Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Size-selected gold clusters on porous titania as the most “gold-efficient” heterogeneous catalysts
Andreas
Dollinger
a,
Lukas
Stolch
a,
Yuan
Luo
a,
Martin
Beck
a,
Christoph H.
Strobel
a,
Matthias
Hagner
a,
Stefan
Dilger
b,
Malin
Bein
b,
Sebastian
Polarz
*b,
Gerd F.
Gantefoer
*a,
Young-Dok
Kim
c and
Sebastian
Proch
*a
aFachbereich für Physik, Universität Konstanz, Universitätsstrasse 10, 78464 Konstanz, Germany. E-mail: gerd.gantefoer@uni-konstanz.de; sebastian.proch@uni-konstanz.de
bFachbereich für Chemie, Universität Konstanz, Universitätsstrasse 10, 78464 Konstanz, Germany. E-mail: sebastian.polarz@uni-konstanz.de
cDepartment of Chemistry, Sungkyunkwan University, Suwon 440-746, Korea
First published on 8th April 2014
Abstract
Research on homogeneous and heterogeneous catalysis is indeed convergent and finds subnanometric particles to be at the heart of catalytically active species. Here, monodisperse gold clusters are deposited from the gas phase onto porous titania generating well-defined model systems and the resulting composite materials exhibit a sharp size-dependency on the number of gold atoms per cluster and exceptionally high-turnovers toward the bromination of 1,4-dimethoxybenzene are observed. This indicates that the deliberate generation of active centres is of utmost importance for the creation of the most “gold-efficient” catalysts.
Introduction
In 19851/872 first subtle clues were found that gold presents an element as valuable for chemists as for jewellers. Today it has become ubiquitous in heterogeneous (epoxidation,3 oxidative coupling of anilines,4 selective oxidation with dioxygen,5 CO oxidation6) and homogeneous (a multitude of organic transformations7) catalysis alike. This in turn leads to a situation where gold as a catalyst has to compete with gold as an investment especially in times of “uncertainty”.8 So if gold is successfully to be used as a catalyst no gold must be wasted. As per definition the most atom-efficient catalyst system has to consist exclusively of active sites9 preferably with only a single metal atom per site. The latter requirement can be fulfilled for molecular catalysts applied in the homogeneous phase, ultimately enzymes.10 However, there are natural limits for the application of molecular catalysts, for instance at high process temperatures or in the absence of solvents. Thus, solid-state compounds applied in the heterogeneous state represent a distinct and important class of catalysts. Unfortunately, it is currently out of range to tailor make solid-state catalysts with such high atom-efficiency as described above. Research in this field is vital in particular for heterogeneous gold catalysts.An interesting connection between molecular and solid-state gold catalysts was revealed by Corma et al. Impressive TONs (turn over numbers), approaching the efficiency of enzymes, have recently been observed for the ester-assisted hydration of alkynes and bromination of aromatics catalysed by AuCl in solution. However, Corma et al. uncovered a deeper understanding regarding the active sites of Au(+1) and Au(+3) catalysts, as it was found that subnanometric gold clusters (1–10 atoms) are responsible for catalytic activity.11 These findings are convergent with the invaluable work of Hutchings et al. on heterogeneous gold catalysts showing that CO oxidation is actually catalysed by subnanometric bilayer clusters (1–10 atoms) and larger particles merely act as spectator species.6 The group of Corma et al. also tried to transfer their findings to heterogeneous catalysis by preparing Au5- and Au8-PAMAM (poly[amidoamine])12 and used them in the above reactions. Unfortunately results obtained were TONs two orders of magnitude lower compared with homogenous clusters in solution, leaving a TON of 102 for the bromination of 1,4-dimethoxybenzene with N-bromosuccinimide (NBS).11 This drop in TON is most likely due to encapsulation of clusters by the PAMAM dendrimer template. However, dendrimer template directed synthesis routes12,13 are currently the only wet chemical methods to make size-selected metal clusters, apart from “magic” number clusters, we are aware of.
