Strategic functionalization of bromine and nitrogen at the bay region of perylene induces a heavy atom effect and promotes intersystem crossing
Pandiselvi
Durairaj
,
Sonia
Das
and
Sunandan
Sarkar
 *
*
Department of Chemistry, National Institute of Technology, Tiruchirappalli, Tamil Nadu 620015, India. E-mail: ssarkar@nitt.edu
First published on 28th October 2025
Abstract
Spin–orbit coupling (SOC)-assisted intersystem crossing (ISC) plays a pivotal role in designing metal-free organic systems for optoelectronic applications. We computationally designed a new structural motif, i.e., 12-bromo-1-azaperylene (12-BAP), which incorporates three crucial factors that promote rapid ISC: a change in state character during singlet–triplet conversion (1ππ* → 3nπ*), a pronounced heavy atom effect (HAE), and a favourable energy alignment between the 1ππ* and 3nπ* states. Notably, the close spatial positioning of the heavy Br atom near the site of orbital angular momentum change intensifies the HAE. As a result, a strong SOC (89 cm−1) between the 1ππ* and 3nπ* states drives ultrafast ISC (1012 s−1). In contrast, azaperylene and its 12-hydroxy derivatives exhibit limited ISC efficiency owing to the higher energetic position of the 3nπ* state. Br substitution at the 12-position of 1-azaperylene yields the highest SOC and fastest ISC among all Br-substituted variants. Efficient triplet-state generation is crucial for the development of high-performance organic phosphorescent emitters and photodynamic therapeutic agents. The 12-BAP motif promotes ISC, providing a versatile scaffold for the rational design of functional organic materials.
Introduction
Harvesting triplet excitons in organic materials enable a wide range of optoelectronic applications, including organic light-emitting devices (OLEDs) and singlet oxygen (1O2) production for photodynamic therapy (PDT).1–3 ISC is a unimolecular photophysical process that converts singlet to triplet excitons (S1 → Tn). Although ISC is a spin-forbidden transition, it becomes feasible through SOC, which leads to mixing of singlet and triplet states.4 Hence, increasing SOC strength is crucial for achieving an efficient triplet population. For example, organometallic compounds of Pt(II), Ir(III), and Ru(II) metals often exhibit near-unity triplet quantum yields due to their large SOC generated by the heavy nucleus.5,6 However, their large-scale production and vast application remain unattainable owing to their scarcity, high cost, and cytotoxicity. Researchers have focused on developing metal-free, purely organic materials capable of efficient triplet exciton generation through structural and crystal engineering to overcome these challenges.7–14Heavier non-metal chalcogens and halogens enhance SOC and ISC in organic materials, reducing dependence on heavy metals.15–21 Organic systems featuring aromatic carbonyls or nitrogen-containing heterocycles (bearing lone-pair electrons) facilitate ISC via pronounced SOC, which originates from a change in the nature of transition orbitals (nπ* ↔ ππ*) between initial and final excited states.22,23 This process is known as the El-Sayed-allowed transition for the spin-flip process.4,23 Since SOC implies that the spin (S) and orbital (L) angular momentum of an electron are coupled, a spin-flip transition must follow the orbital angular momentum change (ΔL) according to the law of angular momentum conservation.20 A change in orbital character between the initial and final states can produce nonzero SOC, thereby allowing spin-forbidden singlet-to-triplet transitions. High SOC is associated with the nπ* ↔ ππ* transition because the nonbonding (n) orbital lies perpendicular to the π-orbital, enabling electron spin flip via a 90° orbital rotation. In contrast, the ππ* → ππ* transition in a planar system exhibits negligible SOC and is known as an El-Sayed-forbidden process, since spin flip cannot occur without a change in orbital orientation. The increase in SOC within twisted π-conjugated frameworks is attributed to π-orbital mixing and the resultant effective orbital angular momentum.24,25 Accordingly, non-planarity promotes state interactions that reinforce the ππ* → ππ* transition as an El-Sayed-allowed process.
Over the past decade, the Kim group has developed a strategy to enhance room-temperature phosphorescence (RTP) through non-covalent halogen bonding between the carbonyl moiety of 4-bromobenzaldehyde and the Br atom of a neighbouring molecule (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯Br).7–9 A heavy Br nucleus near the nπ* site activates the intermolecular heavy atom effect (HAE), promoting ISC and RTP. Importantly, their crystal design principle imparts rigidity to the system, reducing energy loss via molecular vibrations. Sarkar et al. demonstrated an alternative approach wherein substitution of a heavy Br atom proximal to the C
O⋯Br).7–9 A heavy Br nucleus near the nπ* site activates the intermolecular heavy atom effect (HAE), promoting ISC and RTP. Importantly, their crystal design principle imparts rigidity to the system, reducing energy loss via molecular vibrations. Sarkar et al. demonstrated an alternative approach wherein substitution of a heavy Br atom proximal to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group orbitals enhanced ISC in ortho-bromobenzaldehyde.26 This enhancement originates from intramolecular HAE, which operates effectively through lateral interactions between the orbitals of the Br atom and the C
O group orbitals enhanced ISC in ortho-bromobenzaldehyde.26 This enhancement originates from intramolecular HAE, which operates effectively through lateral interactions between the orbitals of the Br atom and the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group. To date, various systems featuring heavy halogens near ΔL sites have been developed, wherein inter- or intramolecular HAEs play a pivotal role in boosting SOC and promoting the triplet-state population.27–36 In this work, we focused on intramolecular HAE, facilitated by strategic substitution that places the heavy atom in close proximity to the key chromophoric orbitals.
O group. To date, various systems featuring heavy halogens near ΔL sites have been developed, wherein inter- or intramolecular HAEs play a pivotal role in boosting SOC and promoting the triplet-state population.27–36 In this work, we focused on intramolecular HAE, facilitated by strategic substitution that places the heavy atom in close proximity to the key chromophoric orbitals.
