Ding-Xing Wang, Hui Wang, Yao Xu, Chi Zhang and Chao Feng
Org. Chem. Front., 2023,10, 2147-2154
DOI:
10.1039/D3QO00281K,
Research Article
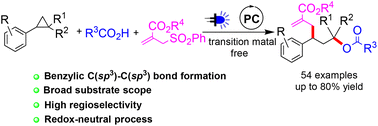
A photoredox catalysed 1,3-oxylallylation of aryl cyclopropanes was accomplished by reaction with carboxylic acids and allyl sulfones. The redox-neutral reaction proceeded in a highly regioselective manner under mild conditions with good functional group compatibility. Simple operation and successful application to the late-stage functionalization of several natural product and pharmaceutical molecule related carboxylic acids add extra merits to the current protocol.