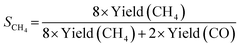Regulating protonation paths for enhanced photocatalytic CO2 methanation by coupling Pt sites on WO2.9/TiO2†
Jiajun
Du
a,
Jun
Deng
a,
ChangAn
Zhou
 a,
Hairong
Yue
a,
Hairong
Yue
 a,
Chong
Liu
a,
Chong
Liu
 a,
Patrik
Schmuki
b,
Štěpán
Kment
c and
Xuemei
Zhou
a,
Patrik
Schmuki
b,
Štěpán
Kment
c and
Xuemei
Zhou
 *a
*a
aSchool of Chemical Engineering, Sichuan University, No. 24 South Section 1, Yihuan Road, Chengdu, 610065, People's Republic of China. E-mail: xuemeizhou@scu.edu.cn
bDepartment of Materials Science and Engineering, Friedrich-Alexander-University Erlangen-Nurnberg, Martensstraße 7, 91058 Erlangen, Germany
cRegional Centre of Advanced Technologies and Materials, Czech Advanced Technology and Research Institute, Palacký University, Křížkovského 511/8, 779 00 Olomouc, Czech Republic
First published on 4th June 2025
Abstract
CO2 methanation via photocatalysis with water vapor is a sustainable technique for reducing CO2 emission but is challenged by the high energy barrier associated with the initial adsorption, activation and protonation of CO2 molecules. In this work, a substoichiometric WO2.9 thin film with strong Lewis acidity was coated on TiO2 microspheres, followed by the deposition of Pt cocatalysts on WO2.9 with controlled Pt single atoms and clusters (Pt–WO2.9/TiO2). The methane production rate reached 10.74 μmol h−1 g−1 with a selectivity of 99.8%, which was ∼40 times higher than that of bare TiO2 (0.27 μmol h−1 g−1). The high methane production rate was attributed to the synergy of Pt sites on the WO2.9/TiO2 heterojunction, where the Pt clusters facilitated water dissociation, thereby providing H* through hydrogen spillover on the surface, and the presence of a substoichiometric WO2.9 surface further enhanced the spillover process. The high density of active H* promoted the protonation pathway for CO2 activation (CO2 → COOH+ → *COOH), which improved the adsorption of the essential intermediate *CO on Pt single atoms and displayed a significantly reduced energy barrier for the protonation reaction of C1 intermediates, resulting in a mixed reaction pathway. This work provides new insights into a mechanism to regulate the reaction path to facilitate efficient photocatalytic CO2 methanation.
1. Introduction
Methane is the main component of natural gas and is widely used as a clean energy source or raw material for bulk production of chemicals.1 In industry, CH4 is synthesized using CO2 and H2 as raw materials via the Sabatier reaction,2 but the carbon emission and energy consumption of the process need to be improved. With the increasing demand for natural gas in the coming decades,3 CH4 synthesis via a green and sustainable approach is highly promising, with photocatalysis emerging as a method to achieve CO2 methanation from solar energy. However, owing to the high thermodynamical stability of CO2, it is challenging to achieve high methane yield via photocatalysis.4 Moreover, the conversion from CO2 to CH4 is an eight-electron reduction reaction that requires a high density of protons and electrons, resulting in relatively slow reaction kinetics.5 In addition, the competing reaction for CO2 reduction is the recombination of protons to form hydrogen. Therefore, photocatalysts that can accumulate photogenerated electrons and promote the migration of protons to carbon intermediates may enhance methane production.6The substoichiometric tungsten oxides, such as WO2.72, W24O68 and WO2.9, are stable in air,7 and the light absorption of WO3−x can be extended to the near-infrared region owing to the defect states below the conduction band minimum (CBM).8,9 The VO (oxygen vacancy) and unsaturated metal coordination sites pair can be used as Lewis acid sites to adsorb and activate CO2 molecules by reducing the activation energy barrier of CO2 molecules.10 In addition, W(VI) sites in WO3−x can be used as electron reservoirs by reducing them to W(V).11 The tungsten oxide with oxygen deficiency (VO) as substoichiometric tungsten oxides (WO3−x) are introduced to construct a heterojunction with TiO2 to improve the charge transfer kinetics of titania and increase the electron density on the surface. Titania is the most investigated semiconductor in photocatalysis due to its high abundance, high chemical stability and resistivity, but it is limited by its overall low reaction rate. In this work, mesoporous TiO2 microspheres were used as a support for substoichiometric tungsten oxide loading. The mesoporous structure and high surface area can provide abundant exposed active sites for surface reduction reactions and facilitate the construction of heterojunctions with intimate interfacial contact. The closely contacted anatase/rutile phase junctions, combined with highly crystalline anatase and rutile nanoparticles, may synergistically promote charge separation and transport.12,13
For methane production, noble metal cocatalysts can effectively enhance photocatalytic activity because of their electron transfer ability.14–16 However, hydrogen evolution reaction, which is a competing reaction in CO2 reduction, can also be accelerated by noble metals as well.17 Previous studies have shown that the coordination environment,16,18 size and distribution19 of Pt can effectively inhibit hydrogen evolution. It was reported in the 1960s that hydrogen spillover from noble metals to the oxide support occurred at the interface between the metal and the support.20 For example, it has recently been demonstrated that Pt nanoparticles (Pt NPs) dissociate H2O into absorbed H atoms (*H) and spillover from Pt NPs to the CO2 adsorption sites on the support to produce methane.16
Moreover, the activation of CO2 during photoreduction involves two different pathways: activation by electrons (CO2 → ·CO2−) and protonation (CO2 → COOH+). Recently, experiments have demonstrated that CO2 activation via protonation on semiconductor photocatalysts, forming an O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O–H+ intermediate,21 can significantly decrease the reaction energy barrier when such an intermediate undergoes further multiple proton-electron coupling reactions. Therefore, it is essential to design photocatalysts that favor the protonation of CO2. In this work, substoichiometric WO2.9 was fabricated on TiO2 microspheres for photocatalytic CO2 methanation, where the deposition of Pt cocatalysts and substoichiometric WO2.9 with good distribution can be expected to control the hydrogen spillover and improve the methane production rate (Scheme 1).
