Sustainable hydrogen production from electroplating wastewater over a solar light responsive photocatalyst
Abstract
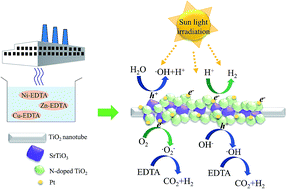
An environmentally friendly and sustainable photocatalytic hydrogen production system was successfully developed using ethylenediaminetetraacetic acid (EDTA) found in the wastewater from electroplating plants as the photo-excited hole scavenger and a solar light responsive multi-junction material as the photocatalyst. Additionally, a facile method of metal removal was established to increase the photocatalytic hydrogen production efficiency. The influences of the metal concentration, pH of the electroplating wastewater, and the photocatalyst concentration on the efficacy of hydrogen production were studied in detail. The results show that before metal removal, the unchelated metal ions and/or the metal–EDTA chelates block the active sites of the photocatalyst, resulting in suppressed adsorptions of H+ ions and/or H2O, which results in negligible hydrogen production efficiency. Through the metal removal process developed in this study, most of the metals could be removed and most of the EDTA could be retained. The removal efficiencies of copper, nickel, and zinc ions were all higher than 90%, facilitating better reaction between the liberated EDTA molecules and the photo-excited holes, improved charge separation, and enhanced hydrogen production efficiency. The system showed economically optimal hydrogen production when the reaction pH was maintained at pH = 6 and the photocatalyst concentration was maintained at 2 g L−1 under simulated sunlight irradiation.

 Please wait while we load your content...
Please wait while we load your content...