Polyaromatic hydrocarbons, such as perylenes, are emerging as robust photosensitizers due to superior photostability and light absorption.37 Rachford et al. reported fluorescence quenching via efficient ISC in Pt(II) complexes, where the Pt(II) center is coordinated in the bay region of two perylenediimide (PDI) units.38 Heavy-metal-free strategies have been developed for triplet exciton formation to circumvent the need for expensive metals. For example, Tiley et al. revealed ultrafast triplet formation upon replacing oxygen atoms with heavier sulfur in PDI.16 Lee et al. recently developed a thione-derived PDI–peptide bioconjugate that showed rapid ISC and served as a photosensitizer for bioimaging and PDT.39 Wu et al. reported an ISC quantum yield exceeding 90% in fused di-PDI systems, which efficiently functioned as photosensitizers for 1O2 generation.40 Modifying the bay region of perylenes by Br atoms distorts the π-system, offering an effective approach to enhancing SOC and accelerating ISC.41,42
Interestingly, bay-region engineering of perylenes affords strategic control over optoelectronic properties. For example, nitrogen doping at these sites enhances molar absorptivity, fluorescence efficiency, and host–guest interactions.43 Gryko et al. reported 1-azaperylene as a bright fluorophore with high optical emission,44 while its hydroxy derivative (12-hydroxy-1-azaperylene) showed reduced fluorescence due to excited-state intramolecular proton transfer (ESIPT).45 Hirono et al. synthesized mono- to tetra-azaperylenes via bay-position nitrogen substitution on perylene and observed a progressive decline in the fluorescence quantum yield with increasing nitrogen content due to enhanced nonradiative decay.46
In this work, we discussed the photophysical properties of a newly designed structural scaffold, in which halogens (F, Cl, and Br) are substituted at the 12-position of 1-azaperylene. The key objective is to elucidate how the heavy elements near the n-orbital of pyridinic-N promote the ISC and quench the fluorescence. Fig. 1 displays the pristine azaperylene (AP) and its 12-substituted derivatives (12-XAP). We estimated the fluorescence and ISC rate constants of these systems using theoretical and computational methods to predict the possible deactivation of the S1 state. We further examined how the substitution of Br at different positions in AP modulates the electronic structure and provides a rationale for designing metal-free spin-conversion scaffolds via HAE.
Computational methods
Density functional theory (DFT) was used to optimize the ground state geometry for all systems.47,48 Excited state properties were computed by employing time-dependent DFT (TDDFT) with Tamm–Dancoff approximation (TDA).49–52 TDDFT often underestimates the energy of triplet states, which leads to inaccurate predictions of the energy gap between singlet and triplet states. The TDA approach overcomes this issue, offering more reliable results.52 The range-separated hybrid (RSH) density functional ωB97X-D was employed in this study.53,54 The ωB97X-D functional incorporates 22.2% short-range and 100% long-range exact Hartree–Fock (HF) exchange and offers balanced accuracy for predicting excitation energies for both local and nonlocal (e.g., charge-transfer) excitons. We used the 6-31+G(d,p) basis set for geometry optimization and excitation energy calculations for all systems.55 The state-tracking method was used to optimize the geometry of higher triplet excited states.56 The SG-2 quadrature grid was employed for all calculations to ensure consistent numerical integration. A tight SCF convergence criterion (level 8) was used, corresponding to a wavefunction error below 10−8, which guarantees reliable self-consistent field convergence. All molecules were treated with C1 symmetry throughout all calculations. State characterization was performed based on orbital contributions and natural transition orbital (NTO) analysis, allowing the assignment of excited states according to their dominant electronic character (e.g., π → π*, n → π*). The Q-Chem 5.4 package was used to perform all the calculations in the gas phase.57The fluorescence quantum yield (Φfl) governs photoluminescence efficiency by quantifying the radiative and nonradiative decay rate constants, which is defined as
 | (1) |
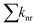 refers to the sum of nonradiative decay rate constants. Typically, kfl is high when the S1 state has high OS, while substantial SOC between S1 and adjacent triplet states accelerates ISC, increasing the triplet population. Thus, the estimation of ISC rate constants is crucial for a comprehensive understanding of the photophysical behaviour of AP and 12-XAP. The fluorescence rate constants (kfl) were estimated using the Strickler–Berg equation:12,58
refers to the sum of nonradiative decay rate constants. Typically, kfl is high when the S1 state has high OS, while substantial SOC between S1 and adjacent triplet states accelerates ISC, increasing the triplet population. Thus, the estimation of ISC rate constants is crucial for a comprehensive understanding of the photophysical behaviour of AP and 12-XAP. The fluorescence rate constants (kfl) were estimated using the Strickler–Berg equation:12,58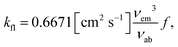 | (2) |
 | (3) |
SOC introduces relativistic correction to the non-relativistic Hamiltonian, typically included through perturbative treatment. SOC enables state mixing and is thus essential for the description of key photophysical properties in molecular systems, for example, ISC. The Breit–Pauli Hamiltonian is utilized to compute the SOC matrix elements between non-relativistic electronic states. The form of the Breit–Pauli spin–orbit (SO) operator (in atomic units) is61–64
 | (4) |
and the second term represents the two-electron spin–own-orbit and spin–other-orbit (SOO) operator, in which h2 is expressed as
Here, α0 and ZA denote the fine structure constant and nuclear charge of the Ath nucleus, respectively; rAi and rij are the distances between the ith electron and the Ath nucleus, and between electrons i and j, respectively; p and s are the momentum and spin operators.
The first term in eqn (4) captures one-electron SO effects, which grow strongly with Z and dominate in heavy atoms, while the two-electron term is appreciable in the case of light-atom systems, as it carries an opposite sign and a magnitude comparable to that of the one-electron term. However, a full evaluation of SOC is computationally expensive due to its two-electron term.65 The computational burden can be mitigated by approximating the two-electron term with an effective one-electron SO operator of mean-field character.62 Alternatively, two-electron effects are treated empirically in the one-electron SO operator by replacing the nuclear charge with effective value,66 and accordingly eqn (4) is reduced to an effective one-electron SO operator of the form:
 | (5) |
Koseki, Gordon, and co-workers notably determined Zeff values for main group elements using the multiconfiguration self-consistent field method with relativistic effective core potentials (MCSCF/SBK).66 They also formulated the scaling factor (Zeff) using the MCSCF method with all-electron basis sets as follows:
| Zeff = ymZA | (6) |
| y1 = 0.45 + 0.05n for Li–F |
| y2 = 0.98 − 0.01n for Na–Cl |
| y3 = 1.21 − 0.03n for K, Ca, Ga–Br |
| y4 = 1.24 for Rb, Sr, In–I |
Chiodo and Russo derived the scaling factor via all-electron DFT calculations using the B3LYP functional and DZVP basis set.69 Their formula defines the scaling factor as follows:
| Zeff = ymZA | (7) |
| y1 = 0.2516 + 0.0626n for B–F |
| y2 = 0.7213 + 0.0144n for Al–Cl |
| y3 = 0.8791 + 0.0039n for Ga–Br |
| y4 = 0.9228 + 0.0017n for In–I. |
Their study revealed that the FSS of diatomic systems calculated using Zeff (B3LYP/DZVP) showed excellent agreement with full-operator results and experimental data. In this work, SOC matrix elements between singlet and triplet states were evaluated using the effective one-electron SO operator (eqn (5)) and Chiodo-derived Zeff values (eqn (7)). The ωB97X-D functional and aug-cc-pVTZ all-electron basis set were used for these calculations.
