Photophysical divergence driven by π-spacer variations in the anthracene–cyanostilbene architecture
Cherumannil
Femina
 a,
Norifumi
Yamamoto
a,
Norifumi
Yamamoto
 b,
Nelliyulla Kappumchalil
Ramya
b,
Nelliyulla Kappumchalil
Ramya
 a,
Pookkottu K.
Sajith
a,
Pookkottu K.
Sajith
 *a and
Reji
Thomas
*a and
Reji
Thomas
 *a
*a
aDepartment of Chemistry, Farook College (Autonomous), Research Centre of University of Calicut, Kozhikode 673632, Kerala, India. E-mail: femi.fana@gmail.com; norifumi.yamamoto@it-chiba.ac.jp; ramyank2012@gmail.com; pksajith@farookcollege.ac.in; pksajisiv@gmail.com; reji@farookcollege.ac.in
bDepartment of Applied Chemistry, Faculty of Engineering, Chiba Institute of Technology, 2-17-1, Tsudanuma, Narashino, Chiba 275-0016, Japan
First published on 22nd October 2025
Abstract
A series of four anthracene-incorporated cyanostilbene derivatives with donor–π–acceptor (D–π–A) architectures was synthesized to investigate the effect of a phenyl π-spacer. Compounds without the spacer (2a, 2b) showed low quantum yields (ΦF = 0.05–0.25) in various solvents, negative solvatochromism, aggregation-induced enhanced emission (AIEE), and red-shifted mechanofluorochromism. In contrast, those with the spacer (3a, 3b) exhibited high quantum yields (ΦF = 0.75–0.85), positive solvatochromism, aggregation-induced emission (AIE), and blue-shifted mechanofluorochromism, highlighting the key role of the spacer in modulating photophysical properties. Quantum chemical calculations reveal that the compounds incorporating a phenyl π-spacer exhibit conformational flexibility between syn- and anti-conformers in both the ground and excited states, due to an energetically more accessible transition state relative to analogues lacking the spacer. The findings in this study underscore the importance of strategic structural engineering in anthracene-based cyanostilbene systems, which profoundly influences their photophysical properties, paving the way for their potential use in future optoelectronic applications.
Introduction
Tuning of the photophysical characteristics of organic luminescent materials under ambient conditions is an area of fundamental research and is extensively applied in the field of optoelectronics.1–5 The conventional π-conjugated compounds without any functional modifications have simple photophysical properties with respect to their structural and functional moieties. Taking this into account, it is necessary to acquire more specific optoelectronic performances, which can be achieved by designing and constructing smart molecules in which the photophysical properties can be tuned in a controlled manner.6 Therefore, novel molecular designs with tunable photophysical properties can serve as promising candidates for the development of versatile optoelectronic devices.7–11 The construction and design of such smart molecular structures possessing stimuli-responsive fluorescence switching, such as mechanochromism, thermochromism, vapochromism etc., has received unprecedented attention.12–16Organic luminogens, which are made up of molecules containing electron donor (D) and electron acceptor (A) units, have drawn considerable recognition due to their effectiveness in obtaining emissive compounds owing to their push–pull intramolecular electronic behaviour. Such systems have a wide range of applications in the fields of sensors, fluorescent probes, organic solar cells, and hybrid materials, etc.17–22 The donor (D) and the acceptor (A) units are linked through π-conjugated spacers, creating an efficient path for the transport of charge carriers from the donor to the acceptor by the intramolecular charge transfer (ICT) mechanism. ICT systems based on cyanostilbenes as acceptor units have been widely explored over the past decades and employed as fluorescent probes to indicate the micro-environmental changes.23–26 Most of the studies in these classes of molecules focused on the proper management and utilization of the phenomena for achieving bioimaging, energy harvesting and display applications. The π-conjugated structures endow ample aggregation modes in the solid state, leading to enhanced solid-state emission, while the ICT phenomenon enhances the emission properties of these molecules in solution. Therefore, the design and synthesis of donor–acceptor-based molecules opens great promise to afford bright emissive materials with tunability in solution and the solid state.25
Among various π-conjugated materials, cyanostilbene-based organic fluorophores have drawn interest due to their extraordinary emissive properties resulting from their rigid and twisted geometry along with their tendency to form emissive aggregates in the solid state. Recent reports have shown that the cyanostilbene derivatives with alkyl chains provide flexible solid-state structures with precise control over molecular packing and aggregation, wherein various external stimuli such as heat, light, pressure, etc. have resulted in tunable optoelectronic properties.27–30 Various attempts have been made by supramolecular chemists to design and synthesize molecular systems with multi–stimuli responsive emission switching.31,32 In this regard, some of the researchers have focused on constructing molecular systems capable of mechanofluorochromism (MFC) properties, which have significant importance in the development of smart devices with potential in the field of sensing and imaging.33,34
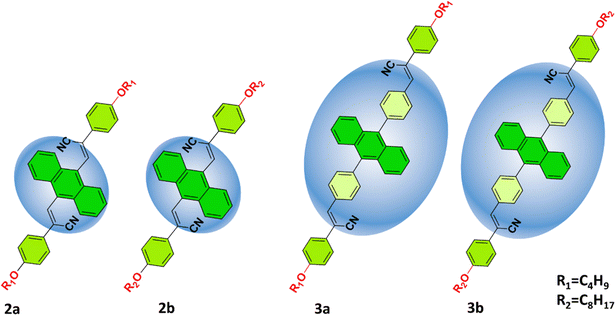
There are several notable examples of cyanostyryl-based donor–acceptor systems with triphenylamine,35 carbazole,33 fluorene36 and perylene as donors showing multi–stimuli responsive photoluminescence properties. However, cyanostyryl systems incorporating anthracene,37,38 a well-known chromophore with excellent photophysical properties, as the donor moiety remain relatively scarce in the literature. In our previous study, we reported the ICT and multi–stimuli responsive emission behaviour of alkyl chain variants in a symmetrically modified anthracene–dicyanostyryl D–π–A system, where the donor and acceptor units are separated by a phenyl spacer.23 Building on these findings, the present work investigates the influence of the π-spacer on the intramolecular charge transfer properties of their homologues (see Fig. 1, 3a and 3b) in comparison to their molecular counterpart where the anthracene moiety is directly linked to the cyanostyryl acceptor unit (see Fig. 1, 2a and 2b). This study aims to compare the role of donor–acceptor linkages in these molecules in spite of enhanced conjugation in the molecular system with an additional phenyl ring. The intrinsic properties of the core chromophore, independent of alkyl chain variations, are represented by compound 2 (comprising 2a and 2b) and compound 3 (comprising 3a and 3b). The present study reports contrasting photophysical behaviour of two series of compounds, including the rarely observed negative solvatochromism in compound 2. Both the compounds showed aggregation induced emission and mechanofluorochromism in the solid state. Herein, we investigated the structural origin of the photophysical divergence of the compounds supported by structural data obtained from X-ray diffraction and TD-DFT calculations.