O–H+ intermediate,21 can significantly decrease the reaction energy barrier when such an intermediate undergoes further multiple proton-electron coupling reactions. Therefore, it is essential to design photocatalysts that favor the protonation of CO2. In this work, substoichiometric WO2.9 was fabricated on TiO2 microspheres for photocatalytic CO2 methanation, where the deposition of Pt cocatalysts and substoichiometric WO2.9 with good distribution can be expected to control the hydrogen spillover and improve the methane production rate (Scheme 1).
 | ||
| Scheme 1 Schematic of the synthesis of TiO2 microspheres, WO2.9/TiO2, and Pt–WO2.9/TiO2 photocatalyst. | ||
2. Experimental
2.1 Chemicals and materials
Pluronic F127 ((H(OC2H4)x(OC3H3)y(O2H4)zOH), Mw = 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1), H2PtCl6·6H2O, WCl6 (99.9%), tetrahydrofuran (THF, 99.9%), sodium bicarbonate (99%) and tetrabutyltitanate (TBOT, 99%) were purchased from Adamas. Acetic acid (AcOH, 99%), absolute ethyl alcohol (C2H5OH, 99.8%), and hydrochloric acid (HCl, 37 wt%) were purchased from Chron in Chengdu. Ultrapure water (resistivity: 17.38 MΩ cm) was used in the synthetic process. All reagents were used as received without further purification.
000 g mol−1), H2PtCl6·6H2O, WCl6 (99.9%), tetrahydrofuran (THF, 99.9%), sodium bicarbonate (99%) and tetrabutyltitanate (TBOT, 99%) were purchased from Adamas. Acetic acid (AcOH, 99%), absolute ethyl alcohol (C2H5OH, 99.8%), and hydrochloric acid (HCl, 37 wt%) were purchased from Chron in Chengdu. Ultrapure water (resistivity: 17.38 MΩ cm) was used in the synthetic process. All reagents were used as received without further purification.
2.2 Synthesis of the catalysts
2.3 Material characterization
The morphology of all samples was observed on a field emission scanning electron microscope (FESEM) (JEOL, JSM-7500, Japan). Transmission electron microscope (TEM) images were acquired with a Japan JEOL-F200 at an acceleration voltage of 200 kV. Samples were dispersed in ethanol, and 10 μL suspension was dropped on an ultra-thin carbon film supported by a 300-mesh copper TEM grid and dried for 1 h under an IR light. Powder X-ray diffraction (XRD) patterns were recorded on a Rigaku MiniFlex600 instrument equipped with a Cu Kα radiation (λ = 0.154178 nm) in the 2θ range of 10–80° with a step size of 0.01°. The lattice spacing d was calculated using Bragg's law: 2d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ = nλ. The Rietveld refinement line of TiO2 was obtained using Maud software, in which the crystal structure models of anatase (9008213.cif) and rutile (9004141.cif) were introduced, respectively, and background and scale parameters, basic phase parameters, microstructure parameters, crystal structure parameters and all parameters for texture were adjusted in turn to calculate the ratio of rutile and anatase in TiO2 microspheres. X-ray photoelectron spectroscopy (XPS) was performed on a Thermo Scientific K-Alpha instrument with a monochromatic Al Kα X-ray source (12 kV, 6 mA). The vacuum in the analysis room is 5.0 × 10−7 mbar. The binding energies of all the spectra were calibrated to the binding energy of C 1s at 284.6 eV.
θ = nλ. The Rietveld refinement line of TiO2 was obtained using Maud software, in which the crystal structure models of anatase (9008213.cif) and rutile (9004141.cif) were introduced, respectively, and background and scale parameters, basic phase parameters, microstructure parameters, crystal structure parameters and all parameters for texture were adjusted in turn to calculate the ratio of rutile and anatase in TiO2 microspheres. X-ray photoelectron spectroscopy (XPS) was performed on a Thermo Scientific K-Alpha instrument with a monochromatic Al Kα X-ray source (12 kV, 6 mA). The vacuum in the analysis room is 5.0 × 10−7 mbar. The binding energies of all the spectra were calibrated to the binding energy of C 1s at 284.6 eV.
2.4 Photocatalytic reduction of CO2
The photocatalytic CO2 reduction experiments were performed using a solid–vapor reaction in a Pyrex glass batch reactor with a window on top for light illumination (Labsolar 6A, Beijing Perfect light, China). The reaction temperature was controlled at approx. 10 °C by circulating cooling reagents at 5 °C. A 300 W Xe-lamp equipped with an AM 1.5 filter was placed on top and the optical power density was 100 mW cm−2. 20 mg sample was dispersed in 1 mL of DI water and dipped on a porous ceramic substrate (Beijing Perfect Light Technology Co., Ltd) with a 30 mm diameter, then dried at 80 °C for 30 minutes. After drying, the substrate loaded with the catalyst was placed on the quartz support in the center of the reactor, 10 cm above the bottom of the reactor. The reactor was filled with 50 mL DI water at the bottom. After the reactor was sealed, it was continuously vacuum-treated for 10 minutes and purged three times with high-purity CO2 (99.999%) gas, where CO2 gas was bubbled through the water at the bottom of the reactor and continuously flowed until the pressure inside the reactor reached ∼50 KPa. During irradiation, the products in the gas phase at different reaction times were analyzed by an online gas chromatograph (GC2030, SHIMADZU, Japan) with a barrier discharge ionization detector (BID) using high-purity helium (99.999%) as the carrier gas and Carboxen@1010 PLOT (30 m × 0.53 mm) as a chromatographic column. In blank experiments, in which there was no light source or photocatalyst, high-purity N2 was used instead of CO2.The selectivity of CO2 reduction (SCO2) and CH4 (SCH4) are calculated using the following equations:
2.5 Photoelectrochemical measurements
The photoelectrochemical measurements of TiO2, Pt–TiO2, WO2.9/TiO2, and Pt–WO2.9/TiO2 were performed in a three-electrode quartz electrolytic cell using an electrochemical workstation (Autolab PGSTAT 204A Instruments by Metrohm, Utrecht, Netherlands). The counter electrode was Ag/AgCl and the reference electrode was a platinum sheet (1 × 1 cm). The working electrode was prepared by the spin-coating method. 5 mg of the samples and 20 μL Nafion solution were placed into 0.5 mL of ethanol and sonicated for 1 h to obtain a uniform suspension, which was then uniformly coated on the glassy carbon electrode and dried in a vacuum oven at 80 °C. 0.5 M Na2SO4 solution was used as the electrolyte.The transient photocurrent spectra were tested at a bias of 0.5 V with an on/off period of 40 s for each transient. The EIS measurements were carried out in the frequency range of 0.01–105 Hz at a bias of 0.5 VOCP. Mott–Schottky (M–S) curves were obtained at frequencies of 600 Hz, 800 Hz, 1000 Hz and 1200 Hz in the potential range of −1.5–1.2 V.
