Silver nanoclusters: synthesis, structures and photoluminescence†
Yun-Peng
Xie
 *a,
Yang-Lin
Shen
a,
Guang-Xiong
Duan
a,
Jun
Han
a,
Lai-Ping
Zhang
b and
Xing
Lu
*a,
Yang-Lin
Shen
a,
Guang-Xiong
Duan
a,
Jun
Han
a,
Lai-Ping
Zhang
b and
Xing
Lu
 *a
*a
aState Key Laboratory of Materials Processing and Die & Mould Technology, School of Materials Science and Engineering, Huazhong University of Science and Technology (HUST), Wuhan 430074, China. E-mail: xieyp@hust.edu.cn; lux@hust.edu.cn
bCollege of Chemistry and Chemistry Engineering, Xinxiang University, Xinxiang 453000, China
First published on 27th May 2020
Abstract
Metal nanoclusters (NCs) consist of tens to hundreds of metal atoms with a diameter of <2 nm, and have attracted significant attention due to their unique molecule-like properties, such as well-defined molecular structures, explicit HOMO–LUMO transitions, quantized charge and strong luminescence emission. Various robust synthetic protocols have been successfully applied to the preparation of metal NCs. Among metal NCs, Au NCs stay at the frontline of this research, and more structural characteristics, particular optical, catalytic and electronic properties, and related technical applications of Au NCs have been discovered in recent years. By taking guidelines from Au NC research, Ag NCs have recently received increasing attention. In this review article, we first survey recent advances in developing efficient synthetic methods for Ag NCs, highlighting the underlying physical and chemical properties that make the delicate control of their sizes and surfaces possible. In the following section, we discuss recent advances in the structural determination of Ag NCs, such as Ag25(2,4-DMBT)18 (2,4-DMBT: 2,4-dimethylbenzenethiolate), Ag29(1,3-BDT)12 (1,3-BDT: 1,3-benzenedithiolate), and Ag44(SR)30 (R = PhCO2H2, PhF, PhF2 or PhCF3). Structural determination will help to gain deep insight into the structure–property relationships at the molecular level. In the last part, we highlight some examples of Ag NCs to demonstrate their photoluminescence properties, which offer potential applications as photodetectors and in sensing and bio-imaging. We give a brief outlook on the future development of Ag NCs from the viewpoint of synthesis and applications.
1. Introduction
Atomically precise metal nanoclusters (NCs) are ultrasmall particles with core sizes below 2 nm, and they are in between the atomic regime and plasmonic metal nanoparticles.1–7 Such metal NCs exhibit dramatically unique electronic and optical properties, such as molecule-like energy gaps, strong photoluminescence (PL) and high catalytic properties.8–15 Among metal NCs, noble metal NCs have drawn tremendous attention from the scientific community because of their unique structures and correlation with versatile applications.3–7 A number of magic size Au and Ag NCs with precise formulas such as Au25(SCH2CH2Ph)18,16–18 Au38(SCH2CH2Ph)24,19–21 and Au102(SPhCO2H2)4422 as well as Ag25(2,4-DMBT)18,23 Ag29(1,3-BDT)12,24 and Ag44(SR)30 (R = PhCO2H2, PhF, PhF2 or PhCF3)25,26 are known. Organic ligands such as thiolates, phosphines, and alkynyls are usually used to cap the surface in order to prevent aggregation and to facilitate the isolation of target Au and Ag NCs.6,7,27 These ligands not only influence the formation processes of Au and Ag NCs but also determine their structures and thus sizes, shapes and eventual properties.Among the noble metal NCs reported so far, Ag NCs are particularly attractive because of the unique physical properties of Ag NCs, such as their strong luminescence and ultra-small size. Such properties provide good platforms to construct luminescent probes for bio-imaging and sensing applications.28,29 However, silver in the zero-valent state is more reactive and easier to oxidize than gold, which makes it more difficult to prepare Ag NCs and investigate their properties as compared with the rather intensively studied gold analogues. Thus, the accessibility of high-quality Ag NCs with well-defined size, structure and surface is crucial for both fundamental and applied science.
Recently, a number of efficient strategies have been developed for the synthesis of Ag NCs with tailorable physico-chemical properties, and also in quantities large enough for practical applications.28,29 With delicate design of synthetic methods such as direct reduction, chemical etching and ligand exchange, many mature processes can be used to prepare high-quality Ag NCs with novel and even unprecedented properties. On the other hand, some techniques such as UV-vis absorption spectroscopy, PL emission spectroscopy, electrospray ionization mass spectrometry (ESI-MS), single crystal X-ray crystallography (SC-XRD), etc. have been used to characterize the physio-chemical properties and determine the total structures of Ag NCs, forming a central research direction in nanoscience.28–30 The atomically precise nature of their structures enables the investigation of the structure–property relationship, which may further optimize their performance. One of the most important spectroscopic properties is the luminescence, which is particularly useful in biological applications. Tunable PL properties of Ag NCs by controlling the core size and the nature of ligands have been reported during the last five years.31,32 It is therefore a focus of this review article to provide a detailed discussion on the synthesis, structures and photoluminescent properties of Ag NCs.
