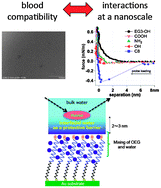
The mechanism underlying the bioinertness of the self-assembled monolayers of oligo(ethylene glycol)-terminated alkanethiol (OEG-SAM) was investigated with protein adsorption experiments, platelet adhesion tests, and surface force measurements with an atomic force microscope (AFM). In this work, we performed systematic analysis with SAMs having various terminal groups (–OEG, –OH, –COOH, –NH2, and –CH3). The results of the protein adsorption experiment by the quartz crystal microbalance (QCM) method suggested that having one EG unit and the neutrality of total charges of the terminal groups are essential for protein-resistance. In particular, QCM with energy dissipation analyses indicated that proteins absorb onto the OEG-SAM via a very weak interaction compared with other SAMs. Contrary to the protein resistance, at least three EG units as well as the charge neutrality of the SAM are found to be required for anti-platelet adhesion. When the identical SAMs were formed on both AFM probe and substrate, our force measurements revealed that only the OEG-SAMs possessing more than two EG units showed strong repulsion in the range of 4 to 6 nm. In addition, we found that the SAMs with other terminal groups did not exhibit such repulsion. The repulsion between OEG-SAMs was always observed independent of solution conditions [NaCl concentration (between 0 and 1 M) and pH (between 3 and 11)] and was not observed in solution mixed with ethanol, which disrupts the three-dimensional network of the water molecules. We therefore concluded that the repulsion originated from structured interfacial water molecules. Considering the correlation between the above results, we propose that the layer of the structured interfacial water with a thickness of 2 to 3 nm (half of the range of the repulsion observed in the surface force measurements) plays an important role in deterring proteins and platelets from adsorption or adhesion.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...