Chemical recycling of polyester textile wastes: shifting towards sustainability†
Théo
El Darai
,
Alexandra
Ter-Halle
,
Muriel
Blanzat
,
Guillaume
Despras
,
Valérie
Sartor
,
Guillaume
Bordeau
,
Armand
Lattes
,
Sophie
Franceschi
,
Stéphanie
Cassel
,
Nadia
Chouini-Lalanne
,
Emile
Perez
 ,
Christophe
Déjugnat
,
Christophe
Déjugnat
 * and
Jean-Christophe
Garrigues
* and
Jean-Christophe
Garrigues
 *
*
Laboratoire SOFTMAT (IMRCP), Université de Toulouse, CNRS UMR 5623, Université Paul Sabatier, 118 route de Narbonne, 31062 Toulouse CEDEX 9, France. E-mail: jean-christophe.garrigues@univ-tlse3.fr; christophe.dejugnat@univ-tlse3.fr
First published on 22nd May 2024
Abstract
Amongst all synthetic polymers used in the clothing industry, polyethylene terephthalate (PET) is the most widely used polyester, its fibres representing half the total PET global market (in comparison bottle PET being less than a third). Compared to bottle PET, the recycling of fabric PET fibres represents a challenge, both due to intrinsic structural differences (chain length and crystallinity) and to the presence of various additives (dyes, protection or finishing agents). Effective waste management requires addressing these additives through elimination or recycling processes. This review article aims to give an overview about all the existing means to recycle PET fibres. Textile recycling encompasses primary (closed-loop), secondary (mechanical), tertiary (chemical), and quaternary (incineration with energy recovery) processes. Mechanical recycling faces challenges due to PET's characteristics, including lower molecular weight and additives. Chemical recycling, particularly solvolysis processes (hydrolysis in neutral, acidic, or alkaline media, alcoholysis, glycolysis, aminolysis or enzymatic hydrolysis), offers a more advanced approach and will be described in detail, focusing both on the specific recycling of fibres when available and enlightening the advantages and drawbacks of each method. To discuss the environmental impact of each process, a quantitative analysis was conducted by defining the experimental domain represented by the temperature range and reaction time, and then calculating the energy-saving coefficient, as a green metric adapted to the diversity of textile PET recycling processes and data provided in the literature. This coefficient allows for discussing the relevance of using complex or non-renewable catalysts in processes, the positioning of enzymatic pathways, and the choice of reaction mechanisms applicable to the industry. A prospective approach was employed to identify key criteria for future advancements in green recycling. Subsequently, a comparative analysis of depolymerisation methods will be presented within the context of sustainable development goals (SDGs), green chemistry, and green metrics. Finally, using ε factors, this analysis will facilitate the detection and highlighting of pathways that show the most promise in terms of greening PET recycling.
1. Introduction
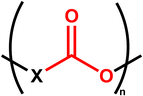
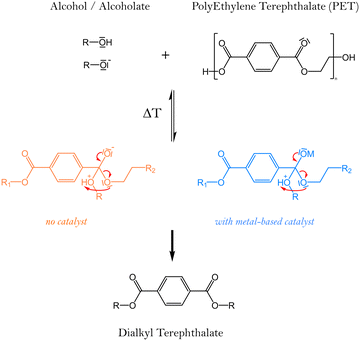
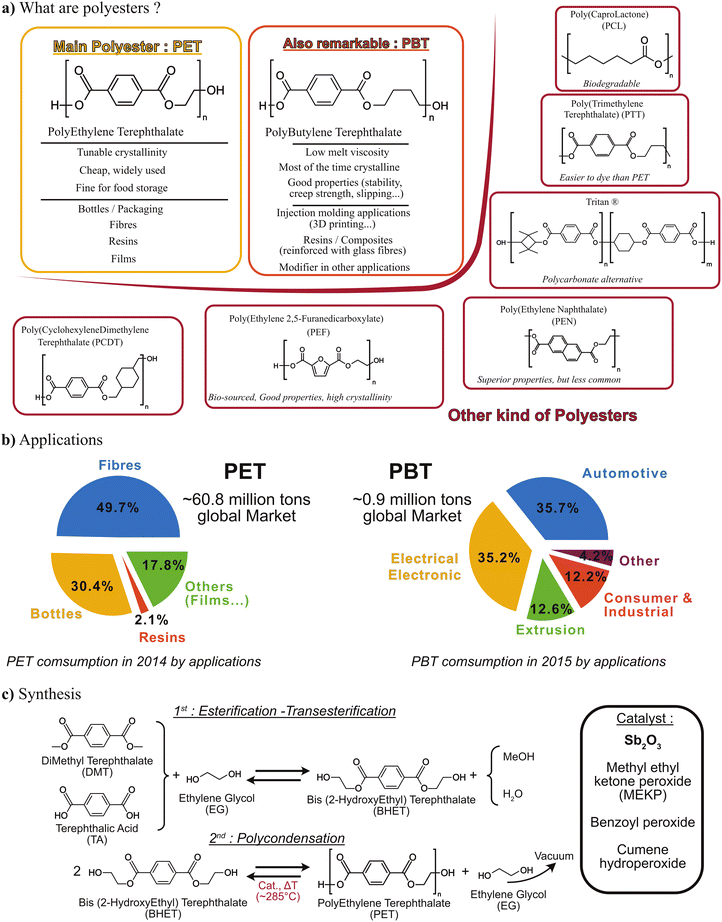
Textile recycling is crucial for reducing the carbon footprint of the industry worldwide. This review aims to discuss the challenges of textile waste recycling, to identify sustainable methods for the environment. Therefore, this review will present the materials used to produce textile fibres, especially polyesters, their properties, and the challenges related to the chemical recycling of these fabrics, which contain various specific additives that must be taken into account in depolymerisation reactions. The depolymerisation methods currently described in the literature will then be presented and detailed. To understand the environmental challenges and obstacles related to a transition to sustainability in textile recycling processes, we will analyse the specific characteristics of this industry, the market, and the volumes involved, and then we will link all this data to sustainable development goals and green chemistry principles. Finally, all depolymerisation methods will be compared using green metrics.Recycling involves the reuse and reprocessing of used clothing, fabrics and clothing scraps generated during manufacturing. Within the broad spectrum of recycling, and depending on the chemical nature of textiles, chemical recycling will be a future solution for producing chemicals to reform new polymers, enabling a true circular economy that limits waste and the environmental impact of this industry.1 Textiles typically include biodegradable components such as plant fibres (cotton, flax, jute, …), animal fibres (wool, silk, cashmere, …) and alongside non-biodegradable materials like polypropylene, polyethylene, nylon, or other synthetic fibres. Most textile fibres currently on the market are made of polyesters. The modern definition of polyesters includes all polymers containing an ester function in the backbone of their repetition unit, so they can be written as described in Fig. 1.
Such structure allows many different kinds of polyesters and properties. However, only a few polyesters are used in the textile industry (Fig. 2a), and one in particular concentrates the majority of polyester production efforts: polyethylene terephthalate (PET). After initial works on alkyds resins during the second part of 19th century, a major step in polyester history occurred in the 1928–1934 period by Carothers’ team for the DuPont de Nemours group, leading to the synthesis of many macromolecules containing esters.2–4 A few years after, in 1941, Whinfield and Dickson succeeded in forming a polyester from ethylene glycol and terephthalic acid, and PET was born.5 Soon after this first synthesis, PET fibres were commercialized by the Imperial Chemical Industries (ICI) under the tradename Terylene™.
 | ||
| Fig. 2 (a) Some common commercial polyester types. (b) Market share by applications of virgin PET/PBT in 2014/2015 respectively.8 (c) PET main synthesis from diMethyl therephthalate (DMT) or terephthalic acid (TA). | ||
Then DuPont de Nemours, which previously worked on polyesters, managed to purchase ICI's patent rights in 1945 and created the new polyester fibre Dacron™.6 In France, this fibre was developed in 1954 by the company Rhodiaceta under the tradename Tergal™. Eastman Chemical followed, and in 1958 created another polyester fibre called Kodel™. Since that date, and until the end of the 1970s, polyester fabrics developed rapidly, with more or less success.7
1.1. PET among polyesters
Among industrial polyesters, PET presents three major advantages exploited in three main applications: it is cheap, can be either amorphous (for transparent bottles) or semi-crystalline (for fibres), and finally is well suited for food and beverage storage (for cling films). These three applications represent roughly 98% of virgin PET world consumption in 2014 (Fig. 2b). PET is also the most produced polyester worldwide; as a comparison, polybutylene terephthalate (PBT) is second in terms of global production with 60 times lower tonnage. Indeed, the uses of PBT and other polyesters are mostly focused on other, often more technical, applications which require specific characteristics. Also, other polyesters such as Polyethylene 2,5-furandicarboxylate (PEF) or Polycaprolactone (PCL) are greener alternatives to PET, but they are more expensive and less used currently. Therefore, nowadays polyesters mainly refer to PET.8,9They constitute modern materials that benefit from many technological innovations, both in terms of fibres (chemistry and spinning), and weaving methods. Polyester microfibre development has also contributed to the development of new fabrics and clothing.10
1.2. From PET synthesis and properties to textiles
Polyester synthesis consists mainly of a polycondensation of a carboxylic acid with an alcohol at relatively high temperature and under vacuum (Fig. 2c). While many metallic or organic catalysts suit this reaction, the main industrial catalyst used is antimony trioxide (Sb2O3), which presents significant environmental and human health risks, depending on the exposure.11 For more details about polyesters synthesis, we invite you to read Gubbels et al. and Scheirs and Long.8,9With regard to the physicochemical properties, polyester is a thermoplastic polymer which can be fused and remolded, allowing fibres to be produced and recycled. These polyester fibres are manufactured as continuous filaments either from a polymer in the granule form (batch process), or by continuous polymerisation. In the manufacturing process, the molten polymer is immediately solidified and then drawn at a temperature above its glass-transition temperature (Tg = 69 °C.) to improve the polymer chains orientation and increase the polyester fibres strength which are a combination of crystalline and non-crystalline regions. To form fibres, polyester is melt-spun through spinnerets.12 The resulting fibres are stretched, combined into yarn, and then woven. The hydrophobic nature of the polymer confers water-repellent properties on the fibres. The resulting fabrics are easy to clean, dry quickly, do not crease, and are mildew resistant. Polyester fabrics are also strong, resistant to stretching, shrinking, and abrasion.13,14 In the 1970s, polyester fabrics were different from those of today, mainly because of the weaving (double knit), and were intended more for inexpensive leisurewear.7
The technical qualities and the low production cost of polyester fibres mean that clothing made with this fibre is very present, and constitutes the essential of clothing to be recycled. The numerous additives existing today also are important in the rise of polyester fabrics.
2. The challenges of PET recycling
2.1. Depolymerisation
Depolymerisation reactions are thermochemical methods which require the control of the various reaction stages to optimize recycling efficiency. To limit the environmental impact, catalysts are used to lower temperatures and improve selectivity to obtain monomers from PET.15 Various strategies have been developed to lower the reaction temperature and enable depolymerisation at room temperature. But solubilizing polymers often requires the use of solvents with a very high environmental impact.16 In the case of textiles, depolymerized compounds are commonly used as raw chemical materials, fuels or lubricants, but are rarely reused as precursors for new polymers.17 This can be explained by the nature of textiles, which contain additives to give them their properties.2.2. Additives
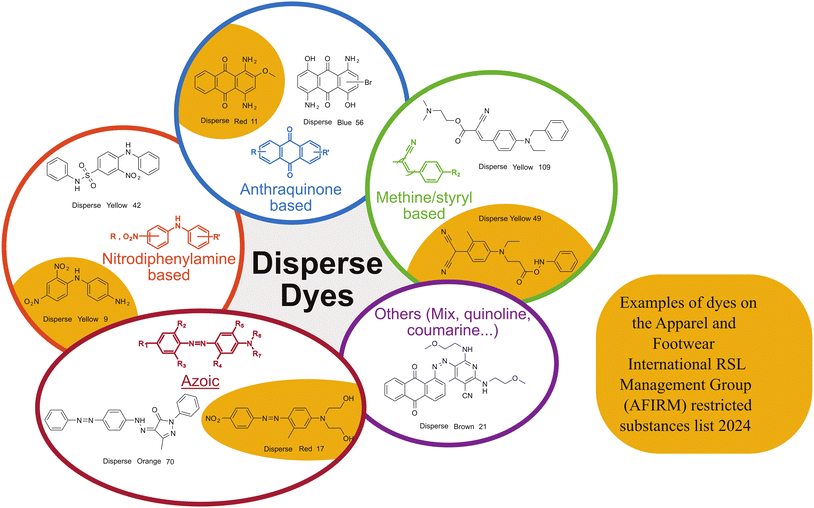
Whatever the recycling technique, it is necessary to take into account the many additives that may be present in polyester fibres. These additives, if not removed, can affect recycling performance and reduce the quality of recycled products in comparison with bottle grade polyesters.18 In the textile industry, additives are essential, whether to treat natural or synthetic fibres.19 These additives provide many features: colouring, protection against ultraviolet light, oil and water repellent, fire resistance, etc. In the textile sector, these chemical additives are mainly introduced in the fabric treatment process, such as dyeing and finishing.20 Also, the additives used are often tenacious, to resist friction and numerous washes. This particularity makes them difficult to eliminate for a recycling process.Dyes are the most common additives used in the textile industry.21 In this area, synthetic fibres, including polyesters, are treated in a special way.22 Polyester is a partially crystalline hydrophobic material, without very reactive functions, for which the traditional dyes of natural fibres (wool, cotton), cannot be used. In this case, special dyes (disperse dyes) must be used, as well as an appropriate dyeing technique.23,24 Such dyes developed for polymeric targets can represent some health/environmental risks (Fig. 3),25 are highly water consuming,26 and might interfere with chemical recycling process if not removed. So that some industries do not include dyed or tinted plastics in their process.27 The polyester fibres are dyed in the mass by direct dye penetration (Fig. 4), using a carrier, a solvent (swelling) and preferentially operating above the Tg. During the dyeing process, various auxiliaries can be used (fixing agent, stirring agent, dyeing acid etc.),21,22 which are generally removed after rinsing, although some can persist on the fibres. Printed fabrics, especially on polyesters, are more and more common.28 Printing techniques use suitable inks, but also auxiliaries (thickeners, emulsifiers, fixing agents…) which can persist in the fibre.
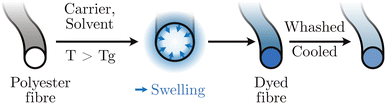 | ||
| Fig. 4 Representation of the transfer mechanism of dispersed dyes to PET fibres.24 | ||
Textile finishing consists of using a finishing agent on the fabric, which will modify the surface properties and provide new fabric functions.29 For example, we can use a water-repellent or oil-repellent finish using an agent that will modify the fibre surface characteristics, so that the fabric will be protected from water (waterproof) or oil (dirt). There are many other agents such as those improving the fastness to rubbing, antistatic agents, anti-slipping agents, anti-creasing agents, agents improving the solidity to light, antibacterial agents, flame retardants, ⋯.29 As the use of high performance textiles has grown, the need for chemical finishes has grown accordingly. Most of the additives used are bonded to the polyester fibre by weak interactions, but some are covalently bonded, like co-monomers (polyols,30,31 phthalates⋯32–34). Copolymers with the ethylene terephthalate unit including a brominated or phosphorus part are also used as Flame-Retardant.35 In all of these cases, the additives will persist in the material regardless of the recycling type. Indeed, fabrics based on polyester fibres are complex mixtures comprising numerous additives of different natures, and in unknown proportions. This complicates recycling, and in particular chemical recycling where some of these additives could interfere. Consequently, an additive (dyes and finishing agents) elimination step (even partial), is recommended before recycling. Also, such a step isolates the additives to treat them separately for their elimination or recycling. Some additives are expensive, or may cause health or environmental problems. Recent innovations include the production of green additives, such as flame retardants derived from bio-macromolecules obtained from lignin,36 or the introduction of bio-sourced functionalities such as antimicrobials on polyester fabrics.37
2.3. Polyesters fibres waste management
Fabrics were conceived to be durable and resistant to degradation. As a consequence, textile fibres are persistent in nature and lead to a pollution estimated to have major ecological and sanitary consequences.38,39 The alternative to a constant increase in demand for textile production – accompanied by a substantial proportion being released into the natural environment – is dematerialisation, substitution, reuse, material recycling, waste-to-energy transformation, and conversion technologies. These options must be carefully considered to design the best solutions to the environmental challenges imposed by plastic production and general use, and should be considered independently for each application.40Among the solutions cited, recycling is an option. Generally, recycling rates are calculated from collected litter and does not consider mismanaged waste so their constant progression does not reflect a better control of textile-derived plastic pollution. In 2017, Geyer et al. estimated that recycling only represented 9% of the plastic ever manufactured while 22% was mismanaged (Fig. 5). This figure could be considered in view of the material recycling rate in Europe which is estimated to have reached 30.6% in 2021.
