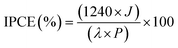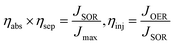Control of crystallization dynamics via vanadium–polyethylene glycol interactions toward high-performance BiVO4 photoanodes
Jihong
Min
a,
Donggu
Kim
a,
Hyewon
Park
b,
Jinwoo
Chu
a,
Nakyung
Kim
a,
Gihun
Jung
a,
Choongman
Moon
a,
Chang Hyun
Kim
a,
Yungi
Nam
a,
Sungbin
Yang
a,
Jeong Young
Park
 b and
Byungha
Shin
b and
Byungha
Shin
 *a
*a
aDepartment of Materials Science and Engineering, Korea Advanced Institute of Science and Technology (KAIST), Daejeon 34141, Republic of Korea. E-mail: byungha@kaist.ac.kr
bDepartment of Chemistry, Korea Advanced Institute of Science and Technology (KAIST), Daejeon 34141, Republic of Korea
First published on 16th October 2025
Abstract
BiVO4 is a promising photoanode material for photoelectrochemical (PEC) water splitting, but its short hole diffusion length significantly limits charge separation efficiency in thick films, which otherwise offer superior light absorption. To overcome this limitation polyethylene glycol (PEG) assisted metal–organic deposition (MOD) has been widely employed to create nanostructured BiVO4 films. However, reproducibility of morphology and PEC performance under identical PEG conditions remains challenging. Here, we reveal that such reproducibility issues stem from changes in PEG-V ion interactions due to variations in the vanadium oxidation state (V4+ → V5+), affecting crystallization kinetics. By developing an Acoustic Energy Assisted Aging (AEA) process to precisely control V oxidation states, we achieved reproducible morphology control from nanoporous structures to isolated large-grain morphologies under identical PEG conditions. Optimized isolated large-grain BiVO4 films exhibited outstanding crystallinity, ideal composition, enhanced optical absorption, and superior charge transport, delivering high photocurrent densities of 5.8 mA cm−2 (SOR, 1.23 V vs. RHE) and 5.4 mA cm−2 (OER with NiFeOOH catalyst). These results represent the highest PEC performance reported for MOD-derived BiVO4 films, highlighting a novel strategy for highly reproducible and efficient nanostructured photoanodes.
1 Introduction
Bismuth vanadate (BiVO4, hereafter BVO) has garnered significant attention for photoelectrochemical (PEC) hydrogen production applications due to its high chemical stability, cost-effectiveness, and favorable band-edge alignment for the oxygen evolution reaction (OER).1,2 BVO possesses a bandgap of approximately 2.4 eV, theoretically enabling a high photocurrent density of about 7.5 mA cm−2 and thus allowing a solar-to-hydrogen (STH) conversion efficiency approaching ∼10%.3 However, a fundamental limitation of BVO is its intrinsically low carrier mobility, particularly the short hole diffusion length,4 which necessitates maintaining an ultrathin film thickness for efficient charge transport. While such thin films minimize the diffusion distance for charge separation and collection, the restricted film thickness severely limits optical absorption, posing challenges in achieving higher PEC performance.5To overcome this limitation, numerous strategies have been reported to effectively enhance charge transport at a sufficient film thickness capable of adequate photon absorption. Notable approaches include doping with transition metals such as Mo5+ and W6+ to facilitate polaron hopping,6 and introducing electron transport layers like SnO2 and WO3 to induce favorable band bending for improved charge separation.1,7 However, thick films still demand long carrier diffusion lengths; thus, these strategies have not fundamentally overcome the intrinsic trade-off between enhanced light absorption and increased carrier diffusion distances in thicker BVO films.
Another approach is forming nanostructured (e.g., nanoflakes, nanoporous structures) BVO to effectively reduce carrier diffusion distances while maintaining sufficient film thickness for light absorption.8–10 Nanostructured BVO offers a substantially higher density of active reaction sites than a compact film, significantly shortening the distance photogenerated charges must travel to reach the surface.10,11 Additionally, internal light scattering inherent in the nanostructures enhances the optical path length, thereby improving overall light absorption efficiency.9 However, conventional nanostructured films often suffer from smaller grain sizes and increased grain boundary areas, resulting in higher surface defect concentrations and poor crystallinity, which may act as charge recombination centers.12 These structural limitations can significantly hinder charge transport efficiency.13 Moreover, excessively small grains can cause Rayleigh scattering, which rather diminishes optical absorption, particularly in the longer wavelength region.14–16 Hence, there remains a crucial need to develop novel BVO nanostructures capable of simultaneously optimizing light absorption and charge transport properties.
In the metal–organic decomposition (MOD) method, one of the most widely used approaches for synthesizing high-quality BVO, a structural modifier such as Polyethylene Glycol (PEG) is often employed to achieve desirable nanostructured morphologies. Previous studies have shown that PEG delays the crystallization kinetics of BVO, promoting the formation of porous structures.17 An increase in the molecular weight (i.e., longer chain length) or concentration of PEG typically leads to higher film porosity and smaller grains.9,18 Because of the issues, such as charge recombination at grain boundaries and Rayleigh scattering, associated with nanostructures with small grains, recent studies have introduced low molecular weight (shorter chain length) PEG, which facilitates the formation of isolated nanoparticles with larger grain sizes. Such isolated large grain structures retain the advantage of shortened charge diffusion pathways to the reaction surface, while simultaneously enhancing optical absorption and enhanced charge separation efficiency compared to conventional nanoporous films of comparable thickness.18,19 However, in practice, reliably and reproducibly obtaining the desired morphology through MOD synthesis remains challenging, even under identical PEG conditions (i.e., molecular weight and concentration).
In this study, we identified variations in the oxidation state of vanadium ions (V4+ or V5+) within the vanadium precursor during BVO film fabrication as a key source of reproducibility issues. In our previous study, we demonstrated that aging the vanadium precursor solution for the duration as long as three days under ambient conditions resulted in an optimal oxidation state, leading to BVO films with (Bi + V)/O stoichiometry optimized for enhanced electrochemical performance.20 However, this extended aging process is inherently inefficient and poses practical challenges, including concentration changes in vanadium ions due to natural evaporation of a volatile MeOH solvent and inconsistent precursor oxidation arising from environmental fluctuations such as temperature and humidity. These issues become even more critical when PEG is incorporated because both the morphology and chemical composition of the BVO films become highly sensitive to the oxidation state of the vanadium precursor. To address this, we introduced an Acoustic Energy Assisted Aging (AEA) method which is a faster, yet controlled and reliable aging strategy. AEA enables rapid and uniform oxidation of the vanadium precursor by continuously supplying high-pressure O2 and H2O. This approach significantly shortens the aging duration to just several tens of minutes, while ensuring excellent reproducibility in controlling the vanadium oxidation states.
