θ = 43.6° suggesting the formation of the Ru3Sn7(420) alloy phase (Fig. S2(a) in the ESI†), whilst the (imp)Ru–(1.30)Sn@Al2O3 sample revealed Ru0 species only (Fig. S2(b) in the ESI†). To gain insight into the existence of metallic Ru, Ru–Sn, and SnOx species, X-ray photoelectron spectroscopy (XPS) analysis was performed on representative fresh and spent (op)Ru–(1.30)Sn@Al2O3 catalysts. The XPS spectra and the binding energies (BEs) of Ru 3d5/2 and Sn 3d5/2 in the fresh and spent (op)Ru–(1.30)Sn@Al2O3 are summarised in Fig. 3 and Table 1.
 | ||
| Fig. 3 XPS spectra of fresh and spent (op)Ru–(1.30)Sn@Al2O3 samples: (A) wide range spectra, (B) Ru 3d5/2, (C) Sn 3d5/2, and (D) Al 2p after reduction with H2 at 400 °C for 2 h. | ||
| Sample | Binding energy (eV) | ||||
|---|---|---|---|---|---|
| Ru0 3d5/2 | Ruδ+ 3d5/2 | Sn 3d5/2 | Sn 3d3/2 | Al 2p | |
| Fresh | 277.73 | 278.21 | 483.65 (Sn0) | 491.37 | 71.57 |
| 282.77 (C 1s) | 484.25 | 492.64 | 72.33 | ||
| 485.11 (Sn4+) | 493.10 (Sn4+) | ||||
| Spent | 277.75 | 278.61 | 482.89 | 491.66 | 71.65 |
| 282.60 (C 1s) | 484.17 | 492.68 | 72.41 | ||
| 485.22 (Sn4+) | 493.73 (Sn4+) | ||||
The signal of the Ru 3d3/2 orbital overlaps with that of the C 1s orbital; therefore, the Ru 3d5/2 peak was used to determine the chemical states of Ru species in all the catalyst. The BEs of Ru 3d5/2 in both fresh and spent (op)Ru–(1.30)Sn@Al2O3 catalysts were observed at 277.82 eV and 277.75 eV, respectively, corresponding to the Ru0 species. A peak between 278.61 and 278.68 eV was also observed, which can be attributed to the oxidative Ruδ+ species.31 For the BE of Sn 3d, two centred peaks were identified: Sn 3d5/2 and Sn 3d3/2 at 484.66 eV and 493.01 eV, respectively, corresponding to oxidized Sn species (Snδ+).32 Additionally, there are weak shoulder peaks at 482.02 eV, 486.81 eV, and 494.18–494.37 eV detected in both fresh and spent (op)Ru–(1.30)Sn@Al2O3 samples, corresponding to reduced tin species (Sn0) and Sn4+, respectively. The BE of Ru 3d5/2 in the bimetallic Ru–Sn alloy was noticeably negatively shifted relative to that of monometallic Ru nanoparticles (BE Ru 3d5/2 = 279.75 eV).33 The negatively shifted Ru 3d5/2 and Ru 3d3/2 peaks in spent (op)Ru–(1.30)Sn@Al2O3 indicate the charge transfer from Sn to Ru, resulting in the Ru atoms becoming negatively charged in the Ru–Sn alloy. Consequently, the affinity of Ru atoms in the Ru–Sn alloy towards C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C was greatly suppressed, leading to the inhibition of THFalc formation as clearly evidenced by the reaction profiles. Moreover, the BEs of Al 2p in Al2O3 (ranging from 71.57 to 71.65 eV) and Al(OH)3 (72.33 to 72.44 eV) for both fresh and spent (op)Ru–(1.30)Sn@Al2O3 samples are clearly observed, with a slight decrease in the intensity of those species. This suggest that the Al2O3 or Al(OH)3 species may leach into the reaction mixture.28
C was greatly suppressed, leading to the inhibition of THFalc formation as clearly evidenced by the reaction profiles. Moreover, the BEs of Al 2p in Al2O3 (ranging from 71.57 to 71.65 eV) and Al(OH)3 (72.33 to 72.44 eV) for both fresh and spent (op)Ru–(1.30)Sn@Al2O3 samples are clearly observed, with a slight decrease in the intensity of those species. This suggest that the Al2O3 or Al(OH)3 species may leach into the reaction mixture.28
The physico-chemical properties of the representative catalysts, including bulk compositions, Brunauer–Emmet–Teller (BET) specific surface area (SBET), H2 uptake (derived from H2-TPR data), and total acidity (derived from NH3-TPD spectra), are summarised in Table 2. The SEM–EDS images and spectra of typical fresh and spent (op)Ru–(1.30)Sn@Al2O3 samples are presented in Fig. S3 and S4, in the ESI.† The N2-adsorption–desorption and porosity profiles of three types of alumina supported Ru–(x)Sn alloy catalysts are shown in Fig. S5, in the ESI.† The Ru particle size estimated from the H2 uptake using the proposed equation of Suib et al.34 for the (op)Ru–(1.30)Sn@Al2O3 sample was 7.4 nm (entry 4). These are comparable with the average Ru nanoparticles obtained from HRTEM measurements (2.30 nm and 2.37 nm for fresh and spent, respectively) (see Fig. S6(a), in the ESI†) and the Scherrer equation at 2θ = 39.8° (30.5 nm).
