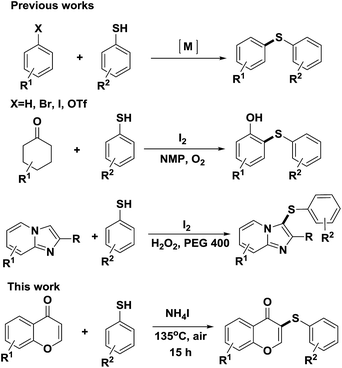
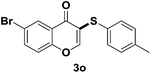
Generation of ArS-substituted flavone derivatives using aryl thiols as sulfenylating agents†
Wannian Zhaoa,
Ping Xieb,
Zhaogang Biana,
Aihua Zhou*a,
Haibo Ge*c,
Ben Niua and
Yingcai Dinga
aPharmacy School, Jiangsu University, Xuefu Road 301, Zhenjiang, Jiangsu 212013, People's Republic of China. E-mail: ahz@ujs.edn.cn
bScientific Information Research Institute, Jiangsu University (Library), Xuefu Road 301, Zhenjiang, Jiangsu 212013, People's Republic of China
cDepartment of Chemistry and Chemical Biology, Indiana University Purdue University Indianapolis, Indianapolis, IN 46202, USA. E-mail: geh@iupui.edu
First published on 2nd July 2015
Abstract
The regioselective and metal-free sulfenylation of flavones was achieved under aerobic conditions with ammonium iodide salt as an inducer instead of traditional iodine/oxidant combinations. This method enables the generation of various ArS-substituted flavone derivatives in good to excellent yields under environmentally friendly conditions, which significantly enriches current flavone chemistry.
The construction of the C–S bond is a very important transformation in organic synthesis,1 because the C–S bond is widely present in natural products and drug molecules.2 Therefore, many researchers have devoted their efforts to developing highly efficient, regioselective, and environmentally friendly C–S bond formation methods.3 Traditionally, the C(sp3)–S bond can be constructed easily by the condensation of an alkyl halide with a metal thiolate.4 Recently, transition metal catalyzed C(sp2)–S bond formations were successfully developed via cross couplings between aryl halides and various sulfenylating reagents such as thiols (Scheme 1),5 sulfonyl chlorides,6 disulfides,7 sodium sulfinates,8 and sulfonyl hydrazides.9 However, these processes require the use of prefunctionalized halides and expensive catalysts.10
Recently, transition metal-catalyzed C–S bond formation via direct C–H bond functionalization has risen as an efficient alternative method, which allows direct conversion of C–H into C–S bonds. Although this method is highly efficient and atom-economic, these reactions still suffer from the high loading of transition metal catalysts, additives and harsh reaction conditions sometimes.11
Very recently, metal-free iodine-induced sulfenylation method to construct C(sp2)–S bond was successfully developed (Scheme 1).12 This method doesn't need the use of any toxic and transitional metal catalysts, and proceeded well under environmentally friendly reaction conditions, generating thioesters in good yields. Despite this significant progress, the development of a new metal-free sulfenylation method is still highly desirable.
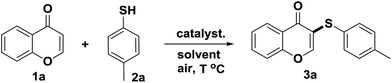
Herein, we developed a new and regioselective sulfenylation method in which a clean and colorless ammonium iodide salt was used as an reaction inducer instead of direct usage of purple iodine. In this study, air was used as the oxidant instead of pure oxygen and some oxidants (Scheme 1);12 and cheap aryl thiols were used as sulfenylating agents. To the best of our knowledge, there are no such reports to date. In this paper, this ammonium iodide-induced sulfenylation method was directly applied to electron-rich flavones to generate flavone derivatives which may be potentially valuable in drug discovery.