The precise oxidation control afforded by AEA allowed us to regulate the interaction between vanadium ions and PEG molecules, enabling fine-tuned morphological control. By varying the AEA treatment duration of vanadium precursor solutions, we demonstrated a wide range of morphological tunability in BVO films—from small-grained nanoporous structures to isolated large-grained structures. Notably, morphology variations induced by changes in vanadium oxidation state in the precursor have a critical impact on the performance of the resulting PEC devices. Under the optimized AEA conditions, we successfully fabricated BVO thin films with well-defined isolated large-grain structures that exhibited superior optical absorption and enhanced charge transport, yielding a remarkable photocurrent density of 5.8 mA cm−2 at 1.23 V (vs. RHE) during the sulfate oxidation reaction (SOR), representing the highest performance reported to date among MOD-derived BVO photoanodes. Our study highlights that the oxidation state of vanadium ions within the precursor solution plays a critical role in determining the resulting film morphology. This finding emphasizes the need for careful control of the vanadium ion oxidation states to achieve controlled and reproducible morphologies.
2 Results and discussion
2.1 Development of acoustic energy assisted aging (AEA) process
In the synthesis of BVO films via MOD, separate precursor solutions are prepared by dissolving the bismuth precursor, bismuth nitrate pentahydrate (Bi(NO3)3·5H2O), in acetic acid and the vanadium precursor, vanadium acetylacetonate (VO(acac)2), in methanol (MeOH), respectively. MeOH in the vanadium precursor solution facilitates the oxidation of adjacent vanadium precursor by breaking V–O–V bonds, thereby converting the oxidation state of vanadium ions from V4+ to V5+.21,22 This oxidation, which progresses over time, is commonly referred to as “aging” and the degree of oxidation of vanadium ions increases with the duration of aging. Our previous study has demonstrated that variations in the oxidation state of the vanadium precursor significantly affect the chemical composition and thus the performance of resulting BVO photoelectrodes.20 In the present study, we introduced acoustic energy to substantially reduce the aging duration, ultimately shortening it to 50 min. The AEA process involves placing a vial containing the vanadium precursor solution into a bath filled with 20 mL of deionized (DI) water and applying ultrasonic energy at a frequency of 35 kHz (Fig. S1).As the oxidation of VO(acac)2 proceeds in MeOH, the absorption spectrum of the vanadium precursor solution undergoes a distinct red-shift, accompanied by a visible color transition from blue to green, and eventually to brown.20Fig. 1a illustrates the photos of the vanadium precursor solutions under different aging conditions exhibiting different colors. Under the ambient conditions, the solution aged for 70 min exhibited minimal color change, maintaining its original blue hue, indicative of negligible changes in the vanadium oxidation state. In contrast, the solution aged for the same duration of 70 min under the AEA conditions demonstrated a pronounced color transition from blue to green and finally to brown, signifying significant changes in the vanadium oxidation states (the bottom photo of Fig. 1a). UV-vis absorbance spectra of these vanadium precursor solutions aged under identical timeframes (Fig. 1b and c) show that the absorbance edge undergoes a substantial red-shift in the AEA-treated solutions compared to those aged under ambient conditions. These results clearly point to the accelerated oxidation process of the vanadium precursor by the AEA method, which typically requires several days under ambient conditions.
To elucidate the mechanism underlying the accelerated oxidation of VO(acac)2 during the AEA process, we investigated the oxidation pathways of VO(acac)2 in MeOH. It is well established that VO(acac)2 in solution can oxidize from V4+ to V5+ in the presence of sufficient H2O and O2, following the reaction described in eqn (1):21
| 4VO(acac)2 + 4MeOH + 2H2O + O2 → 4VO(OH)(OMe)(acac) + 4H(acac) | (1) |
When acoustic energy is supplied to a liquid medium, numerous microscopic bubbles (cavitation bubbles) form within the solution. Driven by acoustic vibrations, these bubbles progressively grow by rectified diffusion (absorbing dissolved gases from the surrounding liquid) and coalescence (merging with nearby bubbles). Eventually, when these bubbles reach their resonance size, they undergo violent collapse, generating localized conditions of extremely high pressure (hundreds of atmospheres) and high temperature (thousands of Kelvin).23 Given that the vanadium precursor solution contains dissolved H2O and O2, these gases are trapped in high-pressure bubbles formed by acoustic cavitation. Thus, it is expected that under these localized extreme conditions, the increased availability of reactive species (H2O and O2) would accelerate the oxidation of V4+ ions. Several previous studies also have reported that acoustic energy promotes electron transfer reactions between metals and organic solvents such as MeOH, further facilitating oxidation reactions.24
Additionally, the AEA process in this study was conducted by simultaneously applying thermal energy via a heated water bath; therefore, two major factors must be responsible for the accelerated oxidation: increased supply of reactive gases (H2O and O2) through acoustic cavitation and enhanced reaction kinetics resulting from elevated temperatures. This was confirmed by the comparison of vanadium precursor solutions aged under different conditions. Five vanadium precursor solutions were prepared and subjected to aging for 70 min under varying environmental conditions (the bottom panel of Fig. 1a): reference (aging conducted under ambient conditions with no heat or acoustic energy applied), N2-purged (ambient conditions with N2 purging to exclude dissolved O2 and H2O), heat-only (a heated bath at 50 °C without acoustic energy), acoustic-only (a lower temperature bath at 15 °C with acoustic energy only), and acoustic + heat (a heated bath at 50 °C with simultaneous application of acoustic energy and heat). Although aging at even higher temperatures may further enhance the oxidation rate, considering the boiling point of MeOH (64.7 °C), an excessively elevated temperature could lead to solvent evaporation and result in fluctuations in precursor concentration which would degrade the reproducibility of synthesized BVO films.
As shown in Fig. 1c, the absorbance spectra of vanadium precursors aged under different conditions demonstrated a notably larger red-shift compared to the reference sample when either acoustic energy or heat was applied. This indicates that applying either acoustic or thermal energy alone can accelerate oxidation compared to ambient conditions. Notably, the precursor solution exposed to both acoustic energy and heat simultaneously exhibited the largest degree of red-shift, suggesting that both acoustic and thermal energies contribute to accelerating the vanadium precursor oxidation. The N2-purged precursor solution exhibited rather a blue-shift compared to the reference sample (Fig. 1c), which is attributed to the substantially reduced oxidation of VO(acac)2 due to the decreased amounts of dissolved H2O and O2 present in the solution. These results suggest that acoustic energy accelerates the oxidation of the vanadium precursor primarily by enhancing the supply of H2O and O2 to the solution.
