Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Are there specific sub-classes of halogen bonds? A statistical analysis of the interactions in crystal structures†
Sławomir J.
Grabowski
 ab
ab
aFaculty of Chemistry, University of the Basque Country and Donostia International Physics Center (DIPC), P.K. 1072, 20080 Donostia, Spain. E-mail: slawomir_grabowski@ehu.eus
bIKERBASQUE, Basque Foundation for Science, 48011 Bilbao, Spain. E-mail: s.grabowski@ikerbasque.org
First published on 27th November 2025
Abstract
Different sub-classes of so-called halogen bonds are analysed, i.e. fluorine, chlorine, bromine and iodine bonds. These interactions are compared with analogous hydrogen bonds. For this purpose, Cambridge Structural Database (CSD) searches were performed to find specific structures where systems linked by C–X⋯O and C–X⋯N interactions occur (X is a halogen centre). Besides, CSD searches were carried out to find the corresponding structures with C–H⋯O and C–H⋯N interactions. These types of halogen bonds and two types of hydrogen bonds are compared with the use of statistical approaches. The X⋯O/N and H⋯O/N bond distances as well as the C–X⋯O/N and C–H⋯O/N bond angles are also compared. Four complex samples analysed theoretically (MP2/6-311++G(d,p) calculations were performed) in an earlier study, which are linked by I–I⋯N, N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–I⋯N, Br–Br⋯N and N
C–I⋯N, Br–Br⋯N and N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Br⋯N interactions, are also considered in this study. For these theoretical results, two parameters are analysed statistically: the binding energy and electron density at the I⋯N or Br⋯N bond critical points. It is also noted in this study that the names applied commonly for various types of interactions so far can be replaced by other more informative names.
C–Br⋯N interactions, are also considered in this study. For these theoretical results, two parameters are analysed statistically: the binding energy and electron density at the I⋯N or Br⋯N bond critical points. It is also noted in this study that the names applied commonly for various types of interactions so far can be replaced by other more informative names.
Introduction
The analysis of intra- and inter-molecular interactions is often a subject of various studies since they are crucial in numerous chemical phenomena as well as physical and biological processes.1–6 Many of these interactions may be also treated as the preliminary stages of chemical reactions such as the proton transfer in the case of hydrogen bonds7 or the SN2 reaction in the case of tetrel bonds.8 Furthermore, it was claimed in early studies that molecular species or molecular fragments that change during specific reactions may be found in crystal structures, and each fragment from the structure corresponds to the frozen stage of the chemical reaction considered.9–12 In other words, if there is a sufficient number of crystal structures containing a given molecular fragment, it is possible to trace the course of a specific chemical reaction, more precisely to trace the geometrical changes occurring during the reaction under consideration. This type of analysis is known as the structure-correlation method.13Owing to the above-mentioned issues, there has been an increase in the studies on interactions, and their classifications. For example, the majority of interactions that initiate chemical reactions may be classified as the Lewis acid–Lewis base interactions, where noticeable or even significant electron charge transfers are observed from the base unit to the acid unit.6,14 There are various concepts to explain the mechanisms of interactions. For example, it was explained that there are areas of electron charge depletion in molecular species, σ-holes and π-holes, often characterised by a positive electrostatic potential (EP) in the case of sufficient electron charge depletion.4,15–17 These areas may act as electrophiles in interactions and chemical reactions. Hence, interactions may be categorized into σ-hole and π-hole bonds.4,15–17
However, the names of interactions usually come from the name of the center acting as the Lewis acid site, and it will be the name of the element or the name of the group in the periodic table.6,14,18 Hence, names such as the hydrogen bond, lithium bond and beryllium bond are known, and also the halogen bond, pnictogen bond and triel bond, respectively. Certainly, not all the names of interactions are mentioned here. These names of interactions are used most often, whereas names derived from the Lewis base centre are rather rare, where one may mention the hydride bond19–21 or halide bond.22 The discussion on the multitude and variety of names of interactions as well as their diversity has been a subject of several recent studies.14,23–26 The proposal to simplify and unify the names of interactions has also been the subject of a recent study,27 which is discussed later in this article.
The so-called halogen bonds are the subject of this study. The first studies on halogen bonds, although this term was not used there, appeared over two hundred years ago. One can mention probably the first synthetized complex that is linked by a halogen bond, I2⋯NH3.28,29 In later studies, this type of interaction was discussed occasionally. For example, a statistical analysis of the geometry of halogen bonds based on structures30 taken from the Cambridge Structural Database, CSD,31,32 was performed. The Lewis acid properties of halogen centres, even though they are electronegative elements, was discussed. These properties were explained by the anisotropy of the van der Waals radii of the halogen centres,33 or the ellipsoidal shape of the electron density around the halogen centres,34 and finally the acidic properties of the halogen centres were explained by the σ-hole concept introduced by Politzer and co-workers.4,15–17,35,36
It is worth noting that at the beginning of this century, the number of studies on halogen bonds, XBs, including review articles and monographs,37–40 increased rapidly. Notably, the important contributions by Resnati and co-workers can be mentioned. It was pointed out that XB is an effective tool in crystal engineering,41 where the similarities between the halogen bond and hydrogen bond interactions were discussed based on crystal structures,42 and the difference between the halogen bond and an interaction where the halogen centre acts as a nucleophile was reported.43 The latter interaction can be named a halide bond.22 The hydrocarbons as effective binding sites with anions through halogen bonds were analysed and numerous examples of this situation in crystal structures were presented.38 The crystal structures with halogen bonds where the fluorine centre acts as an electrophile were analysed and supported by theoretical calculations.44 It is worth mentioning the studies on the role of interactions among halogen bonds in catalysis.45,46
A variety of halogen bond interactions exists, as similarly observed for hydrogen bonds. However, in the case of halogen bonds, their diversity is much greater. Firstly, different elements of the 17th group may participate in halogen bonds, while only one element, hydrogen, participates in hydrogen bonds. In both cases, XBs and HBs, the halogen or hydrogen centre participating in an interaction is positively charged, respectively, or to be more precise, the centre interacts with nucleophiles through the area characterised by the positive electrostatic potential, EP.15–17 If not, it interacts with electrophiles through the negative EP area, and thus a hydride bond or halide bond exists, respectively.14 However, the situation is not so clear, where the electrostatic potential is usually related to the electrostatic part of the energy of interaction15–17 but other terms also constitute this energy; other attractive terms (conventionally assumed to be negative) such as dispersion and polarization ones may be important to stabilize the complex linked by any type of interaction.14
The dihalogen bond, DXB,22 can be considered a type of halogen bond, just like the dihydrogen bond, DHB, which is a special case of the hydrogen bond.47–49 Again, in the former case, the diversity is much greater than in the latter case. In the first case, interactions between different elements of the same group, such as Cl⋯Br and I⋯Br, can be observed; interactions such as Cl⋯Cl and I⋯I between the same elements are also taken into account. Different names for various dihalogen bonds were used in previous studies, including the halogen–halogen bond50–52 or halogen–halide bond.53 Regarding the DHB interaction, contact between two H-atoms characterized by opposite charges is observed, H+δ⋯−δH. In the case of halogens, they may possess dual characteristics, which may act as Lewis acid centres, and at the same time Lewis base centres. Hence, the diversity of dihalogen bonds is also connected with different orientations of groups in contact, R1–X1⋯X2–R2.37 The interactions of X Lewis acid centres with X− anions are also possible.22,54,55 Finally, one can mention issues such as multivalent Lewis acid X-centres56 and bifurcated halogen bonds.57–59 The former one does not occur for hydrogen bonds, while the second one does, and thus bifurcated hydrogen bonds have been the subject of numerous studies.60
Taking into account all the above-mentioned issues related to the diversity of halogen bonds, the question arises whether the use of the term halogen bond is justified for such a wide spectrum of interactions. Thus, the aim of this study is to discuss this topic. For this purpose, Cambridge Structural Database31,32 searches were performed to find crystal structures that present different types of halogen bonds, i.e., fluorine, chlorine, bromine and iodine bonds. Then, statistical approaches were used to assess whether there were significant differences between these types of halogen bonds. Additionally, in this study, an analysis of theoretical results is performed regarding bromine and iodine bonds using the above-mentioned statistical techniques.
Methodology
Cambridge Structural Database searches
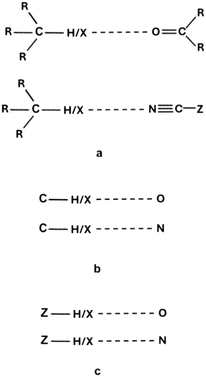
CSD31,32 searches were performed here to find crystal structures that contain systems linked by C–X⋯O, and C–X⋯N interactions. Here, X is a halogen centre, i.e., F, Cl, Br and I. R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 systems were considered in the case of the former type of interactions; Rs are centres directly connected to carbon atoms, which are H (or D), O, N, or C centres, and may be further connected with other atoms or groups. In the case of C–X⋯N interactions, R3CX⋯N
CR2 systems were considered in the case of the former type of interactions; Rs are centres directly connected to carbon atoms, which are H (or D), O, N, or C centres, and may be further connected with other atoms or groups. In the case of C–X⋯N interactions, R3CX⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems are considered; Rs are the same centres as the former sample, while Z is chosen to be any atom connected with the N
CZ systems are considered; Rs are the same centres as the former sample, while Z is chosen to be any atom connected with the N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C group. In both systems, R3CX⋯O
C group. In both systems, R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CX⋯N
CR2 and R3CX⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ, the R-substituents in R3CX or O
CZ, the R-substituents in R3CX or O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 do not have to be the same (Scheme 1a).
CR2 do not have to be the same (Scheme 1a).
