Imine-tautomers of aminothiazole derivatives: intriguing aspects of chemical reactivities†
Abstract
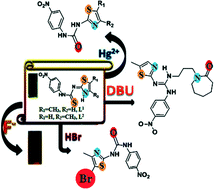
Intriguing reactivities of positional isomers 1-(5-methylthiazol-2-yl)-3-(4-nitrophenyl)thiourea (L1) and 1-(4-methylthiazol-2-yl)-3-(4-nitrophenyl)thiourea (L2) towards environment polluting ions such as mercuric, fluoride, bromide and solvents provide scope to study various self-assemblies formed under different conditions and allied applications. The solid state structures of L1 and L2 revealed that both adopt the imine form. However, the number of symmetry independent molecules in asymmetric units as well as the packing patterns of these two compounds have distinguishable features. The asymmetric unit of L1 has three symmetry non-equivalent molecules. The packing patterns of these two positional isomers differ as L1 has two arrays of dimeric self-assemblies in contrast to the single array found in the packing of L2. The reactivity of L1 and L2 with solvents was explored through crystal structure elucidation. The compound L1 forms solvates with N,N′-dimethylacetamide or dimethylsulphoxide. The structural analysis of solvates of L1 provided avenues to study conformational adjustments. An unusual example of a metastable solvate of L1 with N,N′-dimethylacetamide was isolated. A ring-opening reaction of 1,8-diazabicyclo [5.4.0] undec-7-ene with L1 has been observed. The compound L2 underwent hydrolysis cum bromination with hydrobromic acid in dimethylsulphoxide. The reactivities of L1 and L2 towards mercuric and fluoride ions contributed to chemodosimetric signal transductions. The study establishes the impact of subtle changes in structure, reaction and crystallization conditions, which can influence pre-designed crystal engineering.

 Please wait while we load your content...
Please wait while we load your content...