Based on our previous work, which systematically investigated the effect of precursor composition on the electrochemical performance of solution-processed BVO photoanodes and identified a Bi/V molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 as optimal, we adopted this ratio in the precursor solutions for the MOD process.25 However, despite using the same precursor ratio in the MOD solutions, we observed a decrease in the Bi/V ratio and an increase in oxygen content in the final BVO films as the degree of vanadium ion oxidation in the precursor solution, as revealed by X-ray photoelectron spectroscopy (XPS) and inductively coupled plasma-optical emission spectroscopy (ICP-OES) analysis of samples subjected to varying AEA treatment durations (Fig. 1d, e and S2, S3). The Bi/V ratio of the 0 min BVO film (i.e., without the AEA treatment) was found to be approximately 2.5, significantly deviating from the stoichiometric composition of BVO (Bi
1 as optimal, we adopted this ratio in the precursor solutions for the MOD process.25 However, despite using the same precursor ratio in the MOD solutions, we observed a decrease in the Bi/V ratio and an increase in oxygen content in the final BVO films as the degree of vanadium ion oxidation in the precursor solution, as revealed by X-ray photoelectron spectroscopy (XPS) and inductively coupled plasma-optical emission spectroscopy (ICP-OES) analysis of samples subjected to varying AEA treatment durations (Fig. 1d, e and S2, S3). The Bi/V ratio of the 0 min BVO film (i.e., without the AEA treatment) was found to be approximately 2.5, significantly deviating from the stoichiometric composition of BVO (Bi![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V = 1
V = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). This deviation likely arises because, without the AEA treatment, VO(acac)2 is not adequately dissolved in MeOH, leaving vanadium predominantly in the less oxidized V4+ state. Vanadium ions in the V4+ state are less favorable for the incorporation into the BVO lattice, resulting in a relatively low vanadium content within the final film. Upon increasing the AEA treatment duration, VO(acac)2 became fully dissolved in the precursor solution, and the oxidation of V4+ to V5+ was facilitated. Consequently, the vanadium content in the BVO films gradually increased with increasing AEA treatment duration. Bulk Bi/V ratios of the BVO films were measured by ICP-OES (Fig. S3). The trends in bulk Bi/V ratios measured matched those in the surface Bi/V ratio determined by XPS, indicating that compositional changes induced by varying AEA treatment durations persist throughout the entire BVO film. XPS analysis also reveals an increasing trend in oxygen content with extended AEA treatment duration (Fig. 1e). It has been reported that incomplete oxidation of V4+ in the precursor solutions leads to the formation of oxygen vacancies in the final films.26–30 The AEA treatment increases the amount of oxidized V5+ ions in the precursor, leading to a reduction in oxygen vacancy concentration in the final BVO films. Notably, when the precursor solution was subjected to approximately 50 min of AEA treatment, the resulting BVO film exhibited a (Bi + V)/O ratio close to the stoichiometric value (∼2).
1). This deviation likely arises because, without the AEA treatment, VO(acac)2 is not adequately dissolved in MeOH, leaving vanadium predominantly in the less oxidized V4+ state. Vanadium ions in the V4+ state are less favorable for the incorporation into the BVO lattice, resulting in a relatively low vanadium content within the final film. Upon increasing the AEA treatment duration, VO(acac)2 became fully dissolved in the precursor solution, and the oxidation of V4+ to V5+ was facilitated. Consequently, the vanadium content in the BVO films gradually increased with increasing AEA treatment duration. Bulk Bi/V ratios of the BVO films were measured by ICP-OES (Fig. S3). The trends in bulk Bi/V ratios measured matched those in the surface Bi/V ratio determined by XPS, indicating that compositional changes induced by varying AEA treatment durations persist throughout the entire BVO film. XPS analysis also reveals an increasing trend in oxygen content with extended AEA treatment duration (Fig. 1e). It has been reported that incomplete oxidation of V4+ in the precursor solutions leads to the formation of oxygen vacancies in the final films.26–30 The AEA treatment increases the amount of oxidized V5+ ions in the precursor, leading to a reduction in oxygen vacancy concentration in the final BVO films. Notably, when the precursor solution was subjected to approximately 50 min of AEA treatment, the resulting BVO film exhibited a (Bi + V)/O ratio close to the stoichiometric value (∼2).
2.2 Morphological evolution of BiVO4 films depending on the interaction strength between V ions and PEG
We formed nanostructured BVO thin films by incorporating PEG into the precursor solution. PEG is widely recognized as an effective structure-directing agent, capable of inducing nanopore formation within thin films and tuning their morphological characteristics.8,9,18 Previous studies have shown that by controlling the molecular weight and concentration of PEG, both the porosity and grain size of BVO films can be effectively modulated. These morphological changes are attributed to the strong dative bonds formed between the ether groups (O–C–O) present in PEG molecules and metal ions in the precursor solution, which restrict the diffusion of metal ions and inhibit grain growth during crystallization.31,32 Consequently, increasing PEG concentration or employing longer PEG chain lengths further inhibit metal ion diffusion, ultimately leading to nanostructured BVO films with increased porosity and smaller grains.In this study, we maintained a fixed PEG molecular weight of 200 and a vanadium precursor-to-PEG ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.2. However, we observed significant morphological differences depending on the oxidation state of the vanadium precursor, despite the identical PEG conditions. VO(acac)2 within the vanadium precursor solution decomposes to yield vanadium ions (V4+ or V5+), and the proportion of V5+ ions progressively increases with prolonged AEA treatment due to enhanced oxidation. Upon the oxidation from V4+ to V5+, vanadium ions lose one electron from the [Ar]3d1 configuration, resulting in empty 3d orbitals that facilitate stronger and more stable coordination bonds with the lone pair electrons of oxygen atoms in the PEG molecules. Therefore, we anticipated that such strengthened interactions between V5+ ions and PEG molecules effectively suppress ion diffusion and reduce the crystallization rate, thus inducing morphology changes.
0.2. However, we observed significant morphological differences depending on the oxidation state of the vanadium precursor, despite the identical PEG conditions. VO(acac)2 within the vanadium precursor solution decomposes to yield vanadium ions (V4+ or V5+), and the proportion of V5+ ions progressively increases with prolonged AEA treatment due to enhanced oxidation. Upon the oxidation from V4+ to V5+, vanadium ions lose one electron from the [Ar]3d1 configuration, resulting in empty 3d orbitals that facilitate stronger and more stable coordination bonds with the lone pair electrons of oxygen atoms in the PEG molecules. Therefore, we anticipated that such strengthened interactions between V5+ ions and PEG molecules effectively suppress ion diffusion and reduce the crystallization rate, thus inducing morphology changes.
