Improved tetracycline degradation via integrated peroxymonosulfate activation and photocatalytic self-Fenton using hybrid Type-II/Z-scheme Se/g-C3N4/Bi2WO6 carboxymethyl cellulose hydrogels
Emmanuel Sebastian
Kunnel
 a,
Sanmitra Ajay
Manker
a,
Ashish
Kumar
b,
Akash
Balakrishnan
a,
Sanmitra Ajay
Manker
a,
Ashish
Kumar
b,
Akash
Balakrishnan
 c,
Chandra Shekhar
Pati Tripathi
c,
Chandra Shekhar
Pati Tripathi
 b,
Brajesh Kumar
Dubey
*d and
Suverna
Trivedi
*a
b,
Brajesh Kumar
Dubey
*d and
Suverna
Trivedi
*a
aDepartment of Chemical Engineering, Indian Institute of Technology Kharagpur (IITKGP), West Bengal-721302, India. E-mail: trivedi@che.iitkgp.ac.in; strivedi@che.iitkgp.ac.in
bDepartment of Physics, Institute of Science, Banaras Hindu University, Varanasi, Uttar pradesh-221005, India
cDepartment of Chemical Engineering, Saintgits College of Engineering(Autonomous), 686532, Kerala
dDepartment of Civil Engineering, Indian Institute of Technology Kharagpur (IITKGP), West Bengal-721302, India. E-mail: bkdubey@civil.iitkgp.ac.in
First published on 7th October 2025
Abstract
Developing efficient integrated advanced oxidation processes (AOPs) is vital for sustainable treatment of antibiotic-contaminated water. In this work, a novel 3D photocatalyst was engineered by embedding a ternary Se/g-C3N4/Bi2WO6 (SGB) heterojunction into a carboxymethyl cellulose hydrogel, yielding a stable and reusable SGB hydrogel system. Structural, optical, electrochemical, and photoelectrochemical analyses confirmed a hybrid Type-II/Z-scheme heterojunction, reducing the bandgap to 1.74 eV and enhancing charge separation. The synergistic effects of the ternary interface and hydrogel matrix enabled efficient in situ H2O2 generation (716 μM in water; 958 μM with isopropanol), facilitating a self-Fenton-like reaction. Upon coupling with peroxymonosulfate (PMS) activation, the system achieved 93.86% tetracycline degradation within 30 minutes. Radical scavenging and trapping experiments revealed a multi-radical degradation pathway involving ˙OH, SO4˙−, O2˙−, and 1O2, with their roles modulated by pH. At higher pH, PMS activation via O2˙−/e− favored SO4˙− and 1O2 generation, while lower pH conditions promoted H2O2/˙OH production and hole oxidation. LC-MS analysis confirmed the stepwise degradation of tetracycline into low-mass intermediates, supporting the proposed mechanism. Toxicity analysis further demonstrated that the transformation products exhibited reduced ecological risk, confirming the environmental safety of the process. The SGB hydrogels exhibited excellent stability and reusability, retaining 72.14% degradation efficiency after 12 cycles and retaining performance across a broad pH range. This study introduces a novel photocatalytic platform integrating Type-II/Z-scheme charge transfer, photoelectrochemical performance, multiple AOP pathways, and progressive detoxification within a hydrogel matrix for sustainable pharmaceutical pollutant remediation.
1. Introduction
Freshwater is indispensable. Yet demand from water-intensive industries such as textiles, petrochemicals, pharmaceuticals, and food processing continues to climb, accelerating both consumption and pollution.1,2 Decades of overexploitation and mismanagement have eroded supplies to the point where nearly one-fifth of global withdrawals now exceed sustainable limits, according to the United Nations.3 At the same time, population growth and expanding healthcare have driven an unprecedented rise in pharmaceutical and antibiotic use.4,5 Since conventional wastewater treatment plants struggle to remove these compounds, they persist as “emerging contaminants,” fostering antibiotic-resistant bacteria (ARB) and resistance genes (ARGs).6 Tetracycline (TC), a widely used, low-cost antibiotic in human medicine, livestock, and aquaculture exemplifies the problem. It is environmentally persistent, frequently detected in surface and groundwater, and implicated in escalating antimicrobial resistance.7 Conventional remediation methods such as adsorption, membrane filtration, and biological degradation are often costly, inefficient, or too selective to effectively remove diverse classes of antibiotics. Advanced oxidation processes (AOPs), in contrast, can mineralize organic pollutants into CO2 and H2O through the generation of highly reactive oxygen species (ROS).8 However, standalone AOPs often suffer from slow reaction kinetics, interference from complex water matrices, and risks of secondary pollution, such as the generation of iron sludge.9 Therefore, there is an urgent need for robust, integrated AOP technologies capable of efficiently degrading antibiotics like TC across a range of water conditions.10In response, photocatalytic self-Fenton (PSF) technologies have emerged as a versatile solution by coupling heterogeneous photocatalysis with Fenton-type ˙OH production, without the need for externally added H2O2 or Fe2+. This simplifies operation and minimizes sludge formation.11 The core of PSF lies in the photocatalytic generation of H2O2, which is an environmentally benign oxidant with increasing global demand, directly from O2 and H2O under ambient conditions and visible-light irradiation.12,13 This approach avoids the harsh reaction conditions, high costs, and environmental impacts associated with the anthraquinone method.14,15 Despite these advantages, PSF efficiency decreases in alkaline media, which limits its standalone applicability.16,17 Sulfate-radical AOPs activated by peroxymonosulfate (PMS) offer complementary advantages, including higher redox potential, faster reaction kinetics, and longer radical lifetimes over a wider pH range. Additionally, they enhance charge-carrier separation by scavenging photogenerated electrons. The integration of PSF and PMS in a hybrid, photocatalyst-assisted system (PSF-PMS) holds significant promise. This approach not only enables the generation of diverse ROS such as ˙OH and SO4˙− under varying pH conditions but also maintains operational simplicity and sustainability. Although some studies have explored this strategy,18,19 the development of advanced photocatalysts that combine strong visible-light absorption, efficient charge separation, and effective H2O2 generation alongside PMS activation remains largely unexplored for large-scale water treatment applications.
Photocatalysis employs light-activated materials to generate charge carriers for energy conversion and environmental remediation. Conventional photocatalysts such as titanium dioxide (TiO2) and zinc oxide (ZnO) mainly absorb ultraviolet light, accounting for less than 5% of the solar spectrum, and suffer from rapid recombination of photogenerated charge carriers. Although metal sulfides offer visible-light activity, their toxicity limits practical use. These limitations have motivated the development of visible-light-responsive photocatalysts with low toxicity and enhanced charge separation. Bismuth tungstate (Bi2WO6, BWO) partially meets these criteria due to its non-toxic nature, thermal stability, and visible-light absorption. However, pure BWO experiences fast recombination of photogenerated electrons and holes and only moderate light harvesting, requiring coupling with other semiconductors to improve performance.16 Heterojunction construction enables spatial separation of electrons and holes by directing them to different materials, reducing recombination. Type-II heterojunctions promote efficient charge transfer but weaken redox potential, while Z-scheme heterojunctions retain strong redox power but generate fewer charge carriers. Combining both architectures can balance charge transfer efficiency and redox strength.20,21 Graphitic carbon nitride (g-C3N4, GCN) was selected as the first partner due to its 2.7 eV bandgap, visible-light response, and conduction band potential suitable for two-electron oxygen reduction to H2O2, a green oxidant. Defects in g-C3N4 further improve electron transport.7 Selenium was chosen as the second partner because of its narrow bandgap (∼1.7–1.9 eV), long carrier lifetime, high mobility, broad visible-light absorption, and low toxicity.22 A composite of these three materials is expected to form a ternary heterojunction that integrates Type-II and Z-scheme charge transfer pathways. In this system, BWO provides strong visible-light absorption, GCN drives H2O2 production, and selenium enhances conductivity and light absorption. This design maximizes solar energy utilization, preserves high redox ability, and offers a stable, recyclable platform for AOPs. To our knowledge, such a combination has not been explored for use in PMS-PSF.
In this study, ternary Se/g-C3N4/Bi2WO6 (SGB) hydrogels were synthesized using a simple ionic cross-linking method to enhance photocatalytic performance. Various characterization techniques such as FE-SEM, XRD, FTIR, XPS, DRS, and transient photocurrent and electrochemical analysis confirmed the successful formation of a hybrid Type-II/Z-scheme heterojunction that promotes efficient and rapid charge separation. The integration of photocatalysis with PMS activation enabled high catalytic activity across a wide pH range, effectively mitigating the typical activity loss under alkaline conditions. The optimization of catalyst loading, PMS concentration, and pollutant (TC) concentration identified conditions for near-complete degradation of TC. Mechanistic investigations through radical-quenching tests, electron spin resonance (ESR) spectroscopy, and LC-MS analysis of degradation intermediates revealed a ROS cascade involving ˙OH, SO4˙−, and O2˙−, driven by the dual charge-transfer pathways of the heterojunction. The SGB hydrogel exhibited excellent stability and recyclability over 12 consecutive cycles, demonstrating robust performance, easy recovery, and minimal secondary pollution. Overall, this eco-friendly SGB hydrogel presents a promising and scalable platform for sustainable water treatment by effectively combining photocatalysis with PMS activation for antibiotic degradation.