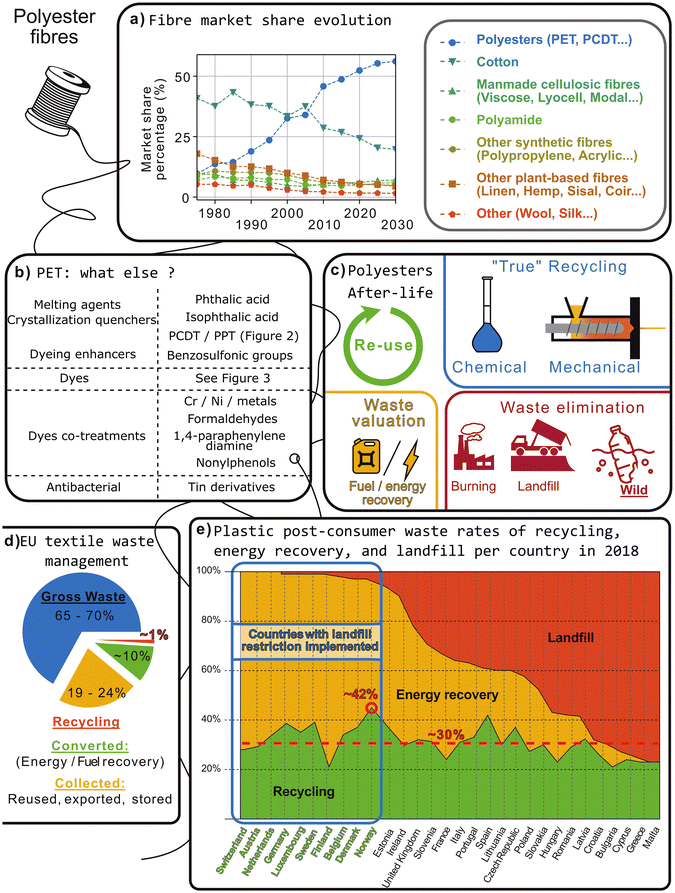 | ||
| Fig. 5 (a) Previous evolution and prospects of the textile market share according to Textile Exchange.68 (b) List of additives commonly found in PET fibres. Some are under restrictions according to the country. (c) The different non-storage ending of plastics. (d) Current textile waste management in Europe, gross waste meaning no clear public management and include storage and export.44 (e) Global plastic management in Europe, including textile and non-textile plastic waste.69 By comparison with (d), we can observe that textile waste are particularly poorly treated. | ||
Little is known about the proportion of synthetic fibres in waste and it varies from country to country. For example, it accounted for 5.8% of the total landfilled municipal solid waste in 2018 in the USA, corresponding approximately to 17 million tonnes (US Environmental Protection Agency). With rapid growth and evolution in fashion trends,41 textile production and waste generation rates have increased substantially lately,42 and tonnages are expected to increase by 50% by 2030.43 It is estimated that synthetic fibres were almost not recycled in 2020.44 This low score evidences the specificity of fibres recycling.
Synthetic fibres recycling is limited, firstly, because collection, separation, and sorting are very challenging. Sorting is the most important step but contrary to a rather well-organized collection of domestic plastic waste, textiles do not have a dedicated recycling sector. For example, post-consumer textile collection rates are 11% in Italy and 75% in Germany, while some countries do not have textile-recycling systems.45 As for recycling, less than 1% of total production was recycled in a closed loop (recycled into the same or similar quality applications).46 Most of the recycled textiles were recycled into other, lower-value applications.46 Recycling rates do not take into account the number of stages involved to perform recycling, nor the different recycling routes.
Generally speaking, textile recycling covers four distinct processes:
- Primary recycling or closed loop recycling, is when recyclable materials are mechanically processed to create a product that performs a similar function. In this process, textile fibres are reused without a loss of properties. By various mechanical means (crushing, grinding, de-weaving, stretching), fibres can be recovered from textile waste. A second treatment series is then applied (cutting, shredding), depending on the fibre origin and the fabric composition, to obtain recycled materials. The cost of these new fibres and the life cycle assessment of these recycled materials must include the environmental impact of the cutting/shredding and washing operations.
- Secondary recycling is also known as mechanical recycling. It is also a mechanical process, but to make new products. This process will involve more separation and purification stages of textile fibres to produce new materials, but they may not necessarily be reused to produce fabrics. Indeed, the fibre polymers, if not modified in their elemental composition, undergo structural changes, often resulting in a decrease in molecular weight because of chain breakages. If fabrics are composed of different fibre chemical families, for example, natural (like cotton) and synthetic (like polyester), it is necessary to separate the two elements by selectively solubilizing one of the two components or by using enzymes. Treatment processes for additives are often required to eliminate them and make the materials homogeneous (colour, composition). For instance, it is possible to separate cotton from polyester in fabrics through treatment without toxic organic solvents, using ionic liquids to dissolve the cellulose in cotton, isolate the polyester, and produce new cotton fibres through dry-jet spinning.47
- Tertiary recycling or chemical recycling is an advanced technology process. The polymer is converted into smaller molecules, through chemical processes, which are used as feedstocks for the production of new polymers, chemicals, or fuels. The chemical processes are tailored to the polymers found in tissues intended for recycling. For polyesters, various catalytic reactions such as hydrolysis, glycolysis, etc., are encountered and will be detailed further in this review. In the pursuit of environmentally-friendly method development, attention should be given to the catalysts involved, solvents, energy consumption, as well as the production of chemical species that may impact the environment.
- Quaternary recycling involves fabric incineration with energy recovery. This route is an alternative to the low recyclability of materials48 and remains quite common to reduce waste quantity by harnessing the energy contained in the materials.
Synthetic textile recycling routes are typically either mechanical or chemical and less frequently thermal. But in many cases recycling often consists of a combination of the three processes.49
For polyester fibres, the first route is abandoned at the industrial scale.50 Plastic bottles, made of what is called bottle-grade PET, are well engaged in the secondary recycling route. For example, about 50% of PET plastic bottles were recycled in the EU in 2022;51 this rate meets the average for most industrialized countries.52 Synthetic fibres are not recycled via the mechanical process, because fibre-grade PET presents distinct physico-chemical characteristics: lower molecular weight and intrinsic viscosity. Another reason why synthetic fibres cannot be mechanically recycled is the presence of high levels of additives. Also, the residual presence of other polymers prevents mechanical recycling, therefore the sorting of textile fibres is not well developed in any country.53
3. Chemical recycling of polyester fibres
3.1. Global overview
Massive textile production is a major concern for developing eco-friendly recycling technologies. In recent years, global textile fibre consumption reached a record 100 million tonnes in 2016. Within this fibre proliferation, PET has emerged as the leading player, representing over 50% of global consumption in 2018.54 Alongside this phenomenal sectoral expansion, there has been a significant increase in discarded textiles, leading to major environmental challenges. Indeed, conventional disposal methods, primarily landfilling and incineration, have resulted in significant ecological issues, highlighting the urgent need to find sustainable solutions for managing these textile wastes.55 PET is a robust, suitable for moulding polymer that can be easily melted to generate new fibres. However, the ductility of PET during a simple mechanical recycling process is significantly altered after three cycles. Consequently, only a small portion of PET is recycled for its original application, with most (50 to 77%) being transformed into fibres used in the production of mixed materials such as carpeting.56Faced with these pressing environmental challenges, alternative chemical, physicochemical, or biocatalytic recycling routes for PET fibres have become an essential solution. These recycling pathways must offer a sustainable alternative to traditional disposal methods, promoting the reuse of these valuable textile resources. Given the quantities of waste generated, chemical recycling of PET emerges as a particularly green promising approach. The nature of the textiles that make up these waste materials will significantly influence the choice of recycling processes to be implemented and their environmental impact. Textile waste is classified into three categories based on the production stage and use: pre-consumer, post-consumer, and industrial waste.57 To optimize sustainability, textile waste is then recycled at the end of its life to produce new materials or monomers,58 thus closing the production loop to return to the initial monomers.59
Chemical recycling of post-consumer textiles is suitable for various industrial and commercial applications, removing accumulated contaminants in the original fibres, dyes, and all additives, to produce fibres with properties identical to the original ones. Chemical recycling allows for the transformation of the PET chain into monomers (depolymerisation). In some processes, PET polymers can also be randomly broken into shorter chain fragments (oligomers). This PET degradation is achieved either by solvolysis (degradation by solvents) or by pyrolysis (thermal degradation in the absence of oxygen or air, or under vacuum).
Pyrolysis is widely used in industrial settings for the production of gases or fuels as part of the chemical recycling of various plastic waste.60 However, for PET, this depolymerisation method is less suitable for eco-friendly processes as it primarily generates carbon dioxide and carbon monoxide during the thermal decomposition.61
Carbonisation is another thermal valorisation technique of PET with a view to produce carbon materials including activated carbon.62,63 This technique has recently received much attraction since it can lead to green energy and sustainable environmental applications (batteries, water electrolysis, pollutant remediation, CO2 capture, etc.).64 PET-containing textiles and blends can also be treated by hydrothermal carbonisation yielding activated carbon with applications to pollutant adsorption and smart textiles conception.65–67
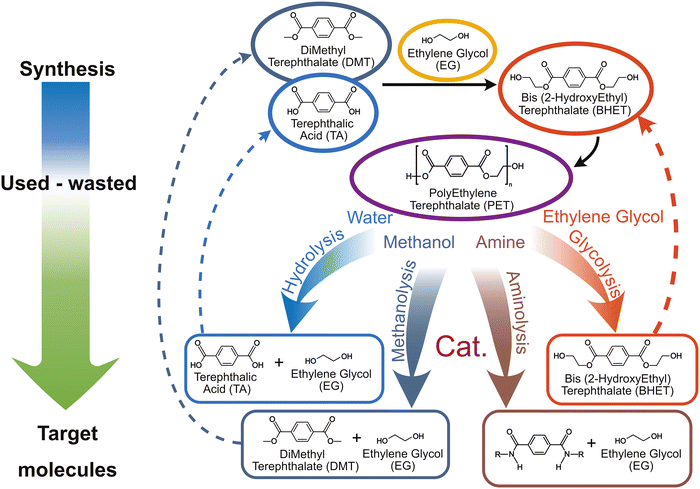
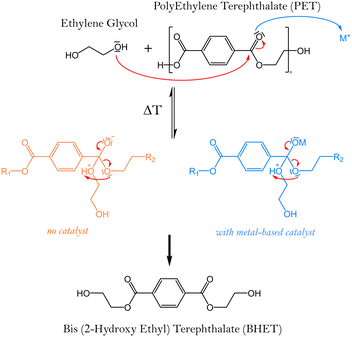
Solvolysis, requires water use, alcohols (mainly methanol or ethylene glycol), or amines. The reaction mechanism for PET depolymerisation consists of three nucleophilic substitution steps.50Fig. 6 illustrates the reaction pathways for the four most commonly used solvents to carry out this depolymerisation reaction: water (hydrolysis), methanol (methanolysis), ethylene glycol (glycolysis), and amines (aminolysis). The corresponding produced monomers can then be re-used for the synthesis of new PET.
These depolymerized PET waste materials are then regenerated and refined into emerging monomers (or oligomers) or transformed into new products. The regeneration of monomers or oligomers is followed by product purification until it reaches an acceptable quality level, suitable for reproducing PET materials or transforming them into new products.
In this recycling method, the material is first prepared as in the case of secondary recycling, up to cutting it into flakes or pellets. Then, it is chemically processed to produce monomers, oligomers, and mixtures thereof (dimers, trimers, etc.), such as bis(2-hydroxyethyl) terephthalate (BHET), terephthalic acid (TPA), glycols, dimethyl terephthalate (DMT), chemicals or fuels, petroleum liquids, and gases.70 The produced monomers can be processed to create new PET packaging. Regenerated monomers can be mixed with virgin materials to improve quality. Monomer/polymer purification can be achieved through distillation, crystallisation, drying, and some additional chemical reactions.
In the following sections we will describe the different chemical and enzymatic methods used for PET depolymerisation. Most are currently dedicated to PET from bottle origin and have been comprehensively described in this field in many recent reviews. Here we will rather focus on PET issued from textile fibres. Complementary to last reviews on “Polyester fabric recycling”, which comprehensively detail the mechanical and technical recycling technologies of textiles71 and blends containing PET,72 we aim here at emphasizing the current progresses in greening PET depolymerisations especially if it turns out that fibres have not yet been considered in the general PET recycling. Hydrolysis methods (in neutral, acidic, and alkaline conditions) will be first presented before describing alcoholysis and glycolysis (a peculiar case of alcoholysis). Aminolysis and enzymatic processes will be exposed next. The economic aspects of recycling PET wastes will then be considered by presenting the current industrial approaches developed worldwide.
3.2. Hydrolysis in neutral media
Usually, two strategies are performed for PET hydrolysis in neutral media, uncatalysed or catalysed pathways. Uncatalysed PET hydrolysis achieves PET degradation with only water. Temperature, pressure, PET/water ratio, reaction time, PET source, PET particle size, energy source, and/or water phases are the studied parameters to improve PET conversion and TPA yields. Already, the phase water is important in the efficiency of PET degradation. Best TPA yield was obtained in saturated liquid water compared to compressed liquid or supercritical fluid.73 Subcritical water conditions were also employed to investigate by NMR the PET depolymerisation chemical pathway. The analysis of PET depolymerisation products and the green metrics indicate the best reaction conditions depending on the interested products.74 O. N. Onwucha et al. developed uncatalysed hydrolysis under neutral conditions offering high hydrolysis yields and the production of large easily filtered TPA crystals and requiring only several water washes.75 This represents a significant advantage compared to the use of strong acid treatment to precipitate and recover TPA in most catalysed hydrolysis reactions in neutral media. The high TPA yields obtained (85 to 98%) required long reaction times (6 to 24 hours) and high PET/water ratios. This latter requirement is contrary to most of the previous reports.76–78Most of the catalysts used during catalysed neutral PET hydrolysis are metallic however organic catalysts were also developed. The TPA molecule shows efficient catalytic properties on PET hydrolysis with a PET conversion of 100% and TPA yield of 95% at 220 °C with 180 min. It can be easily recovered and reused up to eight times allowing easier product separation in a wastewater free process.79 Synergistic effects in the presence of other polymers like polypropylene or cellulose were examined. TPA yields increased in their presence at 250 °C for 30 minutes. Different experiments were completed to understand the nature of the chemical interactions at the origin of this synergy.80 Stanica-Ezeanu et al. used common salts like NaCl, CaCl2, NaHCO3 or KHCO3 as catalysts instead of the well-known metal acetate catalysts to hydrolyze PET and obtained similar or better results. They also studied PET kinetic degradation with marine water and estimated the time of waste PET depolymerisation in a marine environment. The depolymerisation rate is controlled by the surface water temperature and not on salt concentrations.81
PET depolymerisation under microwave irradiation in water with or without catalyst was also developed. PET depolymerisation can reach 90% with zinc sulfate catalyst at 200 °C and a microwave power of 250 W during 210 minutes and even 100% in pure water at a microwave power of 600 W and 20 bar during 120 minutes.82,83 Quaternary ammonium polytungstophosphate catalyst ([(CH3)3N(C16H33)]3PW12O40), a dual functional phase transfer catalyst, exhibits good catalytic performances on PET hydrolysis in neutral media with 100% PET conversion and 93% TPA yield at only 145 °C for 2 h. Moreover, it could be reused without a significant activity decrease for at least three cycles.84 To improve the catalyst reusability, Yan et al. developed Ni/γ-Al2O3 which exhibits 97% TPA yield with optimal experimental conditions. It remains active also after three cycles, but it can be regenerated by calcination.85
As a major aromatic compound, PET can also be converted back into arenes (toluene and xylene) by a Co/TiO2 catalyst inducing both depolymerisation and hydrodeoxygenation reactions,86 or to alkanes with TiO2 nanoparticle supported Ru catalyst.87 Catalytic conversion of PET into H2 fuel in pure water using Ru-5ZnO/mesoporous carbon catalyst was reported. It combines PET depolymerisation and in situ aqueous phase reforming.88
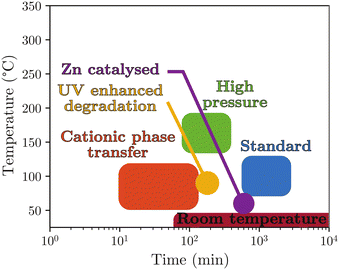
In summary, PET hydrolysis in neutral media can be achieved with or without a catalyst but often requires high temperatures and reaction times, which represents major disadvantages from the perspective of developing sustainable depolymerisation of PET, particularly in the case of textiles (Fig. 7).
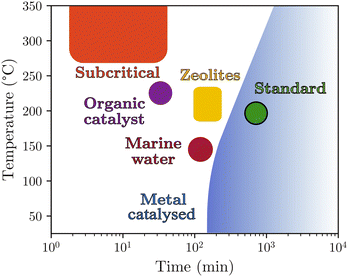 | ||
| Fig. 7 Classification of hydrolysis methods in neutral media sorted by typical reaction time and temperature. | ||
3.3. Hydrolysis in acidic media
PET hydrolysis in acidic media is a way to recover terephthalic acid and ethylene glycol from waste polymers mainly involving PET bottles, flakes, or powders, and reviewed recently.89 However, the case of PET textile fibres remains poorly considered, with almost no example of green PET depolymerisation under acidic conditions. In this reaction, the ester bonds in PET are cleaved by water, and hydrolysis is catalysed by acids and H+ donors.Although the catalytic aspect of this reaction could at first glance be interesting in a green chemistry approach, it requires large amounts of highly concentrated strong acids at high temperatures. The main acids involved are sulfuric acid,89 and nitric acid.90 In the latter case, ethylene glycol is oxidized into more valuable oxalic acid. These processes are costly, difficult to handle, and raise major concerns about recyclability;91 this could explain why this mechanism has not been preferred over other hydrolytic processes, especially in the case of PET textile fibres. Nevertheless, kinetic studies were performed with these acids to obtain reaction mechanism information.
