Microcavity-assisted microfluidic physical sensors: materials, structures, and multifunctional applications
Xinyi
Qu
ab,
Jianfeng
Ma
 ab,
Degong
Zeng
ab,
Jinan
Luo
ab,
Jingzhi
Wu
ab,
Chuting
Liu
ab,
Zhikang
Deng
ab,
Lvjie
Chen
ab,
Rongkuan
Han
ab,
Yancong
Qiao
ab,
Degong
Zeng
ab,
Jinan
Luo
ab,
Jingzhi
Wu
ab,
Chuting
Liu
ab,
Zhikang
Deng
ab,
Lvjie
Chen
ab,
Rongkuan
Han
ab,
Yancong
Qiao
 *ab and
Jianhua
Zhou
*ab and
Jianhua
Zhou
 *ab
*ab
aKey Laboratory of Sensing Technology and Biomedical Instruments of Guangdong Province, School of Biomedical Engineering, Shenzhen Campus of Sun Yat-sen University, Shenzhen, 518107, China. E-mail: qiaoyc3@mail.sysu.edu.cn; zhoujh33@mail.sysu.edu.cn
bKey Laboratory of Sensing Technology and Biomedical Instruments of Guangdong Province, School of Biomedical Engineering, Sun Yat-sen University, Guangzhou, 510275, China
First published on 7th January 2026
Abstract
Microfluidic sensing has long been dominated by chemical approaches that usually rely on fluorescent labels or specific reagents to achieve high specificity. However, these methods often require complex preparation and suffer from limited real-time capability, challenges that become more pronounced in wearable and portable platforms. In contrast, physical sensing offers a complementary route by detecting variations in mechanical, acoustic, optical, or thermal properties directly, enabling label-free, faster, and more robust operation. Under this background, microcavity architectures stand out as one promising option among various physical sensing designs. By spatially confining and enhancing physical signals at the miniature scale, microcavities can sharpen detection resolution and extend dynamic range. These gains are further elevated through the use of tailored materials and are reinforced by fabrication strategies that deliver precise geometry control and adaptable functionality. Harnessing such features, microcavity-based systems have been leveraged in fields ranging from high-resolution tactile sensing in soft robotics to wearable healthcare and human-machine interaction. This review surveys recent progress in materials, fabrication methods, and sensing mechanisms for microcavity-assisted microfluidic physical sensors, and discusses future directions toward broader adoption and scalable deployment.
Introduction
With the rapid advancement of micromachining technology and micro/nano-scale manufacturing processes, microfluidic technology has progressively emerged as an indispensable key technology in the biomedical field, owing to its remarkable advantages, such as high throughput,1 minimal sample consumption,2 rapid analysis,3 and precise control.4 Microfluidic chips are capable of integrating complex experimental procedures into miniaturized devices while substantially enhancing detection efficiency and accuracy,5 thereby demonstrating immense potential in disease diagnosis,6 drug screening,7,8 and biomolecular analysis.9,10In microfluidic sensing systems, chemical or biological sensing approaches represent the predominant sensing mechanisms, typically depending on fluorescent markers or specific reagents.11–13 By contrast, microfluidic sensing approaches based on physical principles have garnered increasing attention from researchers in recent years, due to their label-free nature, rapid response, and high stability, and have achieved substantial progress in wearable devices,14,15 flexible electronics,16 and biomedical engineering.17 Physical sensing techniques, such as mechanical,14 acoustic,18 optical,19 and thermal sensing,20 enable real-time, highly sensitive, and non-invasive detection by directly monitoring the physical properties or state changes of substances within fluids.
In microfluidic chips, the design of functional architectures determines the system's manipulation precision and signal transduction efficiency. Consequently, researchers have progressively developed various active or semi-active components to meet diverse application needs, such as flexible diaphragms for fluid regulation,21 magnetic/electric valves for flow path switching,22,23 and micropillar arrays to enhance the reaction interface.24 While these structures offer distinct advantages across specific application scenarios, in the trend of pursuing higher sensitivity, faster response times, and increased wearability, they struggle to simultaneously meet these demands. Under this backdrop, microcavity structures, capable of significantly enhancing signals at the micro and nanoscale, are increasingly attracting scholarly attention. A microcavity is a closed or semi-closed structure that can effectively confine and enhance optical, acoustic, and mechanical signals at the miniature scale.25,26 Owing to its spatial confinement characteristics,19 the microcavity enhances the detection system's responsiveness to weak signals and demonstrates unique advantages in improving the sensitivity, response speed, and detection limit of physical signal sensing. In particular, sealed airbag-based microcavity structures with active pressurization capabilities offer enhanced mechanical tunability and conformal skin-sensor interaction, enabling continuous and controllable signal output, which is highly advantageous for physiological signal acquisition and multimodal wearable sensing.27
The integration of microcavity structures with microfluidic sensing technology further extends the functionality and application scope of microfluidic chips. Currently, microcavity-assisted microfluidic sensing shows a diversified trend in both material architectures and functional implementation, encompassing lightweight, high specific-surface-area (SSA) cavities based on porous polymers28 or carbon materials,29 microchannels with self-healing liquid metal (LM) capable of dual-mode electrical and mechanical response,30,31 electret membrane cavities with prolonged charge retention,32 and closed/semi-closed polymeric cavities.33 These architectures not only improve sensitivity and dynamic range in mechanical sensing via active pressurization or passive resonance, but also facilitate efficient thermoacoustic and electroacoustic conversion in acoustic transduction, and enhance photothermal coupling efficiency in optical and thermal modalities. In addition, certain microcavities function as patterned encapsulation layers, or utilize microfluidic platforms as tools to assist in sensor assembly and scalable manufacturing.34 Consequently, they empower wearable healthcare systems with physiological signal monitoring;35,36 equip flexible robotic and human-machine interaction (HMI) end-effectors with multimodal tactile perception and touchless recognition;37,38 and enable ultra-wide-range sensing,39 acoustic communication,18 and optoelectronic multifunctional detection for environmental and industrial monitoring.40Fig. 1 provides an overview of the key materials and manufacturing processes of microcavity-assisted microfluidic physical sensors, along with their functional roles across diverse application domains. However, existing technologies face challenges such as high integration complexity, limited material options, insufficient long-term stability, and inadequate reusability. Consequently, systematically investigating the design principles, material selection, and performance optimization methods of microcavity-assisted microfluidic physical sensing holds significant scientific importance and practical application value.
 | ||
| Fig. 1 Schematic of the materials, fabrication, and diverse applications of microcavity-assisted microfluidic physical sensors. It covers porous polymer and carbon-based materials, liquid metal microchannel architectures, electret-based membranes, and sealed polymer cavities, illustrating their functional roles in augmenting mechanical, acoustic, optical, and thermal transduction, along with the corresponding fabrication methodologies, and presenting representative application domains, including wearable healthcare monitoring, HMI technologies, and environmental monitoring. Reproduced with permission from ref. 15. Copyright (2024) Elsevier, ref. 28. Copyright (2023) Wiley-VCH GmbH, ref. 29. Copyright (2025) AAAS, ref. 30. Copyright (2025) Wiley-VCH GmbH, ref. 32. Copyright (2024) Elsevier, ref. 35. Copyright (2024) Springer Nature, ref. 37. Copyright (2024) American Chemical Society, ref. 39. Copyright (2023) American Chemical Society, ref. 46. Copyright (2024) Wiley-VCH GmbH, ref. 49. Copyright (2024) Springer Nature, ref. 50. Copyright (2023) Springer Nature, ref. 51. Copyright (2024) American Chemical Society, ref. 58. Copyright (2024) American Chemical Society, ref. 96. Copyright (2023) American Chemical Society, and ref. 97. Copyright (2025) American Chemical Society. | ||
This review focuses on microcavity-assisted physical sensors from three perspectives: material selection, structural design, and application cases. It systematically discusses the critical role of microcavity structures in enhancing sensing performance and optimizing detection principles. Specifically, the application of microcavities in biomedical detection is emphasized comprehensively, aiming to provide valuable references and guidance for the design and development of microfluidic physical sensors. Therefore, the structure of the subsequent chapters is organized as follows: chapter 2 concentrates on the materials and fabrication principles of microcavity sensors, providing a systematic review of flexible polymers, LMs, carbon-based nanomaterials, and other functional material systems. It also analyzes how fabrication strategies, including laser processing, sacrificial templating, 3D printing, and laminated encapsulation, precisely define cavity geometries, enhance mechanical robustness, and facilitate scalable integration. Chapter 3 elucidates the device-level coupling between microcavity architectures and sensing mechanisms. It begins by analyzing how structural elements enable high gain, energy autonomy, and multimodal outputs within resistive, capacitive, and triboelectric modalities. Subsequently, it examines acoustic transduction, highlighting how different cavities enhance gain in thermoacoustic and electroacoustic conversion. The discussion then extends to optical and thermal domains. Chapter 4 summarizes current challenges in long-term stability, high-throughput fabrication, and intelligent data integration, and looks ahead to the development directions and application prospects of microcavity technology in wearable healthcare, HMI, environmental monitoring, and extreme condition sensing.