Results and discussion
We calculated the absorption (Eab) and emission (Eem) energies, specifically the vertical excitation energy of the S1 state at both ground and S1 state geometries, for AP and its derivatives (12-XAP) (Fig. 1). In our earlier study, the Eab and Eem of perylene were reported as 3.428 eV and 3.035 eV, respectively.42 Replacing the bay C–H unit with pyridinic N has minimal impact on both the Eab and Eem of AP, with values of 3.464 eV and 3.063 eV, respectively. Hirono et al. reported through their spectroscopic analysis that the optical spectra of perylene and AP exhibited remarkable similarity.46 Nevertheless, substituents at the 12-position of AP can modulate electron distribution within the π-system, offering potential avenues for tuning its optoelectronic properties. Table 1 highlights the correlation between the electronic characteristics of the substituents and their influence on optical properties.| Molecule | E ab | f ab | E em | f em | k fl |
|---|---|---|---|---|---|
| AP | 3.464 | 0.635 | 3.063 | 0.680 | 2.47 × 108 |
| 12-HAP | 3.383 | 0.591 | 3.059 | 0.621 | 2.30 × 108 |
| 12-FAP | 3.533 | 0.647 | 3.126 | 0.690 | 2.61 × 108 |
| 12-CAP | 3.507 | 0.626 | 3.100 | 0.671 | 2.49 × 108 |
| 12-BAP | 3.493 | 0.611 | 3.087 | 0.657 | 2.42 × 108 |
Hydroxy substitution at the 12-position of AP (12-HAP) resulted in a 0.08 eV redshift in Eab compared to AP, while a slight redshift was observed in Eem. This trend aligns with previous experimental observations, though the experimentally measured values exhibited significantly larger redshifts.44 The redshift is underestimated because the TDDFT calculations were performed without accounting for dielectric medium effects, whereas the experimental measurements were conducted in various solvents. Our computational data revealed that the redshift observed in 12-HAP originates from an increase in its HOMO energy level, attributed to the strong electron-donating effect of the OH group (Fig. S1). Furthermore, the OH group promoted more extensive π-conjugation in the HOMO of 12-HAP relative to AP. In the past, Deperasińska et al. reported a ∼0.28 eV energy separation between Eab and Eem in 12-HAP from gas-phase TDDFT-B3LYP calculations.45 Our TDDFT calculations using the ωB97X-D functional in the gas phase estimated a 0.32 eV energy difference, closely matching the previous result.
In contrast, halogen substitution at the 12-position of AP (12-FAP, 12-CAP, and 12-BAP) exhibited blueshifts in both Eab and Eem compared to AP. This effect arises from the electron-withdrawing nature of halogens, which influences FMOs and their energies (Fig. S1). The 0.07 eV blueshift in 12-FAP, the most pronounced among the halogenated derivatives, stems from the higher electronegativity of F. While larger halogens like Cl and Br typically reduce the energy gap relative to F, resulting in a less pronounced effect. The S1 state of AP and 12-XAP originated from the HOMO → LUMO transition (Table S1), exhibiting a high transition dipole moment and substantial oscillator strength (OS). Consequently, their fluorescence rate constants (kfl ≈ 108 s−1) are significant, as kfl is directly proportional to OS according to the Strickler–Berg relationship.12,58 The experimentally determined kfl values for perylene and AP are 108 s−1, indicating that the calculated kfl value for AP aligns well with experimental results.46 Prior experimental studies have demonstrated that AP and 12-HAP exhibit strong fluorescence.45,46 To date, the synthesis and spectroscopic analysis of 12-halogenated AP derivatives have not been reported. This work used computational methods to predict the photophysical behaviour of 12-halogenated AP derivatives.
Our prior computational study on bromo-substituted perylenes, conducted using a comparable protocol, showed good agreement with experimental observations.42 An experimental study reported high Φfl for perylene and its ortho- and peri-bromo derivatives (P-oBr and P-pBr), while bay-substitution (P-bBr) led to marked fluorescence quenching.70 These trends were well rationalized through computational analysis, as summarized in Table 2. The calculated kfl values for perylene and P-pBr are in good agreement with experimental measurements. To explain the high Φfl observed for perylene, P-pBr, and P-oBr, the computational study revealed that the corresponding kisc values are negligible or 2–3 orders of magnitude lower than the kfl value. Conversely, the low Φfl of P-bBr is theoretically linked to a two-order increase in ISC, promoted by high SOC resulting from bay-induced π-twisting. In the present work, our calculations are consistent with the experimentally measured kfl and explained that the high Φfl of AP originated from its suppressed ISC.
To investigate the nonradiative decay via the ISC process, we first examined the potential ISC channels of AP and 12-XAP. SOC enables mixing between the unperturbed S1 and Tn states, allowing the singlet state to acquire partial triplet character and vice versa. This admixture allows spin-forbidden ISC to occur with enhanced probability. The extent of this mixing, denoted by λ, is primarily determined by the energy difference between Tn and S1 (ΔST) and the SOC strength. λ can be defined from the first-order perturbation theory as
Possible ISC channels for AP and 12-XAP were identified based on these two factors computed at the ωB97X-D/aug-cc-pVTZ level and are summarized in Table S2. The SOC matrix elements were computed using the effective one-electron SO operator (eqn (5)) with Chiodo-derived Zeff values presented in Table S3.