Hydrolysis in acidic media follows a modified shrinking core model where successive layers are depleted from the bulk PET, starting from the surface.92,93 The reaction can be monitored using complementary techniques to document both the changes in the polymer crystallinity (using WAXS and DSC) and the molecular alterations occurring during hydrolysis and formation of lower molecular weight species (by 13C NMR).94 To take into account recyclability concerns, the acid used can be issued from waste batteries.95
Besides these harsh conditions, other greener methods have also been considered and developed recently. PET recycling in mixed textiles could be considered in the case of polycotton or cotton-based blends (mixed PET and cellulose fibres) by selective hydrolysis of cellulose in 1 mol L−1 aqueous HCl or HNO3 at reflux for a few hours.96 PET depolymerisation can also be performed under mild conditions using solid acid catalysts in methanol. This environmentally friendly methanolysis produced dimethylterephthalate (DMT) from PET powder or textile fibres after a 2 h reaction at 160 °C.97 A very recent promising approach is acetolysis using acetic acid, which has been exemplified with various PET sources including fibres (yarns).98 This reaction was executed at 280 °C for 2 h leading to terephthalic acid and ethylene glycol diacetate after precipitation. Both components can be reused as is for further PET synthesis, offering a low-carbon process for complete upcycling of waste PET from different sources including textiles.
Other greener approaches were also developed for PET hydrolysis under acidic conditions and recycling, mainly applied on bottles or flakes. Among them, the use of the sulfated TiO2 solid super acid catalyst in supercritical CO2,99 or the use of acidic zeolites under microwave irradiation were proposed.77 Easily recoverable organic acids are also efficient, such as poly(styrene sulfonic acid) (PSS),100p-toluenesulfonic acid (PTSA),101 or even terephthalic acid itself.79 Such simple organic acids can also be useful in acido-alcoholysis.102 Finally, Brønsted acid-based ionic liquids,103,104 and deep eutectic solvents,105 represent another green alternative for PET hydrolysis in acidic media. These processes are promising and pave the way for future applications for PET waste textiles degradation and upcycling.
In brief, PET depolymerisation in acidic media often requires the use of strong, concentrated mineral acids (at high temperatures), which do not appear compatible with the development of sustainable processes. Alternative methods using weaker acids or softer acidic conditions still need improvements to consider eco-friendly depolymerisation of PET textile wastes.
3.4. Hydrolysis in alkaline media
Hydrolysis of PET in alkaline media has been widely reported in the literature, mainly involving PET bulk materials, pellets, or bottles.106,107 In contrast, hydrolysis of PET textile fibres under alkaline conditions remains poorly described, especially when considering a green chemistry approach.PET depolymerisation by alkalis is based on the nucleophilic cleavage of ester functions, ending in the formation of ethylene glycol and terephthalate salt. In this reaction, the alkali is a reactant and not a catalyst as it is fully consumed. This could appear problematical from the green chemistry perspective, where catalytic processes are preferred over stoichiometric ones.
Moreover, when different kinds of polymeric materials are engaged together in tissues, the complete selective hydrolysis of PET can be useful to recover other polymer fibres such as cellulose for further recycling. This material recycling approach is an economically and ecologically viable solution,115,116 as it could potentially be implemented at the industrial scale. In this process, the mixed fabric was treated with 5% aqueous NaOH for 1–24 hours at 90 °C for selective PET hydrolysis and cellulose recovery. The same method was also successfully applied to the selective hydrolysis of PET over PVC in coated woven fabrics.117
Other additives were more efficient then cationic surfactants for improved PET hydrolysis in alkaline media, for instance ionic liquid [BMIM]Cl.129 Phase transfer catalysts, such as benzyltributylammonium chloride (BTBAC), are also useful and PET hydrolysis can be completed in less than an hour in 10% NaOH solution at 90 °C, allowing selective separation of cotton and PET in mixed textiles.130 Besides organic additives, inorganic particles were considered for enhancing PET alkaline hydrolysis. TiO2 particles can favor depolymerisation and promote degradation and breaking load loss of medical textiles,131 because of the facilitated diffusion of hydroxide ions through the surface defects of polymeric material induced by the particles. Moreover, inorganic Bi2O3@N-TiO2 composite particles were also used as photocatalysts for the enhanced degradation of PET fibre-based microplastics in alkaline media,132 representing an eco-friendly remediation mechanism.
It appears from these studies that the depolymerisation of PET in alkaline media has been much more developed than other hydrolytic methods (neutral, acidic). Lower temperatures and reaction times can be reached and eco-friendly methods are being developed, making this approach attractive for future environmentally compatible industrial processes.
3.5. Alcoholysis
Early works on model systems showed a greater small alcohols reactivity and in particular methanol in transesterification.141,142 According to this intuitive result, methanol was the first and most studied non-EG alcohol for PET alcoholysis.
A first important depolymerisation route is the catalyst-free depolymerisation, which is mainly performed under supercritical conditions at typically >220 °C/>6 MPa within tens of minutes.143–147 While most works focus on methanol, recent studies focused on a greener alternative: ethanol use in pressurized conditions.148,149 Thus, Lozano-Martinez and co-workers obtained about 94% PET degradation in 30 min at 275 °C/40 bar, and more recently, the effect of adding metal-based catalyst in the process was studied by Yang et al. (92% of degradation within 60 min at 270 °C/8 MPa with ZnO/Al2O3).150
High temperature alcoholysis (>100 °C) in subcritical (but still pressurized) conditions also represents a significant amount of research. Recent efforts demonstrated the possibility to use bio-based catalyst with decent results. Laldinpuii et al. demonstrated that bamboo leaf ash use at 200 °C for 2 h allows a decent retrieving monomers yield (78%),151 and Gangotena et al. also used pectin derivatives to obtain an almost full depolymerisation at 160 °C for 4 h.152 Zeolites can also present nice alternatives to the classical metal-based catalysts, showing similar performances (>90% within 30 min at 200 °C, MgO/NaY catalyst).153 Ionic liquids also work pretty well in this temperature/time range,154,155 even focusing originally on fibres waste.156,157 Thus, Liu et al. and Bui et al. obtained almost full depolymerisation within 8 h at 180–205 °C. More recent works accelerated the reaction time, with only slightly decreased performances (78% degradation yield in 30 min at 195 °C).154 Another alcoholysis field working within minutes at relatively high temperature is the microwave-driven depolymerisation.158 However, this approach gives better results for hydrolysis or glycolysis than methanolysis,159 and is often coupled with alkaline salt (which will be described later on).160–162 Some innovative works used bio-sourced fatty alcohol combined with microwave to degrade polyesters.163
Low temperature alcoholysis (<100 °C) mainly involved solvent mixture doped with a catalyst. Solvent toxicity, the rarity of the catalyst and the reaction end time are keys factor to compare those methods. The co-solvent choice is critical, and two main molecules seems to be privileged: THF164,165 and dichloromethane.18,166 However most of those works exhibit long reaction times (tens of hours), in comparison with high temperature processes, and particularly with microwave-assisted alcoholysis.
Going further, both Nikje et al. and Arias et al. published two works combining alcoholysis, degradation under alkaline conditions, and microwaves, drastically reducing the reaction time (below 5 minutes at the optimum).160,161,171
From a textile view point, either alkaline in alcohol or water are more efficient on PET fibres than on other polyester types.172
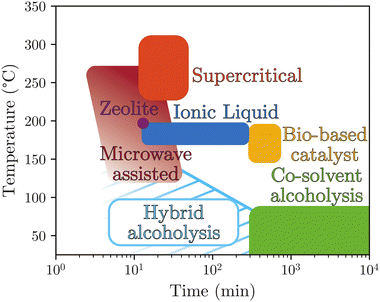
To summarize the chemical processes related to alcoholysis in an experimental domain aimed at identifying their environmental impact, Fig. 10 illustrates the positioning of each through reaction time and operational temperature found in the cited literature. Considering reaction time as a crucial parameter for industrial development, five processes stand out, allowing for short times ranging from 10 to 100 minutes: Hybrid alcoholysis, microwave-assisted alcoholysis, under supercritical conditions, using zeolite or ionic liquid catalysis. Conversely, long reaction times are observed when using biobased catalysts or co-solvents. Regarding reaction temperatures, high temperatures (>125 °C) are employed for five processes, with only cosolvent-assisted alcoholysis and hybrid alcoholysis conducted at reduced temperatures, in the range of 50 °C to 100 °C. The overall environmental impact can be easily assessed through these two parameters.
Despite some historical limitations (moisture sensitive, high temperature/pressure if no catalyst/co-solvent), alcoholysis recent improvements exhibit competitive reaction times and temperatures, especially when coupled with alkaline salts. Works can still be done to improve sustainability by offering greener co-solvents or reducing the alkaline salt amount used in the reaction; however alcoholysis remains an important approach for a greener recycling of polyester fibres. Indeed some industrial processes have already been developed (see section 4).
3.6. Glycolysis
Glycolysis, which has been patented since 1965, is the most commonly used method for PET recycling.173 During glycolysis, PET undergoes a transesterification reaction with an excess glycol amount under inert atmosphere and temperatures around 200 °C.This process leads to the PET depolymerisation and the formation of oligomers and bis-(hydroxyethyl)terephthalate (BHET) (Fig. 6).174
While the main focus was on achieving high-purity BHET, the depolymerisation of PET to create oligomers and polyols can have various applications in new materials including epoxy resins, vinyl esters, and unsaturated polyesters.175–178 Over the years, significant efforts have gone into improving and optimizing the glycolysis process, resulting in its adoption by several industrial companies, including IBM, GARBO SRL, Ioniqa, and Goodyear.179
Through extensive research on PET recycling via glycolysis, scientists discovered that this process is slow and requires a catalyst for faster reactions. Moreover, without a catalyst, PET depolymerisation towards BHET was not complete and resulted in a mixture of oligomers and monomers. To address these challenges, researchers have focused on increasing the reaction rate and yield of BHET monomers by developing efficient catalysts and optimizing reaction conditions. So far, different methods were used for PET glycolysis, including solvent-assisted glycolysis (use of not eco-friendly organic solvents), supercritical glycolysis (supercritical conditions require very high temperatures and pressures)180 and the most popular and the greenest process which is the catalysed glycolysis. Kinetics was also studied in the 90′s, showing the important roles of the catalyst and the temperature, which should be maintained relatively high (between 180 °C and 250 °C typically) to achieve full PET depolymerisation.78,181,182
Among the catalysts-assisted PET glycolysis, mainly three catalysts families are found in the literature: homogeneous catalysis (including metal salt-based catalysts and organocatalysts), ionic liquid-based catalysts, and heterogeneous catalysts (including zeolites, metal oxides, and other nanoparticles).
However, conventional glycolysis methods described above, that are commonly used for recycling PET from bottles, are often poorly efficient when it comes to recycling PET from heterogeneous waste textiles.191 Indeed, PET textile fibres with high crystallinity make it difficult for glycolysis agents to penetrate, so that depolymerisation gradually and slowly takes place from the surface to the bulk. Among metal salts catalysts proposed on fibres, titanium(IV)-phosphate showed greater catalytic activity than Zn(OAc)2 when applied on fibres with respectively 61 and 67% yields. Lei et al. proposed a feeding process using tin chloride (SnCl2) that allows a significant increase in the PET fibres conversion of 92.5% and yields of 70.4% compared to 28% and 14.4% respectively with the one-step reaction. In this study, the PET fibres were added stepwise at regular intervals. The authors suggest that this method allows a rapid transformation of PET into a homogeneous system, thereby accelerating the reaction efficiency.192
As with the use of metal salt catalysts, very few examples of glycolysis were performed on PET fibres. In a study with different PET material sources, Bush et al. showed that carpet fibres underwent PET full conversion and the greatest efficiency, compared to other PET sources, in BHET production (52%) using Gd salts combined with 1-ethyl-3-methylimidazolium acetate ([EMIM]OAc). These results were attributed to the large polymer surface area, even in the presence of additives and dyes.198 Liu et al. used amino acid ionic liquid catalyst and determined the optimal conditions by response surface methodology (RSM) to obtain 84.5% BHET yields on PET fibres.199
Some recent examples of polyester textile wastes depolymerisation were reported using nanoparticles. Vinitha et al. obtained BHET with 90% yield using silver-doped zinc oxide nanoparticles under microwave irradiation for 30 minutes at 180 °C.205 Guo et al. demonstrated that Perkalite F100®, which is an aluminium magnesium layered double hydroxide nanocatalyst, efficiently worked for PET depolymerisation with a BHET yield over 80% after a pre-degradation step.206 Similarly Mg–Al double oxides nanocatalysts were also used to perform fibre to fibre recycling at 240 °C with a BHET yield of about 80%.207
3.7. Aminolysis
PET solvolysis can occur upon treatment with amines or ammonia, hence providing terephthalamide derivatives. Because of the increased nucleophilicity of amine functions, in comparison to hydroxy groups, the aminolytic process is favored over alcoholysis and thus requires milder operating conditions.Typical aminolytic depolymerisation reactions are carried out using primary amines at temperatures ranging from 20 °C to 200 °C and generally under atmospheric pressure and neat conditions. The use of homogeneous or heterogeneous catalysts, sometimes combined with microwave irradiation, allows the aminolysis efficiency to improve by reducing the reaction times, reducing operating temperature ranges, and improving monomeric product yields.
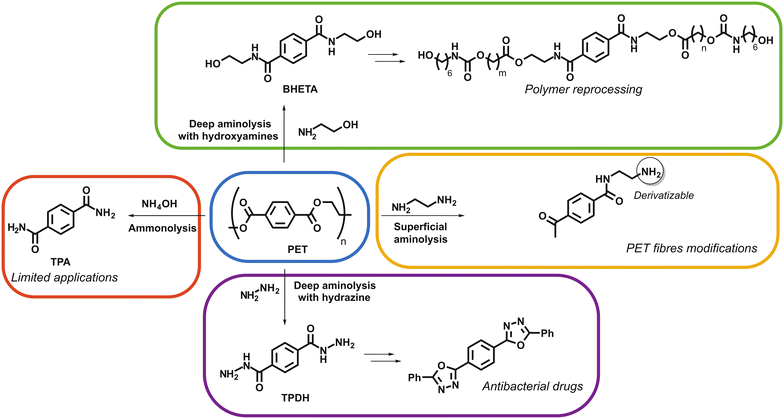
Importantly, one must distinguish two main approaches in PET aminolysis. The polymer can be partially degraded, to obtain lower molecular weight fragments, or even only superficially reacted. This allows modification of PET fibres towards different or even improved mechanical and/or physicochemical properties. Conversely, deep aminolysis of PET, i.e. complete depolymerisation, leads to the formation of terephthalamide-based monomers. This approach is especially interesting for upcycling, either by transforming the obtained monomers for uses as small molecules or for polymer reprocessing. To date, only superficial aminolysis processes have been developed at the industrial scale, while deep aminolysis still runs only at the laboratory scale.
In a seminal study, Farrow et al. carried out partial aminolysis of PET fibres in the presence of aqueous methylamine at room temperature.211 Starting materials of different crystallinities and orientations were subjected to aminolysis during various times and the weight loss and the crystallinity of the resulting fibres were assessed. From these experiments, the authors concluded that the aminolytic degradation proceeds in three main steps: (1) fast attack at amorphous regions, with small changes in weight and crystallinity; (2) chains scissions leading to an important weight decrease and increase in degree of crystallinity; (3) gradual decrease in degradation rate through a slower attack at both amorphous and crystalline regions. Later, several papers reported on the use of aminolysis (using methyl- to n-butylamine) to increase crystallinity of PET fibres to study their morphologies and mechanical properties.212–215 Overall, these studies showed that aminolytic partial degradation leads to molecular weight and samples weight decreases along with a diminution of mechanical resistance. For instance, Collins et al. established a linear relationship between the average molecular weight and the ultimate tenacity of aminolyzed PET fibres.213 Interestingly, Holmes treated PET fibres either with aqueous or gaseous n-butylamine and observed by SEM that surface of water-aminolyzed fibres developed radial cracks while axial cracks appeared in case of gaseous aminolysis. Fibres with axial cracks broke at reduced breaking loads than those presenting radial defects.212
Superficial aminolysis methods were developed, mostly using ethylenediamine, to enable surface derivatisation while preventing polymer degradation.216–220 Hence, it is then possible to easily install various functional moieties onto the surface of PET-based fabrics. For instance, dying, introduction of hydrophilic functions (e.g. zwitterionic polymer brushes) for antifouling purpose or of antibacterial groups were achieved. Those modifications did not significantly affect the mechanical properties of the treated fabrics compared to virgin fibres.