In this review article, we first survey the robust synthetic methods such as direct reduction, chemical etching and ligand exchange for high-quality Ag NCs reported during the last five years. Then we discuss recent advances in the determination of the crystal structure of Ag NCs with different sizes and well-defined molecular formulas. In the next section, we highlight some examples relating to the PL properties of Ag NCs. Some factors such as the core size, capping ligand, heterometal atom and temperature governing the PL properties of Ag NCs are addressed. Finally, a brief conclusion and an outlook on the future research challenges for Ag NC research will be provided.
2. Synthesis of Ag CNs
Compared to the synthesis of Au NCs, the synthesis of Ag NCs is more challenging due to their relative susceptibility in solution under atmospheric conditions. Thus, more delicate control is required to synthesize a well-defined composition of Ag NCs. Some successful attempts have been recently reported.28,29 Some characterization techniques including laser desorption ionization (LDI), matrix assisted laser desorption ionization (MALDI), electrospray ionization (ESI) mass spectrometry (MS), SC-XRD and post-synthetic separation methodologies such as size exclusion chromatography (SEC) and polyacrylamide gel electrophoresis (PAGE) are also used to determine the composition and structures of Ag NCs.33The synthesis of Ag NCs can be roughly classified into three categories: direct reduction of silver precursors in the presence of desired ligands, chemical etching and postsynthetic ligand-exchange.
2.1. Direct reduction
The direct reduction method has been successfully used to prepare Ag NCs in both organic and aqueous media. This synthetic process includes rapid reductive growth of intermediate Ag NCs and slow size focusing to monodisperse Ag NCs in a reducing agent.34 NaBH4 is commonly used as the reducing agent to synthesize Ag NCs with a variety of ligands such as thiolates, alkynyls, DNAs, peptides, proteins and polymers. A few Ag NCs, including thiol-protected Ag NCs (such as Ag25(2,4-DMBT)1823 and Ag29(1,3-BDT)1224) and alkynyl-protected Ag NCs (such as Ag74(PhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)4435 and Ag51(tBuC
C)4435 and Ag51(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)3236) have been successfully synthesized by using this method. However, the reduction kinetics with NaBH4 are generally fast and this leads to the formation of polydisperse Ag NCs. Thus, several methods can be used to slow down the reduction kinetics of NaBH4. For example, the solution pH,37 the concentration of reducing agents,38,39 and the solvent40 can be used to adjust the reducing capability of NaBH4. Another efficient way to slow down the reduction kinetics for Ag NC formation is to replace NaBH4 with other mild reducing agents such as formic acid and DMF.41,42 Some other techniques, such as light,43 ultra-sonication,44 and electricity,45 can also be used to create a mild reducing environment for the formation of Ag NCs.
C)3236) have been successfully synthesized by using this method. However, the reduction kinetics with NaBH4 are generally fast and this leads to the formation of polydisperse Ag NCs. Thus, several methods can be used to slow down the reduction kinetics of NaBH4. For example, the solution pH,37 the concentration of reducing agents,38,39 and the solvent40 can be used to adjust the reducing capability of NaBH4. Another efficient way to slow down the reduction kinetics for Ag NC formation is to replace NaBH4 with other mild reducing agents such as formic acid and DMF.41,42 Some other techniques, such as light,43 ultra-sonication,44 and electricity,45 can also be used to create a mild reducing environment for the formation of Ag NCs.
2.2. Chemical etching
Some Ag NCs can also be produced via a chemical etching process, where a relatively larger Ag nanoparticle (NP) is etched to form small Ag NCs.46–48 Compared to the direct reduction method, there are fewer successful attempts reported involving the chemical etching process, since in general the latter is more time-consuming and often produces Ag NCs at lower yields. Such constraints can be partially addressed by optimizing the etching conditions, including the etching time, reaction temperature, and the ratio of etchant to Ag precursors.An efficient synthetic protocol requires a mild etching environment that makes possible controlled formation of Ag NCs in the reaction solution. For example, an interfacial etching process was used to digest the as prepared Ag NPs to form two luminescent Ag NCs protected by mercaptosuccinic acid (H2MSA).49 During the reaction, Ag@(H2MSA) NPs were employed as starting materials and converged to a mixture of Ag8 and Ag7 NCs in an aqueous–organic biphasic system, and such a mixture was separated using gel electrophoresis. Another example is the synthesis of the red luminescent Ag38 NC through the etching of large citrate-protected Ag@citrate NPs by adding excess mercaptosuccinic acid.50 During the reaction, there is a disappearance of the plasmon feature at 420 nm, implying that that Ag@citrate NPs were converted to Ag NCs. The route provides nearly pure Ag38 NCs, and no byproducts were detected.