The following criteria to choose the above-mentioned systems were applied: 3D coordinates determined, no disordered structures, no errors, no polymeric structures, no powder structures, R-factor less or equal to 5%, e.s.d for CC bonds less than or equal to 0.005 Å and only single crystal structures. In the case of F⋯O, Cl⋯O, Br⋯O, and I⋯O, 11, 92, 76, and 32 crystal structure links result from the searches performed, respectively. In contrast, for F⋯N, Cl⋯N, Br⋯N, and I⋯N, 4, 14, 7, and 1 crystal structure links result from the searches, respectively. It seems that in the case of X⋯N interactions, a greater number of structures is needed to perform the statistical analyses. Hence, for these interactions, the CSD searches were repeated with the same criteria as before and only one parameter changed, R-factor less than or equal to 7.5%. In these searches, for F⋯N, Cl⋯N, Br⋯N, and I⋯N, 6, 15, 8, and 1 crystal structure links were found, respectively. One can see only one crystal structure containing the R3CI⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ system, thus the I⋯N interactions are not considered in further statistical analysis. However, it is also worth mentioning that for each crystal structure, more than one system of the specific interaction may occur. This is why the number of systems in the samples applied for statistical analyses may be greater than the corresponding number of crystal structures, which is presented later in this study.
CZ system, thus the I⋯N interactions are not considered in further statistical analysis. However, it is also worth mentioning that for each crystal structure, more than one system of the specific interaction may occur. This is why the number of systems in the samples applied for statistical analyses may be greater than the corresponding number of crystal structures, which is presented later in this study.
The results of the CSD searches conducted here allow the comparation of halogen bond sub-classes, i.e. fluorine, chlorine, bromine and iodine bonds. Another aim of this study is to compare these sub-classes with the corresponding types of hydrogen bonds. Hence, additional CSD searches were performed to find R3CH⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CH⋯N
CR2 and R3CH⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems. To obtain results of similar accuracy as the former searches, the same search criteria were applied here and only neutron diffraction structures were considered to have the positions of H-atoms determined with similar accuracy as the other heavier atoms.61 The number of crystal structures found in which the R3CH⋯O
CZ systems. To obtain results of similar accuracy as the former searches, the same search criteria were applied here and only neutron diffraction structures were considered to have the positions of H-atoms determined with similar accuracy as the other heavier atoms.61 The number of crystal structures found in which the R3CH⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CH⋯N
CR2 and R3CH⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems occur was 79 and 12, respectively. An R-factor less than or equal to 5% and 7.5%, respectively, was chosen for the above-mentioned searches. If an R-factor less than or equal to 5% is applied for the R3CH⋯N
CZ systems occur was 79 and 12, respectively. An R-factor less than or equal to 5% and 7.5%, respectively, was chosen for the above-mentioned searches. If an R-factor less than or equal to 5% is applied for the R3CH⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ search, only 4 crystal structures meeting the adopted criteria are found.
CZ search, only 4 crystal structures meeting the adopted criteria are found.
Other CSD searches were performed here; C–H/X⋯O and C–H/X⋯N systems without constraints on connections of the C, O and N centres (Scheme 1b), and a more general pair of systems, i.e., Z–H/X⋯O and Z–H/X⋯N, where Z is any element (Scheme 1c). R ≤ 5% is fixed for these new searches, while the other search conditions are the same as that for the R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CX⋯N
CR2 and R3CX⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ pair of systems. In all Z/C–H⋯O/N searches, only neutron diffraction results were considered. Besides, for all searches performed in this study (Scheme 1a–c), the following restrictions were introduced on intermolecular H/X⋯O/N distances and Z/C–H/X⋯O/N angles: distances not greater than the corresponding sum of van der Waals radii (the van der Waals radii inserted in CSD62 were applied in this study) and angles in the range of 90° and 180°. This range is often fixed for hydrogen bonds, which do not occur rather for narrower angles.60 On the other hand, for halogen bonds, this range is acceptable since the Lewis acid properties of X-centres are revealed in the elongation of Z/C–X bonds or close to this direction; thus, for angles lower than 90°, their Lewis base properties are usually revealed.15,16,34
CZ pair of systems. In all Z/C–H⋯O/N searches, only neutron diffraction results were considered. Besides, for all searches performed in this study (Scheme 1a–c), the following restrictions were introduced on intermolecular H/X⋯O/N distances and Z/C–H/X⋯O/N angles: distances not greater than the corresponding sum of van der Waals radii (the van der Waals radii inserted in CSD62 were applied in this study) and angles in the range of 90° and 180°. This range is often fixed for hydrogen bonds, which do not occur rather for narrower angles.60 On the other hand, for halogen bonds, this range is acceptable since the Lewis acid properties of X-centres are revealed in the elongation of Z/C–X bonds or close to this direction; thus, for angles lower than 90°, their Lewis base properties are usually revealed.15,16,34
If one considers only searches with R ≤ 5%, the Z–H/X⋯O and Z–H/X⋯N samples contain C–H/X⋯O and C–H/X⋯N samples, respectively, and the latter ones contain R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CX⋯N
CR2 and R3CX⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ samples.
CZ samples.
Statistical methods
Population and sample are key terms in numerous statistical approaches.63 The term “sample” was mentioned several times earlier here. The term “population” is understood as a collection of objects (observations) under study, for example, parameters of molecular systems, such as intermolecular distances, may be considered. A population may consist of finite or infinite objects, at least the number of objects may be large. Hence, it is usual to analyse a part of the population, i.e., sample, to gain information about the population.For example, the population of C–Br⋯O halogen bonds in R3C–Br⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 complexes may be considered (Scheme 1a), particularly their geometrical parameters such as distances and angles may be analysed. These are all halogen bonds, and thus their number may be large and not useful to analyse. Besides, the part of complexes is not characterised by precise parameters determined by experimental methods, thus only the part of C–Br⋯O halogen bonds of the above-mentioned complexes, i.e., the sample, may be employed for analysis. Numerous samples of distances and samples of angles of halogen and hydrogen bonds in complexes presented in Scheme 1(a–c) are discussed in the next sections.
CR2 complexes may be considered (Scheme 1a), particularly their geometrical parameters such as distances and angles may be analysed. These are all halogen bonds, and thus their number may be large and not useful to analyse. Besides, the part of complexes is not characterised by precise parameters determined by experimental methods, thus only the part of C–Br⋯O halogen bonds of the above-mentioned complexes, i.e., the sample, may be employed for analysis. Numerous samples of distances and samples of angles of halogen and hydrogen bonds in complexes presented in Scheme 1(a–c) are discussed in the next sections.
Descriptive statistics is performed in this study. It concerns well-known quantities such as mean and median values, standard deviation, mean deviation, first and third quartiles, as well as minimum and maximum values;63,64 hence, these terms are not described here. However, statistical tests were also applied to analyse various samples. These tests are Wilk–Shapiro and Kolmogorov–Smirnov tests to check if the populations are characterised by normal distributions and Welch T-test and Mann–Whitney U-test, which are applied to compare distributions of samples, and consequently distributions of populations between them.64,65
It is important to check if the sample considered meets the criteria of normal distribution, given that only in this case these parameters as the mean value, standard deviation and others may be analysed.66 The Wilk–Shapiro test (WS) is more powerful than the Kolmogorov–Smirnov (KS) test, and even estimated as the most powerful test among those often applied to check the normality of distributions.67 The sample size is understood as the number of observations in the sample considered. There are also other methods to check if a random sample of independent observations comes from a population with a normal distribution, which are graphical methods such as histograms and Q–Q-plots (normal quantile–quantile plots).68
Proving that samples apply to normal distributions is also essential because for these samples, one can use further parametric tests that are more powerful than analogues nonparametric tests applied for samples not characterized by normal distributions.64,66 Hence, to compare samples, particularly if they follow the same distributions, two tests were applied here, the Welch T-test (W) and the Mann–Whitney U-test (MW) for samples of normal and non-normal distributions, respectively.64
In the statistical tests, the null and alternative hypotheses are stated, marked usually as H0 and H1, respectively.66 In the WS and KS tests, H0 states that observations of the sample considered follow a specified distribution, particularly a normal distribution, which is the case in this study, while H1 states that the data do not follow this distribution. In the W and MW tests, H0 states that observations of samples considered do not differ, while H1 states that these observations differ from each other.64–66 For example, two samples of C–Br⋯O and C–Cl⋯O angles may be considered, and the null hypothesis states that these samples are not different from each other. Rejecting this hypothesis and accepting the alternative hypothesis means that these two samples are different from each other. The latter may mean that Br⋯O and Cl⋯O halogen bonds are different interactions.
Another quantity attributed to testing of samples is the significance level, which is often marked as α. The latter quantity is the probability related to rejecting the null hypothesis in the testing approach. A value of α equal to 5% is often chosen. If the null hypothesis is rejected at this level of significance, it means that the probability to reject the true null hypothesis is at most 5% or less. In the testing procedure, it is better to choose a lower α value since the rule is to reject the null hypothesis and accept the alternative hypothesis.66 However, if there are no grounds to reject H0, it does not mean that H0 is statistically accepted. This only means that statistically, there are no grounds to reject it.66 Hence, the goal of the WS and KS tests is the acceptance of the alternative hypothesis that the sample is not of the normal distribution and to accept the alternative hypothesis for the W and MW tests that the samples are different from each other.
Results and discussion
Descriptive statistics of C–H⋯O/N and C–X⋯O/N interactions in crystal structures
The statistics concerning X⋯O, X⋯N, H⋯O, and H⋯N intermolecular distances for the R3CH/X⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CH/X⋯N
CR2 and R3CH/X⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems (Scheme 1a) are given in Table 1, while the statistics of the corresponding C–X⋯O, C–X⋯N, C–H⋯O, and C–H⋯N angles are presented in Table 2 (Tables S1–S3 in SI, contain additional information on these systems, respectively).