Deep aminolysis, i.e. complete depolymerisation towards monomers, developed in the 2000′s. Most of the corresponding papers report on catalyst-aided processes, while there are fewer studies dealing with uncatalysed reactions. In 2001, Spychaj et al. described aminolysis of PET flakes from bottles in the presence of triethanolamine or polyamines at 210 °C.221 The maximum depolymerisation conversion was reached after 150 minutes of heating. However, the molecular weight of the products was not characterized (only hydroxyl numbers and viscosity were measured), and it is thus not possible to conclude on the process efficiency as a deep aminolytic degradation. In a more recent study by Hoang and Dang, treatment of flakes with ethylenediamine at 100 °C provided oligomers (tri- to nonamer) that were precisely characterized.222 Also, the trimer to other oligomers ratio could be tuned via modifying the amine/PET ratio, up to quantitative degradation. Quantitative aminolysis to monomers was achieved by Soni et al., by submitting PET flakes to treatment with methyl-, ethyl-, or n-butylamine at room temperature during a maximum time of 45 days.223
Catalysed aminolysis of PET was extensively studied over the past two decades. Acetate, sulfate, carbonate, and bicarbonate salts of metals such as sodium, potassium, zinc, or lead are among the most used catalysts. In 2003, Goje et al. reported the deep degradation of PET powder (50–500 μm particles) in the presence of hydrazine hydrate, DMSO, cyclohexylamine and lead acetate, at 66 °C. Terephthalamide dihydrazide (TPDH) was isolated in a quantitative yield after 3 h and using 127.5 μm particles. The authors claimed that adding cyclohexyalamine to the reaction medium increased the reaction rate, although this effect was not rationalized in the paper. Further studies dealing with hydrazine-mediated aminolysis of PET flakes from bottles in the presence of benign sodium salts as the catalyst were also published. Conventional heating afforded TPDH (85% yield) in 3 h,224 while microwave heating (domestic microwave oven) led to a similar result in only 5–20 minutes.225 TPDH is an interesting monomer as it can be further used as starting material to prepare value-added molecules, such as plasticizers for polyvinylchloride (PVC),226 antibacterial agents,227 or textile dyes.228 However, hydrazine hydrate is a very hazardous and toxic compound, which must preclude its use in PET upcycling.
An interesting alternative to hydrazine hydrate is to employ hydroxyamines (e.g. ethanolamine) as degradation agents. Indeed, the obtained monomers are robust, thanks to the amide bonds, and the presence of end hydroxyl groups allows for further functionalisation or polymer reprocessing. Combination of ethanolamine with sodium acetate or bicarbonate furnished excellent yields in bis(2-hydroxy ethylene)terephthalamide (BHETA) monomer, under conventional (170 °C, 8 h) or microwave heating (700 W, 5–7 min), even using PET fibres as starting material.229–231 Very recently, Pastore and colleagues depicted the synthesis of UV-curable poly(urethane acrylates) from terephthalamide derivatives obtained by NaOAc/microwave-catalysed aminolysis of PET flakes with newly synthesized β-hydroxyamines.232 Degradation of PET to BHETA was described by Achilias et al. under microwave irradiation but without the aid of a catalyst. In this detailed study, the best results were obtained by performing the reaction under irradiation at constant power of 75 W or 100 W, respectively in 20 or 3 minutes.233
Fukushima et al. reported in 2013 on the use of 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) as an effective organocatalyst to convert PET flakes to monomers with various primary amines at temperatures ranging from 45 °C to 190 °C.234 In particular, ethanolamine gave the best result, with a yield of 93% in BHETA by running the depolymerisation at 120 °C for 2 h. Later, Demarteau et al. combined TBD with methanesulfonic acid (MSA) to achieve PET aminolysis in a few minutes (8–15 min) at 180 °C with ethanolamine (93%) or N-methylethanolamine (87%).235 The monomer derived from N-methylethanolamine-mediated degradation was next used for preparing poly(ester-amide)s. Nica et al. derivatized TBD with 4-(methoxycarbonyl)benzoate to prepare a new and effective organocatalyst (M4HPP). Using M4HPP on PET flakes in the presence of ethanolamine or ethylenediamine at 190 °C afforded quantitative yields of monomeric products in only 3–10 minutes.236 Deep eutectic solvents as well as ionic liquids are known as green catalysts in a range of applications including PET lysis. Musale et al. utilized choline chloride·2 ZnCl2 to convert PET flakes to BHETA nearly quantitatively, in refluxing ethanolamine for 90 minutes.237 When the aminolysis was carried out with diethanolamine and the resulting mixture was treated with 25% hydrochloric acid, terephthalic acid was isolated in 86% yield. The ionic liquid Hmim. TfO was also successfully used with ethanolamine (110 °C, 40 minutes) to afford BHETA in 89% yield.238
Tawfik et al. studied heterogeneous catalysis of PET aminolysis with dibutyltin oxide (DBTO) to produce BHETA from PET flakes at 62% yield after 4 h of heating in refluxing ethanolamine. With a view to make the process greener, the same authors exposed the PET/ethanolamine/DBTO mixtures to sunlight, keeping the vessel in a sand bath. Quantitative degradation was however achieved after 60 days of exposure.239 Examples of heterogeneous catalysis by β-zeolites, montmorillonite KSF (clay) and Sn-doped ZnO particles were also reported (in ethanolamine at 150–170 °C), each method providing very good BHETA yields.240,241
Besides amine-mediated depolymerisation, ammonolysis, the process using ammonia as the nucleophilic reagent, was also investigated. The reported studies described the use of liquor ammonia, at high (20 bar) or atmospheric pressure and at 40–180 °C, in the presence of a catalyst (zinc acetate or CTAB).242,243 However, despite high reported yields, the ammonolysis process is scarcely used because the major product, namely teraphthalamide, presents a limited interest, in particular for polymer reprocessing.
In conclusion, aminolysis presents a high potential for PET upcycling and PET fibres functionalisation rather than recycling. The major drawback of this approach is the necessity to use large amounts of eco-unfriendly amines, sometimes under heating at temperatures over 150 °C. These are probably the main reasons why aminolysis is a process restricted so far to the laboratory scale. Despite these limitations, the different reported methods (Fig. 12), and the variety of available amines, including functional ones, offer many possibilities to generate new monomers for polymer chemistry or other applications. The development of new catalysts/eco-friendly processes and the use of more acceptable amines should lead to consider aminolysis of PET textiles suitable for industrial applications.
3.8. Enzymatic PET depolymerisation
As PET is an ester-linked polymer, whole microorganisms or purified enzymes such as esterases could be used to achieve a green and sustainable polymer hydrolysis into its monomers via a more environmentally friendly process, therefore avoiding harsh chemicals and/or energy requiring processes.244However, because of its rigid aromatic structure, PET has long been regarded as non-biodegradable. The high stability of the polymer's backbone, its crystallinity (especially in textile fibres) and hydrophobicity are some of the main factors which restrict polymer biodegradability.245–247
All these features make them even more difficult to biodegrade than bottle-PET or aliphatic polyesters. Enzymes are sensitive to the polymer-chain flexibility, which relies on the polymer structure (aliphatic or aromatic) or on its crystallinity, which is a limitation for the enzyme active site accessibility.248
Some groups have tried to enhance the PET biodegradability by synthesising copolymers with readily hydrolysable aliphatic polyesters (Koshti et al., i.e.).249–251 Nevertheless, it does not address the recycling problem of the already existing PET wastes.
Up to recent years, most of the abundant literature concerning the enzymatic treatment of fabrics, including PET fibres, was for the modification of their surface properties. Enzymes can be efficiently used as competitive alternatives to the chemical surface modification of fabrics (e.g. wettability, de-sizing, scouring, bleaching, dyeing and finishing, for which the existing alternatives use very harsh chemicals whose environmental disposal causes many problems).252–255
It is only recently that PET-hydrolases were identified,256,257 with the first report of a hydrolase from Thermobifida fusca less than 20 years ago. Since then, several PET-hydrolases and cutinases (cutine hydrolases) have been described, among which the promising leaf compost cutinase (LCC).
The limitation for enzyme use lies in the fact that PET has to be hydrolysed at temperatures greater than its glass transition temperature, where the polymer chains become flexible, making them more accessible to the enzyme active sites. That implies the search for thermophilic PET hydrolases.257
High crystallinity can also be overcome by using moist-solid reaction mixtures as will be further discussed in the “enzymes in the recycling of textile PET fibres” part.258
Aggregation problems have sometimes been reported, which were addressed by cutinases (LCC) glycosylation.259
And sometimes the association of two different enzymes was necessary because of incomplete reactions forming both monomers and oligomers which blocked the reaction completion.260
Another limitation for enzyme use of is the long reaction times which can hardly be reduced by heating because of the heat sensitivity of PET-hydrolases.
Using computer-aided enzyme engineering, Tournier et al. created a modified leaf-branch compost cutinase (LCC) with increased thermal stability that allowed them to achieve a minimum of 90% PET degradation in less than 10 h, which is the best result obtained so far by enzymatic hydrolysis.261 They also showed that the biologically recycled PET obtained exhibit the same properties as petrochemical PET.
However, these modified enzymes were efficient in the degradation of bottle-PET samples. An on-line biodegradation/recycling of PET bottles in under development.
It would be of great interest to check their efficiency towards the degradation of PET textiles fibres, containing dyes or mixed with other fibre types.
To recycle textile waste, it is sometimes necessary to separate the cotton and PET components. One of the components should be depolymerized or degraded while the other component should be maintained.
Two different strategies are proposed to recycle mixed polyester-cotton textile wastes.
In the first, only the cellulose was enzymatically hydrolysed and the PET fibres were recycled without depolymerisation, which provided a clean and sustainable way of PET separation. The recovered PET from textile waste fibre were re-spun into new fibres by melt spinning.263
In the second, alkaline pre-treatment was effective for depolymerizing PET at 70–95 °C temperatures. Three products were obtained from the process: cotton cellulose, TPA, and an aqueous phase containing EG. Terephthalic acid (TPA) can be precipitated by adjusting the aqueous phase pH.121 However, the separation of TPA and EG was not considered in this study.
Enzymatic hydrolysis does not always require fibre separation thanks to the high substrate enzyme specificity. Also, it allows the depolymerisation under mild and green conditions (e.g., aqueous reaction media, atmospheric pressure, and temperatures up to 65–70 °C).
And despite the recent breakthrough in protein engineering that allowed the elaboration of PET hydrolases such as modified LCC with increased thermal stability and high activity, still these enzymes are not very efficient in the hydrolysis of high-crystallinity PET, such as spun PET fibres compared to bottle PET.264,265 The necessary melt-amorphisation step of PET prior to the enzymatic hydrolysis is energy demanding and restricts its use to rather clean PET wastes, which is not the case for textile wastes.
High-crystallinity PET (up to 46%) in mixed PET/cotton textiles could be directly and selectively depolymerized to terephthalic acid (TPA) by using a commercial cutinase from Humicola insolens under moist-solid reaction conditions (i.e., very high solid loading), with gentle mechanical mixing (mechano-enzymology) instead of standard aqueous solutions, affording up to 30% TPA yield.266
The process can be readily combined with cotton depolymerisation through simultaneous or sequential application of cellulase enzymes providing up to 83% glucose yield without any negative influence on the TPA yield, showing the potential for both simultaneous and stepwise depolymerisation of PET and cotton under mild, environmentally benign conditions.
The authors compared terephthalic acid (TPA) production from petrol to that from enzymatic PET depolymerisation, which revealed a 69% lesser energy requirement and 17% lesser greenhouse gas emissions for the latter.
Although enzymatic processes for depolymerizing bottle grade PET are rapidly developing on an industrial scale, they are not easy to transpose to PET textiles. The high crystallinity of PET and the need for prior treatments make this application much more complicated. Due to the high energy inputs on these pre-treatments and the very long reaction times required, the depolymerisation of textile PET by enzymatic processes still requires major improvements to be considered a sustainable route for textile recycling.
4. The economic aspects of recycling polyester wastes
The industrial developments for the chemical recycling of PET are particularly advanced for alcoholysis reactions, and two companies operate in this sector:- Evonik© has expertise in high temperature depolymerisation using an autoclave.27 However, they seem to focus on uncoloured bottle recycling, producing high quality monomers, suggesting some limitations because of polyester contaminants. However supercritical ethanol is well suited for multilayer packaging treatment, and therefore offers a wider range of perspectives than only bottle PET.267 So, it might be possible to use it for fibre treatment.
- Loop© recently patented several processes, based on a hybrid approach of alcoholysis/alkaline in chlorinated solvent.268,269
- Glycolysis is also involved: Ioniqa Company has developed a glycolysis process using an ionic liquid as a catalyst, based on paramagnetic nanoparticles that can be separated using a magnet. This process is currently being applied to post-consumer waste.
4.1 Recycled PET market, feasibility and chain sourcing
In any recycling project, it is crucial to assess the commercial potential of products derived from the chemical recycling of PET and their valorisation.270–272 This approach is essential, and begins with a market analysis based on several points:1. Understanding the demand dynamics for recycled products, especially in packaging, textiles, and construction sectors.
2. Competitive analysis concerning virgin ingredients used in PET manufacturing, and thus the market share for recycled ingredients, which may entail higher costs.
3. Regulatory considerations, particularly in packaging, that could influence the adoption of PET products derived from recycled ingredients.
4. Market viability assessment, including the availability and reliability of the PET recycling supply chain (collection, sorting, and processing). This aspect is crucial for evaluating the necessary recycling infrastructure.
5. The industrial success of the recycling process will also depend on consumers’ perception of the quality, safety, and efficiency of recycled PET products, as well as their environmental benefits. Innovative aspects of recycled products and marketing strategies can further enhance consumer appeal. Educational initiatives can promote the acceptance and adoption of recycled PET. Implementing targeted marketing campaigns can raise consumer awareness and promote the advantages of recycled PET products.
The viability of a recycling industry needs also a fine analysis the sourcing of PET to be recycled. A primary source of PET includes beverage bottles, food containers, and polyester textiles, primarily from post-consumer waste streams. By-products from various industries, such as packaging manufacturers, textile producers, and beverage companies, contribute to a secondary PET supply. Chemical recycling benefits from by-products generated in industrial processes like polyester manufacturing, where PET scrap and off-spec products can also be considered. The supply chain for sourcing PET includes also, waste management facilities, collection networks, logistics and transportation and sorting and cleaning facilities (processes to remove contaminants). Ultimately, the industrial viability of PET chemical recycling will require supportive policies and regulations, to encourage the adoption of recycled PET products. In conclusion, a comprehensive understanding of the market readiness for recycled PET products is essential, identifying both barriers and opportunities for stakeholders to drive sustainable growth and consumer acceptance.
4.2 Scale up of the recycling processes and industrial development
The transition from lab-scale to industrial-scale production for key technologies involves meticulous evaluation of various parameters. Scalability, profitability, regulatory compliance, and technological adaptability all need to be assessed. Comprehensive cost–benefit analyses are imperative to assess economic viability and the potential return on investment. Pilot tests must be carried out to validate scalability and optimize production processes before full-scale implementation. Implementation timeframes and resource requirements must be carefully defined to streamline the transition process. The integration of sustainable practices and environmental impact assessments should be an integral part of the transition strategy.Cooperation between laboratory and industrial teams is essential for knowledge transfer and skill development. Overall, a systematic and interdisciplinary approach is imperative to successfully move from laboratory to industrial-scale production of key technologies. Leading companies in PET chemical recycling span several countries, including the United States, Germany, Japan, and the Netherlands. Companies like Loop Industries (USA) and APK AG (Germany) specialize in processing PET, addressing both bottles and fiber. Japan's Teijin Limited and Netherlands-based Ioniqa Technologies are prominent in PET fiber recycling. Teijin processes PET fibers for automotive and apparel industries, with an annual capacity exceeding 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons. Ioniqa employs a proprietary technology to convert colored PET waste into high-grade raw materials.
000 tons. Ioniqa employs a proprietary technology to convert colored PET waste into high-grade raw materials.
In the USA, Loop Industries specialize in depolymerisation of PET by methanolysis since 2019, producing high-quality terephthalic esters.273 They process over 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons annually, contributing to a circular economy. Meanwhile, in France, companies like Carbios employ enzymatic depolymerisation techniques, focusing on both bottles and fibers, with an annual capacity of 40
000 tons annually, contributing to a circular economy. Meanwhile, in France, companies like Carbios employ enzymatic depolymerisation techniques, focusing on both bottles and fibers, with an annual capacity of 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons. The economic balance sheet of these companies shows promising growth, driven by increased demand for sustainable solutions and regulatory incentives. Their innovative approach promises sustainability gains and economic viability. All over the world, new companies are being deployed, as Eastman (USA), Itelyum (Italy), Reliance (India), Jeplan (Japan) and much more.274 Regardless of their interest, these projects demonstrate the feasibility and scalability of chemical recycling of PET. The future of the field looks bright, with investments pouring in and technological advancements paving the way for a greener, more efficient recycling ecosystem.
000 tons. The economic balance sheet of these companies shows promising growth, driven by increased demand for sustainable solutions and regulatory incentives. Their innovative approach promises sustainability gains and economic viability. All over the world, new companies are being deployed, as Eastman (USA), Itelyum (Italy), Reliance (India), Jeplan (Japan) and much more.274 Regardless of their interest, these projects demonstrate the feasibility and scalability of chemical recycling of PET. The future of the field looks bright, with investments pouring in and technological advancements paving the way for a greener, more efficient recycling ecosystem.