To investigate the retardation of crystallization processes according to the oxidation degree of the vanadium precursor, differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) measurements were conducted on precursor solutions subjected to different durations of AEA treatment (Fig. 2b–d). As shown in the DSC (Fig. 2b and c) and TGA (Fig. 2d) results, heat flow changes and mass losses began at approximately 100 °C. The observed heat flow and mass losses at relatively lower temperatures (100–200 °C) can be attributed primarily to the decomposition of residual organic solvents. Meanwhile, exothermic peaks and mass losses at higher temperatures (>230 °C) correspond mainly to the combustion of PEG and crystallization of BVO. After annealing at 450 °C, the final residual mass for all samples remained similar (∼30–40%), indicating that initial conditions such as the residual solvent content and PEG quantity were consistent across the samples.
As evident from the DSC analysis shown in Fig. 2c, the exothermic peaks in the temperature range of 230–300 °C gradually shifted toward higher temperatures with increasing aging duration. A corresponding shift toward higher temperatures in mass loss was also observed in the TGA results (Fig. 2d). Therefore, this observed peak shift can be interpreted as a consequence of more strengthened binding between PEG molecules and oxidized V ions, which further delays the crystallization process. Notably, the DSC curve for the 0 min aging sample exhibited a clear deconvolution into two distinct peaks within the 230–300 °C temperature range. This indicates that, due to insufficient oxidation of the vanadium precursor, weak interaction between V ions and PEG led to PEG combustion and BVO crystallization occurring separately at distinct temperatures. These results demonstrate that fine control of precursor oxidation states can modulate the interaction strength between PEG and metal ions, thereby enabling fine-tuning of crystallization kinetics and ultimately the film morphology.
BVO thin films grown via hydrothermal methods typically undergo crystallization at temperatures exceeding approximately 280 °C, where nucleation and grain growth occur concurrently. Previous studies have reported that only small nuclei are observed at annealing temperatures around 350 °C, whereas well-defined crystalline grains appear after heat treatments at temperatures above 400 °C.33,34 Nucleation predominantly occurs below 350 °C, while grain growth takes place at temperatures above this threshold. Since PEG decomposes below approximately 350 °C, we anticipated that strengthened interactions between V5+ and PEG molecules will slow down the nucleation kinetics occurring at lower temperatures (below 350 °C). At temperatures exceeding 400 °C where rapid grain growth dominates, PEG would have already decomposed, thereby minimizing its influence on grain growth. Based on this rationale, we propose a schematic illustration describing how variations in the oxidation state of V ions influence the crystallization process of BVO (Fig. 2a). In the absence of aging, the influence of PEG is relatively weak, enabling active diffusion of Bi and V ions, thus promoting nucleation. In contrast, extended aging of V ions is expected to have stronger interactions with PEG molecules, thereby retarding nucleation. To verify this mechanism, early-stage microstructures were early-stage microstructures were examined by plan-view after a short isothermal anneal at 350 °C for ∼12 min (nucleation-dominant window) (Fig. S4). Films from aged and non-aged precursors were prepared under otherwise identical conditions. The non-aged film already shows discrete crystalline islands with a higher nucleus number density, whereas the aged film remains predominantly nanoporous with markedly fewer discernible nuclei. These SEM images corroborate a reduced nucleation rate and lower initial nucleus density for the aged precursor, consistent with the DSC/TGA trends in Fig. 2. As fewer nuclei would result in concentrated growth around a limited number of nucleation centers, leading to larger grain sizes in the resultant thin films.35–37 Thus, based on our proposed crystallization mechanism for PEG-added BVO thin films, we expect that sufficiently aged vanadium precursor solution will yield BVO films characterized by isolated and large grained morphology.
The correlation between the degree of vanadium precursor oxidation and BVO film morphology was confirmed by scanning electron microscopy (SEM) images of BVO thin films fabricated using vanadium precursor subjected to varying durations of AEA treatment (Fig. 3). In line with our proposed crystallization mechanism, the particle size of the BVO films gradually increased with prolonged aging duration. The BVO sample prepared without AEA (0 min) exhibited a compact morphology similar to the film fabricated without PEG (w/o PEG). In contrast, the BVO film prepared from the precursor aged for 10 min showed a nanoporous structure characterized by relatively small grains, reflecting enhanced PEG–vanadium ion interactions. For films fabricated from precursors aged for 30 min or longer, well-defined, round-shaped nanoparticles were formed, with grain sizes progressively increasing as AEA treatment time increased further. Particularly, the BVO film prepared from the precursor subjected to 50 min AEA treatment exhibited a morphology consisting of large grains well separated from adjacent grains.
To further elucidate the effect of morphological evolution on the crystallinity of the BVO films, X-ray diffraction (XRD) analysis was performed (Fig. S5). Examining the (112) diffraction peak, which exhibits the strongest intensity in BVO (Fig. S5b), the 0 min AEA sample displayed a relatively low peak intensity and broad full width at half maximum (FWHM). This indicates incomplete crystallization likely arising from insufficient dissolution and oxidation of VO(acac)2 in MeOH. Upon applying AEA treatment of 10 min or longer, clear (112) diffraction peaks were observed, with the FWHM progressively narrowing as the aging duration increased from 10 min to 50 min (Fig. S6). Notably, the 50 min AEA sample exhibited the narrowest FWHM (0.299°), reflecting the highest degree of crystallinity. Fig. S5c highlights an enlarged view of the diffraction peaks around 35°, showing clear peak splitting corresponding to the distinct (200) and (020) planes of monoclinic scheelite BVO, demonstrating high crystallinity of our BVO films.9,38 The clearest peak separation was particularly evident in the film subjected to 50 min of AEA treatment, further emphasizing that the 50 min condition effectively optimizes PEG–V ion interactions to yield a homogeneous, highly crystalline BVO morphology without secondary phases.