2.6 DRIFTS measurements
DRIFTS measurements were conducted using a Thermo Scientific Nicolet FTIR spectrometer equipped with a liquid nitrogen-cooled HgCdTe (MCT) detector. The spectra were obtained by applying an average of 128 scans with a spectral resolution of 2 cm−1. The final spectra were obtained after removing the background spectra.2.7 DFT calculations
All the DFT calculations were performed using the Vienna ab initio simulation package (VASP)22 with the projector augmented wave (PAW) method.23 The exchange-functional is treated using the generalized gradient approximation (GGA) with Perdew–Burke–Ernzerhof (PBE)24 functional. The energy cutoff for the plane-wave basis expansion was set to 450 eV. Partial occupancies of the Kohn–Sham orbitals were allowed using the Gaussian smearing method with a width of 0.2 eV. The WO2.9 (010) and anatase TiO2 (101) surfaces were constructed, where the Pt cluster with 10 Pt atoms was established loading on the surface. In addition, single atom Pt was constructed by replacing the W and Ti atoms on WO2.9 (010) and anatase TiO2 (101) surfaces, respectively. The k-point of 2 × 1 × 1 and 1 × 1 × 1 was used for the calculations on WO2.9 (010) and anatase TiO2 (101) surfaces, respectively. The self-consistent calculations applied a convergence energy threshold of 10−5 eV, and the force convergency was set to 0.05 eV Å−1.The free energy corrections were calculated at 298 K using the following equation:
| ΔG = ΔE + ΔGZPE + ΔGU − TΔS |
3. Results and discussion
3.1 Morphology and structure analysis
In brief, TiO2 microspheres were synthesized via solvent evaporation-driven oriented assembly using tetrabutyltitanate precursors.12 A typical scanning electron microscopy (SEM) image of TiO2 (Fig. 1a) exhibits a regular spheric shape with a diameter of 1.57 ± 0.10 μm, consisting of anatase (JCPDS 21-1272) and rutile (JCPDS 21-1276), with an R (R = IA/IR) of 23.6%, as determined by Rietveld refinement ((Fig. 1b, Table S1 and Fig. S1†). The homojunction between anatase and rutile in the microspheres allows for improved charge-carrier separation efficiency, higher reduction potential of electrons (ECB of rutile ≤0.6 eV) and light absorption ability (Eg of rutile: ∼3.0 eV).26A substoichiometric tungsten oxide film was loaded onto TiO2 microspheres via a hydrothermal method using tungsten hexachloride (WCl6) as a metal precursor in deoxygenated anhydrous ethanol. The morphology of tungsten oxide depends on the concentration of WCl6 in solution, but with TiO2 microspheres as support, the morphology of tungsten oxide is directed by the support, where W6+ ions adsorb on the oxide sites, form nuclei and grow as a film (Fig. S2†).
The XRD spectrum (Fig. S1b†) for WO2.9/TiO2 shows no corresponding diffraction peaks for WO2.9, which may be attributed to the high dispersion of WO2.9 on the surface of TiO2 microspheres. For reference, pure tungsten oxide in a dark blue color was synthesized using the same hydrothermal method. The XRD spectrum of the tungsten oxide (Fig. S3†) shows obvious diffraction peaks at 23.6° and 48.3°, which can be ascribed to the (010) and (020) facets in the WO2.9 phase (JCPDS 05-0386). The Raman spectroscopy (Fig. S4†) revealed characteristic Raman active modes with symmetries Eg at 443 cm−1 and A1g at 609 cm−1 for rutile TiO2, and Eg at 144 cm−1 for anatase. The broad peak at about 230 cm−1 is aroused from the multiple phonons scattering. The attenuation of the peak at 144 cm−1 may be due to the change in the anatase phase in the TiO2 microspheres. The coating amount of WO2.9 was optimized based on the catalytic performance to be 10 wt% (Fig. S5a†).
The deposition of Pt was performed using photochemistry under restricted conditions.27 The ice lattice naturally confines the dispersion of precursors, nucleus formation and crystal size. The density, size, dispersion and allocation of Pt atoms can be fine-regulated by the density of photoelectrons,28 and the Pt precursor concentration. In this work, Pt cocatalysts were deposited as single atoms and nanoclusters on WO2.9 film, reduced by photogenerated electrons under irradiation of ultraviolet light at λ = 275 nm. For reference, Pt cocatalysts were deposited on TiO2 microspheres via the same photochemistry method and annotated as Pt–TiO2. The amount of the Pt precursor was optimized based on the catalytic performance to be 1 wt% (Fig. S5b†).
The diameter of TiO2 microspheres in Pt–WO2.9/TiO2 is 1.61 ± 0.11 μm, where the increase of 40 nm was attributed to the coating of WO2.9 film (Fig. 1b). The lattice fringes at the edge of the microsphere were analyzed to be 0.377 nm, 0.370 nm and 0.364 nm from the HRTEM image (Fig. 1c and S6a†), corresponding to the (010), (302) and (106) crystal planes of WO2.9, respectively.
The atomic dispersion of Pt was analyzed using aberration-corrected high-angle annular dark-field scanning TEM (AC HAADF-STEM) imaging (Fig. 1d and S6b†), which clearly resolves isolated Pt single atoms (marked by orange circles) and uniformly dispersed Pt clusters (marked by red circles) composed of several Pt atoms on WO2.9, with no evidence of Pt nanoparticles exceeding 1 nm in size. The compositions of the elements were analyzed with a uniform distribution of Pt and W ((Fig. 1e). To further clarify the distribution of Pt, CO-adsorption diffuse reflectance infrared Fourier transform spectroscopy (CO-DRIFTS) measurements for Pt–WO2.9/TiO2 are performed (Fig. 1f). First CO coverage saturation is reached with two strong IR peaks for gas phase CO. Then, gas phase CO and physically adsorbed CO were removed, which leaves chemical adsorption of CO with two sets of absorption bands centered at 2103 cm−1 and a broad peak at 2070 cm−1. The former is ascribed to linearly adsorbed CO on single-atom Ptδ+,29 and the latter is assigned to linearly adsorbed CO on Pt0 in Pt particles.30
The CO-DRIFTS spectrum of WO2.9/TiO2 exhibits no clear bands for adsorbed CO (Fig. 1g), indicating that the CO adsorption bands observed for Pt–WO2.9/TiO2 originate from the CO adsorbed on the Pt species. For Pt–TiO2, two sets of CO absorption bands are observed, with one centered at 2119 cm−1 and another broad band centered at 2070 cm−1, where the former peak at 2119 cm−1 is attributed to CO adsorption on single Ptδ+ atoms. Compared with CO on Pt–WO2.9/TiO2 (2103 cm−1), there is a blue shift in the CO adsorption peak on single Ptδ+ sites, indicating that the back donation from WO2.9 to Ptδ+ is stronger than that from TiO2. To evaluate the stability of Pt on TiO2 and WO2.9/TiO2, the photocatalysts were pretreated in Ar/H2 at 100 °C and CO-DRIFTS spectra were collected after the treatment (Fig. S7†).