Table 3 presents the ΔST and SOC between the 1ππ* and 3nπ* states of halogenated AP, revealing basis set sensitivity on singlet–triplet gap and spin–orbit interactions. Inclusion of diffuse functions in the Karlsruhe triple-zeta basis set (def2-TZVPD)71 slightly increased the ΔST without greatly affecting SOC for 12-FAP and 12-CAP. The Dunning correlation-consistent basis set (aug-cc-pVTZ)72 exerted minimal impact on ΔST and showed a 1–1.5 cm−1 reduction in SOC for the heavier analogues (12-CAP and 12-BAP) relative to def2-TZVPD. The aug-cc-pVTZ includes extensive diffuse and high-angular-momentum polarization functions, offering more reliable SOC matrix elements for systems bearing heavier atoms.73 Table S4 summarizes the ΔST and SOC between the 1ππ* and 3nπ* states of 12-BAP for different density functionals. The negative ΔST from PBE0 and B3LYP reflected their underestimation of the 3nπ* state energy, whereas ωB97X-D and CAM-B3LYP provided more reliable energy alignment between 1ππ* and 3nπ* states, as range-separated hybrids better capture the excitation energies of charge-transfer or delocalized states such as nπ*. The SOC values obtained with PBE0 and B3LYP are about 20–30 cm−1 higher than those obtained using ωB97X-D and CAM-B3LYP. A prior theoretical study reported an SOC of 98 cm−1 between the 1ππ* and 3nπ* states in bromine-substituted naphthalenediimide (Br adjacent to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group), calculated using the TDDFT/ωB97X-D/cc-pVTZ method and Chiodo-derived Zeff.74
O group), calculated using the TDDFT/ωB97X-D/cc-pVTZ method and Chiodo-derived Zeff.74
| Molecule | def2-TZVP | def2-TZVPD | aug-cc-pVTZ | |||
|---|---|---|---|---|---|---|
| ΔST | SOC | ΔST | SOC | ΔST | SOC | |
| 12-FAP | 0.44 | 7.21 | 0.46 | 7.15 | 0.47 | 7.09 |
| 12-CAP | 0.22 | 20.27 | 0.23 | 20.04 | 0.24 | 18.95 |
| 12-BAP | 0.13 | 91.44 | 0.14 | 90.51 | 0.15 | 88.92 |
Fig. 2 shows that the T1 state is positioned significantly below the S1 state, leading to a large singlet–triplet energy gap (ΔST) across all systems. The 〈S1|Ĥso|T1〉 is weak due to their identical state character (ππ*). Consequently, T1 is not favourable for ISC due to low singlet–triplet mixing. The T2 state is energetically proximal to S1 for all systems, within the range of ±0.1 eV (Table S2), although SOC is low due to the ππ* nature. Among the several low-lying triplet states, the T6 of AP, T8 of 12-HAP, T4 of 12-FAP and 12-CAP, and T3 of 12-BAP exhibited a different state character (nπ*) than the S1 state (refer to Fig. 2). Thus, these triplet states produced significant SOC with the S1 state, with a notable enhancement observed as the halogen size increases (7 cm−1 for 12-FAP, 19 cm−1 for 12-CAP, and 89 cm−1 for 12-BAP). However, these triplet states are positioned above the S1 state, with a particularly large ΔST of 0.73 eV for AP and over 1.3 eV for 12-HAP at the S1-geometry. In contrast, ΔST decreases with increasing halogen size, with values of 0.47 eV for 12-FAP, 0.24 eV for 12-CAP, and 0.15 eV for 12-BAP. This is because the energy of the non-bonding orbital (n) in 12-HAP is more stabilized than AP, while it increases with the size of the halogens (refer to Fig. S2).
Considering the ΔST and SOC values, the preferred triplet states for ISC are T2 for AP and 12-HAP, T4 for 12-FAP and 12-CAP, and T3 for 12-BAP. Moreover, the proximity of T2 in 12-FAP, 12-CAP, and 12-BAP supports its potential as a viable ISC state. T6 in AP was considered for comparative evaluation due to its significant SOC. We calculated the ISC rate constants (kisc) for these channels in AP and 12-XAP using the Marcus theory.59,60,75 Details of kisc for selected channels are provided in Table S5. The kisc for the S1 → T2 transition in AP is 1.96 × 107 s−1, whereas for S1 → T6, it is 8.06 × 106 s−1. Despite a strong SOC (5.39 cm−1) between S1 and T6, the ISC is slow due to an uphill transition (ΔG ≈ 190 meV) with a high energy barrier of ∼244 meV. The kisc for the S1 → T2 transition in 12-HAP is 6.91 × 107 s−1, three times higher than that of AP due to a lower energy barrier (2.5 meV) and higher SOC (0.46 cm−1) compared to AP. For halogen-substituted AP, the kisc for the S1→T2 transition is ∼107–8 s−1. Although having a low energy barrier (<2 meV), the SOC of T2 associated with S1 is weak (∼0.20 cm−1). The kisc for the S1 → T2 transition of AP and 12-XAP is smaller than or comparable to their fluorescence rate constant (kfl ≈ 108 s−1). Hence, the S1 → T2 ISC channel cannot outcompete the fluorescence of these systems.
Interestingly, the kisc for the S1 → T4 transition in 12-FAP and 12-CAP is 6.19 × 108 s−1 and 6.04 × 1010 s−1, respectively. The S1 → T3 transition in 12-BAP exhibited an ultrafast ISC rate, with a kisc value of 2.22 × 1012 s−1 (Table S5). This improvement of rates is attributed to the enhanced SOC driven by a change in state character following El-Sayed's rule and an energetically favourable process (ΔG < 0). The effect is more pronounced in 12-BAP due to the heavy nucleus of Br near the region of orbital angular momentum change. In addition, the S1 → T3 transition in 12-BAP needs to overcome a minimal energy barrier (5.5 meV) compared to the S1 → T4 transition in 12-FAP and 12-CAP. The Br atom proximal to the pyridinic N in 12-BAP lowered the 3nπ* state energy through sidewise orbital interactions (Fig. 2). Hence, the proximity between the Br atom and pyridinic N in 12-BAP not only enhanced the SOC (89 cm−1) but also tuned the 3nπ* state energy to a favourable position, which enabled the 1ππ* → 3nπ* transition to occur on an ultrafast timescale.
