Diiron monooxygenases in natural product biosynthesis
Abstract
Covering: up to 2017
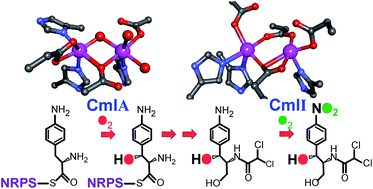
The participation of non-heme dinuclear iron cluster-containing monooxygenases in natural product biosynthetic pathways has been recognized only recently. At present, two families have been discovered. The archetypal member of the first family, CmlA, catalyzes β-hydroxylation of L-p-aminophenylalanine (L-PAPA) covalently linked to the nonribosomal peptide synthetase (NRPS) CmlP, thereby effecting the first step in the biosynthesis of chloramphenicol by Streptomyces venezuelae. CmlA houses the diiron cluster in a metallo-β-lactamase protein fold instead of the 4-helix bundle fold of nearly every other diiron monooxygenase. CmlA couples O2 activation and substrate hydroxylation via a structural change caused by formation of the L-PAPA-loaded CmlP:CmlA complex. The other new diiron family is typified by two enzymes, AurF and CmlI, which catalyze conversion of aryl-amine substrates to aryl-nitro products with incorporation of oxygen from O2. AurF from Streptomyces thioluteus catalyzes the formation of p-nitrobenzoate from p-aminobenzoate as a precursor to the biostatic compound aureothin, whereas CmlI from S. venezuelae catalyzes the ultimate aryl-amine to aryl-nitro step in chloramphenicol biosynthesis. Both enzymes stabilize a novel type of peroxo-intermediate as the reactive species. The rare 6-electron N-oxygenation reactions of CmlI and AurF involve two progressively oxidized pathway intermediates. The enzymes optimize efficiency by utilizing one of the reaction pathway intermediates as an in situ reductant for the diiron cluster, while simultaneously generating the next pathway intermediate. For CmlI, this reduction allows mid-pathway regeneration of the peroxo intermediate required to complete the biosynthesis. CmlI ensures specificity by carrying out the multistep aryl-amine oxygenation without dissociating intermediate products.
- This article is part of the themed collection: Metalloenzymes in Natural Product Biosynthetic Pathways


 Please wait while we load your content...
Please wait while we load your content...