Effect of the ordered meso–macroporous structure of Co/SiO2 on the enhanced activity of hydrogenation of CO to hydrocarbons†
Abstract
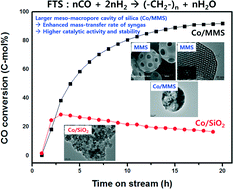
A hierarchically spherical and ordered meso–macroporous silica (MMS) support was applied for the hydrogenation reaction of CO to hydrocarbons using a cobalt-based Fischer–Tropsch synthesis (FTS) catalyst, which was synthesized using dual templates of polystyrene beads for the macroporous structures and an amphiphilic triblock copolymer positioned inside emulsion droplets for the mesoporous structures. A higher surface area with a bimodal pore size distribution on the cobalt-impregnated MMS (Co/MMS) revealed a higher catalytic activity and stability by enhancing the mass transport rate of the heavier hydrocarbons formed inside the macropores with a larger wax entrapping capacity due to their large pore cavity compared with the conventional SiO2 support. The enhanced CO conversion and C5+ selectivity while using the Co/MMS even under a higher space velocity were mainly attributed to the presence of heavy hydrocarbons formed during the FTS reaction in the larger regular macropores' entrapping cavity resulting in the suppression of the wax deposition on the active cobalt sites. Even though the wax deposition seems to be inevitable during the FTS reaction, it was also easily and efficiently removed by intermittently feeding liquid octane during the FTS reaction for an in situ regeneration of the regular meso–macroporous Co/MMS.

 Please wait while we load your content...
Please wait while we load your content...