| Entry | Catalysta | Snb (wt%) | Ru/Snb (molar ratio) | S BET (m2 g−1) | H2d (mmol g−1) | Aciditye (mmol NH3 g−1) | D (%) | d va (nm) |
|---|---|---|---|---|---|---|---|---|
| a The values in the parentheses are the amounts of Sn co-loading (wt%), determined by ICP-AES analysis. b Sn co-loading and Ru/Sn molar ratio were calculated from ICP-AES analysis data. c S BET was derived from N2 adsorption–desorption (BET method) at −196 °C. d H2 uptake was derived from H2-TPR spectra. e Acidity was derived from NH3-TPD spectra. f Metal dispersion (%). g The Ru particle sizes (nm) were also estimated from the H2 uptake data using the proposed equation of Suib et al. with an assumption that the surface area was occupied by an exposed (001) Ru surface.34 h Values in the parentheses are the average crystallites sizes of the Ru3Sn7 alloy (2θ = 39.8°) calculated by the Scherrer equation. n.d. = not determined. | ||||||||
| 1 | (op)Ru@Al2O3 | — | 0 | 207 | 6.58 | 498 | 25.0 | 3.8 |
| 2 | (imp)Ru–(1.23)Sn@Al2O3-500 | 1.23 | 3.6 | 192 | 3.59 | 537 | 14.4 | 6.3 |
| 3 | (cop)Ru–(1.30)Sn@γ-Al2O3 | 1.30 | 3.2 | 158 | 5.48 | 798 | 22.0 | 4.1 |
| 4 | (op)Ru–(1.30)Sn@Al2O3 | 1.30 | 3.4 | 142 | 3.03 | 750 | 12.1 | 7.4 (30.5)h |
| 5 | Rec. (op)Ru–(1.30)Sn@Al2O3 | 1.29 | 3.4 | 82 | n.d. | n.d. | n.d. | n.d. |
The NH3-TPD profiles of the three types of synthesised Ru–(x)Sn@Al2O3 catalysts are shown in Fig. 4. The NH3-TPD spectra were formally divided into three temperature regions for desorption, corresponding to three types of acid sites:35 (1) weak acid sites, ranging from 100 to 200 °C, (2) moderate acid sites, ranging from 200 to 550 °C, and (3) strong acid sites, which are above 550 °C (Fig. S7, in the ESI†). The different synthetic procedures used for Ru–Sn may influence surface acidity, as reflected by the total acidity (measured in μmol NH3 per gram) of each catalyst. The total acidity values for unmodified Ru@Al2O3, (imp)Ru–(1.23)Sn@Al2O3-500, (cop)Ru–(1.30)Sn@γ-Al2O3, and (op)Ru–(1.30)Sn@Al2O3 are 498 μmol NH3 per gram, 537 μmol NH3 per gram, 798 μmol NH3 per gram, and 750 μmol NH3 per gram, respectively (Fig. 4(A) and Table 2). Notably, the concentration of strong acidity in the (cop)Ru–(1.30)Sn@γ-Al2O3 and (op)Ru–(1.30)Sn@Al2O3 samples is significantly higher than that in the (imp)Ru–(1.23)Sn@Al2O3-500 and (op)Ru@Al2O3 samples (Fig. S7 and Table S1, in the ESI†). Since NH3-TPD does not allow the differentiation of Lewis and Brønsted acid sites, pyridine-ATR-IR analysis was conducted on the three synthesised catalysts of (imp)Ru–(1.23)Sn@Al2O3-500, (cop)Ru–(1.30)Sn@γ-Al2O3, and (op)Ru–(1.30)Sn@Al2O3 (Fig. 4(B)). According to the literature on pyridine adsorption peaks in Sn-containing catalysts,36,37 the bands are assigned as follows: the pyridinium ion (PyH+), which forms from the reaction of pyridine with Brønsted acid sites (B), shows a band around 1647 cm−1 (v8a); coordinatively bound pyridines on Lewis acid sites (L) show bands around 1445 (v19b) and 1575 cm−1; and physisorbed or hydrogen-bonded pyridine (H) exhibits bands at near 1437 and 1599 cm−1. The band around 1490 cm−1 represents common vibrations from both PyH+ (B) and coordinatively bound pyridine (L).38 The Py-ATR-IR spectrum of (imp)Ru–(1.23)Sn@Al2O3-500 confirmed the sole presence of Lewis acid sites, indicated by bands at 1435, 1480, and 1580 cm−1.39 In contrast, the (cop)Ru–(1.30)Sn@γ-Al2O3 sample displayed the designed bands at 1647 cm−1, signifying the presence of Brønsted acid sites. These Brønsted acid sites were likely generated by the presence of oxidic tin such as Sn(OH) or SnOx during the catalyst preparation or during the catalytic reaction in the aqueous phase.28,29,40,41
 | ||
| Fig. 4 (A) NH3-TPD and (B) pyridine-ATR-IR profiles of Ru–(1.30)Sn@Al2O3 catalysts synthesised with different approaches after reduction with H2 at 400 °C for 2 h. | ||
2.2. Catalytic reactions
