Self-regulatory factors of embryonic stem cells in co-culture with stromal cells enhance neural differentiation†
Abstract
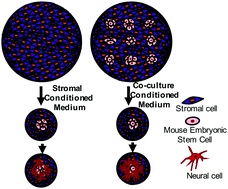
Embryonic stem cells (ESCs), due to their intrinsic capability to generate somatic cells of all three germ layers, are potential sources of neural cells for cell replacement therapies. However, the empirical differentiation protocols and the lack of mechanistic understanding of the neural differentiation of ESCs have limited the utility of ESCs as a developmental model or as a cell source for neural cell populations for replacement therapies. Co-culturing ESCs with stromal cells is one of the extensively used methods to induce neural differentiation. Despite several studies to identify neural inducing factors in stromal cell induced neural differentiation, the self-regulatory effects of ESCs in the neural differentiation process remain unexplored. For the first time, we elucidate the self-regulatory role of mESCs in their neural cell differentiation by supplementing conditioned media from differentiating mESCs to mESC-PA6 co-cultures and quantitatively evaluating the change in neural differentiation. Moreover, we use statistical tools to analyze the expression of various growth and trophic factors and distinguish the factors produced primarily by PA6 cells versus mESCs in co-culture. We observe that addition of the medium containing mESC-secreted factors to a single mESC colony co-cultured with PA6 cells significantly enhances the neural differentiation of mESCs compares to the medium extracted from the stromal cells only. Hierarchical clustering of gene expression data from PA6 and co-cultured mESCs segregates two groups of factors that are produced by the stromal cells and differentiating mESCs. Identifying the major soluble factors that drive and regulate the neural differentiation process in the mESC-PA6 co-culture niche will help understand molecular mechanisms of neural development. Moreover, it can be a major step toward developing novel protocols to differentiate stem cells with mESC derived factor supplementation without using feeder cells and with greater efficiency compared to existing approaches.


 Please wait while we load your content...
Please wait while we load your content...