Design of a live biochip for in situ nanotoxicology studies: a proof of concept
Abstract
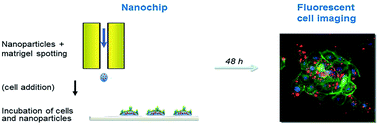
This paper highlights the way in which eukaryotic cell and bacteria based biochips are relevant for nanotoxicological risk evaluation. Here we define NP-biochips as formatted surfaces containing nanoparticles (NPs). They are simple devices which can easily be used to generate quantitative data expressing the effects of NPs on biological material in parallelized medium throughput assays. Firstly we dropped NPs and bacteria onto an agarose layer, using fluorescent bacteria in order to follow by imaging the effects of these NPs on bacterial growth. Secondly we embedded the targeted NPs at precise spot locations in a matrix on which eukaryotic cells can adhere, and followed cell growth. We used titanium dioxide as model NPs for the concept validation. Both types of NP-biochip are realized in order to pattern NPs in 50 or 100 dried 400 μm diameter spots on a glass plate, with less than 0.3% variation in concentration between spots. For anatase TiO2 NPs, we were able to record a non-toxic effect by measuring bacteria or eukaryotic cell survival. NPs are not internalized in bacteria; we thus used hyperspectral imaging to observe NPs on their surfaces. In contrast, NPs translocate in eukaryotic cells so we used fluorescent TiO2 and quantum dots to verify that NPs migrate from the NP-biochip matrix into bronchial cells. In order to illustrate the release of NP from the chip into the cell, we present the dose–response curve in the case of a toxic rutile TiO2 NP. These devices prevent cell and bacteria suffocation that is often observed in standard assays in wells due to NP precipitation. We believe that these tests realized on gel coated biochips are a rather realistic model for NP exposure in situ, imitating bacterial growth in biofilms and eukaryotic cells in tissues.

 Please wait while we load your content...
Please wait while we load your content...