The comparison of kisc for the most effective ISC channels in AP and 12-XAP is shown in Fig. 3. Since the kisc (S1 → T2) in AP and 12-HAP is lower than their kfl, ISC cannot quench their fluorescence. As a result, AP and 12-HAP exhibit intense fluorescence, as observed in previous experiments.45,46 In contrast, halogen substitution at the 12-position of AP increased the ISC rate, primarily guided by the change in state character between S1 and Tn states. The S1 → T4 ISC channel in 12-FAP and 12-CAP originates from the 1ππ* → 3nπ* transition, leading to an increased ISC rate. The ISC rate of 12-FAP is more than twice as fast as its fluorescence rate, which may result in a reduced fluorescence quantum yield. Meanwhile, 12-CAP cannot fluoresce, as its ISC rate is three orders of magnitude faster than its fluorescence rate. The ISC rate for the S1 → T3 channel in 12-BAP is the highest among all the channels and is four orders of magnitude faster than its fluorescence rate. Therefore, ISC effectively quenched the fluorescence in 12-BAP.
We strategically designed a structural motif where the bromine atom proximal to the pyridinic nitrogen in 12-BAP boosts ISC. This enhancement of ISC originates from the interplay of three key factors: (i) the orbital angular momentum change during the 1ππ* → 3nπ* transition; (ii) the heavy atom effect; and (iii) the favourable energy gap between 1ππ* and 3nπ* states. To rationalize the design principles behind efficient triplet population via ISC, we explored how the variation of Br positions in AP influences SOC and ISC. While these positional changes did not affect the fluorescence rate (kfl ≈ 108 s−1; Table S6), they significantly modified the energy spacing and SOC between the 1ππ* and 3nπ* states (Table S7). The 3nπ* state of 2-BAP (T7) was about 1.0 eV above the S1 state at the S1-geometry and had strong SOC (∼35.86 cm−1) with the S1 (1ππ*) state. Shifting the Br atom from the 2- to 3-position in AP lowered the 3nπ* state energy (T6) by 0.15 eV and reduced the SOC by 6 cm−1. A further shift to 4-BAP slightly reduced the 3nπ* state energy (T6) by 0.06 eV, while SOC dropped more sharply by 21 cm−1. The energy spacing between the 1ππ* and 3nπ* (T6) states remained mostly unchanged, fluctuating slightly between 0.68 and 0.78 eV, as Br was placed at various sites ranging from 4 to 11. Meanwhile, the SOC between these states remained within the range of 4.2 to 8.7 cm−1. Substituting Br at the 12-position of AP substantially altered the electronic structure, bringing the 3nπ* state (now T3 in 12-BAP) closer to the 1ππ* (S1) state (ΔST = 0.15 eV). Moreover, the SOC between these states (89 cm−1) reached the highest value among all observed cases.
Fig. 4 illustrates how the SOC, i.e., 〈1ππ*|Ĥso|3nπ*〉, varied with the distance between the Br atom and the pyridinic N. In 2-BAP, R is around 2.75 Å, the nearest distance between Br and N among all the systems. The SOC in 2-BAP is significantly strong (36 cm−1). However, when the Br atom shifts from 2 to 3, the R value increased to ∼4 Å and the SOC reduced to 30 cm−1. When R exceeded 5 Å, with Br positioned between 4 and 11, the SOC sharply decreased to a low value ranging from 4 to 8 cm−1. In 12-BAP, the Br atom is closer to N, with R below 3 Å. Consequently, the SOC is the highest among all the systems, approaching 89 cm−1. Since Ĥso ∝ Z4 (Z is the nuclear charge), a heavy Br atom near the n-orbital of pyridinic-N (at the site of ΔL) can induce strong 〈1ππ*|Ĥso|3nπ*〉 through the HAE. When comparing 2-BAP and 12-BAP, the R value of 2-BAP (2.75 Å) is lower than that of 12-BAP (2.95 Å), while the SOC is higher for 12-BAP. This contrasting behaviour can be rationalized using the NTO plot shown in Fig. 5. In AP, the n-orbital is localized on the pyridinic N, whereas in 2-BAP, the n-orbital extends in-plane with mutual alignment of the p-orbital of Br. This extension is weaker in 3-BAP and absent in 4-BAP to 11-BAP (see Fig. S3). In 12-BAP, the n-orbital exhibited greater spatial extension due to its side-wise association with the in-plane p-orbital of Br. Fig. 5 illustrates that this p-orbital of Br contributed prominently to the hole NTO of the 3nπ* state, in conjunction with the n-orbital of N. This cooperative orbital mixing leads to an expanded hole NTO for the 3nπ* state, which is orthogonal to the hole NTO of the 1ππ* state. Consequently, the SOC matrix element (〈1ππ*|Ĥso|3nπ*〉) is significantly enhanced in 12-BAP.
 | ||
| Fig. 4 Distance between Br and N (R in Å) in n-BAP and the SOC between the 1ππ* and 3nπ* states (〈1ππ*|Ĥso|3nπ*〉 in cm−1). | ||
 | ||
| Fig. 5 Natural transition orbitals of the 1ππ* and 3nπ* states for AP, 2-BAP, 3-BAP, 11-BAP and 12-BAP. | ||
Furthermore, we computed the ISC rates for the nearby 3ππ* state and the 3nπ* state of n-BAP, as summarized in Table S8. The S1 → T2 ISC channel in n-BAP corresponds to the 1ππ* → 3ππ* transition. Despite the weak SOC, the ISC remained energetically favourable (ΔG < 0), yielding kisc in the 105–7 s−1 range. For the alternative ISC channel (1ππ* → 3nπ*), kisc was quite low (∼103 s−1) for 2-BAP and varied between 105–6 s−1 for the other derivatives, except for 12-BAP (where it reached 1012 s−1). Although the SOC was higher for 1ππ* → 3nπ* than for 1ππ* → 3ππ*, the ISC for 1ππ* → 3nπ* remained inefficient across 2-BAP to 11-BAP due to the unfavourable driving force (ΔG > 0) and substantial barrier height (220–480 meV). While strong SOC allows spin-forbidden transitions, it alone does not ensure rapid ISC. In the Marcus framework (refer to eqn (3)), the ISC rate constant scales quadratically with SOC but decays exponentially with ΔG. An uphill singlet–triplet energy gap (ΔG > 0) can therefore outweigh the benefit of large SOC, so both strong coupling and favourable energetic alignment are required for efficient triplet formation. In 12-BAP, the 1ππ* → 3nπ* transition was significantly more efficient, surpassing the 1ππ* → 3ππ* by four orders of magnitude, which was driven by remarkably high SOC (〈1ππ*|Ĥso|3nπ*〉 = 89 cm−1), and an energetically favourable process (ΔG < 0) with a low activation barrier (5.5 meV). Table 4 summarizes the kisc values for the dominant ISC channel in AP and several bromo-substituted derivatives. Notably, Br proximal to the pyridinic N facilitated an ultrafast triplet population via ISC in 12-BAP owing to the enhanced SOC and the optimized energy alignment between the 1ππ* and 3nπ* states.