We examined how varied morphology affects the optical properties of the BVO films. Digital photographs of the BVO films shown in Fig. 4a, the transparency progressively decreases with increasing AEA treatment duration. UV-vis spectroscopy analysis (Fig. 4b) demonstrates that as the average particle size increases, the transmittance in the visible range (350–600 nm) gradually decreases. Notably, at 30 min of AEA treatment, when the morphology transitions from nanoporous to isolated grain structures, a substantial reduction in transparency was observed. In the reflectance spectra (Fig. 4c), the samples aged for 0 min and 10 min exhibited relatively low reflectance, due to higher transmittance. Conversely, samples treated for 30 min or longer, which displayed reduced transmittance, generally showed higher overall reflectance. Interestingly, the large round-shaped grain structures (30 min, 50 min, and 70 min of AEA) exhibit reduced reflectance within the 350–475 nm wavelength range as the grain size increases, which suggests that larger grains enhance forward scattering. As the particle size of BVO films increases and becomes comparable to the wavelength of incident light (approximately 300–520 nm), Mie scattering significantly enhances forward scattering, enabling photons to penetrate deeper into the film and thereby improving overall optical absorption. Fig. 4d presents the absorbance spectra (%), calculated by subtracting the measured reflectance and transmittance from 100%. All BVO films exhibit a distinct absorption edge near 500 nm, which aligns well with previously reported absorption edges for BVO.8,39,40 With the morphological shift from nanoporous to isolated round grains, the combined effects of reduced transmittance and decreased reflectance (due to increased particle size) result in a red-shift of the absorption edge and enhance overall absorbance as the AEA duration increased. Consequently, BVO films with isolated large grain structures demonstrating greater optical absorption properties were achieved with AEA treatments of 50 min or longer.
2.3 PEC performance of BiVO4 thin films
To investigate the impact of morphology changes on PEC performance, BVO photoanodes prepared with varying durations of AEA treatment (0 min, 10 min, 30 min, 50 min, and 70 min) were evaluated for sulfite oxidation reaction (SOR) and oxygen evolution reaction (OER). All photoanodes were prepared with an FTO/SnO2/BVO architecture, and their electrochemical characteristics were assessed under front-side illumination (1 sun, AM 1.5G conditions), meaning that light was incident on the BVO side, using a standard three-electrode configuration. The OER measurements were conducted in 1 M potassium borate (KB, pH = 9) electrolyte, while SOR measurements were performed in the same electrolyte with the addition of 0.3 M sodium sulfite (Na2SO3) as a hole scavenger. The SOR process has a lower thermodynamic equilibrium potential (0.74 V vs. RHE) and faster reaction kinetics compared to OER, enabling photogenerated charge carriers within BVO to readily participate in the electrochemical reaction without significant activation barriers. Thus, evaluating the SOR provides insight into the maximum achievable photocurrent density of BVO in PEC-based OER reactions under ideal catalytic conditions.Linear sweep voltammetry (LSV) results in Fig. 5a demonstrate a significant increase in photocurrent density (Jph) with increasing AEA treatment durations. Remarkably, the BVO film treated with 50 min AEA exhibited a notably high photocurrent density of 5.8 mA cm−2 at 1.23 V vs. reversible hydrogen electrode (RHE), representing the highest reported performance for MOD-derived BVO films to date (Fig. S7). However, further extending the AEA treatment duration to 70 min resulted in decreased photocurrent density. This decline is attributed to excessive particle aggregation during crystallization, which increases grain boundary density and introduces defect sites causing excessive charge recombination, thus impeding efficient charge transport. Consequently, the optimal AEA duration was identified as 50 min. To evaluate the reproducibility of BVO films fabricated via the AEA process, photocurrent densities were measured in SOR electrolyte at 1.23 V vs. RHE across four independent batches (Fig. 5c), each comprising six samples for each AEA treatment condition (0 min, 10 min, 30 min, 50 min, and 70 min). Although some variation in performance among the samples was noted even within the same AEA condition, this variability was much lower compared to previously reported solution-processed BVO films.20,41,42
Furthermore, to assess the intrinsic OER performance of the fabricated BVO photoanodes, linear sweep voltammetry (LSV) was conducted on bare BVO photoanodes without any additional catalysts on the surface (Fig. S8). The 50 min AEA treated BVO film exhibited the highest photocurrent density (2.58 mA cm−2 at 1.23 V vs. RHE). Consistent with the trends observed in the SOR measurements, PEC performance improved progressively with increasing AEA duration up to 50 min, beyond which it began to decline. Additionally, similar trends were observed in incident photon-to-current efficiency (IPCE) measurements conducted under the same OER conditions (Fig. S9).
Photocurrent densities of BVO photoanodes are generally lower under OER conditions compared to those obtained under SOR conditions due to the inherently higher kinetic barriers associated with OER compared to SOR.43,44 Thus, to enhance the OER performance and accurately evaluate the intrinsic activity of BVO photoanodes, we electrodeposited a NiFeOOH OER catalyst layer onto the BVO films using illuminated chronoamperometry (CA). High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and energy-dispersive X-ray spectroscopy (STEM-EDS) confirm a uniform coating of the NiFeOOH layer fully covering the BVO surface (Fig. S10). STEM-EDS mapping also confirms that Ni and Fe elements are uniformly distributed across the surface of the BVO films. As expected, the NiFeOOH layer significantly enhances charge injection efficiency (inj) (Fig. S11). Consequently, among the BVO photoanodes decorated with the NiFeOOH catalyst layer, the film treated with 50 min AEA still exhibited the highest PEC performance, achieving a photocurrent density of 5.4 mA cm−2 at 1.23 V (vs. RHE) for OER.
We investigated the transport properties of photogenerated charge carriers within the bulk of the BVO films by employing electrochemical impedance spectroscopy (EIS). Measurements were carried out under AM 1.5G illumination (1 sun) at 0.74 V vs. RHE, in the same SOR electrolyte used for LSV, with the frequency range set from 100 kHz to 1 Hz. To analyze the electrochemical properties of the BVO films, an equivalent circuit model was employed, consisting of a series resistance (Rs), in series with a parallel combination of a constant phase element (CPE), and a bulk transport/recombination resistance (Rp). In this equivalent circuit, Rp corresponds to the charge transport resistance occurring within the bulk of the BVO, which is equal to the diameter of the semicircle observed in the Nyquist plots (Fig. 5d). As depicted in the Nyquist plots in Fig. 5d, the bulk transport/recombination resistance (Rp) progressively decreased with increasing AEA duration, reaching the lowest value (∼430 Ω cm2) under the 50 min condition, indicating optimal charge transport properties.
In polycrystalline semiconductor films, charge transport is strongly influenced by grain boundaries, which often possess a high density of defects and trap states. These boundaries act as barriers to carrier transport, reducing charge mobility and promoting charge recombination.45,46 Accordingly, to examine the relationship between grain-boundary density and Rp, grain-boundary density was quantified from plan-view SEM (Fig. S12). This metric decreases systematically with increasing AEA duration and reaches a minimum for the 50 min sample (isolated large grains); the extracted Rp follows the same trend, yielding a clear positive correlation between grain-boundary density and bulk resistance. Although the 70 min film exhibits larger grains than the 50 min film, local aggregation increases the boundary density and correspondingly Rp, consistent with the LSV behavior in Fig. 5a. Given that grain boundaries are known to act as barriers to carrier transport and that their density directly correlates with Rp in our dataset, these results indicate that steering the morphology toward large, isolated grains with low grain boundary density enhances bulk charge transport. Additionally, cross-sectional SEM analysis revealed that the BVO films obtained via controlled V5+–PEG interactions had thicknesses around 260 nm (Fig. S13). Previous studies have reported that BVO films exceeding 200 nm typically experience increased recombination and diminished charge transport efficiency due to the extended carrier diffusion pathways to reach electrochemical reaction sites on the surface.34 However, our BVO films with isolated grains display remarkably low bulk charge transport resistances, as evidenced by the EIS measurements, despite being thicker than conventional BVO photoanodes—a feature that enhances light absorption.