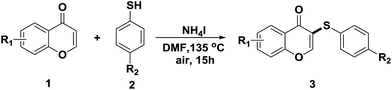
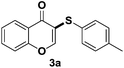
To find the suitable reaction conditions for the ammonium iodide-induced sulfenylation between flavones and aryl thiols, flavone 1a and thiol 2a were used as the representative reactants. Different catalysts/inducers, oxidants, and solvents were screened under different reaction temperature (Table 1). First, CuCl2 and FeCl3 were used as catalysts, and TBHP (tert-butyl hydroperoxide, 70 wt% in water) was employed as an oxidant. Both reactions gave a less than 5% yield or trace amount of expected product 3a (entries 1 and 2) at 80 °C in CH3CN. Using CuI as a catalyst with TBHP also provided a less than 5% yield of 3a (entry 3) in CH3CN. The combination of KI and TBHP only produced a 20% yield of 3a (entry 4). When I2 was employed with TBHP in CH3CN, 45% of 3a was observed (entry 5). The TBAI/TBHP combination in CH3CN didn't afford 3a at all (entry 6), while the combination of NH4I/TBHP in CH3CN afforded a 53% yield of 3a (entry 7). The combination of I2/TBHP or I2/DTBP afforded 3a in 70% and 65% yields, respectively (entries 8 and 9). When the reaction temperature was raised to 135 °C, the combination of NH4I/TBHP in CH3CN gave a 78% yield (entry 10). By converting NH4I into I2, the reaction afforded 75% yield of 3a (entry 11). Using DMF instead of CH3CN afforded 83% yield of 3a (entry 12). Surprisingly, when NH4I was used without TBHP, the reaction also gave an 86% yield of 3a (entry 13). Decreasing the amount of NH4I from 2.0 to 1.2 equivalents led to 64% yield (entry 14). Using CH3CN or THF as a solvent generated 55% yield or trace amount of product (entries 15 and 16). When toluene was used as a solvent, 73% yield of 3a (entry 17) was obtained. No presence of NH4I in DMF didn't produce any product (entry 18). Using H2O2 instead of TBHP as an oxidant in the presence of NH4I afforded 3a in a 65% yield (entry 19). While the combination of I2/H2O2 gave a 70% yield of 3a (entry 20). After the above screening, the suitable conditions selected for the coupling of flavone and aryl thiol are: flavone (1.0 equiv.), thiols (1.2 equiv.), NH4I (2.0 equiv.), DMF is used as a solvent and temperature: 135 °C.
| Entry | Catalyst/inducer | Reagent | Temp. (°C) | Solvent | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: flavone (1 equiv.), aryl thiol (1.2 equiv.), NH4I (2.0 equiv.), aqueous TBHP (70 wt% in water, 2.0 equiv.), metal catalyst (10 mol%), DMF (0.5 mL).b Isolated yields are based on reactant 1a, the reaction was run for 15 hours with air.c NH4I (1.2 equiv.). | |||||
| 1 | CuCl2 | TBHP | 80 | CH3CN | 5 |
| 2 | FeCl3 | TBHP | 80 | CH3CN | Trace |
| 3 | CuI | TBHP | 80 | CH3CN | <5 |
| 4 | KI | TBHP | 80 | CH3CN | 20 |
| 5 | I2 | TBHP | 80 | CH3CN | 45 |
| 6 | TBAI | TBHP | 80 | CH3CN | 0 |
| 7 | NH4I | TBHP | 80 | CH3CN | 53 |
| 8 | I2 | DCP | 80 | CH3CN | 70 |
| 9 | I2 | DTBP | 80 | CH3CN | 65 |
| 10 | NH4I | TBHP | 135 | CH3CN | 78 |
| 11 | I2 | TBHP | 135 | CH3CN | 75 |
| 12 | NH4I | TBHP | 135 | DMF | 83 |
| 13 | NH4I | — | 135 | DMF | 86 |
| 14 | NH4Ic | — | 135 | DMF | 64 |
| 15 | NH4I | — | 135 | CH3CN | 55 |
| 16 | NH4I | — | 135 | THF | Trace |
| 17 | NH4I | — | 135 | Toluene | 73 |
| 18 | — | — | 135 | DMF | 0 |
| 19 | NH4I | H2O2 | 135 | DMF | 65 |
| 20 | I2 | H2O2 | 135 | DMF | 70 |
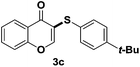
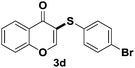
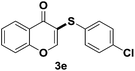
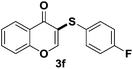
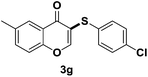
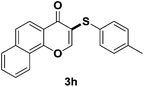
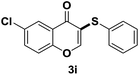
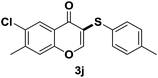
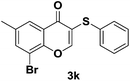
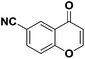
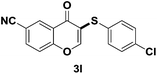
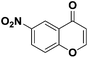
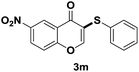
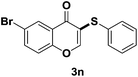
After suitable reaction conditions have been obtained, different aryl thiols were reacted with different flavones which were synthesized based on existing literature.13 From Table 2, it can be found that all flavones with electron-donating functions gave better yields, while flavones with electron-deficient functions gave a little bit lower yields. Based on 1H and 13C-NMR spectra of all products, it was found that –SAr was regioselectively added to the α-position of the ketone function of flavones, and no β-substituted products were isolated.
