Design of CO2 sorbents using functionalized fibrous nanosilica (KCC-1): insights into the effect of the silica morphology (KCC-1 vs. MCM-41)†
Abstract
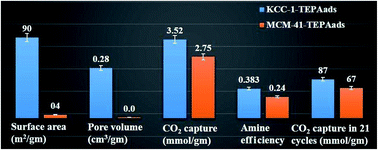
Hybrid materials by functionalization of fibrous nanosilica (KCC-1) were synthesized for efficient CO2 capture, using various amine molecules by physisorption (ads.) as well as covalent attachment. Their CO2 capture efficiency was studied using various parameters, such as CO2 capture capacity, kinetics of adsorption, stability of the sorbent, and regeneration-reuse properties. The best KCC-1 sorbent (i.e., KCC-1-TEPAads) was then studied for the effects of solvents, adsorption temperature and TEPA (tetraethylene pentamine) loading on its textural properties and CO2 capture efficiency and compared with its MCM-41 counterparts. The textural properties of MCM-41 sorbents were drastically affected with loss of more than 99% of their surface area and pore volume, while KCC-1 based sorbents were able to retain a good amount of surface area and pore volume. Notably, the KCC-1-based sorbents exhibited better CO2 capture capacity, rate of adsorption and stability than their MCM-41 counterparts (MCM-41/TEPAads). The total CO2 capture capacity after 21 cycles under humid conditions was 91.5 mmol g−1 for KCC-1-TEPAads but 73.1 mmol g−1 for MCM-41-TEPAads, indicating the superior capacity and stability of the KCC-1-based sorbents even under humid conditions. The better performance of the KCC-1-based sorbents was due to their unique fibrous morphology, accessible surface area and pore volume, which were retained even after functionalization. These results indicate the critical role of the silica morphology as well as the advantages of KCC-1 silica over MCM-41 for the design of efficient CO2 sorbents.
- This article is part of the themed collection: Emerging Investigators 2016: Novel design strategies for new functional materials


 Please wait while we load your content...
Please wait while we load your content...