Perylene diimide-based photocatalysts: from molecular design to emerging applications
Yin
Xiao
a,
Zihe
Chen
a,
Xin
Liu
a,
Xusheng
Wang
 b,
Guixiang
Ding
a,
Zhaoqiang
Wang
a,
Peng
Wang
*c and
Guangfu
Liao
b,
Guixiang
Ding
a,
Zhaoqiang
Wang
a,
Peng
Wang
*c and
Guangfu
Liao
 *a
*a
aCollege of Materials Engineering, Fujian Agriculture and Forestry University, Fuzhou 350002, China. E-mail: liaogf@mail2.sysu.edu.cn
bState Key Laboratory of Bio-based Fiber Materials, School of Materials Science and Engineering, Zhejiang Sci-Tech University, Hangzhou 310018, China
cCenerTech Tianjin Chemical Research & Design Institute Company, Ltd, Tianjin 300131, China. E-mail: lzuwangpeng@163.com
First published on 16th August 2025
Abstract
Perylene diimide (PDI)-based semiconductor materials show significant promise for photocatalytic environmental decontamination and the conversion of energy resources but suffer from inefficient photocarrier separation which greatly limits their activity. Consequently, designing PDI-based photocatalysts to enhance carrier separation has become a major research focus. This persistent challenge has positioned the rational design of PDI-based architectures to enhance carrier dissociation kinetics and elevate functional efficacy as a central focus point of research in the field of contemporary photocatalysis. This review firstly examines recent progress in the rational design of PDI-based photocatalysts and their charge transfer mechanism. Then, advances in the fabrication of PDI photocatalysts and associated electron/hole transfer mechanisms are discussed. It systematically evaluates their enhanced activity in key applications: water splitting, CO2 reduction, N2 fixation, and pollutant degradation etc. Subsequently, the fundamental photocatalytic mechanism inherent to PDI-based materials is scrutinized in depth. Finally, outstanding issues and prospective uses for PDI-based photocatalysts are also discussed. It is believed that this review provides a valuable direction for engineering advanced PDI-based photocatalytic systems.
Wider impactConventional energy production exacerbates global environmental degradation, including climate change and greenhouse effects. Consequently, implementing sustainable renewable energy systems is imperative to mitigating impending energy crises, preserving ecological integrity, and achieving zero-emission targets with solar-driven artificial photosynthesis on advanced semiconductors representing a critical pathway for circular energy cycles. PDI semiconductor photocatalysts are prized for their economic viability, operational stability, superior photochemical responses, and electron-accepting capacity. This review systematically chronicles the evolution of PDI-based photocatalytic systems, combined with contemporary synthesis paradigms with emphasis on structural/functional modifications to advance semiconductor photocatalysis for solar energy harvesting. We further delineate persistent research challenges and strategic future directions. The transformative potential of these advanced materials underscores the imperative for cross-disciplinary convergence between materials chemistry and process engineering. Such synergies will catalyze innovative breakthroughs in semiconductor photocatalysis, accelerating the development of sustainable energy technologies. By consolidating design principles, mechanistic insights, and application landscapes, this work provides a foundational framework for researchers engaged in photocatalytic energy conversion and serves as a blueprint for engineering PDI photocatalysts. |
1. Introduction
Global economic and industrial expansion exacerbates two critical challenges: environmental pollution and energy scarcity.1–8 Semiconductor photocatalysis emerges as a promising technology, efficiently enabling solar-to-chemical energy transformation through photoredox reactions for pollutant degradation,9–11 CO2 reduction,12,13 and H2 production.14,15 This positions it as a key solution for environmental remediation and renewable energy. However, current limitations including short charge carrier lifetimes, insufficient sunlight utilization, poor stability, and low efficiency16 hinder its practical deployment. Recently, organic semiconductor photocatalysts have proliferated due to their molecularly adjustable optoelectronic characteristics, structural versatility and cost-effective synthesis, etc., with representative examples including covalent organic frameworks (COFs),17 metal–organic frameworks (MOFs),18 organic polymers19 and organic supramolecular compounds,20etc. Supramolecular organic semiconductors now constitute a rapidly developing photocatalytic domain, benefiting from precise synthetic control and broad spectral absorption capabilities. Among the organic semiconductors, PDI has garnered significant scientific attention due to its synthetic accessibility, cost-efficiency, and sustained functional integrity under photocatalytic conditions. Nevertheless, their photocatalytic performances remain hampered by inefficient charge separation kinetics and limited operational persistence.21 Dating back to 1913, PDI first served as industrial dyes exhibiting robust durability, chemical resistance, thermal stability, lightfastness, and weatherability. The compound further reveals significant electronic properties beyond pigmentation, including substantial luminescence efficiency, excellent photo-stabilization capacity, and strong electron-accepting capability. Nowadays, PDI demonstrates significant applicability across multiple domains including sensors,22 fluorescent switches,23 fluorescent probes,24 photoconductive materials,25,26 and organic light-emitting diodes (OLEDs),27etc. A landmark 1997 study by Robert et al.28 identified molecular PDI as a photocatalytic photosensitizer. Through photoinitiated energy transfer mechanisms, it produces singlet oxygen that mineralizes phenolic contaminants (e.g., phenol) within controlled pH regimes.Relative to molecular PDI, supramolecular constructs demonstrate advanced photocatalytic behavior, reflecting notable recent advancements in organic photocatalyst design.29–33 Consequently, this has elevated research focus on PDI-based photocatalysts, centering on photogenerated charge behavior, molecular structure–function relationships, and oxidative/reductive reaction mechanisms. Non-covalent interactions – hydrogen bonding, dipole–dipole, π–π stacking, van der Waals, hydrophobic, and electrostatic forces – govern PDI supramolecular assembly. These approaches permit efficient organic photocatalytic architectures through mild, adaptable synthesis, outperforming covalent organic frameworks (COFs) in structural precision and synthetic economy while circumventing elaborate polymerization pathways.
This review analyzes recent progress in PDI-based photocatalytic architectures. Firstly, we introduce the molecular structure of PDI. Secondly, PDI-based photocatalysts are briefly summarized, including modifying the molecular engineering of PDI monomers (such as the substituents of side-chain and bay position), design of PDI polymers, heterojunction engineering (type-II, type-Z and type-S systems), metal deposition/doping, and construction of π–π composite systems, etc. Thirdly, the application of PDI-based photocatalysts including water splitting, CO2 reduction, N2 fixation, and pollutant degradation are summarized. Finally, the analysis concludes by outlining persistent challenges and forward-looking strategies for advanced photocatalyst design. This work aims to establish actionable frameworks for developing high-efficiency PDI materials that enable sustainable energy generation and environmental remediation.
2. Molecular structure of PDI
PDI is a derivative of polycyclic aromatic hydrocarbons,34,35 featuring a perylene core symmetrically functionalized with dual imide groups (–CONHCO–)36 Conventionally synthesized via terminal amidation of 3,4,9,10-perylenetetracarboxylic dianhydride (PTCDA) (Fig. 1a and b), its primary modification sites comprise terminal ‘imide positions’ and peripheral ‘bay positions’ (carbons 1, 6, 7, 12). Usually, the electron density at the nodes of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) of PDI molecules nearly approaches zero, meaning that side-chain motifs do not readily engage in Π-electron conjugation with a perylene ring and fail to significantly affect the overall electronic structures of PDI molecules. Frontier molecular orbital analysis (Fig. 1c) reveals carbon and oxygen dominate HOMO/LUMO composition, while amide nitrogen exhibits negligible orbital contribution.37 Modifications at the bay position alter the intrinsic energy levels and redox potential of PDI molecules, whereas substitutions at the imide position preserve these fundamental electronic properties and, consequently, their spectral absorption and emission characteristics. This methodology enables strategic modulation of imide substituents to probe structure–photoactivity relationships while conserving intrinsic orbital energetics. | ||
| Fig. 1 (a) Molecular structures of PTCDA and (b) PDI which show the numbering of the positions in the ring system. (c) DFT calculations of frontier orbitals of N,N′-dimethyl PDI. Reproduced from ref. 37. Copyright 2011, American Chemical Society. (d) Size of a single PDINH molecule and the π–π stacking distance. Reproduced from ref. 38. Copyright 2016, Wiley. (e) H/J-aggregated PDI photocatalyst diagram representing the effect of (ET) and (EnT). Reproduced from ref. 41. Copyright 2018, Elsevier. | ||
The perylene ring of PDI features a rigid polycyclic π-conjugated framework, driving supramolecular assembly via π-orbital interactions. Typical interplanar distances in these stacked architectures measure 3.4–3.5 Å (Fig. 1d), mirroring graphene's interlayer spacing.38 Computational studies by Zhu et al. employing density functional theory (DFT) reveal enhanced π-stacking reduces PDI's band gap and lowers both HOMO/LUMO energy levels. Unlike monomers, supramolecular PDI exhibits semiconductor-like continuous bands due to non-covalent molecular ordering.39 Würthner et al.40 demonstrated that imide-position substituents modulate stacking configurations through steric and non-covalent effects. PDI aggregates primarily adopt H-aggregate or J-aggregate arrangements. H-aggregates display strong π-orbital overlap and extended conjugation, yielding semiconducting behavior. Conversely, J-aggregates maintain molecular photophysical properties due to reduced π-coupling. Mechanistic studies show H-aggregates facilitate electron transfer (ET): co-facial stacking creates π-delocalized channels enabling rapid electron migration to oxygen. J-aggregates favor energy transfer (EnT) processes. Upon photoexcitation, they efficiently generate triplet states via intersystem crossing, which enables them to act as potent photosensitizers (Fig. 1e). Spectroscopically, H-aggregates exhibit hypsochromically shifted absorption with fluorescence quenching and reduced quantum yields,41 while J-aggregates exhibit bathochromic shifts without significant emission loss42 making them the preferred fluorophores.43 In photocatalysis, H-aggregates demonstrate superior potential due to deeper valence bands, enhanced charge mobility/separation, and stronger oxidative capacity.44 This performance stems from PDI's planar aromatic structure mediating robust intermolecular π–π interactions.
3. Modification of PDI-based photocatalysts
Despite emerging as promising photocatalysts, nevertheless, the photocatalytic performance of PDI supramolecular systems is fundamentally constrained by three principal factors: elevated photogenerated exciton recombination rates, diminished charge transport kinetics, and insufficient oxidative capacity originating from low-lying VB positions.45 Fundamentally, photocatalytic mechanisms comprise three consecutive processes: (i) photoexcitation across the semiconductor bandgap, (ii) spatial separation and transport of photogenerated charge carriers, and (iii) surface redox reactions. Consequently, rational material modifications primarily target two critical objectives: (i) broadening the spectral response range through bandgap engineering and/or sensitization strategies, and (ii) enhancing charge carrier dynamics by minimizing recombination losses while optimizing mobility pathways.46 To overcome these inherent constraints, researchers have proposed many strategies including the design of PDI monomers and polymers, construction of π–π composite systems and heterojunctions system etc. This section provides a critical evaluation of contemporary advancements in enhancing the photocatalytic performance of PDI-based photocatalysts via these methods.3.1. Monomer modification engineering
PDI, an n-type organic semiconductor and high-grade dye, features a polycyclic aromatic structure with an electron-rich perylene core and electron-withdrawing imide groups. This conjugation enables efficient charge carrier migration. PDI monomer architecture – determined by planar conjugation extent, substituent properties, and dipole moment – modulates intermolecular interactions (π–π stacking, electrostatic forces, hydrophobic effects, steric constraints). These interactions govern electronic wavefunction overlap and interchromophoric coupling, enabling precise engineering of supramolecular band structures. In this section, we primarily discuss the influence of substituent groups on the molecular properties of PDI monomers (Table 1 and Fig. 3). Substitution at the imide (N-substitution) and bay positions of PDI molecules serves as a fundamental strategy for precisely tuning their photophysical properties, molecular stacking behavior, and photocatalytic performance.| Photocatalysts | The amount of catalyst (mg) | Morphology | Source of light | Photocatalytic application | Performance | AQY | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | 25 | Nanosheets | Xenon lamp, 300 W, λ > 420 nm | Pollutant | — | 68 | |
| Phenol | 1.23 (h−1) | ||||||
| Catechol | 1.46 (h−1) | ||||||
| BPA | 2.77 (h−1) | ||||||
| 4-CP | 2.08 (h−1) | ||||||
| 4 | 25 | Nanobelts | White LED, 5 W | Pollutant | — | 57 | |
| TC | 0.71 (h−1) | ||||||
| MB | 1.24 (h−1) | ||||||
| RhB | 0.55 (h−1) | ||||||
| 5 | 25 | Ultrathin nanosheets | Xenon lamp, 500 W, λ > 420 nm | Pollutant | 33 | ||
| Phenol | 0.51 (h−1) | ||||||
| OTC | 0.65 (h−1) | ||||||
| EE | 0.55 (h−1) | ||||||
| 6 | 50 | Nanobelts | Xenon lamp, 300 W, λ > 420 nm | H2 production | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 (μmol h−1 g−1) 700 (μmol h−1 g−1) |
2.96 (550 nm) | 62 |
| 7b | 25 | Nanosheets | Xenon lamp, 300/500 W, λ > 420 nm, | Pollutant phenol/O2 production | 1.45 (h−1)/2490 (μmol h−1 g−1) | — | 69 |
| 8 | 25 | 2D layers with flaky | Xenon lamp, 1000 W, λ > 420 nm | Pollutant | — | 30 | |
| MB | 0.262 (h−1) | ||||||
| DCF | 0.172 (h−1) | ||||||
| 9c | 25 | Nanobelt | Xenon lamp, 500 W, λ > 420 nm | Pollutant phenol/O2 production | 3.96 (h−1)/11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 (μmol h−1 g−1) 700 (μmol h−1 g−1) |
— | 56 |
| 10 | 25 | Nanofibers | Xenon lamp, 500 W, λ > 420 nm | Pollutant phenol | 0.129 (h−1) | — | 39 |
| 11 | — | — | White LED array | H2O2 production | — | <1% | 70 |
Alkyl chains, as one of the most prevalent imide side-chain modifications in PDI systems, demonstrate substantial influence on photocatalytic performance through three primary mechanisms: solubility modulation, molecular packing control, and charge transfer regulation. From a solubility perspective, the introduction of alkyl chains effectively attenuates the strong intermolecular π–π interactions characteristic of PDI derivatives. This attenuation effect not only prevents excessive molecular aggregation but also significantly enhances solubility in organic solvents including chloroform and toluene.47–50 The resultant increase in accessible catalytically active sites substantially improves interfacial contact between the photocatalyst and organic substrates. However, the inherent nonpolar nature of alkyl chains presents limitations in polar reaction media.51 Specifically, their weak interactions with aqueous phases and metal oxide surfaces create substantial barriers for efficient charge transfer processes in aqueous photocatalytic systems, as evidenced by reduced quantum yields in water-splitting applications. Regarding molecular organization, alkyl chains – particularly those with optimized length (C6–C12) and branching patterns – induce a distinctive J-type stacking configuration through steric repulsion effects. This packing mode exhibits two critical characteristics: (1) it generates a moderately widened bandgap (2.2–2.5 eV) due to decreased orbital overlap, and (2) facilitates the formation of highly ordered one-dimensional nanostructures.52 Spectroscopically, the J-aggregation induced by alkyl side chains produces a pronounced bathochromic shift in the absorption spectrum. This redshift effect extends the visible light harvesting range to longer wavelengths (λ > 550 nm),53 thereby enhancing solar energy utilization efficiency in optimized systems. While the application of alkyl-modified PDIs in polar reaction environments remains challenging, their unparalleled ability to precisely control fundamental material properties makes them indispensable for photocatalytic applications involving nonpolar substrates. The structure–property relationships established in these systems provide valuable design principles for developing advanced organic photocatalysts. For instance, Wang et al.41 synthesized PDI derivatives with different alkyl chain lengths (H-PDI and J-PDI) via a pH-triggered hydrogelation method, systematically investigating the influence of side-chain substituents on photocatalytic performance. The study revealed that H-PDI with shorter side chains formed face-to-face π–π stacking, known as H-aggregation, exhibiting semiconductor characteristics with a narrowed bandgap of 1.69 eV. This configuration predominantly facilitated electron transfer, generating superoxide radicals and holes, which demonstrated superior activity for phenol degradation under visible light with a rate constant of 0.195 h−1. In contrast, J-PDI with longer side chains adopted a head-to-tail stacking mode, referred to as J-aggregation with a bandgap of 1.78 eV, which promoted energy transfer and efficiently produced singlet oxygen with a high quantum yield of 0.66. Under 600 nm red light irradiation, J-PDI exhibited significantly enhanced inhibition of HeLa cells compared to H-PDI. This work elucidates how side-chain engineering can precisely modulate supramolecular packing to optimize photocatalytic pathways, providing a novel strategy for designing tailored photocatalysts for environmental remediation and antitumor applications.