The (imp)Ru–(1.30)Sn@Al2O3-500 catalyst produced a yield of 75% for 2-pentanone (2-PeO) and 11% for 4,5-DHFM with low yield of 1,5-PeD (13%). The (cop)Ru–(1.30)Sn@Al2O3-500 catalyst achieved an 81% yield of 1,5-PeD, which is comparable to the Ru–(1.30)Sn/γ-Al2O3(E) catalyst that produced 82% 1,5-PeD at 180 °C and H2 30 bar after 3 h, as reported previously.28 Other products included cyclopentanone (CPO), levulinic acid (LA), and a dimer of FFalc ((d)-FFalc). Surprisingly, the (op)Ru–(1.30)Sn@Al2O3 catalyst gave the highest yield of 1,5-PeD (97%), significantly higher than the yields obtained from FFald or FFalc over Ru–(1.30)Sn/ZrO2 or Ru–(1.30)Sn/TiO2 catalysts.29 In addition, the superiority of the (op)Ru–(1.30)Sn@Al2O3 catalyst over other catalyst systems is clearly observed on the reaction rate of 1,5-PeD (mmol 1,5-PeD per gcat per min). The reaction rate over the (op)Ru–(1.30)Sn@Al2O3 was measured at 3.67 mmol 1,5-PeD per gcat per min, while the (cop)Ru–(1.30)Sn@Al2O3-500 and (imp)Ru–(1.30)Sn@Al2O3-500 catalysts showed lower rates of 1.53 mmol 1,5-PeD per gcat per min and 0.22 mmol 1,5-PeD per gcat per min, respectively. This calls for further evaluation of the effect of Sn co-loading, reaction parameters (temperature, initial H2 pressure, and reaction profiles), and recyclability/reusability of the catalysts. Additionally, the possible reaction pathways of 1,5-PeD formation will be systematically investigated.
 | ||
| Fig. 6 Results of FFalc conversion and yields over (op)Ru–(x)Sn/Al2O3 catalysts with different Sn co-loadings. For the reaction conditions, refer to Fig. 5. | ||
Significant differences in product distributions were observed between (op)Ru@Al2O3 catalysts with and without Sn co-loading. The monometallic (op)Ru@Al2O3 catalyst (Ru = ∼4 wt%) produced 25% THFalc, 10% 4,5-DHFM, and 58% by-products, which included CPO, levulinic acid LA, and a dimer of FFalc, based on GC–MS data. This was achieved at 93% conversion of FFalc at 140 °C and 10 bar H2 for 3 h. When a small amount of Sn (0.37 wt%) was introduced to form (op)Ru–(0.37)Sn@Al2O3, the products were shifted to 34% 1,5-PeD, 37% 4,5-DHFM, 19% THFalc, and 6% by-products at 96% conversion of FFalc. Increasing the Sn co-loading to 0.64 wt% to form the (op)Ru–(0.64)Sn@Al2O3 catalyst further enhance the yield of 1,5-PeD to approximately two-folds (71%) at 96% conversion of FFalc, whereas the yields of 4,5-DHFM, THFalc, and by-products were 7%, 15%, 3%, and 7%, respectively. An impressive yield of 1,5-PeD (97%) at 100% conversion of FFalc was obtained over the (op)Ru–(1.30)Sn@Al2O3 (Sn = 1.30 wt%) catalyst whereas undesired THFalc had completely disappeared, indicating that the Ru–(1.30)Sn catalyst is inactive for complete hydrogenation of the furan ring. This can be attributed to the presence of Ru0, Ru3Sn7 alloy, and SnOx species, as clearly evidenced by the XRD patterns (Fig. 2) and XPS spectra of the (op)Ru–(1.30)Sn@Al2O3 catalyst (Fig. 3 and Table 1). Furthermore, the H2 uptake and the metal dispersion data confirmed its high activity and selectivity (Table 1). We propose that the presence of metallic Ru0, Ru3Sn7, Ru0–SnOx, oxidised Sn or SnOx, molecular water, and acidic Al2O3 support work synergistically to catalyse the partial hydrogenation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds and C2–O cleavage of the furan ring in FFalc during the aqueous phase reaction. The Ru0–SnOx species in the intermetallic Ru–Sn served as Brønsted acid or Lewis acid sites for the C2–O cleavage of FFalc or 4,5-DHFM, leading to the high yield of 1,5-PeD.28,42 Additionally, the precise amount of Sn co-loading to form a bimetallic alloy or intermetallic Ni–Sn (ref. 30 and 43) or RANEY® Ni–Sn (ref. 44) catalysts considerably inhibited the hydrogenation of C
C bonds and C2–O cleavage of the furan ring in FFalc during the aqueous phase reaction. The Ru0–SnOx species in the intermetallic Ru–Sn served as Brønsted acid or Lewis acid sites for the C2–O cleavage of FFalc or 4,5-DHFM, leading to the high yield of 1,5-PeD.28,42 Additionally, the precise amount of Sn co-loading to form a bimetallic alloy or intermetallic Ni–Sn (ref. 30 and 43) or RANEY® Ni–Sn (ref. 44) catalysts considerably inhibited the hydrogenation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds to THFalc and selectively catalysed FFald, FFalc, or 2-MeF to form 1,4-PeD in the H2O/ethanol system.45
C bonds to THFalc and selectively catalysed FFald, FFalc, or 2-MeF to form 1,4-PeD in the H2O/ethanol system.45
 | ||
| Fig. 7 Results of the FFalc conversion and yields over the (op)Ru–(1.30)Sn@Al2O3 catalyst as a function of initial H2 pressure. For reaction conditions, refer to Fig. 5. | ||