Previously, work on size-selected gold clusters deposited on TiO2(110) single crystals under UHV conditions found an extreme size-dependent reactivity towards the oxidation of CO, while Au5 is almost inert Au6 and Au7 are very active.14,15 While catalytic activity serves as a valuable probe for conservation of size-dependent properties of soft-landed clusters (<1 eV per atom) XPS measurements of as-deposited clusters gave rise to the same binding energy of 84.7 eV (Au 4f7/2) for all Aun ranging from n = 1 to 7.15 Recently the authors have carried out TPD experiments towards CO oxidation activity on Aun/(TiO2)93/HOPG (n = 5 to 8) composite materials demonstrating that size-dependent properties of catalytic activity observed in single-crystal experiments were preserved on these nanoparticle like TiO2-clusters (diameter approx. 1 nm). Moreover, results indicate that only Au6 and Au7 are active for CO oxidation, creating a situation where coincidental formation of “active species” on high-surface area TiO2 supports is very unlikely.16 These findings led us to believe that size-dependent properties of gold clusters can be probed in real catalytic environments on relevant supports like porous titania. Especially in the light that Corma et al. discovered TONs of 107.11 A realistic value of deposited clusters within a reasonable time frame and without excessive consumption of gold sputter targets (see Scheme 1) is about 1013. This should give rise to the formation of 1020 product molecules, corresponding to millimolar quantities which are easily detected by gas-chromatographic methods.
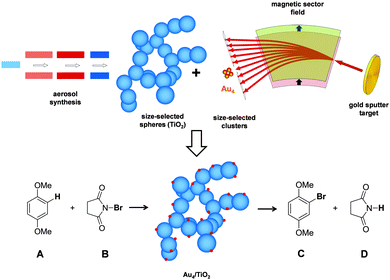 | ||
| Scheme 1 Example of the synthesis of a composite material consisting of size-selected clusters on a highly porous, aerogel-like TiO2, here Au4/TiO2, and its application in catalysis. | ||
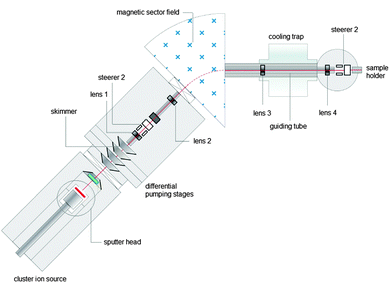
Here, we suggest size-selected cluster deposition as a tool to deliberately create active sites on virtually any catalyst support by purely applying gas phase methods (Scheme 1), which is at the heart of the vision of “nanocatalysis”.17 Highly porous, highly crystalline metal oxide materials can be produced in a controlled fashion facilitating an aerosol-assisted method.18–20 During this process, nanosized primary particles form, which then agglomerate into fractal, highly porous networks. The advantage of these aerogel-like materials is that particles dispersed in the gas-phase are able to penetrate easily and very deep into the porous framework. Thus, in a second step metal clusters are mass-selected in a magnetic sector field and are soft-landed (size-selected clusters) on the metal oxide support, hereby the masses can be selected on an atom-by-atom basis (cluster deposition setup is described in the experimental section). A particular system Aun/TiO2 has been chosen for three reasons: (i) gold is very oxidation resistant and can be transferred from ultra-high vacuum (UHV) to ambient conditions without changes in chemical integrity;21–24 (ii) gold particles on titania exhibit strong metal support interaction (SMSI);25 and (iii) it has been shown that Au/TiO2 is a superior catalyst for homocoupling of iodobenzene compared to Aun-PAMAM.26 Here it will be shown that size-selected clusters do not only act as model systems for catalysis under UHV conditions but also as very efficient catalysts for the bromination of 1,4-dimethoxybenzene in solution.
Experimental
Since this work contains methodology of heterogeneous catalysis in solution and cluster deposition both approaches are explained in detail in the following section.General experimental section
XPS (X-ray photoelectron spectroscopy). XPS spectra have been recorded using an Omicron EA 125 CHA (concentric hemispherical analyzer) with a base pressure in the main chamber of ≈5 × 10−10 mbar using the Mg Kα-line (1253.6 eV). Peak fitting was done via CASA XPS and a Gaussian–Lorentzian fit (GL30), the peak separation for Au 4f was kept at Δ = 3.67 eV.27
SEM (scanning electron microscopy). The highly porous TiO2-layer has been characterized using a Zeiss Cross Beam 1540 XB, which is a combination of a Scanning Electron Microscope (SEM) and a Focused Ion Beam (FIB). To determine the thickness a cross-section was cut into the layer using the FIB and analyzed by SEM.
TEM (transmission electron microscopy). Micrographs have been obtained by dip coating TiO2 primary particles (13.4 nm) onto copper/carbon grids, deposition of Au120 clusters at a cluster current of 200 pA for 2 h, and subsequent investigation within a Jeol ARM instrument.