CZ systems (Scheme 1a) are given in Table 1, while the statistics of the corresponding C–X⋯O, C–X⋯N, C–H⋯O, and C–H⋯N angles are presented in Table 2 (Tables S1–S3 in SI, contain additional information on these systems, respectively).
| Contact | N | n | R MEAN | σ | R MEDIAN | ∑vdW − RMEAN |
|---|---|---|---|---|---|---|
| H⋯O | 79 | 161 | 2.516 | 0.120 | 2.525 | 0.184 |
| F⋯O | 11 | 12 | 2.883 | 0.059 | 2.896 | 0.077 |
| Cl⋯O | 92 | 100 | 3.192 | 0.107 | 3.230 | 0.128 |
| Br⋯O | 76 | 80 | 3.185 | 0.122 | 3.199 | 0.175 |
| I⋯O | 32 | 32 | 3.266 | 0.160 | 3.229 | 0.274 |
| H⋯N | 12 | 37 | 2.658 | 0.132 | 2.672 | 0.202 |
| F⋯N | 6 | 8 | 3.025 | 0.051 | 3.017 | 0.095 |
| Cl⋯N | 15 | 16 | 3.355 | 0.111 | 3.393 | 0.125 |
| Br⋯N | 8 | 8 | 3.350 | 0.159 | 3.386 | 0.170 |
| I⋯N | 1 | 1 | 3.276 | — | 3.276 | — |
| Angle | Θ MEAN | σ | Θ MEDIAN |
|---|---|---|---|
| a Only one C–I⋯N interaction results from the CSD search; thus, the standard deviation is not presented. | |||
| C–H⋯O | 143.7 | 15.9 | 142.0 |
| C–F⋯O | 124.1 | 17.5 | 125.3 |
| C–Cl⋯O | 156.1 | 17.3 | 159.7 |
| C–Br⋯O | 162.8 | 11.9 | 165.8 |
| C–I⋯O | 163.9 | 15.0 | 166.8 |
| C–H⋯N | 140.3 | 16.2 | 140.6 |
| C–F⋯N | 133.0 | 15.5 | 125.1 |
| C–Cl⋯N | 151.8 | 22.6 | 160.3 |
| C–Br⋯N | 150.0 | 32.4 | 165.0 |
| C–I⋯N | 176.7 | 176.7 | |
The parameters in Tables 1 and 2 concern sub-classes of halogen bond interactions. They differ from each other only by the halogen element that acts as the Lewis acid centre, and at least one can expect this is the acidic centre for heavier halogen centres. The statistics of the distances and angles of the C–H⋯O and C–H⋯N hydrogen bonds are included in tables for comparison.
Some parameters such as the mean value and the standard deviation may be applied for samples that meet the condition of a normal distribution. Not all samples of the results collected in Tables 1 and 2 meet this condition, but the mean and standard deviation parameters are presented here for approximate comparison of the interactions that differ by halogen element. Also, the median values are included, which may be compared with the mean values.
Graphical methods: histograms and Q–Q-plots were applied here to check if the samples in Tables 1 and 2 are characterized by normal distributions. These graphical results are presented in Fig. S1–S9 (in SI). None of these histograms present a clear normal distribution for a sample. In the case of the Q–Q plots, the straight lines indicate normal distributions.68 Only in the case of samples with C–H⋯O and C–F⋯O angles (Fig. S1 and S2), respectively, the Q–Q distributions of the points are very close to straight lines.
Given that these graphical results are not clear, Wilk–Shapiro (WS) and Kolmogorov–Smirnov (KS) tests were also performed to check the normality of the distributions, which concern the C–H/X⋯O/N angles and values related to H/X⋯O/N distances, ΔRs, respectively. ΔRs (collected in Table S3) are the differences between the sum of van der Waals radii of the centres in contact and the distance between these centres. These quantities were introduced given that in intermolecular contacts concerning different electron acceptors (halogens or hydrogen), ΔRs may be treated as normalized values.
Results of these tests are presented in Table 3. The sign plus (+) designates that the null hypothesis, H0, is not rejected, while the sign minus (−) designates the rejection of H0 and the acceptance of the alternative hypothesis, H1. In other words, (+) designates that the normal distribution is not rejected, while (−) designates acceptance of the non-normal distribution. The normal distribution of samples is assumed more often according to the KS test than according to the WS test given that the former test is less powerful than the latter one.67 However, in a few cases both tests reject this distribution (H0 is rejected). This is observed for ΔR of the Cl⋯O distance, as well as for the C–Cl⋯O, C–Br⋯O, C–I⋯O, and C–Br⋯N angles. Thus, the conclusions for the mean and standard deviation values of the above-mentioned parameters are burdened with error.
| System considered ↓ | ΔR for H/X⋯O | C–H/X⋯O angle | ΔR for H/X⋯N | C–H/X⋯N angle |
|---|---|---|---|---|
| H⋯O/N | −(+) | +(+) | +(+) | +(+) |
| F⋯O/N | +(+) | +(+) | +(+) | +(+) |
| Cl⋯O/N | −(−) | −(−) | −(+) | −(+) |
| Br⋯O/N | −(+) | −(−) | +(+) | −(−) |
| I⋯O/N | +(+) | −(−) | — | — |
Here, we discuss the results presented in Tables 1 and 2 concerning the C–H/X⋯O/N links (Scheme 1a). In the case of H⋯O and X⋯O contacts, their electron-donating centre is the same, i.e., oxygen, and thus one can analyse the influence of the type of electrophilic centre, halogen or hydrogen, on the properties of the interactions. The shortest RMEAN distance is observed for the C–H⋯O systems, and next a longer mean F⋯O distance is observed, and for the remaining X⋯O contacts, the longest distances occur, i.e. the greatest RMEAN values are observed. It is important that the difference between the RMEAN values of the H⋯O and F⋯O contacts is greater than 3σ (for σ of both types of contacts), and similarly the difference between the RMEAN values of the F⋯O and Cl⋯O contacts is greater than 3σ if the σ of the F⋯O contacts is considered, which is greater than 2σ, but close to 3σ for the σ of Cl⋯O contacts. The differences between the RMEAN values for the Cl⋯O, Br⋯O and I⋯O contacts seem to be negligible. Hence, if one classifies the interactions based on the electron donor–acceptor distances, there are three groups of interactions, C–H⋯O hydrogen bonds, C–F⋯O interactions and C–X⋯O halogen bonds (X = Cl, Br and I).
There are similar observations for the RMEDIAN values. RMEDIAN is equal to 2.53 Å, 2.90 Å, and 3.20–3.23 Å for the H⋯O, F⋯O, and Cl/Br/I⋯O distances, respectively. Hence, the occurrence of three types of interactions is confirmed here. This is in line with former studies where it was pointed out that fluorine is hardly accepted as an electron-accepting centre, while for the remining halogen centres these acceptor properties are observed more often.39 Thus, fluorine bonds should differ significantly from other halogen bonds. This agrees with the σ-hole concept that for heavier halogen centres, stronger σ-holes characterised by greater electrostatic potentials, EPs, occur.4,35,36 This refers to stronger halogen bonds for heavier halogen centres. In the case of fluorine centres, σ-holes, even if they occur, are not as strong and they are often characterised by a negative EP. However, the term fluorine bond is applied from time to time to show the Lewis acid properties of fluorine centres in interactions with nucleophiles.69–71 The fluorine bond was discussed in other studies,72 and even fluorine–fluorine interactions were analysed.73 One can also mention an important study in which the conditions for the existence of fluorine bonds were analysed theoretically.74
It is known that the donor–acceptor distance for intermolecular interactions may be treated as a rough measure of their strength, where stronger interactions occur for shorter distances.6,75 The latter relationships are observed for the same pairs of centres being in contact and for homogeneous samples of interacting species. Hence, to compare the strength of interactions, RMEAN and RMEDIAN are not the best parameters given that different electrophilic centres occur here. The difference between the sum of van der Waals radii of centres being in contact and the distance between them, ΔR, can be treated as an approximate measure of the strength of the interaction. This parameter was described earlier here. However, the ∑vdW − RMEAN values, ΔRMEANs, related to the sub-classes of interactions discussed here are presented in Table 1. The greater the ΔRMEAN, the stronger the interaction. In the case of halogen centres, the lowest ΔRMEAN occurs for fluorine (0.077 Å), and it increases with an increase in the atomic number of the halogen element (Table 1). This difference for bromine is close to that of the H⋯O contact, while for iodine the difference is greater. This is in line with other results, where it was found in several studies that for lighter halogen elements, the halogen bonds are weaker than the corresponding hydrogen bonds, while for heavier halogen elements, such as bromine or iodine, the interactions are comparable to hydrogen bonds or even stronger.76
It is worth noting that there are three types of attractive forces that steer the intermolecular arrangements, i.e., electrostatic, polarization (related to electron charge shifts between interacting species, this term is often named inductive or charge transfer in different interaction energy decomposition schemes) and dispersion.6,14,77 In the case of weak or negligible electrostatic and polarization contributions, the dispersion term is the most important one and the atom–atom intermolecular contacts are close to the corresponding sums of van der Waals radii or even longer. The electron charge shifts are typical for the so-called Lewis acid–Lewis base interactions, especially for halogen bonds, and they are mostly responsible for the shortening of the intermolecular atom–atom contacts.6 Therefore, the greater the above-mentioned ΔR (or ΔRMEAN) parameter, the greater the electron charge shifts between interacting species.
Here, we analyse the H/X⋯N distances (Table 1). Meaningful differences between the RMEAN values of the H⋯N and F⋯N distances as well as between the RMEAN values of the F⋯N and Cl⋯N distances are observed, which are greater than or close to 3σ. A negligible difference is observed for the Cl⋯N and Br⋯N distances. Only one I⋯N interaction results from the CSD search, and thus this interaction is not considered in these comparisons; the results for this single interaction are presented in Table 1 only for comparison. The RMEAN values are very similar to the RMEDIAN values for the H/X⋯N distances.
The ΔRMEAN values for the H/X⋯N contacts change in a similar way as in the case of H/X⋯O contacts, where the lowest values are observed for the F⋯N distances, and an increase in ΔRMEAN is observed with an increase in the atomic number of the halogen element. However, the ΔRMEAN value for the H⋯N contacts is the greatest. Thus, one can see that three types of interactions can be specified, C–H⋯N hydrogen bonds, C–F⋯N interactions, and C–Cl/Br⋯N halogen bonds, similar to interactions with oxygen as the electron donor.