3 Conclusions
In this work, we identify precise control of the oxidation state of vanadium ions within precursor solutions as a critical determinant for reproducible PEG-mediated nanostructuring in MOD-derived BVO photoelectrodes. By introducing an AEA aging strategy, we achieve rapid and accurate regulation of precursor oxidation states. Detailed DSC and TGA analyses reveal that increased vanadium oxidation (V4+ → V5+) in MeOH-based solutions substantially strengthens PEG–V ion interactions, markedly slowing crystallization kinetics during thermal annealing. Consequently, varying the AEA treatment duration induces a systematic morphological evolution of BVO films—from nanoporous structures with fine grains to isolated large-grain morphologies—which profoundly influences optical properties, charge transport, and overall PEC performance. In particular, BVO films fabricated from precursors treated via AEA for 50 minutes exhibit an optimal isolated large-grain structure, characterized by reduced grain boundary density and enhanced forward scattering due to increased grain size, resulting in significantly improved optical absorption and charge transport. Accordingly, these optimized films achieved exceptional photocurrent densities of 5.8 mA cm−2 at 1.23 V vs. RHE under SOR conditions and 5.4 mA cm−2 under OER conditions with a NiFeOOH catalyst, representing state-of-the-art performance among MOD-based BVO photoelectrodes reported to date. This work offers a fundamental strategy to address longstanding reproducibility challenges in solution-based nanostructuring approaches, providing critical insights toward designing high-performance photoelectrochemical materials.4 Experimental
4.1 Acoustic energy assisted aging of vanadium precursor
The acoustic energy-assisted aging (AEA) process was conducted by placing a vial containing the vanadium precursor solution into a water bath filled with 20 mL of deionized (DI) water, followed by exposure to ultrasonic irradiation at a fixed frequency of 35 kHz. The bath temperature was carefully maintained at 50 °C throughout the aging period, with periodic verification using a thermometer. By varying the duration of the AEA treatment, precise control over the oxidation state of the vanadium precursor was achieved.4.2 Fabrication of SnO2/BiVO4 photoanodes
Initially, a thin SnO2 electron-transporting layer (∼70 nm) was prepared on cleaned fluorine-doped tin oxide (FTO, 7 Ω cm−2) substrates using a sol–gel spin-coating method. Specifically, a precursor solution containing 0.2 M tin chloride dihydrate (SnCl2·2H2O, 98%, Sigma-Aldrich) dissolved in isopropanol (99%, Sigma-Aldrich) was spin-coated at 2000 rpm for 30 s. Subsequently, substrates were thermally annealed at 500 °C for 20 min to achieve a dense SnO2 layer.Following this, BiVO4 photoactive layers were fabricated via an optimized metal–organic decomposition (MOD) technique. In detail, the Bi precursor solution was prepared by dissolving 0.5 M Bi(NO3)3·5H2O in acetic acid, and the vanadium precursor was separately prepared by dissolving 0.5 M vanadyl acetylacetonate [VO(acac)2] in methanol. Additionally, to enhance charge carrier mobility, molybdenum (Mo) was incorporated as a dopant, and polyethylene glycol (PEG, molecular weight 200) was added to control film morphology. The Mo doping solution was prepared by dissolving 0.075 mol of Mo precursor in 5 mL of methanol (MeOH) and was subsequently introduced into the vanadium precursor solution at a concentration of 0.25 wt%. PEG was introduced at a volume equivalent to 20% of the vanadium precursor solution. Before mixing, the vanadium precursor underwent controlled acoustic energy assisted aging under different durations. After aging, the Bi and V solutions were combined at an optimized Bi![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V ratio of approximately 1
V ratio of approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. This mixture was spin-coated onto the prepared FTO/SnO2 substrates at 1500 rpm for 20 s. Each spin-coated layer underwent annealing at 480 °C for 30 min in ambient air. The deposition–annealing cycle was repeated four times to obtain BiVO4 films approximately 260 nm thick, which was determined to provide optimal photoelectrochemical performance.
1. This mixture was spin-coated onto the prepared FTO/SnO2 substrates at 1500 rpm for 20 s. Each spin-coated layer underwent annealing at 480 °C for 30 min in ambient air. The deposition–annealing cycle was repeated four times to obtain BiVO4 films approximately 260 nm thick, which was determined to provide optimal photoelectrochemical performance.
4.3 Photoelectrochemical deposition of NiFe-LDH catalyst
The NiFe layered double hydroxide (NiFe-LDH) catalyst was photoelectrochemically deposited using a conventional three-electrode electrochemical cell. The BiVO4/SnO2 single photoanode served as the working electrode, while a platinum coil and an Ag/AgCl reference electrode (3 M KCl) were employed as the counter and reference electrodes, respectively. Illumination was provided by a Xenon arc lamp equipped with an AM 1.5G filter at an intensity of 100 mW cm−2.The deposition procedure involved two sequential electrodeposition steps. Initially, an Fe-based precursor solution was prepared using 0.1 M iron(II) sulfate heptahydrate (FeSO4·7H2O, ≥98%, Sigma-Aldrich), which was purged under nitrogen gas flow for 30 minutes to remove dissolved oxygen. During this step, external potentials of 0.11 V (for the BiVO4/SnO2 electrode) vs. Ag/AgCl were applied under illumination until a total charge density of 400 mC cm−2 was accumulated. Subsequently, a potential of 1.2 V vs. Ag/AgCl was briefly applied under dark conditions for 1 min.
For the subsequent Ni deposition, a solution containing 0.1 M nickel(II) sulfate hexahydrate (NiSO4·6H2O, ≥98%, Sigma-Aldrich) was adjusted to pH 7 using 2 M sodium hydroxide (NaOH). Here, an external bias of −0.1 V (BiVO4/SnO2 electrode) vs. Ag/AgCl was applied until reaching a total charge density of 100 mC cm−2.