GC/MS (gas chromatography/mass spectrometry). GC measurements were done using an Agilent 7890A machine equipped with a Zebron ZB-5MS column with a length of 30 m, an inner diameter of 0.25 mm and a film thickness of 0.25 μm. The temperature program for GC measurements was as follows: (i) 50 °C held for 1 min; (ii) the temperature is raised to 280 °C at a rate of 30 °C min−1; (iii) the temperature is held at 280 °C for another 3 min. MS measurements were performed on an Agilent 5975C apparatus.
Dip coating of aerogel-like TiO2 onto tantalum backing plates. Aerogel-like TiO2 (1 mg) is suspended in 0.2 ml of ethanol (pA) and dip coated onto a 10 mm × 10 mm tantalum backing plate. The ethanol is allowed to evaporate from the sample at room temperature and then it is introduced into the deposition chamber of the cluster deposition apparatus.
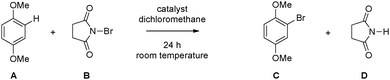
Bromination of 1,4-dimethoxybenzene. 1,4-Dimethoxybenzene (A, 0.5 mmol, 69 mg, see Scheme 1) and N-bromosuccinimide (B, NBS, 0.5 mmol, 89 mg) are placed inside a 5 ml vial and dissolved in 2 ml dichloroethane. The catalyst (blank, AuCl [5 mg], HAuCl4 [5 mg], TiO2 [5 mg], Au4/TiO2 [1 mg], Au7/TiO2 [1 mg], Au8/TiO2 (a) [1 mg], Au8/TiO2, (b) [1 mg], Au9/TiO2 [1 mg], Au120/TiO2 [1 mg]) was added, the vial sealed with a cap, and the reaction stirred for 24 h at room temperature. After that time period had elapsed the vial was reopened and dodecane (0.5 mmol, 64 μl) was added to the mixture as internal standard for GC/MS, the mixture was then filtered, and diluted in hexane prior to the actual GC/MS measurement.11
In order to obtain the number of generated molecules of 1-bromo-2,5-dimethoxybenzene (C, see Scheme 1) GC calibration with the temperature ramp mentioned above has been done. Therefore, 1 mmol of dodecane (internal standard) and 1 mmol are dissolved in hexane and the corresponding chromatogram is measured. The GC/MS method allows unambiguous identification of both substances without having to obtain chromatograms of isolated compounds first. The peak areas of C and dodecane within the chromatogram are linked by the following relationship: 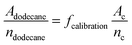 , where A is the area and n the number of moles of the respective substances. This procedure yields the calibration factor
, where A is the area and n the number of moles of the respective substances. This procedure yields the calibration factor 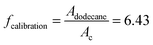 . With this known factor the amount of C within the solution after reaction can be determined by adding a known amount of dodecane. The TONs reported in Table 2 are obtained by dividing the number of product molecules C over (i) the number of clusters in the catalyst (TON per gold cluster) or (ii) the number of gold atoms in the catalyst (TON per gold atom) given in units
. With this known factor the amount of C within the solution after reaction can be determined by adding a known amount of dodecane. The TONs reported in Table 2 are obtained by dividing the number of product molecules C over (i) the number of clusters in the catalyst (TON per gold cluster) or (ii) the number of gold atoms in the catalyst (TON per gold atom) given in units  , respectively. The number of clusters/gold atoms is obtained by measuring and integrating the neutralization current during cluster deposition (see below). This number corresponds to the number of clusters/atoms within the catalyst since all materials obtained from deposition are used as reaction catalysts.
, respectively. The number of clusters/gold atoms is obtained by measuring and integrating the neutralization current during cluster deposition (see below). This number corresponds to the number of clusters/atoms within the catalyst since all materials obtained from deposition are used as reaction catalysts.
Synthesis of aerogel-like TiO2. The liquid precursor Ti(OisoPr)4 is injected into the evaporation zone of the aerosol synthesis setup using a syringe pump from KD Scientific model KS200. The addition of the precursor to the evaporation zone (EZ) via the syringe pump allows us to keep an adjusted concentration of the precursor in the aerosol constant. Tube ovens from Nabertherm were used for the evaporation and the decomposition of the precursor. Exact amounts of gases (N2 and O2) are added using mass flow controllers from Bronkhorst (model el-flow select). Typical flow rates are fnitrogen = 1.2 L min−1 and foxygen = 0.3 L min−1. The precursor concentration is given by the evaporation rate and the volume of the added gases. Typically an evaporation temperature of 90 °C was selected and the concentration of the precursor could be adjusted in the range of 102–106 μg m−3. Formation of titania takes place in the decomposition zone of the set-up, which is heated to 800 °C. Separation of the TiO2 materials from the aerosol is achieved using a thermophoresis unit. The powder X-ray diffraction pattern of the corresponding material is shown in Fig. 2.