2. Materials and methods
2.1. Preparation of SGB hydrogel photocatalysts
GCN was prepared by the thermal polymerization method. This was followed by the preparation of g-C3N4/Bi2WO6 using the hydrothermal route. The detailed procedure is provided in the SI file (Fig. S1). The SGB hydrogel beads were synthesised using a simple wet-impregnation method and blend cross linking strategy. Briefly, equal amounts of GCN/BWO and selenium metal powder were dispersed in 50 mL of distilled water and stirred for 5 h followed by drying overnight at 60 °C. Subsequently, 0.4 g of SGB powder was stirred thoroughly with 4% aqueous carboxymethyl cellulose (CMC) solution (w/v) until homogeneity. This mixture was transferred to 1 M FeCl3 solution dropwise to produce hydrogel beads via phase inversion. Following 12 hours of cross-linking, the synthesized hydrogel beads were readily separated and washed with distilled water until neutral pH was achieved. The resulting SGB hydrogel beads were then stored for subsequent use.2.2. Photocatalyst characterization
Various analytical techniques were employed to thoroughly characterize the SGB hydrogels. The morphology and elemental mapping of synthesized composites and hydrogel beads were studied using scanning electron microscopy (SEM) (Zeiss EVO 60). The crystallinity and purity of the prepared photocatalysts and SGB hydrogels were examined using X-ray diffraction (XRD) (Bruker D2 Phaser). The chemical structure and functional groups of the photocatalyst were investigated using Fourier Transform Infrared Spectroscopy (FTIR) (PerkinElmer Spectrum 100). X-ray photoelectron spectroscopy (XPS) (ULVAC-PHI) was used to investigate the surface chemical composition and to determine the oxidation states of the elements in the SGB hydrogel. The optical characteristics and band gap of the developed photocatalysts were analysed using a UV-visible spectrophotometer (Shimadzu Japan). The photoelectrochemical measurements were carried out using chronoamperometry. The amperometry measurements were carried out by applying a dc voltage of magnitude 0.15 V in 0.1 M Na2SO4 aqueous solution by using an on-off light source. Inductively Coupled Plasma Mass Spectrometry (ICP-MS) (Thermo Scientific iCAP RQ) was used to quantify the amount of Fe leached by the SGB hydrogel photocatalyst.The electrochemical measurements of the developed photocatalysts were conducted using Palm Sense 4 electrochemical workstations. All measurements employed a standard three-electrode setup, where a glassy carbon electrode (GCE) modified with catalysts served as the working electrode, a platinum wire served as the counter electrode, and Ag/AgCl (3.0 M KCl) served as the reference electrode. 0.1 M Na2SO4 aqueous solution was used as the electrolyte. The working electrode was prepared by drop-casting 10 μL of photocatalyst ink (1 mg mL−1 in deionized water with 5% Nafion solution) onto the polished GCE surface. Electrochemical impedance spectra were recorded using a 10 mV sinusoidal perturbation across a frequency range of 0.1–100,000 Hz. Mott–Schottky measurements were conducted within a specific potential window, applying a 10 mV sinusoidal potential at 1000 Hz. The intermediates of TC were measured using liquid chromatography/mass spectrometry (LC/MS) (Waters 2695 Separation Module, USA).
3. Results and discussion
3.1. Characterization of the photocatalysts
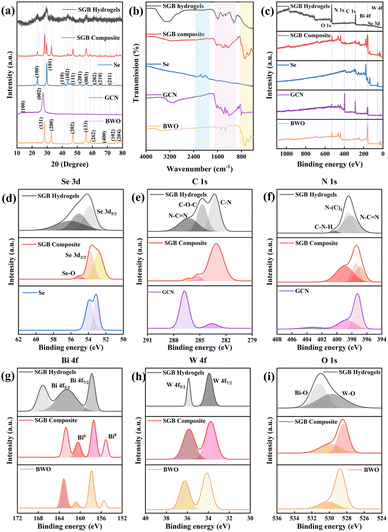
The digital image of the SGB hydrogels, along with an illustrative representation of their preparation, is provided in the SI file (Fig. S1). The surface morphologies and elemental composition of the synthesized photocatalysts were examined by FE-SEM and EDX analysis (Fig. 1). The SEM images of individual BWO and GCN are provided in the SI file (Fig. S2). The SEM micrograph of the SGB heterojunction (Fig. 1(a)) reveals nearly spherical Se particles uniformly deposited on well-defined, flake-like g-C3N4/Bi2WO6 nanosheets (Fig. 1b and c), indicating successful composite formation. In the hydrogel beads (Fig. 1d–f), the SGB heterojunction is encapsulated within raindrop-shaped beads, whose wrinkled surfaces result from water removal during drying. The EDX spectra and elemental composition of GCN, BWO and SGB composites are shown in Fig. S3–S5. As shown in Fig. 1(g–n), EDX elemental mapping of the SGB hydrogels reveals Fe alongside Se, C, N, Bi, W, and O. The uniform distribution of Fe observed in the EDX spectra reflects strong cross-linking between FeCl3 and the SGB-CMC biopolymer network. Thus, SEM and EDX results together confirm that SGB and CMC are seamlessly integrated throughout the hydrogel matrix.The XRD patterns (Fig. 2(a)) confirm the successful synthesis of BWO, as evidenced by characteristic diffraction peaks. Prominent reflections appear at 2θ values of 28.3°, 32.86°, 47.13°, 55.89°, 58.59°, 68.69°, 75.99°, and 78.42°, which correspond to the (131), (200), (202), (133), (262), (400), (102), and (204) crystallographic planes of the orthorhombic BWO phase (JCPDS card no. 39-0256).23 GCN displayed two distinct diffraction peaks at 12.88° and 27.33°, which are attributed to the periodicity of the tri-s-triazine units and the interlayer stacking of the carbon nitride layers, respectively.24 The peaks at 23.56°, 29.73°, 41.29°, 43.69°, 45.45°, 51.72°, 55.86°, 61.42°, 65.28° and 71.49° in the diffraction pattern of Se corresponded to the (100), (101), (110), (102), (111), (201), (003), (202), (210) and (211) reflections of the pure hexagonal phase of selenium crystals.25–27 Important peaks displayed in the diffraction pattern of the SGB composite at 23.50°, 28.36°, 29.70°, 32.83°, 45.21°, 75.99°, and 78.42° with a minor peak shift reveal that individual catalyst components of GCN, BWO and Se are successfully incorporated into the SGB composite. The transformation of the SGB composite into SGB hydrogel beads using CMC resulted in reduced peak intensities and peak broadening, indicating the amorphous nature of the SGB hydrogel beads.
The chemical structure and functional groups of photocatalysts were investigated using FTIR spectra within the 400–4000 cm−1 range (Fig. 2(b)). For BWO, the band situated at 3435 cm−1 is attributed to O–H stretching of the residual hydroxyl group on its surface. The peaks at 1625 and 1384 cm−1 belong to the vibration of absorbed water molecules and N–O vibration of NO3+ from the synthesis procedure. The peaks at 725 cm−1 and 581 cm−1 are attributed to Bi–O, W–O, and W–O–W vibrational stretching in Bi2WO6.28–30 For GCN, several peaks were obtained in the 1200 cm−1 to 1650 cm−1 range which are attributed to CN heterocycles. The broad peak in the 3000 to 3300 cm−1 range is ascribed to NH and NH2 groups. The breathing mode of triazine units gives rise to a peak at 806 cm−1.31
In the case of the SGB hydrogel, the vibrational bands at ∼3500 and 1250 cm−1 have become more intense and broad which could be assigned to the addition of the –OH group from the CMC matrix to the SGB composite. The bands situated at 2800 cm−1 are present in both pristine Se and the SGB hydrogel. The lack of extra peaks indicates that SGB composites and CMC do not form any covalent connections. Rather, a network structure is created by cross-linking Se, GCN, and BWO with CMC, improving the composite hydrogel's mechanical and stability characteristics. In order to incorporate SGB composites into hydrogels, the hydroxyl group of CMC is bonded to the SGB composite by hydrogen bonding.32
X-ray photoelectron spectroscopy (XPS) was employed to analyse the surface chemical composition and oxidation states of elements in the SGB CMC hydrogel. Fig. 2(c) depicts the survey spectra of BWO, GCN, Se, SGB composite and SGB hydrogels. The presence of various elements, such as bismuth (172–152 eV), tungsten (40–30 eV), oxygen (536–524 eV), carbon (291–279 eV), nitrogen (408–394 eV) and selenium (62–50 eV), in the SGB hydrogel suggests the successful formation of the ternary heterojunction. The survey spectra of the SGB hydrogel clearly indicate the presence of C and O spectra with maximum intensities suggesting the surface availability of BWO was reduced with the effective formation of SGB hydrogels due to the loading of CMC and its ubiquitous structural availability of carbon and oxygen. It can also be noted that the predominantly higher presence of carbon in CMC further strengthened the C 1 s spectra of the SGB hydrogel.