2.3. Ligand exchange
The ligand-exchange-induced size/structure transformation process is becoming an important approach in recent years. The peripheral organic ligands have a significant influence on the nuclearity, geometry, bonding and electronic transitions. Depending on the well characterized metal NC species, ligand-exchange may be partial or complete, with or without altering the metal core. In 2014, Bakr and coworkers presented a ligand-exchange method for the rapid and complete thiolate-for-thiolate exchange of Ag44(SR)30.51 Later, they found that the ligand-exchange conversion of Ag35(SG)18 (SG: glutathionate) into Ag44(4-FTP)30 (4-FTP: 4-fluorothiophenol) is also rapid and direct, while the reverse process proceeds slowly through intermediate cluster sizes.52 Meanwhile, the hollow structure of Ag44(4-FTP)30 is converted to non-hollow Ag25(2,4-DMBT)18via a disproportionation mechanism with the ligand-exchange method. The reverse reaction between Ag25(2,4-DMBT)18 and 4-FTP results in Ag44(4-FTP)30 by dimerization followed by a rearrangement reaction.53 Recently, the Pradeep group has performed the rapid transformation of Ag59(2,5-DCBT)32 (2,5-DCBT: 2,5-dichlorobenzenethiolate) to other well-known Ag NCs, Ag44(2,4-DCBT/4-FTP)30, Ag25(2,4-DMBT)18 and Ag29(1,3-BDT)12(PPh3)4 (PPh3: triphenylphosphine), by an exchange reaction with diverse thiol ligands.54Other methods, such as performing the reaction in the solid state or in a gel, can also be used for the synthesis of Ag NCs. For instance, the Pradeep group developed a solid-state method to produce red-emitting thiolated Ag9(H2MSA)7 nanoclusters.55 This method can also synthesize thiolated Ag32(SG)1956 and Ag152(PET)60 (PET: phenylethanethiol),57 and selenolate-protected Ag44(SePh)30 NCs.58 In addition, Chakraborty et al. applied the gel route to generate thiolated Ag25(SG)18 NCs with strong red emission.59 The synthesis of Ag NCs has been summarized in some recent reviews.28,29
3. Structures of Ag NCs
Based on the reported metal NCs with a fully determined structure, it has been found that the stability and properties of metal NCs are influenced by a number of factors including their compositions, core structures and surface functionalities. Hence, it is crucial to control the sizes and geometric structures of the cores and the interfacial structures. Many Au NCs have been structurally determined by SC-XRD.6,7 In contrast, the number of structurally determined Ag NCs has been limited due to their weaker stability, aerial oxidation, and lower purity. Some examples of structurally solved ligand-protected Ag NCs have been reported. More details about structural details are presented in Table S1 (ESI†). Most of the representative structures of Ag NCs could be considered as being built from basic kernel units such as Ag4, Ag6, Ag7 and Ag13 polyhedrons. On the other hand, peripheral ligands including thiols, phosphines, alkynyls or their combination are used to protect Ag NCs. Here, we categorize the important studies of Ag NCs based on different surface ligands.3.1. Thiol-protected Ag NCs
Thiol is the most widely used capping ligand in the shape controlled synthesis of Au and Ag NCs. In the reported thiol-protected Ag NCs with a fully determined structure, most of the representative structures could be categorized into the structure with a keplerate Ag icosahedron core and the structure with atoms arranged in a face-center-cubic (FCC) like pattern.The icosahedron is perhaps the most widely observed structure in metal NCs. The first reported crystal structure of all thiol-protected Ag NCs was that of Ag44(SR)30 (R = PhCO2H2, PhF, PhF2 or PhCF3) in 2013.25,26 The single crystal structure suggests the existence of a Ag32 kernel and six Ag2(SR)5 staples. The Ag32 kernel can further be divided into an icosahedral Ag12 core and a 20-silver-atom dodecahedral shell (Fig. 1a). By using such Ag44 as seeds, larger size Ag50(TBBM)30(dppm)6 (TBBM: 4-tert-butylbenzyl mercaptan; dppm: bis(diphenylphosphino)methane) was obtained by Zhu and coworkers.60
 | ||
| Fig. 1 Total crystal structures of the Ag44(SPhCO2H2)30 and the Ag50(TBBM)30(dppm)6 nanoclusters. Adapted with permission from ref. 60. Copyright 2017, American Chemical Society. | ||
The structure of Ag50 comprises a Ag32 kernel which is retained from Ag44 surrounded by a dodecahedral Ag20 and two symmetrical Ag9(TBBM)15P6 ring motifs (Fig. 1b). Subsequently, the structure of the “golden silver” Ag25(2,4-DMBT)18 NC23 is found to be essentially identical to that of Au25(SCH2CH2Ph)18.16–18 The structure of the Ag25(2,4-DMBT)18 NC has an icosahedral Ag13 kernel which is protected by six dimeric staples (Fig. 2a). The crystal structure of Ag29(1,3-BDT)12(Ph3P)4 protected by a dithiol and monodentate phosphine ligand has been solved by Antoine and coworkers.24 The Ag29 cluster has an icosahedral core similar to that of Ag25(2,4-DMBT)18. The icosahedral core Ag13 is protected with a shell consisting of Ag16S24P4 (Fig. 2b). The shell is composed of four Ag3S6 crowns and four Ag1S3P1.