5. Comparative study
The comparison of chemical recycling methods for textile waste can be conducted according to various sustainability and greenness criteria, which should be defined by linking them to industrial specificities and environmental challenges.5.1 Sustainable development goals and green chemistry
In 2015, the United Nations released 17 objectives known as the Sustainable Development Goals (SDGs). These SDGs were adopted by all Member States of the United Nations (UN) as part of the 2030 Agenda for Sustainable Development, which outlines a 15-year plan to achieve these goals and improve human well-being by addressing crucial issues of social, environmental, and economic sustainability.275,276 Regarding the recycling of textile waste, aiming to develop a circular economy, the developed methods primarily reference three objectives. The first, ensuring access to clean water and adequate sanitation (SDG 6), is linked to fabric washing processes, solvents used, and various reactions involved. The second, promoting responsible consumption and production of resources (SDG 12), is related to the possibility of producing new textiles or materials from textile waste. The third, addressing the effects of climate change (SDG 13), is linked to waste valorisation and consumption of fossil and energy resources.Textile recycling aims to promote a circular economy by organizing the production of materials from waste with equivalent quality over time. In this vein, depolymerisation methods being studied enable the establishment of a long-term supply chain in which today's used garments can become tomorrow's materials. To ensure these three SDGs are met, it is necessary to study the potential environmental impacts of industrial processes implemented in textile recycling. This study is only meaningful in its final industrial application and not at the fundamental stage, which does not consider industrial scales and material and solvent flows. An initial assessment of methods can be made based on SDGs 12 and 13, but publications do not consistently provide all the necessary data for a comparative evaluation. Degradation yields, which characterize the amount of monomer obtained per mass of treated waste, vary from 62% for enzymatic hydrolysis reactions to 100% for many chemical depolymerisation methods. However, many articles do not consistently provide these values (Table S1, ESI†). While it is possible to say that depolymerisation methods align with these goals, it is not possible to classify them qualitatively based on these criteria.
Paul Anastas and John Warner are the pioneers of green chemistry. They developed an approach for designing chemical processes and products that are environmentally friendly.277 This approach seeks to prevent pollution at its source by limiting the use of harmful substances. The principles of green chemistry emphasize the creation of safer chemicals, the use of catalysts rather than stoichiometric reagents, and the reduction of waste production. According to this approach, pollution prevention should be considered at the molecular level, which involves reducing pollution sources throughout the life cycle of chemicals.278 The same considerations as previously mentioned using the SDGs should be taken into account to classify depolymerisation methods in terms of the 12 principles of green chemistry. The lack of comprehensive data on degradation yields, in particular, does not allow for a rigorous evaluation.
5.2 Green metrics
The methods allowing PET depolymerisation are quite diverse, as demonstrated in the previous chapter. To compare these methods, a first approach allows positioning them in a 2D experimental domain depicting the reaction time and temperature of all the cited studies. Fig. 13 illustrates this experimental domain. It appears that certain methods have been developed in very broad domains such as hydrolysis in acidic media or aminolysis. In contrast, glycolysis or enzymatic methods are often developed in more restricted temperature and reaction time ranges.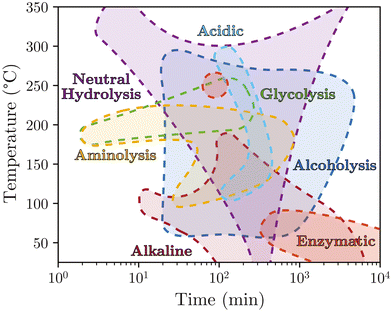 | ||
| Fig. 13 Global mapping of PET depolymerisation methods sorted by typical reaction time and temperature. | ||
Table S1 (ESI†) provides a summary of the cited methods found in the literature, offering quantitative information on material flows between recycled waste, reaction conditions, and depolymerisation yields for monomer production. To rank the reactions based on the data in this table, it is possible to use green metrics. These indices allow for the quantification of contributions from methodological developments in various principles of green chemistry: atom economy, energy efficiency, waste reduction, and so on. The simplest index that can be calculated to classify different reactions is related to atom economy (AE) published in 1991.279 AE is calculated based on stoichiometric data but does not take into account solvent use. In the case of solvolysis reactions, this index is therefore not meaningful, as the solvent will have a considerable effect on various depolymerisation reactions. The second index, E factor, described in 1992, calculates the ratio between the waste mass produced and the product mass purified by different reactions.280 For the calculation of the waste mass generated, a widely used approximation assumes that 90% of the solvents used are industrially recycled, while 10% of the mass of the solvents used becomes waste.
In the case of depolymerisation reactions of textile fibres, this approximation cannot be applied because of the diverse range of methods involved. The production of solvents contaminated with additives, in varying quantities and qualities depending on the recycled fabrics, or the inorganic salts at the end of the hydrolysis reaction, provides no assurance regarding the solvent recyclability. Moreover, in certain reactions, a portion of the solvent is esterified in the depolymerisation process. The combination of these approximations does not allow for a robust classification of depolymerisation methods aimed at recycling PET, especially derived from textile fibres. If calculating the E factor is straightforward to describe a thoroughly validated reaction and has contributed to the success of virtuous reactions,281,282 it is more complex to implement in the case of depolymerisation reactions. Faced with this challenge, Barnard et al. proposed the calculation of an energy economy coefficient (ε) to compare depolymerisation reactions, calculated according to eqn (1):283
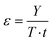 | (1) |
| Study | Year | Code method | Reaction temperature (°C) | Reaction time (min) | Product | Product yield | ε (°C−1 min−1) |
|---|---|---|---|---|---|---|---|
| B. Liu186 | 2018 | MSG | 190 | 1 | BHET | 83.0% | 4.370 × 10−3 |
| S. Nica236 | 2018 | CA | 190 | 3 | BHETA | 100.0% | 1.754 × 10−3 |
| V. Vinitha205 | 2023 | MSG | 150 | 15 | BHETA | 95.00% | 4.222 × 10−4 |
| X. L. Wang169 | 2023 | MA | 80 | 30 | Terephthalic acid | 98.0% | 4.083 × 10−4 |
| V. S. Palekar238 | 2012 | CA | 110 | 40 | BHETA | 89.0% | 2.023 × 10−4 |
| S. Tang153 | 2022 | CoAL | 200 | 30 | DMT | 91.0% | 1.517 × 10−4 |
| D. S. Achilias233 | 2011 | μW AA | 250 | 30 | BHETA | 100.0% | 1.333 × 10−4 |
| P. Lozano Martinez149 | 2021 | SCA | 275 | 30 | TA/DMT/DMT/DET/EG | 94.0% | 1.139 × 10−4 |
| Y. Yang144 | 2002 | SCA | 250 | 40 | DMT | 95.0% | 9.500 × 10−5 |
| R. López-Fonseca189 | 2011 | MSG | 196 | 60 | BHET | 80% | 6.803 × 10−5 |
| H. W. Horn284 | 2012 | AT | 110 | 120 | BAETA | 89% | 6.742 × 10−5 |
| R. M. Musale237 | 2016 | CA | 170 | 90 | BHETA | 97.0% | 6.340 × 10−5 |
| Y. Yang150 | 2023 | SCA | 270 | 60 | DET | 92.0% | 5.679 × 10−5 |
| L. R. Zhang84 | 2013 | NH | 145 | 120 | TPA | 93.0% | 5.345 × 10−5 |
| S. Mishra90 | 2003 | ACH | 120 | 140 | TPA | 87.0% | 5.179 × 10−5 |
| S. Kumagai117 | 2018 | ALH | 180 | 120 | TPA | 100.0% | 4.630 × 10−5 |
| S. Lalhmangaihzuala286 | 2020 | MSG | 190 | 90 | BHET | 79% | 4.620 × 10−5 |
| D. Stanica-Ezeanu81 | 2021 | NH | 205 | 120 | TA; TPA; TGA | 96.0% | 3.902 × 10−5 |
| L. Liu82 | 2005 | NH | 220 | 120 | TA; TPA; TGA | 100.0% | 3.788 × 10−5 |
| F. Quartinello262 | 2017 | MCEH | 250 | 90 | TA | 85.0% | 3.778 × 10−5 |
| M. Imran201 | 2011 | MSG | 300 | 80 | BHET | 90% | 3.750 × 10−5 |
| D. Lei192 | 2022 | MSG | 220 | 90 | BHET | 70.4% | 3.556 × 10−5 |
| L. Liu199 | 2022 | ILG | 193,5 | 125 | BHET | 84.5% | 3.494 × 10−5 |
| Z. Guo206 | 2018 | HCG | 240 | 120 | BHET | 82.0% | 2.847 × 10−5 |
| Y. Peng98 | 2023 | ACH | 280 | 120 | TPA + EG diacetate | 94.0% | 2.798 × 10−5 |
| B. Yan136 | 2023 | ALH | 150 | 240 | TPA | 100.0% | 2.778 × 10−5 |
| Z. Chen191 | 2023 | MSG | 250 | 150 | BHET | 99.7% | 2.659 × 10−5 |
| S. L. Fávaro267 | 2013 | SCA | 255 | 120 | DET | 80.0% | 2.614 × 10−5 |
| W Yang79 | 2021 | NH | 220 | 180 | TPA | 95.0% | 2.399 × 10−5 |
| T. Yoshioka92 | 1994 | ACH | 150 | 300 | TPA | 95.0% | 2.111 × 10−5 |
| Y. S. Parab241 | 2012 | CA | 170 | 240 | BHETA | 86.0% | 2.108 × 10−5 |
| V. Tournier261 | 2020 | EH | 72 | 600 | TPA | 90.0% | 2.083 × 10−5 |
| N. G. Bush198 | 2023 | ILG | 180 | 240 | BHET | 50.0% | 1.157 × 10−5 |
| S. R. Shukla231 | 2006 | CA | 170 | 480 | BHETA | 91.0% | 1.115 × 10−5 |
| S. Liu156,157 | 2013 | ILA | 205 | 480 | DBTP | 95% | 9.654 × 10−6 |
| A. Peterson115 | 2022 | ALH | 90 | 1000 | TPA | 80.0% | 8.889 × 10−6 |
| P. McKeown164 | 2020 | CoAL | 100 | 960 | DMT | 72.0% | 7.500 × 10−6 |
| C. N. Onwucha75 | 2023 | NH | 200 | 1440 | TPA | 98.0% | 3.400 × 10−6 |
Fig. 14 provides the classification of the different depolymerisation methods based on the calculated ε from Table 1.
 | ||
| Fig. 14 Classification of depolymerisation methods for the different studies according to the energy economy coefficient ε. | ||
This figure illustrates that two methods stand out significantly from the others, with a ε coefficient of 4.370 × 10−3 and 1.754 × 10−3 °C−1 × min−1. The first depolymerisation method, metal salts and organocatalysts-assisted glycolysis enables complete depolymerisation of PET (Table S1, ESI†) with a reaction time of 1 minute at 190 °C with a green Zn acetate catalyst. For this method, the source of PET, whether virgin or post-consumer, is not specified. The second method, a catalysed aminolysis (CA), enables complete depolymerisation of PET with a reaction time of 3 minutes at 190 °C, using a monoamide-ester type catalyst: 1,5,7-triazabicyclo [4.4.0]dec-5-ene (TBD). In this method, the degraded polymer is PET waste obtained from post-consumer bottles.
The depolymerized PET is not derived from textile fibres, for these 2 methods and it should be verified whether these conditions are compatible with the structure of PET in fibres. The following are two methods with an ε coefficient approximately four times lesser. The first is an organocatalyst-assisted glycolysis (MSG), using Ag-doped ZnO nanoparticles, and the second is a mixed alcoholysis (MA) using KOH as a catalyst. While the greater simplicity and greener in sourcing is KOH as a catalyst may provide an advantage to this method, it was only developed for PET derived from bottles. In contrast, the MSG method with Ag-doped ZnO nanoparticles was developed for PET derived from fibres. Both of these methods require reaction times ranging from 15 to 30 minutes and temperatures of 80 or 150 °C.
The following 5 methods (CA, CoAL, μW AA, SCA) achieve a calculated ε coefficient ranging between 2 × 10−4 and 1 × 10−4. It is noteworthy that 2 methods involving the same catalysed aminolysis mechanism exhibit an ε coefficient reduced by a factor of 15. The method using ionic liquids as a catalyst showed a reduced BHETA yield (89%) compared to the one using a monoamide-ester type catalyst (100%). Although the operating temperature is less (110 °C), the time required for depolymerisation and the lesser yield are unfavourable for the green ranking associated with the reaction energy efficiency. All other methods analysed in this review exhibit conditions that are unfavourable in terms of energy efficiency, with an ε factor reduced by a factor of 30 to 3000. Regarding enzymatic or mixed methods (MCEH), often presented as a green process in the literature, the lack of data prevents a comprehensive quantitative assessment. For those with available data, the reduced yield (85%), prolonged reaction time, and greater temperature hinder the achievement of a high energy economy coefficient.
6. Conclusions and perspectives
Recycling of PET, particularly PET from textiles, is an ever-evolving field, as demonstrated by the methods presented previously. The comparative study of the environmental impact of the developed reactions, based on the energy efficiency achieved, enables an objective comparison and provides a foundation for future developments of PET depolymerisation methods derived from textile fibres.In the realm of chemical recycling, and to minimize the environmental impact of the reactions involved, there is a series of research into new catalysts or new reaction conditions, used to enhance depolymerisation yield and reduce reaction times and temperatures, all aimed at ensuring a high greenness index.285 Research into new catalysts based on abundant, renewable, and non-toxic resources has shown that PET depolymerisation via glycolysis can be complete, in the presence of an excess of ethylene glycol (EG), at a temperature of 196 °C, using sodium carbonate as a catalyst (López-Fonseca 2011).189 With this catalyst, the reaction time is halved compared to the use of conventional catalysts with a greater environmental impact, such as Mn, Co, Zn, or lead acetate. Organic catalysts, like 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD), appear to be “green” candidates for PET depolymerisation via glycolysis with a large EG excess.284 Current research on new organic catalysts continues through theoretical work and extensive use of modeling.285
Regarding heterogeneous catalysts based on metal oxides, several systems yield promising results in PET recycling. The primary advantage lies in the ease of catalyst recovery, especially for those formulated at the nanoscale, exhibiting a very large surface area, for example, by impregnating silica nanoparticles201 or silver-doped ZnO nanoparticles.205 Other research focuses on the production of new catalytic chemical species from waste valorisation. In PET recycling, a new catalyst produced from orange peel ash, composed of porous materials with a large specific surface area bearing basic sites, shows complete depolymerisation of PET in 90 minutes, with possible catalyst reuse.286
In addition to these methods aimed at depolymerizing PET, there is a series of innovative research focused on producing polymer compatibilizers. These are polymers of variable architecture, such as random copolymers, grafted copolymers, and block copolymers, which can interact with conventional PET from recycling to modify their thermoplastic properties, enabling straightforward fusion/moulding to produce new fibres. Often, the method involves creating mixtures of recycled polymers, for example, PET/PE, PET/PP, and producing a new material by adding an agent that makes the mixture compatible.287,288 As a result, composite fibres were produced from polypropylene (PP) and PET, using a copolymer of PP grafted with acrylic acid (PP-g-AA) as a compatibilizer.289 An approach to modifying PET chains from textile recycling can also be developed to elongate these chains using different chain extenders. These chain extenders can be oxazolines,290 organic phosphites,291 and epoxides.292 The major advantage of these modifications made to PET chains from textile recycling is to homogenize the average PET molecular weights and produce recycled fibres without altering their crystalline structure, allowing for new recycling cycles at the end of the textile's life.293
Finally, PET upcycling is under rapid development with a view to lead to value-added products rather than simple recycling. So far, it has mainly been applied to PET issued from bottles, from packaging, or in pure form and was recently reviewed.294–299 Different valuable substances can be obtained, among them functional TA and EG derivatives,300 organic building blocks and bio-products,301 new polymers such as polyhydroxyalkanoates (PAHs),302 or other valuable bio-based polymers,303–305 and membranes.306 PET upcycling can also produce fuels such as dihydrogen,307,308 foods (bacterial biomass), or functional materials such as metal–organic frameworks (MOFs) or vitrimers.309 The processes involved imply biochemical conversions,301,302 chemical transformations, photocatalysis,310 and electrocatalysis.307
In the case of PET upcycling from textiles and fabrics, examples are currently much rarer. Functional TA derivatives (such as BHET and BHETA) can by produced by direct glycolysis and aminolysis of dyed textiles, catalysed by nanoparticles and under microwave activation.205 Similarly, valuable phthalic acids were obtained from abandoned banners after thermocatalytic treatment using a metal-based alloy favoring decarboxylation and deshydrogenation reactions.311 More elaborated products were also directly produced from (dyed) PET textiles, such as (MOFs).312–314
The many upcycling developments deployed for bottle PET will certainly be applied soon more largely to textiles, supporting circular economy and reducing environmental pollution.
Author contributions
All authors have contributed equally to the drafting of the manuscript and approve the final version.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The work related to this journal has been supported by internal funds from the SMODD team, the CNRS and Toulouse 3, Paul Sabatier University.References
- L. Brivio and F. Tollini, in Towards Circular Economy: Closing the Loop with Chemical Recycling of Solid Plastic Waste, ed. D. Moscatelli and M. Pelucchi, Academic Press, 2022, vol. 60, pp. 215–267 Search PubMed.
- J. W. Hill, J. Am. Chem. Soc., 1930, 52, 4110–4114 Search PubMed.
- W. H. Carothers and J. A. Arvin, J. Am. Chem. Soc., 1929, 51, 2560–2570 Search PubMed.
- W. H. Carothers, J. Am. Chem. Soc., 1929, 51, 2548–2559 Search PubMed.
- J. R. Whinfield and J. T. Dickson, GB Pat, 578079A, 1941 Search PubMed.
- P. W. Morgan, J. Macromol. Sci., Part A: Pure Appl.Chem., 1981, 15, 1113–1131 CrossRef.
- A. Smelik, Fash. Pract., 2023, 15, 279–299 Search PubMed.