The deconvoluted spectra of all the above mentioned elements are shown in Fig. 2(d–i). As seen in Fig. 2(d), the peaks for Se 3d at 53 eV (53.89, 52.82, and 53.06 eV for beads, SGB, and Se powder, respectively) and 55 eV (55.08, 53.75, and 53.89 eV) are attributed to Se 3d5/2 and Se 3d3/2, respectively.33,34 The heterojunction between materials caused an additional Se–O peak at 56.07 eV and 55.03 eV in beads and the SGB composite, respectively.34Fig. 2(e) presents the C 1s XPS spectra. The peak near 283 eV (observed at 283.26, 283.11, and 283.57 eV for the SGB hydrogel, SGB composite, and GCN, respectively) is assigned to graphitic or adventitious carbon and the sp2-hybridized C–N bonds. The peak around 286 eV (286.27, 286.36, and 286.76 eV for the SGB hydrogel, SGB composite, and GCN, respectively) corresponds to the sp2-hybridized carbon in N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and the triazine rings within the aromatic structure of GCN.35,36 Additionally, the C–O–C bond characteristic of carboxymethyl cellulose (CMC) appears at 285.27 eV in the SGB composite and at 284.71 eV in the hydrogel beads.37
N and the triazine rings within the aromatic structure of GCN.35,36 Additionally, the C–O–C bond characteristic of carboxymethyl cellulose (CMC) appears at 285.27 eV in the SGB composite and at 284.71 eV in the hydrogel beads.37
Considering the N 1s spectra, the first peaks (398.17, 396.91, and 397.21 eV for the SGB hydrogel, SGB composite, GCN respectively) in Fig. 2(f) correspond to the sp2 bonded nitrogen in the form of C–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C.38–40 The second peak (398.52, 397.48, and 398.65 eV for the SGB hydrogel, SGB composite, and GCN, respectively) is attributed to the tertiary nitrogen (N–(C)3) groups and the third peak (400, 398.91, and 403.27 eV for the SGB hydrogel, SGB composite, and GCN, respectively) belongs to the surface uncondensed amino groups (C–N–H).41,42 From the XPS spectra of Bi 4f (Fig. 2(g)), the two peaks at approximately 157 eV (157.76, 157.42, and 157.83 eV for the SGB hydrogel, SGB composite, and BWO, respectively) and 163 eV (162.57, 162.72, and 163 eV for the SGB hydrogel, SGB composite, and BWO, respectively) can be ascribed to the binding energies of Bi 4f7/2 and Bi 4f5/2, respectively.43,44 Besides, two less intense peaks at 155 and 160 eV correspond to small quantities of metallic bismuth scattered on the BWO and SGB composite surface.40,45,46 The two deconvoluted peaks (Fig. 2(h)) for W 4f at approx. 34 eV (33.91, 33.78, and 34.13 eV for the SGB hydrogel, SGB composite, and BWO, respectively) and 36 eV (35.87, 35.84, and 36.24 eV for the SGB hydrogel, SGB composite, and BWO, respectively) represent the W 4f7/2 and W 4f5/2 spin–orbit doublets of W6+.18,47,48 The deconvoluted O1s spectra (Fig. 2(i)) revealed peaks at approx. 528 eV and 530 eV, attributed to W–O and Bi–O bonds, respectively, in the SGB composite and BWO.49 However, the intense O 1s peak of the hydrogel is attributed to the higher oxygen content belonging to the CMC. Compared to GCN and BWO components, a minor peak shift is observed in the SGB hydrogel and SGB composite, which reflects electronic interactions and possible charge transfer.50
C.38–40 The second peak (398.52, 397.48, and 398.65 eV for the SGB hydrogel, SGB composite, and GCN, respectively) is attributed to the tertiary nitrogen (N–(C)3) groups and the third peak (400, 398.91, and 403.27 eV for the SGB hydrogel, SGB composite, and GCN, respectively) belongs to the surface uncondensed amino groups (C–N–H).41,42 From the XPS spectra of Bi 4f (Fig. 2(g)), the two peaks at approximately 157 eV (157.76, 157.42, and 157.83 eV for the SGB hydrogel, SGB composite, and BWO, respectively) and 163 eV (162.57, 162.72, and 163 eV for the SGB hydrogel, SGB composite, and BWO, respectively) can be ascribed to the binding energies of Bi 4f7/2 and Bi 4f5/2, respectively.43,44 Besides, two less intense peaks at 155 and 160 eV correspond to small quantities of metallic bismuth scattered on the BWO and SGB composite surface.40,45,46 The two deconvoluted peaks (Fig. 2(h)) for W 4f at approx. 34 eV (33.91, 33.78, and 34.13 eV for the SGB hydrogel, SGB composite, and BWO, respectively) and 36 eV (35.87, 35.84, and 36.24 eV for the SGB hydrogel, SGB composite, and BWO, respectively) represent the W 4f7/2 and W 4f5/2 spin–orbit doublets of W6+.18,47,48 The deconvoluted O1s spectra (Fig. 2(i)) revealed peaks at approx. 528 eV and 530 eV, attributed to W–O and Bi–O bonds, respectively, in the SGB composite and BWO.49 However, the intense O 1s peak of the hydrogel is attributed to the higher oxygen content belonging to the CMC. Compared to GCN and BWO components, a minor peak shift is observed in the SGB hydrogel and SGB composite, which reflects electronic interactions and possible charge transfer.50
The optical properties of the synthesized photocatalysts were studied using UV-visible diffuse reflectance spectroscopy (DRS). The absorption spectra obtained from DRS measurements are shown in Fig. 3(a). The spectra clearly indicate that the formation of the SGB composite significantly enhances light absorption in the visible region compared to pristine GCN and BWO. This improvement is attributed to the presence of selenium, which absorbs visible light more effectively than GCN or BWO, as well as the modified electronic structure resulting from heterojunction formation among the components of the SGB composite. Furthermore, the light absorption capacity increases even more after synthesizing the SGB hydrogel. This enhancement is mainly due to selenium's superior visible light absorption and the incorporation of CMC, which forms a three-dimensional hydrogel network.
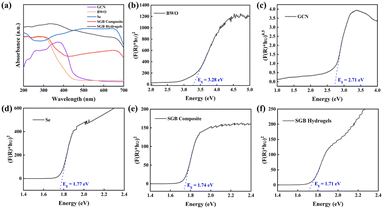 | ||
| Fig. 3 (a) UV-vis absorption spectra and (b–f) the corresponding Tauc plots for the prepared photocatalysts. | ||
The bandgaps of the materials were determined through Tauc plot analysis with the results presented in Fig. 3(b–f). According to the Tauc plots, the bandgap values follow the order of SGB hydrogels (1.71 eV) < SGB composite (1.73 eV) < Se (1.76 eV) < GCN (2.71 eV) < BWO (3.28 eV) (Table S1). The measured band gaps for GCN, BWO, and Se are consistent with values reported in earlier studies.20,51,52 The smaller bandgap of the SGB composite compared to its individual components is likely due to the interaction and overlap of the energy bands of GCN, BWO, and Se, which create new energy levels and enhance visible-light photocatalytic activity.53 Moreover, the interaction through hydrogen bonds between the hydroxyl groups of carboxymethyl cellulose (CMC) and the amino groups of graphitic carbon nitride (g-C3N4) within the SGB hydrogel plays a significant role in tuning the material's optical characteristics.32,54 This study demonstrates that both Se and CMC play key roles in tuning the bandgap and enhancing the performance of the hybrid photocatalyst.