 | ||
| Fig. 2 Core, shell and framework of the Ag25(SPhMe2)18 and the Ag29(BDT)12(TPP)4 nanoclusters. Adapted with permission from ref. 6. Copyright 2016, American Chemical Society. | ||
During the past few years, FCC core structures in metal NCs have attracted great attention due to their key roles in understanding the origin of macroscopic FCC metal materials such as gold, silver, copper, etc.6,61–63 Several FCC-type kernel structures for Ag NCs have been reported thus far.
The FCC unit cell comprises 8 vertices and 6 face centers and, hence, a total of 14 atoms in the unit. For example, the crystal structure of an all thiol-protected [Ag62S12(StBu)32]2+ nanocluster (Ag-NC) shows a complete FCC Ag14 core structure with a Ag48(StBu)32 shell configuration interconnected by 12 sulfide ions, which is similar to the [Ag62S13(StBu)32]4+ (Ag-QD) structure.64,65 In the center of the Ag14 kernel, there is an octahedral Ag6 kernel, which is enclosed by eight equilateral-triangle-shaped planes. Of note, the Ag-QD could be electrochemically reduced into the Ag-NC, via an intermediate compound [Ag62S13(StBu)32]2+ (Ag-inter) (Fig. 3),66 wherein the Ag62 nanocluster template remained unchanged. It can be seen that the Ag-QD, Ag-inter and Ag-NC have a similar Ag36(StBu)32 surface and (AgS)12 subsurface connecting the Ag36(StBu)32 surface with the Ag14 (or Ag14S) cubic core.
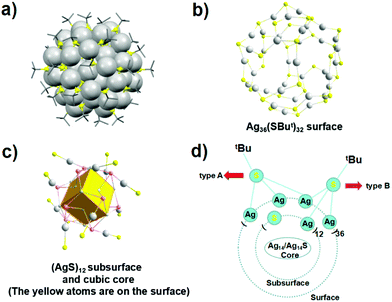 | ||
| Fig. 3 X-ray structure of [Ag62S13(StBu)32]2+. (a) The similar frameworks contained in the Ag-QD, Ag-inter, and Ag-NC. (b) Ag36(StBu)32 shell. (c) Bonding mode of core silver atoms (gray) and sulfur atoms (pink) linked with the shell sulfur atoms (yellow). (d) Two types of ligands. Adapted with permission from ref. 66. Copyright 2016, American Chemical Society. | ||
Recently, Wu et al. reported a new all thiol-protected Ag46S7(2,4-DMBT)24 nanocluster with FCC structure.67 The structure of the Ag46 NC can be viewed as a 38-Ag atom kernel with a sulfur atom in the center, capped by surface motifs including two Ag(2,4-DMBT)3, six Ag(2,4-DMBT)2, six 2,4-DMBT, and six sulfido units. The Ag38 kernel can further be divided into an octahedral Ag6 core and a 32-silver-atom tetradecahedron shell (Fig. 4). This tetradecahedron is made up of eight hexagons and six tetragons.