Deposition of mass-selected Aun−. Aerogel-like TiO2 dip-coated (see above) on tantalum plates has been introduced into the deposition chamber of the cluster setup. The chamber was pumped down to ≈1 × 10−8 mbar and Aun− was deposited onto dip-coated tantalum plates while the neutralization current was measured and integrated to obtain the total number of clusters since they carry a single negative charge. Average deposition currents for the different clusters were 20–100 pA yielding deposition times of several hours. After deposition the samples were removed from the deposition chamber and introduced into another UHV system for XPS analysis.
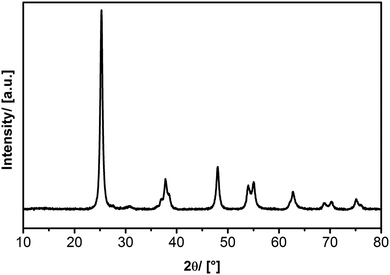 | ||
| Fig. 2 Powder X-ray diffraction (Cu Kα) of titania with a primary particle size of approx. 13 nm in the anatase phase. | ||
Results and discussion
Aerogel-like TiO2 with a primary particle size of 13.4 nm has been used as a support material for size-selected gold clusters. The particle size of titania crystallites is determined from the analysis of peak-broadening of the signals obtained by powder X-ray diffraction (PXRD, see Fig. 2) using the Scherrer-method. In addition, TEM micrographs were checked and showed that the size of the TiO2 nanocrystals is in the same region. However, it should be noted that the value obtained from PXRD provides an average size, whereas there is of course a certain polydispersity in the sample.An innovative approach which permits complementation of real-space techniques for thin-film characterization from reciprocal space is discussed by Perlich et al.29 It involves grazing-incidence small-angle X-ray scattering (GISAXS) data and their evaluation. Unfortunately, we do not have any access to SAXS equipment capable of GI-measurements. Therefore, we could only investigate the porosity of the sample by SEM and TEM (which gives only a qualitative impression) and N2 physisorption analysis, which at least gives quantitative figures for internal surface area, pore volume and pore-size distribution. However, giving a precise value for the porosity 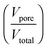 is not possible at the moment. However, it is believed that this is also not critical information for the current contribution, since it is undoubtedly correct that the used, aerogel-like porous TiO2 materials possess high porosity, low density and lots of open pore-space.
is not possible at the moment. However, it is believed that this is also not critical information for the current contribution, since it is undoubtedly correct that the used, aerogel-like porous TiO2 materials possess high porosity, low density and lots of open pore-space.
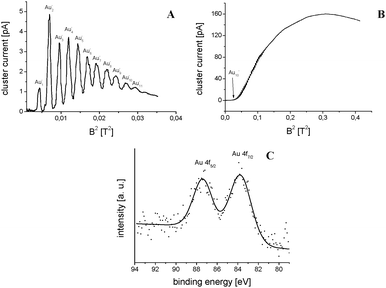
The described material has been suspended in ethanol and dip-coated onto tantalum backing plates to facilitate gold cluster deposition. An SEM investigation of the as-prepared plates shows a thickness of 16 μm and conserved porosity of the support material (Fig. 4). Furthermore the number of TiO2 particle monolayers can be determined to be about 1200. The film has also been investigated by XPS, however, due to considerable film thicknesses charging effects occur. In order to account for charging Ti(+4) 2p3/2, which is to be found at 259.0 eV, has been used as a reference.27 TiO2/Ta plates have been inserted into a UHV deposition chamber and size-selected gold clusters have been deposited. The mass spectrum of these gold species in the gas phase is presented in Fig. 3A and B. It basically shows monodisperse clusters for small species. Deposition currents for gold clusters varied between 20 and 100 pA yielding deposition times of several hours. Numbers of deposited clusters ranged from 3 × 1011 to 8 × 1012, which multiplied by “n” yields the number of gold atoms in the sample (see Table 1).