Electrochemical analysis provides insight into the generation, separation, and transport of electrons and holes across the surface of a photocatalyst, which are the factors that directly impact its catalytic efficiency. Accordingly, electrochemical impedance spectroscopy (EIS) was employed to investigate the interfacial charge-transfer resistance and surface recombination processes. The resulting EIS spectra are presented as Nyquist plots and shown in Fig. 4(a). In such plots, a smaller semicircle indicates lower interfacial charge-transfer resistance and enhanced electron mobility. The measured arc radii followed the order: BWO > GCN > SGB composite > Se > SGB hydrogel, confirming that the incorporation of selenium and embedding the material in a CMC hydrogel significantly reduce interfacial charge-transfer resistance. Complementary insights were obtained from Bode analysis (Fig. 4(b)), where the SGB hydrogel exhibited a shift toward higher peak frequency, indicating improved charge carrier mobility and suppressed recombination. Additionally, the dynamics of interfacial charge transfer and separation were investigated using the transient photocurrent response. A strong photocurrent is generally regarded as evidence of efficient electron–hole pair separation, thereby promoting photocatalytic activity. The current–time response in the dark and under light irradiation is shown in Fig. S6. The photocurrent increases rapidly as soon as the light is switched on, and it instantly decreases once the light is switched off. The photocurrent responses show a stable current cycle. The SGB hydrogel shows a much higher photocurrent response compared to the SGB composite and individual components, suggesting improved charge transfer dynamics and hence photocatalytic efficiency as well.22,55,56
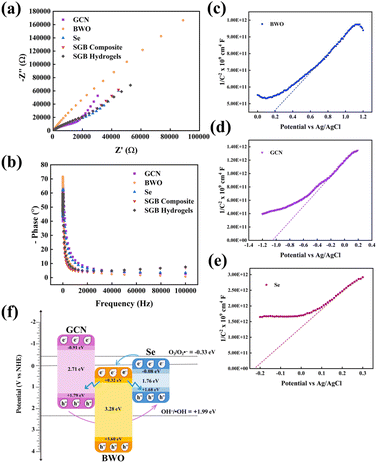 | ||
| Fig. 4 (a) EIS spectra and (b) Bode plot of the prepared photocatalysts along with Mott–Schottky plots of (c) BWO; (d) GCN; and (e) Se; and (f) proposed band structure of the SGB hydrogel. | ||
Two key factors drive this behaviour. First, hydrogen bonding between the SGB composite and CMC forms conductive pathways that facilitate electron transfer. Second, the incorporation of selenium enhances the intrinsic conductivity of the material, further promoting charge transport. Together, these effects lower interfacial resistance, enhance carrier mobility, and suppress electron–hole recombination, thereby endowing the SGB hydrogel with potentially superior photocatalytic performance. To further investigate the electronic band structure of the SGB hydrogel, Mott–Schottky (M–S) analysis was performed on Se, BWO, and GCN to determine their flat-band potentials (Fig. 4(c–e)). For an n-type semiconductor, the flat-band potential measured via Mott–Schottky (M–S) analysis is typically close to the conduction band edge. The positive slopes observed in all M–S plots confirmed the n-type semiconductor nature of Se, BWO, and GCN. From the intercepts on the potential axis (vs. Ag/AgCl reference electrode), the flat-band (conduction band) potentials were estimated as −0.22 eV for Se, +0.18 eV for BWO, and −1.05 eV for GCN. These values were then converted to the normal hydrogen electrode (NHE) scale using eqn (S1), resulting in conduction band potentials of −0.08 eV (Se), +0.32 eV (BWO), and −0.91 eV (GCN). Based on the band gap values obtained from earlier analysis, the corresponding valence band potentials were calculated as +1.68 eV (Se), +3.60 eV (BWO), and +1.79 eV (GCN), all referenced to the NHE.
After estimating the band positions of GCN, BWO, and Se, it becomes clear that the conduction band potentials of BWO (0.32 eV) and Se (−0.08 eV) are more positive than the redox potential of O2/O2˙− (−0.33 eV vs. NHE), which means their electrons are not energetic enough to reduce oxygen molecules (O2) effectively and generate superoxide radicals (O2˙−). Similarly, the valence band potentials of GCN and Se are slightly less positive than the redox potential required for generating hydroxyl radicals (OH˙) from water (H2O/OH˙ +1.99 eV vs. NHE), making it difficult for holes in these materials to drive water oxidation. As a result, the typical Type-II charge transfer mechanism is not efficient for producing (ROS) in this system. On the other hand, electrons collected in the conduction band of GCN can readily reduce O2 to form O2˙−, while holes in the valence band of BWO can oxidize H2O and generate ˙OH, supporting more effective photocatalytic reactions. The inclusion of Se further boosts the number of available charge carriers due to its high conductivity, promoting better charge flow throughout the system. Therefore, adopting a hybrid Type-II/Z-scheme charge transfer pathway is advantageous and proposed (Fig. 4(f)), as it balances efficient charge movement with strong redox activity. In this design, a ternary heterojunction forms among Se, GCN, and BWO within the SGB hydrogel composite. Here, GCN and BWO provide robust charge carriers, while Se ensures smooth charge transport. The hydrogel's interconnected and porous structure not only optimises the availability of these active sites but also improves the diffusion of reactants, leading to enhanced overall catalytic performance.
3.2. Photocatalytic H2O2 production
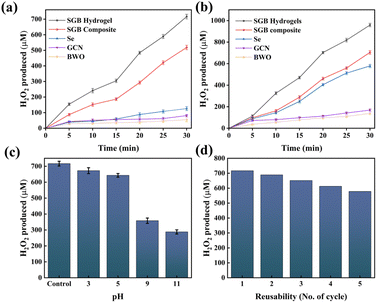
A crucial first step in establishing the PSF-PMS system is confirming the in situ generation of H2O2, which is essential for the subsequent degradation studies to proceed via a self-Fenton mechanism. Dark reaction and photolysis experiments confirmed that no H2O2 was produced in the absence of light or catalyst before the photocatalytic experiments (Fig. 5). Using the SGB hydrogel beads, the photocatalytic generation of H2O2 was carried out for 30 minutes under LED lamp illumination. The corresponding results are shown in Fig. 5(a). The H2O2 production follows the order: SGB hydrogel beads (716 μM) > SGB composite (518 μM) > Se (126 μM) > GCN (81 μM) > BWO (54 μM). The significant increase in H2O2 production by the SGB composite is likely due to its hybrid Type-II/Z-scheme configuration, which ensures effective charge carrier separation and a plentiful supply of charge carriers. The further enhancement of H2O2 production in the SGB hydrogel can be attributed to improved electron transfer facilitated by hydrogen bonding between CMC and the SGB composite, which promotes the increased generation of superoxide (O2˙−). However, with isopropanol used as the sacrificial agent, a notable increase in H2O2 generation was observed in the order: SGB hydrogel beads (958.86 μM) > SGB composite (704 μM) > Se (578 μM) > GCN (168 μM) > BWO (138 μM) (Fig. 5(b)). This enhancement is attributed to isopropanol's ability to inhibit H2O2 decomposition while donating additional electrons. As shown in Fig. 5(c), H2O2 generation by the SGB hydrogels increases with decreasing pH, due to the higher proton availability facilitating the conversion of O2˙− to H2O2. Evaluating photostability and reusability is critical for catalyst viability. As illustrated in Fig. 5(d), the SGB hydrogels retained activity over five consecutive cycles, with H2O2 yield gradually decreasing to 578.2 μM by the fifth run.3.3. Performance evaluation on TC degradation
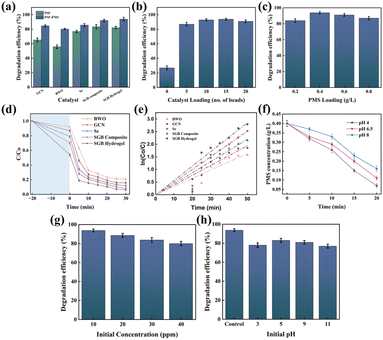
Photocatalytic removal of TC was evaluated under both PSF and PSF–PMS conditions, as illustrated in Fig. 6(a). In every case, incorporating PMS into the PSF system markedly enhanced TC mineralization, highlighting the superior efficacy of the integrated PSF–PMS advanced oxidation process over stand-alone PSF. Focusing on the PSF–PMS configuration, Fig. 6(b) plots TC concentration versus time (20 min dark, 30 min visible light) for each photocatalyst at a PMS loading of 0.4 g L−1. Under these conditions, degradation efficiencies ranked as follows: hydrogel beads (93.86%) > SGB heterojunction (92.12%) > Se (85.57%) > GCN (84.61%) > BWO (80.18%). The SGB-loaded beads achieved 93.86% TC removal within 30 min of illumination—superior to all single-component catalysts. Without PMS, efficiencies declined across the same series: beads (82.22%) > SGB (83.45%) > Se (77.02%) > g-C3N4 (65.21%) > Bi2WO6 (55.84%), confirming that PMS is critical for accelerating TC degradation under LED visible light. The SGB composites showed boosted performance by generating more photogenerated electrons and holes thanks to the formation of the hybrid Type-II/Z-scheme heterojunction. The hydrogel's enhanced activity is largely attributed to the carboxymethyl cellulose (CMC) matrix and cyclic Fe, which promote charge separation and inhibit electron–hole recombination. By contrast, PMS alone produced negligible degradation under identical conditions, as it could not be effectively activated to generate persulfate radicals for TC breakdown. Fig. 6(c) indicates that TC degradation follows a pseudo-first-order kinetics, with the corresponding rate constants and R2 values listed in Table S2. The enhanced catalytic performance arises from rapid charge-carrier transfer, superior charge-separation efficiency, and abundant active sites created by the Type-II/Z-scheme heterojunction among BWO, GCN, and Se. Additionally, the carboxymethyl cellulose (CMC) matrix (i) effectively suppresses electron–hole recombination by facilitating carrier transport, (ii) broadens visible-light absorption and fine-tunes the composite's bandgap, and (iii) slightly boosts the pollutant adsorption during the dark phase of the PSF–PMS process. Table S3 shows a comparison of the TC degradation efficiency of the SGB hydrogels with other reported photocatalysts, demonstrating its better performance.The effect of varying the dosage of SGB hydrogel beads on the efficiency of PSF–PMS-driven TC degradation is presented in Fig. 6(d). Increasing the bead quantity from five to ten improved the degradation efficiency from 86.63% to 93.86% within 30 minutes under visible light irradiation (PMS = 0.4 g L−1). This enhancement is attributed to the increased number of active sites, which promotes the generation of reactive radical species and facilitates more effective utilization of visible light in the PSF–PMS system.