Here, we analyse the changes in the C–H/X⋯O/N angles (Table 2). The differences between samples are not as clear here as in the case of distances (Table 1), where they are greater than or close to 3σ. This is because for angles the σ-values are large and range from 15° to 32.4°. However, it can be noticed again that the differences between C–H⋯O and C–F⋯O angles as well as between C–F⋯O and C–Cl⋯O angles are much greater (greater than σ value) than between the remaining C–X⋯O angles where X = Cl, Br and I. We observe a similar situation regarding angles where the nitrogen centre acts as the electron donor. Hence the division of interactions into three sub-classes, C–H⋯O/N hydrogen bonds, C–F⋯O/N interactions and C–Cl/Br/I⋯O/N halogen bonds, is confirmed here.
There are other interesting observations concerning the differences between these angles (Table 2). The mean angles, ΘMEAN, are equal to 143.7°, 124.1°, and 156.1–163.9° for the C–H⋯O, C–F⋯O and C–Cl/Br/I⋯O systems, respectively. In the case of the C–H⋯N, C–F⋯N and C–Cl/Br⋯N systems, the mean angles, ΘMEAN, are equal to 140.3°, 133.0°, 150.0–151.8°, respectively. Thus, one can see for both great groups differing by the electron donor, an increase in the angle with a change in the Lewis acid centre, following the order of F < H < Cl/Br/I. This order is confirmed by the ΘMEDIAN values (Table 2). In other words, halogen bonds (with Cl/Br/I centres) are more directional than the corresponding hydrogen bonds, while the latter interact more directionally than fluorine bonds. This is consistent with the previous studies, particularly those related to the σ-hole concept, according to which the directionality of halogen bonds is related to the small area of σ-hole in the C–X bond extension at the Lewis acid centre, whereas the directionality of hydrogen bonds is related to the hydrogen that has a positive EP for the entire hemisphere.15,16 In the case of fluorine, σ-hole is often not observed and the entire F-sphere possesses a negative EP. Probably, the latter case strongly influences the ΘMEAN and ΘMEDIAN values for the fluorine bonds.
The above-mentioned division into sub-classes is based on the type of Lewis acid centre, i.e. halogen element; the hydrogen bond is considered in this study for comparison. However, we do not know whether there are differences between sub-classes characterized by the same Lewis acid centre and by different electron donors; an example is the C–Br⋯O and C–Br⋯N pair. It is probably reasonable to classify the last pair, as well as the other pairs with the same Lewis acid centre and different electron donors, as the same interaction type, given that some similarities can be observed between the corresponding subclasses in terms of angles and ΔRMEAN parameters. This will be discussed later in this paper.
Fig. 1–3 present examples of crystal structures containing the R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CX⋯N
CR2 and R3CX⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems analysed here. Fig. 1 shows the fragment of the crystal structure of D-erythro-4-fluoro glutamic acid, where molecules are linked by the C–F⋯O fluorine bonds as well as by N–H⋯O and C–H⋯O hydrogen bonds.78 In the case of the fluorine bond, the F⋯O distance is equal to 2.731 Å, while the C–F⋯O angle is 159.6°. This angle is close to linearity. Besides, the F⋯O distance is shorter by more than 0.2 Å of the corresponding sum of van der Waals radii, i.e., 2.96 Å. This suggests that the C–F⋯O interactions occurring in this crystal structure are classified as directional σ-hole bonds.15,16 It can be seen (Fig. 1) that the oxygen centre in the C–F⋯O interaction is also involved in the N–H⋯O hydrogen bond. Thus, this is a case of a bifurcated interaction where the single oxygen electron donor is involved in hydrogen and fluorine bonds, which may be treated as partial bonds. Bifurcated interactions were reported in earlier studies but both partial interactions were classified as hydrogen bonds,79,80 and two cases were considered, as follows: (1) a common electron donor and two proton donors and (2) a common proton donor and two electron donors. Fig. 1 also presents the C–H⋯O hydrogen bonds between molecules of the crystal structure; however, these interactions characterized by H⋯O distances equal to 2.49 Å seem to be weaker than the N–H⋯O hydrogen bonds, where these distances are much shorter and equal to 1.94 Å.
CZ systems analysed here. Fig. 1 shows the fragment of the crystal structure of D-erythro-4-fluoro glutamic acid, where molecules are linked by the C–F⋯O fluorine bonds as well as by N–H⋯O and C–H⋯O hydrogen bonds.78 In the case of the fluorine bond, the F⋯O distance is equal to 2.731 Å, while the C–F⋯O angle is 159.6°. This angle is close to linearity. Besides, the F⋯O distance is shorter by more than 0.2 Å of the corresponding sum of van der Waals radii, i.e., 2.96 Å. This suggests that the C–F⋯O interactions occurring in this crystal structure are classified as directional σ-hole bonds.15,16 It can be seen (Fig. 1) that the oxygen centre in the C–F⋯O interaction is also involved in the N–H⋯O hydrogen bond. Thus, this is a case of a bifurcated interaction where the single oxygen electron donor is involved in hydrogen and fluorine bonds, which may be treated as partial bonds. Bifurcated interactions were reported in earlier studies but both partial interactions were classified as hydrogen bonds,79,80 and two cases were considered, as follows: (1) a common electron donor and two proton donors and (2) a common proton donor and two electron donors. Fig. 1 also presents the C–H⋯O hydrogen bonds between molecules of the crystal structure; however, these interactions characterized by H⋯O distances equal to 2.49 Å seem to be weaker than the N–H⋯O hydrogen bonds, where these distances are much shorter and equal to 1.94 Å.
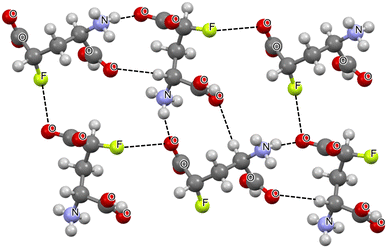 | ||
| Fig. 1 Fragment of the crystal structure of D-erythro-4-fluoro glutamic acid (KINWIN ref code).78 The F⋯O and H⋯O intermolecular contacts are designated by the black narrow lines. | ||
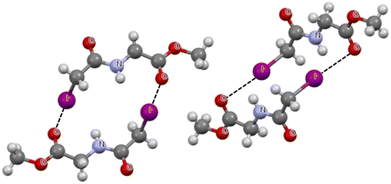 | ||
| Fig. 2 Fragment of the crystal structure of methyl N-(iodoacetyl)glycinate (KIPTIP ref code).81 The I⋯O intermolecular contacts are designated by the black narrow lines. | ||
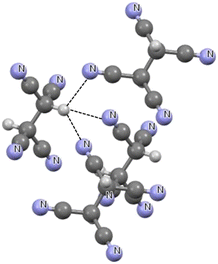 | ||
| Fig. 3 Fragment of the crystal structure of 1,1,2,2-ethanetetracarbonitrile (BUGKIX01 ref code).82 The H⋯N intermolecular contacts are designated by the black narrow lines. | ||
It is worth mentioning that the RMEAN F⋯O distance for the sample containing R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 complexes is equal to 2.88 Å (Table 1), which is close to the sum of the van der Waals radii. This means that the majority of C–F⋯O interactions considered here are rather weak with the dominant or at least important role of dispersion forces. In the sample of R3CF⋯O
CR2 complexes is equal to 2.88 Å (Table 1), which is close to the sum of the van der Waals radii. This means that the majority of C–F⋯O interactions considered here are rather weak with the dominant or at least important role of dispersion forces. In the sample of R3CF⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 systems, the shortest F⋯O distance occurs in the structure of Fig. 1, thus not only dispersion, but also other attractive forces are important here.
CR2 systems, the shortest F⋯O distance occurs in the structure of Fig. 1, thus not only dispersion, but also other attractive forces are important here.
Fig. 2 shows a fragment of the crystal structure of methyl N-(iodoacetyl)glycinate.81 Here, the molecules are connected by C–I⋯O halogen bonds. The centrosymmetric dimers are presented in Fig. 2, where two equivalent C–I⋯O links are observed. The I⋯O distances are equal to 3.398 Å, which is less than the corresponding sum of van der Waals radii by 0.142 Å. This means that this halogen (iodine) bond is stronger than the C–F⋯O interaction discussed above (Fig. 1). The C–I⋯O angle for the iodine bond is equal to 174.4°, and the system is very close to linearity, which indicates that the interaction is classified as a σ-hole bond.
Fig. 3 presents a fragment of the crystal structure of 1,1,2,2-ethanetetracarbonitrile.82 It can be seen that in the trifurcated C–H⋯N hydrogen bonds here, one C–H bond is connected with three nitrogen electron-donating centres. The H⋯N distances are equal to 2.523 Å, 2.783 Å and 2.321 Å with the C–H⋯N angles equal to 115.4°, 119.1° and 140.0°, respectively. However, each case is far from linearity, which is typical for furcated hydrogen bonds.60,79,80,83 In one of the H⋯N contacts of 2.321 Å, a rather strong interaction is detected given that this distance is shorter than the corresponding sum of van der Waals radii by 0.539 Å.
Descriptive statistics of halogen bonds and hydrogen bonds in crystal structures – greater samples
The parameters presented in Tables 1 and 2 (and Tables S1–S3) concern special types of complexes linked by halogen or hydrogen bonds, which are R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CX⋯N
CR2 and R3CX⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems (Scheme 1a). The above discussion shows that the hydrogen-bonded systems as well as the fluorine-bonded ones differ from other interactions, i.e. chlorine, bromine and iodine bonds. Therefore, the three latter interactions may be “covered” by the common name halogen bonds. However, to check whether this “division” covers greater samples, i.e., whether it is more general, other samples are considered here. In the CSD,31,32 the C–H/X⋯O and C–H/X⋯N systems where searched, in which there are no restrictions on the connections of C, O and N centres (Scheme 1b), and another more general pair of systems, i.e., Z–H/X⋯O and Z–H/X⋯N (Z is any centre, see Scheme 1c). The parameters for the new, greater samples are presented in Tables 4 and 5 (Tables S4 and S5 contain additional parameters for these samples, respectively). These tables, similar to Tables 1 and 2, contain parameters such as RMEAN and σ, which are appropriate for samples characterised by a normal distribution. However, tests checking the normality of distributions are not performed here; these greater samples are presented for rough comparison only. More detailed further statistical analyses are carried out only for samples containing the R3CX⋯O
CZ systems (Scheme 1a). The above discussion shows that the hydrogen-bonded systems as well as the fluorine-bonded ones differ from other interactions, i.e. chlorine, bromine and iodine bonds. Therefore, the three latter interactions may be “covered” by the common name halogen bonds. However, to check whether this “division” covers greater samples, i.e., whether it is more general, other samples are considered here. In the CSD,31,32 the C–H/X⋯O and C–H/X⋯N systems where searched, in which there are no restrictions on the connections of C, O and N centres (Scheme 1b), and another more general pair of systems, i.e., Z–H/X⋯O and Z–H/X⋯N (Z is any centre, see Scheme 1c). The parameters for the new, greater samples are presented in Tables 4 and 5 (Tables S4 and S5 contain additional parameters for these samples, respectively). These tables, similar to Tables 1 and 2, contain parameters such as RMEAN and σ, which are appropriate for samples characterised by a normal distribution. However, tests checking the normality of distributions are not performed here; these greater samples are presented for rough comparison only. More detailed further statistical analyses are carried out only for samples containing the R3CX⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3CX⋯N
CR2 and R3CX⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems (see next section of this study).