- E. Gubbels, T. Heitz, M. Yamamoto, V. Chilekar, S. Zarbakhsh, M. Gepraegs, H. Köpnick, M. Schmidt, W. Brügging, J. Rüter and W. Kaminsky, Ullmann's Encyclopedia of Industrial Chemistry, John Wiley & Sons, Ltd, 2018, pp. 1–30 Search PubMed.
- J. Scheirs and T. E. Long, Modern Polyesters: Chemistry and Technology of Polyesters and Copolyesters, Wiley, 2004 Search PubMed.
- B. Henry, K. Laitala and I. G. Klepp, Sci. Total Environ., 2019, 652, 483–494 Search PubMed.
- C. J. Boreiko and T. G. Rossman, Toxicol. Appl. Pharmacol., 2020, 403, 115156 Search PubMed.
- R. Hufenus, Y. Yan, M. Dauner and T. Kikutani, Materials, 2020, 13, 4298 Search PubMed.
- S. Grishanov, in Handbook of Textile and Industrial Dyeing, ed. M. Clark, Woodhead Publishing, 2011, vol. 1, pp. 28–63 Search PubMed.
- Handbook of Technical Textiles, ed. A. Horrocks and S. Anand, CRC Press, 2000 Search PubMed.
- Q. Dong, A. D. Lele, X. Zhao, S. Li, S. Cheng, Y. Wang, M. Cui, M. Guo, A. H. Brozena, Y. Lin, T. Li, L. Xu, A. Qi, I. G. Kevrekidis, J. Mei, X. Pan, D. Liu, Y. Ju and L. Hu, Nature, 2023, 616, 488–494 CrossRef CAS PubMed.
- S. Zhang, W. Xu, R. Du, W. An, X. Liu, S. Xu and Y.-Z. Wang, Chem. Eng. J., 2023, 470, 144032 CrossRef CAS.
- L. T. J. Korley, T. H. Epps, B. A. Helms and A. J. Ryan, Science, 2021, 373, 66–69 CrossRef CAS PubMed.
- F. Tollini, L. Brivio, P. Innocenti, M. Sponchioni and D. Moscatelli, Chem. Eng. Sci., 2022, 260, 117875 CrossRef CAS.
- R. M. Darbra, J. R. G. Dan, J. Casal, A. Àgueda, E. Capri, G. Fait, M. Schuhmacher, M. Nadal, J. Rovira, V. Grundmann, D. Barceló, A. Ginebreda and D. Guillén, in Global Risk-Based Management of Chemical Additives I: Production, Usage and Environmental Occurrence, ed. B. Bilitewski, R. M. Darbra and D. Barceló, Springer Berlin Heidelberg, Berlin, Heidelberg, 2011, pp. 83–107 Search PubMed.
- R. H. Barker, Environ. Health Perspect., 1975, 11, 41–45 CrossRef CAS PubMed.
- N. N. Mahapatra, Textile Dyes, WPI Publishing, 2016 Search PubMed.
- J. Wang, W. Cheng, Y. Gao, L. Zhu and L. Pei, Polymers, 2019, 11, 520 CrossRef PubMed.
- M. Clark, Handbook of textile and industrial dyeing, Woodhead Publishing Limited, 2011 Search PubMed.
- A. Ketema and A. Worku, J. Chem., 2020, 2020, 6628404 Search PubMed.
- Apparel and Footwear International RSL Management (AFIRM) Group, Restricted Substances List, https://afirm-group.com/wp-content/uploads/2024/04/<?pdb_no 2024?>2024<?pdb END?>_AFIRM_RSL_2024_0404_EN.pdf.
- L. R. Manea, A.-P. Bertea and A. Bertea, IOP Conf. Ser.: Mater. Sci. Eng., 2020, 877, 12062 CAS.
- D. Dilba, Long live plastic, https://elements.evonik.com/Research-and-Innovation/long-live-plastic/.
- M. Y. Soliman, H. A. Othman and A. G. Hassabo, J. Text. Color. Polym. Sci., 2021, 18, 247–252 Search PubMed.
- W. D. Schindler and P. J. Hauser, Chemical Finishing of Textiles, Woodhead Publishing, 2004 Search PubMed.
- B. C. Goswami, R. D. Anandjiwala and D. Hall, Textile Sizing, Taylor \& Francis, 2004 Search PubMed.
- M. M. Houck, R. E. Menold and R. A. Huff, Probl. Forensic Sci., 2001, 46, 217–221 Search PubMed.
- B. J. Holland and J. N. Hay, Polymer, 2002, 43, 1797–1804 CrossRef CAS.
- D. M. Connor, S. D. Allen, D. M. Collard, C. L. Liotta and D. A. Schiraldi, J. Appl. Polym. Sci., 2001, 81, 1675–1682 CrossRef CAS.
- B. Li, J. Yu, S. Lee and M. Ree, Polymer, 1999, 40, 5371–5375 Search PubMed.
- E. L. Lawton and C. J. Setzer, Flame-Retardant Polymeric Materials, Springer US, Boston, MA, 1975, pp. 193–221 Search PubMed.
- A. Shukla, V. Sharma, S. Basak and S. W. Ali, Cellulose, 2019, 26, 8191–8208 Search PubMed.
- V. Sharma and S. Wazed Ali, Sustainable Chem. Pharm., 2022, 27, 100645 Search PubMed.
- M. MacLeod, H. P. H. Arp, M. B. Tekman and A. Jahnke, Science, 2021, 373, 61–65 Search PubMed.
- C. M. Rochman, M. A. Browne, B. S. Halpern, B. T. Hentschel, E. Hoh, H. K. Karapanagioti, L. M. Rios-Mendoza, H. Takada, S. Teh and R. C. Thompson, Nature, 2013, 494, 169–171 CrossRef CAS PubMed.
- I. Paul-Pont, J.-F. Ghiglione, E. Gastaldi, A. Ter Halle, A. Huvet, S. Bruzaud, F. Lagarde, F. Galgani, G. Duflos, M. George and P. Fabre, Waste Manage., 2023, 157, 242–248 Search PubMed.
- K. Niinimäki, G. Peters, H. Dahlbo, P. Perry, T. Rissanen and A. Gwilt, Nat. Rev. Earth Environ., 2020, 1, 189–200 Search PubMed.
- F. Uddin, Textile Manufacturing Processes, IntechOpen, Rijeka, 2019 Search PubMed.
- N. Šajn, Environmental impact of the textile and clothing industry. What consumers need to know, https://www.europarl.europa.eu/thinktank/en/document/EPRS_BRI(2019)633143.
- J. Janmark, K.-H. Magnus, M. Strand, N. Langguth and S. Hedrich, McKinsey Co., 2022, 76, https://www.mckinsey.com/industries/retail/our-insights/scalingtextile-recycling-in-europe-turning-waste-into-value Search PubMed.
- D. Watson, A. K. Aare, S. Trzepacz and P. Christine Dahl Petersen, Eur. Cloth. Action Plan, 2018, 89, https://www.ecap.eu.com/wp-content/uploads/2018/07/ECAP-Textile-collection-in-European-cities_full-report_with-summary.pdf Search PubMed.
- A. Morlet, R. Opsomer, S. Herrmann, L. Balmond, C. Gillet and L. Fuchs, Ellen MacArthur Found., 2017, 1–150 Search PubMed.
- S. Haslinger, M. Hummel, A. Anghelescu-Hakala, M. Määttänen and H. Sixta, Waste Manage., 2019, 97, 88–96 Search PubMed.
- R. Kumar, Mater. Today Proc., 2021, 37, 2382–2386 CrossRef CAS.
- G. Sandin and G. M. Peters, J. Cleaner Prod., 2018, 184, 353–365 CrossRef CAS.
- L. Bartolome, M. Imran, B. G. Cho, W. A. Al-Masry and D. H. Kim, Material Recycling - Trends and Perspectives, InTech, 2012, pp. 65–84 Search PubMed.
- A. Grant, V. Lahme, T. Connock and L. Lugal, Zero Waste Eur., 2022, https://zerowasteeurope.eu/library/how-circular-is-pet/ Search PubMed.
- N. A. Rorrer, S. Nicholson, A. Carpenter, M. J. Biddy, N. J. Grundl and G. T. Beckham, Joule, 2019, 3, 1006–1027 CrossRef CAS.
- A. M. Al-Sabagh, F. Z. Yehia, G. Eshaq, A. M. Rabie and A. E. ElMetwally, Egypt. J. Pet., 2016, 25, 53–64 CrossRef.
- R. Geyer, J. R. Jambeck and K. L. Law, Sci. Adv., 2017, 3, e1700782 CrossRef PubMed.
- T. Thiounn and R. C. Smith, J. Polym. Sci., 2020, 58, 1347–1364 Search PubMed.
- A. Rahimi and J. M. García, Nat. Rev. Chem., 2017, 1, 46 CrossRef.
- S. Cuc and M. Vidovic, Oper. Supply Chain Manag. An Int. J., 2014, 4, 108–115 CrossRef.
- I. Boustead, Eco-profiles of the European Plastics Industry - Polyethylene Terephthalate (PET) (Bottle grade), https://www.inference.org.uk/sustainable/LCA/elcd/external_docs/petb_31116f00-fabd-11da-974d-0800200c9a66.pdf.
- J. M. Garcia and M. L. Robertson, Science, 2017, 358, 870–872 CrossRef CAS PubMed.
- L. Dai, N. Zhou, Y. Lv, Y. Cheng, Y. Wang, Y. Liu, K. Cobb, P. Chen, H. Lei and R. Ruan, Prog. Energy Combust. Sci., 2022, 93, 101021 Search PubMed.
- J. M. Encinar and J. F. González, Fuel Process. Technol., 2008, 89, 678–686 CrossRef CAS.
- R. Blanchard and T. H. Mekonnen, J. Environ. Chem. Eng., 2022, 10, 108810 Search PubMed.
- S. Sharifian and N. Asasian-Kolur, J. Anal. Appl. Pyrolysis, 2022, 163, 105496 Search PubMed.
- Z. Chen, W. Wei, B.-J. Ni and H. Chen, Environ. Funct. Mater., 2022, 1, 34–48 Search PubMed.
- G. Duman, J. Water Process Eng., 2021, 43, 102286 CrossRef.
- E.Ü Parmakoğlu, A. Çay and J. Yanık, AATCC J. Res., 2023, 10, 133–143 Search PubMed.
- A. Khan, M. Awais and M. Mohsin, Biomass Convers. Biorefin., 2023, 1–16 Search PubMed.
- S. Opperskalski, A. Franz, A. Patanè, S. Siew and E. Tan, Text. Exch., 2022, 1–118 Search PubMed.
- PlasticEurope, Plastics – the Facts 2020, https://plasticseurope.org/knowledge-hub/plastics-the-facts-2020/.
- A. B. Raheem, Z. Z. Noor, A. Hassan, M. K. Abd Hamid, S. A. Samsudin and A. H. Sabeen, J. Cleaner Prod., 2019, 225, 1052–1064 CrossRef CAS.
- R. B. Baloyi, O. J. Gbadeyan, B. Sithole and V. Chunilall, Text. Res. J., 2023, 94, 508–529 Search PubMed.
- S.-L. Loo, E. Yu and X. Hu, J. Environ. Chem. Eng., 2023, 11, 110482 CrossRef CAS.
- P. Pereira, P. E. Savage and C. W. Pester, ACS Sustainable Chem. Eng., 2023, 11, 7203–7209 CrossRef CAS.
- A. Jaime-Azuara, T. H. Pedersen and R. Wimmer, Green Chem., 2023, 25, 2711–2722 RSC.
- C. N. Onwucha, C. O. Ehi-Eromosele, S. O. Ajayi, M. Schaefer, S. Indris and H. Ehrenberg, Ind. Eng. Chem. Res., 2023, 62, 6378–6385 CrossRef CAS.
- M. J. Kang, H. J. Yu, J. Jegal, H. S. Kim and H. G. Cha, Chem. Eng. J., 2020, 398, 125655 CrossRef CAS.
- C.-Y. Kao, B.-Z. Wan and W.-H. Cheng, Ind. Eng. Chem. Res., 1998, 37, 1228–1234 CrossRef CAS.
- J. R. Campanelli, M. R. Kamal and D. G. Cooper, J. Appl. Polym. Sci., 1994, 54, 1731–1740 CrossRef CAS.
- W. Yang, R. Liu, C. Li, Y. Song and C. Hu, Waste Manage., 2021, 135, 267–274 CrossRef CAS.
- S. Mahadevan Subramanya, Y. Mu and P. E. Savage, ACS Eng. Au, 2022, 2, 507–514 CrossRef CAS.
- D. Stanica-Ezeanu and D. Matei, Sci. Rep., 2021, 11, 4431 CrossRef CAS PubMed.
- L. Liu, D. Zhang, L. An, H. Zhang and Y. Tian, J. Appl. Polym. Sci., 2005, 95, 719–723 CrossRef CAS.
- W. L. Zhang, X. N. Shi, X. Zhang, C. H. Han and D. Zhang, Adv. Mater. Res., 2012, 550–553, 792–797 CAS.
- L. Zhang, J. Gao, J. Zou and F. Yi, J. Appl. Polym. Sci., 2013, 130, 2790–2795 CrossRef CAS.
- M. Yan, Y. Yang, F. Chen, D. Hantoko, A. Pariatamby and E. Kanchanatip, Environ. Sci. Pollut. Res., 2023, 30, 102560–102573 CrossRef CAS.
- S. Hongkailers, Y. Jing, Y. Wang, N. Hinchiranan and N. Yan, ChemSusChem, 2021, 14, 4330–4339 CrossRef CAS PubMed.
- R. Li, W. Zeng, R. Zhao, Y. Zhao, Y. Wang, F. Zhang, M. Tang, Y. Wang, X. Chang, F. Wu and Z. Liu, Nano Res., 2023, 16, 12223–12229 CrossRef CAS.
- H. Su, Y. Hu, H. Feng, L. Zhu and S. Wang, ACS Sustainable Chem. Eng., 2023, 11, 578–586 CrossRef CAS.
- H. Abedsoltan, Polym. Eng. Sci., 2023, 63, 2651–2674 CrossRef CAS.
- S. Mishra, A. S. Goje and V. S. Zope, Polym. React. Eng., 2003, 11, 79–99 CrossRef CAS.
- S. H. Park and S. H. Kim, Fash. Text., 2014, 1, 1 CrossRef.
- T. Yoshioka, N. Okayama and A. Okuwaki, Ind. Eng. Chem. Res., 1998, 37, 336–340 CrossRef CAS.
- T. Yoshioka, T. Motoki and A. Okuwaki, Ind. Eng. Chem. Res., 2001, 40, 75–79 CrossRef CAS.
- G. M. de Carvalho, E. C. Muniz and A. F. Rubira, Polym. Degrad. Stab., 2006, 91, 1326–1332 CrossRef CAS.
- R. K. Al-tamimi, M. N. Khalaf, M. Sabri and L. Sabri, J. Mater. Environ. Sci., 2011, 2, 88–93 CAS.
- C. Costa, A. Viana, C. Silva, E. F. Marques and N. G. Azoia, Waste Manage., 2022, 153, 99–109 CrossRef CAS PubMed.
- B. Ye, R. Zhou, Z. Zhong, S. Wang, H. Wang and Z. Hou, Green Chem., 2023, 25, 7243–7252 RSC.
- Y. Peng, J. Yang, C. Deng, J. Deng, L. Shen and Y. Fu, Nat. Commun., 2023, 14, 3249 CrossRef CAS PubMed.
- X. Li, H. Lu, W. Guo, G. Cao, H. Liu and Y. Shi, AIChE J., 2015, 61, 200–214 CrossRef CAS.
- H. Abedsoltan, I. S. Omodolor, A. C. Alba-Rubio and M. R. Coleman, Polymer, 2021, 222, 123620 CrossRef CAS.
- W. Yang, J. Wang, L. Jiao, Y. Song, C. Li and C. Hu, Green Chem., 2022, 24, 1362–1372 RSC.
- L. Kárpáti, G. Szarka, M. Hartman and V. Vargha, Period. Polytech., Chem. Eng., 2018, 62, 336–344 CrossRef.
- C. Jehanno, I. Flores, A. P. Dove, A. J. Müller, F. Ruipérez and H. Sardon, Green Chem., 2018, 20, 1205–1212 RSC.
- A. S. Amarasekara, J. A. Gonzalez and V. C. Nwankwo, J. Ion. Liq., 2022, 2, 100021 CrossRef.
- A. N. Paparella, S. Perrone, A. Salomone, F. Messa, L. Cicco, V. Capriati, F. M. Perna and P. Vitale, Catalysts, 2023, 13, 1035 CrossRef CAS.
- S. Hashemizad, M. Montazer and A. Rashidi, J. Appl. Polym. Sci., 2012, 125, 1176–1184 CrossRef CAS.
- R. Chen, S. Deng, T. Cui, S. Duan, Q. Jia and L. Zhang, Prog. Rubber, Plast. Recycl. Technol., 2024, 40, 77–97 CrossRef.
- M. J. Collins, S. H. Zeronian and M. Semmelmeyer, J. Appl. Polym. Sci., 1991, 42, 2149–2162 CrossRef CAS.
- A. M. Grancarić and N. Kallay, J. Appl. Polym. Sci., 1993, 49, 175–181 CrossRef.
- S. Kumar and C. Guria, J. Macromol. Sci., Part A: Pure Appl. Chem., 2005, 42, 237–251 CrossRef.
