Facile simulation of carbon with wide pore size distribution for electric double-layer capacitance based on Helmholtz models†
Abstract
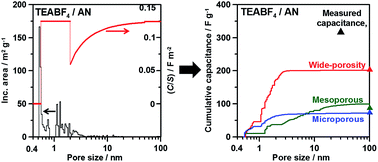
This study reports on a facile method based on Helmholtz models for simulating the electric double-layer capacitance of various forms of carbon in aqueous H2SO4 and KOH and organic tetraethylammonium tetrafluoroborate/acetonitrile electrolytes. The proposed method combines cylindrical pore models for macropores and mesopores with the slit-pore model for micropores exhibiting constant surface-based capacitance (C/S). The pore structures and pore size distribution of carbon are analyzed by using a method based on non-local density functional theory (NLDFT). We then used data related to the capacitance of microporous carbon to evaluate the constant C/S values produced by distinct electrolytes in carbon micropores and to determine the molecule-sieving effect of the micropores. The constant C/S values obtained from the micropores suggest that the effective static dielectric constant at the electrode–electrolyte interface is proportional to the thickness of the ion-solvating layer. The C/S values in mesopores decreased with a decrease in the pore size due to the effects of wall-curvature confinement. For an aqueous electrolyte, the C/S values in micropores are larger than those in mesopores and macropores due to the compactness of the ion-solvating layers, which account for the higher dielectric constant in the micropores. Our simulation results regarding the capacitance values of each carbon are in excellent agreement with experimental data, thereby verifying the reliability of the proposed model. This model is capable of providing reliable, precise predictions of capacitance values and also reveals the mechanism underlying the double-layer formation of distinct pores and the interfacial properties associated with capacitive performance.

 Please wait while we load your content...
Please wait while we load your content...