CZ systems (see next section of this study).
| Contact | N | n | R MEAN | σ | R MEDIAN | ΔRMEAN |
|---|---|---|---|---|---|---|
| H⋯O | 208 | 785 | 2.509 | 0.140 | 2.533 | 0.191 |
| 256 | 1643 | 2.222 | 0.377 | 2.351 | 0.478 | |
| F⋯O | 1525 | 1932 | 2.889 | 0.062 | 2.905 | 0.071 |
| 2529 | 3941 | 2.833 | 0.112 | 2.868 | 0.127 | |
| Cl⋯O | 3980 | 4967 | 3.169 | 0.111 | 3.189 | 0.151 |
| 6408 | 8646 | 3.175 | 0.101 | 3.189 | 0.145 | |
| Br⋯O | 2126 | 2384 | 3.168 | 0.126 | 3.182 | 0.192 |
| 2321 | 2665 | 3.168 | 0.148 | 3.192 | 0.192 | |
| I⋯O | 1064 | 1640 | 3.125 | 0.239 | 3.128 | 0.415 |
| 1132 | 1948 | 3.120 | 0.251 | 3.123 | 0.420 | |
| H⋯N | 50 | 112 | 2.680 | 0.140 | 2.710 | 0.180 |
| 90 | 244 | 2.489 | 0.339 | 2.617 | 0.371 | |
| F⋯N | 1827 | 2394 | 3.028 | 0.071 | 3.044 | 0.092 |
| 4140 | 8493 | 2.975 | 0.113 | 2.999 | 0.145 | |
| Cl⋯N | 2515 | 3242 | 3.341 | 0.120 | 3.374 | 0.139 |
| 7132 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 619 619 |
3.319 | 0.106 | 3.331 | 0.161 | |
| Br⋯N | 850 | 1002 | 3.273 | 0.197 | 3.322 | 0.247 |
| 1324 | 1900 | 3.321 | 0.198 | 3.383 | 0.199 | |
| I⋯N | 869 | 1199 | 3.069 | 0.299 | 2.968 | 0.631 |
| 1038 | 1479 | 3.147 | 0.345 | 3.037 | 0.553 |
| Angle | Θ MEAN | σ | Θ MEDIAN |
|---|---|---|---|
| C–H⋯O | 142.7 | 17.7 | 143.1 |
| Z–H⋯O | 150.0 | 20.9 | 154.1 |
| C–F⋯O | 134.9 | 22.4 | 137.0 |
| Z–F⋯O | 132.4 | 20.4 | 130.4 |
| C–Cl⋯O | 152.5 | 19.6 | 157.8 |
| Z–Cl⋯O | 138.2 | 26.3 | 143.4 |
| C–Br⋯O | 159.6 | 14.8 | 162.6 |
| Z–Br⋯O | 156.0 | 19.6 | 161.4 |
| C–I⋯O | 158.5 | 19.2 | 165.4 |
| Z–I⋯O | 153.4 | 23.7 | 163.4 |
| C–H⋯N | 141.0 | 16.9 | 142.3 |
| Z–H⋯N | 145.3 | 20.2 | 146.1 |
| C–F⋯N | 133.1 | 19.9 | 133.7 |
| Z–F⋯N | 126.0 | 19.0 | 123.0 |
| C–Cl⋯N | 137.8 | 25.4 | 140.8 |
| Z–Cl⋯N | 119.6 | 23.8 | 112.5 |
| C–Br⋯N | 154.2 | 22.5 | 162.4 |
| Z–Br⋯N | 135.9 | 29.8 | 139.9 |
| C–I⋯N | 165.8 | 18.7 | 172.9 |
| Z–I⋯N | 157.6 | 26.8 | 170.9 |
Here, we analyse the C–H/X⋯O and C–H/X⋯N systems (Scheme 1b and upper rows of Tables 4 and 5). The dependencies and differences observed for smaller samples of interactions (Scheme 1a) are also observed here. Regarding the intermolecular distances for systems with the oxygen as the electron donor, there are greater differences between the C–H⋯O and C–F⋯O samples as well as between the C–F⋯O and C–Cl⋯O ones, than between other C–X⋯O (X = Cl, Br, and I) samples. These differences between the RMEAN values for the former pairs of samples are greater than 2σ. Similar differences are observed for the RMEDIAN values. Conversely, the differences between the RMEAN and RMEDIAN values of C–Cl⋯O, C–Br⋯O, and C–I⋯O are negligible. Similar RMEAN values are observed for complexes containing bromine and chlorine, and the same regarding their RMEDIAN values. However, these values are slightly smaller for iodine species than for the Br and Cl complexes, but not by much, i.e., 0.04 Å and 0.05–0.06 Å for RMEAN and RMEDIAN, respectively.
The observations regarding the H/X⋯N distances in the C–H⋯N and C–X⋯N systems are very similar to that concerning the H/X⋯O distances in the C–H⋯O and C–X⋯O systems. The differences between the H⋯N and F⋯N distances as well as between the F⋯N and Cl⋯N distances are greater than 2σ for the RMEAN values; the latter observation is confirmed by the corresponding differences between the RMEDIAN values. Similar RMEAN and RMEDIAN values are observed for chlorine, bromine and iodine species. The I⋯N distances are smaller than the distances for chlorine and bromine, similar to the corresponding distances of the oxygen–electron donating centre.
Here, we consider the ΔRMEAN parameter, which usually increases with an increase in the strength of the interaction. In the case of complexes with oxygen as the electron-donating centre, the values observed for the H, F, Cl, Br and I centre acting as the electron acceptor are 0.191 Å, 0.071 Å, 0.151 Å, 0.192 Å and 0.415 Å, respectively. In the case of the N-centre acting as a donor, the values observed are 0.180 Å, 0.092 Å, 0.139 Å, 0.247 Å and 0.631 Å, respectively. The order of the electrophilic properties is the same for both electron donors, i.e. F < Cl < H < Br < I. However, in the case of the oxygen nucleophilic centre, the ΔRMEAN parameter for bromine and hydrogen is almost the same, while in the case of nitrogen, this parameter for bromine is much greater than for hydrogen.
The changes in the C–H/X⋯O and C–H/X⋯N angles with a change in the electron acceptor centre for two greater samples of complexes (Scheme 1b) are very similar to that related to smaller samples (Scheme 1a). The ΘMEAN and ΘMEDIAN values for both groups of samples (N-centre and O-centre electron-donor groups) are observed to follow the order C–F⋯O/N < C–H⋯O/N < C–Cl⋯O/N < C–Br⋯O/N < C–I⋯O/N. This order is consistent with the σ-hole concept,15,16 as explained earlier. There is only one exception in the case of ΘMEAN values, i.e., the C–Br⋯O angle is slightly greater than the C–I⋯O angle.
The question arises if there are significant differences between related samples that differ only by the electron-donor centre, for example, between the C–Cl⋯O and C–Cl⋯N samples. Concerning the H/X⋯O/N intermolecular distances, the H/X⋯N distances are greater than the corresponding H/X⋯O distances by approximately 0.1 Å. The only difference is for the I⋯O and I⋯N pair given that the RMEAN and RMEDIAN values are greater for the former contacts than for the latter ones.
The results for the last groups of complexes that are linked by the Z–H/X⋯N and Z–H/X⋯O interactions (Scheme 1c) are similar to that for the groups analysed earlier here (Scheme 1a and b), particularly there are similarities with groups where the C–H/X⋯N and C–H/X⋯O systems without additional constraints occur (Scheme 1b). However, there are two main differences that should be briefly discussed here. The RMEAN and RMEDIAN values for the H⋯O and H⋯N contacts are much shorter here than for the smaller samples. This is because the smaller samples include the C–H⋯N and C–H⋯O hydrogen bonds, which are usually classified as weak interactions characterized by long H⋯N and H⋯O distances, respectively.60,79,80,83 In the larger samples containing Z–H⋯N and Z–H⋯O systems, the O–H⋯N and O–H⋯O hydrogen bonds and other strong ones that are characterized by short contacts are considered.7
Another difference in these larger samples concerns the Z–Cl⋯O/N angles, which are smaller than any other interaction type, and even the ΘMEAN and ΘMEDIAN values for the Z–F⋯O/N angles are greater. This may be related to the Z-centre, which may strongly influence the σ-hole located at the chlorine centre. However, this issue should be analysed in detail. Smaller Z–Cl⋯O/N angles may occur for chlorine and nitrogen centres in contact, for which their Lewis acid and Lewis base properties are not well determined.
Fig. 4 presents a fragment of the crystal structure of 1-chloro-2-methyl-1H-benzimidazole84 as an example of the occurrence of a halogen bond where the chlorine centre is covalently bonded with a non-carbon centre, i.e. nitrogen. The chains of 1-chloro-2-methyl-1H-benzimidazole molecules are linked in this structure by chlorine bonds. Fig. 4 presents the part of one of these chains with N–Cl⋯N interactions; the Cl⋯N distances are equal to 2.648 Å, while the N–Cl⋯N angles are 176.3°.