Materials and fabrication strategies
Microcavity structures enhance target physical signals by spatially confining acoustic, photonic, thermal, or mechanical fields. The design of flexible materials, as well as the integration of multifunctional material systems, governs the microcavity's dimensional precision, operational robustness, and functional versatility. Thus, materials engineering and fabrication methodologies remain fundamental to advancing research on microcavity-assisted physical sensing technologies. This section systematically reviews material classifications and representative fabrication techniques.Materials
 | ||
| Fig. 2 Polymer materials for constructing cavity walls and porous sensing layers. a. Exploded view and SEM image of the fully printed sensor array. TPU acts as a spacer layer, providing mechanical support to both the top and bottom surfaces of the sensor. PDMS encapsulation isolates the device from air exposure, thereby preventing oxidation of the electrodes and sensing layer.27 Reproduced with permission from ref. 27. Copyright (2025) Wiley-VCH GmbH. b. SEM image of PDMS foam fabricated using a sacrificial template method, and optical photographs of PDMS foam doped with varying concentrations of silver nanosheets.41 Reproduced with permission from ref. 41. Copyright (2025) Elsevier. c. Schematic illustration of the porous PDMS formation process and optical image of the porous sensing unit. CB absorbs heat from infrared laser irradiation, which initiates a cross-linking reaction in the PDMS precursor, resulting in solidification. Concurrently, laser-induced thermal decomposition of glucose hydrate generates steam, promoting the formation of internal pores within the PDMS matrix.44 Reproduced with permission from ref. 44. Copyright (2023) Wiley-VCH GmbH. d. Fabrication process of CNT/PU conductive sponge. SR releases hydrogen gas during the foaming process, leading to pore formation within the conductive sponge. By adjusting the SR ratio, CNT/PU sponges with varying pore sizes and densities can be obtained.42 A refers to the chemical blowing agent, and B refers to the curing agent, which are the two components of SR. Reproduced with permission from ref. 42. Copyright (2023) Elsevier. | ||
 | ||
| Fig. 3 LM sensing layers with diverse structural configurations. a. LM sensors with spiral, serpentine, and wave-shaped microchannels.31 Reproduced with permission from ref. 31. Copyright (2025) Wiley-VCH GmbH. b. A schematic diagram and optical image of a single embedded LM-ME sensing unit, together with a digital photograph of the LM-ME array in its pristine state.46 Reproduced with permission from ref. 46. Copyright (2024) Wiley-VCH GmbH. c. A photograph and side view of a breathable encapsulated LM foam-based soft stress sensor. The device consists of an integrated, delamination-free structure composed of PDMS foam in the top and bottom layers, and LM-coated PDMS foam in the middle layer.47 Reproduced with permission from ref. 47. Copyright (2023) Wiley-VCH GmbH. | ||
 | ||
| Fig. 4 Various sensors fabricated from carbon-based materials. a. Schematic and SEM images of three-dimensional graphene foam used for constructing thermoacoustic loudspeakers.51 Reproduced with permission from ref. 51. Copyright (2024) American Chemical Society. b. A photograph of a snail-shaped cavity loudspeaker worn on the human ear. The device is connected to the bottom of the cavity inlet to generate sound waves, and the acoustic signals are amplified and directed toward the ear.29 Reproduced with permission from ref. 29. Copyright (2025) AAAS. c. Photographs of a graphene aerogel sensor in ultra-stretchable condition and under 400% tensile strain.54 Reproduced with permission from ref. 54. Copyright (2023) Royal Society of Chemistry. d. An optical microscopy image of a strain-insensitive CNT–PDMS tactile sensing layer. Each sensing unit has a dome-shaped architecture.55 Reproduced with permission from ref. 55. Copyright (2025) Wiley-VCH GmbH. e. A cross-sectional schematic of a graphene/PMMA closed-cavity ultrasonic sensor. An air gap of 105 μm is formed by the suspended membrane and the silicon substrate.56 Reproduced with permission from ref. 56. Copyright (2023) MDPI. f. A schematic illustration of a tri-modal sensor unit for refractive index, temperature, and pressure sensing, based on an SNG coupled resonator. The graphene layer is deposited inside the dual-ring and inverted-T resonator structures.57 Reproduced with permission from ref. 57. Copyright (2024) IEEE. g. A schematic of a PETA/GO composite film optical microcavity sensor.58 Reproduced with permission from ref. 58. Copyright (2024) American Chemical Society. h. A schematic of a thin-film soft pressure sensor composed of a substrate, electrodes, and sensing layer, along with a diagram showing the micropore network formed within the sensing layer.59 Reproduced with permission from ref. 59. Copyright (2023) Wiley-VCH GmbH. i. A schematic of a dual-enzyme conductive microcavity, with black regions representing CNT buckypapers. A 3D exploded view illustrates the concept of microcavity.60 Reproduced with permission from ref. 60. Copyright (2024) MDPI. | ||
Fabrication techniques
Fabrication strategies for microcavity-enabled sensors are transitioning from traditional planar micromachining toward three-dimensional, customizable manufacturing paradigms. | ||
| Fig. 5 Fabrication techniques for microcavity-assisted physical sensors. a. Laser processing method. Reproduced with permission from ref. 44. Copyright (2023) Wiley-VCH GmbH. b. Sacrificial templating method. Reproduced with permission from ref. 41. Copyright (2025) Elsevier. c. 3D printing techniques. Reproduced with permission from ref. 29. Copyright (2025) AAAS. d. Laminated packaging and system integration. Reproduced with permission from ref. 27. Copyright (2025) Wiley-VCH GmbH. | ||
Microcavity-assisted physical sensors exhibit diversity in both materials and fabrication techniques. Researchers design sensors and microcavities based on application scenarios. By reasonably selecting and combining different materials, such as polymers, LMs, carbon-based materials, and other special materials, it is possible to achieve higher sensitivity, faster response, and improved stability in microcavity designs. Simultaneously, the synergy of advanced fabrication techniques enables precise control over cavity geometry, pore size distribution, and wall surface functionalization across micron to millimeter scales. These microstructures are further integrated with self-powered mechanisms, signal conditioning modules, and artificial intelligence (AI)-driven algorithms, collectively establishing a robust foundation for the development of next-generation, high-sensitivity, multi-parametric, and large-area manufacturable microfluidic physical sensors.
Sensing principle, structure, and application of microcavity-assisted sensors
Building upon these advanced processes and materials, researchers have developed various microfluidic sensors for physical signal detection. Practical applications reveal that different materials exhibit distinct compatibility preferences in sensor architecture and operational principles, due to their inherent differences in electrical, mechanical, and thermal properties. For instance, elastic polymers are commonly used to construct flexible resistive and capacitive sensors owing to their excellent compressibility and dielectric characteristics. LMs demonstrate high conductivity and flow reconfigurability, making them widely applicable in resistive microchannel structures. Ultra-thin graphene's superior electrical and thermal conductivity plays a pivotal role in acoustic resonance and thermoacoustic transduction sensing. To clarify the coupling pathways between material systems and sensor architectures, Table 1 systematically outlines commonly used material systems for microcavity-assisted physical sensors, summarizing the applicability and functional emphases of mainstream material systems in microcavity construction. These materials support diverse sensing mechanisms, including resistive, capacitive, triboelectric, acoustic, and optical/thermal sensing. Researchers typically need to select optimal structural configurations and sensing mechanisms based on practical application requirements.| Material categories | Specific materials | Features | Selected properties | Preferred sensing mechanism | Typical application scenarios | |
|---|---|---|---|---|---|---|
| Polymer | Elastomer | PDMS, Ecoflex | High elasticity, low young's modulus, large deformability | Young's modulus (E): 100–2000 kPa;68 relative permittivity (εR): 2.6–2.8 (ref. 69 and 70) | Resistive, capacitive, triboelectric, acoustic, and photothermal | Compressible microcavities, multimodal sensing, electronic skin, health monitoring, and wearable motion tracking |
| PU | With balanced strength and flexibility, good thermal stability | Young's modulus (E): 1–200 MPa;71 thermal degradation temperature: >300°C (ref. 72) | ||||
| Engineering plastic | PI | With good mechanical strength, thermal stability, and strong film-forming properties | Young's modulus (E): 5.89 GPa;73 relative permittivity (εR): 2.1–2.94;74 glass transition temperature (Tg): >350°C (ref. 75) | Capacitive (electret-based), resistive | Microcavity-assisted capacitive sensor substrate, flexible circuit support, laser-micromachined encapsulation | |
| PET | Young's modulus (E): 1.07 GPa;76 relative permittivity (εR): 3.1;77 glass transition temperature (Tg): 80°C (ref. 77) | |||||
| Hydrophilic polymer | PVA | Water-soluble, highly biocompatible | Young's modulus (E): 0.04–7 GPa (ref. 78) | Resistive | Flexible biosensing layer, capacitive/ion-conductive dielectric, skin/physiological signal acquisition | |
| Fluorinated polymer | PVDF | Chemically stable, hydrophobic, some with piezoelectric or charge-holding characteristics | Piezoelectric coefficient (d33): 13–28 pC/N, relative permittivity (εR): 12 (ref. 79) | TENG-based piezoelectric electret sensing | Non-contact touch sensing, motion/posture recognition, remote interaction, humidity monitoring | |
| PTFE/PFA composite film | Piezoelectric coefficient (d33): 23.8 pC/N (ref. 36) | |||||
| LM | EGaIn: gallium–indium alloy (75.5% Ga, 24.5% in) | Liquid at ambient conditions, excellent electrical conductivity, mechanical deformability, and strong compatibility with soft elastomers | Electrical conductivity (σ): 3.4 × 106 S m−1, melting point (Tm): 15.7 °C (ref. 80) | Resistive (microchannel strain-based) | 3D pressure mapping, strain sensing, bioelectrical monitoring, wearable e-skin | |
| Galinstan: gallium–indium–tin alloy (68% Ga, 21.5% in, 10.5% Sn) | Electrical conductivity (σ): 3.8 × 106 S m−1, melting point (Tm): −19 °C (ref. 80) | |||||
| Carbon-based material | Graphene | Excellent electrical and thermal conductivity, two-dimensional structure | Thermal conductivity (κ): 5300 W m−1 K−1, electron mobility (μ): 3.5×105 cm2 V−1 s−1, SSA: 2630 m2 g−1 (ref. 81) | Acoustic, thermoacoustic, resistive, triboelectric, optical | Thermoacoustic speaker, acoustic sensing, micro-pressure detection | |
| CNT | One-dimensional flexible conductor combining conductivity and mechanical flexibility | Thermal conductivity (κ): 3500 W m−1 K−1;82 electron mobility (μ): 2800–5400 cm2 V−1 s−1;83 SSA: 1315 m2 g−1 (ref. 84) | Resistive, triboelectric, thermal | Strain sensing, conductive film | ||
| Other functional materials | PZT | High piezoelectric response, strong electromechanical coupling, and consistent response | Piezoelectric coefficient (d33): 750 pC/N (ref. 85) | Triboelectric, capacitive, photothermal | Passive sensing, non-contact interaction, flexible HMI | |
| Hydrogel | Hydrophilic and flexible, with ionic conductivity and excellent biocompatibility | Water content: 70–90 wt%;86 ionic conductivity: 10−5–30 S cm−1 (ref. 87) | Capacitive sensing (flexible electrolyte-based), electrochemical sensing | Skin-interface sensing, physiological signal detection, flexible iontronic devices | ||
Mechanical sensing
Resistive sensing
Luo et al. integrate a PDMS-based spherical sealed microcavity with a laser-induced graphene (LIG) conductive layer to develop a high-performance flexible resistive sensor tailored for non-invasive, single-vessel blood pressure monitoring (Fig. 6a).35 The encapsulated air cavity functions as a mechanical buffer, significantly enhancing the electromechanical transduction efficiency while mitigating fatigue-induced degradation of the LIG conductive network under sustained or high-intensity loading. When coupled with a dual-stage convolutional neural network, the system achieves clinically relevant accuracy, with a diastolic pressure error of less than 0.5 mmHg and a systolic pressure error of less than 2.1 mmHg, in estimating blood pressure from a single vessel, thus offering a cost-effective and non-invasive approach to wearable hemodynamic monitoring.