Experimental and computational methods
All the chemicals and reagents required for the synthesis of anthracene–cyanostilbene derivatives (2a to 3b) were purchased from commercial suppliers and used without further purification. All of the compounds were synthesized by following the reported procedures with modifications and the synthesized compounds were characterized using 1H NMR and 13C NMR spectroscopy in CDCl3 with TMS as an internal standard (see Fig. S1–S6). Compounds 2b and 3b were further characterized using single-crystal X-ray diffraction. The single crystals of compounds 2b (orange-yellow crystal) and 3b (green crystal) were grown by slow evaporation at room temperature from the respective dichloromethane and ethyl acetate solutions. Fig. S7 shows the ORTEP of compounds 2b and 3b drawn at 50% probability. The mass spectral data were obtained from electron spray ionization mass spectroscopy (ESI-MS), model-micromass QTof MicroTM and a Maxis Impact. UV-visible spectroscopic studies were carried out on a Cary 5000 high-performance UV-Vis-NIR spectrophotometer with superb photometric performance in the 175–3300 nm range. The fluorescence lifetime and the steady state fluorescence studies were conducted on an FLS1000-xS-t fluorescence lifetime spectrometer and on an Edinburgh FLS-1000 photoluminescence spectrometer attached with an integrating sphere. All the fluorescence spectra were recorded with a quartz cuvette of path length 1 cm at a slit width of 1 nm. Absolute fluorescence quantum yield of the compounds in both solid and solution states was recorded on an LQE-100 LED photo-luminescence quantum yield measurement system. The single crystal X-ray diffraction data were measured on a Bruker AXS Kappa Apex2 diffractometer. The powder X-ray diffraction intensities were measured on a Panalytical Aeris Research diffractometer.All quantum chemical calculations were performed using the Gaussian 16, Revision C.02 program package.39 The geometries of the ground state (S0) and the first singlet excited state (S1) were fully optimized in the gas phase using density functional theory (DFT) and its time-dependent extension (TD-DFT), respectively. The long-range corrected ωB97-XD functional was employed in conjunction with the 6-311+G(d) basis set for all atoms. Frequency calculations were subsequently carried out at the same level of theory to verify that each optimized structure was a true energy minimum, characterized by the absence of imaginary frequencies. The Gibbs free energies (ΔG) of these conformers were computed at 298.15 K and 1 atm based on the results of the frequency analyses under the harmonic oscillator approximation. The transition state (TS) structures connecting the syn and anti-conformers were also located and were confirmed to be first-order saddle points by the presence of a single imaginary frequency corresponding to the isomerization coordinate. Vertical absorption energies (S0 → S1) and their corresponding wavelengths were calculated by performing TD-DFT calculations on the S0-Min geometries. To simulate the fluorescence process, the geometries of the first singlet excited state (S1) were optimized for all conformers. Emission wavelengths (S1 → S0) were then determined by computing the vertical transition energies from these S1-Min structures.
Results and discussion
Solution state photophysical properties
In order to explore the role of extended conjugation and alkyl chain variation on the solution state photophysical properties of the newly synthesized anthracene–cyanostyrene derivatives 2 and 3, we investigated the photophysical properties of these compounds from their dilute solutions in THF (1 µM) (Fig. 2). The UV-visible absorption spectra of compounds 3a and 3b showed comparable absorption spectra with maxima centered around 397 nm with shoulder bands centered at 378 nm and 342 nm, respectively. The compounds 2a and 2b also exhibited similar absorption spectra with maxima centered at 409 nm (see Table 1). The emission spectra were recorded from the respective THF solution at room temperature and upon 370 nm excitation, 2a and 2b exhibited emission bands at 415 and 431 and a major charge transfer (CT) band at 545 nm. In homologues 3, the emission spectra showed a single band at 485 nm with high quantum yield values (ΦF) of 0.87 and 0.82, respectively. Conversely, compound 2 showed moderate ΦF of around 0.05. This enhancement in ΦF and life-time values (Table 1) is ascribed to the incorporation of the spacer group phenyl ring, which facilitates enhanced ICT compared to 2a and 2b.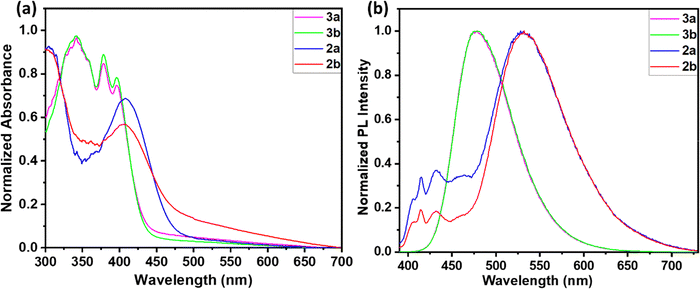 | ||
| Fig. 2 Normalized (a) UV-visible absorption and (b) emission spectra of the compounds 2a-3b recorded from THF solutions (1 µM), λex = 370 nm. | ||
| Compounds | λ abs (nm) | λ em (nm) | τ (ns) | Φ F |
|---|---|---|---|---|
| 2a | 408 | 545 | 1.22 | 0.05 |
| 2b | 409 | 553 | 0.75 | 0.06 |
| 3a | 342, 379, 397 | 485 | 2.72 | 0.87 |
| 3b | 343, 378, 398 | 485 | 2.68 | 0.82 |
Solvatochromism
To examine how D–π–A features and solvent polarity influence the photoluminescence, we recorded the emission spectra of compounds 2 and 3 in solvents ranging from hexane to DMF. Fig. 3(a) and (b) show the photoluminescence spectra recorded for the compounds 2a and 2b in solvents with varying polarity from hexane to DMF. The photoluminescence spectra of these compounds in all the solvents showed three distinct emission bands. The high-energy minor shoulder peaks observed at 415 and 431 nm appear consistently, irrespective of the solvent used.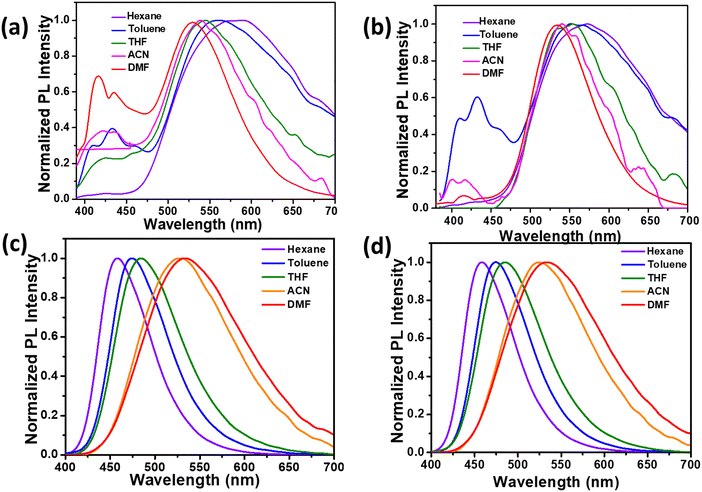 | ||
| Fig. 3 Normalized emission spectra of (a) 2a, (b) 2b, (c) 3a, and (d) 3b in solvents with different polarities. | ||
Conversely, the band corresponding to the CT state showed a blue shift (584 nm to 524 nm) on increasing solvent polarity from hexane to DMF. The observed negative solvatochromic shift in 2 implies that the dipole moment in the excited state is lower compared to the corresponding ground state. In contrast, the emission spectra recorded for the compound 3 (see Fig. 3(c) and (d)) for solvents of varying polarity showed a single band corresponding to ICT. The single emission band ranging from 460 nm (in hexane) to 535 nm (in DMF), along with reduced intensity on increasing solvent polarity, clearly indicates that the emissions are exclusively from the CT state. Furthermore, we compared the solvent dependent intensity variation in both the series of compounds by taking 2b and 3b as representatives (see Fig. S8). Despite the contrasting solvatochromic photoluminescence behaviour, the spectra of the compounds showed a gradual decrease in the emission intensity in response to increased solvent polarity attributed to the stabilization of the CT state.40,41 A detailed comparison of the quantum yield in these molecules showed significant differences among the series.