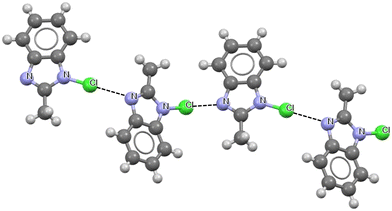 | ||
| Fig. 4 Fragment of the crystal structure of 1-chloro-2-methyl-1H-benzimidazole (GEKPIZ ref code).84 The Cl⋯N intermolecular contacts are designated by the black narrow lines. | ||
Fig. 5 presents a fragment of the crystal structure of potassium hydrogen dicrotonate.85 This figure shows that the crotonic acid molecule is linked with the crotonate anion by a charge-assisted O–H⋯O hydrogen bond.86 The hydrogen bond is very strong as the H⋯O distance is equal to 1.348 Å and the O–H⋯O angle is 178.0°, and thus this arrangement is close to linear. These strong O–H⋯O hydrogen bonds in crystal structures influence the mean H⋯O distance of the sample analysed in this study (Scheme 1c). The H⋯O and H⋯N mean distances are shorter than the corresponding distances in the C–H⋯O and C–H⋯N systems (Scheme 1a and b). Fig. 5 presents the potassium cation and two above-mentioned links that may be named hydrogen dicrotonate species.
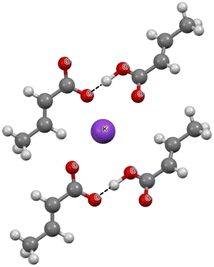 | ||
| Fig. 5 Fragment of the crystal structure of potassium hydrogen dicrotonate (KHCROT01 ref code).85 The H⋯O intermolecular contacts are designated by black narrow lines. | ||
Descriptive statistics leads to numerous valuable conclusions. The parameters related to intermolecular contacts such as mean and median distances indicate that fluorine bonds differ significantly from that of other halogens, such as chlorine, bromine and iodine. However, the parameter related to the shortening of the intermolecular contact below the corresponding sum of van der Waals radii, ΔRMEAN, clearly indicates that the strength of the halogen bond increases with an increase in the atomic number of the halogen centre. Therefore, it can be concluded that there is a greater differentiation of halogen bonds into sub-classes, which is related to the type of element.
Conclusions based on the C–X⋯O/N angles are not unambiguous because the standard deviations for their mean values are large, making it difficult to detect clear differences between angles referring to different X-centers. Besides, there are differences between interactions characterised by the same Lewis acid centre and differing by the electron donor. The X⋯O distances are shorter than the X⋯N distances, and the reverse situation is observed for the I⋯O and I⋯N pair of interactions sometimes (for samples presented in Scheme 1b).
The samples of systems linked by hydrogen bonds were considered here for comparison. The hydrogen bonds correspond approximately to the bromine bonds, the iodine bonds are stronger, while the remaining halogen bonds are weaker than their corresponding hydrogen bonds.
Sub-classes of halogen bonds-statistical tests for systems in crystal structures
In this section, statistical tests were used to compare the analysed samples. Firstly, the samples of the R3C–H/X⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CR2 and R3C–H/X⋯N
CR2 and R3C–H/X⋯N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CZ systems (Scheme 1a) are analysed given that they concern similar structures in which the C–H/X⋯O/N contacts are influenced by similar environments. The C–I⋯N system is not considered here given that only one contact of this type was obtained from the CSD search. Also, larger samples with greater internal heterogeneity were not further analyzed in this study (Scheme 1b and c). The results of the application of the Mann–Whitney U-test (MW test) for the smallest samples are presented in Table 6.
CZ systems (Scheme 1a) are analysed given that they concern similar structures in which the C–H/X⋯O/N contacts are influenced by similar environments. The C–I⋯N system is not considered here given that only one contact of this type was obtained from the CSD search. Also, larger samples with greater internal heterogeneity were not further analyzed in this study (Scheme 1b and c). The results of the application of the Mann–Whitney U-test (MW test) for the smallest samples are presented in Table 6.
| Contact | C–H⋯O/N | C–F⋯O/N | C–Cl⋯O/N | C–Br⋯O/N | C–I⋯O/N |
|---|---|---|---|---|---|
| C–H⋯O/N | −(−) | −(−) | −(−) | +(−) | −(−) |
| C–F⋯O/N | +(+) | −(−) | +(−) | −(−) | −(−) |
| C–Cl⋯O/N | +(+) | +(+) | −(−) | −(−) | −(−) |
| C–Br⋯O/N | +(+) | +(+) | +(+) | −(−) | −(+) |
| C–I⋯O/N | — | — | — | — | — |
The MW nonparametric test was applied given that some of the sub-samples analysed here cannot be classified as normally distributed (see Table 3). The samples of ΔR values related to H/X⋯O/N distances and the samples of the C–H/X⋯O/N angles are considered. It can be assumed that if the null hypothesis, H0, is not rejected (+), then there is no reason from a statistical point of view to reject the statement that samples belong to the same type of interaction, while if we accept the alternative hypothesis, H1 (−), then there are different types of interactions. The weakness of this approach is that it relies on a single parameter (here, ΔR related to distance or Θ, the angle) and applies to arbitrarily selected samples. However, this test can be also applied to other parameters for the same pairs of samples, not only two parameters that are considered in this study.
Here, we look at the results collected in Table 6. Firstly, we look at pairs where the samples differ only by the type of electron donor, nitrogen or oxygen centre. It is surprising that for all pairs of samples of distances and angles, the null hypothesis is rejected. This means that the alternative hypothesis is assumed that in each pair the samples come from different populations. This means that all pairs of C–X⋯O and C–X⋯N interactions cannot be classified as the same type of interaction, and the same concerns the C–H⋯O and C–H⋯N pair of hydrogen bonded systems. This seems to be surprising given that the names of interactions usually come from the type of Lewis acid centre, which is the name of the group as the halogen bond or the name of the element as the hydrogen bond, for example. However, the results of the statistical tests indicate that the type of electron donor may differentiate the interactions.
Here, we compare the hydrogen bond with the sub-classes of halogen bonds, i.e. fluorine, chlorine, bromine and iodine bonds. In the case of angles, the sample of the C–H⋯O hydrogen-bonded systems and any sub-class of halogen bonded systems do not belong to the same population, given that in all these cases, the null hypothesis is rejected and the alternative hypothesis is accepted (see the negative signs in parentheses in the first line of Table 6). In the case of ΔRs, only two samples corresponding to H⋯O and Br⋯O contacts can be classified as belonging to the same population, given that from a statistical point of view there is no reason to reject the null hypothesis (this is the only positive sign that appears in first line of Table 6). At first glance, the latter finding seems to be surprising. However, it is well known that halogen bonds with lighter halogen centres are weaker than the corresponding hydrogen bonds, while in the case of heavier halogen centres, the corresponding halogen bonds are comparable in strength with hydrogen bonds or they are even stronger, and the latter occurs for bromine and iodine atoms acting as the electrophilic sites.76
A distinct situation is observed for the C–H⋯N hydrogen bond, which forms pairs with all sub-classes of halogen bonds that belong to the same populations. This concerns both values related to distances, ΔRs, and angles (see the first left column of Table 6, except for the first top element). This is an interesting observation for all sub-classes of interactions with the nitrogen electron-donor centre. Even all combinations of pairs, for both ΔRs and angles, belong to the same populations. This means that from a statistical point of view, one cannot reject the hypothesis that all C–H/X⋯N interactions belong to the same population.
One can see that the C–F⋯O interaction is a rather separate sub-class belonging to a separate population. The only pair that may be assumed to belong to the same population, C–F⋯O and C–Cl⋯O, concerns distances. There is only one more pair of sub-classes of the halogen bond with an oxygen–electron donor that may be assumed to belong to the same population, C–Br⋯O and C–I⋯O, and it concerns angles.
Sub-classes of halogen bonds-statistical tests for systems calculated theoretically
The statistical tests are applied here for systems calculated theoretically in a former study.87 These are complexes of substituted pyridines with D—X molecules (X = Br, I and D = X and CN). These complexes are linked by X⋯N halogen bonds, given that the halogen centre acts here as the electron acceptor, while the nitrogen centre is the electron donor. Four samples of complexes were analysed in this study, which differ by their Lewis acid unit, i.e., Br2, I2, BrCN and ICN. In other words, the complexes are connected by Br–Br⋯N, I–I⋯N, NCBr⋯N and NCI⋯N links, which determine the samples considered; the same derivatives of pyridine are Lewis base units in these samples.The results for further statistical analysis of the complexes described above were taken from the SI of ref. 87.88 These are the binding energies, |Ebin|s (absolute values are considered here), and the electron densities at the X⋯N bond critical points, BCPs, calculated at the MP2/6-311++G(d,p) level of theory. Here, different parameters are considered than those considered for the experimental results discussed in the previous sections of this study. Geometric parameters related to distances and angles were considered earlier.