| Molecule | |Vsoc| | ΔG | E r | E a | k isc |
|---|---|---|---|---|---|
| AP | 0.20 | −42.5 | 114.3 | 11.3 | 1.96 × 107 |
| 2-BAP | 0.13 | −10.3 | 109.0 | 22.3 | 5.55 × 106 |
| 3-BAP | 0.23 | −16.3 | 109.5 | 19.8 | 1.91 × 107 |
| 11-BAP | 0.18 | −32.5 | 116.2 | 15.1 | 1.37 × 107 |
| 12-BAP | 88.92 | −436.5 | 546.4 | 5.5 | 2.22 × 1012 |
Conclusions
In conclusion, we computationally designed a new structural motif, i.e., 12-BAP, by functionalizing a bromine atom near the nitrogen at the bay site of AP to enhance ISC. The HAE from Br significantly strengthened SOC (〈1ππ*|Ĥso|3nπ*〉 = 89 cm−1), enabling ultrafast ISC (1012 s−1) that outcompetes fluorescence. In contrast, AP and 12-HAP remained fluorescent due to their higher 3nπ* state energies. Br substitution at the 2-, 3-, and 12-positions increased the 〈1ππ*|Ĥso|3nπ*〉 via HAE, with 12-BAP showing the strongest effect. Despite its shortest Br⋯N distance, 2-BAP exhibited lower SOC than 12-BAP because the stronger lateral interaction in 12-BAP leads to greater extension of the hole NTO. This lateral interaction in 12-BAP also lowered the 3nπ* state energy, facilitating rapid ISC, while 2-BAP retained a higher, less favourable energy for the 3nπ* state, which limited its ISC efficiency. Thus, the strategic design of 12-BAP integrates three key elements that collectively facilitate ultrafast ISC: (i) the orbital angular momentum change during singlet–triplet conversion (1ππ* → 3nπ*), (ii) a pronounced HAE, and (iii) a narrow energy gap between the 1ππ* and 3nπ* states. The development of efficient phosphorescent organic materials relies on promoting ISC from the S1 to the triplet state. Moreover, in PDT, the photosensitizer transfers its triplet energy to ground-state molecular oxygen (3O2), generating reactive singlet oxygen (1O2). Hence, ISC-mediated conversion of singlet to triplet excitons plays a crucial role in both organic optoelectronic performance and PDT efficacy. This work introduces a design principle for enhancing ISC, and we anticipate that the proposed spin-conversion strategy will significantly advance both metal-free OLEDs and PDT.Author contributions
PD contributed to data curation, methodology, formal analysis, investigation, visualization, and manuscript writing and revision. SD was involved in data curation, methodology, formal analysis, and visualization. SS led the project's conceptualization and supervision, and contributed to methodology, investigation, formal analysis, and manuscript writing and revision.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Frontier molecular orbitals of AP and 12-XAP at S0 geometry, energy level diagram for frontier molecular orbitals at S1 geometry, natural transition orbitals of n-BAP at S1 geometry (Fig. S1–S3); excitation energies of AP and 12-XAP at S0 and S1 geometries; ΔST, SOC and singlet–triplet mixing coefficient between S1 and Tn states of AP and its derivatives; scaling factors (Zeff) utilized in effective one-electron spin–orbit operator, comparison of ΔST and SOC for 12-BAP across density functionals, details of ISC rate constants for AP and its derivatives, fluorescence rate constant of n-BAP (Table S1–S8); Cartesian coordinates for the optimized S0, S1 and Tn geometries of AP and 12-XAP (Table S9–S23). See DOI: https://doi.org/10.1039/d5cp03115j.Acknowledgements
PD and SD gratefully thank NIT Tiruchirappalli and the Ministry of Education (Government of India) for their research fellowships. SS acknowledges the funding from the DST-SERB project (CRG/2021/002893). We thank DST-FIST (SR/FST/CS-II/2018/64) for providing access to the HPC facility.References
- C. W. Tang and S. A. VanSlyke, Appl. Phys. Lett., 1987, 51, 913–915 CrossRef CAS.
- M. A. Baldo, D. F. O'Brien, Y. You, A. Shoustikov, S. Sibley, M. E. Thompson and S. R. Forrest, Nature, 1998, 395, 151–154 CrossRef CAS.
- T. C. Pham, V. N. Nguyen, Y. Choi, S. Lee and J. Yoon, Chem. Rev., 2021, 121, 13454–13619 CrossRef CAS PubMed.
- S. K. Lower and M. A. El-Sayed, Chem. Rev., 1966, 66, 199–241 CrossRef CAS.
- J. Zhao, W. Wu, J. Sun and S. Guo, Chem. Soc. Rev., 2013, 42, 5323–5351 RSC.
- M. Schulze, A. Steffen and F. Würthner, Angew. Chem., Int. Ed., 2015, 54, 1570–1573 CrossRef CAS PubMed.
- O. Bolton, K. Lee, H.-J. Kim, K. Y. Lin and J. Kim, Nat. Chem., 2011, 3, 205–210 CrossRef CAS PubMed.
- O. Bolton, D. Lee, J. Jung and J. Kim, Chem. Mater., 2014, 26, 6644–6649 CrossRef CAS.