4.4 Material characterizations
The crystallographic structures of BiVO4 films were characterized using a versatile thin-film X-ray diffractometer (Rigaku Ultima IV). Surface and cross-sectional morphologies of the prepared BiVO4 films were observed through scanning electron microscopy (SEM) analyses using Hitachi SU5000 and ZEISS Sigma 300 instruments. Transmission electron microscopy (TEM) investigations were performed using a Talos F200X instrument operated at an accelerating voltage of 200 kV. The chemical composition and surface states of the BiVO4 films were examined by X-ray photoelectron spectroscopy (XPS) analysis utilizing a multipurpose spectrometer (Sigma Probe, Thermo VG Scientific) and ICP-OES (Thermo Scientific, iCAP 6300). Additionally, simultaneous thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) were conducted to investigate the thermal behavior and crystallization kinetics of the precursors. These analyses were carried out using a simultaneous TGA-DSC analyzer (LABSYS Evo, Setaram, France). Optical characteristics, including transmittance, reflectance, and absorbance of the BiVO4 films and vanadium precursors were evaluated using ultraviolet-visible (UV-vis) spectroscopy (SolidSpec-3700 spectrophotometer, Shimadzu).4.5 Photoelectrochemical characterizations
Photoelectrochemical (PEC) characterization of the fabricated photoelectrodes was conducted in a conventional three-electrode cell setup using an undivided quartz cell connected to a potentiostat (Bistat, Biologic Science Instruments). A platinum coil and an Ag/AgCl (3 M KCl) electrode were employed as counter and reference electrodes, respectively. All potentials measured against the Ag/AgCl electrode were converted to reversible hydrogen electrode (RHE) scale using the following equation:| ERHE = EAg/AgCl + 0.197 V + (0.0591 V × pH) |
A 300 W xenon arc lamp (Newport, model 66902) equipped with an AM 1.5G optical filter was utilized to provide simulated sunlight. Prior to each experiment, the irradiance intensity was calibrated to 100 mW cm−2 using a calibrated silicon photodiode (Newport, model 91150 V).
Electrochemical measurements were conducted in a potassium borate (KB, 1 M, pH ≈ 9) buffered electrolyte. For sulfite oxidation reaction (SOR) experiments, the KB electrolyte was supplemented with sodium sulfite (Na2SO3, 0.3 M). Linear sweep voltammetry (LSV) and cyclic voltammetry (CV) measurements were recorded starting from the open-circuit potential (OCP) toward anodic potentials at a fixed scan rate of 20 mV s−1. Additionally, to minimize oxygen interference, the electrolyte solutions were purged thoroughly with high-purity argon gas for at least 30 minutes prior to initiating water oxidation measurements.
4.6 Incident photon-to-current efficiency (IPCE) measurement
IPCE measurements of the BiVO4 were performed using a 300 W xenon lamp (ABET Technologies, model 10500) coupled with a monochromator (DONGWOO OPTRON, Mmac 200) set at a bandwidth of 10 nm. The measurements were conducted under a three-electrode configuration at an applied potential of 1.23 V vs. RHE. Prior to measurement, the illumination intensity was carefully calibrated using a silicon photodiode detector (Bunkokeiki, BS-500BK). The electrolyte solution within an undivided quartz cell was thoroughly purged with Ar for 30 minutes to eliminate dissolved oxygen. The IPCE was calculated according to the following equation:where λ is the wavelength of the incident monochromatic light (nm), J is the measured photocurrent density (mA cm−2), and P is the incident monochromatic light intensity (mW cm−2).
Bulk charge separation efficiency considering light absorption (ηabs × ηsep) and charge injection efficiency (ηinj).
The charge separation efficiency considering light absorption (ηabs × ηsep) and injection efficiency (ηinj) were calculated using the following equations:
Author contributions
J. M. conceived and designed the study and drafted the manuscript. D. K. provided the BiVO4 photoelectrodes and contributed to materials synthesis. H. P., J. Y. P., J. C., N. K., Y. N., and S. Y. conducted PEC measurements and additional characterizations. J. M., G. J., C. M., and C. H. K. contributed to analysis and discussion of experimental results. B. S. supervised the entire project. All authors contributed to manuscript revision and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Additional raw data are available from the corresponding author upon reasonable request.The data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta06109a.
Acknowledgements
This work was supported by the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (No. RS-2025-02315803). This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Ministry of Strategy and Finance (No. RS-2021-NR057811).References
- S. Byun, B. Kim, S. Jeon and B. Shin, J. Mater. Chem. A, 2017, 5, 6905–6913 RSC.
- X. Shi, H. Jeong, S. J. Oh, M. Ma, K. Zhang, J. Kwon, I. T. Choi, I. Y. Choi, H. K. Kim, J. K. Kim and J. H. Park, Nat. Commun., 2016, 7, 11943 CrossRef CAS.
- C. Ros, T. Andreu and J. R. Morante, J. Mater. Chem. A, 2020, 8, 10625–10669 RSC.
- M. Ziwritsch, S. Müller, H. Hempel, T. Unold, F. F. Abdi, R. Van De Krol, D. Friedrich and R. Eichberger, ACS Energy Lett., 2016, 1, 888–894 CrossRef CAS.
- K. J. McDonald and K. S. Choi, Energy Environ. Sci., 2012, 5, 8553–8557 RSC.
- S. K. Cho, H. S. Park, H. C. Lee, K. M. Nam and A. J. Bard, J. Phys. Chem. C, 2013, 117, 23048–23056 CrossRef CAS.
- S. Y. Chae, C. S. Lee, H. Jung, O.-S. Joo, B. K. Min, J. H. Kim and Y. J. Hwang, ACS Appl. Mater. Interfaces, 2017, 9, 19780–19790 CrossRef CAS PubMed.
- H. He, S. P. Berglund, A. J. E. Rettie, W. D. Chemelewski, P. Xiao, Y. Zhang and C. B. Mullins, J. Mater. Chem. A, 2014, 2, 9371–9379 RSC.
- C. Feng, Z. Jiao, S. Li, Y. Zhang and Y. Bi, Nanoscale, 2015, 7, 20374–20379 RSC.
- E. Samuel, B. Joshi, M. W. Kim, M. T. Swihart and S. S. Yoon, Nano Energy, 2020, 72, 104648 CrossRef CAS.
- C. Nanoparticle, A. Aadenan, N. Affiqah, A. Mohamad, F. Mohamad, N. Mohd and N. Daud, Improving Photocatalytic Activities of LaFeO3 Photocathode by Chromium-Incorporated Nanoparticle, 2024, pp. 775–790 Search PubMed.
- Y. K. Kho, W. Y. Teoh, A. Iwase, L. Mädler, A. Kudo and R. Amal, ACS Appl. Mater. Interfaces, 2011, 3, 1997–2004 CrossRef CAS.
- A. Iwase and A. Kudo, J. Mater. Chem., 2010, 20, 7536–7542 RSC.