The influence of PMS dosage on TC degradation in the PSF-PMS system was studied over a range of 0.2 to 0.8 g L−1, with the results shown in Fig. 6(e). Efficient activation of PMS is necessary to generate sufficient sulfate radicals and other reactive oxygen species for effective TC oxidation. Increasing the PMS dosage from 0.2 to 0.4 g L−1 improved the TC degradation efficiency from 84.71% to 93.86%. However, the TC degradation efficiency dropped from 93.86% to 87.43% when the PMS dosage was increased to 0.8 g L−1. The SGB beads' active sites severely restrict the PMS's activation capacity, hence a greater quantity of PMS can fully utilize the active sites. The catalytic active sites might not satisfy PMS requirements at larger dosages. When too much PMS is present in the solution, it reacts with HSO5−, takes part in the oxidation reaction, and reduces the efficiency of TC breakdown. TC breakdown may be adversely affected by the excess PMS's ability to scavenge hydroxyl and sulphate radicals.57 The potassium iodide-based spectrophotometric technique was used to determine the remaining PMS concentration in the deteriorated TC solution. According to Fig. 6(f), the optimal PMS dosage of 0.4 g L−1 was not used during the first photodegradation period (t = 0). After 30 minutes of using the photo-assisted PSF-PMS system, the PMS concentration decreased to 0.1 g L−1 as the duration increased. The efficient activation and use of PMS for TC breakdown were confirmed by this work. Fig. 6(g) examines the effect of the initial TC concentration (10–40 mg L−1). As the starting concentration increased, the degradation efficiency declined from 93.86% to 80.57%. At higher pollutant loads, intermediate byproducts and an excess of substrate molecules likely compete for active sites on the SGB beads, reducing overall breakdown rates.
As shown in Fig. 6(h), the impact of initial pH is a crucial factor that needs to be considered when creating an effective PSF-PMS system. It was discovered that the TC stock solution had a natural pH of 6.1. 0.1 M HCl and NaOH were added to the TC to change its original pH. From 3 to 11, the TC degradation efficiency increased, whereas in the extreme basic area, it decreased. The reduced performance of SGB beads under acidic conditions within the PSF-PMS system is attributed to the acidity of PMS. At low pH, the degradation efficiency of TC decreases due to increased electrostatic repulsion between the positively charged catalyst surface and TC molecules, as well as the suppressed activation of HSO5− ions caused by the high concentration of H+ ions. Conversely, at alkaline pH (∼11), less reactive SO52− ions replace HSO5− and experience repulsion from the negatively charged catalyst surface, further lowering catalytic activity. These combined effects inhibit TC degradation under both strongly acidic and basic conditions in the PSF-PMS system.
3.4. Environmental implications
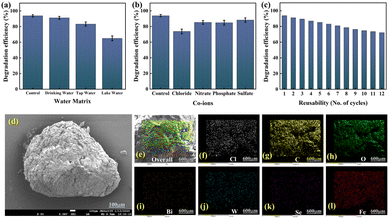
Assessing the practical applications of hydrogels is crucial for advancing energy-efficient and sustainable wastewater treatment methods. Interfering ions present in water play a significant role in influencing the photocatalytic degradation of TC. Fig. 7(a) illustrates TC removal in the presence of various co-ions, including chloride, nitrate, phosphate, and sulphate, in a reaction medium. Compared to the control, a notable decrease in degradation efficiency of the SGB hydrogel was observed, primarily due to the inhibitory effect of chloride ions (73.72%), which is attributed to the formation of Cl˙, Cl2˙, and OCl˙ radicals that quench ˙OH and sulphate radicals. Nitrate ions (85.28%) reduce the efficiency by quenching reactive oxygen species (ROS). Similarly, phosphate ions (84.93%) impair photocatalytic degradation due to their interaction with ROS and persulfate radicals. Sulphate ions (88.31%) also hinder the process by scavenging hydroxyl radicals. These results confirm that co-ions interfere with TC degradation during photocatalysis.The complex composition of real water can affect the photocatalytic degradation of TC. To study this, different water samples such as domestic tap water, drinking water, and lake water were used, each containing 10 ppm of TC. The degradation experiments were conducted for 30 minutes under LED light. As shown in Fig. 7(b), the degradation efficiencies were 90.26% in drinking water, 83.34% in tap water, and 65.12% in lake water. The lower degradation in tap and lake water is due to the presence of heavy metals, co-ions like chloride and nitrate, and other minerals that interfere with the process. Additionally, organic matter in lake water competes for ROS, further reducing TC degradation.
To determine whether beads can be commercialized, their photostability and reusability must be evaluated. At the end of each experiment, SGB hydrogel beads were recovered by filtration and immediately reused in the subsequent TC degradation test. According to Fig. 7(c), the SGB hydrogel beads showed a recyclability up to 12 cycles. After 12 consecutive cycles, the degradation efficiency of the SGB hydrogel beads in the visible light-driven PSF-PMS system declined from 93.86% to 72.14%. This moderate reduction in catalytic activity is attributed to the surface adsorption of TC and its intermediate products within the pores and on the surface of the hydrogel beads, which also induced slight structural alterations. The SEM image of the recycled SGB beads after the 12th cycle is presented in Fig. 7(d), while the corresponding EDS elemental mappings are shown in Fig. 7(e–l), revealing minor morphological changes consistent with wear and tear. ICP-MS analysis confirmed minimal Fe leaching from the hydrogel matrix, with concentrations decreasing from 20 ppb after the first cycle to 17 ppb after the 12th cycle, indicating excellent structural integrity and negligible iron leaching during repeated use. Furthermore, XRD analysis (Fig. S6) confirmed that the crystal structure of the SGB beads remained intact after repeated use. So, it can be said that the SGB beads are appropriate for the ongoing reclamation of persistent organic pollutants due to their increased stability, reusability, and recoverability. In addition to this, a comprehensive comparison has also been carried out with recent photocatalysts in the existing literature based on energy consumption and cost (Table S3). Compared to reported catalysts, SGB hydrogels, having moderate costs material wise, show significant performance with minimal energy consumption.
3.5. Mechanistic insights
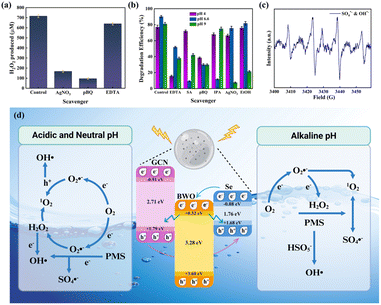
To understand the degradation mechanism of the SGB hydrogel, various scavengers were used to selectively capture reactive species: p-benzoquinone (p-BQ) for superoxide radicals (O2˙−), sodium azide (SA) for singlet oxygen (1O2), EDTA-Na for photogenerated holes (h+), isopropyl alcohol (IPA) and ethanol (EtOH) for hydroxyl (˙OH) and sulfate radicals (SO4˙−), and silver nitrate (AgNO3) for photogenerated electrons (e−). Initial quenching tests with p-BQ, EDTA-Na, and AgNO3 were performed to study the role of these species in the in situ production of H2O2 during peroxysulfate (PSF) activation by the SGB hydrogel at natural pH (Fig. 8(a)). H2O2 is central to the PSF mechanism, acting as the critical link between photogenerated electrons and Fenton-like reactions that generate subsequent oxidants. Under neutral conditions, the PSF system produces approximately 716 μM of H2O2 without scavengers. Introducing p-BQ to trap superoxide radicals reduces H2O2 yield drastically, confirming superoxide's role as the essential intermediate in converting dissolved oxygen to peroxide. Similarly, AgNO3, a scavenger of photogenerated electrons, diminishes H2O2 production as well, validating the electron-driven pathway.However, singlet oxygen (1O2) dominates TC degradation under near-neutral conditions and is formed by two parallel routes (Fig. 8(b)). First, O2·− formed by the one-electron reduction of dissolved O2 disproportionate in water to produce H2O2 and 1O2 as by-products. Second, PMS can lose an electron to form SO5˙−, which then generates singlet oxygen (1O2) either through self-decomposition or by reacting with water. Additionally, PMS can self-decompose to SO52−, which reacts with H2O to produce superoxide (O2˙−), and the rearrangement of O2˙− similarly yields 1O2 alongside sulfate anions. These two routes together strengthen high 1O2 generation, as confirmed by sodium azide quenching. Hydroxyl radicals constitute the second most abundant species. They originate from two sources. First via the catalytic activation of in situ H2O2 and second through the direct oxidation of water by photogenerated holes. The combined ˙OH pool works synergistically with 1O2 to oxidize electron-rich sites in TC. Superoxide radicals play an intermediary yet crucial role. They form both via direct single-electron transfer to O2 and through the reaction of PMS anion radicals (SO52−) with water, generating O2˙−, SO4˙−, and H+. Subsequent superoxide conversion produces H2O2 and feeds back into 1O2 formation. Thus, O2·− serves as a bridge, sustaining both the 1O2 and ˙OH pathways. Together, these intertwined routes establish a robust, multi-pathway oxidation network that underpins the exceptional TC degradation at pH 6.6.