As a result, we have eight samples of parameters for complexes with four types of links. Firstly, it was checked whether the samples come from normally distributed populations. Graphical methods: histograms and Q–Q-plots were applied to check if these samples are characterized by normal distributions (Fig. S10–S13 in SI). Again, none of these graphical approaches present a clear normal distribution of a sample. This is why two tests were applied here, the Wilk–Shapiro, WS, test68,89 and the Kolmogorov–Smirnov, KS, one.68 The former test is stronger than the latter one.68,90 The results in Table 7 show that only in one case, for the complexes linked by Br–Br⋯N interactions, the sample of binding energies is not normally distributed according to the WS test, and thus the null hypothesis is rejected here and the alternative hypothesis that this sample has a different than normal distribution is assumed. However, the KS test shows that the assumption the sample is normally distributed cannot be rejected, similar to all the remaining samples.
| Parameter | N⋯Br–Br | N⋯BrCN | N⋯I–I | N⋯ICN |
|---|---|---|---|---|
| |Ebin| | −(+) | +(+) | +(+) | +(+) |
| ρ BCP | +(+) | +(+) | +(+) | +(+) |
Other tests were applied here to check if the samples described above belong to the same populations. This is checked for four samples of binding energies and four samples of electron density values. The results are presented in Table 8. The pairs of samples characterised by the same parameter, |Ebin| or ρBCP, are considered.
| Parameter | N⋯Br–Br | N⋯BrCN | N⋯I–I | N⋯ICN |
|---|---|---|---|---|
| N⋯Br–Br | — | −(−) | +(+) | +(+) |
| N⋯BrCN | −(−) | — | −(−) | −(−) |
| N⋯I–I | −(−) | −(−) | — | +(+) |
| N⋯ICN | −(−) | −(−) | −(−) | — |
The two tests applied here, the Welch T-test and Mann–Whitney U-test, show the same results. Here, we describe the results concerning pairs of samples of binding energy. In the case of pairs characterised by the same type of electron-accepting centre, bromine or iodine, the results are surprising rather. The tests show that the Br–Br⋯N and NCBr⋯N samples do not belong to the same population despite their same type of the electrophilic centre, bromine. The null hypothesis is rejected here, and the alternative hypothesis is accepted. On contrary, in the case of the I–I⋯N and NCI⋯N samples, the null hypothesis that they come from the same population is not rejected. It is also strange that the Br–Br⋯N sample forms pairs that may be assumed to come from the same populations with the I–I⋯N and NCI⋯N samples, despite the different electron-accepting centres present in these pairs, i.e., bromine and iodine. Another situation is observed for the NCBr⋯N with I–I⋯N and NCBr⋯N with NCI⋯N pairs, where the null hypothesis is rejected, and the alternative one is accepted that samples come from different populations.
An unexpected situation occurs for pairs of samples characterized by the electron density at the BCP, ρBCP, parameter. In the case of all pairs of samples, the null hypothesis is rejected and H1 is accepted; there is no pair of samples that may come from the same population. In other words, the classification of interactions in the same group for the systems discussed in this section does not make sense. Even the same type of electron donor and electron acceptor, for Br⋯N or for I⋯N, does not guarantee the same type of interaction. This may lead to the more general conclusion that any previously known interaction, such as halogen bond or hydrogen bond, does not possess specific characteristics. These names only say that in the former interaction, any halogen centre acts as the electron acceptor, while in the latter interaction, this is the hydrogen centre. Both above-mentioned centres are electrophiles according to the definitions of the corresponding interactions.21,91,92 The name of the interactions most often applied in various studies refer to the name of the electrophilic centre.14
Conclusions
Statistical approaches were applied here to analyse samples of complexes linked by halogen and hydrogen bonds. The experimental results on systems taken from the Cambridge Structural Database as well as the theoretical results from MP2-6-311++G(d,p) calculations were analysed using statistical approaches. Some results agree with known findings from earlier studies on these interactions; however other findings are very surprising. Some of the findings of this study are presented below.- Descriptive statistics show different geometrical parameters for C–F⋯O/N fluorine bonds than for other C–X⋯O/N halogen bonds.
- The analysis of the geometrical parameters for greater samples of structures with halogen bonds shows that there are differences between the Z–Cl⋯O/N interactions, where Z is any centre and the other C–Cl⋯O/N chlorine bonds. This may relate to numerous factors; among them, the specific properties of chlorine centres connected with Z-atoms.
- The Mann–Whitney U-test for pairs of samples of ΔR parameters based on the X/H⋯O/N distances shows that the C–H⋯O and C–Br⋯O systems belong to the same population. This is in line with former studies, which indicate that the fluorine and chlorine bonds are much weaker than the corresponding hydrogen bonds, while the bromine bonds are stronger or comparable to the corresponding hydrogen bonds.
- The Mann–Whitney U-test test shows that none of the pairs of corresponding C–H/X⋯O and C–H/X⋯N samples belong to the same population. This means that the type of interaction is strongly connected with the type of electron donor, oxygen or nitrogen in the case of the samples analysed in this study.
- Other results on the structures taken from CSD suggest that the classification of interactions mainly based on the type of Lewis acid centre in a contact with the electron donor does not make sense. This is confirmed by the theoretical results employed in this study from the former article.87,88
Summarizing the findings based on the results presented here, it can be seen that the most useful approach is the descriptive statistical analysis, particularly for normally distributed populations, given that in this case, numerous statistical parameters may be applied, such as mean value, standard deviation or quartiles. This analysis provided numerous interesting observations here. The other statistical tools, as tests, for example, lead to ambiguous results.
It is worth noting that approaches applied in this study may be useful for other analyses where different parameters, for example, geometric, energetic and topological, are compared. These statistical tools may be also useful to analyze molecular structures, interrelations between the above-mentioned parameters, as well as chemical processes.
It is important that based on this study one, can draw a certain conclusion that using existing names for interactions does not make sense. The designations of interactions based on the names of the centres in contact were proposed recently,27 where the Br⋯O interaction analysed in this study is one example. If one assumes that the Lewis acid centre appears at the left site of the mark and the Lewis base centre at the right, the last designation corresponds to halogen bond. This is even more informative than the name “halogen bond” given that it informs of the bromine–oxygen acceptor–donor interaction. The H⋯O mark, another example, is related to the hydrogen bond, where oxygen plays the role of electron donor. However, C–H⋯O and O–H⋯O hydrogen bonds often differ significantly in their properties. Hence, the centres neighbouring the atoms in contact may be indicated, for example (O)H⋯O or (O)H⋯O(C). The last term may correspond to an interaction between carboxylic acids. Alternatively, the (C)Br⋯O mark informs of a halogen bond where the acceptor centre is connected with a carbon. It was also indicated in recent studies that interactions lead to changes in configurations if they are strong enough.93,94 For example, tetrahedral tetrel centres may interact with electron donors, which are tetrel bonds that often lead to a change in the tetrahedral configuration to trigonal bipyramid. However, the same tetrahedral–trigonal bipyramid change is observed for pnictogen or chalcogen centres if the lone electron pairs are considered in the configurations analysed. Thus, maybe the change in conformation is more important in the description (or mark) of the interaction than the name of the central Lewis acid centre?
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ce00826c.References
- Molecular Interactions. From van der Waals to Strongly Bound Complexes, ed. S. Scheiner, John Wiley & Sons, Chichester, 1997 Search PubMed.
- H.-J. Schneider, Angew. Chem., Int. Ed., 2009, 48, 3924–3977 CrossRef CAS PubMed.
- P. Hobza and K. Müller-Dethlefs, Non-Covalent Interactions, Theory and Experiment, Royal Society of Chemistry, Thomas Graham House, Science Park, Milton Road, Cambridge, United Kingdom, 2010 Search PubMed.
- P. Politzer, K. E. Riley, F. A. Bulat and J. S. Murray, Comput. Theor. Chem., 2012, 998, 2–8 CrossRef CAS.
- Intermolecualr Interactions in Crystals, ed. J. Novoa, Royal Chemical Society, United Kingdom, 2018 Search PubMed.
- S. J. Grabowski, Lewis Acid – Lewis Base Interactions: Mechanisms and Related Phenomena, World Scientific, Singapore, 2025 Search PubMed.
- S. J. Grabowski, Chem. Rev., 2011, 11, 2597–2625 CrossRef.
- S. J. Grabowski, Phys. Chem. Chem. Phys., 2014, 16, 1824–1834 RSC.
- J. D. Dunitz, Analysis of the Structure of Organic Molecules, Cornell University Press, Ithaca, 1979 Search PubMed.
- H. B. Bürgi, Angew. Chem., Int. Ed. Engl., 1975, 14, 460–473 CrossRef.
- H. B. Bürgi and J. D. Dunitz, Acc. Chem. Res., 1983, 16, 153–161 CrossRef.
- H. B. Bürgi, J. D. Dunitz and E. Shefter, J. Am. Chem. Soc., 1973, 95, 5065–5067 CrossRef.
- Structure Correlation, ed. H. B. Burgi and J. D. Dunitz, VCH, Weinheim, 1994 Search PubMed.
- S. J. Grabowski, Phys. Chem. Chem. Phys., 2017, 19, 29742–29759 RSC.
- P. Politzer, J. S. Murray and T. Clark, Phys. Chem. Chem. Phys., 2010, 12, 7748–7757 RSC.
- P. Politzer, J. S. Murray and T. Clark, Phys. Chem. Chem. Phys., 2013, 15, 11178–11189 RSC.
- T. Clark, J. S. Murray and P. Politzer, Phys. Chem. Chem. Phys., 2018, 20, 30076–30082 RSC.
- I. Alkorta, J. Elguero and A. Frontera, Crystals, 2020, 10, 180 CrossRef CAS.
- I. Alkorta, I. Rozas and J. Elguero, Chem. Soc. Rev., 1998, 27, 163–170 RSC.
- S. J. Grabowski, W. A. Sokalski and J. Leszczynski, Chem. Phys. Lett., 2006, 422, 334–339 CrossRef CAS.
- S. J. Grabowski, Chem. Commun., 2024, 60, 6239–6255 RSC.
- S. J. Grabowski, J. Phys. Chem. A, 2011, 115, 12340–12347 CrossRef CAS.
- R. H. Crabtree, Chem. Soc. Rev., 2017, 46, 1720–1729 RSC.
- R. Taylor, Cryst. Growth Des., 2024, 24, 4003–4012 Search PubMed.
- L. Brammer, A. Peuronen and T. M. Roseveare, Acta Crystallogr., Sect. C: Struct. Chem., 2023, 79, 204–216 Search PubMed.
- E. Arunan, P. Metrangolo, G. Resnati and S. Scheiner, Cryst. Growth Des., 2024, 24, 8153–8158 CAS.
- S. J. Grabowski, Molecules, 2021, 26, 4939 CrossRef CAS PubMed.
- J. J. Colin and H. F. Gaultier de Claubry, Ann. Chim., 1814, 90, 87–100 Search PubMed.
- J. J. Colin, Ann. Chim., 1814, 91, 252–272 Search PubMed.
- N. Ramasubbu, R. Parthasarathy and P. Murray-Rust, J. Am. Chem. Soc., 1986, 108, 4308–4314 CrossRef CAS.