X-ray photoelectron spectroscopy (XPS) was used to analyze the chemical species of the photocatalysts. The XPS survey spectra of TiO2, Pt–TiO2, WO2.9/TiO2 and Pt–WO2.9/TiO2 demonstrate that no impurity elements were present in the as-synthesized samples, except for Pt, W, O, Ti and C (Fig. S8†). The XPS Ti 2p spectrum of TiO2 shows four symmetric peaks after deconvolution of the spectrum, with a pair of binding energy at 458.4 eV and 464.1 eV for Ti 2p3/2 and Ti 2p1/2 of Ti4+, and another pair at 456.9 eV and 462.6 eV representing Ti 2p3/2 and Ti 2p1/2 for Ti3+, with an area ratio of 36.5% for Ti3+ (Fig. 2a).
With the coating of WO2.9, for the Ti 2p spectra of WO2.9/TiO2 and Pt–WO2.9/TiO2, a doublet peak at 458.4 eV for Ti 2p3/2 and 464.1 eV for Ti 2p1/2 of Ti4+ can be detected, where the shoulder peak of Ti3+ vanishes, which may be attributed to the oxidation during hydrothermal reaction in solution. During the formation of WO2.9, electrons are preferentially extracted from Ti3+-associated oxygen vacancies in TiO2, thereby oxidizing Ti3+ to Ti4+.
For Pt–TiO2, the shoulder peak for Ti3+ vanished, possibly because of Pt deposition on the vacancy sites. However, the binding energy of Ti4+ shifts to lower energy (∼0.30 eV), suggesting either the defects partially exist, or the electrons transfer from Pt to TiO2 at the interface, which may induce upward surface band bending of TiO2.
In the W 4f XPS spectrum of WO2.9/TiO2, two doublets can be deconvoluted-peaks with a binding energy of 35.2 eV and 37.3 eV attributed to W6+, and another doublet at 33.9 eV and 36.0 eV attributed to W5+ (Fig. 2b). With Pt deposition, the peak of W6+ for Pt–WO2.9/TiO2 shifts to higher binding energy (∼0.13 eV), indicating a decrease in the electron density of the W species, probably due to the transfer to the Pt sites. The shoulder peak for W5+ exists after the photodeposition of Pt, with an area ratio of 13.7%, which is close to the ratio for W5+ in WO2.9/TiO2 (13.0%) (Table S2†). Compared with the change in Ti 2p for Pt–TiO2 after deposition of Pt, the defect sites in WO2.9 remain for Pt–WO2.9/TiO2, suggesting a stable structure of substoichiometric WO2.9.
The Pt 4d5/2 spectrum of Pt–WO2.9/TiO2 was resolved into three components, Pt4+ at 316.8 eV, Pt2+ at 314.4 eV, and Pt0 at 311.6 eV (Fig. 2c). Compared with the Pt 4d5/2 spectrum for Pt–TiO2, the binding energies of Pt2+ show a shift to lower binding energy (∼0.20 eV, 314.6 eV), indicating an overall increase in the electron density of Pt on Pt–WO2.9/TiO2. For both samples, the Pt2+ species is dominant (∼55%), but the concentration of Pt0 species in Pt–TiO2 (22.6%) is higher than that in Pt–WO2.9/TiO2 (15.7%) (Table S2†).
The O 1s XPS spectrum (Fig. 2d) of TiO2 shows three components at 527.8 eV, 529.6 eV, and 531.7 eV, possibly corresponding to electron-rich oxygen (OR < 529.0 eV), lattice O (OL, Ti–O), and hydroxyl O (OC, –OH), respectively.31 With the coating of WO2.9, for the O 1s spectra of WO2.9/TiO2 and Pt–WO2.9/TiO2, the peak related to electron-rich oxygen disappeared, which may be attributed to the oxidation during the hydrothermal reaction in solution. An additional peak at ∼531.2 eV appears, which is characteristic of the oxygen-deficient tungsten oxide (OV). Compared with the O 1s spectra for TiO2 and WO2.9/TiO2, the O 1s spectra for Pt–TiO2 and Pt–WO2.9/TiO2 show a shift to lower binding energy, which may be due to photoreduction and the interaction between the surface and deposited Pt.
The Mott–Schottky spectra show that the flat band (Efb) potential of TiO2 and WO2.9 is −0.85 and 0.10 V vs. Ag/AgCl (about −0.65 and 0.30 V vs. NHE, ENHE (V) = EAg/AgCl + 0.197), respectively. (Fig. 3a) Furthermore, for n-type semiconductors, the conduction band minimum (CBM) is typically ∼0.2 V more negative than the flat-band potential. Consequently, the CBM positions of TiO2 and WO2.9 were measured to be −0.85 V and 0.10 V (vs. NHE), respectively. The optical band gap was obtained from Tauc plots according to the following equation:
| (αhν)n = A(hν − Eg) |
In situ XPS Ti 2p and W 4f spectra under illumination (Fig. 3c and d) demonstrate that the Ti 2p peaks shifted +0.36 eV to higher binding energy, and the W 4f peaks shifted −0.18 eV, accompanied by an increase in W5+ content (13.7 → 24.0%). These observations confirm the directional electron transfer from TiO2 to WO2.9, consistent with a type II heterojunction mechanism.
3.2 Photocatalytic CO2 reduction and photoelectrochemical performance
The above XPS analysis demonstrates the electronic interactions between WO2.9 and Pt, and between WO2.9 and TiO2, which may contribute to the photocatalytic methane production. The photocatalytic CO2 reduction performance of the Pt–WO2.9/TiO2 composites was evaluated and measured, leading to the production of CO, CH4, and H2 in the gas phase. Each experiment was repeated three times for the reference samples as TiO2, Pt–TiO2, and WO2.9/TiO2, to ensure reproducibility. The comparison of CH4, H2 and CO yield over reaction time under illumination is shown in Fig. S10.†For TiO2, the yield of CH4 after 6 hours is 1.61 μmol g−1 (Fig. 4a), which increased to 3.68 μmol g−1 after coating with WO2.9 for WO2.9/TiO2. For Pt–WO2.9/TiO2, the yield of CH4 reaches 64.41 μmol g−1, which is 17.5 times higher than that of WO2.9/TiO2. However, for Pt–TiO2, the CH4 yield is 37.63 μmol g−1, which is about half of that for Pt–WO2.9/TiO2.