Aromatic or rigid side chains, such as phenyl, naphthyl, or cycloalkyl groups (e.g., 4-tert-butylphenyl), exert a distinct influence on the photocatalytic performance of PDI molecules through their impact on molecular stacking, stability, and electronic interactions.52 These rigid aromatic moieties strengthen intermolecular π–π stacking via additional aromatic interactions, which enhances charge delocalization across the PDI backbone and improves photostability by reducing structural fluctuations under light irradiation – an advantage for long-term catalytic reactions. However, their large steric volume and strong intermolecular interactions led to poor solubility in both organic solvents and aqueous media, limiting the processability of PDI and potentially causing excessive aggregation that reduces the accessible catalytic active sites.54 In terms of molecular packing, aromatic side chains induce a face-to-face H-type stacking mode with a relatively small d-spacing (approximately 3.5 Å), which narrows the band gap of PDI, enabling stronger absorption of visible light. Nevertheless, the tight and rigid stacking increases carrier scattering, resulting in lower charge mobility compared to alkyl-modified PDI. This trade-off between strong light absorption and moderate charge mobility makes aromatic-modified PDI particularly suitable for photocatalytic reactions requiring high oxidizing power and selectivity. Overall, aromatic or rigid side chains play a unique role in optimizing PDI's performance in selective organic synthesis and reactions demanding high stability.55 For example, Zhu et al.56 implemented a σ-spacer length optimization strategy to augment charge mobility in imidazole-alkyl-perylene diimide (IMZ-alkyl-PDI) photocatalysts with donor–spacer–acceptor (D–σ–A) architecture through precise π–π stacking distance regulation (Fig. 2a). Among the series – non-alkylated (C0IPDI), ethyl-bridged (C2IPDI), and propyl-modified (C3IPDI) – the ethyl linkage achieved minimal π–π separation (3.19 Å) by steric minimization between donor/acceptor units, elucidating intrinsic photocarrier transport mechanisms. C2IPDI demonstrated exceptional photocatalytic enhancement: 32-fold greater phenol degradation efficiency versus IMZ-PDI, alongside a 271-fold increase in oxygen evolution (Fig. 2b and c). Moreover, Sun and coworkers33 engineered an ultrathin porous hp-PDI-NA photocatalyst via nicotinic acid terminal substitution (Fig. 2d). It demonstrated 3.5-fold higher visible-light phenol degradation activity than nano-PDI, achieving near-complete mineralization. The catalyst retained 98% activity after 5 cycles, and effectively mineralized antibiotics (oxytetracycline) and hormones (ethinylestradiol) (Fig. 2e). Combined characterization/theory revealed its enhanced performance stems from a bi-planar conformation and hierarchically porous nanosheet morphology. NA substitution reduces steric hindrance, strengthens π–π conjugation, and shortens interlayer spacing, thereby boosting carrier separation/transport and structural stability (Fig. 2f). Although aromatic substituents further extend conjugation, narrowing the bandgap for enhanced visible-light absorption which offers critical advantages including improved processability for homogeneous composite formation, tunable stacking distances for efficient charge transport, and increased surface hydrophilicity to facilitate pollutant adsorption and water activation, bulky groups may disrupt π-conjugation and increase charge transport resistance, excessive intermolecular interactions can reduce active site accessibility, and chemically unstable substituents (e.g., certain alkyl amines) may degrade under prolonged irradiation, compromising catalytic durability. To enhance photocatalytic performance, functional group modifications are employed.
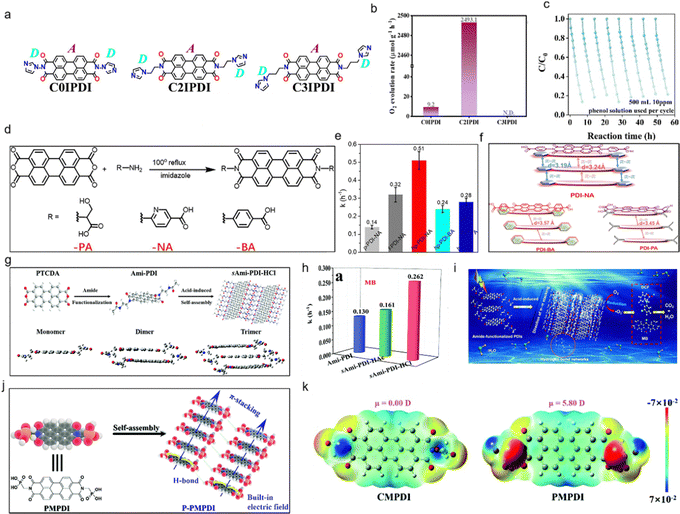 | ||
| Fig. 2 (a) Different σ lengths of molecular structures about CnIPDI. (b) Comparison of photocatalytic O2 evolution rate on CnIPDI. (c) Cyclic stability of C2IPDI. Reproduced from ref. 56. Copyright 2023, Wiley. (d) The synthesis method of PDI-NA molecular and reference catalyst. (e) TOC removal rate and mineralization rate constant over different photocatalysts. (f) A schematic diagram of PDI-NA, PDI-BA and PDI-PA. Reproduced from ref. 33. Copyright 2022, Elsevier. (g) The synthesis process and structure of sAmi-PDI-HCl (over) and the DFT calculations regarding geometries of sAmi-PDI-HCl monomers, dimers, and trimers (below). (h) The kinetics constants toward MB. (i) The mechanism for the photocatalytic degradation process when using amide-functionalized supramolecular PDI. Reproduced from ref. 30 Copyright 2020, the Royal Society of Chemistry. (j) The synthesis of supramolecular P-PMPDI. (k) Diagram of molecular dipoles and electron distribution in PDI derivatives. Reproduced from ref. 62. Copyright 2019, the Royal Society of Chemistry. | ||
Polar functionalized chains, such as those bearing carboxyl (–COOH)39,57 or amine (–NH2)58 groups (e.g., glycine or polyethylene glycol derivatives), play a crucial role in regulating the photocatalytic performance of PDI molecules through their influence on solubility, intermolecular interactions, and heterojunction formation. These polar groups significantly enhance the water solubility of PDI, addressing the issue of poor dispersibility in aqueous systems that limits the accessibility of catalytic active sites, thereby facilitating contact with water-soluble pollutants and improving reaction kinetics. Additionally, the polar nature of these side chains enables strong hydrogen bonding or electrostatic interactions with metal oxides (e.g., TiO2, BiOCl, etc.) or metal ions (e.g., Zn2+),59 which is critical for the formation of stable heterojunctions; such heterojunctions promote efficient interfacial charge transfer, reducing the recombination rate of photogenerated electrons and holes. For instance, carboxyl-functionalized PDI can form a close heterojunction with BiOCl,60 where the interfacial charge transfer is accelerated, significantly increasing the generation of hydroxyl radicals (˙OH) and resulting in a threefold enhancement in phenol degradation efficiency compared to alkyl-modified PDI. However, these polar side chains can also induce excessive intermolecular interactions, leading to H-type aggregation in some cases, which may narrow the visible-light absorption range and increase electron–hole recombination, thereby partially offsetting the positive effects. In terms of molecular stacking, the polar interactions between these side chains tighten the π–π stacking, which narrows the band gap of PDI, allowing for better utilization of visible light. Overall, polar functionalized chains are particularly advantageous in aqueous photocatalytic systems and heterojunction-based catalytic systems, despite their potential to induce unfavorable aggregation, their ability to enhance solubility and promote charge transfer makes them indispensable in optimizing PDI's photocatalytic performance. For example, Li et al.30 engineered an intralayer polarization field within amide-functionalized PDI supramolecular assemblies (sAmi-PDI) (Fig. 2g). π–π stacking and hydrogen bonding synergistically enhance polarization while constructing electron–hole transfer bridges and accelerating carrier separation. The acidic medium optimized electrostatic interactions and provided abundant electron donors/acceptors, boosting self-assembly efficiency. Benefitting from these effects, sAmi-PDI exhibited twofold-enhanced photocatalytic activity in pollutant degradation. The polarization field – originating from supramolecular networks – enables rapid carrier migration, establishing a green synthesis paradigm for high-performance PDI photocatalysts (Fig. 2h and i). Guo et al.61 synthesized engineered an oxygen-deficient PDI supramolecular system (R-Ov-PDI) to optimize hole migration kinetics. Photoinduced holes were preferentially trapped at anionic defect centers, triggering an attack on the C–N bond. Remarkably, visible-light-driven benzylamine photooxidation achieved a benchmark efficiency of 31.3 mmol g−1 h−1 with >99% imine selectivity. Moreover, Kong et al.62 engineered the preparation of a non-covalent self-assembled phosphoric acid-substituted PDI (PMPDI) (Fig. 2j). Functionalization with electron-withdrawing terminal groups (Fig. 2k) enhanced photocatalytic performance through improved exciton dissociation and extended photon harvesting range. Pu et al.57 synthesized a series of asymmetrically structured PDI supramolecular photocatalysts (PDI-CH3, PDI-NH2, and PDI-COOH) via terminal group modification of imide positions, aiming to enhance the internal electric field (IEF) through molecular dipole engineering. Density functional theory (DFT) calculations revealed that the electron-withdrawing –COOH group in PDI-COOH induced the largest dipole moment (2.3257 D), followed by PDI-NH2 (1.1715 D) and PDI-CH3 (0.0034 D), directly correlating with IEF intensity (PDI-COOH: 8.4 × PDI-CH3). This enhanced IEF significantly improved charge separation efficiency from 4.6% (PDI-CH3) to 11.2% (PDI-COOH), as confirmed by photoelectrochemical tests and surface photovoltage spectroscopy. The work demonstrates that asymmetric molecular design amplifies IEF to simultaneously boost oxidative and reductive photocatalytic activities, offering a universal strategy for organic photocatalyst optimization.
3.2. Polymer modification engineering
The limited stability of PDI supramolecular materials, stemming from weak non-covalent interactions, presents a significant challenge. Replacing these interactions with directional covalent linkages between PDI monomers would simultaneously enhance structural integrity and preserve the uninterrupted π-delocalization channels essential for rapid electron migration. Zhang's group71 fabricated crystalline urea-PDI materials, achieving an oxygen evolution rate of 3223.9 μmol μmol g−1 h−1 with an apparent quantum yield (AQY) of 3.86% at 450 nm illumination (Fig. 4a and b). The combined effect of crystallinity and molecular dipole moment established a potent IEF. This configuration facilitated effective charge separation and sustains photocatalytic activity for over 100 hours (Fig. 4c). Subsequently, in Cao's work72 1D Co-UPDI nanocrystals were synthesized via Co2+-UPDI assembly, achieving a record POE rate in AgNO3 colloids, which was 10 times higher than that of UPDI alone. Co–N coordination enhances H-stacking rigidity and enabled in situ CoOOH formation. Wu et al.73 prepared non-continuous conjugated semiconductor EDA-PTCDA nanosheets using an uncomplicated solvothermal approach assisted by acidification (Fig. 4d). Significant molecular dipole anisotropy enabled efficient partitioning and trapping of photogenerated electron–hole pairs. This polymer demonstrates stable, continuous reactive oxygen species (ROS) production under natural sunlight, conferring effective microbicidal action against both Gram-positive and Gram-negative bacterial strains (Fig. 4e). Huang's74 group synthesized a 3D porous PDI-CTS polymer photocatalyst with a donor–acceptor (D–A) structure. It exhibited a remarkable bisphenol A degradation rate (0.343 min−1) through persulfate radical generation. Donor–acceptor synergy accelerated interfacial charge migration, creating a larger dipole moment and a 6.9-fold stronger IEF than pure PDCTA, greatly facilitating photogenerated carrier separation. Recently, Huang et al.75 synthesized m-, p-, and o-PDI polymers by coupling PDI with benzene diamines at distinct positions to enhance charge carrier separation (Fig. 4f). Distinct linkage configurations modulated specific surface area, band energetics, and charge transport behavior. Among these, m-PDI demonstrated maximal interfacial exposure and the most negative VB position, while its distinctive architecture enhanced ofloxacin (OFL) adsorption affinity and electron transfer kinetics. Consequently, m-PDI achieved a 0.07481 min−1 OFL degradation rate (60 min, light) with robust stability in aquatic environments (Fig. 4g and h). Recent representative studies (Fig. 5 and Table 2) highlight three principal merits of (PDI)-based polymers: (1) inherent high crystallinity substantially improves charge transport efficiency; (2) significant molecular dipoles facilitate formation of intensified IEF, enabling accelerated movement of photoinduced charges; (3) relative to supramolecular PDI systems, covalently bonded architectures demonstrate enhanced structural integrity. | ||
| Fig. 4 (a) Solid 13C NMR spectrum of urea-PDI. (b) The photocatalytic oxygen evolution with urea-PDI. (c) Mechanism diagram of the urea-PDI polymer photocatalyst. Reproduced from ref. 71. Copyright 2020, Wiley. (d) EDA-PTCDA synthetic methods and solid state 13C NMR spectrum. (e) Quantitative analysis of intracellular ROS before and after natural light irradiation. Reproduced from ref. 73. Copyright 2023, Wiley. (f) Diagram of photocatalyst synthesis. (g) Comparison of degradation rate constants of three materials. (h) UV-vis absorption spectra of m-PDI, p-PDI and o-PDI. Reproduced from ref. 75. Copyright 2024, Elsevier. | ||
| Photocatalysts | The amount of catalyst | Morphology | Source of light | Photocatalytic application | Performance | AQY | Ref. |
|---|---|---|---|---|---|---|---|
| 1a | — | Uniform nanosheet | AM1.5G | Water splitting | Photocurrent density 115.1 (μA cm−2) | — | 76 |
| 2 | 25 | Ultrathin sheets | Xenon lamp, 300 W | O2 | 5110.25 (μmol h−1 g−1) | 2.15 (420 nm) | 77 |
| 3 | 25 | Nanobelt | Xenon lamp, 300 W, λ > 420 nm | O2 | 3223.9 (μmol h−1 g−1) | 3.86 (450 nm) | 71 |
| 4 | 10 | Layer structure | LED lamp, 100 W | Pollutant Cr | 2.04 (h−1) | — | 78 |
| 5 | Nanosheets | Natural light source | Antibacterial Escherichia coli/Staphylococcus aureus | within 60/45 min | — | 73 |
3.3. Heterojunction engineering
While a narrow bandgap enhances solar energy utilization in photocatalysts, it concomitantly promotes photoinduced charge recombination. Conversely, wide bandgaps suppress recombination and preserve strong redox potentials for charge carriers, yet excessively wide gaps limit broad-spectrum photon harvesting. To resolve this trade-off – termed the ‘single-component bottleneck’ – heterojunction engineering provides an effective strategy. Heterojunction photocatalysts comprise two or more distinct semiconductor components, establishing an IEF at their material interfaces. This IEF serves as the primary impetus for photogenerated charge transfer, effectively prolonging carrier lifetimes while minimizing recombination rates.79–81 This approach increases the lifetime of photo-generated charges and reduces their recombination. In addition, the construction of a heterojunction can also optimize the band positions and facilitate surface catalytic reactions. Contemporary advances in PDI-based heterojunctions utilize these principles to boost photocatalytic efficiency for solar fuel production and pollutant degradation. These systems are categorized primarily by charge transfer mechanism into three classes: type-II, Z-scheme, and S-scheme heterojunctions incorporating PDI. The subsequent portion methodically reviews seminal recent developments in PDI-based heterostructure photocatalysts.Bismuth-based photocatalysts have emerged as prominent materials due to their extended light-harvesting range, modifiable energy band characteristics, and unique electronic properties. However, persistent limitations include inadequate photon capture efficiency and structural instability. A promising strategy to mitigate these constraints involves constructing heterostructures through integration of PDIs with these photocatalytic systems. For instance, Zhang et al.82 synthesized Bi2WO6/PDI heterojunctions via water bath heating. Under visible light, the Bi2WO6/PDI heterojunction exhibited higher phenol degradation rates than pure Bi2WO6 or self-assembled PDI, and doubled the oxygen production rate of pure PDI (Fig. 7b and c). The enhanced activity stems from the formation of an n–n type-II heterojunction which has a staggered band alignment facilitating charge separation (Fig. 7a).