At an initial H2 pressure of 5 bar, the main products were 45% 1,5-PeD and 37% 4,5-DHFM, alongside a small amount of 8% by-products (LA, CPO, and a dimer of FFalc) at 90% conversion of FFalc. As the initial H2 pressure was increased to 10 bar, an outstanding yield of 1,5-PeD (97%) was obtained at 100% conversion of FFalc. At an initial H2 pressure of 20 bar, the yield of 1,5-PeD remained the same at 10 bar, although the quantity of THFalc gradually increased at the higher H2 pressure. This suggests that the elevated pressure promotes the complete hydrogenation of FFalc to produce THFalc. It has been reported that the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond hydrogenation in FFald or FFalc to THFalc over Sn or In modified-Ni or Ru based catalysts tends to occur preferentially at high concentration of H2.44,46 In fact, the 1,5-PeD yields slightly decreased to 92% and 87% at the initial H2 pressures of 30 and 40 bars, respectively, while the amounts of THFalc increased significantly to 5% and 12%. Thus, we concluded that the optimised initial H2 pressure is between 10 and 20 bar.
C bond hydrogenation in FFald or FFalc to THFalc over Sn or In modified-Ni or Ru based catalysts tends to occur preferentially at high concentration of H2.44,46 In fact, the 1,5-PeD yields slightly decreased to 92% and 87% at the initial H2 pressures of 30 and 40 bars, respectively, while the amounts of THFalc increased significantly to 5% and 12%. Thus, we concluded that the optimised initial H2 pressure is between 10 and 20 bar.
 | ||
| Fig. 8 Results of FFalc conversion and yields over the (op)Ru–(1.30)Sn@Al2O3 catalyst as a function of reaction temperatures. For reaction conditions, refer to Fig. 5. | ||
At reaction temperatures of 100 °C and 120 °C, the conversions of FFalc were 54% and 79%, respectively, producing 1,5-PeD (27–65%) and 4,5-DHFM (14–17%) without any formation of THFalc. When the reaction temperature was increased to 140 °C, an extremely high yield of 1,5-PeD (96%) was obtained with only 4% of undesired product. However, further increasing the reaction temperature to 160 °C and 180 °C, the yield of 1,5-PeD slightly decreased to 91% and 86%, respectively, followed by a small amount of THFalc (5–9%) as the result of over hydrogenation of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of FFalc. In addition, undesired by-products, including LA, CPO, 2-MeF, and 2-MeTHF, were also observed at these temperatures. It is well known that the formation of 2-MeF following the furan ring hydrogenation to 2-MeTHF occurs efficiently during the catalytic conversion of FFalc over mono- and bimetallic catalysts at higher temperature or at a low pressure of H2.44,47
C bond of FFalc. In addition, undesired by-products, including LA, CPO, 2-MeF, and 2-MeTHF, were also observed at these temperatures. It is well known that the formation of 2-MeF following the furan ring hydrogenation to 2-MeTHF occurs efficiently during the catalytic conversion of FFalc over mono- and bimetallic catalysts at higher temperature or at a low pressure of H2.44,47
| Entry | Catalystsa | Conv.b (%) | Yieldb (%) | r (rate of reaction) | ||||
|---|---|---|---|---|---|---|---|---|
| 1,5-PeD | 4,5-DHFM | CPO (CPL) | THFalc | Othersc | ||||
| Reaction conditions: catalyst, 50 mg; substrate (FFalc), 2.0 mmol; solvent (H2O), 3 ml; initial H2 pressure, 10 bar; 140 °C, 3 h.a The values in the parentheses are the loading amounts of Sn (wt%).b Conversion and yield were determined by GC (FID) using an internal standard technique.c By-products include LA and a dimer of FFalc ((d)-FFalc) (based on the GC-MS data).d r (rate of reaction; mmol 1.5-PeD per gcat per min).e The reactant was the reaction mixture obtained from a reaction run using the (op)Ru–(2.56)Sn@Al2O3 catalyst (entry 4). The carbon balance was more than 96% for all the catalysts. | ||||||||
| 1 | (cop)Ru–(1.30)Sn/γ-Al2O3(E) | 66 | 44 | 22 | 0 | 0 | 0 | 1.25 |
| 2 | (cop)Ru–(1.30)Sn/γ-Al2O3(H) | 68 | 19 | 20 | 9 | 0 | 19 | 0.85 |
| 3 | (cop)Ru–(1.30)Sn/γ-Al2O3(M) | 82 | 58 | 6 | 7 | 5 | 5 | 1.28 |
| 4 | (op)Ru–(2.56)Sn@Al2O3 | 100 | 0 | 82 | 0 | 0 | 18 | n.d. |
| 5e | (op)Ru–(1.30)Sn@Al2O3 | 100 | 75 | 13 | 0 | 0 | 12 | 2.22 |
| 6 | As prepared (op)Ru–(1.30)Sn@Al2O3 | 99 | 35 | 60 | 0 | 0 | 4 | 1.10 |
| 7 | (cop)Sn/Al2O3-500 | 35 | — | — | — | — | 35 | n.d. |
| 8 | Al2O3-500 | 15 | — | — | — | — | 15 | n.d. |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond and ring opening of the C–O bond (entry 11, Table S2, and Fig. S13, in the ESI†).