Compared with the methane production rate reported in the literature, the value reported in this work is among the top values (Table S4†). In the reference experiments, there was no detection of the gas product, e.g. CH4 or CO, in the experiments without CO2 (using substituted N2), without light irradiation, or as a photocatalyst (Fig. S11†).
In addition, the H2 yield is 3.34 μmol g−1 for TiO2 and 0.80 μmol g−1 for WO2.9/TiO2, suggesting that the WO2.9/TiO2 heterojunction significantly improved the overall CO2 conversion rate and suppressed the competing reaction for hydrogen evolution. Moreover, the hydrogen yield is 41.12 μmol g−1 for Pt–TiO2, which is 4.8 times higher than that for Pt–WO2.9/TiO2 (8.47 μmol g−1), indicating that the deposition of Pt promotes the hydrogen evolution reaction, but with WO2.9, hydrogen evolution can be effectively inhibited.
The selectivity of CO2 reduction (SCO2) and CH4 (SCH4) are calculated based on the yield, where SCO2 refers to the selectivity for electron consumption CO2 reduction (see Fig. S12†) in competition with hydrogen evolution, and SCH4 refers to the methane selectivity among CH4 and CO (Fig. 4b). The SCO2 is 67.1% for TiO2 microspheres, which increases significantly to 95.0% with the loading of WO2.9 and reaches a remarkably high value of 96.8% for Pt–WO2.9/TiO2. However, SCO2 for Pt–TiO2 decreases to 78.6%, which suggests that the presence of WO2.9 facilitates the electron transfer to CO2 reduction reaction.
The stability of Pt–WO2.9/TiO2 was evaluated by performing six consecutive reaction cycles (6 hours for each). The average methane production rate in the fifth cycle was 73.3% of the first cycle (Fig. S13†), showing a slight decrease in the activity, which may stem from partial deactivation of the Pt active sites or surface contamination by intermediates.
To understand the impact of Pt sites on the CO2 reduction reaction, Pt NP–WO2.9/TiO2 was prepared by annealing Pt–WO2.9/TiO2 in N2 to convert Pt to nanoparticles. The methane production rate of Pt NP–WO2.9/TiO2 significantly decreased to 5.25 μmol g−1 h−1, and the hydrogen production rate increased to 19.19 μmol g−1 h−1 (Fig. S14†), that gives a SCO2 of 52.4%. These results indicate that the competitive reaction of hydrogen evolution is favoured on Pt nanoparticles.
The SCH4 among CH4 and CO for TiO2 was 94.9%, that is like SCH4 for WO2.9/TiO2 (96.2%). In the presence of Pt, the SCH4 is 99.8% for Pt–WO2.9/TiO2, 99.7% for Pt–TiO2, and 99.9% for Pt NP–WO2.9/TiO2, which suggests that Pt can significantly improve the methane selectivity, possibly contributing to the proton production and spillover during the reaction.
The photocurrent density of Pt–WO2.9/TiO2 (1.77 μA cm−2) is nearly 3 times as compared to TiO2 (0.56 μA cm−2), demonstrating a significant improvement in the separation and transfer of photogenerated charge carriers (Fig. 4c). The electrochemical impedance spectroscopy (EIS) spectra show that the charge transfer resistance of Pt–WO2.9/TiO2 is 15.31 kOhm, which is lower than the Rct of Pt–TiO2 (27.92 kOhm, Table S5†). The lower charge transfer resistance favoured the transport and separation of photogenerated electron–hole pairs (Fig. 4d). The coating of WO2.9 on TiO2 not only extends the absorption edge but improves the separation efficiency of photogenerated carriers.
After five cycles of experiments, the post-reaction photocatalyst was analysed. A slight decrease in photocurrent density (Fig. S15a†) and an increase in charge transfer resistance (Fig. S15b†) are in line with a decrease in photocatalytic activity, possibly due to partial deactivation of the Pt active sites.
3.3 CO2 photoreduction mechanism
Except for the charge transfer kinetics, the surface reaction rate also determines the overall reaction rate. The reaction intermediates during the photocatalytic methane production were thus detected by in situ DRIFTS measurements (Fig. 5 and S16†). After the adsorption equilibrium of CO2 and water vapor, the DRIFTS for Pt–WO2.9/TiO2 (Fig. 5a, bottom spectrum) show the peaks at 1273 cm−1, 1372 cm−1, 1424 cm−1, 1700 cm−1, that could be attributed to the features of the *COOH intermediates, that is, OH deformation, C–O stretch, symmetric stretch and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretch, respectively. The signals appeared at 1067 cm−1, 1464 cm−1, 1150 cm−1 can be assigned to *CHO, *CH2O and *OCH3, respectively. Furthermore, the *CO absorption band at ∼2077
O stretch, respectively. The signals appeared at 1067 cm−1, 1464 cm−1, 1150 cm−1 can be assigned to *CHO, *CH2O and *OCH3, respectively. Furthermore, the *CO absorption band at ∼2077![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cm−1 can be detected, indicating abundant CO intermediates were generated on the surfaces after the adsorption of CO2 and the *CHO intermediate originated from *CO. The bands at 2987 cm−1 to 2898 cm−1 can be attributed to the –CH3/–CH2 stretch and the wide band at 1637 cm−1 can also be observed, which is attributed to physically adsorbed H2O. The results suggest that the dissociation of CO2, H2O and proton reactions occur on the surface after the adsorption of CO2 and H2O in the dark.
cm−1 can be detected, indicating abundant CO intermediates were generated on the surfaces after the adsorption of CO2 and the *CHO intermediate originated from *CO. The bands at 2987 cm−1 to 2898 cm−1 can be attributed to the –CH3/–CH2 stretch and the wide band at 1637 cm−1 can also be observed, which is attributed to physically adsorbed H2O. The results suggest that the dissociation of CO2, H2O and proton reactions occur on the surface after the adsorption of CO2 and H2O in the dark.
 | ||
| Fig. 5 In situ DRIFTS spectra recorded during the photocatalytic CO2 reduction of (a) Pt–WO2.9/TiO2 and (b) TiO2. (c) Proposed pathways for CO2 methanation. | ||
After illumination of the Xe lamp with AM 1.5 filter, the DRIFTS feature for *CO at ∼2077![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cm−1 and the bands at 2987 to 2898 cm−1 for –CH3/–CH2 stretching almost vanishes, suggesting that the intermediates were consumed during the reaction. The intensity for the *CHO at 1067 cm−1, *OCH3 at 1150 cm−1 and *CH2O at 1464 cm−1 decrease during the 40 min illumination duration. However, the intensity of the signals for the *COOH intermediate features increased over the illumination time, that is OH deformation at 1273 cm−1, symmetric stretch at 1424 cm−1, and C
cm−1 and the bands at 2987 to 2898 cm−1 for –CH3/–CH2 stretching almost vanishes, suggesting that the intermediates were consumed during the reaction. The intensity for the *CHO at 1067 cm−1, *OCH3 at 1150 cm−1 and *CH2O at 1464 cm−1 decrease during the 40 min illumination duration. However, the intensity of the signals for the *COOH intermediate features increased over the illumination time, that is OH deformation at 1273 cm−1, symmetric stretch at 1424 cm−1, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretch at 1700 cm−1. Based on the above analysis, the pathway of CO2 methanation on Pt–WO2.9/TiO2 photocatalyst is a mixed pathway (Fig. 5c).