 | ||
| Fig. 7 (a) The possible reaction mechanism of Bi2WO6/PDI composite materials. (b) Photocatalytic degradation of 5 ppm phenol. (c) Oxygen evolution from water by PDI and the 50%-Bi2WO6/PDI sample in the presence of an electron acceptor. Reproduced from ref. 82 Copyright 2018, Elsevier. (d) The possible photocatalytic mechanism of the CPM-2 composite under visible light irradiation. Reproduced from ref. 83. Copyright 2019, Elsevier. (e) Possible photocatalytic mechanism of the BiOBr/Bi4O5Br2/PDI system. Reproduced from ref. 84. Copyright 2022, Elsevier. (f) Degradation mechanism of TC in the system of visible light/6%PDI/FM88B/H2O2. (g) Ammoniation reaction between PDI and FM88B. (h) Effect of H2O2 concentration on the degradation efficiency of TC over 6%PDI/FM88B. Reproduced from ref. 85. Copyright 2024, Elsevier. | ||
Research in photocatalysis indicates the considerable potential of nonmetallic carbon-based semiconductors due to their superior optoelectronic characteristics and eco-compatibility. Type-II heterojunctions between PDIs and carbon materials have also gained increasing attention for applications. Particularly noteworthy are n-type graphitic carbon nitride (g-C3N4) photocatalysts, which represent the most extensively investigated system in this category. Li et al.83 synthesized a g-C3N4/PDI@NH2-MIL-53(Fe) (CPM) type-II heterojunction via thermal polymerization, surface growth, and solvothermal methods (Fig. 7d). The CPM heterojunction demonstrated exceptional visible-light photocatalytic activity with H2O2, degrading aqueous pollutants rapidly. This superior performance stems from the optimized type-II heterojunction. Close interfacial contact and aligned band structures between g-C3N4/PDI and NH2-MIL-53(Fe) enhance charge separation.
Ternary or multicomponent systems enhance carrier separation efficiency through synergistic effects. Wang et al.84 developed a visible-light-responsive BiOBr/Bi4O5Br2/PDI dual heterojunction photocatalyst for efficient degradation of endocrine-disrupting chemicals (EDCs) in water (Fig. 7e). The aligned energy bands between Bi4O5Br2 and BiOBr promote heterojunction formation, improving space-charge separation. Simultaneously, PDI loading extends the photo-response range while facilitating carrier transfer and separation. Within this dual-heterojunction system: (1) electrons migrate to BiOBr's CB. (2) Holes accumulate on PDI's VB. This charge separation enables H2O oxidation to O2 followed by ˙O2− formation through reduction. Notably, photocatalytic activity persists significantly under anoxic conditions.
Metal–organic frameworks (MOFs) constitute porosity-defined crystalline solids formed by metal nodes cross-linked to organic molecular bridges. Their extensive surface exposure, abundant coordination centers, facile surface functionalization, and tunable porosity enable widespread applications in environmental remediation and catalytic processes. Wu et al.85 successfully modified PDI onto NH2-MIL-88B(Fe, Mn) (FM88B) via water bath heating, forming a PDI/FM88B type-II heterojunction (Fig. 7f). This modification, confirmed by amide bond formation (Fig. 7g), enhanced structural stability and created an efficient interfacial electron transfer pathway. In the photo-Fenton system, 6%-PDI/FM88B achieved 89% tetracycline (TC) degradation under 30 min visible light (Fig. 7h). The performance enhancement arises from synergistic photocatalysis-Fenton processes, wherein the type-II heterojunction directs photoinduced electron transfer from PDI (donor) to FM88B (acceptor), while the interfacial IEF crucially promotes e−–h+ separation and directional migration – further amplified by the heterostructure's strong light absorption.
Despite demonstrating effective photogenerated charge segregation, it still exhibits limitations: (1) in type II heterojunctions, electrons transfer from the CB of the narrow-bandgap semiconductor to the CB of the wide-bandgap semiconductor, while holes migrate from the VB of the wide-bandgap to the VB of the narrow-bandgap semiconductor. This results in preserved carriers with both reduced redox potentials compared to the individual semiconductors. (2) The light absorption range of type II heterojunctions is fundamentally constrained by the narrow-bandgap component, leading to inefficient broadband solar energy utilization. (3) In type II heterojunctions formed solely through physical contact, interfacial defects or gaps introduce additional charge-transfer resistance, significantly compromising carrier separation efficiency. (4) The preserved charge carriers with diminished redox potentials tend to accumulate and recombine in the absence of sacrificial agents for hole/electron consumption, ultimately degrading catalytic efficiency. Consequently, the type-II mechanism remains controversial.
For example, the π–π stacking in the NDINH/PDINH supramolecular system which was developed by Xu et al.95 creates a giant IEF (Fig. 9a), significantly enhancing charge separation and carrier lifetime. This IEF drives a direct Z-scheme charge transfer pathway, preserving strong redox potentials for both half-reactions. Concurrently, the NDINH coating attenuates backscattered electromagnetic fields across PDINH surfaces, enhancing UV-light utilization efficiency. Exceptional full-spectrum photocatalytic overall water splitting (OWS) activity is enabled by these synergistic effects, achieving H2 and O2 evolution rates of 317.2 and 154.8 μmol g−1 h−1 respectively. This performance is further evidenced by a high O2 evolution rate of 2.61 μmol g−1 h−1 (with AgNO3) and 0.13% solar-to-hydrogen efficiency (Fig. 9b and c). Dai et al.96 developed a 3D PANI/PDI direct Z-scheme photocatalytic system. Retained electrons in PANI's CB and holes in PDI's VB drove oxygen radical formation. Meanwhile, a novel PDI/FePc heterojunction featuring strong π–π interactions was synthesized via a self-assembled method97 (Fig. 9d). This structure demonstrated enhanced visible-light photocatalytic degradation of tetracycline hydrochloride (TC), achieving removal rates 3-fold and 87.5-fold higher than pristine PDI and FePc, respectively and exhibiting superior oxidation kinetics, evidenced by a lower Tafel slope (131.1 mV dec−1) versus PDI (228.6 mV dec−1) (Fig. 9e and f). The observed boost originates from π-conjugated interactions minimizing layer-to-layer separation within the molecular assembly, consequently enhancing charge separation and transport efficiency.
 | ||
| Fig. 9 (a) Schematic diagram of the NDINH/PDINH Z-scheme electron transfer process. (b) The overall water splitting performance over different catalysts under full-spectrum light irradiation. (c) Time course of photocatalytic overall water splitting over NDINH/PDINH. Reproduced from ref. 95. Copyright 2023, Wiley. (d) PDI/FePc Z-scheme electron transfer mechanism. (e) Photocatalytic degradation of TC activities over PDI, FePc and PDI/FePc heterojunctions. (f) The corresponding Tafel plots. Reproduced from ref. 97. Copyright 2023, Elsevier. | ||
Strategic engineering of electronic band gaps in hybrid organic–inorganic materials through covalent bonding enables precise fabrication of S-scheme heterojunctions. For example, through electrostatic interactions, the modification of PDI with Ag2S nanoparticles by Yang et al.104 was confirmed by TEM and HRTEM characterization, revealing uniform dispersion of Ag2S on PDI surfaces with established heterojunction interfaces (Fig. 11b). The S-scheme Ag2S/PDI heterojunction (Fig. 11a) exhibited superior photocatalytic activity, achieving 94% phenol degradation within 2 hours – significantly outperforming individual Ag2S or PDI components (Fig. 11c). Due to its enhanced oxidation capability, the composite also facilitates in situ water oxidation for oxygen evolution. Chen et al.105 fabricated an organic–inorganic dual S-scheme heterojunction In2O3/PDI/In2S3 (denoted IO/PDI/IS) photocatalyst through a synergistic approach combining solvent-induced self-assembly and electrostatic driving forces. The rational design leverages complementary band structures and an intensified IEF (Fig. 11e), enabling a defect-mediated dual S-scheme charge transfer pathway within the IO/PDI/IS architecture (Fig. 11d). This configuration demonstrates exceptional efficacy in degrading recalcitrant organic pollutants, including lignin and antibiotics. Notably, the system achieved an 80.9% mineralization rate for sodium lignosulfonate (SL), highlighting its advanced oxidative capability. Recently, Li et al.106 developed an organic–inorganic S-scheme heterojunction photocatalyst by incorporating Nb5+-substituted BiVO4 (introducing oxygen vacancies, Ovs) with β-alanine-functionalized PDI supramolecules (Fig. 11f). This strategic modification reduced the bandgap and enhanced visible-light absorption. The composite demonstrated superior degradation efficiency for persistent aquatic pollutants including TC, RhB, SMX, and phenol. The enhanced photocatalytic activity stems from synergistic effects: Ovs facilitate charge separation, while interfacial Nb–O and Bi–O bonds maintain strong redox potentials (Fig. 11g).
 | ||
Fig. 11 (a) Mechanism of Ag2S/PDI. (b) The HR-TEM image of Ag2S/PDI (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6). (c) Visible light irradiation (where B-PDI represents bulk-PDI, N-PDI represents nano-PDI, and 1 0.6). (c) Visible light irradiation (where B-PDI represents bulk-PDI, N-PDI represents nano-PDI, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x represents the mass ratio of Ag2S to PDI). Reproduced from ref. 104. Copyright 2019, the Royal Society of Chemistry. (d) Schematic illustration of the charge migration between IO/IS and IO/PDI/IS. (e) The calculation of IEF intensity of IS, IO/IS and IO/PDI/IS. Reproduced from ref. 105. Copyright 2023, Wiley. (f) Schematic illustration of the facile solvothermal preparation of PNBVO composite materials. (g) Photocatalytic electron transfer mechanism of PNBVO. Reproduced from ref. 106. Copyright 2025, Elsevier. x represents the mass ratio of Ag2S to PDI). Reproduced from ref. 104. Copyright 2019, the Royal Society of Chemistry. (d) Schematic illustration of the charge migration between IO/IS and IO/PDI/IS. (e) The calculation of IEF intensity of IS, IO/IS and IO/PDI/IS. Reproduced from ref. 105. Copyright 2023, Wiley. (f) Schematic illustration of the facile solvothermal preparation of PNBVO composite materials. (g) Photocatalytic electron transfer mechanism of PNBVO. Reproduced from ref. 106. Copyright 2025, Elsevier. | ||
In general, hybridizing PDI supramolecular photocatalysts with different semiconductors to form a heterojunction structure has shown to be an effective strategy mainly through extending the light absorption for photo-excitation and reducing the recombination of photo-generated carriers. However, to realize successful and reasonable construction of PDI-based heterojunction, several aspects need to be considered: (1) matched energy band potentials, allowing effective transfer and spatial separation for charge carriers from one semiconductor to another; (2) synthetic feasibility, adapted to PDI supramolecular photocatalysts’ fabrication condition; (3) structure and activity stability, essential for their future application.
3.4. Metal deposition/doping and co-catalyst engineering
Depositing noble metals onto photocatalysts constitutes an effective approach for enhancing photocatalytic efficiency,107 primarily through Schottky or Ohmic junction formation that modulates photogenerated charge transfer dynamics. Critically, plasmonic noble metal nanoparticles (e.g., Au, Ag, Cu) exploit localized surface plasmon resonance (LSPR) effects, wherein collective electron oscillations significantly boost photon absorption capacity.108Miao et al.109 fabricated PDI@Au NPs via electrostatic adsorption, demonstrating enhanced visible-light phenol photodegradation kinetics. This performance enhancement stems from synergistic effects: (1) Au nanoparticle surface plasmon resonance (SPR). (2) Resonance energy transfer (RET) with PDI collectively broadens visible-light utilization; (3) Au's lower Fermi level facilitates efficient electron–hole separation. For another example, Liu's group110 achieved homogeneous deposition of platinum quantum dots (Pt QDs) on PDI supramolecular nanorods through a facile in situ reduction protocol (Fig. 12a). The optimized 1 wt% Pt QDs/PDI composite exhibited substantially enhanced photocatalytic activity, demonstrating 6.2-fold greater phenol degradation efficiency versus pristine PDI under visible light (Fig. 12b). Critically, the mild synthetic conditions preserved the nanorods' structural and electronic integrity. This performance enhancement stems from Pt QDs functioning as electron-shuttling mediators that efficiently capture and transfer photogenerated electrons, thereby accelerating charge separation kinetics.
 | ||
| Fig. 12 (a) Diagram for the photocatalytic mechanism of Pt QDs/PDI. (b) Photocatalytic degradation performance of Pt QD/PDI composites. Reproduced from ref. 110. Copyright 2021, the Royal Society of Chemistry. (c) The photocatalytic mechanism of the TiO2/CuPDIsm composite. (d) The photodegradation ratios of the samples toward TC. Reproduced from ref. 113. Copyright 2023, the Royal Society of Chemistry. (e) The photodegradation ratios of the samples toward MB. (f) The photocatalytic mechanism of the SA-PDI composite. (g) Amoxicillin removal performances of supramolecular catalysts in photocatalytic oxidation. Reproduced from ref. 114. Copyright 2023, Elsevier. | ||
Compared to metal deposits, metal doping enables atomic-level dispersion of active sites, preventing deactivation caused by metal particle agglomeration.111,112 This strategy significantly increases the mass-specific density of catalytically active sites in the catalyst. For instance, Liang et al.113 synthesized Cu-doped PDI supramolecules (CuPDIsm) with a 1D structure and integrated them with TiO2 to form a heterojunction photocatalyst (Fig. 12c). Cu incorporation enhanced visible-light absorption and specific surface area. Crucially, Cu2+ coordination bridges and H-type π–π stacking significantly accelerated intramolecular electron transfer within CuPDIsm. Furthermore, photoinduced electrons migrated from CuPDIsm to TiO2via interfacial hydrogen bonding and electronic coupling, promoting charge separation. Consequently, the TiO2/CuPDIsm composite exhibited exceptional visible-light photodegradation activity, achieving 89.87% tetracycline and 97.26% methylene blue removal (Fig. 12d and e). Recently, Burcu Palas et al.114 synthesized silver- and cobalt-doped PDI supramolecular photocatalysts and evaluated their efficacy for amoxicillin removal from aqueous solutions. Both bulk PDI and self-assembled PDI (SA-PDI) were functionalized with Ag or Co at 1 wt% and 10 wt% loadings (Fig. 12f). Photocatalytic reaction parameters were optimized at pH 4.6, a catalyst loading of 0.52 g L−1, and an initial amoxicillin concentration of 10.3 mg L−1. Under these visible-light conditions, the optimal system achieved 51.8% amoxicillin degradation efficiency (Fig. 12g).