- M. S. Kwon, D. Lee, S. Seo, J. Jung and J. Kim, Angew. Chem., Int. Ed., 2014, 53, 11177–11181 CrossRef CAS PubMed.
- D. Lee, O. Bolton, B. C. Kim, J. H. Youk, S. Takayama and J. Kim, J. Am. Chem. Soc., 2013, 135, 6325–6329 CrossRef CAS PubMed.
- M. S. Kwon, Y. Yu, C. Coburn, A. W. Phillips, K. Chung, A. Shanker, J. Jung, G. Kim, K. Pipe and S. R. Forrest, Nat. Commun., 2015, 6, 8947 CrossRef PubMed.
- M. S. Kwon, J. H. Jordahl, A. W. Phillips, K. Chung, S. Lee, J. Gierschner, J. Lahannabcd and J. Kim, Chem. Sci., 2016, 7, 2359–2363 RSC.
- S. Hirata, K. Totani, J. Zhang, T. Yamashita, H. Kaji, S. R. Marder, T. Watanabe and C. Adachi, Adv. Funct. Mater., 2013, 23, 3386–3397 CrossRef CAS.
- S. Xiong, Y. Xiong, D. Wang, Y. Pan, K. Chen, Z. Zhao, D. Wang and B. Z. Tang, Adv. Mater., 2023, 35, 2301874 CrossRef CAS PubMed.
- R. D. Pensack, Y. Song, T. M. McCormick, A. A. Jahnke, J. Hollinger, D. S. Seferos and G. D. Scholes, J. Phys. Chem. B, 2014, 118, 2589–2597 CrossRef CAS PubMed.
- A. J. Tilley, R. D. Pensack, T. S. Lee, B. Djukic, G. D. Scholes and D. S. Seferos, J. Phys. Chem. C, 2014, 118, 9996–10004 CrossRef CAS.
- G. He, B. D. Wiltshire, P. Choi, A. Savin, S. Sun, A. Mohammadpour, M. J. Ferguson, R. McDonald, S. Farsinezhad, A. Brown, K. Shankar and E. Rivard, Chem. Commun., 2015, 51, 5444–5447 RSC.
- R. Acharya, S. Cekli, C. J. I. Zeman, R. M. Altamimi and K. S. Schanze, J. Phys. Chem. Lett., 2016, 7, 693–697 CrossRef CAS PubMed.
- W. Shao, H. Jiang, R. Ansari, P. M. Zimmerman and J. Kim, Chem. Sci., 2022, 13, 789–797 RSC.
- W. Shao and J. Kim, Acc. Chem. Res., 2022, 55, 1573–1585 CrossRef CAS PubMed.
- E. Hamzehpoor, C. Ruchlin, Y. Tao, C.-H. Liu, H. M. Titi and D. F. Perepichka, Nat. Chem., 2023, 15, 83–90 CrossRef CAS PubMed.
- D. R. Kearns and W. A. Case, J. Am. Chem. Soc., 1966, 88, 5087–5097 CrossRef CAS.
- E. Lim and J. M. Yu, J. Chem. Phys., 1966, 45, 4742–4743 CrossRef CAS.
- K. Schmidt, S. Brovelli, V. Coropceanu, D. Beljonne, J. Cornil, C. Bazzini, T. Caronna, R. Tubino, F. Meinardi, Z. Shuai and J. L. Brédas, J. Phys. Chem. A, 2007, 111, 10490–10499 CrossRef CAS PubMed.
- S. Sarkar, J. D. Protasiewicz and B. D. Dunietz, J. Phys. Chem. Lett., 2018, 9, 3567–3572 CrossRef CAS PubMed.
- S. Sarkar, H. P. Hendrickson, D. Lee, F. DeVine, J. Jung, E. Geva, J. Kim and B. D. Dunietz, J. Phys. Chem. C, 2017, 121, 3771–3777 CrossRef CAS.
- E. Lucenti, A. Forni, C. Botta, L. Carlucci, C. Giannini, D. Marinotto, A. Pavanello, A. Previtali, S. Righetto and E. Cariati, Angew. Chem., Int. Ed., 2017, 56, 16302–16307 CrossRef CAS PubMed.
- Y. Lee, R. M. Malamakal, D. M. Chenoweth and J. M. Anna, J. Phys. Chem. Lett., 2020, 11, 877–884 CrossRef CAS PubMed.
- W. Shao, J. Hao, H. Jiang, P. M. Zimmerman and J. Kim, Adv. Funct. Mater., 2022, 32, 2201256 CrossRef CAS.
- Y. He, J. Wang, Q. Li, S. Qu, C. Zhou, C. Yin, H. Ma, H. Shi, Z. Meng and Z. An, Adv. Opt. Mater., 2023, 11, 2201641 CrossRef CAS.
- K. Chen, Y. Xiong, D. Wang, Y. Pan, Z. Zhao, D. Wang and B. Z. Tang, Adv. Funct. Mater., 2024, 34, 2312883 CrossRef CAS.
- Y. Tani, K. Miyata, E. Ou, Y. Oshima, M. Komura, M. Terasaki, S. Kimura, T. Ehara, K. Kubo, K. Onda and T. Ogawa, Chem. Sci., 2024, 15, 10784–10793 RSC.
- C. Demangeat, M. Remond, T. Roisnel, C. Quinton and L. Favereau, Chem. – Eur. J., 2024, 30, e202401506 CrossRef CAS PubMed.
- K. Vinod, S. D. Jadhav and M. Hariharan, Chem. – Eur. J., 2024, 30, e202400499 CrossRef CAS PubMed.
- D. Shikichi, T. Ehara, M. Komura, K. Onda, K. Miyata and Y. Tani, J. Chem. Phys., 2025, 162, 121101 CrossRef CAS PubMed.
- S. M. Wagalgave, A. A. Kongasseri, U. Singh, A. Anilkumar, S. N. Ansari, S. K. Pati and S. J. George, J. Am. Chem. Soc., 2025, 147, 15591–15601 CrossRef CAS PubMed.
- J. T. Markiewicz and F. Wudl, ACS Appl. Mater. Interfaces, 2015, 7, 28063–28085 CrossRef CAS PubMed.
- A. A. Rachford, S. Goeb and F. N. Castellano, J. Am. Chem. Soc., 2008, 130, 2766–2767 CrossRef CAS PubMed.