Under strongly acidic conditions, photogenerated holes drive most of the oxidation, directly attacking TC and producing hydroxyl radicals by oxidizing water (Fig. 8(b)). Leftover electrons reduce dissolved oxygen into superoxide, which both oxidizes contaminants and converts to H2O2 and singlet oxygen. This superoxide-mediated cascade also feeds back into hydroxyl radical pools via H2O2 activation. Meanwhile, photogenerated electrons partially reduce silver ions, confirming their availability but minor oxidative contribution. Hydroxyl radicals arise not only from holes but also from catalytic H2O2 and PMS activation. A small fraction of PMS molecules self-decay to singlet oxygen, supplementing oxidative power. Sulfate radicals form through one-electron PMS reduction but play the least role under acidic pH. Together, holes initiate oxidation and ˙OH formation; electrons generate superoxide that sustains both 1O2 and ˙OH pathways; and minor SO4˙− adds additional radical flux—synergistically delivering efficient TC degradation in acidic media.
Under alkaline conditions, photogenerated electrons drive most of the degradation (Fig. 8(b)). These electrons rapidly reduce PMS into sulfate radicals, which directly attack TC and convert water into hydroxyl radicals. Meanwhile, excess electrons reduce dissolved O2 into superoxide, which both contributes to degradation and regenerates downstream oxidants. Photogenerated electrons accumulate at the catalyst surface and activate PMS, producing a high flux of sulfate radicals. Their oxidative power is the strongest here, in contrast to pH 4 (where holes dominate) and pH 6.6 (where singlet oxygen leads). Sulfate radicals then oxidize TC itself and convert water molecules into hydroxyl radicals, supplementing the oxidative network. At pH 9.0, their role surpasses that of hydroxyls, unlike at pH 6.6 where ˙OH ranks second, and at pH 4 where ˙OH is mid-level. Superoxide serves as an intermediary: generated by the electron reduction of oxygen, and it helps sustain both sulfate and hydroxyl radical formation and, to a lesser extent, singlet-oxygen pathways. This contrasts with pH 6.6, where superoxide chiefly feeds singlet-oxygen production, and with pH 4, where it follows hole processes.
Electron paramagnetic resonance (EPR) spin-trapping confirmed multiple radicals in the PSF–PMS hybrid system. With DMPO as a spin trap, Fig. S7 shows a quartet at ∼3430 G, indicating O2˙− formation under light. Fig. 8(c) reveals DMPO-adduct signals of SO4˙− (and its decay products) plus a smaller ˙OH quartet, demonstrating concurrent sulfate and hydroxyl radical generation. These EPR results, matching scavenger data, verify that O2˙− (from photocatalytic O2-reduction), SO4˙− (from PMS activation), and ˙OH (from H2O2 activation and sulfate pathways) co-operate in a multi-radical oxidation mechanism.
Building on the band alignment and Mott–Schottky results (Fig. 4(c–e)) and measured bandgaps, we propose a hybrid Type-II/Z-scheme mechanism in the SGB hydrogel (Fig. 8(d)).20,21 Under visible-light irradiation, each semiconductor absorbs photons to generate electron–hole pairs. At the Se/g-C3N4 interface, the Se conduction band (CB) lies above g-C3N4 CB, driving Se-CB electrons into g-C3N4's CB via a Type-II junction, while g-C3N4 VB holes migrate into Se's VB. Likewise, at the Se/Bi2WO6 interface, Se-CB electrons transfer into Bi2WO6's higher CB, and Bi2WO6 VB holes funnel into Se's VB. These two Type-II steps spatially separate carriers, lowering recombination rates and guiding electrons and holes toward distinct regions. Concurrently, a Z-scheme recombination occurs between electrons in Bi2WO6's CB and holes in g-C3N4's VB.55 This selective interfacial recombination retains the most energetic carriers, which are electrons in g-C3N4's CB and holes in Bi2WO6's VB, thereby preserving strong redox potentials. Crucially, this hybrid configuration maximizes the availability of free photogenerated charge carriers by preventing wasteful bulk recombination while maintaining carrier populations at optimal energy levels. Electrons accumulating in g-C3N4's CB not only reduce O2 to superoxide radicals (O2˙−) but also directly participate in TC degradation through electron transfer reactions. Similarly, holes concentrated in Bi2WO6's VB both oxidize H2O to hydroxyl radicals (˙OH) and directly attack TC molecules via hole-mediated oxidation. Selenium's role as a conductive mediator is pivotal. Its narrow bandgap and high intrinsic conductivity create electron highways that accelerate charge transport across interfaces, dramatically increasing the population of free carriers available for both ROS generation and direct pollutant attack.52,58 Furthermore, Se extends visible-light absorption, boosting overall carrier generation while its high mobility ensures rapid delivery of electrons into reactive sites, amplifying both indirect ROS pathways and direct charge-transfer degradation mechanisms. Photoelectrochemical validation confirms this enhanced charge availability as the SGB hydrogel exhibits the highest transient photocurrent and lowest charge-transfer resistance, indicating maximum free carrier density and mobility. EPR spin-trapping detects abundant O2˙−, SO4˙−, and –OH formation, while scavenger studies reveal that both ROS-mediated pathways and direct carrier reactions contribute significantly to TC degradation.
This design retains strong redox potentials by ensuring that the most reactive carriers participate in reduction and oxidation reactions. The hybrid charge migration mechanism is substantiated by parallels with recent literature, such as the switching from Type-II to Z-scheme in via interfacial band bending, and ternary systems where semiconductors (like Se) boost conductivity while serving as a relay for charge transport.20,21,59,60 Selenium's semiconductor properties and high mobility enable rapid electron flow, reduce recombination, and act as a conductive bridge between the two main photocatalysts.58,61 Under light irradiation, EPR further detects robust ROS production (O2˙−, SO4˙−, and ˙OH), confirming the proposed charge migration and oxidative species pathways. The system also engages a self-Fenton-like loop, in which O2˙− intermediates produce H2O2in situ, which is readily photo-activated by electrons, yielding additional ˙OH. In particular, in the presence of peroxymonosulfate (PMS), both PMS and generated H2O2 mutually activate to diversify the ROS profile ensuring the efficient production of SO4˙−, O2˙−, 1O2, and ˙OH across a wide pH window. These species, alongside photogenerated holes and electrons that are abundant thanks to the Type-II/Z-scheme configuration, drive effective TC decomposition into benign products. Overall, the Se/g-C3N4/Bi2WO6 photocatalyst leverages the hybrid Type-II/Z-scheme arrangement to maximize charge separation and availability while preserving redox strength, as directly evidenced by both electrochemical, transient photocurrent and spectroscopic data along with literature and scavenger studies. This mechanism underpins the observed enhancement in photocatalytic performance, sustained ROS generation, and pollutant mineralization across diverse reaction conditions.