 | ||
| Fig. 4 (a) The central Ag6 octahedron with a central sulfur; (b and c) the Ag6S@Ag32 kernel; and (d) the crystal facets of the FCC Ag38 kernel. S yellow, Ag other colors. Adapted with permission from ref. 67. Copyright 2018, John Wiley & Sons, Inc. | ||
Phosphine was usually used as an auxiliary ligand with thiol or alkyl to produce some novel metal NCs. Introducing phosphine not only enhances the yield and stability of Ag NCs but it also assists with the growth of high-quality single crystals. Thiol and phosphine ligands have excellent compatibility in protecting Ag NCs. Zheng et al. reported the single crystal structure of a mixed ligand protected Ag14 cluster in 2012.68 As the smallest FCC-type Ag NC reported to date, the Ag14(SC6H3F2)12(PPh3)8 cluster contains an octahedral Ag64+ core, which is encapsulated by eight cubically arranged Ag(SC6H3F2)2PPh3 tetrahedrons that share one corner between them (Fig. 5a). Another important characteristic of this cluster is that all the thiolate ligands bind to three Ag atoms and no staple motifs are found. Later, a helical FCC structure was observed in the Ag23(PPh3)8(SC2H4Ph)18 nanocluster (Fig. 5b).69 Ag23 has a bioctahedral Ag11 core, which is viewed as two Ag6 building blocks fused together by vertex sharing. Due to a slight distortion in the vertex-sharing Ag11 core, Ag23 has a chiral structure.
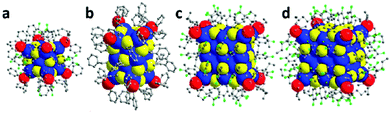 | ||
| Fig. 5 X-ray structures of Ag cubes: Ag14(SC6H3F2)12(PPh3)8, Ag23(PPh3)8(SC2H4Ph)18, Ag38(SPhF2)26(PnBu3)8, and Ag63(SPhF2)36(PnBu3)8. Adapted with permission from ref. 62. Copyright 2019, Royal Society of Chemistry. | ||
When fusing four of such simple Ag14 FCC cubes together via face sharing, a square like Ag38(SPhF2)26(PnBu3)8 metal framework was obtained (Fig. 5c).70 Further aggregation of another four Ag14 FCC cubes or one more square-like Ag38(SPhF2)26(PnBu3)8 gives rise to the cubic-structured Ag63(SPhF2)36(PnBu3)8 (Fig. 5d).71
The Zang group prepared a FCC Ag14 NC protected by face-capping 1,2-dithiolate-o-carborane ligands. Site-specific surface modification of the Ag14 NC with pyridyl-type ligands affords highly thermostable NCs.72 Moreover, by using a progressively optimized ligand-bridging approach, various 1D-to-3D silver cluster-assembled materials are predesigned and obtained (Fig. 6). This strategy not only greatly improves the stability but also modulates the emission properties of the target materials.
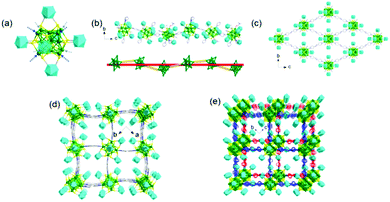 | ||
| Fig. 6 Structural represention of the (a) 1,2-dithiolate-o-carborane-capped Ag14 NC; (b) 1D helix; (c) 2D grid network; (d) 3D porous framework and (e) 2-fold interpenetrated porous 3D frameworks. Adapted with permission from ref. 72. Copyright 2018, American Chemical Society. | ||
Another series with FCC kernels pertains to the box-like Ag46(2,5-DMBT)24(PPh3)8 and Ag67(2,4-DMBT)32(PPh3)8 NCs.73–75 The crystal structure of Ag67(2,4-DMBT)32(PPh3)8 was reported by Alhilaly et al.75 The Ag67 structure consists of a Ag23 kernel protected by a layer of Ag44S32P8 arranged in the shape of a box (Fig. 7). Unlike the common Ag13 icosahedron geometry, the Ag23 kernel was formed through a cuboctahedron sharing opposite square faces with two Ag8 crowns and then capped by two silver atoms at the open crown positions. This crowning of the Ag13 cuboctahedron leads to the box-shape growth of the Ag67 cluster. The entire cluster is stabilized by 8 AgS3P motifs and 8 bridging thiolates. Of note, after removing a block of the Ag21(SR)8 unit from Ag67(SR)32(PPh3)8, a box-like structure of Ag46(SR)24(PPh3)8 was predicted. Experimentally, the Ag46(2,5-DMBT)24(PPh3)8 NC has recently been synthesized and crystallized (Fig. 8).73,74 Ag46 has a Ag14 core with a FCC structure which is protected by a Ag32S24P8 shell.