3.5. Others
The hierarchical organization of PDI supramolecular systems is primarily mediated by π–π stacking, which dictates both structural integrity and optoelectronic properties. Intensified stacking interactions promote long-range π-conjugation and molecular orbital hybridization – key determinants for optimizing charge carrier mobility and separation efficiency in organic semiconductor architectures.115 Based on the π–π stacking interactions in PDI supramolecular materials, researchers have integrated PDI with complementary π-conjugated organic systems to construct a larger π–π composite system. For example, Wei et al.95 developed a π–π-stacked NDINH/PDINH supramolecular photocatalyst via rapid solution assembly. The strong intermolecular π-interactions induce a pronounced IEF and efficient charge transport. This optimized π-stacking configuration achieves full-spectrum overall water splitting with H2/O2 evolution rates of 317.2/154.8 μmol g−1 h−1 and exceptional 32-hour stability, demonstrating the pivotal role of π–π molecular engineering in photocatalyst design. Dai et al.116 fabricated a 3D PANI/PDI heterojunction photocatalyst via in situ growth. The PANI framework enhances mechanical robustness and provides abundant reactive sites/mass transport pathways. Strong π–π interactions establish an extended delocalized π-system and favorable heterojunction, significantly promoting charge carrier separation. Consequently, tetracycline degradation rates increased 15.3- and 17-fold versus pristine PDI and PANI, respectively, with sustained activity over 75 h in continuous flow.As a π-conjugated organic component, PDI supramolecular architectures can integrate with some other highly π-conjugated materials to build a π–π composite system to enhance interplanar coupling, and reduced stacking distances correlate with increased π-electron delocalization and orbital density superposition. These electronic configurations demonstrably facilitate charge carrier migration and separation, ultimately boosting photocatalytic efficiency and operational stability.
4. Photocatalytic application
Due to their high stability and narrow band gap, PDIs exhibit strong optical responses across the visible to near-infrared spectrum, enabling their widespread application in photocatalysis such as energy storage, energy conversion, and environmental protection. In this section, we present a succinct overview of their applications in photocatalytic water splitting, CO2 reduction, N2 fixation and pollutant degradation.4.1. Water splitting
Photocatalytic technology plays a significant role in developing green energy and addressing energy and environmental challenges. In recent years, photocatalytic water splitting has attracted increasing attention in the global energy and environmental crisis due to its clean and environmentally friendly characteristics.100,117–120 However, the widespread application of conventional inorganic photocatalysts (e.g., TiO2) is hindered by limitations including poor visible-light utilization, low quantum yield, and high cost.121 To overcome these constraints, the development of novel, efficient photocatalytic materials is actively pursued. Notably, PDI-based photocatalysts have rapidly emerged as a research focus, particularly for photocatalytic water splitting. Their prominence stems from exceptional light-harvesting capacity, high electron mobility, robust chemical and photochemical stability, and the unique ability to precisely tailor band structures and surface properties through molecular engineering.| Photocatalyst | Application | Light source | Amount of catalyst | Photocatalytic performance | AQY | Ref. |
|---|---|---|---|---|---|---|
| P-PMPDI | H2 production | 300 W Xe lamp, λ > 420 nm | 50 mg | 1170 μmol g−1 h−1 | 2.96 (550 nm) | 62 |
| Zn0.5Cd0.5S/PDIs | H2 production | Solar simulator (AM 1.5) | 50 mg | 1320 μmol g−1 h−1 | — | 124 |
| PDI-phthalic | H2 production | 300 W Xe lamp, λ > 420 nm | 25 mg | 1100 μmol g−1 h−1 | — | 69 |
| N-APDIs | H2 production | 500 W Xe lamp, λ > 420 nm | 20 mg | 61![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 490 μmol g−1 h−1 490 μmol g−1 h−1 |
5.9 (420 nm) | 127 |
| S-APDIs | H2 production | Hg lamp, 500 W | 20 mg | 900 μmol g−1 h−1 | — | 132 |
| g-C3N4/PDIs | H2 production | 300 W Xe lamp, λ > 420 nm | 10 mg | 1600 μmol g−1 h−1 | — | 176 |
| TATF-COF/PUP | H2 production | 350 W Xe lamp, λ > 420 nm | 5 mg | 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 μmol g−1 h−1 500 μmol g−1 h−1 |
19.7 (420 nm) | 177 |
| GQDs/PDIs | H2 production | 500 W Xe lamp, λ > 420 nm | 25 mg | 1600 μmol g−1 h−1 | 0.5 (420 nm) | 178 |
| P-PMPDIs-Zr | H2 production | 300 W Xe lamp, λ > 420 nm | 50 mg | 504![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 μmol g−1 h−1 600 μmol g−1 h−1 |
11.7 (420 nm) | 125 |
| CN-PDI | H2 production | 450 nm LED light source | 5 mg | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 μmol g−1 h−1 700 μmol g−1 h−1 |
5.8 (450 nm) | 179 |
| TiO2/PDIs | H2 production | 300 W Xe lamp, λ > 420 nm | 50 mg | 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 μmol g−1 h−1 700 μmol g−1 h−1 |
— | 180 |
| g-C3N4/Pt/PDIs | H2 production | 400 W Xe lamp, λ > 420 nm | 25 mg | 150 μmol g−1 h−1 | 0.31 (420 nm) | 181 |
| PDIs/TiO2 | H2 production | 300 W Xe lamp, λ > 365 nm | 100 mg | 1200 μmol g−1 h−1 | 70.69 (365 nm) | 182 |
| PDI-NH | O2 production | 300 W Xe lamp, (420 nm cutoff filter) | 15 mg | 40.6 mmol g−1 h−1 | 10.4 (400 nm) | 132 |
| p-Ag2S/n-PDI | O2 production | 500 W xenon lamp, 420 nm cut-off filter | 25 mg | 34 μmol g−1 h−1 | — | 104 |
| Oxamide-PDI | O2 production | 300 W full-spectrum xenon lamp, (783 mW cm−2) | 0.025 g | 5110.25 μmol g−1 h−1 | 2.15 (420 nm) | 77 |
| PDI/Co3O4/Pt | O2 production | 300 W Xe lamp, 420 nm cut-off filter | 15 mg | 24.4 mmol g−1 h−1 | 6.9 (420 nm) | 133 |
| PT-CB | O2 production | 300 W Xe lamp, (420 nm cut-off filter) | 5 mg | 966.28 μmol g−1 h−1 | — | 183 |
| 3D MXene/GO/PDI | CO2 reduction | 350 W xenon lamp for UV-vis irradiation | 10 mg | 711 μmol g−1 h−1 (HCHO) | — | 142 |
| 3%Au/PHI-PDI | CO2 reduction | 300 W Xe lamp | 30 mg | 122.65 μmol g−1 h−1(CO) | — | 143 |
| Cu@PDI(30%)-NZU67 | CO2 reduction | 300 W Xe lamp UV cut-off filter (λ > 420 nm) | 50 mg | 941.28 μmol g−1 h−1 (ETHO) | — | 147 |
| BOPDI | N2 fixation | 300 W Xe lamp (λ > 420 nm) | 20 mg | 74 μmol g−1 h−1 | 1.29 (450 nm) | 159 |
| PDIMA-2 | N2 fixation | 300 W Xe lamp (λ > 400 nm) | 20 mg | 49.9 μmol g−1 h−1 | 1.07 (420 nm) | 160 |
| 30% PDI/10H-CNv | N2 fixation | 300 W xenon lamp, simulating sunlight (AM 1.5G) | 10 mg | 519.2 μmol g−1 h−1 | — | 161 |
| 3D PANI/PDI | TC removal | 5 W LED lamp, (420 nm cut-off filter) | 25 mg | 0.5265 h−1 | — | 116 |
| CNPDI | TC removal | 250 W xenon lamp, (420 nm cut-off filter) | — | 0.026 min−1 | — | 178 |
| I-PDI/PEDOT-M | TC removal | 250 W xenon lamp | 20 mg | 0.0087 min−1 | — | 170 |
| FM88B | TC removal | 300 W xenon lamp λ > 420 nm | 7 mg | 0.067 min−1 | — | 85 |
| BN/PDI-2-350 | TC removal | Visible light, xenon lamp | 15 mg | — | — | 184 |
| PDI (5.0%)/BiOCl-BiPO4 | RhB/TC removal | Visible light irradiation | 25 mg | 0.037 min−1/0.0135 min−1 | — | 185 |
| MNP30/PDS/Vis | SMX removal | 300 W xenon lamp, cut-420 nm filter | — | 0.8873 min−1 | — | 186 |
| BWGP-2 | BPA removal | 300 W Xe lamp | — | 55% | — | 187 |
However, the photocatalytic H2 production performance of pure PDIs is severely limited by inherent challenges including unfavorable band structure, rapid charge-carrier recombination, insufficient surface active sites, and mass transfer constraints.62,125 To address these limitations, modification strategies such as side chain regulation, elemental doping, and heterojunction construction are commonly employed to significantly enhance their photocatalytic activity.
Constructing heterojunctions is a common strategy to enhance PDI-based photocatalytic hydrogen production. This approach can effectively promote the separation and transfer of photogenerated electron–hole pairs through the IEF formed at the heterojunction interface, reduce the recombination probability of carriers, and extend their lifetime. Additionally, heterojunctions can broaden the light absorption range of PDI-based materials, allowing them to harvest more solar energy and further boost the photocatalytic hydrogen production performance. For example, Zhu and co-workers126 designed a novel co-assembled material for photocatalytic H2 production through π–π stacking interactions of TPPS/PDI organic semiconductors with a D–A interface (Fig. 13a and b). The TPPS/PDIs exhibit a remarkable photocatalytic H2 production rate of 30.36 mmol g−1 h−1, attributed to its pronounced IEF that facilitates efficient charge separation and a giant interfacial potential gradient that extends the lifetime of photoexcited carriers. To broaden the absorption spectrum and enhance the separation of photoinduced charges, Yang et al.104 reported the fabrication of p-Ag2S/n-PDIs via a synergistic strategy combining hydrogen bonding, π–π stacking interactions, and a two-step electrostatic self-assembly process (Fig. 13c). Ag2S enhances π–π orbital overlap in PDI assemblies, facilitating directional photoelectron transport along the 1D π-conjugated pathways. Concurrently, Ag2S extends the visible-light harvesting capacity, thereby promoting photochemical conversion efficiency. Finally, p-Ag2S/n-PDIs heterojunctions exhibit superior ultraviolet-light, visible-light and full spectrum photocatalytic performance.
 | ||
| Fig. 13 (a) The molecular formula and electrostatic potential distribution of TPPS and PDI. (b) Left: The frontier molecular orbital distribution of TPPS and PDI at the interface; right: schematic diagram of interfacial interactions of co-assembly supramolecular TPPS/PDI. Reproduced from ref. 126. Copyright 2022, Wiley. (c) Schematic illustration of the synthesis of the self-assembled PDI and Ag2S/PDI composite. Reproduced from ref. 104. Copyright 2019, the Royal Society of Chemistry. (d) Proposed electron transfer mechanism of supramolecular R-APDI for photocatalytic H2 production. Reproduced from ref. 127. Copyright 2022, Elsevier. | ||
In the context of the water splitting reaction mechanism, the suppression of electron–hole recombination emerges as a pivotal factor for enhancing photocatalytic activity. Additionally, both metal and non-metal element doping strategies have been demonstrated to effectively boost the photocatalytic H2 production performance of PDIs, thereby offering promising avenues for optimizing their catalytic efficiency in energy conversion systems. For example, the metal-doped supramolecular P-PMPDI-Zr125 exhibited exceptional hydrogen evolution activity under visible light irradiation, achieving a remarkable rate of 50.46 mmol g−1 h−1, which is 4.34 times higher than that of the Zr-free cationic counterpart. Notably, P-PMPDI-Zr maintains substantial photocatalytic activity even at longer wavelengths, demonstrating an apparent quantum yield (AQY) of 11.70% at 630 nm, along with excellent stability. These results highlight that metal doping serves as a facile and effective strategy to broaden the absorption spectrum and enhance charge transfer in supramolecular systems. Furthermore, doping with Co, Ni, and Cu also significantly improves the HER performance compared to pristine P-PMPDI, further validating the universality of this approach. Xu et al.127 successfully incorporated non-metallic elements N, S, and Se into PDIs (polyimide derivatives), yielding N-APDI, S-APDI, and Se-APDI samples. These heteroatom-annulated PDI supramolecules exhibited a substantially higher H2 production compared to non-annulated APDI counterparts. Mechanistically, this enhanced photocatalytic activity is attributed to two key factors (Fig. 13d). The incorporation of heteroatoms strengthens the molecular dipole moment, thereby intensifying the IEF to facilitate the separation and migration of photogenerated charge carriers. Simultaneously, heteroatom annulation generates additional active sites that optimize the hydrogen evolution reaction (HER) kinetics, promoting more efficient H2 production.