3.6. Tetracycline degradation pathway and toxicity analysis
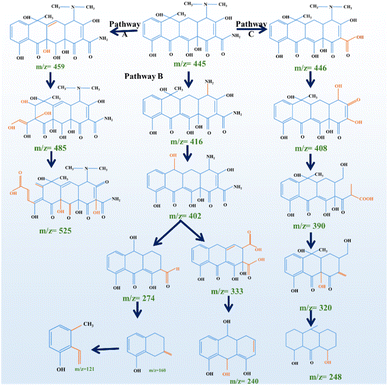
To elucidate the transformation mechanism of TC under LED-activated PMS oxidation using Se/g-C3N4/Bi2WO6@CMC hydrogels, high-resolution mass spectrometry (HRMS) was employed to identify the intermediate species formed during the photocatalytic degradation process. The analysis revealed three major degradation pathways, denoted as Pathways A, B, and C, each initiated by distinct radical-mediated reactions such as hydroxylation, dealkylation, deamination, and oxidative ring cleavage (Fig. 9).18In Pathway A, the degradation of TC begins with the hydroxylation of the parent compound (m/z = 455), forming A1 (m/z = 459). This is followed by extensive oxidative functionalization, resulting in the formation of A2 (m/z = 485), which possesses multiple hydroxyls and possibly carbonyl groups. The transformation proceeds to A3 (m/z = 525), indicative of a highly oxidized intermediate with significant structural fragmentation and incorporation of oxygen atoms. This pathway suggests that sulfate and hydroxyl radicals attack electron-rich sites, facilitating deep oxidation and breakdown into highly polar, possibly biodegradable species.62
Pathway B initiates with N-demethylation and the deamination of the dimethylamino group, converting TC (m/z = 445) to B1 (m/z = 416). This is followed by further oxidation to B2 (m/z = 402), a pivotal intermediate that branches into two sub-pathways: (i) One route leads to B3a (m/z = 274) through ring cleavage and oxidative degradation, (ii) the other produces B3b (m/z = 333) via partial oxidation of the aromatic framework. These intermediates are further transformed into smaller products such as B4 (m/z = 160) and B5 (m/z = 121), which are likely phenolic and carboxylic acid derivatives, indicating extensive ring opening and mineralization. This pathway demonstrates that both partial oxidation and complete ring destruction co-occur depending on radical availability and the molecular structure.7,18
In Pathway C, the initial transformation of TC (m/z = 445) to C1 (m/z = 446) occurs via radical-induced hydroxylation. Progressive oxidation leads to C2 (m/z = 408) and C3 (m/z = 390), potentially involving dealkylation and incorporation of carboxylic groups. The pathway culminates in C4 (m/z = 248), indicating partial ring cleavage and transformation into low molecular weight carboxylated species.36,62 Compared to Pathway A and B, Pathway C appears to preserve parts of the ring structure longer but still facilitates conversion into biodegradable compounds.
Furthermore, the Ecological Structure Activity Relationships (ECOSAR) Predictive Model was utilized to assess the acute and chronic toxicity potential of degradation products. The ECOSAR assessment (Fig. S10) classified TC as a high acute toxicity pollutant (LC50/EC50 < 10 mg L−1), corroborating its ecological risk. Classification was performed according to the Globally Harmonized System of Classification and Labelling of Chemicals (GHS).63 The oxidative ring-opening followed by functionalization that reduces hydrophobicity and bioaccumulation seems to be an effective detoxification mechanism, as most of the degradation products (P2, P5, P6, P7, P10, and P12–P15) were non-toxic in the model (LC50/EC50 > 100 mg L−1). Moderate toxicity (1–100 mg L−1) was retained by a few intermediates (P1, P4, P9, and P11) and algae, which can be explained by the presence of aromatic or pharmacophoric residues. Chronic toxicity predictions similarly highlighted transient risks for P8 and P9; however, their rapid transformation prevents persistence and cumulative effects. Above all, the method effectively breaks down TC and significantly reduces the ecotoxicological impact of its by-products, indicating both effectiveness and ecological protection as key advantages.
4. Conclusions
This study demonstrates a highly effective strategy for tetracycline degradation through the integration of PMS activation with reusable Se/g-C3N4/Bi2WO6 (SGB) photocatalytic hydrogel beads. The ternary heterojunction was synthesized via thermal polycondensation, hydrothermal growth, and wet impregnation, and subsequently immobilized within a CMC matrix. Under visible LED irradiation, the hydrogel beads achieved 93.86% removal of a 10 ppm TC solution within just 30 minutes using 400 mg L−1 PMS, while retaining high catalytic activity over 12 consecutive cycles. The Type II/Z-scheme band alignment in the SGB heterojunction significantly enhanced charge-carrier separation, suppressed electron–hole recombination, and facilitated the generation of reactive oxygen species, including O2˙−, SO4˙−, and ˙OH radicals. The synergistic effect of photocatalysis and PMS activation enabled rapid pollutant degradation, efficient visible-light utilization, easy catalyst recovery, and excellent long-term stability.Based on the degradation intermediates detected by LC-MS, three possible degradation pathways were proposed, involving sequential hydroxylation, N-demethylation, deamination, and ring cleavage. The identified intermediates (e.g., m/z = 248, 160, 121) indicate efficient molecular fragmentation and mineralization. Moreover, the photodegradation process exhibited a stepwise reduction in molecular complexity, suggesting progressive detoxification, as supported by the structural transformation of the intermediates. These findings provide a practical blueprint for the design of sustainable and reusable photocatalysts for advanced antibiotic removal in wastewater treatment.
Author contributions
Emmanuel Sebastian Kunnel: writing – original draft, formal analysis, data curation, conceptualization. Sanmitra Ajay Manker: writing – original draft, software, formal analysis. Akash Balakrishnan: validation, software. Ashish Kumar: formal analysis, validation. Chandra Shekhar Pati Tripathi: formal analysis, validation writing – review & editing. Brajesh Kumar Dubey: supervision, project administration. Suverna Trivedi: supervision, resources, project administration, investigation, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing interests.Data availability
The data that support the findings of this study are available from the corresponding author, S. Trivedi, upon reasonable request.Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta05936d.
Acknowledgements
ESK gratefully acknowledges the Ministry of Education for the PhD fellowship. ST thanks the Sponsored Research & Industrial Consultancy (SRIC) Cell of IIT Kharagpur for providing the FSRG CEC funding. CSPT acknowledges the IoE Cell at Banaras Hindu University for the seed grant award under the IoE Development Scheme (Scheme No. 6031). The authors also acknowledge central research facilities IITKGP and Department of chemical engineering for the use of Instrumentation facilities.References
-
E. S. Kunnel, S. A. Mankera, A. Kumar, A. Balakrishnan, C. S. P. Tripathi, B. K. Dubey, S. Trivedi, in Emerging Contaminants and Associated Treatment Technologies, Springer International Publishing, Cham, 2022, pp. 231–261 Search PubMed
.
- J. Lei, H. Yang, B. Weng, Y. Zheng, S. Chen, P. W. Menezes and S. Meng, Adv. Energy Mater., 2025, 15, 2500950 CrossRef CAS
.
- A. Bhatt, P. Arora and S. K. Prajapati, J. Environ. Chem. Eng., 2020, 8, 104429 CrossRef CAS PubMed
.
- S. P. Shanmugavel and G. Kumar, Environ. Pollut., 2024, 341, 122842 CrossRef PubMed
.
- Y. Zhang, J. Li, S. Jiao, Y. Li, Y. Zhou, X. Zhang, B. Maryam and X. Liu, Sci. Total Environ., 2024, 929, 172734 CrossRef CAS PubMed
.
- G. Gopal, S. A. Alex, N. Chandrasekaran and A. Mukherjee, RSC Adv., 2020, 10, 27081 RSC
.
- A. Balakrishnan, M. Chinthala, R. K. Polagani and D.-V. N. Vo, Environ. Res., 2023, 216, 114660 CrossRef CAS PubMed
.
- B. Weng, M. Zhang, Y. Lin, J. Yang, J. Lv, N. Han, J. Xie, H. Jia, B.-L. Su and M. Roeffaers, Nat. Rev. Clean Technol., 2025, 1–15 Search PubMed
.
- F. Yan, L. An, X. Xu, W. Du and R. Dai, Sci. Total Environ., 2024, 906, 167737 CrossRef CAS PubMed
.
- A Kumar, Langmuir, 2025, 41(23), 14765–14777 CrossRef CAS PubMed
.
- A. Balakrishnan, E. S. Kunnel, N. Dayanandan, H. Tripathy, M. Chinthala, A. Kumar and D.-V. N. Vo, J. Environ. Chem. Eng., 2025, 13, 117171 CrossRef CAS
.
- A. Hayat, Z. Ajmal, A. Y. A. Alzahrani, S. B. Moussa, M. Khered, N. Almuqati, A. Alshammari, Y. Al-Hadeethi, H. Ali and Y. Orooji, Coord. Chem. Rev., 2025, 522, 216218 CrossRef CAS
.
- X. Han, W. Chen, M. Xu, T. Bai and B. Li, Sep. Purif. Technol., 2024, 345, 127397 CrossRef CAS
.
- A. Balakrishnan, M. Chinthala, A. Kumar, N. Dayanandan and S. Trivedi, Int. J. Hydrogen Energy, 2025, 98, 1020–1033 CrossRef CAS
.
- L. Wang, J. Zhang, Y. Zhang, H. Yu, Y. Qu and J. Yu, Small, 2022, 18, 2104561 CrossRef CAS
.
- J. Ma, N. Ding and H. Liu, Sep. Purif. Technol., 2023, 324, 124628 CrossRef CAS
.
- P. P. Singh, G. Pandey, Y. Murti, J. Gairola, S. Mahajan, H. Kandhari, S. Tivari and V. Srivastava, RSC Adv., 2024, 14, 20492–20515 RSC
.