 | ||
| Fig. 4 SEM image of aerogel-like TiO2 dip-coated onto a tantalum backing plate. The thickness has been determined by cross-section and is shown as green bars in the image. | ||
| Entry | Composite material | Number of Au clusters | Number of Au atoms |
|---|---|---|---|
| 1 | Au4/TiO2 | 2.7 × 1011 | 1.1 × 1012 |
| 2 | Au7/TiO2 | 1.3 × 1012 | 9.3 × 1012 |
| 3 | Au8/TiO2 (a) | 1.9 × 1012 | 1.5 × 1013 |
| 4 | Au8/TiO2 (b) | 8.1 × 1012 | 6.5 × 1013 |
| 5 | Au9/TiO2 | 1.1 × 1012 | 1.0 × 1013 |
| 6 | Au120/TiO2 | 1.6 × 1012 | 1.9 × 1014 |
A close packed monolayer of gold, e.g., Au(111), contains about 1.5 × 1015 atoms per cm2 which in the observed deposition spot (circular with 3 mm in diameter, 7 × 10−2 cm2) translates into ≈1 × 1014 atoms within the spot per monolayer. The lower limit for detection of surface species by XPS is about 1% of a monolayer corresponding to ≈1 × 1012 atoms on the top monolayer of the TiO2 film. For small clusters, especially Au4−, this cannot be achieved since these small clusters are able to penetrate deep into the porous film structure (≈1200 ML). The huge number of monolayers in addition to the small amount of gold clusters deposited makes observation of an XPS signal very unlikely, except for the largest clusters, Au120−, which is exactly what is found. In the Au120/TiO2 composite the number of gold atoms is about one order of magnitude higher in comparison (see Table 1). On top of that these large clusters might have problems penetrating into the network of TiO2 and unlike the other clusters accumulate on the top TiO2 layer making detection via XPS possible (Fig. 3C). The binding energy after charge correction yields a value of 83.7 eV for Au 4f7/2 which is close to the bulk value of 84.0 eV.27 This is to be expected for such big clusters.
The same cross-section SEM technique should in principle be useful to determine the penetration depths of the clusters into the TiO2 layer. The problem with this approach is EDX sensitivity with a lower value of 0.5 to 1 mass%. For samples provided here 1 mg of TiO2 was used which means that 10 μg would be needed to be deposited per sample if the cluster ion beam is focused down maybe 1 μg or less. On the Au4 sample around 0.4 ng gold was deposited, 3 ng for Au7, 5 ng for Au8, 3 ng for Au9 and 60 ng for Au120, respectively. Fig. 3B presents a mass spectrum over the complete range of the respective magnetron sputter source which unequivocally shows that larger clusters are much easier to make than smaller ones. So an attempt was made to deposit large clusters in order to achieve enough material to meet EDX sensitivity criteria. Au120 was deposited for 2 h onto 1 mg of TiO2 suspended in ethanol on a TEM grid which yielded a total amount of approximately 300 ng which was still too low to be detected by EDX, however, the corresponding TEM micrograph of the sample in Fig. 5 demonstrates massive coagulation of these clusters which should not be the case if it was possible for this size to penetrate deeply into the TiO2 network. On the other hand it was not possible to find any evidence for smaller clusters within TEM micrographs due to their very low mass and deposition currents. We believe these measurements tentatively show the low penetration depths of large Au120 clusters into the titania framework.
Composite materials presented in Table 1 have been used as catalysts for the bromination of 1,4-dimethoxybenzene (1) using NBS (2). The Aun/TiO2 materials were removed from the tantalum backing plate and transferred directly into reaction vessels containing 1 and 2 dissolved in dichloroethane which have been stirred for 24 h at room temperature (see the Experimental section). The conversion of the reaction has been checked via gas chromatography. The results are found in Table 2. Compared to previous results11,30 achieved TONs with size-selected gold clusters on titania are extremely high, while a TON of 102 is described in the literature TONs as high as 108 are found for Au7/TiO2 (Table 2, entry 6, Fig. 6). This gap in TON should be ascribed to the “naked” nature of the clusters obtained by “soft-landing” in UHV compared to size-selected clusters created by dendrimer templates. Clusters formed within a dendrimer become encapsulated in the template which acts as a large ligand with multiple binding sites12,13 preventing leaching into solution but also decreasing exposure to reactants. A closer look shows that composite materials Aun/TiO2 (n = 4, 7, 9) exhibit similar TONs based on the number of gold atoms (Table 2, entries 5, 6, 9 and Fig. 6) which might be indicative of dissolution of clusters into the reactant solution facilitating behavior as described by J. Oliver-Meseguer et al.11 Due to its d10s1 valence electron configuration gold is considered a “simple” metal and can be treated by the Jellium model,31 according to which Au4, Au7, and Au9 do not exhibit any particular stability and thus are prone to show this type of behavior. On the other hand Au8 (Table 2, entries 7 and 8 and Fig. 6) is a very stable magic number cluster completely filling the 1p cluster valence orbital and indeed the TON per atom is two orders of magnitude lower compared to the previous examples. This is indicative of a stable cluster which remains on the surface (in the pores) giving rise to less exposure to reactant molecules and in turn lower TONs per gold atom. The large Au120 clusters have similar low TONs per gold atom which can be blamed on the reduced solubility of larger clusters compared to smaller ones (no particular stability according to the Jellium model).