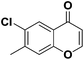
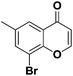
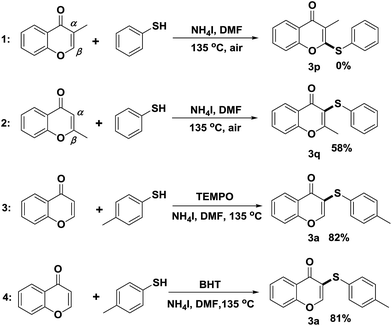
To further explore how the substituents on α and β-position of flavones influence the regioselective sulfenylation, two methyl-substituted flavones were synthesized with methyl function on α and β-position of the flavone (see Scheme 2, entries 1 and 2). When the α-position reaction site was blocked, no any expected product was isolated, indicating that the regioselectivity of this sulfenylation is very good, and the reaction only happened on the α-position of flavone. When the methyl group on the β-position of flavone, a decreased sulfenylation yield was observed, possibly due to the steric effects caused by neighbouring methyl function (Scheme 3).
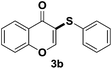
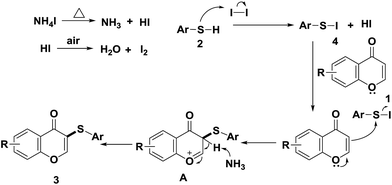
To determine if a radical process is involved in this sulfenylation protocol, TEMPO (2,2,6,6-tetramethypiperidine) and BHT (butylated hydroxytoluene) were used as radical scavengers in the reaction of producing 3a. In the presence of TEMPO or BHT, 3a was still produced in good yields (Scheme 2). This fact indicated that radical intermediate is not involved in the sulfenylation process. Based on the above results and existing literature,12a,14 a plausible nucleophilic substitution reaction mechanism is proposed below. At 135 °C, NH4I was split into NH3 and HI, and the resulting HI was further oxidized by air to generate iodine. Thiophenol was then reacted with iodine to form electrophilic species ArS–I, which reacted further with electron-rich flavone 1 to give reaction intermediate A. After the loss of a proton from intermediate A, final product 3 was obtained.
Conclusion
In summary, an efficient and regioselective ammonium iodide-induced sulfenylation of flavones is herein reported with different thiophenols as sulfenylating agents, generating various ArS-substituted flavone derivatives in good to excellent yields under metal-free conditions. This method has improved the current reported protocol represented by using iodine as a catalyst. This method enriches current flavone chemistry, making it a highly valuable and practical approach in pharmaceutical industry. Investigation on biological activities of flavone derivatives is currently underway. The method is also quite suitable for compound library production.Acknowledgements
Aihua Zhou acknowledge Jiangsu University for financial support (1281290006).Notes and references
- (a) M. Iwasaki, M. Iyanaga, Y. Tsuchiya, Y. Nishimura, W. Li, Z. Li and Y. Nishihara, Chem.–Eur. J., 2014, 20, 2459 CrossRef CAS PubMed; (b) W. Ali, S. Guin, S. K. Rout, A. Gogoi and B. K. Patel, Adv. Synth. Catal., 2014, 356, 3099 CrossRef CAS PubMed; (c) H. Xi, B. Deng, Z. Zong, S. Lu and Z. Li, Org. Lett., 2015, 17, 1180 CrossRef CAS PubMed; (d) C. Shen, P. Zhang, Q. Sun, S. Bai, T. S. A. Hor and X. Liu, Chem. Soc. Rev., 2015, 44, 291 RSC; (e) H. Wang, L. Wang, J. Shang, X. Li, H. Wang, J. Gui and A. Lei, Chem. Commun., 2012, 48, 76 RSC; (f) C.-P. Zhang and D. A. Vicic, J. Am. Chem. Soc., 2012, 134, 183 CrossRef CAS PubMed.