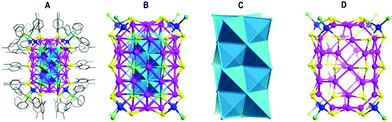 | ||
| Fig. 7 (A) Total structure of Ag67(SPhMe2)32(PPh3)8. (B) The structure of Ag67S32P8 obtained by disconnecting carbon atoms in A. (C) Ag23 metal core. (D) The structure of the NC without the Ag23 metal core, i.e., Ag44(SPhMe2)32(PPh3)8. Adapted with permission from ref. 75. Copyright 2017, American Chemical Society. | ||
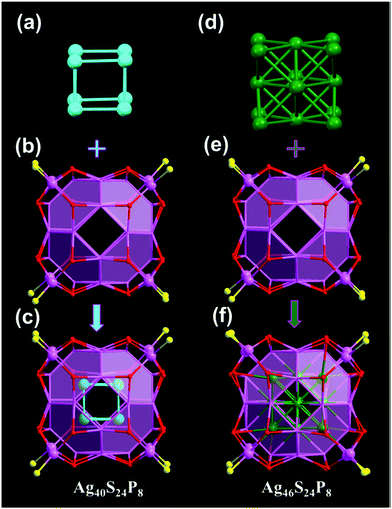 | ||
| Fig. 8 Core, shell and framework of Ag40(2,4-DMBT)24(PPh3)8 and Ag46(2,5-DMBT)24(PPh3)8. Adapted with permission from ref. 73. Copyright 2018, American Chemical Society. | ||
The Ag40(2,4-DMBT)24(PPh3)8 and Ag46(2,5-DMBT)24(PPh3)8 NCs share the same shell of Ag32S24P8, while the metal cores are arranged into different types (Fig. 8).73,74 In contrast to the Ag46 nanocluster, Ag40 presents a newly found loose Ag8 core with a simple-cubic structure. Interestingly, a cavity exists between the Ag8 core and the inner layer of the protecting shell, but no such cavity exists in Ag46. In Zhu's work, they successfully transformed Ag40 to Ag46via a ligand exchange strategy.73 Notably, the intermediate Ag nanocluster, [Ag43(2,5-DMBT)25(PPh3)4], was also obtained.73 The framework of Ag43 has a two-shelled Ag12@Ag20 core, which is protected by four kinds of units, including Ag2S5P, Ag4S8P, Ag2S4P and Ag3S6P, and two S bridge bonds were found on the surface of this nanocluster.
Recently, Zheng and coworkers reported a detailed structural and spectroscopic characterization of Ag40(DMBT)24(PPh3)8H12 (Ag40H12).76 In contrast to the Ag40 NC, the metal framework of Ag40H12 also consists of identical Ag8@Ag32S24P8. Based on a detailed analysis of the structural features and 1H and 2H NMR spectra, the positions of the 12 hydrides were determined to be residing on the 12 edges of the cubic core.
Among thiolate protected metal NCs, chiral thiolate protected metal NCs with different electronic configurations are of great importance in nanoscience and nanotechnology owing to their chiro-optical properties and applications in asymmetric drugs, sensors, and catalysts. For example, a range of structures and properties of chiral thiolate protected Au NCs including Au20(SPhtBu)16,77 Au28(SPhtBu)20,78 Au38(SCH2CH2Ph)24,21 Au102(p-MBA)44,22 and Au133(SPhtBu)5279 have recently been studied.
We herein highlight recent findings on chiral thiolate-protected Ag NC clusters. A few chiral thiolate protected Ag NCs such as Ag16(dppe)4(SPhF2)14 (dppe: 1,2-bis(diphenylphosphino)ethane),80 Ag32(dppe)5(SPhCF3)24,80 Ag32(dppm)5(SAdm)13Cl8,82 Ag45(dppm)4(StBu)16Br12,82 and Ag33(SCH2CH2Ph)24(PPh3)483 have been characterized by SCXRD, in which all the surface organic ligands are achiral.
In 2003, Zheng et al. reported two chiral AgNCs, Ag16(dppe)4(SPhF2)14 and Ag32(dppe)5(SPhCF3)24,80 which are protected by achiral diphosphine and thiolate ligands. The clusters have core–shell structures with a multinuclear Ag unit encapsulated in a shell containing a Ag(I)–thiolate–diphosphine complex. In Ag16(dppe)4(SPhCF2)14, a Ag8 core is encapsulated in a shell of Ag8(dppe)4(SPhCF2)14, while the structure of Ag32(dppe)5(SPhCF3)24 shows that it possesses a Ag22 core protected by one Ag6(dppe)3(SPhCF3)12, two Ag2(dppe)(SPhCF3)4 and four (SPhCF3) units. As shown in Fig. 9, both Ag16 and Ag32 conform to C2 symmetry, and their chirality is caused by the asymmetric arrangements of the tetrahedral [AgS3P] coordination units on the surface.