C bond and ring opening of the C–O bond (entry 11, Table S2, and Fig. S13, in the ESI†).
 | ||
| Fig. 9 Time profiles for the one-pot conversion of FFalc and yields over the (op)Ru–(1.30)Sn@Al2O3 catalyst. | ||
At an early reaction time of 0.5 h, about 48% of FFalc was converted to 21% 1,5-PeD, 26% 4,5-DHFM and 4% LA + (d)-FFalc. Both the conversion of FFalc and yield of 1,5-PeD increased simultaneously as the reaction time was prolonged to 1 h. The maximum yield of 4,5-DHFM and LA + (d)-FFalc reached 23% and 7%, respectively, at 0.5–0.75 h, after which they plateaued after 2 h. The reaction was completed (100% conversion of FFalc) after 1.5 h, with the yield of 1,5-PeD gradually increasing to maximum (97%) after 4 h, leaving a remaining amount of 3% LA. These results indicate that the formation of 1,5-PeD occurs at a faster rate than that of 4,5-DHFM. Once 4,5-DHFM is formed on the catalyst surface, it rapidly undergoes hydrogenolysis via C2–O scission to form 1,5-PeD on Ru3Sn7 and Ru–SnOx species. The partial hydrogenation of the furan ring was also affected by the concentration of adsorbed H2 on the surface, as indicated by results in Fig. 7 regarding the effect of the initial H2 pressure.
As shown in Fig. 9, 4,5-DHFM was clearly observed at the early stage of the reaction but completely disappeared after 6 h. Moreover, the amounts of by-products slightly increased during the initial phase of the reaction (0.5–0.75 h), remaining unchanged (∼3%) even after 6 h. It is possible that the formation of an oligomer from FFalc and LA occurred due to the surface acidity of Ru–SnOx or Al2O3 as the reaction time was prolonged.50 Therefore, it can be presumably concluded that the formation of 1,5-PeD from FFalc follows a typical pattern of consecutive reaction pathways. To support this suggestion, specific reaction conditions (140 °C and 2.5 bar H2 for 0.5–3.0 h in a trace amount of H2O (2–3 drops)) were used and monitored using ATR-IR spectroscopy, and the results are shown in Fig. 10.
The ATR-IR spectra of the reaction mixture showed that an asymmetric band of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C in 4,5-DHFM was observed at 1633 cm−1 and the band intensified over time. Firstly, adsorbed FFalc on the Ru3Sn7 or Ru–SnOx surface of (op)Ru–(1.30)Sn@Al2O3 was partially hydrogenated to form the 4,5-DHFM intermediate. The formed 4,5-DHFM was further activated by Brønsted acid sites of Ru–SnOx or Al2O3 support at the C2–O bond, while the terminal –OH group attached to SnOx or Al2O3. These promoted the cleavage of the C2–O bond, leading to the formation of penta-2-ene-1,5-diol, which is rapidly hydrogenated to form 1,5-PeD as the final product (Scheme 2).13,28,51 While our mechanistic interpretations are supported by ex situ characterization and literature, future research could incorporate in situ DRIFTS and XAS/EXAFS to provide deeper insights into the adsorption configurations of intermediates like 4,5-DHFM and the real-time evolution of C–O bond cleavage on Ru–Sn sites.
C in 4,5-DHFM was observed at 1633 cm−1 and the band intensified over time. Firstly, adsorbed FFalc on the Ru3Sn7 or Ru–SnOx surface of (op)Ru–(1.30)Sn@Al2O3 was partially hydrogenated to form the 4,5-DHFM intermediate. The formed 4,5-DHFM was further activated by Brønsted acid sites of Ru–SnOx or Al2O3 support at the C2–O bond, while the terminal –OH group attached to SnOx or Al2O3. These promoted the cleavage of the C2–O bond, leading to the formation of penta-2-ene-1,5-diol, which is rapidly hydrogenated to form 1,5-PeD as the final product (Scheme 2).13,28,51 While our mechanistic interpretations are supported by ex situ characterization and literature, future research could incorporate in situ DRIFTS and XAS/EXAFS to provide deeper insights into the adsorption configurations of intermediates like 4,5-DHFM and the real-time evolution of C–O bond cleavage on Ru–Sn sites.