The fabrication of a PDI heterojunction also presents a dual-pronged advantage: it enhances the efficiency of charge separation and maintains the strong redox capability of photocatalysts, thereby realizing efficient photocatalytic hydrogen generation. For example, Yu and co-workers128 successfully designed an inorganic–organic S-scheme heterojunction. As illustrated in Fig. 14a, ZIS nanosheets are grown onto the disc-shaped MIL-125-PDI surface, forming unique hollow nanodiscs with a hierarchical architecture. This configuration endows the material with abundant surface active sites, a tailored electronic structure, and a spatially segregated redox interface. Experimental results and theoretical calculations consistently indicate that the staggered band alignment and work function disparity between MIL-125-PDI and ZIS give rise to the formation of an IEF. This electric field, in turn, governs the pathways of charge transfer and consequently improves the efficiency of charge separation (Fig. 14b).
 | ||
| Fig. 14 (a) Synthetic route for MIL-125-PDI/ZIS. (b) Schematic of the proposed mechanism of charge transfer over MIL-125-PDI/ZIS under simulated sunlight irradiation. Reproduced from ref. 128. Copyright 2024, Wiley. | ||
Heterojunction engineering represents a key strategy for establishing robust IEFs, which drive directional charge migration to overcome kinetic bottlenecks in photocatalytic O2 evolution by spatially isolating redox sites. For instance, Yang et al.104 successfully constructed an efficient full-spectrum responsive p-Ag2S/n-PDI heterojunction with a photocatalytic O2 production rate of about 34.6256 mmol g−1 h−1 (Fig. 15a and b). The enhanced photocatalytic performance can be primarily ascribed to several key factors facilitated by Ag2S: firstly, it optimizes the π–π stacking degree within PDI, significantly improving the mobility of photo-generated electrons along the quasi-one-dimensional stacking channels. Secondly, Ag2S broadens light absorption, thereby boosting the conversion efficiency of light into chemical energy. Furthermore, the intrinsic IEF formed at the Ag2S/PDI interface favors the efficient separation of photo-induced charge carriers. This synergistic effect, coupled with the formation of a p-Ag2S/n-PDI heterojunction, generates a greater quantity of active species compared to pristine PDI, ultimately leading to a substantially enhanced oxidation capability.
 | ||
| Fig. 15 (a) The synthesis of the self-assembled PDI and Ag2S/PDI composite. (b) Amount of O2 evolved by Ag2S/PDI. Reproduced from ref. 104. Copyright 2019, the Royal Society of Chemistry. (c) XRD spectrum of PDI-NH. (d) Schematic illustration of PDI-NH. Reproduced from ref. 132. Copyright 2022, Wiley. (e) Amount of O2 evolved by oxamide-PDI. Reproduced from ref. 77. Copyright 2023, Wiley. (f) AQY, (g) relative IEF and (h) amount of O2 evolved by PDI/Co3O4/Pt. Reproduced from ref. 133. Copyright 2023, American Chemical Society. | ||
Nowadays, enhancing the crystallinity of PDIs during their molecular assembly via non-covalent interactions represents a critical strategy for boosting photocatalytic efficacy. This crystalline ordering facilitates efficient charge carrier transport by augmenting the intrinsic IEF. For example, Zhu et al.132 synthesized a highly crystalline PDI supramolecular photocatalyst (PDI-NH) via an imidazole solvent method (Fig. 15c). The catalyst exhibits a breakthrough oxygen evolution rate with high apparent quantum yield, which is 1353 times higher than the low crystalline PDI-NH (Fig. 15d). This crystallinity originates from ordered self-assembly via π–π stacking and hydrogen bonding within the molten imidazole.
PDI supramolecular assembly, held together by weak non-covalent interactions, often suffer from poor structural stability. Therefore, designing PDI polymers is another efficient strategy to improve the O2 production rate. Liu's group77 synthesized a highly-crystallized linear conjugated polymer, Oxamide-PDI, by alternating copolymerization of PTCDA with hydrophilic oxamide. The more extended π-conjugation of perylene-cored PTCDA endows oxamide-PDI with a larger value of interaction energy for π–π stacking. Featuring a planar perylene core with extended conjugation and a polar dicarbonyl bridging group, oxamide-PDI formed rigid 1D ordered stacks. This structure endowed exceptional π–π stacking-mediated exciton splitting and robust intramolecular charge transfer capabilities through the bridging group. Notably, the highly crystalline oxamide-PDI achieved remarkable solar-driven O2 evolution efficiency under mild pH conditions without co-catalysts (Fig. 15e).
A notable aspect of the aforementioned research is its demonstration of efficacy in the absence of oxygen evolution reaction (OER) co-catalysts. Nevertheless, the strategic incorporation of such co-catalysts remains highly advantageous, as they facilitate the extraction and trapping of photogenerated charges, furnish additional redox-active sites, and reduce both the reaction overpotential and activation energy barrier associated with surface oxygen evolution. Consequently, the strategic design of efficient semiconductor/co-catalyst composite materials represents an essential approach for enhancing photocatalytic performance in the OER. Li et al.133 constructed a dual cocatalysts-modified PDI polymer (PDI/Co3O4/Pt) for facilitating photocatalytic O2 evolution performance to improve the solar utilization. The Co3O4, acting as superior active sites, contributed to lower the barrier of the water oxidation reaction, and the IEFs of cocatalysts and PDI drive separation and transfer of photogenerated charges. As a consequence, DI/Co3O4/Pt exhibited strong stability and a photocatalytic O2 evolution rate of 24.4 mmol g−1 h−1, which is 5.4 times higher than that of pure PDI. The apparent quantum yield of the O2 evolution reaction reaches 6.9% at 420 nm and remains 1.2% at 590 nm. In the system, Co3O4 provides the dominant effects for photocatalytic O2 evolution reactions, and Pt mainly plays a role in charge transfer (Fig. 15f and g).
4.2. CO2 reduction
The photocatalytic conversion of CO2 and water into chemicals and fuels using light energy, emulating natural photosynthesis, is considered to be among the most promising methods for decreasing atmospheric CO2 levels.134–137 Induced photosynthesis via the photocatalytic CO2 reduction reaction (CO2RR) to value-added chemicals is a long-lasting way to address energy and ecological problems.138–140 The oxidative half-reaction in photocatalytic systems predominantly involves water oxidation to generate oxygen (O2) or hydrogen peroxide (H2O2). Despite the inherent merits of CO2 photocatalytic conversion technology, its practical implementation confronts significant challenges. It is still a multi-step process demanding the concurrent fulfilment of both thermodynamic and kinetic criteria. Thermodynamically, the redox potentials of the reaction intermediates must align favorably with the band edge positions of the photocatalyst. Kinetically, the substantial energy barriers associated with multi-electron transfer processes must be overcome. Consequently, an efficient photocatalyst for the overall reduction of CO2 necessitates both exceptional photogenerated charge separation efficiency and appropriately positioned conduction band minimum and valence band maximum to drive the reduction and oxidation half-reactions, respectively. Therefore, developing highly active photocatalytic systems is crucial for enhancing the reaction rate of photocatalytic CO2 reduction. Compared to inorganic semiconductors, organic semiconductors offer distinct advantages, including chemically tunable optoelectronic properties, robust photochemical and thermal stability, adjustable band energy levels, flexible morphological and structural design, facile synthetic modulation, and elemental abundance. Among diverse organic semiconductors, PDI exhibits a broad spectral response range, environmental benignity, and low cost, leading to its widespread application in fields such as fluorescent probes, sensors, transistors, and photocatalytic systems.For example, Hu and co-workers141 engineered ZrO2-supported PDI photosensitizers via salicylic acid anchors (Fig. 16a). Coupled with Re(bpy)(CO)3Cl/TEOA in DMF, the composite demonstrated competitive CO2-to-CO TONs under white LED (100 mW cm−2), attributed to stable surface grafting and facilitated electron transfer at minimal catalyst usage (Fig. 16b and c). The strong anchoring of salicylic acid on the surface of ZrO2 and efficient electron transfer at low catalyst concentrations make ZrO2/PDIs a promising candidate for CO2 photoreduction applications.
 | ||
Fig. 16 (a) Strategy for photogeneration of a potent and long-lived ZrO2(e−). (b) TON of CO and H2 by Re(bpy)(CO)3Cl and ZrO2|PDI as a function of irradiation time. (c) Photocatalytic CO2 reduction TON under 520 nm irradiation or broad band irradiation with wavelengths greater than 500 nm. Reproduced from ref. 141. Copyright 2022, American Chemical Society. (d) Schematic illustration of the MXene/GO/PDI composite aerogels. (e) MF generation yield of samples changing over time under the irradiation for 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) h. (f) MF generation rate of samples under the irradiation for 4 h. (f) MF generation rate of samples under the irradiation for 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) h. Reproduced from ref. 142. Copyright 2022, Elsevier. (g) Proposed photocatalytic mechanism for Au/PHI-PDI. (h) Time-dependent CO evolution performances. (i) Stability test over the 3%Au/PHI-PDI. Reproduced from ref. 143. Copyright 2025, Elsevier. (j) Proposed photocatalytic mechanism for Cu@PDI(30%)-NZU67. (k) Photocatalytic activity: Cu@PDI(30%)-NZU67 (50 mg), irradiation wavenumber = 420 nm, TEOA (0.3 M), and MeCN/deionized H2O (4 h. Reproduced from ref. 142. Copyright 2022, Elsevier. (g) Proposed photocatalytic mechanism for Au/PHI-PDI. (h) Time-dependent CO evolution performances. (i) Stability test over the 3%Au/PHI-PDI. Reproduced from ref. 143. Copyright 2025, Elsevier. (j) Proposed photocatalytic mechanism for Cu@PDI(30%)-NZU67. (k) Photocatalytic activity: Cu@PDI(30%)-NZU67 (50 mg), irradiation wavenumber = 420 nm, TEOA (0.3 M), and MeCN/deionized H2O (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v). (l) AQE results of Cu@PDI(30%)-NZU67. Reproduced from ref. 147. Copyright 2024, Elsevier. 1 v/v). (l) AQE results of Cu@PDI(30%)-NZU67. Reproduced from ref. 147. Copyright 2024, Elsevier. | ||
While the VB position of PDI offers a strong oxidation capability, the positive CB potential results in an insufficient thermodynamic driving force for reduction. Furthermore, rapid recombination of photogenerated charge carriers significantly limits the practical application of PDI. Consequently, PDI-based heterojunction engineering overcomes this limitation through two synergistic mechanisms: (1) tailored energy band alignment and (2) optimized photogenerated carrier separation, ultimately boosting CO2-to-fuel conversion efficiency when utilizing H2O as the reductant. For example, Wu et al.142 fabricated novel 3D MXene/GO/PDI aerogels via self-assembly (Fig. 16d), establishing an electron transfer network through π–π stacking that enhanced electron delocalization. The optimized aerogel achieved a formaldehyde (HCHO) production rate of 771.1 μmol g−1 h−1 in photocatalytic CO2 reduction, an 8-fold enhancement over MXene/PDI (Fig. 16e and f). Characterization confirmed a strongly coupled interface via π–π cross-linking, generating a robust IEF and narrowed bandgap. Concurrently, a Z-scheme heterojunction formed between MXene/GO and PDI due to their aligned band structures. This dual configuration accelerated photogenerated electron transfer and enhanced interfacial redox capabilities.
Non-covalent heterojunctions, characterized by the absence of robust chemical bonding, typically exhibit compromised interfacial carrier transport efficiency. This limitation adversely impacts photocatalytic CO2 reduction performance. In contrast, covalently bonded heterojunctions feature strong covalent linkages between semiconductor components, providing directional charge transfer channels that enhance photocatalytic activity. Yang et al.143 constructed Au/PHI-PDI with synergistic junctions: (1) covalent S-scheme heterojunction enabling rapid intralayer charge transfer via PHI-PDI bonds, and (2) Schottky junction creating vertical IEF for interlayer charge transport (Fig. 16g). The covalent connection between PHI and PDI constructs a fast charge transfer channel, which is beneficial to boost intralayer charge separation and migration. Furthermore, the formed Schottky junction could generate a vertical IEF, which enhances interlayer charge transport (Fig. 16h and i). As a result, this architecture boosted charge utilization efficiency, yielding 122.65 μmol g−1 h−1 CO over 3%Au/PHI-PDI surpassing PHI-PDI and PHI by factors of 2.77 and 9.24 respectively.
Currently, the integration of PDI with organic frameworks has also begun to be investigated in the field of photocatalytic CO2 reduction, such as in metal–organic frameworks (MOFs), covalent organic frameworks (COFs), etc. For example, research on MOFs containing PDI has focused on applications in sensing144 and photocatalytic degradation,145,146 and few studies have studied their potential for CO2 conversion. Recently, Altalbawy and co-workers147 incorporated a PDI chromophore moiety into a Cu@PDI(30%)-NZU67 and applied the resulting material for CO2 conversion of ETHO for the first time (Fig. 16j–l). From a MOF engineering perspective, anchoring PDI within secondary building units (SBUs) proved strategically superior to conventional linker-based integration. For another, Zhu and colleagues148 developed a cobalt-metalated, one-dimensional covalent organic framework with ABC stacking (PP-COF-Co). This framework integrates PDI as a photosensitizing unit and 1,10-phenanthroline moieties as metal-coordinating sites which exhibit a 57-fold increase in photocatalytic CO2 reduction activity compared to its pristine analogue.
4.3. N2 fixation
Ammonia (NH3) serves as an essential industrial chemical for manufacturing explosives and fertilizers, underpinning critical societal infrastructure.149–151 Conventionally, industrial-scale NH3 production employs the Haber–Bosch process, where Fe-based catalysts mediate N2 and H2 conversion under elevated temperatures and pressures,152–155 with an annual global energy consumption of ∼2% and responsibility for 1.6% of anthropogenic CO2 emissions.156,157 Photocatalytic nitrogen reduction reaction (PNRR) represents an emerging sustainable alternative to the conventional Haber–Bosch process. However, the exceptionally strong nonpolar N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N triple bond in N2 molecules possesses significant challenges for efficient photocatalytic fixation.158 Furthermore, existing photocatalysts exhibit sluggish charge transfer kinetics, resulting in unsatisfactory N2 fixation activity. Therefore, developing green and efficient ammonia synthesis technologies is essential for sustainable global development.
N triple bond in N2 molecules possesses significant challenges for efficient photocatalytic fixation.158 Furthermore, existing photocatalysts exhibit sluggish charge transfer kinetics, resulting in unsatisfactory N2 fixation activity. Therefore, developing green and efficient ammonia synthesis technologies is essential for sustainable global development.
PDI represents a prototypical n-type organic semiconductor among diverse organic semiconductor materials, which is famous for its exceptional visible-light absorption capacity and robust chemical stability with great potential in photocatalytic N2 fixation. In the study conducted by Yang and coworkers,159 notably, the BOPDI photocatalyst achieved an NH3 production rate of 74.0 μmol g−1 h−1 without sacrificial agents or cocatalysts, representing an 11-fold enhancement over conventional PDIs. The apparent quantum yield (AQY) for NH3 generation reached 1.29% under 450 nm monochromatic irradiation. Mechanistic studies attribute this performance to the intensified IEF, which directs electron migration toward embedded catalytic units while localizing photoinduced holes on benzene/perylene moieties (Fig. 17a–c). This spatial charge separation creates long-lived intermediate states: electrons activate N2 to form NH3, while holes drive H2O oxidation to O2. This work constitutes the first demonstration of PDI-based composites for photocatalytic N2 fixation to ammonia. In another study, Wang et al.160 established a facile solution-phase self-assembly strategy to generate IEFs in PDI-triazine-based polymers (Fig. 17d). The optimized PDIMA-2 photocatalyst achieves exceptional nitrogen fixation performance (49.90 μmol g−1 h−1) without co-catalysts or sacrificial agents – surpassing pristine PDI by ∼10-fold and outperforming carbon materials (Fig. 17e and f). Notably, significant NH3 production persists under 750 nm monochromatic irradiation. Extended near-infrared absorption originates from triazine-PDI π–π stacking-induced LUMO–HOMO orbital overlap. Mechanistic studies reveal the IEF arises from face-to-face molecular dipoles and π-stacking arrangements, dramatically enhancing carrier separation/migration. Recently, Cui and his co-workers161 constructed Z-scheme PDI/10H-CNv heterojunctions via in situ condensation (Fig. 17g). Nitrogen vacancies and curled pores in 10H-CNv boosted N2 adsorption sites, while the enhanced IEF directed electron–hole separation. This synergy achieved 519.2 μmol g−1 h−1 NH3 and 135.9 μmol g−1 h−1 (Fig. 17h and i).