O stretch at 1700 cm−1. Based on the above analysis, the pathway of CO2 methanation on Pt–WO2.9/TiO2 photocatalyst is a mixed pathway (Fig. 5c).
For the DRIFTS of TiO2 (Fig. 5b), after the adsorption of CO2, the peaks of monodentate carbonates at 1518 cm−1 and 1437 cm−1 (m-CO32−), bidentate carbonates at 1587 cm−1, and 1327 cm−1 (b-CO32−) can be detected, as well as *CO at 2077 cm−1, physically adsorbed H2O at 1638 cm−1 and –CH3/–CH2 at 2987 to 2898 cm−1. After illumination of AM1.5 for 40 min, the signal of the IR peaks related to b-CO32− and *CO intermediates completely vanishes, but the intensity for the bands at 1518 and 1437 cm−1 for m-CO32− increases over illumination time. Moreover, the intensity of the characteristic peak at 1700 cm−1 of *COOH (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) hardly changes with illumination. The above phenomena indicate that light excitation enhances the adsorption and activation ability of TiO2 for CO2 gas (CO2 to adsorbed carbonate species), but the protonation process of the intermediates is limited, resulting in low catalytic efficiency. In addition, CO2 methanation on TiO2 occurs via the carbene pathway. The formation of the *CO intermediate needs to overcome a high energy barrier to form the *C intermediate, so CH4 production through the mixed pathway is favoured instead of the carbene pathway.34 Hence, the CH4 yield and selectivity of Pt–WO2.9/TiO2 are higher than those of TiO2, consistent with the yield and reaction rate detected in the CO2 photoreduction experiments.
O) hardly changes with illumination. The above phenomena indicate that light excitation enhances the adsorption and activation ability of TiO2 for CO2 gas (CO2 to adsorbed carbonate species), but the protonation process of the intermediates is limited, resulting in low catalytic efficiency. In addition, CO2 methanation on TiO2 occurs via the carbene pathway. The formation of the *CO intermediate needs to overcome a high energy barrier to form the *C intermediate, so CH4 production through the mixed pathway is favoured instead of the carbene pathway.34 Hence, the CH4 yield and selectivity of Pt–WO2.9/TiO2 are higher than those of TiO2, consistent with the yield and reaction rate detected in the CO2 photoreduction experiments.
The DRIFTS spectra of Pt–TiO2 and WO2.9/TiO2 were acquired as a reference (Fig. S16†). Compared to TiO2, the peaks of b-CO32− disappear and the intensity of peaks related to m-CO32− significantly decreased in the DRIFTS spectra of WO2.9/TiO2 acquired after adsorption and under illumination, and the intensity of the peak related to *COOH (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) significantly increased with the extension of illumination time (Fig. S16a†). The *COOH species is a key intermediate in the photoreduction of CO2 to CO or CH4, and its formation and adsorption behavior significantly affect the yield and selectivity of the CO2 reduction reaction relative to the hydrogen evolution reaction.
O) significantly increased with the extension of illumination time (Fig. S16a†). The *COOH species is a key intermediate in the photoreduction of CO2 to CO or CH4, and its formation and adsorption behavior significantly affect the yield and selectivity of the CO2 reduction reaction relative to the hydrogen evolution reaction.
For the DRIFTS of Pt–TiO2 in the dark and under illumination (Fig. S16b†), the intensity of the peaks related to *COOH (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) also increased. The peak corresponding to *CH3O at 1150 cm−1 appears after adsorption of CO2 and the intensity decreased over time under illumination, demonstrating that the Pt sites regulate the CO2 methanation pathway from carbene to mixed pathway.
O) also increased. The peak corresponding to *CH3O at 1150 cm−1 appears after adsorption of CO2 and the intensity decreased over time under illumination, demonstrating that the Pt sites regulate the CO2 methanation pathway from carbene to mixed pathway.
Based on the above discussion and analysis, it is clear that the most significant adsorption peak of the *COOH species appears for Pt–WO2.9/TiO2, but no observation of the adsorption peak corresponding to the carbonate species before and after illumination suggests that the synergy between WO2.9 and Pt sites can promote the conversion of CO2 into the *COOH intermediate. The absence of carbonate intermediates indicates that the reaction followed a proton-induced pathway for CO2 reduction (CO2 → COOH+ → *COOH).
In addition to the photoelectric performance and spectroscopic analysis, it can be determined that the formation of WO2.9/TiO2 heterojunction and the loading of Pt sites on the WO2.9 film significantly improve the light absorption and achieve a higher separation efficiency of photogenerated carriers. Furthermore, the strong Lewis acidity of the WO2.9 film, the electron-rich nature of the Pt sites and the hydrogen spillover channel from Pt clusters (Pt NPs) to Pt single atoms (Pt1) optimize the adsorption and activation processes of CO2, significantly promoting the generation of key intermediates for CO2 methanation, such as *COOH and *OCH3. Consequently, the highest CH4 production and selectivity in Pt–WO2.9/TiO2 are simultaneously achieved.
Hydrogen spillover occurs between noble metal particles and oxide supports (particularly reducible support), which allows the migration of active hydrogen atoms from metal particles (where a hydrogen atom is generated) to the support.35,36 In the hydrogen evolution reaction, the hydrogen spillover effect can significantly enhance reaction rates.37–39 Therefore, the rational design of spillover pathways on the catalyst can effectively improve CO2 hydrogenation rates by suppressing competitive HER.16,40
To further evaluate the hydrogen spillover assisted by WO2.9, density functional theory (DFT) calculations were performed using Pt/TiO2 and Pt–WO2.9/TiO2. Fig. 6a shows that on the Pt–WO2.9 surface, H* preferentially absorbs at the Pt/WO2.9 interface with ΔGH values of −0.17 eV (site 1) and −0.08 eV (site 2), where the negative values indicate spontaneous hydrogen adsorption. The ΔGH at the W top site (site 3, between Pt NPs and Pt1) was 0.15 eV, resulting in a thermodynamic energy barrier from site 2 to site 3 of 0.23 eV. On the surface of Pt–TiO2, the values of ΔGH on site 1 and site 2 were −0.25 eV and −0.50 eV, respectively, indicating spontaneous hydrogen bonding at the interface between the Pt cluster and TiO2. The ΔGH for H* adsorption on the Ti top (site 3) was 0.11 eV, leading to a considerably higher thermodynamic energy barrier (0.61 eV) from site 2 to site 3, compared to the Pt–WO2.9/TiO2, which may hinder hydrogen spillover from site 2 to site 3 on the surface of TiO2.