| Entry | Substrate | Conversiona (%) | Productsa (% yield) | |||
|---|---|---|---|---|---|---|
| Reaction conditions: catalyst, 50 mg; substrate, 2.0 mmol; solvent (H2O), 3 ml; initial H2 pressure, 10 bar; 140 °C, 3 h.a Conversion and yield were determined by GC (FID) using an internal standard technique. Values in the parentheses are the yield of the product. The carbon balance was more than 98% for all the reactions. | ||||||
| 1 |
|
100 ± 0.49 |
 (65) (65) |
 (20) (20) |
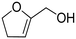 (9) (9) |
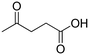 (6) (6) |
| 2 |
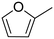
|
97 ± 0.62 |
 (57) (57) |
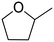 (34) (34) |
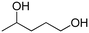 (6) (6) |
— |
| 3 |
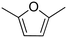
|
74 ± 0.43 |
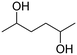 (34) (34) |
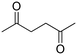 (9) (9) |
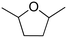 (21) (21) |
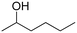 (10) (10) |
| 4 |
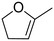
|
65 ± 0.51 |
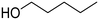 (42) (42) |
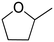 (20) (20) |
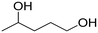 (3) (3) |
— |
| 5 |
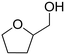
|
No reaction | — | — | — | — |
3. Conclusion
In conclusion, we have developed a facile one-pot synthesis of the confined structure Ru3Sn7 alloy on alumina ((op)Ru–(x)Sn@Al2O3) catalysts which demonstrated exceptionally rapid and selective hydrogenolysis of FFalc towards 1,5-PeD under mild conditions (140 °C, 10–20 bar H2, 3–6 h). For comparison, two types of (cop)Ru–(1.30)Sn/γ-Al2O3 and (imp)Ru–(1.30)Sn/Al2O3 catalysts were also synthesised using coprecipitation (cop) and wet impregnation (imp), respectively. The (imp)Ru–(1.30)Sn@Al2O3-500 produced only 13% yield of 1,5-PeD, whilst the (cop)Ru–(1.30)Sn@Al2O3-500 catalyst produced 81% 1,5-PeD; meanwhile, the (op)Ru–(1.30)Sn@AL2O3 catalyst afforded an outstanding yield of 97% 1,5-PeD, with a productivity of 3.67 mmol 1,5-PeD per gcat per min. This result represents the highest yield and selectivity for 1,5-PeD compared to the state-of-the-art catalysts in the one-pot conversion of FFalc to diols under milder conditions. In the (op)Ru–(1.30)Sn@Al2O3 catalyst, the presence of Ru3Sn7 or Ru–SnOx facilitates the partial hydrogenation of the furan ring in FFalc to form 4,5-DHFM. The acidic species of SnOx or Snn+ or acidic Al2O3 promote the activation of ring opening processes via C2–O cleavage, while Ru0 is used for the following hydrogenation, leading to the high yield of the final product 1,5-PeD. This synthetic strategy can guide the design of more-efficient, selective, and stable catalysts for the catalytic direct conversion of furfural or furfuryl alcohol to 1,5-pentanediol.Data availability
The data supporting this article have been included as part of the ESI.†Author contributions
Rodiansono: conceptualisation, methodology, writing – original draft, writing – review & editing, and supervision. Atina Sabila Azzahra and Edi Mikrianto: formal analysis (XPS and XRD), investigation, and experiment. Indri Badria Adilina, Kiky Corneliasari Sembiring, Gagus Ketut Sunnardianto, and Ahmad Afandi: writing – review & editing, supervision, and formal analysis (XPS, NH3-TPD, H2-TPR, and N2-adsorption analyses).Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors acknowledge The Indonesian Endowment Funds for Education (LPDP) through BRIN-RIIM2 scheme (contract number of 79/IV/KS/11/2022), DRPTM-Kemendiktisaintek through Regular Fundamental scheme (contract number of 056/E5/PG.02.00.PL/2024), and LPPM-ULM through Internal Fundamental scheme (contract number of 1374.95/UN8.2/PG/2024) for financial support. We also acknowledge the facilities and scientific and technical support from the Advanced Chemical Characterization Laboratory, National Research, and Innovation Agency through E- Layanan Sains - BRIN.Notes and references
- M. Schlaf, Dalton Trans., 2006, 4645–4653 RSC.
- R. Gérardy, D. P. Debecker, J. Estager, P. Luis and J. C. M. Monbaliu, Chem. Rev., 2020, 120, 7219–7347 CrossRef.
- Y. Zhu, W. Zhao, J. Zhang, Z. An, X. Ma, Z. Zhang, Y. Jiang, L. Zheng, X. Shu, H. Song, X. Xiang and J. He, ACS Catal., 2020, 10, 8032–8041 CrossRef CAS.
- M. J. Gilkey, A. V. Mironenko, L. Yang, D. G. Vlachos and B. Xu, ChemSusChem, 2016, 9, 3113–3121 CrossRef CAS PubMed.