| Entry | Catalyst | Conversion [%] | TON per gold clusterc | TON per gold atomd |
|---|---|---|---|---|
a 5 mg was added.
b 1 mg was added.
c In units of 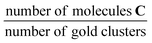 .
d In units of .
d In units of 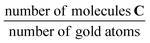 . .
|
||||
| 1 | None (blank) | ≈0 (Trace amounts) | 0 | 0 |
| 2 | AuCla | ≈100 | Undefined | 23 |
| 3 | HAuCl4a | ≈100 | Undefined | 33 |
| 4 | TiO2a | ≈0 (Trace amounts) | 0 | 0 |
| 5 | Au4/TiO2b | 4.4 | 5.1 × 107 | 1.3 × 107 |
| 6 | Au7/TiO2b | 52.0 | 1.2 × 108 | 1.7 × 107 |
| 7 | Au8/TiO2 (a)b | 3.2 | 5.1 × 106 | 6.4 × 105 |
| 8 | Au8/TiO2 (b)b | 16.0 | 6.5 × 106 | 8.1 × 105 |
| 9 | Au9/TiO2b | 46.0 | 1.3 × 106 | 1.4 × 107 |
| 10 | Au120/TiO2b | 20.0 | 3.6 × 107 | 3.0 × 105 |
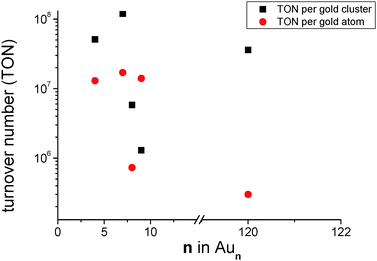 | ||
Fig. 6 Graphical representation of the content of Table 2 showing the TON per gold cluster (squares) and the TON per gold atom (filled circles) plotted against the number of atoms in the cluster for Au4/TiO2, Au7/TiO2, Au8/TiO2, and Au9/TiO2 in units of 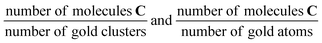 , respectively. Most striking is the difference in TON per gold atom between n = 8 and 120 and n = 4, 7, 9. , respectively. Most striking is the difference in TON per gold atom between n = 8 and 120 and n = 4, 7, 9. | ||
Direct sputter deposition from a target onto a substrate does not allow the landing of intact monodisperse pre-formed entities from the gas-phase onto the sample contrary to soft-landing of size-selected particles. It has to be admitted that sputter deposition is much easier to be conducted and much faster than the latter method. Moreover, the ingenious method of GISAXS is able to characterize film growth under sputter conditions in real-time, results obtained by Schwartzkopf et al.32 show four-stage growth and the corresponding thresholds with submonolayer resolution. This knowledge allows tremendous control over film properties and cluster size in the nanometer range. Work conducted by Valden et al.33 shows that gold catalysts on titania for CO oxidation at exposure times of 10 Torr CO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O2 (2
O2 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) for 120 min (7.2 × 1010 L) show best reaction rates for islands with a diameter between 2.5 and 3.5 nm. These islands show a thickness of only two layers and their activity is blamed on the high aspect ratio and related quantum size effects introducing a band-gap into these islands. Employing the GISAXS method should facilitate exceptionally simple preparation of CO oxidation catalysts in this pressure regime. However, if the reactant exposure is lowered down to 0.2–600 L only clusters Au6 and Au7 show catalytic activity on TiO2 which was presented in a recent contribution (see above),16i.e., indeed every atom counts. Larger and smaller clusters would be a waste of gold under such conditions. Similar results were found by J. Oliver-Meseguer et al. for the bromination of aromatics using gold clusters in solution as described above. Although sputter deposition of gold clusters is unmatched in simplicity and duration for the physical preparation of heterogeneous gold catalysts it is believed that the full potential of the non-scalable sub-nano regime can only be exploited by size-selected cluster deposition under soft-landing conditions so far.