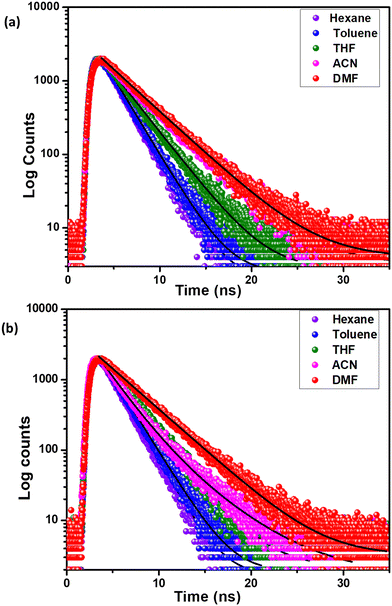
Interestingly, the compound 3 showed approximately 3 to 10 times enhancement in the quantum yield in different solvents in comparison to compound 2 (see Table 2). The molecules 2a and 2b showed reasonable quantum yield in nonpolar solvents hexane (0.24 and 0.25) and toluene (0.17 and 0.18), respectively, while in polar solvents the compounds showed negligible quantum yield values indicating the stabilization of the CT states. The quantum yield enhancement in molecules 3a and 3b in comparison to molecules 2a and 2b indicates the influence of ICT in determining the photoluminescence of these molecules. The compounds 2a and 2b in polar solvents like acetonitrile and DMF showed poor emission and therefore the lifetime was not recorded with reliability. It is noteworthy that the molecules 2a and 2b showed a decrease in lifetime and quantum yield on increasing the solvent polarity, while molecules 3a and 3b showed a constant increase in lifetime (see Fig. 4) moving from nonpolar to polar solvents (see Table 2). In general, the radiative decay constants for all the compounds showed a diminishing trend on increasing the solvent polarity indicating the stabilization of the CT states in polar solvents.
| Compound | Solvent | λ abs (nm) | λ em (nm) | τ (ns) | Φ F | k r | k nr |
|---|---|---|---|---|---|---|---|
| 2a | Hexane | 407 | 584 | 2.57 | 0.24 | 0.09 | 0.296 |
| Toluene | 408 | 560 | 1.53 | 0.17 | 0.11 | 0.542 | |
| THF | 408 | 545 | 1.22 | 0.05 | 0.04 | 0.779 | |
| Acetonitrile | 406 | 539 | — | 0.01 | — | — | |
| DMF | 410 | 524 | — | 0.05 | — | — | |
| 2b | Hexane | 407 | 572 | 1.90 | 0.25 | 0.13 | 0.395 |
| Toluene | 410 | 564 | 1.60 | 0.18 | 0.11 | 0.513 | |
| THF | 408 | 553 | 0.75 | 0.06 | 0.08 | 1.253 | |
| Acetonitrile | 406 | 541 | — | 0.01 | — | — | |
| DMF | 411 | 535 | — | 0.05 | — | — | |
| 3a | Hexane | 376, 395 | 458 | 1.99 | 0.80 | 0.40 | 0.100 |
| Toluene | 379, 400 | 474 | 2.14 | 0.79 | 0.37 | 0.098 | |
| THF | 379, 397 | 485 | 2.72 | 0.87 | 0.32 | 0.048 | |
| Acetonitrile | 378, 396 | 525 | 3.70 | 0.80 | 0.22 | 0.054 | |
| DMF | 380, 400 | 533 | 3.78 | 0.77 | 0.20 | 0.061 | |
| 3b | Hexane | 375, 395 | 460 | 1.97 | 0.75 | 0.38 | 0.127 |
| Toluene | 377, 397 | 475 | 2.09 | 0.80 | 0.38 | 0.096 | |
| THF | 378, 398 | 485 | 2.68 | 0.82 | 0.31 | 0.067 | |
| Acetonitrile | 378, 397 | 523 | 2.83 | 0.81 | 0.29 | 0.067 | |
| DMF | 380, 400 | 535 | 3.74 | 0.78 | 0.21 | 0.059 |
Solid state photoluminescence
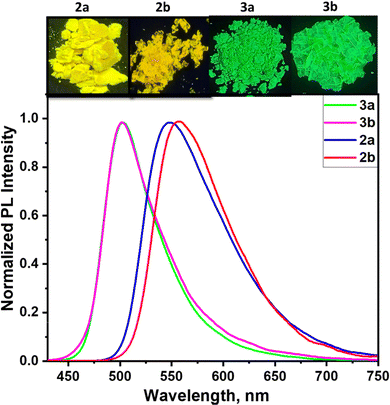
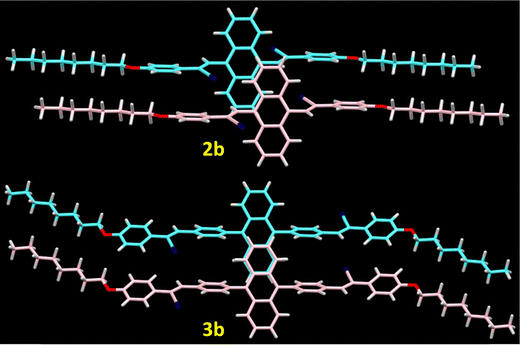
Following the distinct solution state properties of the newly synthesized compounds, the investigation focused on evaluating the solid-state luminescent behaviour by analysing their photoluminescence spectra recorded from crystalline samples. The combined spectra are presented in Fig. 5 with the corresponding photoluminescence data summarized in Table 3. The photoluminescence spectra of both compounds 2 and 3 showed significant difference in the photoluminescence maxima in response to the molecular packing and difference in conjugation. Despite the presence of a phenyl π-spacer in 3a and 3b, the molecules 2a and 2b showed maximum red shifted emission from their crystalline samples. The red-shifted emission in 2a and 2b could be due to the molecular aggregation in the solid state.42A detailed analysis using single-crystal X-ray diffraction studies revealed the molecular arrangements in the crystals of compounds 2b and 3b (Fig. 6). Both 2b and 3b showed different levels of twist in the molecular geometry with twist angles of 81° and 69°, respectively. As seen in Fig. S9, molecule 2b showed C–H⋯N and C–H⋯O interaction (H⋯N, 2.549 Å and H⋯O 2.472 Å) and π⋯π stacking interaction involving the edges of the anthracene with a distance of 3.686 Å. The intermolecular interactions in 3b include a C–H⋯π (2.873 Å) interaction along with π⋯π (3.593 Å) stacking involving the terminal phenyl rings. In comparison, molecule 2b showed a slipped molecular arrangement comprising a J-aggregate along the molecular long axis, while molecule 3b resulted in a pair of aggregates slipped along the molecular short axis (Fig. 6). The molecule 3b with non-cofacial π–π stacking gives rise to strong orbital overlap between the aromatic rings, leading to red shifted emission compared to the spectra recorded from THF solution. The red-shifted absorption in the solid state compared to solutions confirms J-aggregate formation in the solid state (Fig. S10). In both these molecules, the intramolecular twist restricts the cofacial π–π stacking limiting the aggregation caused quenching, where a similar observation was made in the case of 9,10-distyrylanthracene derivatives.38,43