 | ||
| Fig. 6 Closed airbag-assisted microcavity-based sensing systems. a. A flexible resistive pressure sensor composed of a spherical PDMS microcavity and a laser-induced graphene layer, enabling noninvasive single-vessel blood pressure monitoring.35 Reproduced with permission from ref. 35. Copyright (2024) Springer Nature. b. A fully printed flexible sensor array with integrated airbag microcavities for simultaneous pulse monitoring at three radial artery positions via multi-stage active pressurization.27 Reproduced with permission from ref. 27. Copyright (2025) Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. c. A multi-channel wearable sensing platform combining silicone airbags and ionic conductive films for synchronized pulse acquisition and real-time blood pressure estimation.33 Reproduced with permission from ref. 33. Copyright (2024) Springer Nature. d. A 3D pulse sensing array with multiple airbags and machine learning-assisted signal processing for atrial fibrillation classification.88 Reproduced with permission from ref. 88. Copyright (2025) American Chemical Society. | ||
Beyond blood pressure monitoring, actively pressurized sensing mechanisms have also been widely applied in pulse diagnostics. Drawing inspiration from traditional Chinese medicine (TCM) palpation techniques, Wang et al. develop a fully printed, flexible sensor array that integrates an airbag-based microcavity array for simultaneous acquisition of pulse signals at the “Cun”, “Guan”, and “Chi” positions (Fig. 6b).27 The microcavity structure supports multi-phase active pressurization, effectively emulating the dynamic “palpation” process to enhance the conformal interface between the skin and sensor surface, while modulating the sensing depth to capture pulse waveforms at varying vascular layers. The system exhibits consistent performance across diverse participants, body postures, and activity states, demonstrating the microcavity's engineering adaptability under complex pressure modulation conditions.
Based on the concept of pulse diagnosis, Zhao et al. introduce a multi-channel, actively pressurized flexible pulse monitoring platform, integrating silicone-based airbag cavities with flexible ion-conductive membrane arrays to enable synchronized pressure measurements at three distinct diagnostic points (Fig. 6c).33 Coupled with a neural network model based on nine static pressure segments and pulse wave amplitude, the platform provides real-time predictions of systolic and diastolic blood pressures.
In addition, Cao et al. expand the multi-airbag structure to a three-dimensional, dynamically controlled pulse array platform, incorporating a machine learning model to develop an atrial fibrillation classification system, thereby achieving the digitalization of traditional pulse diagnosis data (Fig. 6d).88 This platform integrates active pressurization, adaptive deformation, and AI-assisted diagnostic capabilities, marking a paradigm shift in microcavity-based health sensing systems from simulation-based sensing to cognitive diagnostic applications.
The sealed airbag microcavity structure modulates the sensor state through active deformation control, thereby enhancing both the sensitivity and reliability of flexible resistive sensors, while offering a systematic design pathway for the high-precision acquisition of complex physiological signals. The structure's tunability, coupled with its continuous and controllable signal response, positions it as one of the most prominent application paradigms in contemporary microcavity-assisted flexible microfluidic sensing technologies.
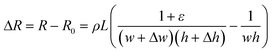 | (1) |
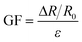 | (2) |
In the field of resistive sensing, utilizing LM-filled microchannels to fabricate high-sensitivity pressure sensors is a widely adopted technique. Wu et al. introduce an innovative structural design in which convex microarrays (CMs) are embedded within the LM channel (Fig. 7a).89 By integrating convex structures into the PDMS microchannel, the degree of deformation in the channel's cross-section during pressurization is effectively enhanced, leading to increased sensitivity to 4.3 × 10−2 kPa−1, while maintaining a measurement range of up to 340 kPa. The device has been successfully applied in pulse detection, finger bending, gait analysis, and gesture recognition scenarios.
 | ||
| Fig. 7 Microchannel-based resistive sensors with LM. a. A soft resistive sensor integrating CMs within a PDMS-LM microchannel to enhance cross-sectional deformation and improve sensitivity, applied in pulse monitoring, gait analysis, and gesture recognition.89 Reproduced with permission from ref. 89. Copyright (2023) IEEE. b. A dome-shaped microchannel sensor that differentiates multi-axis pressure distribution (X, Y, Z) and supports intelligent stress recognition through machine learning models.90 Reproduced with permission from ref. 90. Copyright (2024) Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. c. A Peano-type fractal microchannel structure that enhances stretchability and sensing density by optimizing the conductive path geometry of the LM.15 Reproduced with permission from ref. 15. Copyright (2023) Springer Nature. d. A pressure-sensitive structure based on the reversible contact behavior of LMDs, enabling dynamic resistance switching for applications such as physiological signal imaging and throat motion monitoring.30 Reproduced with permission from ref. 30. Copyright (2025) Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
Gul et al. develop a soft sensor using LM with a dome-shaped design, which, when combined with AI algorithms, leads to the development of a flexible sensor system capable of three-dimensional stress differentiation (Fig. 7b).90 The modular LM channel structure can resolve loading discrepancies in the X, Y, and Z directions. By incorporating machine learning models, this system achieves advanced posture and motion recognition, offering innovative perspectives for flexible robotics and multimodal HMI.
Subsequently, Luo et al. employ geometrically tunable structures to optimize the deformation response of microchannels and introduce Peano fractal microchannels (Fig. 7c).15 The Peano fractal not only increases the strain sensing density per unit area, but also through the optimization of structural geometry, delays the fracture point of the LM's conductive path, thus expanding the effective detection range. Their study highlights the importance of embedded structures in regulating conductive pathways within microchannels and demonstrates that engineering the geometry of microcavities can achieve a synergistic optimization of sensitivity, stretchability, and stability.
Building on this, Dong et al. introduce a novel pressure sensing structure based on the reversible contact behavior of liquid metal droplets (LMDs). By precisely controlling the microchannel geometry and electrolyte filling, they achieve controllable contact and disconnection of LMDs under varying pressures, leading to stable resistance changes and enabling “switch-like” dynamic resistance regulation (Fig. 7d).30 It has been successfully applied to complex physiological signal sensing scenarios, including two-dimensional image reconstruction and pharyngeal motion detection.