- Y.-L. Lee, Y.-T. Chou, B.-K. Su, C.-C. Wu, C.-H. Wang, K.-H. Chang, J.-A. A. Ho and P.-T. Chou, J. Am. Chem. Soc., 2022, 144, 17249–17260 CrossRef CAS PubMed.
- Y. Wu, Y. Zhen, Y. Ma, R. Zheng, Z. Wang and H. Fu, J. Phys. Chem. Lett., 2010, 1, 2499–2502 CrossRef CAS.
- J. Sunny, E. Sebastian, S. Sujilkumar, F. Würthner, B. Engels and M. Hariharan, Phys. Chem. Chem. Phys., 2023, 25, 28428–28436 RSC.
- P. Durairaj, D. Mukkonathil and S. Sarkar, J. Phys. Chem. A, 2024, 128, 10193–10201 CrossRef CAS PubMed.
- Y. Wu, X. Liu, J. Liu, G. Yang, Y. Deng, Z. Bin and J. You, J. Am. Chem. Soc., 2024, 146, 15977–15985 CrossRef CAS PubMed.
- D. T. Gryko, J. Piechowska and M. Gałęzowski, J. Org. Chem., 2010, 75, 1297–1300 CrossRef CAS PubMed.
- I. Deperasińska, D. T. Gryko, E. Karpiuk, B. Kozankiewicz, A. Makarewicz and J. Piechowska, J. Phys. Chem. A, 2012, 116, 2109–2116 CrossRef PubMed.
- A. Hirono, H. Sakai, S. Kochi, T. Sato and T. Hasobe, J. Phys. Chem. B, 2020, 124, 9921–9930 CrossRef PubMed.
- P. Hohenberg and W. Kohn, Phys. Rev., 1964, 136, B864–B871 CrossRef.
- W. Kohn and L. J. Sham, Phys. Rev., 1965, 140, A1133–A1138 CrossRef.
- M. E. Casida, C. Jamorski, K. C. Casida and D. R. Salahub, J. Chem. Phys., 1998, 108, 4439–4449 CrossRef CAS.
- S. Hirata and M. Head-Gordon, Chem. Phys. Lett., 1999, 314, 291–299 CrossRef CAS.
- J. S. Sears, T. Koerzdoerfer, C.-R. Zhang and J.-L. Brédas, J. Chem. Phys., 2011, 135, 151103 CrossRef PubMed.
- M. J. G. Peach, M. J. Williamson and D. J. Tozer, J. Chem. Theory Comput., 2011, 7, 3578–3585 CrossRef CAS PubMed.
- J.-D. Chai and M. Head-Gordon, Phys. Chem. Chem. Phys., 2008, 10, 6615–6620 RSC.
- J.-D. Chai and M. Head-Gordon, J. Chem. Phys., 2008, 128, 084106 CrossRef PubMed.
- M. M. Francl, W. J. Pietro, W. J. Hehre, J. S. Binkley, M. S. Gordon, D. J. DeFrees and J. A. Pople, J. Chem. Phys., 1982, 77, 3654–3665 CrossRef CAS.
- K. D. Closser, O. Gessner and M. Head-Gordon, J. Chem. Phys., 2014, 140, 134306 CrossRef PubMed.
- E. Epifanovsky, A. T. B. Gilbert, X. Feng, J. Lee, Y. Mao, N. Mardirossian, P. Pokhilko, A. F. White, M. P. Coons and A. L. Dempwolff, et al. , J. Chem. Phys., 2021, 155, 084801 CrossRef CAS PubMed.
- S. J. Strickler and R. A. Berg, J. Chem. Phys., 1962, 37, 814–822 CrossRef CAS.
- R. A. Marcus, Rev. Mod. Phys., 1993, 65, 599–610 CrossRef CAS.
- Q. Ou and J. E. Subotnik, J. Phys. Chem. C, 2013, 117, 19839–19849 CrossRef CAS.
- D. G. Fedorov and M. S. Gordon, J. Chem. Phys., 2000, 112, 5611–5623 CrossRef CAS.
- S. Kotaru, P. Pokhilko and A. I. Krylov, J. Chem. Phys., 2022, 157, 224110 CrossRef CAS PubMed.
- J. Ehrman, E. Martinez-Baez, A. J. Jenkins and X. Li, J. Chem. Theory Comput., 2023, 19, 5785–5790 CrossRef CAS PubMed.
- J. J. Talbot, T. P. Cheshire, S. J. Cotton, F. A. Houle and M. Head-Gordon, J. Phys. Chem. A, 2024, 128, 7830–7842 CrossRef CAS PubMed.
- A. Van Yperen-De Deyne, E. Pauwels, V. Van Speybroeck and M. Waroquier, Phys. Chem. Chem. Phys., 2012, 14, 10690–10704 RSC.
- S. Koseki, M. S. Gordon, M. W. Schmidt and N. Matsunaga, J. Phys. Chem., 1995, 99, 12764–12772 CrossRef CAS.
- S. Koseki, M. W. Schmidt and M. S. Gordon, J. Phys. Chem., 1992, 96, 10768–10772 CrossRef CAS.
- S. Koseki, M. W. Schmidt and M. S. Gordon, J. Phys. Chem. A, 1998, 102, 10430–10435 CrossRef CAS.
- S. G. Chiodo and N. Russo, J. Comput. Chem., 2008, 30, 832–839 CrossRef PubMed.
- F. Lewitzka, H.-G. Lühmannsrijben, M. Strauch and W. Lüttke, J. Photochem. Photobiol. A, 1991, 61, 191–200 CrossRef CAS.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- J. Dunning and H. Thom, J. Chem. Phys., 1989, 90, 1007–1023 CrossRef.
- M. K. Lee, P. Hoerner, W. Li and H. B. Schlegel, J. Chem. Phys., 2020, 153, 244109 CrossRef CAS PubMed.
- J. Liu, Z. Lan and J. Yang, Phys. Chem. Chem. Phys., 2021, 23, 22313–22323 RSC.
- P. Durairaj, K. Samuthirapandi and S. Sarkar, J. Phys. Chem. A, 2023, 127, 3330–3338 CrossRef CAS PubMed.
| This journal is © the Owner Societies 2025 |