Aggregation induced emission (AIE)
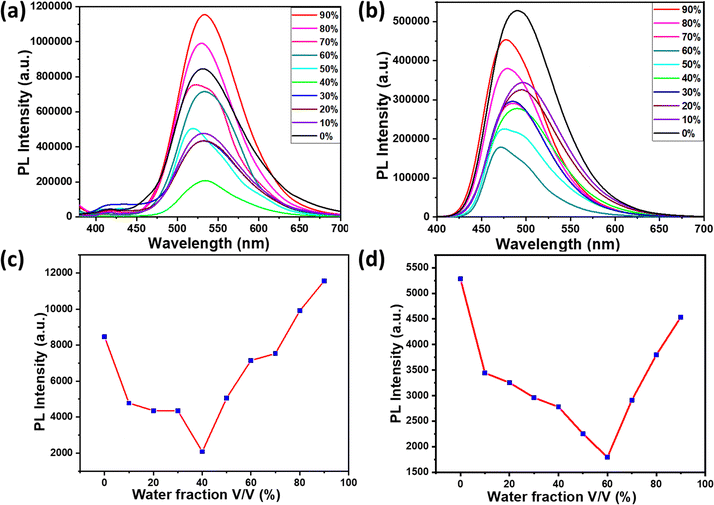
The study further explored the aggregation-induced emission (AIE) in both the classes of compounds, taking 2b and 3b as representative molecules. The aggregation properties of the compounds were investigated by comparing the emission spectra recorded from respective THF solutions containing different water fractions. Fig. 7(a) shows the emission spectra recorded for 2a from the THF–water mixtures. Up to 40% water fraction, the compound 2b showed a sharp decrease in spectral intensity of the CT band. The decreased emission intensity along with blue shift on increasing the water fraction can be attributed to the stabilization of the HOMO on increasing the solvent polarity. Increasing the water fraction above 40% showed an increase in emission intensity with a slight blue shift compared to the emission spectrum recorded from THF solution, where this is clear evidence of aggregation-induced emission enhancement (AIEE) in compound 2b (see Fig. 7(c)). The compound 3b on increasing water fraction showed a gradual decrease in emission intensity with a red shift as represented in solvent-dependent studies. A further increase of water fraction above 60% resulted in blue-shifted emission with enhanced emission intensity (see Fig. 7(b) and (d)), where this observation can be ascribed to aggregation induced emission (AIE) in these molecules. The blue shifted emission band in 2a on aggregation can be attributed to the formation of aggregates similar to H-aggregates, while the redshifted emission in compound 3b is originating from aggregates similar to J-aggregates.44–46Stimuli responsive emission
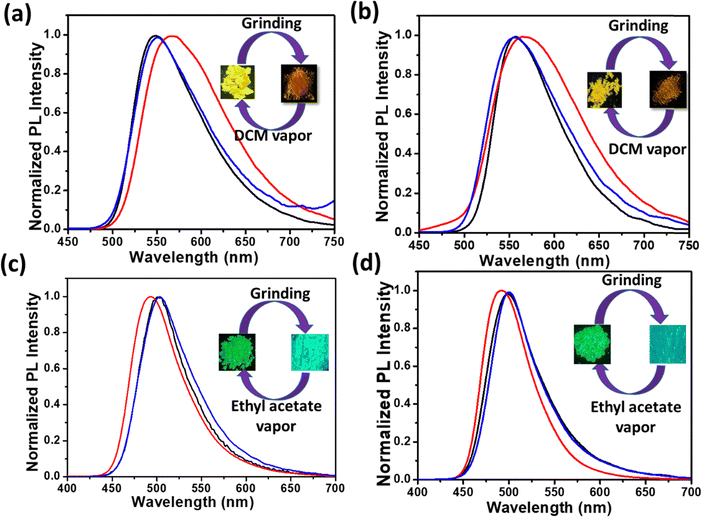
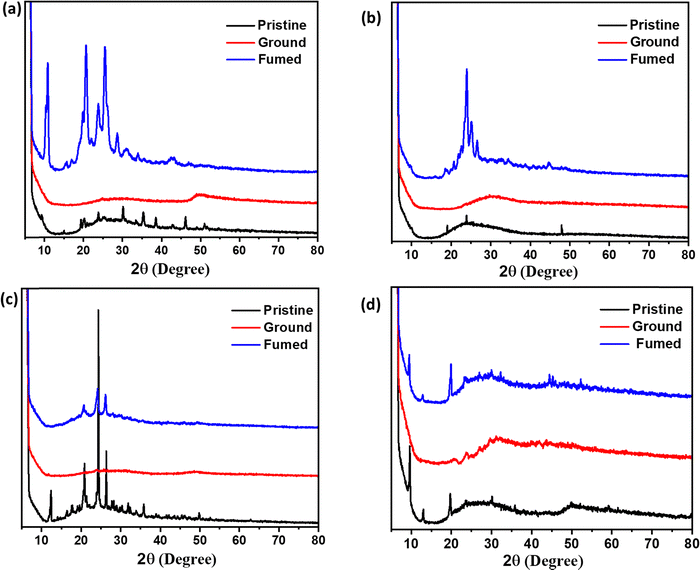
The newly synthesized compounds were explored for multi–stimuli responsive photoluminescence properties. All the compounds showed mechanofluorochromism (MFC) in response to applied stress. To test the possible mechanofluorochromic properties of 2a and 2b, the solid-state photoluminescence changes of the compounds were examined before and after grinding. The emission spectrum of 2a exhibited a red shifted emission (Δλem = 20 nm) along with an emission colour change of yellow to orange indicating an obvious MFC (see Fig. 8(a)). A subsequent fuming of the ground sample with dichloromethane vapours converted the ground sample from orange to yellow emission (see Fig. 8(b)). In contrast, the MFC studies of compounds 3a and 3b showed blue-shifted emission (Δλem = 12 nm) compared to the pristine samples accompanied by a change in emission colour from green to cyan. The ground samples on fuming with ethyl acetate vapours resulted in recovery of emission analogous to the emission of the pristine sample (Fig. 8(c) and (d)).In order to explore the structural origin of the stimuli-responsive emission of all the compounds, powder X-ray diffraction studies were carried out on different phases. Fig. 9(a) and (b) show the powder X-ray diffractograms recorded from different phases of 2a and 2b. The pristine samples of both the compounds were crystalline in nature as indicated by the sharp peaks in the diffractogram. The compounds on grinding resulted in crystal to amorphous transitions as represented by broad featureless diffractograms in Fig. 9(a) and (b). The diffractogram recorded from the DCM fumed sample in both cases showed peaks corresponding to solvent-induced crystallisation similar to the pristine sample. Similarly, the diffractograms of 3a and 3b also indicate the crystalline nature of their pristine samples. The ground samples showed the formation of amorphous phase under applied stress. Interestingly, the ground samples on fuming with ethyl acetate showed recovery of the pristine phase as indicated by the sharp peaks resembling those of the pristine samples. This study demonstrates the distinct multiple stimuli-responsive nature of the two series of compounds, especially with sensitivity towards vapours of dichloromethane and ethyl acetate.