 | ||
| Fig. 17 (a) Plausible mechanism for the photocatalytic N2 fixation over BOPDI under visible light irradiation. (b) NH3 evolution rates over the as-prepared BOPDI catalysts. (c) Apparent quantum efficiency (AQY) of BOPDI. Reproduced from ref. 159. Copyright 2022, Elsevier. (d) Schematic diagram of the synthetic route. (e) Ammonia synthesis rates over different samples. (f) Diagram of the photocatalytic nitrogen fixation mechanism over PDIMA-2. Reproduced from ref. 160. Copyright 2022, Elsevier. (g) Direct Z-scheme heterojunction of 10H-CNv. (h) Average NH4+ yields for different photocatalysts. (i) Gibbs free energy diagrams of the photocatalytic reduction of ammonia by N2 in 30% PDI/10H-CNv composites. Reproduced from ref. 161. Copyright 2024, Elsevier. | ||
4.4. Pollutant degradation
Photocatalysis represents a crucial, economical, and effective strategy for addressing environmental pollution.162–164 Organic semiconductors serve as promising photocatalysts due to their facile synthesis, low cost, tunable functionalization, earth abundance, and robust photochemical stability. PDI, a conventional organic semiconductor widely used in dyes, solar cells, and optoelectronic devices, has recently gained significant attention for photocatalytic applications. In environmental remediation, PDI-based nanocomposites effectively degrade diverse aqueous organic pollutants including antibiotics and phenolic compounds (Table 3). Theoretical calculations reveal PDI's frontier molecular orbitals: LUMO and HOMO energies derive from carbon and oxygen atomic orbitals, with nitrogen atoms acting as nodal points in π-orbital wavefunctions. Consequently, PDI's electronic structure primarily depends on π–π stacking interactions.165 When a PDI-based photocatalyst is excited under illumination, photogenerated electrons in the CB reduce adsorbed O2 to generate strongly oxidizing superoxide radicals (˙O2−). Simultaneously, VB holes migrate to the material surface, producing hydroxyl radicals (˙OH) and directly participating in oxidation. These reactive species (˙O2−, ˙OH, and h+) subsequently mineralize organic pollutants into non-toxic inorganic compounds and low-toxicity small molecules.Antibiotics and pharmaceutical metabolites accumulating in aquatic environments represent persistent ecological threats due to structural complexity and degradation resistance.166–169 Conventional water treatment technologies fail to ensure complete elimination. Heterojunction photocatalysts incorporating PDI offer transformative solutions through extended spectral utilization and enhanced carrier separation efficiency. Current advances include strategically designed type-II, Z-scheme, and S-scheme architectures via controlled integration of PDI with complementary semiconductors, such as g-C3N4, metal–organic frameworks, and metal oxides. These designs not only broaden the light absorption range, but also markedly enhance photocatalytic activity through interfacial charge transfer mechanisms. This section systematically reviews the latest advancements in PDI-based heterojunctions for the degradation of antibiotics and drug residues, elucidates the underlying mechanisms driving performance enhancement, and explores potential directions for future technological optimization.
For example, an imprinted PDI/PEDOT type-II heterojunction photocatalyst film (I-PDI/PEDOT-M) was engineered via N-methylpyrrolidone (NMP)-induced surface self-corrosion assisted rapid spin-coating170 (Fig. 18a). Under 1 h visible light irradiation, the degradation efficiencies for tetracycline (TC) and ciprofloxacin (CIP) reached 73.7% and 5.0% respectively. Notably, TC degradation efficiency exceeded CIP by 14.65-fold (Fig. 18b). This pronounced divergence originates from the type-II heterojunction between PDI and PEDOT, which enables directional carrier separation that sustains superior photocatalytic activity in I-PDI/PEDOT-M. Mao et al.171 successfully synthesized a PDI/WO3/α-Fe2O3, PWF composite photocatalyst with a dual Z-scheme heterojunction via the pulsed laser ablation in liquid (PLAL) technique (Fig. 18c). Photocatalytic performance evaluation revealed that the PWF composite achieved 94.2% TC removal efficiency under 180 min of irradiation using a 15 W low-pressure mercury lamp (λ = 254 nm) (Fig. 18d). The enhanced photocatalytic activity can be primarily attributed to two key factors: (1) dual mechanisms drive performance enhancement; (2) augmented adsorption capacity via site density/surface charge optimization; (3) band-engineered dual Z-scheme heterojunction enabling synergistic photoconversion-charge separation efficiency gains.
 | ||
| Fig. 18 (a) Synthesis process schematic of I-PDI/PEDOT-M. (b) The photodegradation rate of TC and CIP by different powder materials. Reproduced from ref. 170. Copyright 2023, Elsevier. (c) Flow chart of pulsed laser preparation of PDI/WO3/α-Fe2O3 composites. (d) Total removal rate of TC in different samples. Reproduced from ref. 171. Copyright 2025, Elsevier. (e) Schematic illustrations of the PDIBr/A10 photocatalytic mechanism. (f) Photodegradation efficiency of A10 base composites about BPA. Reproduced from ref. 172. Copyright 2024, Elsevier. (g) Photocatalytic degradation mechanism of rhodamine B over PDISA/AgBr composite under visible light. (h) Degradation curves of RhB. Reproduced from ref. 175. Copyright 2024, Elsevier. | ||
Besides antibiotics, PDI-based photocatalysts can also be employed for the degradation of phenols. In a related study, through solvent-exchange self-assembly, Zha et al.172 constructed Z-scheme PDIBr/TiO2(A10) heterojunctions. The composite demonstrated dual-mode BPA degradation: 71.04% (sacrificial-agent-free) vs. 71.7% (persulfate-assisted) under visible light (Fig. 18e). The composite demonstrated dual-mode BPA degradation of 71.04% (sacrificial-agent-free) vs. 71.7% (persulfate-assisted) under visible light (Fig. 18f). Critical enhancement derives from interfacial H-aggregates with π–π orbital overlap that enable directional charge transport and inhibit recombination. Spectroscopic evidence verifies the Z-scheme mechanism as the principal BPA degradation route.
In addition to phenols and antibiotics, PDI-based materials can also be used to degrade other pollutants, such as rhodamine b, methylene blue and methyl orange etc. Zhang et al.173 successfully fabricated PDI/BiO2-type-II heterojunctions through ultrasonic-assisted self-assembly technology. The materials exhibited remarkable photocatalytic performance under visible light irradiation, achieving a remarkable degradation efficiency. The key factor behind the enhanced performance was the IEF in type-II heterojunctions, which effectively drives the spatial separation of photogenerated carriers. Zhang et al.174 achieved efficient synthesis of PDI/Bi2O4 type-II heterojunction photocatalysts using water bath heating coupled with ultrasonic dispersion. Remarkably, the 5% PDISA/Bi2O4 material degraded 98.6% of RhB and 97.0% of methylene blue within 25 min under visible light. The key mechanism involved an IEF formed at the heterojunction interface, which critically accelerated the separation and migration of photogenerated carriers, leading to superior photocatalytic performance. Furthermore, Xu et al.175 fabricated PDISA/AgBr type-II organic–inorganic heterojunctions via chemical co-precipitation (Fig. 18g). Attributed to the synergistic effects of type-II heterojunctions – enhancing photogenerated carrier separation and broadening light absorption – the PDISA/AgBr-40 composite achieved 97.8% RhB degradation in 20 min under visible light (Fig. 18h).
5. Conclusions and outlook
This review comprehensively examines recent advancements in enhancing the photocatalytic performance of PDI-based composites through strategic structural engineering and functionalization. Following an analysis of the fundamental molecular architecture and electronic properties of PDI, we discuss several synthetic methods for fabricating PDI-based photocatalysts, correlating their physicochemical properties with performance metrics. Subsequently, we systematically evaluate the deployment of these materials across diverse photocatalytic applications, with particular emphasis on mechanistic insights governing charge transfer pathways. Due to their tunable band structures and exceptional photostability, PDI-based systems demonstrate significant promise in renewable energy conversion and environmental remediation. Notwithstanding these merits, persistent challenges in carrier recombination kinetics, scalability, and long-term stability necessitate still further investigation to realize their full technological potential. Specifically, as follows:(1) PDI-based heterojunctions are predominantly assembled through electrostatic assembly, covalent/non-covalent conjugation, or semiconductor surface adsorption. Nevertheless, progressive disruption of interfacial chemical integrity during extended photocatalytic operation compromises charge transfer kinetics and catalytic efficacy. Strategic reinforcement of these interfaces through rational engineering constitutes an essential research priority to ensure operational longevity and practical deployment of PDI heterojunction systems.
(2) Further exploration of PDI's surface/structural properties is essential to optimize its optical and photocatalytic performance. Unique morphologies, exemplified by high-surface-area PDI nanosheets, offer advantages including large specific surface area, reduced charge recombination, and enhanced light utilization. While conventional nanostructures (nanosheets, nanowires, nanorods) are well-understood, high-performance configurations integrating elevated specific surface area with enhanced active site exposure – exemplified by quantum dots, hollow tubes, and hollow spheres – necessitate further exploration.
(3) Current PDI-based composites predominantly absorb visible light, while the near-infrared (NIR) region (43% of solar spectrum) remains underutilized. Substantial research efforts should therefore focus on developing advanced PDI photocatalysts capable of effective NIR light harvesting to enhance solar energy conversion efficiency.
(4) Advancing the rational design and functional efficacy of PDI-based photocatalysts necessitates a comprehensive mechanistic elucidation of their photocatalytic processes. Beyond fundamental charge carrier dynamics, rigorous investigation into the thermodynamics and microkinetics of surface-mediated catalytic reactions is imperative. State-of-the-art in situ spectroscopic characterization coupled with first-principles computational modelling provides indispensable tools for such fundamental inquiries. These methodologies will unravel critical structure–function relationships, ultimately enabling the precision engineering of PDI-based photocatalysts with exceptional quantum efficiency and reaction specificity.
(5) The established synthetic routes for PDI monomers and polymers remain procedurally intricate, cost-intensive, associated with environmental risks toxic modifiers/solvents, and poor reactor compatibility due to aggregation-induced clogging. To address these challenges, recent advances employ biocompatible functionalization and solvent-free synthesis to minimize toxicity precluding scalable industrial manufacturing and e.g., 3D-printed monoliths to enhance dispersibility. These synergistic strategies achieve cost-effective, environmentally benign (OECD-compliant), and industrially adaptable PDI systems, demonstrating scalable potential for environmental remediation and energy conversion applications.
(6) Current research on PDI supramolecular photocatalysts remains predominantly confined to laboratory-scale investigations. Translational implementation for authentic environmental remediation scenarios merits prioritized exploration.
(7) The growing adoption of additive manufacturing in catalyst engineering enables precise reconfiguration of macroscopic architectures, offering unprecedented control over mass transport and light harvesting dynamics. Integrating 3D printing technologies with molecularly tailored PDI systems constitutes a promising frontier for designing spatially programmable photocatalysts – where supramolecular organization and reactor geometry synergistically optimize quantum efficiency and scalability.
The future development of PDI-based photocatalysis presents both transformative opportunities and critical challenges. This review establishes a framework for designing high-performance PDI systems, emphasizing four key metrics, i.e., photocatalytic activity, structural stability, reaction selectivity, and visible-light harvesting capacity. Deeper integration of theoretical simulations with experimental validation will accelerate mechanistic understanding and material innovation. With sustained interdisciplinary efforts, PDI-based photocatalysts are poised to enable paradigm-shifting applications in sustainable chemistry.
Author contributions
Initiation and conceptualization: G. L. and P. W. Methodology and formal analysis: Y. X., Z. C., and X. L. Investigation: Y. X., G. D., X. W., and Z. W. Funding acquisition: G. L. Project administration: G. L. Supervision: G. L., and P. W. Writing – original draft: Y. X., Z. C., and X. L. Writing – review and editing: G. L. and P. W.Conflicts of interest
The authors declare that they have no competing interests.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 52203110), the Youth Talent Support Program of China Association for Science and Technology (Grant No. 2023QNRC0692), and the Natural Science Foundation of Fujian (Grant No. 2024J01403).References
- J. Zhang, D. Yan, G. Ding, X. Wang, C. Li, S. Zhong, Y. Yu, L. Shuai and G. Liao, Angew. Chem., Int. Ed., 2025, e202511448, DOI:10.1002/anie.202511448.
- G. Liao, E. Sun, E. B. G. Kana, H. Huang, I. A. Sanusi, P. Qu, H. Jin, J. Liu and L. Shuai, Carbohydr. Polym., 2024, 341, 122351 CrossRef PubMed.
- G. Ding, J. Zhang, D. Yan, Y. Yu, L. Shuai, L. Chen and G. Liao, Nano Lett., 2025, 25, 8984–8992 CrossRef CAS.
- J. Zhang, Y. Yang, G. Ding, Z. Wang, P. Wang, C. Li and G. Liao, Chem. Eng. J., 2025, 505, 159165 CrossRef CAS.
- S. Liu, Y. Guo, S. Yi, S. Yan, C. Ouyang, F. Deng, C. Li, G. Liao and Q. Li, Sep. Purif. Technol., 2023, 307, 122727 CrossRef CAS.
- J. Fang, M. Chen, X. Wang, Z. Huang, S. Zhao, P. Wang, Q. Li and G. Liao, Chem. Eng. J., 2025, 516, 164208 CrossRef CAS.
- C. Li, G. Ding, P. Wang, K. Liu, B. Yang and G. Liao, Dalton Trans., 2025, 54, 889–897 RSC.
- Y. Liu, S. Huang, X. Huang and D. Ma, Mater. Horiz., 2024, 11, 1611–1637 RSC.
- Y. Ahmed, K. R. Dutta, P. Akhtar, M. A. Hossen, M. J. Alam, O. A. Alharbi, H. AlMohamadi and A. W. Mohammad, Beilstein J. Nanotechnol., 2025, 16, 264–285 CrossRef CAS PubMed.
- C. Li, H. Lu, G. Ding, Q. Li and G. Liao, Catal. Sci. Technol., 2023, 13, 2877–2898 RSC.
- Z. Wang, G. Ding, J. Zhang, X. Lv, P. Wang, L. Shuai, C. Li, Y. Ni and G. Liao, Chem. Commun., 2024, 60, 204–207 RSC.
- M. Devaraj and X. Zhou, Coord. Chem. Rev., 2025, 532, 216509 CrossRef CAS.
- G. Liao, Y. He, H. Wang, B. Fang, N. Tsubaki and C. Li, Device, 2023, 1, 100173 CrossRef.
- C. Li, B. Cheng, H. Lu, G. Ding, Z. Jiang and G. Liao, Inorg. Chem., 2023, 62, 6843–6850 CrossRef CAS.
- G. Liao and M. Wu, Innovation Energy, 2024, 1, 100047 CrossRef CAS.
- M. Faraji, M. Yousefi, S. Yousefzadeh, M. Zirak, N. Naseri, T. H. Jeon, W. Choi and A. Z. Moshfegh, Energy Environ. Sci., 2019, 12, 59–95 RSC.
- T. Luo, L. Gilmanova and S. Kaskel, Coord. Chem. Rev., 2023, 490, 215210 CrossRef CAS.
- C. Li, N.-Y. Huang, Y. Yang, Q. Xu and G. Liao, Coord. Chem. Rev., 2025, 524, 216292 CrossRef.
- W. Zhang, C. Shu, H. Cui, Q. Wan, C.-T. Au, B. Yi and H. Yang, Macromol. Rapid Commun., 2023, 44, 2300012 CrossRef PubMed.
- Y. Guo, B. Liu, J. Zhang, G. Wang, C. Pan, H. Zhao, C. Wang, F. Yu, Y. Dong and Y. Zhu, Appl. Catal., B, 2024, 340, 123217 CrossRef.
- W. Che, C. Sun, Z. Wu, Y. Sun and Q. Shang, J. Cleaner Prod., 2024, 453, 142235 CrossRef.
- V. Adepu, M. Tathacharya, R. S. Fernandes, A. Tiwari, S. Siraj, S. Kanungo, N. Dey and P. Sahatiya, Adv. Mater. Technol., 2023, 8, 2201633 CrossRef.