- J. He, K. Huang, K. J. Barnett, S. H. Krishna, D. M. Alonso, Z. J. Brentzel, S. P. Burt, T. Walker, W. F. Banholzer, C. T. Maravelias, I. Hermans, J. A. Dumesic and G. W. Huber, Faraday Discuss., 2017, 202, 247–267 RSC.
- K. Tomishige, M. Honda, H. Sugimoto, L. Liu, M. Yabushita and Y. Nakagawa, Carbon Neutrality, 2024, 3, 1–41 CrossRef.
- X. Li and Q. Deng, Trans. Tianjin Univ., 2023, 29, 347–359 CrossRef CAS.
- T. Tong, X. Liu, Y. Guo, M. N. Banis, Y. Hu and Y. Wang, J. Catal., 2018, 365, 420–428 CrossRef CAS.
- Z. Wang, B. Pholjaroen, M. Li, W. Dong, N. Li, A. Wang, X. Wang, Y. Cong and T. Zhang, J. Energy Chem., 2014, 23, 427–434 CrossRef.
- J. Guan, G. Peng, Q. Cao and X. Mu, J. Phys. Chem. C, 2014, 118, 25555–25566 CrossRef CAS.
- S. Liu, Y. Amada, M. Tamura, Y. Nakagawa and K. Tomishige, Green Chem., 2014, 16, 617–626 RSC.
- L. Bruna, M. Cardona-Farreny, V. Colliere, K. Philippot and M. R. Axet, Nanomaterials, 2022, 12, 1–11 CrossRef.
- W. Xu, H. Wang, X. Liu, J. Ren, Y. Wang and G. Lu, Chem. Commun., 2011, 47, 3924–3926 RSC.
- Y. Nakagawa and K. Tomishige, Catal. Today, 2012, 195, 136–143 CrossRef CAS.
- S. Koso, N. Ueda, Y. Shinmi, K. Okumura, T. Kizuka and K. Tomishige, J. Catal., 2009, 267, 89–92 CrossRef CAS.
- D. Sun, S. Sato, W. Ueda, A. Primo, H. Garcia and A. Corma, Green Chem., 2016, 18, 2579–2597 RSC.
- X. Li, P. Jia and T. Wang, ACS Catal., 2016, 6, 7621–7640 CrossRef CAS.
- Z. J. Brentzel, K. J. Barnett, K. Huang, C. T. Maravelias, J. A. Dumesic and G. W. Huber, ChemSusChem, 2017, 10, 1351–1355 CrossRef CAS PubMed.
- J. Tan, Y. Su, X. Hai, L. Huang, J. Cui, Y. Zhu, Y. Wang and Y. Zhao, Mol. Catal., 2022, 526, 112391 CAS.
- J. Peng, D. Zhang, Y. Wu, H. Wang, X. Tian and M. Ding, Fuel, 2023, 332, 126261 CrossRef CAS.
- Q. Shen, Y. Li, F. Wang, X. Zhang, Z. Zhang, Z. Zhang, Y. Yang, C. Bing, X. Fan, J. Zhang and X. He, Mol. Catal., 2024, 556, 113919 CAS.
- H. Liu, Z. Huang, F. Zhao, F. Cui, X. Li, C. Xia and J. Chen, Catal. Sci. Technol., 2016, 6, 668–671 RSC.
- D. Liu, J. Fu, J. Wang, X. Zhu, J. Xu, Y. Zhao and J. Huang, Appl. Surf. Sci., 2024, 642, 158571 CrossRef CAS.
- T. Xiang, D. Dai, X. Li, D. Liu, C. Feng, P. Dai, L. Li, X. Gu and Y. Liu, Appl. Catal. B: Environ., 2024, 348, 123841 CrossRef CAS.
- J. Tan, H. Huang, Y. Zhang, J. Cui, J. Zhang, L. Huang, Y. Wang and Y. Zhu, Green Chem., 2024, 26, 11608–11624 RSC.
- D. Dai, C. Feng, M. Wang, Q. Du, D. Liu, Y. Pan and Y. Liu, Catal. Sci. Technol., 2022, 12, 5879–5890 RSC.
- J. Wang, D. Liu, X. Yao, J. Fu, S. Jia and J. Huang, Appl. Catal., A, 2024, 676, 119675 CrossRef CAS.
- R. Rodiansono, A. S. Azzahra, U. T. Santoso, E. Mikrianto, E. Suarso, K. C. Sembiring, I. B. Adilina, G. K. Sunnardianto and A. Afandi, Catal. Sci. Technol., 2025, 15, 808–821 RSC.
- R. Rodiansono, A. S. Azzahra, E. Mikrianto, A. Ridhoni, I. Mustari, A. Nurfitriani, T. S. D. Bodoi, R. E. Sanjaya, E. Suarso and P. R. Ansyah, Bull. Chem. React. Eng. Catal., 2025, 20, 293–306 CrossRef.
- R. Rodiansono, M. D. Astuti, T. Hara, N. Ichikuni and S. Shimazu, Green Chem., 2019, 21, 2307–2315 RSC.