The contrasting stimuli responsive behaviour of compounds 2 and 3 is further evidenced by their distinct photophysical properties. Table 3 shows the quantum yield, fluorescence lifetime (see Fig. S11) and corresponding radiative and nonradiative decay constants for the different phases of each compound. From the tabulated data, it is evident that on grinding the photoluminescence lifetime and quantum yield decrease as a result of the introduction of new decay pathways. In all the pristine samples, the nonradiative decay is prominent as indicated by nonradiative decay constants (knr). On applying mechanical stress, the radiative decay constants for compounds 2a and 2b showed a substantial decrease that may be due to a more disordered arrangement of molecules in the amorphous state. In contrast, the pristine and ground phases of 3a and 3b showed comparable radiative and nonradiative decay constants indicating the similar molecular arrangement in the pristine and ground phases.
| Compounds | λ em (nm) | Φ F | τ F (ns) | k r (ns−1) | k nr (ns−1) | |
|---|---|---|---|---|---|---|
| 2a | Pristine | 548 | 0.24 | 3.1 | 0.077 | 0.25 |
| Ground | 568 | 0.13 | 3.9 | 0.033 | 0.22 | |
| 2b | Pristine | 555 | 0.10 | 3.3 | 0.030 | 0.27 |
| Ground | 568 | 0.07 | 3.7 | 0.019 | 0.25 | |
| 3a | Pristine | 503 | 0.31 | 3.9 | 0.079 | 0.18 |
| Ground | 492 | 0.26 | 3.0 | 0.087 | 0.25 | |
| 3b | Pristine | 501 | 0.36 | 4.2 | 0.085 | 0.15 |
| Ground | 490 | 0.31 | 3.7 | 0.087 | 0.19 |
Quantum chemical calculations
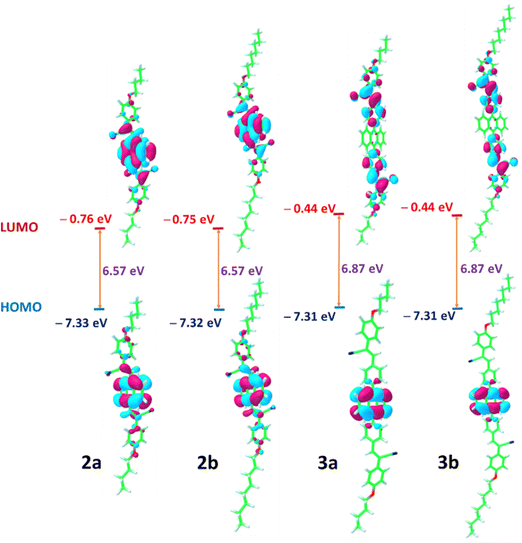
Additional insights into the structural and photophysical properties of the synthesized compounds were gained through DFT and TD-DFT calculations, carried out at the ωB97-XD/6-311+G(d) level of theory.47 The longer λabs values corresponding to the lowest energy transition (S0 → S1) observed for 2, compared to 3, can be rationalized based on their optimized geometries and molecular electrostatic potential (MESP) analyses (Fig. 10–12).48 The optimized geometries show good agreement with the corresponding crystal structures of 2 and 3. In compound 3, the phenyl ring adopts an almost perpendicular orientation relative to the linked anthracene moiety, which disrupts the extent of π-conjugation and leads to a slightly higher HOMO–LUMO gap compared to compound 2.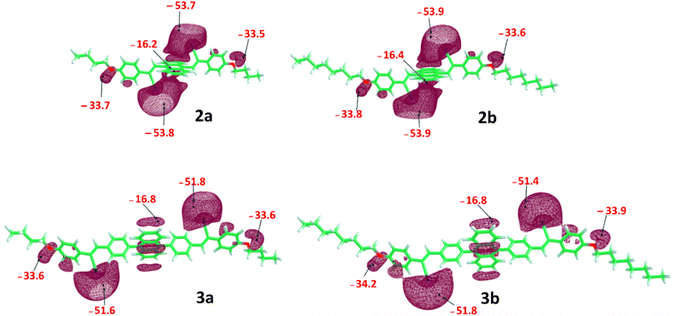 | ||
| Fig. 10 MESP minimum values (Vmin in kcal mol−1) shown in the corresponding isosurface plots (at −12.5 kcal mol−1). | ||
The MESP topology analysis offers a visual representation of the π-electron cloud and lone pair regions in a molecule.49,50 The minimum electrostatic potential values (Vmin) serve as a useful quantitative descriptor for assessing the electron density in these regions.51,52 The MESP plot depicted in Fig. 10 showed that the Vmin for the –CN region in compound 2 is more negative (−53.7 to −53.9 kcal mol−1) than that observed in compound 3 (−51.4 to −51.8 kcal mol−1). In addition, the Vmin value around the central anthracene segment in compound 3 is slightly more negative than that in compound 2, indicating a reduced delocalization of electron density in compound 3 relative to 2, which in turn influences the frontier molecular orbital (FMO) energy levels. The HOMO and LUMO distributions of compound 2 are localized predominantly on the central anthracene unit and cyanostilbene moieties. Instead, the HOMO of compound 3 is largely confined to the anthracene moiety, while the LUMO extends over the cyanostilbene groups, indicating the presence of ICT. The HOMO–LUMO energy gap remains essentially unchanged upon alkyl chain modification, with comparable values observed for compounds 2a and 2b, and a similar trend observed for compounds 3a and 3b (Fig. 11).
