Brandon R. Groves, T. Stanley Cameron and Alison Thompson
Org. Biomol. Chem., 2017,15, 7925-7935
DOI:
10.1039/C7OB01278K,
Paper
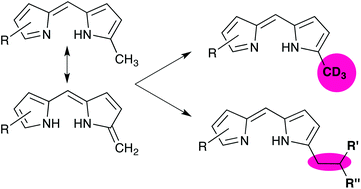
Regioselective reactivity of the 1-methyl group of free-base dipyrrins is explored, including discussion of tautomerism to provide exocyclic alkenyl reactivity. Deuterium is installed so as to generate dipyrrins substituted with deuterated methyl groups. Furthermore, the 1-methyl group reacts to become involved in C–C bonds involving only sp3-hybridised carbon atoms. The isolation of an elusive framework featuring a dipyrrin substituted with a pyrrole in a non-vinylogous fashion is also reported. The use of asymmetric dipyrrins featuring an electron-withdrawing group on one of the pyrrolic units results in regioselective reaction of the alpha-methyl group distal to the electron-withdrawing group.