- C. R. Groom, I. J. Bruno, M. P. Lightfoot and S. C. Ward, Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater., 2016, 72, 171–179 CrossRef CAS.
- R. Wong, F. H. Allen and P. Willett, J. Appl. Crystallogr., 2010, 43, 811–824 CrossRef CAS.
- S. C. Nyburg and W. Wong-Ng, Proc. R. Soc. London, Ser. A, 1979, 367, 29–45 CAS.
- F. Zordan, L. Brammer and P. Sherwood, J. Am. Chem. Soc., 2005, 127, 5979–5989 CrossRef CAS PubMed.
- T. Clark, M. Hennemann, J. S. Murray and P. Politzer, J. Mol. Model., 2007, 13, 291–296 CrossRef CAS.
- P. Politzer, P. Lane, M. C. Concha, Y. Ma and J. S. Murray, J. Mol. Model., 2007, 13, 305–311 Search PubMed.
- M. Formigué and P. Batail, Chem. Rev., 2004, 104, 5379–5418 CrossRef.
- E. Parisini, P. Metrangolo, T. Pilati, G. Resnati and G. Terraneo, Chem. Soc. Rev., 2011, 40, 2267–2278 RSC.
- G. Cavallo, P. Metrangolo, R. Milani, T. Pilati, A. Priimagi, G. Resnati and G. Terraneo, Chem. Rev., 2016, 116, 2478–2601 Search PubMed.
- Halogen Bonding. Fundamentals and Applications, ed. P. Metrangolo and G. Resnati, Springer-Verlag, Berlin, Heidelberg, 2008 Search PubMed.
- P. Metrangolo and G. Resnati, Chem. – Eur. J., 2001, 7, 2511-2519 Search PubMed.
- P. Metrangolo, H. Neukirch, T. Pilati and G. Resnati, Acc. Chem. Res., 2005, 38, 386–395 CrossRef CAS PubMed.
- P. Metrangolo, T. Pilati and G. Resnati, CrystEngComm, 2006, 8, 946–947 RSC.
- P. Metrangolo, J. S. Murray, T. Pilati, P. Politzer, G. Resnati and G. Terraneo, Cryst. Growth Des., 2011, 11, 4238–4246 Search PubMed.
- K. T. Mahmudov, A. V. Gurbanov, F. I. Guseinov and M. F. C. Guedes da Silva, Coord. Chem. Rev., 2019, 387, 32–46 CrossRef CAS.
- K. T. Mahmudov and A. J. L. Pombeiro, Chem. – Eur. J., 2023, 29, e202203861 CrossRef CAS.
- T. B. Richardson, S. de Gala and R. H. Crabtree, J. Am. Chem. Soc., 1995, 117, 12875–12876 CrossRef CAS.
- S. J. Grabowski, W. A. Sokalski and J. Leszczynski, Chem. Phys., 2007, 337, 68–76 CrossRef CAS.
- V. I. Bakhmutov, Dihydrogen Bonds, Principles, Experiments, and Applications, John Wiley & Sons, Hoboken, NJ, USA, 2008 Search PubMed.
- J. Dominikowska and M. Palusiak, Chem. Phys. Lett., 2013, 583, 8–13 CrossRef CAS.
- M. Capdevila-Cortada and J. J. Novoa, CrystEngComm, 2015, 17, 3354–3365 RSC.
- J. Dominikowska, F. M. Bickelhaupt, M. Palusiak and C. Fonseca Guerra, ChemPhysChem, 2016, 17, 474–480 Search PubMed.
- F. F. Awwadi, R. D. Willett, K. A. Peterson and B. Twamley, J. Phys. Chem. A, 2007, 111, 2319–2328 Search PubMed.
- A. Daolio, A. Pizzi, S. K. Nayak, J. Dominikowska, G. Terraneo, P. Metrangolo and G. Resnati, Chem. – Asian J., 2023, 18, e202300520 Search PubMed.
- J. Dominikowska, Cryst. Growth Des., 2025, 25, 3906–3914 CrossRef CAS.
- S. J. Grabowski, Chem. Phys. Lett., 2014, 605–606, 131–136 CrossRef CAS.
- S. Scheiner, Molecules, 2021, 26, 350 CrossRef CAS.
- M. Michalczyk, W. Zierkiewicz and S. Scheiner, Cryst. Growth Des., 2022, 22, 6521–6530 CrossRef CAS.
- K. Kupietz, R. M. Gomola, T. Roisnel, A. Frontera and R. Gramage-Doria, CrystEngComm, 2024, 26, 2131–2135 RSC.
- S. J. Grabowski, Understanding Hydrogen Bonds, Theoretical and Experimental Views, The Royal Society of Chemistry, United Kingdom, Cambridge, 2021 Search PubMed.
- C. C. Wilson, Single Crystal Neutron Diffraction From Molecular Materials, World Scientific Publishing Co. Pre. Ltd., Singapore, 2000 Search PubMed.
- S. Alvarez, Dalton Trans., 2013, 42, 8617–8636 RSC.
- B. Illowsky, S. Dean, D. Birmajer, B. Blount, S. Boyd, M. Einsohn, J. Helmreich, L. Kenyon, S. Lee and J. Taub, Introductory Statistics – 2e, OpenStax, Houston, Texas, 2023 Search PubMed.
- D. J. Sheskin, Handbook of Parametric and Nonparametric Statistical Procedures, Chapman & Hall/CRC, Boca Raton, Florida, 2000 Search PubMed.
- J. F. Deshpande, U. Naik-Nimbalkar and I. Dewan, Nonparametric Statistics, Theory and Methods, World Scientific Publishing, Singapore, 2018 Search PubMed.
- F. Clegg, Simple Statistics: A Course Book for the Social Sciences, Cambridge University Press, Cambridge, 1983 Search PubMed.
- N. M. Razali and Y. B. Wah, Journal of Statistical Modeling and Analytics, 2011, 2, 21–33 Search PubMed.
- M. J. Crawley, Statistics, An Introduction Using R, John Wiley & Sons, United Kingdom, 2015 Search PubMed.
- D. Chopra and T. N. Guru Row, CrystEngComm, 2011, 13, 2175–2186 RSC.
- C. Laurence, J. Graton and J. F. Gal, J. Chem. Educ., 2011, 88, 1651–1657 CrossRef CAS.
- Y.-X. Lu, J.-W. Zou, Q.-S. Yu, Y.-J. Jiang and W.-N. Zhao, Chem. Phys. Lett., 2007, 449, 6–10 CrossRef CAS.
- M. D. Prasanna and T. N. Guru Row, CrystEngComm, 2000, 2, 134–140 RSC.
- M. S. Pavan, K. D. Pasad and T. N. Guru Row, Chem. Commun., 2013, 49, 7558–7560 RSC.
- S. Scheiner, J. Phys. Chem. A, 2020, 124, 7290–7299 CrossRef CAS PubMed.
- P. Gilli, V. Bertolasi, V. Ferretti and G. Gilli, J. Am. Chem. Soc., 1994, 116, 909–915 CrossRef CAS.
- S. J. Grabowski, Phys. Chem. Chem. Phys., 2013, 15, 7249–7259 RSC.
- A. M. Pendas, M. A. Blanco and E. Francisco, J. Chem. Phys., 2006, 125, 184112 CrossRef PubMed.
- M. Hudlicky and J. S. Merola, Tetrahedron Lett., 1990, 31, 7403–7406 CrossRef CAS.
- S. Scheiner, Hydrogen Bonding: A Theoretical Perspective, Oxford University Press, New York, 1997 Search PubMed.
- G. R. Desiraju and T. Steiner, The Weak Hydrogen Bond in Structural Chemistry and Biology, Oxford University Press, New York, 1999 Search PubMed.
- P. Singh, H. B. Singh and R. J. Butcher, J. Organomet. Chem., 2018, 876, 1–9 CrossRef CAS.
- J. P. Declercq, B. Tinant, A. Parfonry, M. van Meerssche, E. Legrand and M. S. Lehmann, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1983, 39, 1401–1405 CrossRef.
- G. J. Jeffrey, An Introduction to Hydrogen Bonding, Oxford University Press, New York, 1997 Search PubMed.
- A. Dhaka, O. Jeannin, E. Aubert, E. Espinosa, M. Fourmigue and le-R. Jeon, Chem. Commun., 2022, 58, 10825–10828 RSC.
- D. R. McGregor, J. C. Speakman and M. S. Lehmann, J. Chem. Soc., Perkin Trans. 1, 1977, 2, 1740–1745 RSC.
- G. Gilli and P. Gilli, J. Mol. Struct., 2000, 552, 1–15 CrossRef CAS.
- A. Pisati, A. Forni, S. Pieraccini and M. Sironi, IUCrJ, 2025, 12, 188–197 CrossRef CAS PubMed.
- Table S3 of supporting information DOI:10.1107/S2052252525000363/pen5006sup1.pdf.
- S. S. Shapiro and M. B. Wilk, Biometrika, 1965, 52, 591–611 CrossRef.
- M. A. Stephens, J. Am. Stat. Assoc., 1974, 69, 730–737 CrossRef.
- E. Arunan, G. R. Desiraju, R. A. Klein, J. Sadlej, S. Scheiner, I. Alkorta, D. C. Clary, R. H. Crabtree, J. J. Dannenberg, P. Hobza, H. G. Kjaergaard, A. C. Legon, B. Mennucci and D. J. Nesbitt, Pure Appl. Chem., 2011, 83, 1637–1641 CrossRef CAS.
- G. R. Desiraju, P. S. Ho, L. Kloo, A. C. Legon, R. Marquardt, P. Metrangolo, P. Politzer, G. Resnati and K. Rissanen, Pure Appl. Chem., 2013, 85, 1711–1713 CrossRef CAS.
- S. J. Grabowski, Sci., 2022, 4, 17 CrossRef CAS.
- S. J. Grabowski, Struct. Chem., 2019, 30, 1141–1152 CrossRef CAS.
Footnote |
| † Dedicated to Professor Resnati, celebrating a career in fluorine and noncovalent chemistry on the occasion of his 70th birthday. |
| This journal is © The Royal Society of Chemistry 2026 |