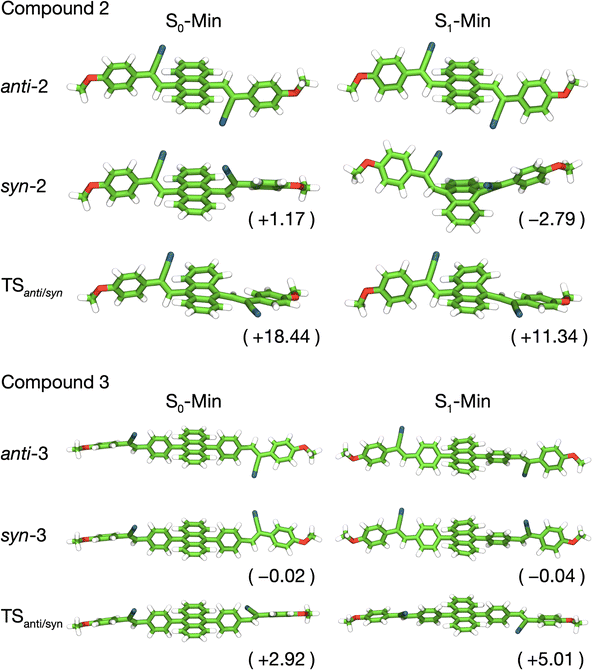
We further investigated the conformational landscapes in the ground (S0) and first singlet excited (S1) states of the synthesized compounds (Fig. 12). To reduce computational cost, the calculations were performed on simplified model structures by using methoxy groups for the alkyl chains. The calculations showed that both compounds 2 and 3 can exist as two primary conformers, syn and anti, based on the orientation of their terminal cyano groups. The optimized ground state geometries are referred to as S0-Min in Fig. 12. A critical difference between the two compounds emerges from the rotational barrier between these conformers in the ground state. For compound 3, the calculated activation barrier for the anti → syn isomerization is merely 2.92 kcal mol−1. This low barrier allows for rapid interconversion at room temperature, meaning that the syn and anti-conformers, which are nearly isoenergetic (ΔG = −0.02 kcal mol−1), exist as a dynamic equilibrium mixture. In stark contrast, the corresponding barrier for compound 2 is 18.44 kcal mol−1. This substantial barrier effectively prevents rotation at room temperature, indicating that the syn and anti-conformers of compound 2 are configurationally locked isomers. Given that the anti-2 conformer is thermodynamically more stable by 1.17 kcal mol−1, it is the predominant species, and photoexcitation will occur from this ground-state population.
In the S1 state, the potential energy surface reveals a complex landscape. While compound 3 shows minimal geometric changes upon excitation, compound 2 exhibits a dramatic transformation. The syn-2 conformer, in particular, undergoes a profound structural change, adopting a significantly bent and non-planar geometry where the central anthracene core is distorted out of its plane. This distorted structure is the thermodynamic global minimum on the S1 potential energy surface for compound 2. The optimized geometries of all conformers in the S1 state, referred to as S1-Min, are shown in Fig. 12.
These computational findings were compared with the experimental absorption and emission spectra (Table S2). For compound 3, which exists as a rapidly equilibrating mixture, the calculated properties of the anti-conformer (absorption at 357 nm, emission at 459 nm) are in good agreement with the experimental data. The calculated blue shift observed for compound 3 relative to compound 2 is consistent with the experimental results, as illustrated in the simulated absorption spectrum shown in Fig. S12. The small Stokes shift is consistent with its rigid, largely planar structure in both the S0 and S1 states.
For compound 2, the calculated absorption for the dominant anti-2 conformer (384 nm) matches the experimental spectrum well. Our calculations predict that emission from the bent, thermodynamically stable syn-2 S1-Min structure would appear at 674 nm. However, this long-wavelength emission is absent in the experimental spectrum. This absence strongly implies that the excited-state isomerization from anti-2 to syn-2 is kinetically hindered, likely by a high activation barrier on the S1 surface, and thus does not occur. Therefore, the major CT emission band observed for compound 2 at approximately 530 nm may be attributed to emission from the locally excited state of the anti-2 conformer. Our calculated emission wavelength for anti-2 is 537 nm, which closely matches the experimental result. The large Stokes shift observed for 2 can thus be attributed to the significant geometric relaxation of the anti-2 conformer itself within its own S1 potential well, without undergoing isomerization. Finally, a minor shoulder is observed in the experimental emission of 2 around 400–450 nm. This higher-energy emission is not assigned to the fully relaxed S1 state. It may be due to emission from the initially populated, unrelaxed excited state of anti-2, occurring prior to complete geometric relaxation, a phenomenon consistent with hot luminescence. Further photophysical studies would be required to definitively assign this feature.
In the present investigation, the comparison of the emission properties of all the molecules rules out the effect of the alkyl chain variation in modulating the photophysical properties in the solution state. On the other hand, the presence and absence of π-spacer impart distinct photophysical properties in both solution and the solid state. In addition to the extended conjugation the π-spacer in compound 3 provides scope for intramolecular free rotation. Meanwhile, the directly linked donor–acceptor in compound 2 results in a conformationally locked state with distinct photophysical properties including negative solvatochromism. The intramolecular twist in both the molecules played a pivotal role in aggregation induced emission by preventing cofacial π-stacking and thereby aggregation caused quenching. Both the series showed unique mechanofluorochromism along with vapochromic reversibility originating from the crystal to amorphous transition and vice versa. With the help of the present molecular design, we demonstrated the impact of conformational restriction on the intramolecular charge transfer properties of anthracene–cyanostyrene donor acceptor systems. In line with these observations, our findings substantiate the role of spacer engineering as a powerful strategy to fine tune the photophysical behaviour of the donor–acceptor systems.
Conclusions
In summary, the present study demonstrates the markedly distinct photophysical properties of anthracene–cyanostilbene derivatives, strongly affected by the nature of the π-spacer. The newly synthesized compounds, 2 (lacking a phenyl spacer) and 3 (containing a phenyl spacer), showed pronounced differences in their fluorescence emission, lifetime, quantum yield, and external stimuli response. Notably, variation in alkyl chain length exerted minimal influence on the photophysical properties, underscoring the dominant effect of π-conjugation tuning via spacer modification. Crystallographic analysis revealed distinct molecular packing and intermolecular interactions that correlate with their emissive characteristics, while theoretical calculations provided insight into the electronic structures and excited-state dynamics. DFT/TD-DFT calculations were employed to investigate the electronic structures and conformational landscapes of compounds 2 and 3 in their S0 and S1 states. The remarkable photoluminescence properties of these compounds offer promising opportunities for further investigation towards sensing and optoelectronic applications.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data underlying this study are available in the supplementary information (SI). The supplementary information file contains synthesis and characterization details of compounds 2a to 3b, crystallographic information table of 2b and 3b, molecular structure and details related to intermolecular interactions, and computational results (TD-DFT studies and conformational analysis), Photophysical studies of compounds 2a to 3b. See DOI: https://doi.org/10.1039/d5cp03033a.CCDC 2417412 and 2417413 contain the supplementary crystallographic data for this paper.53a,b
Acknowledgements
CF acknowledges the Department of Science and Technology (DST) for funding research under Women Scientist Scheme-A (WOS-A), (DST/WOS-A/CS-126/2021). The authors thank DST-FIST, (SR/FIST/COLLEGE/2019/735) Government of India, for providing instrumentation support. We also acknowledge the University of Calicut for access to its instrumentation facilities. RT thanks RUSA-159/RUSA-SPD/2017, Govt. of India, for the research support. We gratefully acknowledge Dr Ratheesh K Vijayaraghavan (IISER- Kolkata) and Dr Chinna Ayya Swamy P. (NIT Calicut) for various photophysical measurements. The quantum chemical calculations were performed at the Research Center for Computational Science, Okazaki, Japan.References
- O. Ostroverkhova, Organic optoelectronic materials: mechanisms and applications, Chem. Rev., 2016, 116, 13279–13412 CrossRef CAS PubMed.