Recent research has introduced the use of micro-cage structures to achieve local regulation of microscopic strain, representing an innovative microcavity configuration. This structure has demonstrated significant advantages in enhancing the resolution and anti-crosstalk capabilities of array sensors, making it highly suitable for multi-pixel pressure sensing systems. Zhang et al. implement micro-cage strain-constrained structures in PDMS using a photo-crosslinking strategy.91 By employing ultraviolet-induced crosslinking inhibition, they introduce locally uncrosslinked regions in the PDMS, thereby forming microcavity units with localized strain characteristics that effectively constrain deformation diffusion within each pixel. The micro-cage structure reduces mechanical crosstalk between array elements by 90.3%, while maintaining an excellent sensitivity of 18.94 kPa−1. Additionally, it achieves a spatial resolution exceeding 4000 ppi and a minimum perceptible weight of 1 g (Fig. 8). This approach offers an effective structural solution for high-density and low-crosstalk flexible array sensors.
 | ||
| Fig. 8 Micro-cage structured stretchable pressure sensor array and its key performance metrics.91 A photo-crosslinking strategy is used to create localized uncrosslinked regions in PDMS, forming micro-cage units that confine strain within individual pixels, reduce crosstalk by 90.3%, and achieve a minimum detectable weight of 1 g. Reproduced with permission from ref. 91. Copyright (2023) Springer Nature. | ||
Capacitive sensing
Based on the aforementioned design principles, Wang et al. develop a dual-modal flexible electronic skin, utilizing digital light processing 3D printing to construct micro-spike electrode structures, and introducing a porous PDMS dielectric layer via a sacrificial template method to create microstructure–microcavity composite capacitive sensing units (Fig. 9a).28 The porous structure not only reduces the dielectric constant, but also enhances compressive deformation response, resulting in a high sensitivity of 1.672 kPa−1 within a pressure range of 0–20 kPa, with mass detection capability down to 1 g and non-contact recognition ability up to 10 cm. Under a similar structural framework, Wu et al. further propose a flexible sensing array integrating porous dielectric layers, micro-spike electrodes, and edge electric field sensing mechanisms (Fig. 9b). The porous sponge structure, made of PDMS with hundreds of micron-sized holes, is filled with ionic liquid and sandwiched between two layers of flexible conductive fabric, forming a highly sensitive variable capacitance structure. Non-contact recognition is enabled by the edge electric field interference mechanism, and dynamic gesture and 3D morphology sensing with >99% accuracy is achieved through machine learning.37
 | ||
| Fig. 9 Porous dielectric layer-based capacitive sensors. a. A dual-responsive electronic skin fabricated via DLP 3D printing, integrating micro-spike electrodes and a porous PDMS dielectric layer to form microstructure–microcavity composite capacitive units.28 Reproduced with permission from ref. 28. Copyright (2023) Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. b. A bimodal sensing array composed of ionic liquid-filled porous PDMS and flexible fabric electrodes, enabling edge electric field modulation for touchless and dynamic pressure sensing with >99% gesture recognition accuracy.37 Reproduced with permission from ref. 37. Copyright (2024) American Chemical Society. | ||
The micron-scale cavities within the porous dielectric layer are crucial in enhancing the compressive response of the device and its sensitivity to edge electric field disturbances. Additionally, they significantly reduce the overall mass, facilitating the maintenance of the device's flexibility and conformability. This microcavity structure, which integrates both performance and lightweight characteristics, serves as a fundamental structural basis for the development of multi-modal flexible sensors.
Liu et al. develop a self-powered flexible non-contact sensor (Fig. 10a), where a fluorinated ethylene propylene (FEP) film and PET substrate composite form a cavity structure. By utilizing corona charging, stable potential and capacitive responses are generated, enabling precise sensing of distances as small as 2 mm. The sensor successfully detects eyelid movements and exhibits a certain degree of resolution for various gestures and spatial positions, showing considerable application potential in the fields of soft robotics and wearable sensing technologies.92
 | ||
| Fig. 10 Electret-based capacitive sensors with non-contact sensing capability. a. A self-powered soft sensor combining FEP film and PET substrate via corona charging to form an electret cavity, enabling sub-millimeter distance resolution and gesture recognition for non-contact positioning.92 Reproduced with permission from ref. 92. Copyright (2024) American Chemical Society. b. A flexible electret sensor platform using a multilayer microcavity structure and variable-spacing electrode array to achieve ultra-wide sensing range (2–180 mm) and 3D posture recognition.39 Reproduced with permission from ref. 39. Copyright (2023) American Chemical Society. c. An electret actuator integrating optical ranging and acoustic feedback, based on an FEP–air–PET sandwich microcavity that supports multimodal long-range HMI with scanning and voice response functions.32 Reproduced with permission from ref. 32. Copyright (2024) Elsevier. | ||
Dai et al. introduce a flexible electret sensing platform integrated with a variable-spacing electrode array. By utilizing a microcavity structure to adjust the equivalent capacitance response curve with respect to distance, they achieve an ultra-wide sensing range (2–180 mm) and multi-angle 3D recognition capabilities (Fig. 10b).39 The device enables non-contact multi-point tracking, object posture recognition, and remote sliding gesture interaction through the coupling of electric field disturbances and structural microcavity variations.
In an extended approach, Xu et al. integrate electret microcavity structures with optical ranging and acoustic feedback functionalities, constructing an intelligent scanner based on an FEP–air–PET sandwich structure. This system enables 3D spatial depth perception and real-time voice feedback within a self-driven HMI system (Fig. 10c).32 The device, without any external power input, utilizes structural vibrations to drive a sound wave emitter and adjust the laser scanning angle, allowing for long-range positioning and navigation up to 250 cm. This serves as a representative example of the application of electret microcavity structures in multimodal interaction.
In a non-contact sensing, the electret structure generates an electric field disturbance, whereas in a contact scenario, it induces a charge coupling response. As a result, when applied to contact-based mechanical sensing environments, the signal output transitions to a triboelectric response.
Triboelectric sensing
Triboelectric sensing relies on the surface charge transfer during the contact-separation process of materials, enabling passive, highly sensitive flexible pressure sensing. When integrated with electret or porous microcavity structures, it enhances the device's output stability, response linearity, and spatial adaptability.Wu et al. develop a flexible, self-powered electret pressure sensor array based on a multilayer structure of e-PTFE, PFA, and FEP, with the core being a composite film featuring a microcavity structure (Fig. 11a).36 The microcavity structure significantly reduces the Young's modulus of the film layer while enhancing sensing performance, achieving a sensitivity of 9 mV N−1 over a range of 1.4–13.6 N. The sensor demonstrates excellent performance in real-time monitoring across various human body scenarios (e.g., sound, pulse, and finger joint activities) and during pig knee joint replacement surgery.
 | ||
| Fig. 11 Triboelectric microcavity-based self-powered sensors. a. A flexible sensor array with an e-PTFE/PFA/FEP multilayer microcavity structure, reducing film stiffness and enhancing pressure sensing performance, applicable in physiological monitoring and surgical scenarios.36 Reproduced with permission from ref. 36. Copyright (2024) Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. b. Fiber filter based on TENG with an annular microcavity structure, enabling airflow-driven electrostatic particle capture for sustainable personal protection.93 Reproduced with permission from ref. 93. Copyright (2025) Elsevier. c. A porous PDMS-based triboelectric sensor with a water-containing gradient microchannel structure, where liquid migration and “water bridge” formation enhance linearity and support 3D tactile and bending sensing.94 Reproduced with permission from ref. 94. Copyright (2024) Springer Nature. | ||
Zhou et al. develop a respiration-driven self-powered PVDF/cellulose acetate (CA) nanofiber filter material, incorporating a porous fiber network created by electrospinning and an annular cavity composed of copper foil, electrode sheets, and elastic supports (Fig. 11b).93 This microcavity structure aids in maintaining a stable airflow channel and electric field gradient, thereby enhancing the electrostatic adhesion capacity generated by the triboelectric effect. It exhibits sustained and efficient capture of submicron particles, demonstrating the role of microcavity structures in self-powered filtration systems.
Porous PDMS materials are frequently utilized in the construction of triboelectric microfluidic sensors. Qin et al. design a sensor based on a water-containing elastomer structure, employing gradient-distributed multi-stage microchannels to create a sandwich-like porous microcavity network (Fig. 11c).94 The microcavity provides both liquid storage and flow paths, and when the sensor nears its compression limit, a “water bridge” forms through liquid extrusion, dynamically adjusting the internal electric field, achieving a wide linear detection range from 5 to 1240 kPa. The porous microcavity improves deformation adaptability and charge migration efficiency, enabling the device to perform three-dimensional tactile recognition and bending angle sensing, thereby expanding the application scope of triboelectric sensing.
The integration of electret and triboelectric microcavity structures highlights a convergent pathway toward self-powered, highly sensitive, and spatially adaptive sensing systems. Through the coupling of permanent charge retention, dynamic air gap modulation, and porous dielectric design, these approaches enable multimodal signal conversion across both contact and non-contact scenarios. Collectively, they demonstrate the synergistic advantages of microcavity-enhanced self-powered sensing technologies in the development of next-generation human–machine interfaces, intelligent wearables, and low-power biomedical diagnostics.