- Z. Yang, Y. Xiong, T. Li, Z. Yang and B. H. Chen, Catal. Commun., 2024, 186, 106822 CrossRef CAS.
- M. J. Hidajat, I. T. Ghampson, G.-N. Yun and D. W. Hwang, Chem. Eng. J., 2023, 461, 141912 CrossRef CAS.
- D. J. Morgan, Surf. Interface Anal., 2015, 47, 1072–1079 CrossRef CAS.
- X. Shen, L.-J. Garces, Y. Ding, K. Laubernds, R. P. Zerger, M. Aindow, E. J. Neth and S. L. Suib, Appl. Catal., A, 2008, 335, 187–195 CrossRef CAS.
- J. Wang, P. A. Chernavskii, Y. Wang and A. Y. Khodakov, Fuel, 2013, 103, 1111–1122 CrossRef CAS.
- E. Tututi-Ríos, H. González, D. A. Cabrera-Munguia, A. Gutiérrez-Alejandre and J. L. Rico, Catal. Today, 2022, 394–396, 235–246 CrossRef.
- A. Rezayan, K. Wang, R. Nie, T. Lu, J. Wang, Y. Zhang and C. C. Xu, Chem. Eng. J., 2022, 429, 132261 CrossRef CAS.
- M. Tamura, K. I. Shimizu and A. Satsuma, Appl. Catal., A, 2012, 433–434, 135–145 CrossRef CAS.
- C. Martin, I. Martin, C. Delmoral and V. Rives, J. Catal., 1994, 146, 415–421 CrossRef CAS.
- L. Ma, H. Wang, C. Zhu, Q. Liu, J. Tan, C. Wang and Z. Liang, ChemSusChem, 2019, 12, 2154–2160 CrossRef PubMed.
- X. Liu, X. Liu, G. Xu, Y. Zhang, C. Wang, Q. Lu and L. Ma, Green Chem., 2019, 21, 5647–5656 RSC.
- R. Ma, X.-P. Wu, T. Tong, Z.-J. Shao, Y. Wang, X. Liu, Q. Xia and X.-Q. Gong, ACS Catal., 2017, 7, 333–337 CrossRef CAS.
- R. Rodiansono, A. S. Azzahra, P. R. Ansyah, S. Husain and S. Shimazu, RSC Adv., 2023, 13, 21171–21181 RSC.
- R. Rodiansono, M. D. Astuti, K. Mustikasari, S. Husain, P. R. Ansyah, T. Hara and S. Shimazu, RSC Adv., 2022, 12, 241–250 RSC.
- R. Rodiansono, M. D. Astuti, S. Husain, A. Nugroho and S. Sutomo, Bull. Chem. React. Eng. Catal., 2019, 14, 529–541 CrossRef CAS.
- R. Rodiansono, M. D. Astuti, D. R. Mujiyanti, U. T. Santoso and S. Shimazu, Mol. Catal., 2018, 445, 52–60 Search PubMed.
- Y. Yang, L. Chen, Y. Chen, W. Liu, H. Feng, B. Wang, X. Zhang and M. Wei, Green Chem., 2019, 21, 5352–5362 RSC.
- P. P. Upare, Y. Kim, K. R. Oh, S. J. Han, S. K. Kim, D. Y. Hong, M. Lee, P. Manjunathan, D. W. Hwang and Y. K. Hwang, ACS Sustainable Chem. Eng., 2021, 9, 17242–17253 CrossRef CAS.
- T. Mizugaki, T. Yamakawa, Y. Nagatsu, Z. Maeno, T. Mitsudome, K. Jitsukawa and K. Kaneda, ACS Sustainable Chem. Eng., 2014, 2, 2243–2247 CrossRef CAS.
- D. Götz, M. Lucas and P. Claus, React. Chem. Eng., 2016, 1, 161–164 RSC.
- Q. Liu, B. Qiao, F. Liu, L. Zhang, Y. Su, A. Wang and T. Zhang, Green Chem., 2020, 22, 3532–3538 RSC.
- Y. L. Louie, J. Tang, A. M. L. Hell and A. T. Bell, Appl. Catal., B, 2017, 202, 557–568 CrossRef CAS.
- T. S. D. Bodoi, A. S. Azzahra, M. R. Ansyari, E. Mikrianto, R. Rodiansono, R. E. Sanjaya and P. R. Ansyah, 1st ICWDGs 2024, AIP Conference Proceedings, 2024, pp. 325–338 Search PubMed.
- A. S. Azzahra, N. Annisa, R. Rodiansono, U. T. Santoso, R. E. Sanjaya and E. Suarsa, 1st ICWSDGs 2024, AIP Conference Proceedings, 2024, pp. 127–138 Search PubMed.
- J. Huo, J.-P. Tessonnier and B. H. Shanks, ACS Catal., 2021, 11, 5248–5270 CrossRef CAS.
- A. P. Amrute, K. Jeske, Z. Łodziana, G. Prieto and F. Schüth, Chem. Mater., 2020, 32, 4369–4374 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Catalyst preparation & characterisation and catalytic reactions. See DOI: https://doi.org/10.1039/d5cy00542f |
| This journal is © The Royal Society of Chemistry 2025 |