Rational molecular design for isoindigo-based polymer semiconductors with high ductility and high electrical performance†
Abstract
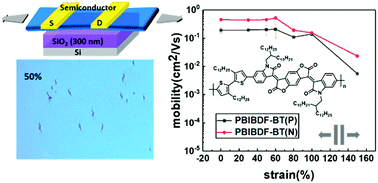
Achieving good electrical properties and ductility of polymer semiconductors has always been challenging. In this work, a series of isoindigo derivative-based conjugated polymers was studied in an effort to gain a better understanding of the influence of polymer main and side chain structures on their electrical and mechanical properties. The results suggested that the introduction of alkyl side chains onto the donors can significantly enhance the mechanical properties of isoindigo-based polymers; however, the electrical properties of the films greatly deteriorated due to the large steric hindrance by the chain. The insertion of strong electron-withdrawing units, such as benzodifurandione, into the isoindigo chain during the synthesis of a bis(2-oxoindolin-3-ylidene)-benzodifuran-dione (BIBDF)-based polymer (PBIBDF-BT) significantly boosted the electrical properties of the films without decreasing their mechanical properties. The crack onset in PBIBDF-BT thin films was observed at 50% tensile strain. In addition, PBIBDF-BT thin films exhibited bipolar transport properties with both electron and hole mobilities greater than 0.1 cm2 V−1 s−1 at 100% strain. It is found that the improvement of PBIBDF-BT performance is attributed to its proper molecular structure. The long alkyl side chains significantly increase the ductility of PBIBDF-BT thin films, and the strong electron-withdrawing BIBDF unit in the main chains enhances the local aggregation, resulting in a significant increase in mobility. These results indicate that the mechanical and electrical properties of conjugated polymers could simultaneously be improved through reasonable molecular design.


 Please wait while we load your content...
Please wait while we load your content...