1) for 120 min (7.2 × 1010 L) show best reaction rates for islands with a diameter between 2.5 and 3.5 nm. These islands show a thickness of only two layers and their activity is blamed on the high aspect ratio and related quantum size effects introducing a band-gap into these islands. Employing the GISAXS method should facilitate exceptionally simple preparation of CO oxidation catalysts in this pressure regime. However, if the reactant exposure is lowered down to 0.2–600 L only clusters Au6 and Au7 show catalytic activity on TiO2 which was presented in a recent contribution (see above),16i.e., indeed every atom counts. Larger and smaller clusters would be a waste of gold under such conditions. Similar results were found by J. Oliver-Meseguer et al. for the bromination of aromatics using gold clusters in solution as described above. Although sputter deposition of gold clusters is unmatched in simplicity and duration for the physical preparation of heterogeneous gold catalysts it is believed that the full potential of the non-scalable sub-nano regime can only be exploited by size-selected cluster deposition under soft-landing conditions so far.
Conclusions
Sharp size-dependencies for catalytic activities are well-known from reactions under UHV conditions with size-selected cluster catalysts,14–16,34,35 and only experiments involving size-selected deposition made it fashionable to think of the exact number of atoms in heterogeneous catalysis. Here it could be shown that the “pressure gap” can be overcome for “soft-landed” gold clusters on TiO2 which act as very efficient bromination catalysts in solution under “real” catalytic conditions. Since sharp size-dependence is also observed and can be explained in terms of the Jellium model. In conclusion size-selected cluster deposition presents an invaluable tool to facilitate deliberate creation of active sites in heterogeneous catalysis which is the idea of nanocatalysis.17 As future steps deposition currents for gold clusters have to be optimized to synthesize sufficient amounts for in-depth mechanistic analysis of catalytic reactions.Acknowledgements
Financial support from the German research foundation (DFG) is gratefully acknowledged (Grant No.: GA 389/24-1 and PO 780/8-1).Notes and references
- G. J. Hutchings, J. Catal., 1985, 96, 292–295 CrossRef CAS.
- M. Haruta, T. Kobayashi, H. Sano and N. Yamada, Chem. Lett., 1987, 405–408 CrossRef CAS.
- M. D. Hughes, Y. J. Xu, P. Jenkins, P. McMorn, P. Landon, D. I. Enache, A. F. Carley, G. A. Attard, G. J. Hutchings, F. King, E. H. Stitt, P. Johnston, K. Griffin and C. J. Kiely, Nature, 2005, 437, 1132–1135 CrossRef CAS PubMed.
- A. Grirrane, A. Corma and H. Garcia, Science, 2008, 322, 1661–1664 CrossRef CAS PubMed.
- M. Turner, V. B. Golovko, O. P. H. Vaughan, P. Abdulkin, A. Berenguer-Murcia, M. S. Tikhov, B. F. G. Johnson and R. M. Lambert, Nature, 2008, 454, 981–983 CrossRef CAS PubMed.
- A. A. Herzing, C. J. Kiely, A. F. Carley, P. Landon and G. J. Hutchings, Science, 2008, 321, 1331–1335 CrossRef CAS PubMed.
- A. S. K. Hashmi, Chem. Rev., 2007, 107, 3180–3211 CrossRef CAS PubMed.
- Y. Oh, D. Gros, C. Alcidi and P. de Grauwe, Economic Policy, CEPS Special Reports, 2010.
- H. S. Taylor, Proc. R. Soc. London, Ser. A, 1925, 108, 105–111 CrossRef CAS.
- G. J. Hutchings, Gold Bull., 2009, 42, 260–266 CrossRef CAS.
- J. Oliver-Meseguer, J. R. Cabrero-Antonino, I. Dominguez, A. Leyva-Perez and A. Corma, Science, 2012, 338, 1452–1455 CrossRef CAS PubMed.
- J. Zheng, C. W. Zhang and R. M. Dickson, Phys. Rev. Lett., 2004, 93, 077402 CrossRef.