Based on the results of CO2 photoreduction experiments, the highest H2 production rate (19.19 μmol g−1 h−1) is observed for Pt NPs on WO2.9/TiO2 without Pt1, suggesting that protons may accumulate on Pt NPs and recombine to form hydrogen gas, which is a competitive reaction process for methane production. With Pt1 and Pt clusters (Pt–WO2.9/TiO2), H2 production rate decreased significantly (1.42 μmol g−1 h−1), and the methane production rate greatly increased (10.74 μmol g−1 h−1). In addition, for 2Pt–WO2.9/TiO2 (with nominal 2.0 wt% Pt precursor), the hydrogen evolution rate was nearly two times higher than that of Pt–WO2.9/TiO2 (Fig. S5b†). Therefore, Pt SAs optimize CO2 adsorption/activation, and Pt clusters enhance H2O dissociation, which synergistically determines the overall activity and selectivity for CO2 methanation.
For the sample without WO2.9 (Pt–TiO2), the methane production rate is 6.27 μmol g−1 h−1 and the H2 production rate is 6.85 μmol g−1 h−1, which may be due to the limited spillover of hydrogen. The experimental results and DFT calculations demonstrate that during the photocatalytic reduction reaction, protons at the Pt clusters transfer from the interface between the Pt clusters and the oxide support to the surface of the oxide, then desorb and participate in the reaction with CO2 molecules on adjacent Pt1, where the spillover is favoured on the WO2.9, in comparison to TiO2. The synergistic effect of Pt clusters and Pt1 and the promotion of WO2.9 on hydrogen spillover led to high efficiency for CO2 methanation and inhibited HER activity.
To further demonstrate the role of the synergistic Pt sites, calculations on the Gibbs free energy pathways of CO2 reduction from adsorbed CO2 to *CHO or CO were performed using Pt–WO2.9/TiO2 and bare WO2.9 (Fig. 6b). On the surface of bare WO2.9, ΔGH value of CO2 to *COOH was 0.66 eV, which decreased to 0.14 eV for Pt–WO2.9/TiO2. The lower energy barrier on the surface of Pt–WO2.9/TiO2 can be ascribed to the electronic structure of Pt1 favouring the adsorption compared to that of W. Moreover, due to the hydrogen spillover to the O atom adjacent to Pt1, an OH bond forms near Pt1, which can significantly weaken the original Pt–O bond (Fig. S17†). Consequently, when a C1 intermediate adsorbs and forms a chemical bond with Pt1, the Pt–O bond strengths further decrease and may break, leading to stable adsorption of the C1 intermediate on the Pt1 site (inset in Fig. 6b).
In addition, on Pt–WO2.9/TiO2, the energy barrier from *CO to *CHO was 0.44 eV, which was significantly lower than the energy barrier for *CO desorption (1.55 eV). Therefore, *CO tends to further protonation reaction rather than desorption of CO on Pt–WO2.9/TiO2. However, in WO2.9, the energy barrier from *CO to desorbed CO molecules was 0.40 eV, which was lower than the energy barrier from *CO to *CHO (0.58 eV). Therefore, *CO intermediates tend to desorb from the WO2.9 surface to free CO molecules, which is in line with the experimental results, in which the CO production rate is higher for WO2.9/TiO2 without Pt presence on the surface.
For Pt–WO2.9/TiO2, CH4 selectivity reaches almost 100% with highest production rate under simulated solar light. Hydrogen spillover from Pt clusters to the vicinity of Pt1 sites not only regulates the coordination environment of Pt1 sites for CO2 adsorption and activation but also provides protons for CO2 methanation.
4. Conclusion
In this work, CO2 molecules were converted to methane using a Pt–WO2.9/TiO2 photocatalyst, achieving a high CH4 selectivity of 99.8% and an average CH4 production rate of 10.74 μmol g−1 h−1 under simulated solar light. Dissociated water on the Pt clusters provides active H* for CO2 methanation through the hydrogen spillover channel, while the Pt single atoms act as the adsorption and activation sites for CO2. The strong Lewis acidity of WO2.9 enhances hydrogen spillover, effectively inhibiting the competitive hydrogen evolution activity. Moreover, the hydrogen spillover influences the coordination of Pt1 for the adsorption of C1 intermediates and reduces the energy barrier for further protonation. The yield of CH4 is thus enhanced by promoting the activation of CO2 molecules and the CH4 production pathways. Therefore, this work provides insights into the atomic design of cost-effective photocatalysts for CO2 methanation.Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 22108179, X. Z., 22308234, C. Z., 22176135 C. L.) and the Postdoctoral Fellowship Program of CPSF (GZC20231775, C. Z.). The authors would like to thank Dr. Yingming Zhu at the Institute of New Energy and Low-Carbon Technology for UV-vis measurements, and Yanping Huang at the Center of Engineering Experimental Teaching, School of Chemical Engineering, Sichuan University.References
- X. Meng, X. Cui, N. P. Rajan, L. Yu, D. Deng and X. Bao, Chem, 2019, 5, 2296–2325 Search PubMed.
- M. Tommasi, S. N. Degerli, G. Ramis and I. Rossetti, Chem. Eng. Res. Des., 2024, 201, 457–482 CrossRef.
- U. Ulmer, T. Dingle, P. N. Duchesne, R. H. Morris, A. Tavasoli, T. Wood and G. A. Ozin, Nat. Commun., 2019, 10, 3169 CrossRef PubMed.
- T. Zhang, X. Han, N. T. Nguyen, L. Yang and X. Zhou, Chin. J. Catal., 2022, 43, 2500–2529 CrossRef CAS.
- X. Li, J. Yu, M. Jaroniec and X. Chen, Chem. Rev., 2019, 119, 3962–4179 CrossRef CAS PubMed.
- J. Fu, K. Jiang, X. Qiu, J. Yu and M. Liu, Mater. Today, 2020, 32, 222–243 CrossRef CAS.