- N. Fabre, T. Fukaminato, I. Ikariko, L. Chocron, A. Brosseau and R. Métivier, Adv. Opt. Mater., 2024, 12, 2400452 CrossRef.
- Y. Ma, C. Hao, Z. Ning, F. Zhang, J. Cui, T. Jiang and Z. Shi, Inorg. Chem. Commun., 2024, 166, 112694 CrossRef.
- Y. H. Koo, Y. Tsutsui, M. Omoto, Y. Yomogida, K. Yanagi, Y. K. Kato, M. A. Hermosilla-Palacios, J. L. Blackburn and S. Seki, J. Phys. Chem. Lett., 2025, 16, 3232–3239 CrossRef PubMed.
- D. Powell and L. Whittaker-Brooks, Mater. Horiz., 2022, 9, 2026–2052 RSC.
- F. Brust, O. Nagler, K. Shoyama, M. Stolte and F. Würthner, Adv. Opt. Mater., 2023, 11, 2202676 CrossRef.
- R. Gerdes, D. Wöhrle, W. Spiller, G. Schneider, G. Schnurpfeil and G. Schulz-Ekloff, J. Photochem. Photobiol. A: Chem., 1997, 111, 65–74 CrossRef CAS.
- Y. Xiao, G. Ding, J. Tao, Z. Wang, Z. Chen, L. Chen, L. Shuai and G. Liao, Nat. Commun., 2025, 16, 7476 CrossRef CAS.
- H. Li, C. Wang, X. Bai, X. Wang, B. Sun, D. Li, L. Zhao, R. Zong and D. Hao, Mater. Chem. Front., 2020, 4, 2673–2687 RSC.
- Q. Zhang, L. Jiang, J. Wang, Y. Zhu, Y. Pu and W. Dai, Appl. Catal., B, 2020, 277, 119122 CrossRef CAS.
- B. Yang, L. Lu, S. Liu, W. Cheng, H. Liu, C. Huang, X. Meng, R. D. Rodriguez and X. Jia, J. Mater. Chem. A, 2024, 12, 3807–3843 RSC.
- Y. Sun, D. Wang and Y. Zhu, Chem. Eng. J., 2022, 438, 135667 CrossRef CAS.
- Z. Wang, Q. Peng, X. Huang, Q. Ma, J. Shao and Q. Shen, Dyes Pigments, 2021, 185, 108877 CrossRef CAS.
- J. Tan, G. Zhang, C. Ge, J. Liu, L. Zhou, C. Liu, X. Gao, A. Narita, Y. Zou and Y. Hu, Org. Lett., 2022, 24, 2414–2419 CrossRef CAS PubMed.
- S. Chen, P. Slattum, C. Wang and L. Zang, Chem. Rev., 2015, 115, 11967–11998 CrossRef CAS.
- C. Huang, S. Barlow and S. R. Marder, J. Org. Chem., 2011, 76, 2386–2407 CrossRef CAS PubMed.
- D. Liu, J. Wang, X. Bai, R. Zong and Y. Zhu, Adv. Mater., 2016, 28, 7284–7290 CrossRef CAS PubMed.
- J. Wang, W. Shi, D. Liu, Z. Zhang, Y. Zhu and D. Wang, Appl. Catal., B, 2017, 202, 289–297 CrossRef CAS.
- F. Würthner, C. R. Saha-Möller, B. Fimmel, S. Ogi, P. Leowanawat and D. Schmidt, Chem. Rev., 2016, 116, 962–1052 CrossRef.
- J. Wang, D. Liu, Y. Zhu, S. Zhou and S. Guan, Appl. Catal., B, 2018, 231, 251–261 CrossRef CAS.
- M. Más-Montoya and R. A. J. Janssen, Adv. Funct. Mater., 2017, 27, 1605779 CrossRef.
- B. Wei, H. Li, H. Chu, H. Dong, Y. Zhang, C.-L. Sun and Y. Li, Langmuir, 2024, 40, 6493–6505 CrossRef CAS.
- H. Wu, L. Xue, Y. Shi, Y. Chen and X. Li, Langmuir, 2011, 27, 3074–3082 CrossRef CAS.
- Y. Xu, X. Zhu, H. Yan, P. Wang, M. Song, C. Ma, Z. Chen, J. Chu, X. Liu and Z. Lu, Chin. J. Catal., 2022, 43, 1111–1122 CrossRef CAS.
- X. Yang, S. Zhang, P. Li, S. Gao and R. Cao, J. Mater. Chem. A, 2020, 8, 20897–20924 RSC.
- H. Langhals, S. Demmig and T. Potrawa, J. Prakt. Chem., 1991, 333, 733–748 CrossRef CAS.
- S. Tatemichi, M. Ichikawa, T. Koyama and Y. Taniguchi, Appl. Phys. Lett., 2006, 89, 112108 CrossRef.
- Y. Liu, M. D. Cole, Y. Jiang, P. Y. Kim, D. Nordlund, T. Emrick and T. P. Russell, Adv. Mater., 2018, 30, 1705976 CrossRef.
- Y.-J. Kim, Y. Lee, K. Park, C. W. Ahn, H.-T. Jung and H.-J. Jeon, J. Phys. Chem. Lett., 2020, 11, 3934–3940 CrossRef CAS PubMed.
- R. Guan, X. Cheng, Y. Chen, Z. Wu, Z. Zhao, Q. Shang, Y. Sun and Z. Sun, Nano Res., 2023, 16, 10770–10778 CrossRef CAS.
- S. Ghosh, X.-Q. Li, V. Stepanenko and F. Würthner, Chem. – Eur. J., 2008, 14, 11343–11357 CrossRef CAS PubMed.
- M. E. Ozser, Mater. Today Commun., 2021, 27, 102446 CrossRef CAS.
- S. Izawa, K. Uchida, M. Nakamura, K. Fujimoto, J. Roudin, J.-H. Lee, T. Inuzuka, T. Nakamura, M. Sakamoto, Y. Nakayama, M. Hiramoto and M. Takahashi, Chem. – Eur. J., 2021, 27, 14081–14091 CrossRef PubMed.
- Z. Li, J. Jiao, W. Fu, K. Gao, X. Peng, Z. Wang, H. Zhuo, C. Yang, M. Yang, G. Chang, L. Yang, X. Zheng, Y. Yan, F. Chen, M. Zhang, Z. Meng and X. Shang, Angew. Chem., Int. Ed., 2024, 63, e202412977 CrossRef.
- W. Liu, C. He, S. Huang, K. Zhang, W. Zhu, L. Liu, Z. Zhang, E. Zhu, Y. Chen, C. Chen and Y. Zhu, Angew. Chem., Int. Ed., 2023, 62, e202304773 CrossRef.
- Y. Pu, F. Bao, D. Wang, X. Zhang, Z. Guo, X. Chen, Y. Wei, J. Wang and Q. Zhang, J. Environ. Chem. Eng., 2022, 10, 107123 CrossRef.
- R. Yan, M. Song, P. Chen, H. Song, C. Fu, H. Peng and S.-F. Yin, J. Colloid Interface Sci., 2023, 651, 68–75 CrossRef PubMed.
- L. Zeng, T. Liu, C. He, D. Shi, F. Zhang and C. Duan, J. Am. Chem. Soc., 2016, 138, 3958–3961 CrossRef.
- X. Gao, K. Gao, X. Li, Y. Shang and F. Fu, Catal. Sci. Technol., 2020, 10, 372–381 RSC.
- Y. Guo, B. Liu, J. Zhang, C. Wang, G. Wang, C. Pan, H. Zhao, Y. Dong and Y. Zhu, Appl. Catal., B, 2024, 350, 123915 CrossRef.
- K. Kong, S. Zhang, Y. Chu, Y. Hu, F. Yu, H. Ye, H. Ding and J. Hua, Chem. Commun., 2019, 55, 8090–8093 RSC.
- T. Weil, T. Vosch, J. Hofkens, K. Peneva and K. Müllen, Angew. Chem., Int. Ed., 2010, 49, 9068–9093 CrossRef.
- T. T. Clikeman, E. V. Bukovsky, X.-B. Wang, Y.-S. Chen, G. Rumbles, S. H. Strauss and O. V. Boltalina, Eur. J. Org. Chem., 2015, 6641–6654 CrossRef.
- F. Zhang, Y. Ma, Y. Chi, H. Yu, Y. Li, T. Jiang, X. Wei and J. Shi, Sci. Rep., 2018, 8, 8208 CrossRef PubMed.
- B. Zhang, S. Wang, S. Min, K. Li, L. Kang and W. Lin, Mol. Catal., 2024, 560, 114116 Search PubMed.
- M.-H. Lin, M.-H. Fang, Q. Liao and M.-J. Lin, Dyes Pigments, 2023, 220, 111730 CrossRef.
- Y. Zhang, D. Wang, W. Liu, Y. Lou, Y. Zhang, Y. Dong, J. Xu, C. Pan and Y. Zhu, Appl. Catal., B, 2022, 300, 120762 CrossRef.
- Y. Sheng, W. Li, Y. Zhu and L. Zhang, Appl. Catal., B, 2021, 298, 120585 CrossRef.
- M. Gryszel, T. Schlossarek, F. Würthner, M. Natali and E. D. Głowacki, ChemPhotoChem, 2023, 7, e202300070 CrossRef.
- Z. Zhang, X. Chen, H. Zhang, W. Liu, W. Zhu and Y. Zhu, Adv. Mater., 2020, 32, 1907746 CrossRef PubMed.
- A. Cao, R. Li, X. Xu, W. Huang, Y. He, J. Li, M. Sun, X. Chen and L. Kang, Appl. Catal., B, 2022, 309, 121293 CrossRef.
- X. Wu, B. Hu, D. Li, B. Chen, Y. Huang, Z. Xie, L. Li, N. Shen, F. Yang, W. Shi, M. Chen and Y. Zhu, Angew. Chem., Int. Ed., 2023, 62, e202313787 CrossRef PubMed.
- S. Huang, H. Zhang, W. Li, L. Liu, J. Xu, M. Chong, J. Li and Y. Zhu, Appl. Catal., B, 2024, 347, 123790 CrossRef.
- C. Huang, W. Yu, N. Fang, C. He, Y. Chu and B. Lai, Sep. Purif. Technol., 2025, 360, 131146 CrossRef.
- Y.-J. Chen, J.-Z. Zhang, Z.-X. Wu, Y.-X. Qiao, L. Zheng, F. Wondu Dagnaw, Q.-X. Tong and J.-X. Jian, Angew. Chem., Int. Ed., 2024, 63, e202318224 CrossRef.
- D. Liu, X. Yang, P. Chen, X. Zhang, G. Chen, Q. Guo, H. Hou and Y. Li, Adv. Mater., 2023, 35, 2300655 CrossRef PubMed.
- L. Yang, Y. Fu, F. Sun, M. Deng, C. Zhang, N. Li, D. Hao, Q. Wang and G. Zhuang, J. Colloid Interface Sci., 2023, 639, 472–483 CrossRef PubMed.
- H. Wang, L. Zhang, Z. Chen, J. Hu, S. Li, Z. Wang, J. Liu and X. Wang, Chem. Soc. Rev., 2014, 43, 5234–5244 RSC.
- Y.-P. Yuan, L.-W. Ruan, J. Barber, S. C. Joachim Loo and C. Xue, Energy Environ. Sci., 2014, 7, 3934–3951 RSC.
- S. J. A. Moniz, S. A. Shevlin, D. J. Martin, Z.-X. Guo and J. Tang, Energy Environ. Sci., 2015, 8, 731–759 RSC.
- K. Zhang, J. Wang, W. Jiang, W. Yao, H. Yang and Y. Zhu, Appl. Catal., B, 2018, 232, 175–181 CrossRef.
- Y. Li, Y. Fang, Z. Cao, N. Li, D. Chen, Q. Xu and J. Lu, Appl. Catal., B, 2019, 250, 150–162 CrossRef.
- H. Wang, Y. Zhou, J. Wang, A. Li and P. François-Xavier Corvini, Chem. Eng. J., 2022, 433, 133622 CrossRef.
- M. Wu, H. Yang, Q. Wu, Z. He and S. Wang, J. Environ. Chem. Eng., 2024, 12, 112246 CrossRef.
- G. Liao, C. Li, X. Li and B. Fang, Cell Rep. Phys. Sci., 2021, 2, 100355 CrossRef.
- H. Zhu, C. Zhang, K. Xie, X. Li and G. Liao, Chem. Eng. J., 2023, 453, 139775 CrossRef CAS.
- C. D. Jaeger and A. J. Bard, J. Phys. Chem., 1979, 83, 3146–3152 CrossRef CAS.
- H. Tada, T. Mitsui, T. Kiyonaga, T. Akita and K. Tanaka, Nat. Mater., 2006, 5, 782–786 CrossRef CAS.
- Q. Huang, Q. Zhang, S. Yuan, Y. Zhang and M. Zhang, Appl. Surf. Sci., 2015, 353, 949–957 CrossRef CAS.
- J. Yu, S. Wang, J. Low and W. Xiao, Phys. Chem. Chem. Phys., 2013, 15, 16883–16890 RSC.
- Y. Yuan, R.-T. Guo, L.-F. Hong, X.-Y. Ji, Z.-D. Lin, Z.-S. Li and W.-G. Pan, Mater. Today Energy, 2021, 21, 100829 CrossRef CAS.
- P. Zhou, J. Yu and M. Jaroniec, Adv. Mater., 2014, 26, 4920–4935 CrossRef CAS.
- G. Zhang, Z. Wang and J. Wu, Nanoscale, 2021, 13, 4359–4389 RSC.
- X. Xu, L. Meng, J. Zhang, S. Yang, C. Sun, H. Li, J. Li and Y. Zhu, Angew. Chem., Int. Ed., 2024, 63, e202308597 CrossRef CAS PubMed.
- W. Dai, L. Jiang, J. Wang, Y. Pu, Y. Zhu, Y. Wang and B. Xiao, Chem. Eng. J., 2020, 397, 125476 CrossRef CAS.
- K. Shi, M. Zhou, F. Wang, X. Li, W. Huang, K. Lu, K. Yang and C. Yu, Chemosphere, 2023, 329, 138617 CrossRef CAS.
- C. Li, B. Cheng, J. Shen, H. Wang, M. Yi, P. Gu, R. Liu, G. Liao and Z. Jiang, Sep. Purif. Technol., 2025, 360, 131116 CrossRef CAS.
- S. Liu, Y. Zhang, Y. Guo, Z. Cheng, M. Yuan, Z. Xu, G. Liao and Q. Li, J. Colloid Interface Sci., 2025, 686, 45–62 CrossRef CAS PubMed.
- C. Li, H. Lu, G. Ding, T. Ma, S. Liu, L. Zhang and G. Liao, Chin. J. Catal., 2024, 65, 174–184 CrossRef CAS.
- J. Low, J. Yu, M. Jaroniec, S. Wageh and A. A. Al-Ghamdi, Adv. Mater., 2017, 29, 1601694 CrossRef.
- J. Wei, G. Zhang, S. Xie, Z. Zhang, T. Gao, M. Zhang and X. Li, Angew. Chem., Int. Ed., 2025, 64, e202500441 CrossRef CAS.
- F. Wu, Y. Tang, Y. Pan, J. Han, W. Xing, J. Zhang, G. Wu and Y. Huang, Small, 2025, 2500670, DOI:10.1002/smll.202500670.
- J. Yang, H. Miao, W. Li, H. Li and Y. Zhu, J. Mater. Chem. A, 2019, 7, 6482–6490 RSC.