- A. Balakrishnan, M. Chinthala, A. Kumar, D. Barceló and S. Rtimi, Chem. Eng. J., 2025, 503, 158378 CrossRef CAS
.
- B. An, J. Liu, B. Zhu, F. Liu, G. Jiang, X. Duan, Y. Wang and J. Sun, Chem. Eng. J., 2023, 478, 147344 CrossRef CAS
.
- W. Shi, C. Liu, M. Li, X. Lin, F. Guo and J. Shi, J. Hazard. Mater., 2020, 389, 121907 CrossRef CAS
.
- E. M. Hashem, M. A. Hamza, A. N. El-Shazly, S. A. Abd El-Rahman, E. M. El-Tanany, R. T. Mohamed and N. K. Allam, Chemosphere, 2021, 277, 128730 CrossRef CAS
.
- K. Zhong, H. Gao, J. Feng, Y. Zhang and K. Lai, J. Mater. Sci., 2019, 54, 10632–10643 CrossRef CAS
.
- Y. Chen, Y. Zhang, C. Liu, A. Lu and W. Zhang, Int. J. Photoenergy, 2012, 2012, 1–6 Search PubMed
.
- K. V. Suryaa, A. Balakrishnan, M. Chinthala, K. B. Devi, H. Tripathy, A. Kumar, T. M. Aminabhavi and S. Rtimi, Chem. Eng. J., 2025, 505, 159470 CrossRef
.
- N. Srivastava and M. Mukhopadhyay, Bioprocess Biosyst. Eng., 2015, 38, 1723–1730 CrossRef CAS
.
- L. Y. Cruz, D. Wang and J. Liu, J. Photochem. Photobiol., B, 2019, 191, 123–127 CrossRef CAS PubMed
.
- V. Cittrarasu, D. Kaliannan, K. Dharman, V. Maluventhen, M. Easwaran, W. C. Liu, B. Balasubramanian and M. Arumugam, Sci. Rep., 2021, 11, 1032 CrossRef CAS
.
- H. Dong, Y. Yin and X. Guo, Environ. Sci. Pollut. Res., 2018, 25, 11754–11766 CrossRef CAS PubMed
.
- R. Wang, Z. Jiang, L. Xu and C. Liu, J. Mater. Sci.: Mater. Electron., 2021, 32, 6931–6941 CrossRef CAS
.
- D. Trixy Nimmy Priscilla, R. Radha, A. Chitra and S. K. Geetha, J. Cluster Sci., 2024, 35, 875–889 CrossRef CAS
.
- L. Meng, C. Zhao, H. Chu, Y.-H. Li, H. Fu, P. Wang, C.-C. Wang and H. Huang, Chin. J. Catal., 2024, 59, 346–359 CrossRef CAS
.
- T. Shan, J. Li, S. Wu, H. Wu, F. Zhang, G. Liao, H. Xiao, L. Huang and L. Chen, Chem. Eng. J., 2023, 478, 147509 CrossRef CAS
.
- G. Lu, H. Yang, J. Zhang, B. Lin and J. Xu, Sep. Purif. Technol., 2025, 355, 129642 CrossRef CAS
.
- H. Liu, R. Suo, W. Li, L. Luo, H. Yang, J. Chen and C.-Z. Lu, Sep. Purif. Technol., 2024, 346, 127439 CrossRef CAS
.
- Z. U. Rehman, M. Bilal, F. K. Butt, S. U. Rehman, Z. Asghar, K. Zheng, Y. Zhang, X. Xu, J. Hou and X. Wang, Sep. Purif. Technol., 2024, 350, 128001 CrossRef CAS
.
- A. Balakrishnan, K. V. Suryaa, M. Chinthala and A. Kumar, J. Colloid Interface Sci., 2024, 669, 366–382 CrossRef CAS
.
- Y. Cai, D. Li, S. Peng and H. Liu, Colloid Polym. Sci., 2025, 303, 287–300 CrossRef CAS
.
- S. Shen, W. Zhong, Z. Wang, Z. Lin and S. Feng, R. Soc. Open Sci., 2019, 6, 181886 CrossRef CAS PubMed
.
- S. Kang, X. Liu, Z. Wang, Y. Wu, M. Dou, H. Yang, H. Zhu, D. Li and J. Dou, Environ. Res., 2023, 232, 116345 CrossRef CAS PubMed
.
- A. Kumar, V. Arya, A. Pathak, S. Trivedi, D. Guin and C. S. P. Tripathi, Langmuir, 2025, 41, 14765–14777 CrossRef CAS PubMed
.
- F. Qiao, J. Wang, S. Ai and L. Li, Sens. Actuators, B, 2015, 216, 418–427 CrossRef CAS
.
- Y. Duan and L. Liu, J. Chem. Technol. Biotechnol., 2023, 98, 247–256 CrossRef CAS
.
- J. R. Shallenberger, C. M. Smyth, R. Addou and R. M. Wallace, Surf. Sci. Spectra, 2019, 26, 014015 CrossRef
.
- K. Bhakar, S. Roy, N. A. Rajpurohit and D. Kumar, ACS Appl. Nano Mater., 2024, 7, 28145–28159 CrossRef CAS
.
- X. Su, L. Hou, L. Xia, X. Yu, J. Guo, Y. Zhu and Y. Zhang, J. Mater. Sci., 2019, 54, 4559–4572 CrossRef CAS
.
- Y. He, J. Li, K. Li, M. Sun, C. Yuan, R. Chen, J. Sheng, G. Leng and F. Dong, Chin. J. Catal., 2020, 41, 1430–1438 CrossRef CAS
.
- H. Li, N. Li, M. Wang, B. Zhao and F. Long, R. Soc. Open Sci., 2018, 5, 171419 CrossRef PubMed
.
- S. Rajendran, T. Chellapandi, V. UshaVipinachandran, D. Venkata Ramanaiah, C. Dalal, S. K. Sonkar, G. Madhumitha and S. K. Bhunia, Appl. Surf. Sci., 2022, 577, 151809 CrossRef CAS
.
- Z. Qin, L. Zhu, X. Ge, C. Li, X. Wang and X. Liu, Inorg. Chem. Commun., 2025, 171, 113618 CrossRef CAS
.
- A. S. Correa, L. G. Rabelo, W. S. Rosa, N. Khan, S. Krishnamurthy, S. Khan and R. V. Gonçalves, Energy Adv., 2023, 2, 123–136 RSC
.
- H. Tripathy, A. Balakrishnan, M. Chinthala and A. Kumar, Ind. Eng. Chem. Res., 2024, 63, 20125–20143 CrossRef CAS
.
- S. N. F. M. Nasir, H. Ullah, M. Ebadi, A. A. Tahir, J. S. Sagu and M. A. Mat Teridi, J. Phys. Chem. C, 2017, 121, 6218–6228 CrossRef CAS
.
- W. Ma, Y. Zhang, L. Chen, X. Xie, S. Yuan, Z. Qiu, G. Zhu and J. Guo, Int. J. Biol. Macromol., 2024, 281, 136559 CrossRef CAS
.
- A. Balakrishnan, E. S. Kunnel, R. Sasidharan, M. Chinthala and A. Kumar, ACS Sustainable Chem. Eng., 2024, 12, 5169–5185 CrossRef CAS
.
- Y. Li, H. Zhang, S. Yao, C. Chao, Q. Chen, J. Chen, F. Fan, H. Jia and M. Dong, J. Water Process Eng., 2024, 63, 105490 CrossRef
.
- C. Mo, L. Zhou, J. Zheng, B. Liang, H. Huang, G. Huang, J. Liang, S. Li, M. Junaid, J. Wang and K. Huang, Chemosphere, 2024, 359, 142286 CrossRef CAS PubMed
.
- L. Zeng, C. Huang, Y. Tang, C. Wang and S. Lin, Ultrason. Sonochem., 2024, 106, 106886 CrossRef CAS
.
- M. Bigdeli Tabar, H. Azimi and R. Yousefi, Appl. Surf. Sci., 2023, 622, 156912 CrossRef CAS
.
- Z.-F. Huang, J. Song, X. Wang, L. Pan, K. Li, X. Zhang, L. Wang and J.-J. Zou, Nano Energy, 2017, 40, 308–316 CrossRef CAS
.
- T. Zhou, G. Dong, J. Geng, K. Han and C. Xiao, Appl. Surf. Sci., 2024, 655, 159592 CrossRef CAS
.
- B. Palanivel and A. Mani, ACS Omega, 2020, 5, 19747–19759 CrossRef CAS PubMed
.
- X. He, T. Kai and P. Ding, Environ. Chem. Lett., 2021, 19, 4563–4601 CrossRef CAS PubMed
.
- M. Wang, G. Lu, R. Jiang, T. Dang and J. Liu, J. Colloid Interface Sci., 2022, 622, 995–1007 CrossRef CAS PubMed
.
| This journal is © The Royal Society of Chemistry 2025 |