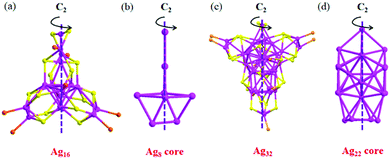 | ||
| Fig. 9 Core and framework of Ag16(dppe)4(SPhF2)14 and Ag32(dppe)5(SPhCF3)24 NCs. Adapted with permission from ref. 27. Copyright 2018, American Chemical Society. | ||
As a further development, the synthetic recipe of Ag16 and Ag32 was modified by replacing dppe with dppp (1,3-bis(diphenyphosphino)propane). A pair of optically pure enantiomers Ag78(dppp)6(SPhCF3)42 (R/S-Ag78) were synthesized.81 The molecular architecture of the R/S-Ag78 NCs can be described as a Ag@Ag21@Ag44@Ag12(dppp)6(SPhCF3)24(SPhCF3)18 core–shell structure (Fig. 10). The Ag@Ag21 kernel displays D3 symmetry and can be described as three mutually inter penetrating icosahedra. The 3-fold axis and three 2-fold axes pass through the center Ag atoms. The Ag@Ag21 kernel is encapsulated in a Ag44 shell whose structure can be rationalized. The predetermined chirality in the Ag78 cluster originates in the chiral arrangement of the surface-protecting units.
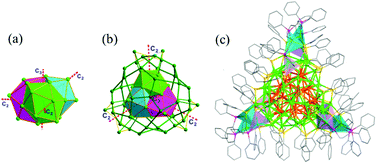 | ||
| Fig. 10 Core, shell and overall structures of Ag78(dppp)6(SPhCF3)42. Adapted with permission from ref. 82. Copyright 2017, American Chemical Society. | ||
Two multi-ligand-protected chiral Ag NCs, Ag32(dppm)5(SAdm)13Cl8 and Ag45(dppm)4(StBu)16Br12, have been synthesized and structurally characterized (Fig. 11).82 Ag32 possesses an achiral Ag13 icosahedral kernel, and Ag45 also has an achiral core of 23 Ag atoms, but the cores are protected by chiral shells, Ag19S13Cl8P10 and Ag22S16P8Br12, respectively. It is interesting to note that the coplanar fusion of Ag13 units into Ag23 constitutes the metal core of the Ag45 nanocluster.
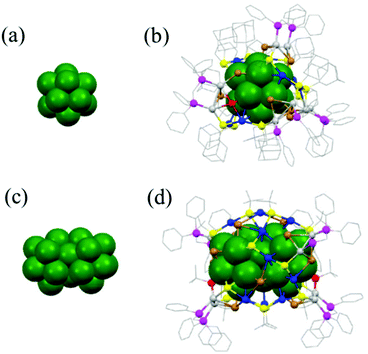 | ||
| Fig. 11 Core and overall structures of Ag32(dppm)5(SAdm)13Cl8 and Ag45(dppm)4(StBu)16Br12 NCs. Adapted with permission from ref. 81. Copyright 2017, Royal Society of Chemistry. | ||
Remarkably, the asymmetric distribution of the three types of ligands (thiolate, phosphine, and halogen) on the cluster surface induces chirality that can transfer from the ligand shell to the inner metal core, thereby resulting in an intrinsic chiral structure.
Very recently, Chen et al. discovered a new chiral crystal structure of the Ag33(SCH2CH2Ph)24(PPh3)4 NC (Fig. 12).83 The Ag33 nanocluster contains a Ag13 icosahedral core and a chiral shell of Ag20S24P4 composed of –SR–Ag–SR– motifs and –Ag–P terminals. Pd(PPh3)4 played a crucial role in the formation of Ag33 but not by replacing silver atoms to form alloys.
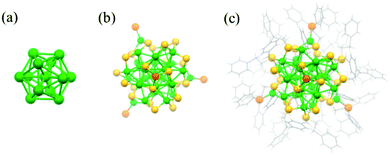 | ||
| Fig. 12 Core, shell and framework of Ag33(SCH2CH2Ph)24(PPh3)4. Adapted with permission from ref. 83. Copyright 2019, American Chemical Society. | ||
The shell-by-shell mode is the most common growth mode for nanoparticles, as reflected in different-size spherical nanoparticles. This growth mode allows the isotropic three-dimensional expansion of the particle size and is expected to apply to the structures of giant nanoclusters. Indeed, this mode has been observed in many metal NCs.6,61 The Zheng and Jin groups solved the crystal structures of a series of plasmonic twinned silver nanoclusters, such as Ag136(SPhtBu)64Cl3Ag0.45,84 Ag141X12(SAdm)40 (X = Cl, Br, I and SAdm = 1-adamantanethiolate),85 Ag146Br2(SPhiPr)80,86 Ag206(SCy)68F2Cl2 (Cy: cyclohexanethiolate),87 and Ag374(SPhtBu)113Br2Cl2,84 which can be described as 5-fold twinned cores enclosed within related structurally distinctive Ag–SR complex shells (Fig. 13).