- R. Chen, H. Lou, Y. Pang, D. Yang and X. Qiu, Small, 2024, 20, 2306354 CrossRef CAS.
- N. Li, Y. Niu, W. An, F. Ruan, H. Wu, B. Hui, Y. Wang and G. Fan, Appl. Catal., B, 2025, 369, 125141 CrossRef CAS.
- F. Zhang, H. Peng, S. Jiang, C. Wang, X. Xu and L. Wang, Environ. Sci. Poll. Res., 2019, 26, 8226–8236 CrossRef CAS.
- S. Linic, P. Christopher and D. B. Ingram, Nat. Mater., 2011, 10, 911–921 CrossRef CAS PubMed.
- H. Miao, J. Yang, Y. Wei, W. Li and Y. Zhu, Appl. Catal., B, 2018, 239, 61–67 CrossRef CAS.
- D. Liu, L. Chen, W. Chen, M. Qin and S. Wei, Dalton Trans., 2021, 50, 4008–4016 RSC.
- B. Yang, L. Lu, Q. Zhang, G. Ding, G. Liao, M. Zhang, X. Liu, R. D. Rodriguez and X. Jia, Chem. Eng. J., 2025, 509, 161236 CrossRef CAS.
- C. Li, X. Liu, G. Ding, P. Huo, Y. Yan, Y. Yan and G. Liao, Inorg. Chem., 2022, 61, 4681–4689 CrossRef CAS PubMed.
- Y. Liang, W. Gui, Z. Yang, K. Cheng, X. Zhou, C. Yang, J. Xu and W. Zhou, RSC Adv., 2023, 13, 11938–11947 RSC.
- B. Palas and G. Ersöz, J. Mol. Liq., 2024, 394, 123717 CrossRef CAS.
- Y. Wang, H. Xu and X. Zhang, Adv. Mater., 2009, 21, 2849–2864 CrossRef CAS.
- W. Dai, L. Jiang, J. Wang, Y. Pu, Y. Zhu, Y. Wang and B. Xiao, Chem. Eng. J., 2020, 397, 125476 CrossRef CAS.
- H. Zhu, L. Gou, C. Li, X. Fu, Y. Weng, L. Chen, B. Fang, L. Shuai and G. Liao, Device, 2024, 2, 100283 CrossRef.
- C. Li, X. Liu, P. Huo, Y. Yan, G. Liao, G. Ding and C. Liu, Small, 2021, 17, 2102539 CrossRef CAS.
- Y.-J. Zhang, J.-Z. Cheng, Y.-Q. Xing, Z.-R. Tan, G. Liao and S.-Y. Liu, Mater. Sci. Semicond. Proc., 2023, 161, 107463 CrossRef CAS.
- A. Muzammil, R. Haider, W. Wei, Y. Wan, M. Ishaq, M. Zahid, W. Yaseen and X. Yuan, Mater. Horiz., 2023, 10, 2764–2799 RSC.
- G. Liao, X. Tao and B. Fang, Matter, 2022, 5, 377–379 CrossRef.
- G. Liao, Y. Gong, L. Zhang, H. Gao, G.-J. Yang and B. Fang, Energy Environ. Sci., 2019, 12, 2080–2147 RSC.
- P. Chen, L. Blaney, G. Cagnetta, J. Huang, B. Wang, Y. Wang, S. Deng and G. Yu, Environ. Sci. Technol., 2019, 53, 1564–1575 CrossRef PubMed.
- T. Sun, J. Song, J. Jia, X. Li and X. Sun, Nano Energy, 2016, 26, 83–89 CrossRef.
- H. Ding, Z. Wang, K. Kong, S. Feng, L. Xu, H. Ye, W. Wu, X. Gong and J. Hua, J. Mater. Chem. A, 2021, 9, 7675–7683 RSC.
- J. Yang, J. Jing, W. Li and Y. Zhu, Adv. Sci., 2022, 9, 2201134 CrossRef PubMed.
- H. Xu, Z. Wang, S. Feng, X. Liu, X. Gong and J. Hua, Int. J. Hydrogen Energy, 2023, 48, 8071–8081 CrossRef.
- W. Yu, N. Fang, Z. Liu, Y. Chu and B. Lai, Small, 2024, 20, 2407104 CrossRef.
- Z. Chen, D. Yan, X. Wang, G. Ding, Z. Wang, Y. Xiao, X. Liu, P. Wang, L. Chen, L. Shuai and G. Liao, ACS Catal., 2025, 15, 13568–13580 CrossRef.
- J. Li, J. Fang, H. Liang, Z. Wei, J. Fang, L. Qiu, X. Lu, F. Yang and G. Zeng, Chem. Eng. J., 2025, 164628 CrossRef.
- S. Peng, R. Wang, Y. Yang, S. Wang, E. Liang, B. Han, J. Li, X. Yu and Q. Zhang, Macromol. Rapid Commun., 2025, 46, 2400967 CrossRef CAS PubMed.
- Y. Sheng, W. Li, L. Xu and Y. Zhu, Adv. Mater., 2022, 34, 2102354 CrossRef CAS.
- W. Li, Z. Wei, Y. Sheng, J. Xu, Y. Ren, J. Jing, J. Yang, J. Li and Y. Zhu, ACS Energy Lett., 2023, 8, 2652–2660 CrossRef CAS.
- G. Ding, C. Li, L. Chen and G. Liao, Energy Environ. Sci., 2024, 17, 5311–5335 RSC.
- G. Liao, G. Ding, B. Yang and C. Li, Precis. Chem., 2024, 2, 49–56 CrossRef CAS.
- K. Liu, M. A. Nawaz and G. Liao, Coord. Chem. Rev., 2025, 535, 216611 CrossRef CAS.
- G. Ding, C. Li, Y. Ni, L. Chen, L. Shuai and G. Liao, EES Catal., 2023, 1, 369–391 RSC.
- Z. Chen, G. Ding, Z. Wang, Y. Xiao, X. Liu, L. Chen, C. Li, H. Huang and G. Liao, Adv. Funct. Mater., 2025, 35, 2423213 CrossRef CAS.
- F. Tian, X. Wu, J. Chen, X. Sun, X. Yan and G. Liao, Dalton Trans., 2023, 52, 11934–11940 RSC.
- K. Liu, Y. Liao, P. Wang, X. Fang, J. Zhu, G. Liao and X. Xu, Nanoscale, 2024, 16, 11096–11108 RSC.
- Z. Zhao, F. Niu, P. Li, H. Wang, Z. Zhang, G. J. Meyer and K. Hu, J. Am. Chem. Soc., 2022, 144, 7043–7047 CrossRef CAS PubMed.
- W. Wu, H. Bi, Z. Zhang, L. Sun, R. Wei, L. Gao, X. Pan, J. Zhang and G. Xiao, Colloids Surf. A, 2023, 657, 130486 CrossRef CAS.
- R. Yang, Q. Li, Z. Ma, S. Liu, D. Tian, D. Li and D. Jiang, Chem. Eng. J., 2025, 506, 160043 CrossRef CAS.
- R. Dalapati, M. Hunter, M. Sk, X. Yang and L. Zang, ACS Appl. Mater. Interfaces, 2024, 16, 32344–32356 CrossRef CAS.
- S. Zheng, H. Du, L. Yang, M. Tan, N. Li, Y. Fu, D. Hao and Q. Wang, J. Hazard. Mater., 2023, 447, 130849 CrossRef CAS.
- Y. Li, Y.-H. Li, P. Wang, C. Zhao, C.-Y. Tang, S.-J. Gao and C.-C. Wang, J. Environ. Chem. Eng., 2023, 11, 109205 CrossRef CAS.
- F. M. A. Altalbawy, P. Sharma, F. H. Alsultany, A. Kumar, K. P. Vinay, I. S. Alalaq, M. Chahar, U. S. Altimari, A. M. Jabbar, M. M. Alam and L. H. Alzubaidi, J. Mol. Struct., 2025, 1326, 141088 CrossRef CAS.
- C. Zhu, C. Gong, D. Cao, L.-L. Ma, D. Liu, L. Zhang, Y. Li, Y. Peng and G. Yuan, Angew. Chem., Int. Ed., 2025, 64, e202504348 CrossRef CAS.
- M. Philippi, K. Kitzinger, J. S. Berg, B. Tschitschko, A. T. Kidane, S. Littmann, H. K. Marchant, N. Storelli, L. H. E. Winkel, C. J. Schubert, W. Mohr and M. M. M. Kuypers, Nat. Commun., 2021, 12, 4774 CrossRef PubMed.
- S. Zhang, Y. Zhao, R. Shi, C. Zhou, G. I. N. Waterhouse, Z. Wang, Y. Weng and T. Zhang, Angew. Chem., Int. Ed., 2021, 60, 2554–2560 CrossRef.
- Z. Lu, J. Zhang, Y. Wang, Y. Yu and L. Kong, Mater. Horiz., 2025, 12, 3286–3300 RSC.
- M. Sharma, A. Kumar, D. Sajwan, K. Kumari, B. P. Mishra and V. Krishnan, Adv. Sustainable Syst., 2025, 9, 2400903 CrossRef.
- J. S. Anderson, J. Rittle and J. C. Peters, Nature, 2013, 501, 84–87 CrossRef.
- G. Liao, C. Li, S.-Y. Liu, B. Fang and H. Yang, Phys. Rep., 2022, 983, 1–41 CrossRef.
- G. Liao, C. Li, S.-Y. Liu, B. Fang and H. Yang, Trends Chem., 2022, 4, 111–127 CrossRef.
- Q. Dong, X. Li, Y. Duan, X. He, X. Liang, F. Yu and C. Wang, Appl. Catal., B, 2025, 366, 125042 CrossRef.
- D. Zhu, L. Zhang, R. E. Ruther and R. J. Hamers, Nat. Mater., 2013, 12, 836–841 CrossRef PubMed.
- Q. Li, D. Shen, Z. Xiao, X. Liu, X. Xu, M. Wu, W. Wang, L. Liu, Q. Li and X. Li, Small, 2025, 21, 2411665 CrossRef PubMed.
- S. Yang, X. Deng, P. Chen, G. Li, Q. Wang, Q. Wang and S.-F. Yin, Chem. Eng. J., 2022, 441, 136084 CrossRef CAS.
- Q. Wang, J. Cao, P. Chen, S. Yang, C. Fu, F. Liu and S.-F. Yin, Appl. Catal., A, 2023, 649, 118978 CrossRef CAS.
- D. Cui, X. Yang, Y. Liu, M. Li, C. Wang and F. Li, Sep. Purif. Technol., 2025, 356, 130035 CrossRef CAS.
- Y. Qiao, C. Sun, J. Jian, T. Zhou, X. Xue, J. Shi, G. Che and G. Liao, J. Mol. Liquids, 2023, 385, 122383 CrossRef CAS.
- G. Ding, Z. Wang, J. Zhang, P. Wang, L. Chen and G. Liao, EcoEnergy, 2024, 2, 22–44 CrossRef CAS.
- C. Du, J. Xu, G. Ding, D. He, H. Zhang, W. Qiu, C. Li and G. Liao, Nanomaterials, 2023, 13, 3066 CrossRef CAS PubMed.
- W. Zhou, G. Liu, B. Yang, Q. Ji, W. Xiang, H. He, Z. Xu, C. Qi, S. Li, S. Yang and C. Xu, Sci. Total Environ., 2021, 780, 146483 CrossRef CAS PubMed.
- Q. Wu, J. Wang, Z. Wang, Y. Xu, Z. Xing, X. Zhang, Y. Guan, G. Liao and X. Li, J. Mater. Chem. A, 2020, 8, 13685–13693 RSC.
- S. Liu, F. Deng, Y. Guo, C. Ouyang, S. Yi, C. Li, G. Liao and Q. Li, ACS Appl. Nano Mater., 2024, 7, 889–903 Search PubMed.
- G. Liao, Y. Gong, L. Zhong, J. Fang, L. Zhang, Z. Xu, H. Gao and B. Fang, Nano Res., 2019, 12, 2407–2436 CrossRef CAS.
- G. Liao, J. Fang, Q. Li, S. Li, Z. Xu and B. Fang, Nanoscale, 2019, 11, 7062–7096 RSC.
- Z. Lu, B. Li, B. Wei, G. Zhou, Y. Xu, J. Zhang, H. Chen, S. Hua, C. Wu and X. Liu, Sep. Purif. Technol., 2023, 314, 123609 CrossRef CAS.
- Z. Mao, P. Luo, J. Ling, X. Zhu, K. Sun, Y. Cao, D. Zhu and W. Liu, J. Alloys Compd., 2025, 1011, 178195 CrossRef CAS.
- K. Zha, L. Li, J. Zhang, S. Tang, X. Li, J. Hai, D. Fan, M. Li, Y. Liu and Z. Lu, J. Photochem. Photobiol. A, 2024, 451, 115517 CrossRef CAS.
- X. Zhang, L. Shi and Y. Zhang, J. Taiwan Inst. Chem. Eng., 2022, 132, 104111 CrossRef CAS.
- X. Zhang, L. Shi, L. Yao and L. Cui, Mater. Res. Bull., 2022, 146, 111589 CrossRef CAS.
- T. Xu, S. Zhang, W. Zhang and L. Shi, Opt. Mater., 2024, 147, 114656 CrossRef CAS.
- H. Miao, J. Yang, Y. Sheng, W. Li and Y. Zhu, Solar RRL, 2021, 5, 2000453 CrossRef CAS.
- Z. Liang, R. Shen, P. Zhang, Y. Li, N. Li and X. Li, Chin. J. Catal., 2022, 43, 2581–2591 CrossRef CAS.
- J. Yang, H. Miao, J. Jing, Y. Zhu and W. Choi, Appl. Catal., B, 2021, 281, 119547 CrossRef CAS.
- C. Ye, J.-X. Li, H.-L. Wu, X.-B. Li, B. Chen, C.-H. Tung and L.-Z. Wu, ACS Appl. Mater. Interfaces, 2018, 10, 3515–3521 CrossRef CAS PubMed.
- L. Liu, J. Liu, S. Zong, Z. Huang, X. Feng, J. Zheng and Y. Fang, Int. J. Hydrogen Energy, 2022, 47, 39486–39498 CrossRef.
- S. Chen, C. Wang, B. R. Bunes, Y. Li, C. Wang and L. Zang, Appl. Catal., A, 2015, 498, 63–68 CrossRef.
- X. Li, X. Lv, Q. Zhang, B. Huang, P. Wang, X. Qin, X. Zhang and Y. Dai, J. Colloid Interface Sci., 2018, 525, 136–142 CrossRef.
- J. Chen, W. Lin, J. Lin and Y. Wang, J. Mater. Chem. A, 2025, 13, 1095–1101 RSC.
- X. Ji, X. Liu, Y. Guo and J. Zhang, Chem. Eng. J., 2021, 425, 131260 CrossRef.
- H. Zhuang, F. Wang, K. Shi and K. Yang, Catalysts, 2023, 13, 688 CrossRef.
- Y. Jia, L. Duan, H. Li, C. Zhang, Q. Gao, H. Zhang, S. Li and M. Li, Sep. Purif. Technol., 2025, 358, 130292 CrossRef.
- Z. Zhang, J. Liu, P.-Y. Gu, R. Ji, L. Jin, S. Zhou, J. He, D. Chen, Q. Xu and J. Lu, Sep. Purif. Technol., 2022, 287, 120539 CrossRef.
| This journal is © The Royal Society of Chemistry 2025 |