Acoustic sensing
Microcavity architectures play a pivotal role in acoustic energy harvesting and transduction. They effectively capture acoustic energy, augment sound pressure, and adjust the frequency response, thereby efficiently converting weak acoustic vibrations into readily measurable and processable electrical or optical outputs, thereby constituting an effective pathway to enhancing the sensitivity of acoustic sensing.Chen et al. propose an acoustic sensor (Fig. 12a) based on GO/DNA aerogel.52 This sensor capitalizes on the porous microcavity structure to detects faint acoustic vibrations with high sensitivity and converts them into measurable resistance changes. It delivers an effective response bandwidth of 300–4000 Hz and a sensitivity of 74.4 kPa−1, exhibiting highly synchronized responses to animal sounds, human voices, and environmental noise. Zhong et al. propose a highly sensitive acoustic sensor (Fig. 12b) based on PI/GO foam.95 This sensor converts the tiny vibration energy, produced by the repeated reflection of sound waves within porous microcavity, into electrical signals, enabling the identification of both biological and non-biological sounds via distinct peaks. Liu et al. propose a highly sensitive graphene-based Fabry–Perot acoustic sensor (Fig. 12c).96 By reducing the pressure change within the enlarged backing air cavity (EBC), the equivalent acoustic pressure acting on the diaphragm was increased, achieving an exceptionally high mechanical sensitivity of 187.32 nm Pa−1 @ 16 kHz. Active sensors require both a sound source and a sensor. Therefore, in terms of the sound source, Ren et al. report a wearable acoustic device based on graphene thermoacoustic resonance and a 3D-printed conch-inspired spiral cavity (Fig. 12d).29 Tuning the cavity height affords control over the operating resonant frequency, yielding pronounced resonance enhancement at 5.4 kHz, with the sound pressure level (SPL) rising from 32 dB to 71 dB.
 | ||
| Fig. 12 Acoustic sensing systems and device architectures. a. Acoustic sensor based on GO/DNA aerogel.52 Reproduced with permission from ref. 52. Copyright (2025) Royal Society of Chemistry. b. Highly sensitive acoustic sensor based on PI/GO foam.95 Reproduced with permission from ref. 95. Copyright (2025) American Chemical Society. c. Graphene-based F–P acoustic sensor using the EBC.96 Reproduced with permission from ref. 96. Copyright (2023) American Chemical Society. d. Wearable acoustic device based on graphene thermoacoustic resonator.29 Reproduced with permission from ref. 29. Copyright (2025) AAAS. | ||
Other sensing
In addition to the aforementioned mainstream systems, microcavity-assisted sensing technologies based on optical and thermal principles also play a vital role in physical sensing within microfluidic systems. Fan et al. demonstrate an ultraviolet (UV)–near-infrared (NIR) dual-narrowband photodetector applicable to solar UV intensity monitoring and PPG signal detection (Fig. 13a).97 By incorporating a microcavity structure, the device leverages optical interference effects to enhance its optical response at the resonant wavelength, allowing selective transmission of 330 nm UV light (transmittance >80%) while suppressing light in other spectral ranges. Zhu et al. report an organic PPG sensor that integrates a microcavity organic light-emitting diode (OLED) and an annular organic photodetector (OPD) (Fig. 13b).63 By tuning the cavity length between the semi-transparent and reflective metal electrodes, the angular emission profile of the OLED is controlled, enhancing arterial light absorption, reducing baseline drift, and optimizing the pulsating signal. Movaghgharnezhad et al. design a highly sensitive broadband visible-light detection system based on a 3D heterogeneous multiscale porous graphene structure (Fig. 13c).98 The interconnected multilevel porous microcavity improves photoresponsivity to 4.4 mA W−1, representing a three-order-of-magnitude enhancement over monolayer graphene photodetectors. Li et al. propose a multifunctional phase-change aerogel for wearable thermal management (Fig. 13d).99 Leveraging a cross-linked continuous skeleton and preserved porous cavity structure, the material enhances heat storage capacity while maintaining the intrinsic properties of aerogels, such as thermal insulation, adsorption, and mechanical flexibility. The resulting thermal conductivity reaches 29.6 mW m−1 K−1. | ||
| Fig. 13 Optical and thermal sensors. a. UV–NIR dual narrowband detector.97 Reproduced with permission from ref. 97. Copyright (2025) American Chemical Society. b. An organic PPG sensor integrating a microcavity OLED and an annular OPD.63 Reproduced with permission from ref. 63. Copyright (2024) Wiley-VCH GmbH. c. A high-sensitivity photodetector based on three-dimensional heterogeneous multiscale porous graphene.98 Reproduced with permission from ref. 98. Copyright (2023) Elsevier. d. A multifunctional phase-change aerogel featuring both aerogel properties and thermal regulation performance.99 Reproduced with permission from ref. 99. Copyright (2024) Wiley-VCH GmbH. | ||
Summary and Prospect
Microcavity-assisted microfluidic physical sensors expand the capabilities of flexible sensors in precise measurement and multimodal signal processing through sophisticated cavity design, rational combinations of various flexible materials, and fabrication techniques suitable for mass production. This work provides a comprehensive analysis of microcavity architectures in microfluidic physical sensors, focusing on material optimization, representative structural typologies, and mechanisms for multimodal sensing. Special attention is given to their role in enhancing performance across resistive, capacitive, and triboelectric channels, emphasizing the pivotal contributions of microcavities to signal amplification, strain modulation, and multimodal signal integration. By integrating functional materials, such as polymers, porous materials, LMs, and carbon-based nanomaterials, microcavity structures substantially improve the sensitivity and temporal responsiveness of sensors, while simultaneously conferring superior mechanical flexibility, environmental adaptability, and system-level integrability. These attributes position microcavity-enabled devices as promising candidates for applications in health diagnostics, interactive systems, and environmental monitoring.In contrast to conventional sensing architectures, microfluidic platforms exhibit distinct advantages in the context of physical signal detection. On one hand, the enclosed and controllable channels and cavity structures provide a stable three-dimensional support environment for microcavity units, helping to confine deformation regions and improve response repeatability. On the other hand, the design flexibility of microfluidic channels and their ability to manipulate fluid media make them naturally compatible with materials, such as LMs100 and porous gels,101 thereby enhancing the sensor's dynamic response and adaptability to complex environments. Microfluidic systems inherently possess the capacity to precisely modulate and transmit a wide range of physical stimuli, including pressure,102 acoustic signals,103 temperature,104 and fluidic cues,105 thereby establishing a foundational infrastructure for integrated multiparametric sensing.
Although significant advancements have been achieved, microcavity-enabled microfluidic sensing platforms continue to encounter challenges. The stability and long-term reliability of microcavity structures still depend on further optimization of material systems and packaging processes. Moreover, scalable and standardized high-throughput fabrication techniques for large-area production remain underdeveloped, thereby constraining the widespread implementation of such sensors in industrial and clinical applications. In addition, the convergence of multimodal sensing and AI algorithms remains predominantly at the experimental validation phase, with few cases of mature, systematic deployment.
Future research may advance along three principal directions. At the materials level, research focuses on novel composite systems that exhibit stretchability, autonomous self-healing, self-powering capabilities, and responsiveness to multiple physical stimuli. Efforts also aim to enhance their environmental tolerance under extreme conditions, while incorporating biodegradable and biocompatible materials to address the specific requirements of wearable and implantable medical devices. From a manufacturing standpoint, integrating techniques, such as laser micromachining, 3D printing, and soft lithography with automated encapsulation enables high-throughput, standardized, and large-area production, while the processing precision still requires further improvement. Furthermore, the development of multi-material co-fabrication and programmable structural manufacturing strategies addresses the growing demand for personalized and adaptive sensing solutions. Additionally, integrating physics-based model with machine learning enables the parametric design and predictive performance evaluation of microcavity architectures, thereby accelerating device design workflows. Regarding application and system integration, advancing the deep convergence of microcavity sensing with AI algorithms and low-power wireless communication is essential. Custom optimization should be performed for diverse application scenarios, including healthcare, HMIs, environmental monitoring, and industrial diagnostics, while pursuing developments toward high-density sensor arrays, minimized inter-channel crosstalk, and enhanced spatial resolution. Such progress ultimately facilitates the realization of multimodal, intelligent sensing architectures. Meanwhile, integrating physicochemical approaches is emphasized, wherein the inherent advantages of chemical sensing are harnessed in synergy with physical modalities to enable multimodal and multi-throughput data acquisition, thereby supplying richer datasets to support algorithmic training and inference. For instance, hydrogels not only enable multimodal chemical sensing,106 but are also commonly used in physical sensing applications.38 By harnessing the material's capacity to adapt to diverse sensing mechanisms, it becomes possible to design multifunctional sensing systems. Similarly, by augmenting organ-on-a-chip platforms with physical sensing capabilities to monitor internal parameters, researchers can provide more effective guidance for experimental design and optimization.
Microcavity-assisted sensors possess a solid foundation for industrialization and strong potential for real-world deployment. In the field of wearable healthcare, the integration of enclosed microcavity structures and multimodal sensing algorithms enables noninvasive health assessments such as continuous blood pressure and pulse monitoring, applicable to smart health patches, postoperative rehabilitation monitoring, and chronic disease management systems.107,108 In HMI and intelligent interfaces, structures involving porous electrets and LM microchannels are widely applicable in non-contact operating systems, AI-based interactive platforms, and tactile sensors for soft robotic end-effectors.109–114 The incorporation of AI accelerates the advancement of microcavity sensor technologies. By using machine learning and computational modeling, researchers systematically explore material combinations and cavity designs with enhanced efficiency, thus reducing the research cycle; simultaneously, intelligent algorithms at the application-level function to integrate multimodal signals and discern complex patterns, which in turn enhances support for health monitoring and HMI. As demand grows for precise monitoring of controlled microenvironments, these systems can be further extended to applications in environmental sensing and microscale fluidic regulation. In extreme condition sensing, advanced materials including graphene aerogels and microstructured conductive foams enable precise detection in challenging settings involving high or low temperatures and high pressures.115 Combined with scalable fabrication techniques such as soft lithography and 3D printing, it is expected to establish low-cost, mass-producible industrial platforms to support the rapid development of flexible electronics and digital healthcare.116
As research progresses, the focus of microcavity-assisted microfluidic physical sensors shifts from single performance optimization to more complex system integration and engineering applications. In the future, with the continuous progress of materials, fabrication processes, and intelligent algorithms, microcavity-assisted microfluidic sensing technology is expected to continue playing a role in intelligent perception systems and become a key technological enabler in fields of biomedicine, flexible electronics, and novel HMI.
