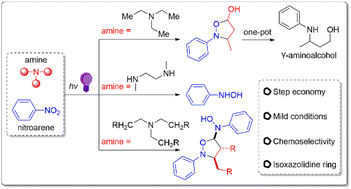
Reduction of nitroarenes constitutes an important route to high value-added products. It is highly desirable but remains a challenge that this process occurs with selectivity under mild conditions. In this work, we developed a photochemical strategy for the mild and selective reduction of nitroarenes using amines as the reducing agent. Furthermore, the photo-induced redox reaction between nitroarenes and amines triggers a cascade process, forming isoxazolidine derivatives with high efficiency. This important skeleton was previously synthesized via the use of unstable aldehydes and hydroxylamine, which not only limits the introduction of functional groups but also complicates practical operation. This work provides an efficient way for the construction of isoxazolidine derivatives, and opens up new opportunities for the application of the photoactivation of nitroarenes and amines.