- W. Tsen, K. Hsiao and Y. Shu, J. Appl. Polym. Sci., 2009, 113, 1822–1827 CrossRef CAS.
- H. Tavanai, J. Text. Inst., 2009, 100, 633–639 CrossRef CAS.
- Y. Kawahara, T. Yoshioka, W. Takarada, T. Kikutani and M. Tsuji, J. Fiber Sci. Technol., 2016, 72, 9–16 CrossRef.
- A. Sarno, K. Olafsen, S. Kubowicz, F. Karimov, S. T. L. Sait, L. Sørensen and A. M. Booth, Environ. Sci. Technol. Lett., 2021, 8, 250–255 CrossRef CAS.
- A. Peterson, J. Wallinder, J. Bengtsson, A. Idström, M. Bialik, K. Jedvert and H. de la Motte, Sustainability, 2022, 14, 7272 CrossRef CAS.
- J. Bengtsson, A. Peterson, A. Idström, H. de la Motte and K. Jedvert, Sustainability, 2022, 14, 6911 CrossRef CAS.
- S. Kumagai, S. Hirahashi, G. Grause, T. Kameda, H. Toyoda and T. Yoshioka, J. Mater. Cycles Waste Manag., 2018, 20, 439–449 CrossRef CAS.
- T. Kaurin, T. Pušić and M. Čurlin, Polymers, 2022, 14, 3088 CrossRef CAS PubMed.
- J. M. Kang, M. G. Kim, J. E. Lee, J. W. Ko, I. J. Kim, J. Y. Lee, D. J. Lee, S. I. Ko, D. H. Jung and S. G. Lee, Polymers, 2020, 12, 1243 CrossRef CAS PubMed.
- N. Prorokova, A. Chorev, S. Kuzmin, S. Vavilova and V. Prorokov, Chem. Chem. Technol., 2014, 8, 293–302 CrossRef CAS.
- A. Boondaeng, J. Keabpimai, P. Srichola, P. Vaithanomsat, C. Trakunjae and N. Niyomvong, Polymers, 2023, 15, 1964 CrossRef CAS PubMed.
- E. Gholamzad, K. Karimi and M. Masoomi, Chem. Eng. J., 2014, 253, 40–45 CrossRef CAS.
- S. Youn and C. H. Park, Text. Res. J., 2019, 89, 959–974 CrossRef CAS.
- E. S. Kim, C. H. Lee and S. H. Kim, J. Appl. Polym. Sci., 2009, 112, 3071–3078 CrossRef CAS.
- J. H. Lee, S. H. Park, K. W. Oh, C. H. Lee and S. H. Kim, Polym. Int., 2012, 61, 657–663 CrossRef CAS.
- S. Niu, T. Wakida, S. Ogasawara, H. Fujimatsu and S. Takekoshi, Text. Res. J., 1995, 65, 771–775 CrossRef CAS.
- S. M. Gawish, S. Mosleh and A. M. Ramadan, J. Appl. Polym. Sci., 2002, 85, 1652–1660 CrossRef CAS.
- I. Čorak, A. Tarbuk, D. Đorđević, K. Višić and L. Botteri, Materials, 2022, 15, 1530 CrossRef PubMed.
- R. M. Musale and S. R. Shukla, J. Text. Inst., 2017, 108, 467–471 CrossRef CAS.
- A. Palme, A. Peterson, H. de la Motte, H. Theliander and H. Brelid, Text. Cloth. Sustain., 2017, 3, 4 CrossRef.
- M. Rahman and G. C. East, Text. Res. J., 2009, 79, 728–736 CrossRef CAS.
- D. Zhou, L. Wang, F. Zhang, J. Wu, H. Wang and J. Yang, Adv. Sustainable Syst., 2022, 6, 2100516 CrossRef CAS.
- V. Ramopoulos, Energy-efficient, scalable and modular industrial microwave applicator for high temperature alkaline hydrolysis of PET, KIT Scientific Publishing, Karlsruhe, 2023 Search PubMed.
- V. Štrukil, ChemSusChem, 2021, 14, 330–338 CrossRef PubMed.
- S. Zhang, Y. Xue, Y. Wu, Y.-X. Zhang, T. Tan and Z. Niu, Chem. Sci., 2023, 14, 6558–6563 RSC.
- B. Yan, S. Zhang, M. Zhang, Y. Yu, T. Qin, L. Tang, Y. Liu, W. Wu and Q. Mei, Chem. Eng. J., 2023, 474, 145697 CrossRef CAS.
- H. Gruschke, W. Hammerschick and H. Medem, US Pat, 3403115A, 1968 Search PubMed.
- R. Lotz, G. Wick and C. Neuhaus, US Pat, 3321510A, 1967 Search PubMed.
- E. Heisenberg, E. Siggel and R. Lotz, US Pat, 3037050A, 1962 Search PubMed.
- J. Otera, Chem. Rev., 1993, 93, 1449–1470 CrossRef CAS.
- P. R. Fehlandt and H. Adkins, J. Am. Chem. Soc., 1935, 57, 193–195 CrossRef CAS.
- G. B. Hatch and H. Adkins, J. Am. Chem. Soc., 1937, 59, 1694–1696 CrossRef CAS.
- M. Goto, H. Koyamoto, A. Kodama, T. Hirose and S. Nagaoka, J. Phys.: Condens. Matter, 2002, 14, 136–144 Search PubMed.
- Y. Yang, Y. Lu, H. Xiang, Y. Xu and Y. Li, Polym. Degrad. Stab., 2002, 75, 185–191 CrossRef CAS.
- T. Sako, T. Sugeta, K. Otake, N. Nakazawa, M. Sato, K. Namiki and M. Tsugumi, J. Chem. Eng. JAPAN, 1997, 30, 342–346 CrossRef CAS.
- Q. Liu, R. Li and T. Fang, Chem. Eng. J., 2015, 270, 535–541 CrossRef CAS.
- M. Genta, F. Yano, Y. Kondo, W. Matsubara and S. Oomoto, Mitsubishi Heavy Ind. Ltd., Tech. Rev., 2003, 40, 1–4 Search PubMed.
- K. Yu, J. Liu, J. Sun, Z. Shen and J. Yin, J. Supercrit. Fluids, 2023, 194, 105837 CrossRef CAS.
- P. Lozano-Martinez, T. Torres-Zapata and N. Martin-Sanchez, ACS Sustainable Chem. Eng., 2021, 9, 9846–9853 CrossRef CAS.
- Y. Yang, F. Chen, T. Shen, A. Pariatamby, X. Wen, M. Yan and E. Kanchanatip, Process Saf. Environ. Prot., 2023, 173, 881–892 CrossRef CAS.
- Z. T. Laldinpuii, V. Khiangte, S. Lalhmangaihzuala, C. Lalmuanpuia, Z. Pachuau, C. Lalhriatpuia and K. Vanlaldinpuia, J. Polym. Environ., 2022, 30, 1600–1614 CrossRef CAS.
- P. A. Gangotena, S. Ponce, Á. Gallo-Córdova, D. A. Streitwieser and J. R. Mora, ChemistrySelect, 2022, 7, e202103765 CrossRef CAS.
- S. Tang, F. Li, J. Liu, B. Guo, Z. Tian and J. Lv, J. Environ. Chem. Eng., 2022, 10, 107927 CrossRef CAS.
- M. Ma, S. Wang, Y. Liu, H. Yu, S. Yu, C. Ji, H. Li, G. Nie and S. Liu, J. Appl. Polym. Sci., 2022, 139, e52814 CrossRef CAS.
- T. T. Le Bui, D. A. Nguyen, S. V. Ho and H. T. N. Uong, J. Appl. Polym. Sci., 2016, 133, 1–11 Search PubMed.
- S. Liu, L. Zhou, L. Li, S. Yu, F. Liu, C. Xie and Z. Song, J. Polym. Res., 2013, 20, 310 CrossRef.
- S. Liu, Z. Wang, L. Li, S. Yu, C. Xie and F. Liu, J. Appl. Polym. Sci., 2013, 130, 1840–1844 CrossRef CAS.
- A. Kržan, J. Appl. Polym. Sci., 1998, 69, 1115–1118 CrossRef.
- M. N. Siddiqui, H. H. Redhwi and D. S. Achilias, J. Anal. Appl. Pyrolysis, 2012, 98, 214–220 CrossRef CAS.
- M. M. A. Nikje and F. Nazari, Polimery, 2009, 54, 635–638 CrossRef.
- M. M. A. Nikje and F. Nazari, Adv. Polym. Technol., 2006, 25, 242–246 CrossRef.
- D. S. Achilias, in Microwave-assisted Polymer Synthesis, ed. R. Hoogenboom, U. S. Schubert and F. Wiesbrock, Springer International Publishing, Cham, 2016, pp. 309–346 Search PubMed.
- H. Beneš, J. Slabá, Z. Walterová and D. Rais, Polym. Degrad. Stab., 2013, 98, 2232–2243 CrossRef.
- P. McKeown, M. Kamran, M. G. Davidson, M. D. Jones, L. A. Román-Ramírez and J. Wood, Green Chem., 2020, 22, 3721–3726 RSC.
- J. Tang, X. Meng, X. Cheng, Q. Zhu, D. Yan, Y. Zhang, X. Lu, C. Shi and X. Liu, Ind. Eng. Chem. Res., 2023, 62, 4917–4927 CrossRef CAS.
- D. D. Pham and J. Cho, Green Chem., 2021, 23, 511–525 RSC.
- L.-C. Hu, A. Oku, E. Yamada and K. Tomari, Polym. J., 1997, 29, 708–712 CrossRef CAS.
- L. Cosimbescu, D. R. Merkel, J. Darsell and G. Petrossian, Ind. Eng. Chem. Res., 2021, 60, 12792–12797 CrossRef CAS.
- X.-L. Wang, W.-L. An, R. Du, F. Tian, Y. Yang, X. Zhao, S. Xu and Y.-Z. Wang, J. Environ. Chem. Eng., 2023, 11, 109434 CrossRef CAS.
- X.-L. Wang, W.-L. An, F. Tian, Y. Yang, X. Zhao, P.-P. Xu, S. Xu and Y.-Z. Wang, ACS Sustainable Chem. Eng., 2020, 8, 16010–16019 CrossRef CAS.
- J. J. R. Arias and W. Thielemans, Green Chem., 2021, 23, 9945–9956 RSC.
- H. İ İçoğlu, Tekst. ve Konfeksiyon, 2022, 32, 57–64 CrossRef.
- J. T. Macdowell, US Pat, 3222299A, 1965 Search PubMed.
- H. Yue, Y. Zhao, X. Ma and J. Gong, Chem. Soc. Rev., 2012, 41, 4218–4244 RSC.
- U. R. Vaidya and V. M. Nadkarni, J. Appl. Polym. Sci., 1989, 38, 1179–1190 CrossRef CAS.
- U. R. Vaidya and V. M. Nadkarni, J. Appl. Polym. Sci., 1988, 35, 775–785 CrossRef CAS.
- U. R. Vaidya and V. M. Nadkarni, Ind. Eng. Chem. Res., 1988, 27, 2056–2060 CrossRef CAS.
- U. R. Vaidya and V. M. Nadkarni, J. Appl. Polym. Sci., 1987, 34, 235–245 CrossRef CAS.
- K. Ghosal and C. Nayak, Mater. Adv., 2022, 3, 1974–1992 RSC.
- M. Imran, B.-K. Kim, M. Han, B. G. Cho and D. H. Kim, Polym. Degrad. Stab., 2010, 95, 1686–1693 CrossRef CAS.
- J.-W. Chen and L. Chen, J. Appl. Polym. Sci., 1999, 73, 35–40 CrossRef CAS.
- D. Gintis, Makromol. Chem., Macromol. Symp., 1992, 57, 185–190 CrossRef CAS.
- S. N. Tong, D. S. Chen, C. C. Chen and L. Z. Chung, Polymer, 1983, 24, 469–472 CrossRef CAS.
- S. C. Kosloski-Oh, Z. A. Wood, Y. Manjarrez, J. P. de los Rios and M. E. Fieser, Mater. Horiz., 2021, 8, 1084–1129 RSC.
- S. Baliga and W. T. Wong, J. Polym. Sci., Part A: Polym. Chem., 1989, 27, 2071–2082 CrossRef CAS.
- B. Liu, X. Lu, Z. Ju, P. Sun, J. Xin, X. Yao, Q. Zhou and S. Zhang, Ind. Eng. Chem. Res., 2018, 57, 16239–16245 CrossRef CAS.
- N. D. Pingale and S. R. Shukla, Eur. Polym. J., 2008, 44, 4151–4156 CrossRef CAS.
- D.-T. Van-Pham, Q.-H. Le, T.-N. Lam, C.-N. Nguyen and W. Sakai, Polym. Degrad. Stab., 2020, 179, 109257 CrossRef CAS.
- R. López-Fonseca, I. Duque-Ingunza, B. de Rivas, L. Flores-Giraldo and J. I. Gutiérrez-Ortiz, Chem. Eng. J., 2011, 168, 312–320 CrossRef.
- K. Fukushima, O. Coulembier, J. M. Lecuyer, H. A. Almegren, A. M. Alabdulrahman, F. D. Alsewailem, M. A. Mcneil, P. Dubois, R. M. Waymouth, H. W. Horn, J. E. Rice and J. L. Hedrick, J. Polym. Sci., Part A: Polym. Chem., 2011, 49, 1273–1281 CrossRef CAS.
- Z. Chen, H. Sun, W. Kong, L. Chen and W. Zuo, Green Chem., 2023, 25, 4429–4437 RSC.
- D. Lei, X.-L. Sun, S. Hu, H. Cheng, Q. Chen, Q. Qian, Q. Xiao, C. Cao, L. Xiao and B. Huang, Ind. Eng. Chem. Res., 2022, 61, 4794–4802 CrossRef CAS.
- H. Wang, Z. Li, Y. Liu, X. Zhang and S. Zhang, Green Chem., 2009, 11, 1568–1575 RSC.
- Q. Wang, X. Yao, S. Tang, X. Lu, X. Zhang and S. Zhang, Green Chem., 2012, 14, 2559–2566 RSC.
- Y. Liu, X. Yao, H. Yao, Q. Zhou, J. Xin, X. Lu and S. Zhang, Green Chem., 2020, 22, 3122–3131 RSC.
- A. P. Abbott, G. Capper, D. L. Davies, H. L. Munro, R. K. Rasheed and V. Tambyrajah, Chem. Commun., 2001, 1, 2010–2011 RSC.
- Q. Wang, X. Yao, Y. Geng, Q. Zhou, X. Lu and S. Zhang, Green Chem., 2015, 17, 2473–2479 RSC.
- N. G. Bush, C. H. Dinh, C. L. Catterton and M. E. Fieser, RSC Sustain., 2023, 1, 938–947 RSC.
- L. Liu, H. Yao, Q. Zhou, D. Yan, J. Xu and X. Lu, ACS Eng. Au, 2022, 2, 350–359 CrossRef CAS.
- S. R. Shukla, V. Palekar and N. Pingale, J. Appl. Polym. Sci., 2008, 110, 501–506 CrossRef CAS.
- M. Imran, K. Lee, Q. Imtiaz, B.-K. Kim, M. Han, B. G. Cho and D. H. Kim, J. Nanosci. Nanotechnol., 2011, 11, 824–828 CrossRef CAS PubMed.
- I. Yunita, S. Putisompon, P. Chumkaeo, T. Poonsawat and E. Somsook, Chem. Pap., 2019, 73, 1547–1560 CrossRef CAS.
- Q. Suo, J. Zi, Z. Bai and S. Qi, Catal. Lett., 2017, 147, 240–252 CrossRef CAS.
- I. Cano, C. Martin, J. A. Fernandes, R. W. Lodge, J. Dupont, F. A. Casado-Carmona, R. Lucena, S. Cardenas, V. Sans and I. de Pedro, Appl. Catal., B, 2020, 260, 118110 CrossRef CAS.
- V. Vinitha, M. Preeyanghaa, M. Anbarasu, B. Neppolian and V. Sivamurugan, Environ. Sci. Pollut. Res., 2023, 30, 75401–75416 CrossRef CAS PubMed.
- Z. Guo, K. Lindqvist and H. de la Motte, J. Appl. Polym. Sci., 2018, 135, 46285 CrossRef.
- Z. Guo, M. Eriksson, H. de la Motte and E. Adolfsson, J. Cleaner Prod., 2021, 283, 124579 CrossRef CAS.
- B. Shojaei, M. Abtahi and M. Najafi, Polym. Adv. Technol., 2020, 31, 2912–2938 CrossRef CAS.
- G. P. Karayannidis and D. S. Achilias, Macromol. Mater. Eng., 2007, 292, 128–146 CrossRef CAS.
- D. Paszun and T. Spychaj, Ind. Eng. Chem. Res., 1997, 36, 1373–1383 CrossRef CAS.
- G. Farrow, D. Ravens and I. Ward, Polymer, 1962, 3, 17–25 CrossRef CAS.
- S. A. Holmes, J. Appl. Polym. Sci., 1996, 61, 255–260 CrossRef CAS.
- M. J. Collins, S. H. Zeronian and M. L. Marshall, Macromol. Sci., Part A: Pure Appl.Chem., 1991, 28, 775–792 CrossRef.
- M. S. Ellison, L. D. Fisher, K. W. Alger and S. H. Zeronian, J. Appl. Polym. Sci., 1982, 27, 247–257 CrossRef CAS.