- K. H. Ye, H. Li, D. Huang, S. Xiao, W. Qiu, M. Li, Y. Hu, W. Mai, H. Ji and S. Yang, Nat. Commun., 2019, 10, 1–9 CrossRef CAS.
- D. K. Lee and K. S. Choi, Nat. Energy, 2018, 3, 53–60 CrossRef CAS.
- C. Lin, C. Dong, S. Kim, Y. Lu, Y. Wang, Z. Yu, Y. Gu, Z. Gu, D. K. Lee, K. Zhang and J. H. Park, Adv. Mater., 2023, 35, 1–8 Search PubMed.
- S. Bi, Z. Yao, X. Han, C. Bi, X. Wang, Q. Chen, Y. Wang, R. Wang, K. Asare-Yeboah, Z. He and R. Song, Electron. Mater. Lett., 2024, 20, 711–724 CrossRef CAS.
- S. G. Shim, J. Tan, H. Lee, J. Park, J. Yun, Y. S. Park, K. Kim, J. Lee and J. Moon, Chem. Eng. J., 2022, 430, 133061 CrossRef CAS.
- L. Li, H. Tang, Y. Chen, R. Yang, D. Tian and Z. Chen, Electron. Mater. Lett., 2020, 16, 481–490 CrossRef CAS.
- S. Byun, G. Jung, Y. Shi, M. Lanza and B. Shin, Adv. Funct. Mater., 2020, 30, 1–7 CrossRef.
- R. Gryboś, A. Samotus, N. Popova and K. Bogolitsyn, Transition Met. Chem., 1997, 22, 61–64 CrossRef.
- N. Ndiaye, M. Thiam, B. Ngom, O. Sakho, M. Chaker, N. Manyala and A. Beye, Am. J. Nanomater., 2017, 5, 59–67 CrossRef CAS.
- S. Manickam, D. Camilla Boffito, E. M. M. Flores, J. M. Leveque, R. Pflieger, B. G. Pollet and M. Ashokkumar, Ultrason. Sonochem., 2023, 99, 106540 CrossRef CAS.
- P. Cintas, A. Barge, S. Tagliapietra, L. Boffa and G. Cravotto, Nat. Protoc., 2010, 5, 607–616 CrossRef CAS PubMed.
- S. Byun, G. Jung, S. Y. Moon, B. Kim, J. Y. Park, S. Jeon, S. W. Nam and B. Shin, Nano Energy, 2018, 43, 244–252 CrossRef CAS.
- S. Wang, P. Chen, J. H. Yun, Y. Hu and L. Wang, Angew. Chem., Int. Ed., 2017, 56, 8500–8504 CrossRef CAS PubMed.
- Y. Zhang, Y. Guo, H. Duan, H. Li, C. Sun and H. Liu, Phys. Chem. Chem. Phys., 2014, 16, 24519–24526 RSC.
- Y. Zhou, L. Zhang, L. Lin, B. R. Wygant, Y. Liu, Y. Zhu, Y. Zheng, C. B. Mullins, Y. Zhao, X. Zhang and G. Yu, Nano Lett., 2017, 17, 8012–8017 CrossRef PubMed.
- X. An, T. Li, B. Wen, J. Tang, Z. Hu, L. M. Liu, J. Qu, C. P. Huang and H. Liu, Adv. Energy Mater., 2016, 6, 1–10 Search PubMed.
- B. J. Trześniewski, I. A. Digdaya, T. Nagaki, S. Ravishankar, I. Herraiz-Cardona, D. A. Vermaas, A. Longo, S. Gimenez and W. A. Smith, Energy Environ. Sci., 2017, 10, 1517–1529 RSC.
- Y. Zhao, J. Wei, H. Li, Y. Yan, W. Zhou, D. Yu and Q. Zhao, Nat. Commun., 2016, 7, 1–9 Search PubMed.
- D. B. Pedersen, M. Z. Zgierski, S. Anderson, D. M. Rayner, B. Simard, S. Li and D. S. Yang, J. Phys. Chem. A, 2001, 105, 11462–11469 CrossRef.
- K. R. Yoon, J. W. Ko, D. Y. Youn, C. B. Park and I. D. Kim, Green Chem., 2016, 18, 944–950 RSC.
- A. Chemseddine, K. Ullrich, T. Mete, F. F. Abdi and R. Van De Krol, J. Mater. Chem. A, 2016, 4, 1723–1728 RSC.
- R. Gottesman, I. Peracchi, J. L. Gerke, R. Irani, F. F. Abdi and R. van de Krol, ACS Energy Lett., 2022, 7, 514–522 CrossRef.
- A. Chianese and H. J. Kramer, Industrial Crystallization Process Monitoring and Control, 2012 Search PubMed.
- S. Wang, B. Liu, Q. Wang, Z. Gong, P. Zhang, T. Wang and J. Gong, Adv. Funct. Mater., 2024, 2403019, 1–9 Search PubMed.
- S. Tokunaga, H. Kato and A. Kudo, Chem. Mater., 2001, 13, 4624–4628 CrossRef.
- X. Lu, K. hang Ye, S. Zhang, J. Zhang, J. Yang, Y. Huang and H. Ji, Chem. Eng. J., 2022, 428, 131027 CrossRef.
- R. Li, H. Han, F. Zhang, D. Wang and C. Li, Energy Environ. Sci., 2014, 7, 1369–1376 RSC.
- Y. Liang, T. Tsubota, L. P. A. Mooij and R. Van De Krol, J. Phys. Chem. C, 2011, 115, 17594–17598 CrossRef.
- K. Sayama, A. Nomura, T. Arai, T. Sugita, R. Abe, M. Yanagida, T. Oi, Y. Iwasaki, Y. Abe and H. Sugihara, J. Phys. Chem. B, 2006, 110, 11352–11360 CrossRef CAS PubMed.
- C. Zachäus, F. F. Abdi, L. M. Peter and R. Van De Krol, Chem. Sci., 2017, 8, 3712–3719 RSC.
- M. Barroso, C. A. Mesa, S. R. Pendlebury, A. J. Cowan, T. Hisatomi, K. Sivula, M. Grätzel, D. R. Klug and J. R. Durrant, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 15640–15645 CrossRef CAS.
- C. Santato, M. Odziemkowski, M. Ulmann and J. Augustynski, J. Am. Chem. Soc., 2001, 123, 10639–10649 CrossRef CAS PubMed.
- C. A. Bignozzi, S. Caramori, V. Cristino, R. Argazzi, L. Meda and A. Tacca, Chem. Soc. Rev., 2013, 42, 2228–2246 RSC.
| This journal is © The Royal Society of Chemistry 2025 |