- K. Yamamoto, T. Imaoka, W. J. Chun, O. Enoki, H. Katoh, M. Takenaga and A. Sonoi, Nat. Chem., 2009, 1, 397–402 CrossRef CAS PubMed.
- S. S. Lee, C. Y. Fan, T. P. Wu and S. L. Anderson, J. Am. Chem. Soc., 2004, 126, 5682–5683 CrossRef CAS PubMed.
- S. Lee, C. Y. Fan, T. P. Wu and S. L. Anderson, J. Chem. Phys., 2005, 123, 124710 CrossRef PubMed.
- X. Tang, J. Schneider, A. Dollinger, Y. Luo, A. S. Worz, K. Judai, S. Abbet, Y. D. Kim, G. F. Gantefor, D. H. Fairbrother, U. Heiz, K. H. Bowen and S. Proch, Phys. Chem. Chem. Phys., 2014, 16, 6735–6743 RSC.
- R. Schlögl and S. B. Abd Hamid, Angew. Chem., Int. Ed., 2004, 43, 1628–1637 CrossRef PubMed.
- S. Dilger, C. Hintze, M. Krumm, C. Lizandara-Pueyo, S. Deeb, S. Proch and S. Polarz, J. Mater. Chem., 2010, 20, 10032–10040 RSC.
- S. Dilger, C. Lizandara-Pueyo, M. Krumm and S. Polarz, Adv. Mater., 2012, 24, 543–548 CrossRef CAS PubMed.
- D. Lehr, D. Grossmann, W. Gruenert and S. Polarz, Nanoscale, 2014, 6, 1698–1706 RSC.
- D. C. Lim, R. Dietsche, M. Bubek, G. Ganteför and Y. D. Kim, ChemPhysChem, 2006, 7, 1909–1911 CrossRef CAS PubMed.
- D. C. Lim, R. Dietsche, G. Gantefor and Y. D. Kim, Chem. Phys. Lett., 2008, 457, 391–395 CrossRef CAS PubMed.
- D. C. Lim, R. Dietsche, G. Gantefor and Y. D. Kim, Chem. Phys., 2009, 359, 161–165 CrossRef CAS PubMed.
- D. C. Lim, R. Dietsche, G. Ganteför and Y. D. Kim, Appl. Surf. Sci., 2009, 256, 1148–1151 CrossRef CAS PubMed.
- D. W. Goodman, Catal. Lett., 2005, 99, 1–4 CrossRef CAS.
- M. Boronat, A. Leyva-Pérez and A. Corma, Acc. Chem. Res., 2014, 47, 834–844 CrossRef CAS PubMed.
- J. F. Moulder, W. F. Stickle, P. E. Sobol and K. D. Bomben, Handbook of X-ray photoelectron spectroscopy, Physical Electronics, Inc., 1995 Search PubMed.
- H. Haberland, M. Karrais, M. Mall and Y. Thurner, J. Vac. Sci. Technol., A, 1992, 10, 3266–3271 CAS.
- J. Perlich, G. Kaune, M. Memesa, J. S. Gutmann and P. Müller-Buschbaum, Philos. Trans. R. Soc., A, 2009, 367, 1783–1798 CrossRef CAS PubMed.
- F. Mo, J. M. Yan, D. Qiu, F. Li, Y. Zhang and J. Wang, Angew. Chem., Int. Ed., 2010, 49, 2028–2032 CrossRef CAS PubMed.
- W. A. de Heer, Rev. Mod. Phys., 1993, 65, 611–676 CrossRef CAS.
- M. Schwartzkopf, A. Buffet, V. Korstgens, E. Metwalli, K. Schlage, G. Benecke, J. Perlich, M. Rawolle, A. Rothkirch, B. Heidmann, G. Herzog, P. Muller-Buschbaum, R. Rohlsberger, R. Gehrke, N. Stribeck and S. V. Roth, Nanoscale, 2013, 5, 5053–5062 RSC.
- M. Valden, X. Lai and D. W. Goodman, Science, 1998, 281, 1647–1650 CrossRef CAS.
- W. E. Kaden, T. P. Wu, W. A. Kunkel and S. L. Anderson, Science, 2009, 326, 826–829 CrossRef CAS PubMed.
- Y. Watanabe, X. Wu, H. Hirata and N. Isomura, Catal. Sci. Technol., 2011, 1, 1490–1495 CAS.
| This journal is © the Owner Societies 2014 |