- T. Paik, M. Cargnello, T. R. Gordon, S. Zhang, H. Yun, J. D. Lee, H. Y. Woo, S. J. Oh, C. R. Kagan, P. Fornasiero and C. B. Murray, ACS Energy Lett., 2018, 3, 1904–1910 CrossRef CAS.
- K. Manthiram and A. P. Alivisatos, J. Am. Chem. Soc., 2012, 134, 3995–3998 CrossRef CAS PubMed.
- H. Khan, M. G. Rigamonti and D. C. Boffito, Appl. Catal., B, 2019, 252, 77–85 CrossRef CAS.
- C. Lu, J. Li, J. Yan, B. Li, B. Huang and Z. Lou, Appl. Mater. Today., 2020, 20, 100744 CrossRef.
- F. Riboni, L. G. Bettini, D. W. Bahnemann and E. Selli, Catal. Today, 2013, 209, 28–34 CrossRef CAS.
- Y. Liu, R. Che, G. Chen, J. Fan, Z. Sun, Z. Wu, M. Wang, B. Li, J. Wei, Y. Wei, G. Wang, G. Guan, A. A. Elzatahry, A. A. Bagabas, A. M. Al-Enizi, Y. Deng, H. Peng and D. Zhao, Sci. Adv., 2015, 1, e1500166 CrossRef PubMed.
- L. Yang, J. Du, J. Deng, N. H. M. Sulaiman, X. Feng, C. Liu and X. Zhou, Small, 2024, 20, 2307007 CrossRef.
- J. Ran, M. Jaroniec and S.-Z. Qiao, Adv. Mater., 2018, 30, 1704649 CrossRef PubMed.
- C. Dong, C. Lian, S. Hu, Z. Deng, J. Gong, M. Li, H. Liu, M. Xing and J. Zhang, Nat. Commun., 2018, 9, 1252 CrossRef PubMed.
- Y. Wang, J. Zhao, Y. Li and C. Wang, Appl. Catal., B, 2018, 226, 544–553 CrossRef.
- S. Xie, Y. Wang, Q. Zhang, W. Deng and Y. Wang, ACS Catal., 2014, 4, 3644–3653 CrossRef.
- H. Wang, L. Zhang, Y. Zhou, S. Qiao, X. Liu and W. Wang, Appl. Catal., B, 2020, 263, 118331 CrossRef.
- X. Shi, Y. Huang, Y. Bo, D. Duan, Z. Wang, J. Cao, G. Zhu, W. Ho, L. Wang, T. Huang and Y. Xiong, Angew. Chem., Int. Ed., 2022, 61, e202203063 CrossRef.
- L. Kuai, Z. Chen, S. Liu, E. Kan, N. Yu, Y. Ren, C. Fang, X. Li, Y. Li and B. Geng, Nat. Commun., 2020, 11, 48 CrossRef.
- S. Yin, Y. Zhou, Z. Liu, H. Wang, X. Zhao, Z. Zhu, Y. Yan and P. Huo, Nat. Commun., 2024, 15, 437 CrossRef.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef.
- P. E. Blöchl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- J. P. Perdew, J. A. Chevary, S. H. Vosko, K. A. Jackson, M. R. Pederson, D. J. Singh and C. Fiolhais, Phys. Rev. B: Condens. Matter Mater. Phys., 1992, 46, 6671–6687 CrossRef.
- C. Guo, X. Tian, X. Fu, G. Qin, J. Long, H. Li, H. Jing, Y. Zhou and J. Xiao, ACS Catal., 2022, 12, 6781–6793 CrossRef.
- C. Gao, T. Wei, Y. Zhang, X. Song, Y. Huan, H. Liu, M. Zhao, J. Yu and X. Chen, Adv. Mater., 2019, 31, 1806596 CrossRef.
- H. Wei, K. Huang, D. Wang, R. Zhang, B. Ge, J. Ma, B. Wen, S. Zhang, Q. Li, M. Lei, C. Zhang, J. Irawan, L.-M. Liu and H. Wu, Nat. Commun., 2017, 8, 1490 CrossRef.
- A. Kubiak, Sci. Rep., 2024, 14, 13827 CrossRef PubMed.
- W. Zhang, H. Wang, J. Jiang, Z. Sui, Y. Zhu, D. Chen and X. Zhou, ACS Catal., 2020, 10, 12932–12942 CrossRef.
- K. Ding, A. Gulec, A. M. Johnson, N. M. Schweitzer, G. D. Stucky, L. D. Marks and P. C. Stair, Science, 2015, 350, 189–192 CrossRef.
- M. Kan, C. Yang, Q. Wang, Q. Zhang, Y. Yan, K. Liu, A. Guan and G. Zheng, Adv. Energy Mater., 2022, 12, 2201134 CrossRef.
- L. Li, X. Dai, C. Cheng, F. Chen, S. M. Wabaidur, W. Wang and Y. Hu, Chem. Eng. J., 2025, 507, 160101 CrossRef.
- L. Li, H. Liu, C. Cheng, X. Dai, F. Chen, J. Ning, W. Wang and Y. Hu, ACS Catal., 2024, 14, 10204–10213 CrossRef.
- Ş. Neaţu, J. A. Maciá-Agulló, P. Concepción and H. Garcia, J. Am. Chem. Soc., 2014, 136, 15969–15976 CrossRef PubMed.
- W. C. Conner, Jr. and J. L. Falconer, Chem. Rev., 1995, 95, 759–788 CrossRef.
- M. D. Marcinkowski, A. D. Jewell, M. Stamatakis, M. B. Boucher, E. A. Lewis, C. J. Murphy, G. Kyriakou and E. C. H. Sykes, Nat. Mater., 2013, 12, 523–528 CrossRef PubMed.
- Z. Gu, M. Li, C. Chen, X. Zhang, C. Luo, Y. Yin, R. Su, S. Zhang, Y. Shen, Y. Fu, W. Zhang and F. Huo, Nat. Commun., 2023, 14, 5836 CrossRef.
- J. Li, J. Hu, M. Zhang, W. Gou, S. Zhang, Z. Chen, Y. Qu and Y. Ma, Nat. Commun., 2021, 12, 3502 CrossRef.
- J. Li, H.-X. Liu, W. Gou, M. Zhang, Z. Xia, S. Zhang, C.-R. Chang, Y. Ma and Y. Qu, Energy Environ. Sci., 2019, 12, 2298–2304 RSC.
- P. Liu, Z. Huang, X. Gao, X. Hong, J. Zhu, G. Wang, Y. Wu, J. Zeng and X. Zheng, Adv. Mater., 2022, 34, 2200057 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5cy00167f |
| This journal is © The Royal Society of Chemistry 2025 |