- A. C. Grimsdale, K. Leok Chan, R. E. Martin, P. G. Jokisz and A. B. Holmes, Synthesis of light-emitting conjugated polymers for applications in electroluminescent devices, Chem. Rev., 2009, 109, 897–1091 CrossRef CAS PubMed.
- M. Pope and C. E. Swenberg, Electronic processes in organic crystals and polymers, Oxford University Press, 1999 Search PubMed.
- B.-K. An, J. Gierschner and S. Y. Park, π-Conjugated cyanostilbene derivatives: A unique self-assembly motif for molecular nanostructures with enhanced emission and transport, Acc. Chem. Res., 2012, 45, 544 CrossRef CAS PubMed.
- X. Chen, D. Tan and D.-T. Yang, Multiple-boron–nitrogen (multi-BN) doped π-conjugated systems for optoelectronics, J. Mater. Chem. C, 2022, 10, 13499–13532 RSC.
- C. Femina, T. Yamagami, N. Yamamoto, R. Thomas and P. K. Sajith, Theoretical insights into aggregation-induced emission of bis(cyanostyryl)pyrrole derivatives, Phys. Chem. Chem. Phys., 2025, 27, 8478–8487 RSC.
- W. Qin, Z. Yang, Y. Jiang, J. W. Y. Lam, G. Liang, H. S. Kwok and B. Z. Tang, Construction of efficient deep blue aggregation-induced emission luminogen from triphenylethene for nondoped organic light-emitting diodes, Chem. Mater., 2015, 27, 3892–3901 CrossRef CAS.
- S.-Y. Chen, Y.-W. Chiu and G.-S. Liou, Substituent effects of AIE-active α-cyanostilbene-containing triphenylamine derivatives on electrofluorochromic behavior, Nanoscale, 2019, 11, 8597–8603 RSC.
- Y. Sagara, M. Karman, E. Verde-Sesto, K. Matsuo, Y. Kim, N. Tamaoki and C. Weder, Rotaxanes as mechanochromic fluorescent force transducers in polymers, J. Am. Chem. Soc., 2018, 140, 1584–1587 CrossRef CAS.
- Y. Wang, X. Tan, Y.-M. Zhang, S. Zhu, I. Zhang, B. Yu, K. Wang, B. Yang, M. Li, B. Zou and S. X.-A. Zhang, Dynamic behavior of molecular switches in crystal under pressure and its reflection on tactile sensing, J. Am. Chem. Soc., 2015, 137, 931–939 CrossRef CAS PubMed.
- C. Femina, P. K. Sajith, K. Remya, R. Thomas and R. V. Solomon, Theoretical insights into the structural and optical properties of D−π–A-based cyanostilbene systems of α and β variants, ACS Omega, 2024, 9, 22764–22776 CrossRef CAS PubMed.
- S. Gouthaman, A. Jayaraj, M. Sugunalakshmi, G. Sivaraman and C. A. S. P, Supramolecular self-assembly mediated aggregation-induced emission of fluorene-derived cyanostilbenes: multifunctional probes for live cell-imaging, J. Mater. Chem. B, 2022, 10, 2238–2250 RSC.
- G. Huang, X. Chang, Y. Jiang, B. Lin, B. S. Li and B. Z. Tang, Multi-stimuli responsive cyanostilbene derivatives: pH, amine vapor sensing and mechanoluminescence, Mater. Chem. Front., 2020, 4, 1720–1728 RSC.
- Y. Zhang, H. Li, G. Zhang, X. Xu, L. Kong, X. Tao, Y. Tian and J. Yang, Aggregation-induced emission enhancement and mechanofluorochromic properties of α-cyanostilbene functionalized tetraphenyl imidazole derivatives, J. Mater. Chem. C, 2016, 4, 2971–2978 RSC.
- B. Wang and C. Wei, Stimuli-responsive fluorescence switching of cyanostilbene derivatives: ultrasensitive water, acidochromism and mechanochromism, RSC Adv., 2018, 8, 22806–22812 RSC.
- G. Fan and D. Yan, Positional isomers of cyanostilbene: two-component molecular assembly and multiple-stimuli responsive luminescence, Sci. Rep., 2014, 4, 4933 CrossRef CAS PubMed.
- X. Wang, Z. Ding, Y. Ma, Y. Zhang, H. Shang and S. Jiang, Multi-stimuli responsive supramolecular gels based on a D–π–A structural cyanostilbene derivative with aggregation induced emission properties, Soft Matter, 2019, 15, 1658–1665 RSC.
- C. Dou, L. Han, S. Zhao, H. Zhang and Y. Wang, Multi-stimuli-responsive fluorescence switching of a donor−acceptor π-conjugated compound, J. Phys. Chem. Lett., 2011, 2, 666–670 CrossRef CAS.
- S. Rana, S. Vaidyanathan and S. Patel, Aggregation induced emission (AIE) based donor–π–acceptor fluorophores: an approach to fabricate acidochromic sensors and white light emitting diodes, J. Mater. Chem. C, 2024, 12, 14148–14164 RSC.
- G. Qian, B. Dai, M. Luo, D. Yu, J. Zhan, Z. Zhang, D. Ma and Z. Y. Wang, Band gap tunable, donor−acceptor−donor charge-transfer heteroquinoid-based chromophores: near infrared photoluminescence and electroluminescence, Chem. Mater., 2008, 20, 6208–6216 CrossRef CAS.
- B. M. Samrudhi, H. V. Barkale, D. Devadiga, N. Dey and T. N. Ahipa, Donor-acceptor-based conjugated fluorescent probes for volatile acid detection, ChemistrySelect, 2024, 9, e202401790 CrossRef.
- J. Zhao, C. Yao, M. U. Ali, J. Miao and H. Meng, Recent advances in high-performance organic solar cells enabled by acceptor–donor–acceptor–donor–acceptor (A–DA′D–A) type acceptors, Mater. Chem. Front., 2020, 4, 3487–3504 RSC.
- C. Femina, M. Shanthil, P. K. Sajith and R. Thomas, Anthracene-incorporated cyanostilbene based donor–acceptor systems: intramolecular charge transfer and aggregation induced emission, New J. Chem., 2023, 47, 13810–13819 RSC.
- Z. Jiang, H. Zhao, W. Wu, K. Chen, H. Yu, T. Wang, X. Huang, N. Wang, L. Zhou and H. Hao, Multi-stimuli responsive organic polymorphic crystals: anisotropic elasticity and plasticity, mechanochromism and photomechanical motions, J. Mater. Chem. C, 2023, 11, 4375–4383 RSC.
- Z. Huang, F. Tang, F. He, L. Kong, J. Huang, J. Yang and A. Ding, Pyrene and triphenylamine substituted cyanostyrene and cyanostilbene derivatives with dual-state emission for high-contrast mechanofluorochromism and cell imaging, Org. Chem. Front., 2022, 9, 5118–5124 RSC.
- P. Athira, R. Nelliyulla Kappumchalil, A. R. Sachin, M. Yoosuf, R. Thomas and G. Gopakumar, Intramolecular charge transfer and stimuli-responsive emission in cholesterol-appended phenothiazine–cyanostyryl-based donor–acceptor systems, J. Phys. Chem. A, 2024, 128, 3935–3946 CrossRef CAS PubMed.