- (a) M. Halim, D. J. Yee and D. Sames, J. Am. Chem. Soc., 2008, 130, 14123 CrossRef CAS PubMed; (b) M. S. C. Pedras, Q.-A. Zheng and S. Strelkov, J. Agric. Food Chem., 2008, 56, 9949 CrossRef CAS PubMed; (c) X. Wang, L. Cui, N. Zhou, W. Zhu, R. Wang, X. Qian and Y. Xu, Chem. Sci., 2013, 4, 2936 RSC; (d) A. J. Wilson, J. K. Kerns, J. F. Callahan and C. J. Moody, J. Med. Chem., 2013, 56, 7463 CrossRef CAS PubMed.
- (a) J.-K. Qiu, W.-J. Hao, D.-C. Wang, P. Wei, J. Sun, B. Jiang and S.-J. Tu, Chem. Commun., 2014, 50, 14782 RSC; (b) L. D. Tran, I. Popov and O. Daugulis, J. Am. Chem. Soc., 2012, 134, 18237 CrossRef CAS PubMed; (c) E. L. Tyson, M. S. Ament and T. P. Yoon, J. Org. Chem., 2012, 78, 2046 CrossRef PubMed; (d) F.-L. Yang, F.-X. Wang, T.-T. Wang, Y.-J. Wang and S.-K. Tian, Chem. Commun., 2014, 50, 2111 RSC; (e) Y. Yang, L. Tang, S. Zhang, X. Guo, Z. Zha and Z. Wang, Green Chem., 2014, 16, 4106 RSC; (f) M. Yoshimatsu, K. Ohta and N. Takahashi, Chem.–Eur. J., 2012, 18, 15602 CrossRef CAS PubMed.
- (a) J. R. Campbell, J. Org. Chem., 1964, 29, 1830 CrossRef CAS; (b) T. Migita, T. Shimizu, Y. Asami, J.-I. Shiobara, Y. Kato and M. Kosugi, Bull. Chem. Soc. Jpn., 1980, 53, 1385 CrossRef CAS; (c) B. C. Ranu and T. Mandal, J. Org. Chem., 2004, 69, 5793 CrossRef CAS PubMed.
- (a) C.-K. Chen, Y.-W. Chen, C.-H. Lin, H.-P. Lin and C.-F. Lee, Chem. Commun., 2010, 46, 282 RSC; (b) A. Correa, O. G. Mancheño and C. Bolm, Chem. Soc. Rev., 2008, 37, 1108 RSC; (c) M. A. Fernández-Rodríguez, Q. Shen and J. F. Hartwig, J. Am. Chem. Soc., 2006, 128, 2180 CrossRef PubMed; (d) P. F. Larsson, A. Correa, M. Carril, P. O. Norrby and C. Bolm, Angew. Chem., 2009, 121, 5801 CrossRef PubMed; (e) M. Sayah and M. G. Organ, Chem.–Eur. J., 2011, 17, 11719 CrossRef CAS PubMed; (f) C. Yu, C. Zhang and X. Shi, Eur. J. Org. Chem., 2012, 10, 1953 CrossRef PubMed.
- (a) M. Chen, Z.-T. Huang and Q.-Y. Zheng, Chem. Commun., 2012, 48, 11686 RSC; (b) O. Saidi, J. Marafie, A. E. Ledger, P. M. Liu, M. F. Mahon, G. Kociok-Köhn, M. K. Whittlesey and C. G. Frost, J. Am. Chem. Soc., 2011, 133, 19298 CrossRef CAS PubMed; (c) D. Zhang, X. Cui, Q. Zhang and Y. Wu, J. Org. Chem., 2015, 80, 1517 CrossRef CAS PubMed.
- (a) W. Ge and Y. Wei, Green Chem., 2012, 14, 2066 RSC; (b) D. Huang, J. Chen, W. Dan, J. Ding, M. Liu and H. Wu, Adv. Synth. Catal., 2012, 354, 2123 CrossRef CAS PubMed; (c) P. Sang, Z. Chen, J. Zou and Y. Zhang, Green Chem., 2013, 15, 2096 RSC; (d) S. Zhang, P. Qian, M. Zhang, M. Hu and J. Cheng, J. Org. Chem., 2010, 75, 6732 CrossRef CAS PubMed.
- (a) M. Yang, H. Shen, Y. Li, C. Shen and P. Zhang, RSC Adv., 2014, 4, 26295 RSC; (b) F. Xiao, H. Chen, H. Xie, S. Chen, L. Yang and G.-J. Deng, Org. Lett., 2014, 16, 50 CrossRef CAS PubMed; (c) Y. Xu, X. Tang, W. Hu, W. Wu and H. Jiang, Green Chem., 2014, 16, 3720 RSC.