Author contributions
Jianhua Zhou contributed to the conceptualization and funding acquisition for the project. He played a crucial role in project administration and supervision, and contributed to the review and editing of the manuscript. Yancong Qiao was responsible for the conceptualization of the study, overseeing the project administration and supervision. He also contributed significantly to writing and reviewing the manuscript. Xinyi Qu led the investigation process and was primarily responsible for writing the original draft. She contributed to the visualization of the study, and participated in the conceptualization of the study. Jianfeng Ma was involved in the investigation phase of the study and made contributions to the visualization aspects, including data representation and graphical elements. Degong Zeng contributed to the investigation phase, providing valuable input in the research process. He also helped with the review and editing of the manuscript. Jinan Luo was involved in the investigation and participated in the review and editing process of the manuscript. Jingzhi Wu, Chuting Liu, Zhikang Deng, Lvjie Chen, and Rongkuan Han: these authors contributed to the review and editing of the manuscript, providing insightful feedback and revisions to improve clarity, structure, and consistency, ensuring the final version met academic standards.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research data, software, or code are generated or analyzed in this study. All data supporting this review are from previously published sources, which are cited within the article.Acknowledgements
This work is supported by National Natural Science Foundation of China (No. 62201624, 32000939, 21775168, 22174167, 51861145202, U20A20168, 12302119), Shenzhen Medical Research Fund (No. A2403041), the Guangdong Basic and Applied Basic Research Foundation (No. 2024A1515012056; 2019A1515111183), Shenzhen Science and Technology Program (No. RCBS20221008093310024, JCYJ20230807111120043, RCBS20210706092407002; JCYJ20220818102014028), the Research Funds from Sun Yat-sen University (No. 2024_76200_B25890), the Fundamental Research Funds for the Central Universities, Sun Yat-sen University (No. 24xkjc034). The authors are also thankful for the support from Key Laboratory of Sensing Technology and Biomedical Instruments of Guangdong Province (No. 2020B1212060077).References
- S. Li, Y. Zhang, J. Liu, X. Wang, C. Qian, J. Wang, L. Wu, C. Dai, H. Yuan, C. Wan, J. Li, W. Du, X. Feng, Y. Li, P. Chen and B. Liu, Small, 2024, 20, 2401848 Search PubMed.
- S. R. Little, Z. Leung, A. B. V. Quach, A. Hirukawa, F. Gholizadeh, M. Hajiaghayi, P. J. Darlington and S. C. C. Shih, Adv. Mater. Technol., 2023, 8, 2300719 CrossRef CAS.
- Y. Xie, L. Xu, J. Zhang, C. Zhang, Y. Hu, Z. Zhang, G. Chen, S. Qi, X. Xu, J. Wang, W. Ren, J. Lin and A. Wu, Mater. Horiz., 2024, 11, 5752–5767 Search PubMed.
- J. S. Y. Low, H. F. Teh, T. M. Thevarajah, S. W. Chang and S. M. Khor, Biosens. Bioelectron., 2025, 270, 116949 CrossRef CAS PubMed.
- W. Qiu and S. Nagl, ACS Sens., 2021, 6, 1147–1156 CrossRef CAS PubMed.
- Z. Zhu, S. Li, D. Wu, H. Ren, C. Ni, C. Wang, N. Xiang and Z. Ni, Lab Chip, 2022, 22, 2097–2106 RSC.
- A. Bhusal, E. Dogan, H.-A. Nguyen, O. Labutina, D. Nieto, A. Khademhosseini and A. K. Miri, Biofabrication, 2022, 14, 014103 Search PubMed.
- Z. Yang, Z. Zhou, T. Si, Z. Zhou, L. Zhou, Y. R. Chin, L. Zhang, X. Guan and M. Yang, Small, 2023, 19, 2207194 CrossRef CAS PubMed.
- X. Xiang, F. Li, Q. Ye, Y. Shang, M. Chen, J. Zhang, B. Zhou, H. Suo, Y. Ding and Q. Wu, Sens. Actuators, B, 2022, 358, 131517 Search PubMed.
- X. Liu, Y. Wang, Y. Du, J. Zhang, Y. Wang, Y. Xue, J. Zhao, L. Ge, L. Yang and F. Li, Chem. Eng. J., 2024, 486, 150233 CrossRef CAS.
- X. Tong, X. Lin, N. Duan, Z. Lv, Z. Wang and S. Wu, Sens. Actuators, B, 2023, 397, 134691 CrossRef CAS.
- Z. He, J. Huang, W. Shen, X. Lei, Y. Zhang, L. Zhu, X. Shen, D. Zhang, D. Yu and M. Zhou, ACS Appl. Mater. Interfaces, 2023, 15, 24913–24922 CrossRef PubMed.
- X. Zhao, Y. He, S. Shao, Q. Ci, L. Chen, X. Lu, Q. Liu and J. Chen, ACS Sens., 2024, 9, 2413–2420 Search PubMed.
- S. Pan, T. Zhang, C. Zhang, N. Liao, M. Zhang and T. Zhao, Lab Chip, 2024, 24, 1668–1675 Search PubMed.
- Y. Luo, H. Fan, X. Lai, Z. Zeng, X. Lan, P. Lin, L. Tang, W. Wang, Y. Chen and Y. Tang, Biosens. Bioelectron., 2024, 246, 115905 Search PubMed.
- C. Zhang, J. Chen, C. Xu, T. He, X. Zhang, J. Zhang, X. Sun, B. Xu, Y. Zhu and H. Yang, Nat. Commun., 2025, 16, 1404 Search PubMed.
- M. Yuan, Z. Liu, X. Wu, H. Gou, Y. Zhang, X. Ning, W. Li, Z. Yao, Y. Wang, W. Pei and H. Chen, Sens. Actuators, A, 2023, 354, 114250 Search PubMed.
- J. Li, L. Bo, T. Li, P. Zhao, Y. Du, B. Cai, L. Shen, W. Sun, W. Zhou and Z. Tian, Adv. Mater. Technol., 2024, 9, 2400572 Search PubMed.
- Y. Zheng, J. Sun, Y. Ma, H. Zhang, Z. Cui, G. G. Paschos, X. Song, Y. Tao, P. Savvidis, W. Kong, L. Wen, S. Bian and M. Sawan, Adv. Sci., 2025, 12, 2410125 Search PubMed.
- M. Wagih, J. Shi, M. Li, A. Komolafe, T. Whittaker, J. Schneider, S. Kumar, W. Whittow and S. Beeby, Nat. Commun., 2024, 15, 452 CrossRef PubMed.
- R. You, Q. Fan, Z. Wang, W. Xing, Y. Wang, Y. Song, X. Duan, R. You and Y. Wang, Research, 2024, 7, 0314 CrossRef PubMed.
- A. M. Moran, V. T. Vo, K. J. McDonald, P. Sultania, E. Langenbrunner, J. H. V. Chong, A. Naik, L. Kinnicutt, J. Li and T. Ranzani, Commun. Eng., 2024, 3, 117 CrossRef PubMed.
- X. Li, T. Chen, Z. Zheng, J. Gao, Y. Wu, X. Wu, T. Jiang, Z. Zhu and R. X. Xu, Small, 2024, 20, 2404952 CrossRef.
- P. Lemke, L. Schneider, W. Kunz, A. L. Rieck, P. S. Jäger, A. Bruckmann, B. Nestler, K. S. Rabe and C. M. Niemeyer, Adv. Funct. Mater., 2024, 34, 2313944 CrossRef.
- K. J. Vahala, Nature, 2003, 424, 839–846 Search PubMed.
- M. K. Zalalutdinov, J. T. Robinson, J. J. Fonseca, S. W. LaGasse, T. Pandey, L. R. Lindsay, T. L. Reinecke, D. M. Photiadis, J. C. Culbertson, C. D. Cress and B. H. Houston, Nat. Commun., 2021, 12, 3267 Search PubMed.
- X. Wang, G. Wu, X. Zhang, F. Lv, Z. Yang, X. Nan, Z. Zhang, C. Xue, H. Cheng and L. Gao, Adv. Mater., 2025, 37, 2410312 CrossRef PubMed.
- H. L. Wang, T. Chen, B. Zhang, G. Wang, X. Yang, K. Wu and Y. Wang, Small, 2023, 19, 2206830 CrossRef PubMed.
- Y.-H. Wei, Z.-F. Guo, Y.-F. Wang, T. Lin, W.-W. Hou, S.-W. Duan, L.-Q. Tao, H. Tian, Y. Yang and T.-L. Ren, Sci. Adv., 2025, 11, eadv2801 CrossRef PubMed.
