Cryptomelane K1.33Mn8O16 as a cathode for rechargeable aqueous zinc-ion batteries†
Abstract
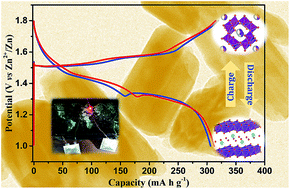
Aqueous rechargeable zinc-ion batteries have immense commercial potential in the primary battery market, which is largely occupied by alkaline zinc/manganese oxide batteries. These storage systems are desirable for large-scale applications due to their superior safety, high capacity, fast ionic conductivity and low cost. Pursuing new Mn-based cathodes for Zn-ion batteries, cryptomelane with a specific chemical composition of K1.33Mn8O16 was synthesized using a simple sonochemical technique. The formation of phase pure tetragonal K1.33Mn8O16 with I4/m symmetry was confirmed by X-ray diffraction. When tested as a cathode for Zn-ion batteries, cryptomelane K1.33Mn8O16 demonstrated a high reversible capacity of 312 mA h g−1 at a rate of C/10. Robust cycling stability was observed retaining more than 80% of initial capacity after 650 cycles with ∼99% coulombic efficiency. The intercalation chemistry of Zn–MnO2 systems is very rich involving polymorphism and phase transformations during charge/discharge cycles. Synergizing ex situ X-ray diffraction and spectroscopy with the electrochemical titration technique, we have examined the detailed mechanism of (de)intercalation of Zn2+ into cryptomelane K1.33Mn8O16, which is proposed as a potential cathode for Zn-ion batteries.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...