- (a) X. Li, X. Xu, P. Hu, X. Xiao and C. Zhou, J. Org. Chem., 2013, 78, 7343 CrossRef CAS PubMed; (b) X. Li, Y. Xu, W. Wu, C. Jiang, C. Qi and H. Jiang, Chem.–Eur. J., 2014, 20, 7911 CrossRef CAS PubMed; (c) M. Zhang, P. Xie, W. Zhao, B. Niu, W. Wu, Z. Bian, C. U. Pittman Jr and A. Zhou, J. Org. Chem., 2015, 80, 4176 CrossRef CAS PubMed; (d) J. Zhang, Y. Shao, H. Wang, Q. Luo, J. Chen, D. Xu and X. Wan, Org. Lett., 2014, 16, 3312 CrossRef CAS PubMed.
- (a) C. Dai, Z. Xu, F. Huang, Z. Yu and Y.-F. Gao, J. Org. Chem., 2012, 77, 4414 CrossRef CAS PubMed; (b) S. Mao, Y.-R. Gao, X.-Q. Zhu, D.-D. Guo and Y.-Q. Wang, Org. Lett., 2015, 17, 1692 CrossRef CAS PubMed; (c) S. Tang, Y. Wu, W. Liao, R. Bai, C. Liu and A. Lei, Chem. Commun., 2014, 50, 4496 RSC; (d) Z. Wu, H. Song, X. Cui, C. Pi, W. Du and Y. Wu, Org. Lett., 2013, 15, 1270 CrossRef CAS PubMed; (e) Y. Xu, P. Liu, S.-L. Li and P. Sun, J. Org. Chem., 2015, 80, 1269 CrossRef CAS PubMed; (f) X. Zhao, E. Dimitrijevic and V. M. Dong, J. Am. Chem. Soc., 2009, 131, 3466 CrossRef CAS PubMed.
- (a) K. Inamoto, Y. Arai, K. Hiroya and T. Doi, Chem. Commun., 2008, 5529 RSC; (b) K. Inamoto, C. Hasegawa, K. Hiroya and T. Doi, Org. Lett., 2008, 10, 5147 CrossRef CAS PubMed; (c) L. L. Joyce and R. A. Batey, Org. Lett., 2009, 11, 2792 CrossRef CAS PubMed; (d) R. Samanta and A. P. Antonchick, Angew. Chem., Int. Ed., 2011, 50, 5217 CrossRef CAS PubMed; (e) C. Shen, H. Xia, H. Yan, X. Chen, S. Ranjit, X. Xie, D. Tan, R. Lee, Y. Yang and B. Xing, Chem. Sci., 2012, 3, 2388 RSC; (f) R. Xu, J.-P. Wan, H. Mao and Y. Pan, J. Am. Chem. Soc., 2010, 132, 15531 CrossRef CAS PubMed; (g) Y. Yang, W. Hou, L. Qin, J. Du, H. Feng, B. Zhou and Y. Li, Chem.–Eur. J., 2014, 20, 416 CrossRef CAS PubMed; (h) J. Zhu, Z. Chen, H. Xie, S. Li and Y. Wu, Org. Lett., 2010, 12, 2434 CrossRef CAS PubMed.
- (a) M.-A. Hiebel and S. Berteina-Raboin, Green Chem., 2015, 17, 937 RSC; (b) Y. Liao, P. Jiang, S. Chen, H. Qi and G.-J. Deng, Green Chem., 2013, 15, 3302 RSC.
- (a) D. Ameen and T. J. Snape, Synthesis, 2015, 47, 141 CAS; (b) S.-H. Wang, Y. Wang, Y.-Y. Zhu, S.-J. Liu, J. Han, Y.-F. Zhou, D.-W. Li, D. Koirala and C. Hu, Chem. Res. Chin. Univ., 2011, 27, 54 CAS; (c) Y. Zheng, M. Zhao, Q. Qiao, H. Liu, H. Lang and Z. Xu, Dyes Pigm., 2013, 98, 367 CrossRef CAS PubMed.
- F. L. Yang and S. K. Tian, Angew. Chem., Int. Ed., 2013, 52, 4929 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra10763f |
| This journal is © The Royal Society of Chemistry 2015 |