- S. Dong, G. Ma, Z. Xiong, D. Ge, Y. Guo, W. Li and S. Zhang, Adv. Intell. Syst., 2025, 7, 2401019 CrossRef.
- Z. Zhao, X. Zou, J. Zhang and K. W. C. Lai, Adv. Electron. Mater., 2025, 11, 2500124 CrossRef.
- S. Xu, X. Guan, K. Bian, Q. Zhu, N. Dai, X. Zhao, Y. Qiu, S. Zheng, Y. Dong, J. Zhong, Q. Zhong and T. Hu, Nano Energy, 2024, 125, 109553 CrossRef.
- Y. Zhao, Q. Sun, S. Mei, L. Gao, X. Zhang, Z. Yang, X. Nan, H. Zhang, C. Xue and J. Li, Microsyst. Nanoeng., 2024, 10, 77 CrossRef PubMed.
- M. S. Rahman, A. Shon, R. Joseph, A. Pavlov, A. Stefanov, M. Namkoong, H. Guo, D. Bui, R. Master, A. Sharma, J. Lee, M. Rivas, A. Elati, Y. Jones-Hall, F. Zhao, H. Park, M. A. Hook and L. Tian, Sci. Adv., 2025, 11, eads4415 CrossRef PubMed.
- J. Luo, J. Wu, X. Zheng, H. Xiong, L. Lin, C. Liu, H. Liu, H. Tang, H. Liu, F. Han, Z. Liu, Z. Deng, C. Liu, T. Cui, B. Li, T.-L. Ren, J. Zhou and Y. Qiao, Nano Res., 2024, 17, 10058–10068 Search PubMed.
- L. Wu, J. Xue, J. Meng, B. Shi, W. Sun, E. Wang, M. Dong, X. Zheng, Y. Wu, Y. Li and Z. Li, Adv. Funct. Mater., 2024, 34, 2316712 Search PubMed.
- Q. Wu, C. Zhou, Y. Xu, S. Han, A. Chen, J. Zhang, Y. Chen, X. Yang, J. Huang and L. Guan, ACS Sens., 2024, 9, 2091–2100 Search PubMed.
- H. Wang, Q. Ding, Y. Luo, Z. Wu, J. Yu, H. Chen, Y. Zhou, H. Zhang, K. Tao, X. Chen, J. Fu and J. Wu, Adv. Mater., 2024, 36, 2309868 Search PubMed.
- N. Dai, X. Guan, C. Lu, K. Zhang, S. Xu, I. M. Lei, G. Li, Q. Zhong, P. Fang and J. Zhong, ACS Nano, 2023, 17, 24814–24825 Search PubMed.
- H. Yang, X. Cao, Z.-G. Hu, Y. Gao, Y. Lei, M. Wang, Z. Zuo, X. Xu and B.-B. Li, Photonics Res., 2023, 11, 1139–1147 CrossRef.
- G. Mogli, M. Costantini and S. Stassi, Sens. Actuators, A, 2025, 392, 116734 Search PubMed.
- J. Liao, X. Dai, J. Han, J. Yang, Y. Wu, Y. Cao, Y. Qiu, Y. Wang, L.-B. Huang, H. Ni and W. Feng, Nano Energy, 2024, 121, 109252 CrossRef.
- J. Zhang, S. Wei, C. Liu, C. Shang, Z. He, Y. Duan and Z. Peng, Microsyst. Nanoeng., 2024, 10, 19 CrossRef PubMed.
- D. Guo, X. Lei, H. Chen, L. Yi, Y. Li, Y. Zhao, F. Liu and G. J. Cheng, Adv. Sens. Res., 2024, 3, 2300165 CrossRef.
- L. Liu, J. Li, Z. Tian, X. Hu, H. Wu, X. Chen, L. Zhang and W. Ou-Yang, Nano Energy, 2024, 128, 109817 CrossRef.
- H. Li, C. Zhang, H. Xu, Q. Yang, Z. Luo, C. Li, L. Kai, Y. Meng, J. Zhang, J. Liang and F. Chen, Adv. Sci., 2025, 12, 2413233 Search PubMed.
- J. Xu, Z. Wang, X. Wang, Y. Wu, R. Xing, T. Yu, Y. Li, J. Ao, Y. Tao, B. Bai, M. D. Dickey, D. Zhang and J. Yang, Adv. Mater. Technol., 2023, 8, 2201193 CrossRef.
- L. Qin, W. Xia, Y. Wu, X. Huang, A. Nsilani Kouediatouka, H. Li, M. Ding, F. J. Mawignon, S. Lu, K. Cao, P. Yang, Y. Zhang and G. Dong, ACS Appl. Mater. Interfaces, 2024, 16, 58980–58990 CrossRef PubMed.
- Y. Li, B. Liu, P. Xu, J. Liu, X. Dai, A. Yu, T. Wang, L. Guo, T. Guan, L. Song and M. Xu, Nano Res., 2024, 17, 10008–10016 CrossRef.
- Y. Qiu, Z. Zou, Z. Zou, N. K. Setiawan, K. V. Dikshit, G. Whiting, F. Yang, W. Zhang, J. Lu, B. Zhong, H. Wu and J. Xiao, npj Flexible Electron., 2023, 7, 37 CrossRef.
- W. Hou, Y. Wei, Y. Wang, S. Duan, Z. Guo, H. Tian, Y. Yang and T.-L. Ren, ACS Appl. Mater. Interfaces, 2024, 16, 23544–23552 Search PubMed.
- S. Chen, P. Zhang, J. Zhao, K. S. Novoselov and D. V. Andreeva, Nanoscale Horiz., 2025, 10, 1405–1413 Search PubMed.
- Y. Dai, Y. Li, S. Xuan, Y. Dai, T. Xu and H. Yu, ACS Appl. Mater. Interfaces, 2025, 17, 11117–11125 CrossRef PubMed.
- G. Yang, X. Zhang, R. Wang, X. Liu, J. Zhang, L. Zong and H. Yang, Mater. Horiz., 2023, 10, 1865–1874 RSC.
- K. Bae, M. Kim, S. Sim, Y. Kang and J. Kim, Small Methods, 2025, 9, 2401730 CrossRef PubMed.
- J. Xu, G. S. Wood, E. Mastropaolo, P. Lomax, M. Newton and R. Cheung, Micromachines, 2023, 14, 810 CrossRef.
- Z. Zhu and X. Wang, IEEE Sens. J., 2024, 24, 8712–8720 Search PubMed.
- S. Zhang, B. Dong, Z. Wang, Z. Chen, T. Wei, J. Zheng and P. Lin, ACS Appl. Electron. Mater., 2024, 6, 7439–7447 CrossRef.
- K. Tou, K. Nara, T. Sekine, Y. Wang, Y. Takeda, D. Kumaki and S. Tokito, Adv. Sens. Res., 2023, 2, 2300045 CrossRef.
- I. Jeerapan, Y. Nedellec and S. Cosnier, Nanomaterials, 2024, 14, 545 CrossRef PubMed.
- J. Li, H. Jia, J. Zhou, X. Huang, L. Xu, S. Jia, Z. Gao, K. Yao, D. Li, B. Zhang, Y. Liu, Y. Huang, Y. Hu, G. Zhao, Z. Xu, J. Li, C. K. Yiu, Y. Gao, M. Wu, Y. Jiao, Q. Zhang, X. Tai, R. H. Chan, Y. Zhang, X. Ma and X. Yu, Nat. Commun., 2023, 14, 5009 CrossRef PubMed.
- Z. Yang, J. Wang, X. Wan, H. Xu, C. Zhang, X. Lu, W. Jing, C. Guo and X. Wei, Microsyst. Nanoeng., 2024, 10, 177 CrossRef PubMed.
- H. Zhu, H. Yu, J. Zhang, Y. Zhang, L. Zhang, S. Liu and W. Xie, Laser Photonics Rev., 2024, 18, 2400042 CrossRef.
- X. Li, Y. Du, X. Pan, C. Xiao, X. Ding, K. Zheng, X. Liu, L. Chen, Y. Gong, M. Xue, X. Tian and X. Zhang, Adv. Mater., 2025, 37, 2416991 CrossRef PubMed.
- J.-R. Zhang, A. Li, X.-L. Li, Y.-B. Zhao, J.-S. Sun, X.-X. Guo, W. Wang, J. Liu, Y.-L. Zhang and D.-D. Han, ACS Appl. Mater. Interfaces, 2025, 17, 18940–18953 Search PubMed.
- Y. Zhang, H. Duan, G. Li, M. Peng, X. Ma, M. Li and S. Yan, J. Nanobiotechnol., 2022, 20, 246 Search PubMed.
- Y. Liu, C. Zhang, Y. Chen, R. Yin, P. He and W. Zhao, ACS Appl. Mater. Interfaces, 2023, 15, 38572–38580 CrossRef PubMed.
- F. Schneider, T. Fellner, J. Wilde and U. Wallrabe, J. Micromech. Microeng., 2008, 18, 065008 CrossRef.
- W. Wang, G. Ren, M. Zhou and W. Deng, Polymer, 2021, 13, 1075 Search PubMed.
- E. R. Cholleti, J. Stringer, M. Assadian, V. Battmann, C. Bowen and K. Aw, Sensors, 2018, 19, 42 CrossRef PubMed.