- J. R. Overton and S. K. Haynes, J. Polym. Sci., Polym. Symp., 1973, 43, 9–17 CrossRef.
- E. Lorusso, Y. Feng, J. Schneider, L. Kamps, N. Parasothy, T. Mayer-Gall, J. S. Gutmann and W. Ali, Nano Sel., 2022, 3, 594–607 CrossRef CAS.
- E. Lorusso, W. Ali, M. Leniart, B. Gebert, M. Oberthür and J. S. Gutmann, Polymers, 2020, 12, 6 CrossRef CAS PubMed.
- J. Zhou, M. Li, L. Zhong, F. Zhang and G. Zhang, Colloids Surf., A, 2017, 513, 146–152 CrossRef CAS.
- V. Popescu, A. Muresan, O. Constandache, G. Lisa, E. I. Muresan, C. Munteanu and I. Sandu, Ind. Eng. Chem. Res., 2014, 53, 16652–16663 CrossRef CAS.
- T. Ohe and Y. Yoshimura, Sen'i Gakkaishi, 2012, 68, 253–258 CrossRef CAS.
- T. Spychaj, E. Fabrycy, S. Spychaj and M. Kacperski, J. Mater. Cycles Waste Manag., 2001, 3, 24–31 CAS.
- C. N. Hoang and Y. H. Dang, Polym. Degrad. Stab., 2013, 98, 697–708 CrossRef CAS.
- R. K. Soni, S. Singh and K. Dutt, J. Appl. Polym. Sci., 2010, 115, 3074–3080 CrossRef CAS.
- N. George and T. Kurian, Prog. Rubber, Plast. Recycl. Technol., 2016, 32, 153–168 CrossRef.
- Y. S. Parab, N. D. Pingale and S. R. Shukla, J. Appl. Polym. Sci., 2012, 125, 1103–1107 CrossRef CAS.
- R. K. Soni, K. Dutt, A. Jain, S. Soam and S. Singh, J. Appl. Polym. Sci., 2009, 113, 1090–1096 CrossRef CAS.
- V. S. Palekar, A. J. Damle and S. R. Shukla, Eur. J. Med. Chem., 2009, 44, 5112–5116 CrossRef CAS PubMed.
- V. S. Palekar, N. D. Pingale and S. R. Shukla, Color. Technol., 2010, 126, 86–91 CAS.
- G. M. M. Sadeghi, R. Shamsi and M. Sayaf, J. Polym. Environ., 2011, 19, 522–534 CrossRef CAS.
- N. D. Pingale and S. R. Shukla, Eur. Polym. J., 2009, 45, 2695–2700 CrossRef CAS.
- S. R. Shukla and A. M. Harad, Polym. Degrad. Stab., 2006, 91, 1850–1854 CrossRef CAS.
- G. Pastore, R. Giacomantonio, G. Lupidi, F. Stella, R. Risoluti, E. Papa, R. Ballini, F. Sarasini, J. Tirillò, E. Marcantoni and S. Gabrielli, Front. Chem., 2023, 11, 1234763 CrossRef CAS PubMed.
- D. S. Achilias, G. P. Tsintzou, A. K. Nikolaidis, D. N. Bikiaris and G. P. Karayannidis, Polym. Int., 2011, 60, 500–506 CrossRef CAS.
- K. Fukushima, J. M. Lecuyer, D. S. Wei, H. W. Horn, G. O. Jones, H. A. Al-Megren, A. M. Alabdulrahman, F. D. Alsewailem, M. A. McNeil, J. E. Rice and J. L. Hedrick, Polym. Chem., 2013, 4, 1610–1616 RSC.
- J. Demarteau, I. Olazabal, C. Jehanno and H. Sardon, Polym. Chem., 2020, 11, 4875–4882 RSC.
- S. Nica, M. Duldner, A. Hanganu, S. Iancu, B. Cursaru, A. Sarbu, P. Filip and E. Bartha, Rev. Chim., 2018, 69, 2613–2616 CrossRef CAS.
- R. M. Musale and S. R. Shukla, Int. J. Plast. Technol., 2016, 20, 106–120 CrossRef CAS.
- V. S. Palekar, R. V. Shah and S. R. Shukla, J. Appl. Polym. Sci., 2012, 126, 1174–1181 CrossRef CAS.
- M. E. Tawfik, N. M. Ahmed and S. B. Eskander, J. Appl. Polym. Sci., 2011, 120, 2842–2855 CrossRef CAS.
- V. Vinitha, M. Preeyanghaa, M. Anbarasu, G. Jeya, B. Neppolian and V. Sivamurugan, J. Polym. Environ., 2022, 30, 3566–3581 CrossRef CAS.
- Y. S. Parab, R. V. Shah and S. R. Shukla, Curr. Chem. Lett., 2012, 1, 81–90 CrossRef CAS.
- A. Mittal, R. K. Soni, K. Dutt and S. Singh, J. Hazard. Mater., 2010, 178, 390–396 CrossRef CAS PubMed.
- K. P. Blackmon, D. W. Fox and S. J. Shafer, US Pat, 4973746A, 1990 Search PubMed.
- L. Shi and L. Zhu, ChemBioChem, 2024, 25, e202300578 CrossRef CAS PubMed.
- C. Liu, C. Shi, S. Zhu, R. Wei and C.-C. Yin, Biochem. Biophys. Res. Commun., 2019, 508, 289–294 CrossRef CAS PubMed.
- S. Jaiswal, B. Sharma and P. Shukla, Environ. Technol. Innov., 2020, 17, 100567 CrossRef.
- F. Kawai, T. Kawabata and M. Oda, Appl. Microbiol. Biotechnol., 2019, 103, 4253–4268 CrossRef CAS PubMed.
- M. T. Zumstein, D. Rechsteiner, N. Roduner, V. Perz, D. Ribitsch, G. M. Guebitz, H.-P. E. Kohler, K. McNeill and M. Sander, Environ. Sci. Technol., 2017, 51, 7476–7485 CrossRef CAS PubMed.
- R.-J. Müller, I. Kleeberg and W.-D. Deckwer, J. Biotechnol., 2001, 86, 87–95 CrossRef PubMed.
- D. Kint and S. Muñoz-Guerra, Polym. Int., 1999, 48, 346–352 CrossRef CAS.
- R. Koshti, L. Mehta and N. Samarth, J. Polym. Environ., 2018, 26, 3520–3529 CrossRef CAS.
- T. Brueckner, A. Eberl, S. Heumann, M. Rabe and G. M. Guebitz, J. Polym. Sci., Part A: Polym. Chem., 2008, 46, 6435–6443 CrossRef CAS.
- M. Alisch-Mark, A. Herrmann and W. Zimmermann, Biotechnol. Lett., 2006, 28, 681–685 CrossRef CAS PubMed.
- G. Fischer-Colbrie, S. Heumann, S. Liebminger, E. Almansa, A. Cavaco-Paulo and G. M. Guebitz, Biocatal. Biotransform., 2004, 22, 341–346 CrossRef CAS.
- G. M. Gübitz and A. C. Paulo, Curr. Opin. Biotechnol., 2003, 14, 577–582 CrossRef PubMed.
- R. Müller, H. Schrader, J. Profe, K. Dresler and W. Deckwer, Macromol. Rapid Commun., 2005, 26, 1400–1405 CrossRef.
- F. Kawai, Catalysts, 2021, 11, 206 CrossRef CAS.
- S. Kaabel, J. P. D. Therien, C. E. Deschênes, D. Duncan, T. Friščić and K. Auclair, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2026452118 CrossRef CAS PubMed.
- A. N. Shirke, C. White, J. A. Englaender, A. Zwarycz, G. L. Butterfoss, R. J. Linhardt and R. A. Gross, Biochemistry, 2018, 57, 1190–1200 CrossRef CAS PubMed.
- A. Mrigwani, B. Thakur and P. Guptasarma, Green Chem., 2022, 24, 6707–6719 RSC.
- V. Tournier, C. M. Topham, A. Gilles, B. David, C. Folgoas, E. Moya-Leclair, E. Kamionka, M.-L. Desrousseaux, H. Texier, S. Gavalda, M. Cot, E. Guémard, M. Dalibey, J. Nomme, G. Cioci, S. Barbe, M. Chateau, I. André, S. Duquesne and A. Marty, Nature, 2020, 580, 216–219 CrossRef CAS PubMed.
- F. Quartinello, S. Vajnhandl, J. Volmajer Valh, T. J. Farmer, B. Vončina, A. Lobnik, E. Herrero Acero, A. Pellis and G. M. Guebitz, Microb. Biotechnol., 2017, 10, 1376–1383 CrossRef CAS PubMed.
- X. Li, Y. Hu, C. Du and C. S. K. Lin, Waste Biomass Valorization, 2019, 10, 3763–3772 CrossRef CAS.
- J. Shimizu and T. Kikutani, J. Appl. Polym. Sci., 2002, 83, 539–558 CrossRef CAS.
- Y. Fu, W. R. Busing, Y. Jin, K. A. Affholter and B. Wunderlich, Macromolecules, 1993, 26, 2187–2193 CrossRef CAS.
- S. Kaabel, J. Arciszewski, T. H. Borchers, J. P. D. Therien, T. Friščić and K. Auclair, ChemSusChem, 2023, 16, e202201613 CrossRef CAS PubMed.
- S. L. Fávaro, A. R. Freitas, T. A. Ganzerli, A. G. B. Pereira, A. L. Cardozo, O. Baron, E. C. Muniz, E. M. Girotto and E. Radovanovic, J. Supercrit. Fluids, 2013, 75, 138–143 CrossRef.
- H. Essaddam, US Pat, 10640442B2, 2020 Search PubMed.
- H. Essaddam, US Pat, 10087130B2, 2018 Search PubMed.
- K. Locock, J. Deane, E. Kosior, H. Prabaharan, M. Skidmore and O. Hutt, The Recycled Plastics Market: Global Analysis and Trends, https://publications.csiro.au/publications/publication/PIcsiro:EP19308.
- W. D'Ambrières, F. Actions Sci. Reports, 2019, 19, 12–21 Search PubMed.
- F. S. Yildizhan, Engineering Plastics: Market Analysis and Recycling Methods, SciOpen Prepr., 2021 DOI:10.14293/S2199-1006.1.SOR-.PPOYUPU.v1 , https://www.scienceopen.com/hosted-document.
- A. McNeeley and Y. A. Liu, Ind. Eng. Chem. Res., 2024, 63, 3400–3424 CrossRef CAS.
- H. Dijkstra, P. van Beukering and R. Brouwer, Mar. Pollut. Bull., 2021, 162, 111880 CrossRef CAS PubMed.
- Y.-J. Cai and T.-M. Choi, Transp. Res. Part E: Logist. Trans. Rev., 2020, 141, 102010 CrossRef PubMed.
- A. Halpaap and J. Dittkrist, Curr. Opin. Green Sustainable Chem., 2018, 9, 25–29 CrossRef.
- P. T. Anastas, Chem. Rev., 2007, 107, 2167–2168 CrossRef.
- T.-L. Chen, H. Kim, S.-Y. Pan, P.-C. Tseng, Y.-P. Lin and P.-C. Chiang, Sci. Total Environ., 2020, 716, 136998 CrossRef CAS PubMed.
- B. M. Trost, Science, 1991, 254, 1471–1477 CrossRef CAS PubMed.
- R. A. Sheldon, Chem. Commun., 2008, 3352–3365 RSC.
- R. A. Sheldon, ACS Sustainable Chem. Eng., 2018, 6, 32–48 CrossRef CAS.
- R. A. Sheldon, Green Chem., 2017, 19, 18–43 RSC.
- E. Barnard, J. J. Rubio Arias and W. Thielemans, Green Chem., 2021, 23, 3765–3789 RSC.
- H. W. Horn, G. O. Jones, D. S. Wei, K. Fukushima, J. M. Lecuyer, D. J. Coady, J. L. Hedrick and J. E. Rice, J. Phys. Chem. A, 2012, 116, 12389–12398 CrossRef CAS PubMed.
- C. Jehanno, M. M. Pérez-Madrigal, J. Demarteau, H. Sardon and A. P. Dove, Polym. Chem., 2019, 10, 172–186 RSC.
- S. Lalhmangaihzuala, Z. Laldinpuii, C. Lalmuanpuia and K. Vanlaldinpuia, Polymers, 2020, 13, 37 CrossRef PubMed.
- I. A. Ignatyev, W. Thielemans and B. Vander Beke, ChemSusChem, 2014, 7, 1579–1593 CrossRef CAS PubMed.
- C. P. Papadopoulou and N. K. Kalfoglou, Polymer, 2000, 41, 2543–2555 CrossRef CAS.
- X. Si, L. Guo, Y. Wang and K. Lau, Compos. Sci. Technol., 2008, 68, 2943–2947 CrossRef CAS.
- G. P. Karayannidis and E. A. Psalida, J. Appl. Polym. Sci., 2000, 77, 2206–2211 CrossRef CAS.
- B. Jacques, J. Devaux, R. Legras and E. Nield, Macromolecules, 1996, 29, 3129–3138 CrossRef CAS.
- P. Raffa, M.-B. Coltelli, S. Savi, S. Bianchi and V. Castelvetro, React. Funct. Polym., 2012, 72, 50–60 CrossRef CAS.
- W.-J. Wu, X.-L. Sun, Q. Chen and Q. Qian, Polymers, 2022, 14, 510 CrossRef CAS PubMed.
- W. Yang, Appl. Comput. Eng., 2023, 7, 229–233 CrossRef.
- Z. Jia, L. Gao, L. Qin and J. Yin, RSC Sustain., 2023, 1, 2135–2147 RSC.
- A. Kulkarni, G. Quintens and L. M. Pitet, Macromolecules, 2023, 56, 1747–1758 CrossRef CAS.
- K. Chan and A. Zinchenko, J. Cleaner Prod., 2023, 433, 139828 CrossRef CAS.
- J. Mudondo, H.-S. Lee, Y. Jeong, T. H. Kim, S. Kim, B. H. Sung, S.-H. Park, K. Park, H. G. Cha, Y. J. Yeon and H. T. Kim, J. Microbiol. Biotechnol., 2023, 33, 1–14 CrossRef CAS PubMed.
- L. G. Schaerer, R. Wu, L. I. Putman, J. M. Pearce, T. Lu, D. R. Shonnard, R. G. Ong and S. M. Techtmann, Trends Biotechnol., 2023, 41, 184–196 CrossRef CAS PubMed.
- Y. Ogiwara and K. Nomura, ACS Org. Inorg. Au, 2023, 3, 377–383 CrossRef CAS PubMed.
- J. Diao, Y. Hu, Y. Tian, R. Carr and T. S. Moon, Cell Rep., 2023, 42, 111908 CrossRef CAS PubMed.
- P. Liu, Y. Zheng, Y. Yuan, Y. Han, T. Su and Q. Qi, Waste Manage, 2023, 172, 51–59 CrossRef CAS PubMed.
- T. Tiso, T. Narancic, R. Wei, E. Pollet, N. Beagan, K. Schröder, A. Honak, M. Jiang, S. T. Kenny, N. Wierckx, R. Perrin, L. Avérous, W. Zimmermann, K. O'Connor and L. M. Blank, Metab. Eng., 2021, 66, 167–178 CrossRef CAS PubMed.
- P. Ranganathan, Y.-H. Chen, S.-P. Rwei and Y.-H. Lee, Mater. Today Chem., 2022, 26, 101101 CrossRef CAS.
- Y. Zhang, F. Tian, C. Liu, X. Liu, Y. He and Z. Wu, J. Cleaner Prod., 2024, 434, 140048 CrossRef CAS.
- S. Hussain, M. H. Wadgama, A. L. Khan, M. Yasin and F. H. Akhtar, ACS Sustainable Chem. Eng., 2023, 11, 726–732 CrossRef CAS.
- H. Zhou, Y. Ren, Z. Li, M. Xu, Y. Wang, R. Ge, X. Kong, L. Zheng and H. Duan, Nat. Commun., 2021, 12, 4679 CrossRef CAS PubMed.
- H. Su, T. Li, S. Wang, L. Zhu and Y. Hu, J. Hazard. Mater., 2023, 443, 130120 CrossRef CAS PubMed.
- L. Fabrizio, R. Arrigo, M. T. Scrivani, M. Monti and A. Fina, Polymer, 2023, 266, 125618 CrossRef CAS.
- L. Fan, L. Chen, H. Zhang, W. Xu, X. Wang, S. Xu and Y. Wang, Angew. Chem., Int. Ed., 2023, 62, e202314448 CrossRef CAS PubMed.
- C. Park, H. Lee, N. Lee, B. Ahn and J. Lee, J. Hazard. Mater., 2022, 440, 129825 CrossRef CAS.
- J. G. D. Dutra, M. H. de Souza Santana, Y. Ko, M. J. Lis, F. M. Bezerra, M. P. Moises and J. P. Hinestroza, Mater. Circ. Econ., 2022, 4, 24 CrossRef.
- Y. Ko, T. J. Azbell, P. Milner and J. P. Hinestroza, Ind. Eng. Chem. Res., 2023, 62, 5771–5781 CrossRef CAS.
- A. K. Nason, R. T. Jerozal, P. J. Milner and J. Suntivich, ACS Sustainable Chem. Eng., 2023, 11, 18–22 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc00911h |
| This journal is © The Royal Society of Chemistry 2024 |