- S.-Y. Chen, M.-H. Pai and G.-S. Liou, Effects of alkyl chain length and anion on the optical and electrochemical properties of AIE-active α-cyanostilbene-containing triphenylamine derivatives, J. Mater. Chem. C, 2020, 8, 7454–7462 RSC.
- N. Kumar, D. Maity, S. Som and S. Banerjee, Multi-stimuli responsive cyanostilbene assemblies in aqueous media: temporal luminescence modulation through pH-feedback network, Small, 2025, 2503123 CrossRef CAS PubMed.
- L. Zhu and Y. Zhao, Cyanostilbene-based intelligent organic optoelectronic materials, J. Mater. Chem. C, 2013, 1, 1059–1065 RSC.
- C. Hang, H.-W. Wu and L.-L. Zhu, π-Conjugated cyanostilbene-based optoelectric functional materials, Chin. Chem. Lett., 2016, 27, 1155–1165 CrossRef CAS.
- H. Yu, W. Wu, H. Zhao, K. Chen, S. Li, M. Tan, T. Wang, X. Huang, N. Wang and H. Hao, Cyanostyrene derivative with multi-stimuli responsive properties: Multicolor- and high-color-contrast switching in response to force, heat and light, Dyes Pigm., 2023, 220, 111727 CrossRef CAS.
- J. Xiong, K. Wang, Z. Yao, B. Zou, J. Xu and X.-H. Bu, Multi-stimuli-responsive fluorescence switching from a pyridine-functionalized tetraphenylethene AIEgen, ACS Appl. Mater. Interfaces, 2018, 10, 5819–5827 CrossRef CAS PubMed.
- A. Afrin and P. Chinna Ayya Swamy, Symphony of light: AIE and MFC in carbazole-based cyanostilbenes, J. Mater. Chem. C, 2024, 12, 1923–1944 RSC.
- M. Sun, J. Liu, T. Jiang, J. Lin, M. Xu, Y. Zhao, Q. Liu, K. Chen and C. Zeng, A thiophene-substituted cyanostilbene derivative: Understanding the mechanism of its aggregation-induced emission, acidochromism and mechanochromism from the molecular level, J. Mol. Struct., 2025, 1337, 142262 CrossRef CAS.
- A. Shabashini, V. Ramar, B. Karthikeyan, M. K. Panda and G. C. Nandi, Design and synthesis of triphenylamine based cyano stilbenes for picric acid sensing and two photon absorption applications, ChemistrySelect, 2021, 6, 12300–12308 CrossRef CAS.
- D. Zhang, Y. Liu, H. Gao, Q. Chang and X. Cheng, α-Cyanostilbene and fluorene based bolaamphiphiles: synthesis, self-assembly, and AIEE properties with potential as white-light emissive materials and light-emitting liquid crystal displays, J. Mater. Chem. C, 2020, 8, 17474–17481 RSC.
- H. C. Zhang, E. Q. Guo, Y. L. Zhang, P. H. Ren and W. J. Yang, Donor−acceptor-substituted anthracene-centered cruciforms: synthesis, enhanced two-photon absorptions, and spatially separated frontier molecular orbitals, Chem. Mater., 2009, 21, 5125–5135 CrossRef CAS.
- J. He, B. Xu, F. Chen, H. Xia, K. Li, L. Ye and W. Tian, Aggregation-induced emission in the crystals of 9,10-distyrylanthracene derivatives: the essential role of restricted intramolecular torsion, J. Phys. Chem. C, 2009, 113, 9892 CrossRef CAS.
- M. J. Frisch, et al., Gaussian 16, Revision C.02, Gaussian, Inc., Wallingford CT, 2016 Search PubMed.
- A. R. Nair, C. Raksha, R. Heera, M. Gayathri Mohan, P. Manoj and A. Sivan, D-A-D/A chalcones with tunable optical characteristics: Synthesis, photophysical, electrochemical and theoretical investigations, J. Photochem. Photobiol., A, 2024, 451, 115511 CrossRef CAS.
- P. Sumsalee, P. Morgante, G. Pieters, J. Crassous, J. Autschbach and L. Favereau, Negative solvatochromism and sign inversion of circularly polarized luminescence in chiral exciplexes as a function of solvent polarity, J. Mater. Chem. C, 2023, 11, 8514–8523 RSC.
- S. Ma, S. Du, G. Pan, S. Dai, B. Xu and W. Tian, Organic molecular aggregates: From aggregation structure to emission property, Aggregate, 2021, 2, e96 CrossRef CAS.
- Z. Zhao, B. He and B. Z. Tang, Aggregation-induced emission of siloles, Chem. Sci., 2015, 6, 5347–5365 RSC.
- B. Hämisch and K. Huber, Mechanism and equilibrium thermodynamics of H- and J-aggregate formation from pseudo isocyanine chloride in water, Soft Matter, 2021, 17, 8140–8152 RSC.
- H. Piwoński, S. Nozue, H. Fujita, T. Michinobu and S. Habuchi, Organic J-aggregate nanodots with enhanced light absorption and near-unity fluorescence quantum yield, Nano Lett., 2021, 21, 2840–2847 CrossRef PubMed.
- Q. He, A. Basu, H. Cha, M. Daboczi, J. Panidi, L. Tan, X. Hu, C. C. Huang, B. Ding, A. J. P. White, J.-S. Kim, J. R. Durrant, T. D. Anthopoulos and M. Heeney, Ultra-narrowband near-infrared responsive J-aggregates of fused quinoidal tetracyanoindacenodithiophene, Adv. Mater., 2023, 35, 2209800 CrossRef CAS PubMed.
- J.-D. Chai and M. Head-Gordon, Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections, Phys. Chem. Chem. Phys., 2008, 10, 6615–6620 RSC.
- J. Tomasi, Use of the electrostatic potential as a guide to understanding molecular properties, in Chemical Applications of Atomic and Molecular Electrostatic Potentials, ed. P. Politzer and D. G. Truhlar, Springer, US, Boston, MA, 1981, pp. 257–294 Search PubMed.
- S. R. Gadre, C. H. Suresh and N. Mohan, Electrostatic potential topology for probing molecular structure, bonding and reactivity, Molecules, 2021, 26, 3289 CrossRef CAS PubMed.
- C. H. Suresh, G. S. Remya and P. K. Anjalikrishna, Molecular electrostatic potential analysis: A powerful tool to interpret and predict chemical reactivity, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2022, 12, e1601 CAS.
- T. M. Ismail, N. Mohan and P. K. Sajith, Theoretical study of hydrogen bonding interactions in substituted nitroxide radicals, New J. Chem., 2021, 45, 3866–3875 RSC.
- P. K. Anjalikrishna, S. R. Gadre and C. H. Suresh, Electrostatic potential for exploring electron delocalization in infinitenes, circulenes, and nanobelts, J. Org. Chem., 2023, 88, 4123–4133 CrossRef CAS PubMed.
- (a) CCDC 2417412: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2m4j1c; (b) CCDC 2417413: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2m4j2d.
| This journal is © the Owner Societies 2025 |