- M. C. Lopes, V. G. De Castro, L. M. Seara, V. P. A. Diniz, R. L. Lavall and G. G. Silva, J. Appl. Polym. Sci., 2014, 131, 41207 CrossRef.
- H. J. Zo, S. H. Joo, T. Kim, P. S. Seo, J. H. Kim and J. S. Park, Fibers Polym., 2014, 15, 1071–1077 Search PubMed.
- D. Ding, Z. Shang, X. Zhang, X. Lei, Z. Liu, Q. Zhang and Y. Chen, Ceram. Int., 2020, 46, 28363–28372 Search PubMed.
- W. Zhang, L. Wang, S. Liu, F. Guan, M. Jiang, Y. Li, D. Yue, J. Li, X. Liu and Y. Feng, J. Alloys Compd., 2023, 950, 169760 Search PubMed.
- H. Li, X. Wang, Z. Ding, W. Gao, Y. Liu, K. Ma, Z. Hu and Y. Wang, Polymers, 2024, 16, 1188 Search PubMed.
- B. A. Alshammari, M. Hossain, A. M. Alenad, A. G. Alharbi and B. M. AlOtaibi, Polymers, 2022, 14, 1718 Search PubMed.
- P. Yang, F. Tian and Y. Ohki, IEEE Trans. Dielectr. Electr. Insul., 2014, 21, 2310–2317 Search PubMed.
- N. Jain, V. K. Singh and S. Chauhan, J. Mech. Behav. Mater., 2017, 26, 213–222 Search PubMed.
- K. S. Ramadan, D. Sameoto and S. Evoy, Smart Mater. Struct., 2014, 23, 033001 CrossRef.
- M. A. H. Khondoker and D. Sameoto, Smart Mater. Struct., 2016, 25, 093001 CrossRef.
- Y. Qiao, G. Gou, F. Wu, J. Jian, X. Li, T. Hirtz, Y. Zhao, Y. Zhi, F. Wang, H. Tian, Y. Yang and T.-L. Ren, ACS Nano, 2020, 14, 3779–3804 Search PubMed.
- S.-Y. Yue, T. Ouyang and M. Hu, Sci. Rep., 2015, 5, 15440 CrossRef PubMed.
- A. T. Khan, N. Wei, O. Salmela, K. Mustonen, Y. Liao, A. Hussain, E.-X. Ding, M. G. Uddin, H. Jiang, Y. Ohno and E. I. Kauppinen, Carbon, 2025, 239, 120320 CrossRef.
- A. Peigney, Ch. Laurent, E. Flahaut, R. R. Bacsa and A. Rousset, Carbon, 2001, 39, 507–514 CrossRef.
- R. Singampalli, R. Dachepalli, K. Chandra, B. Naidu, S. K. Nagasamudram, K. Srinivas, B. Basha and C. Beera, Biointerface Res. Appl. Chem., 2019, 9, 4205–4216 Search PubMed.
- Y. Yue, ACS Appl. Eng. Mater., 2024, 2, 638–648 Search PubMed.
- S. Ghosh, N. Kumar and S. Chattopadhyay, Asian J. Pharm. Sci., 2025, 20, 101007 Search PubMed.
- Y. Cao, P. Li, Y. Zhu, Z. Wang, N. Tang, Z. Li, B. Cheng, F. Wang, T. Chen and L. Sun, ACS Sens., 2025, 10, 272–282 CrossRef PubMed.
- Y. Wu, S. Li, Z. Zhao, D. Zhang, X. Xiong, T. Yu, M. D. Dickey and J. Yang, IEEE Sens. J., 2023, 23, 9176–9182 Search PubMed.
- O. Gul, J. Kim, K. Kim, H. J. Kim and I. Park, Adv. Mater. Technol., 2024, 9, 2302134 CrossRef.
- Y. Zhang, Q. Lu, J. He, Z. Huo, R. Zhou, X. Han, M. Jia, C. Pan, Z. L. Wang and J. Zhai, Nat. Commun., 2023, 14, 1252 Search PubMed.
- J. Liu, Y. Chen, Y. Liu, C. Wu, Z. Li, Y. Gao, X. Qiu, Y. Wang, X. Guo and F. Xuan, ACS Appl. Mater. Interfaces, 2024, 16, 29188–29197 CrossRef PubMed.
- G. Zhou, Y. Ma, B. Wei, J. Chen, Y. Zhang, S. Hu, J. Li and Y. Wang, J. Colloid Interface Sci., 2025, 696, 137871 CrossRef PubMed.
- S. Qin, P. Yang, Z. Liu, J. Hu, N. Li, L. Ding and X. Chen, Nat. Commun., 2024, 15, 10640 Search PubMed.
- Y. Zhong, Y. Wang, L. Ma, P. He, J. Qin, J. Gao and J. Li, ACS Appl. Mater. Interfaces, 2025, 17, 11154–11163 CrossRef PubMed.
- Y. Liu, C. Li, B. Li, S. Lu, S. Fan, S. Dong, Z. Wan and M. Shen, ACS Appl. Mater. Interfaces, 2023, 15, 51390–51398 CrossRef PubMed.
- Q. Fan, K. Li, H. Zhang, C. Han, Z. Wang, Q. Li, Z. Wan, Y. Zhou, X. Liu and J. Huang, ACS Photonics, 2025, 12, 1084–1094 CrossRef.
- S. Movaghgharnezhad, M. Kim, S. Min Lee, H. Jeong, H. Kim, B. Gak Kim and P. Kang, Mater. Des., 2023, 231, 112019 CrossRef.
- Y. Li, X. Zhao, Y. Tang, X. Zuo and H. Yang, Adv. Funct. Mater., 2024, 34, 2403059 Search PubMed.
- L. Dai, X. Wu, H. Hou, Z. Hu, Y. Lin and Z. Yuan, Lab Chip, 2024, 24, 1977–1986 Search PubMed.
- C. Shen, H. Yang, W. She and Q. Meng, Biotechnol. Bioeng., 2023, 120, 2027–2038 Search PubMed.
- Y. Bai, X. Yu, X. Han, Y. Liu and G. Li, Sens. Actuators, A, 2024, 377, 115683 CrossRef.
- Y. Gao, T. Voglhuber-Brunnmaier, Y. Li, L. Akh, N. H. Patino, A. K. Fajrial, M. Ruzzene, B. Jakoby and X. Ding, Phys. Rev. Lett., 2025, 134, 037002 CrossRef PubMed.
- Z. Huang, T. Chen, Y. Jiang, R. Zhou, Y. Wang, J. Ji, H. Xie, T. Pan, D. Fan, L. Liang, L. Yang, B. Jiang, P. Li, M. Gao, J. Zhu, G. Yao, D. Xue and Y. Lin, npj Flexible Electron., 2025, 9, 52 Search PubMed.
- F. Pappalardo, C. Panarello, S. Quattropani, L. Galluccio, A. Licciardello, R. Ruffino, G. Li-Destri, A. Lombardo, G. Morabito and N. Tuccitto, Lab Chip, 2025, 25, 1707–1717 Search PubMed.
- Q. Ding, H. Wang, Y. Zhou, Z. Zhang, Y. Luo, Z. Wu, L. Yang, R. Xie, B. Yang, K. Tao, S. Pan, F. Liu, J. Fu, F. Huo and J. Wu, Adv. Mater., 2025, 37, 2502369 CrossRef PubMed.
- P. Yi, X. Fu, Y. Liu, X. Zhang, C. Zhang and X. Li, Nano Energy, 2023, 113, 108592 CrossRef.
- K. Zhang, X. An, C. Wang, Y. Wang, Z. Sun, T. Ling, S. Lu and S. Sun, J. Mater. Sci., 2025, 60, 2419–2434 Search PubMed.
- Y. Pi, Q. Liu, Z. Li, D. Zhao, K. Zhang, Z. Liu, B. Zhou, I. M. Lei, Y. Ma and J. Zhong, npj Flexible Electron., 2023, 7, 45 Search PubMed.
- X. Wang, J. Lu, Y. Zhou, Y. Li, Z. Luo, Y. Gui, W. Lu and L. Li, Adv. Funct. Mater., 2025, 2506305 CrossRef.
- Q. Xie, L. Han, J. Liu, W. Zhang, L. Zhao, Y. Liu, Y. Chen, Y. Li, Q. Zhou, Y. Dong and X. Wang, Adv. Healthcare Mater., 2025, 14, 2402010 Search PubMed.
- X. Li, B. Qi, X. Wan, J. Zhang, W. Yang, Y. Xiao, F. Mao, K. Cai, L. Huang and J. Zhou, Nano Energy, 2023, 114, 108652 Search PubMed.
- N. Li, F. Zhan, M. Guo, X. Yuan, X. Chen, Y. Li, G. Zhang, L. Wang and J. Liu, Adv. Mater., 2025, 37, 2419524 CrossRef PubMed.
- G. Ye, Y. Yang, X. Zhang, Q. Wu, Y. Wan, N. Yang and P. Yang, Adv. Funct. Mater., 2025, e10705 Search PubMed.
- Z. Li, X. Li, K. Pang, K. Li, Y. Gao, C. Zhang, J. Lu, Y. Liu, Z. Xu and C. Gao, Nat. Commun., 2025, 16, 6087 Search PubMed.
- S. Tisato, G. Vera, Q. Song, N. Nekoonam and D. Helmer, Nat. Commun., 2025, 16, 6730 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