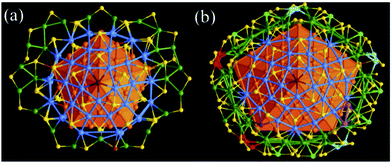 | ||
| Fig. 13 (a) The shell of Ag136 with the bowl-like half J73 related [Ag30(SPhtBu)15Cl] caps highlighted in blue; and (b) the shell of Ag374 with key structure elements highlighted in different colors. Adapted with permission from ref. 84. Copyright 2016 Nature Publishing Group. | ||
These large nanoclusters follow the shell-by-shell growth mode, in which a Ag7 or Ag19 innermost kernel and corresponding growth modes have been observed. For the Ag7-kernel based nanoclusters, Ag146Br2(SPhiPr)80 can be dissected into Ag7 (kernel)@Ag32 (1st shell)@Ag12 (2nd shell)Ag95Br2(SPhiPr)8086 and Ag206(SR)68F2Cl2 follows a Ag7 (kernel)@Ag32 (1st shell)@Ag77 (2nd shell)@Ag90(SCy)68F2Cl2 (motif shell) configuration.87
For the Ag19-kernel based nanoclusters, Ag141(SAdm)40(Cl/Br/I)12 displays a Ag19 (kernel)@Ag52 (1st shell)@Ag70(SAdm)68F2Cl2 (motif shell) three-shell configuration85 and Ag210/211(SPhiPr)71(PPh3)5/6Cl exhibits a Ag19 (kernel)@Ag52 (1st shell)@Ag45 (2nd shell)@Ag89(SPhiPr)71Cl&(Ag-PPh3)5/6 (motif shell) four-shell configuration.88
Unlike the shell-by-shell growth mode in larger metal NCs, many smaller structures have been found to be assembled from small polyhedrons.6 For instance, the structure of Ag22(dppe)4(2,5-DMBT)12Cl4 exhibits a Ag10 kernel, which is composed of two Ag5 units having distorted trigonal bipyramidal geometry (Fig. 14). The Ag10 core is protected by a Ag12(dppe)4(2,5-DMBT)12Cl4 shell, which is formed by four Ag2SP2Cl and four AgS2 staple motifs.89 The Ag22 cluster exhibits crystallization-enhanced PL.
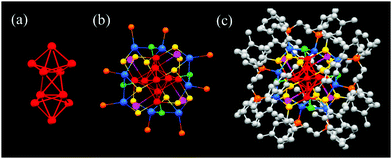 | ||
| Fig. 14 Core, shell and overall structures of Ag22(dppe)4(2,5-DMBT)12Cl4. Adapted with permission from ref. 89. Copyright 2019, American Chemical Society. | ||
3.2. Alkynyl-protected Ag CNs
Beyond thiolate ligands, the alkynyl ligand has been employed for the synthesis of a number of coinage metal clusters.90–92 Recently, some alkynyl-protected Ag NCs have been identified, and they exhibit good stability and crystallizability. For example, Zhang et al. and Xie et al. exhibited the structures of two all alkynyl-protected Ag NCs, Ag74(PhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)44 and Ag51(tBuC
C)44 and Ag51(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)32, respectively.35,36 Their crystal structures both contain three-shell structures. The crystal structure of Ag74 possesses a Ag4 tetrahedron inner core, which is surrounded by the second Ag22 shell (Fig. 15).35 The outermost shell consists of 48 Ag atoms that are enclosed into 12 pentagons and 56 triangles. However, when the phenylacetylene is replaced by tert-butylethynide, the Ag51 NC is prepared. The crystal structure of Ag51 displays a Ag@Ag14@Ag36 three shell structure, which is capped by 32 tert-butylethynide ligands on the surface (Fig. 16).36
C)32, respectively.35,36 Their crystal structures both contain three-shell structures. The crystal structure of Ag74 possesses a Ag4 tetrahedron inner core, which is surrounded by the second Ag22 shell (Fig. 15).35 The outermost shell consists of 48 Ag atoms that are enclosed into 12 pentagons and 56 triangles. However, when the phenylacetylene is replaced by tert-butylethynide, the Ag51 NC is prepared. The crystal structure of Ag51 displays a Ag@Ag14@Ag36 three shell structure, which is capped by 32 tert-butylethynide ligands on the surface (Fig. 16).36
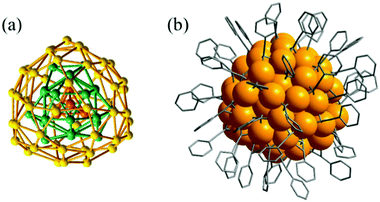 | ||
Fig. 15 (a) The Ag74 core; and (b) overall structure of Ag74(PhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)44. Adapted with permission from ref. 35. Copyright 2017, American Chemical Society. C)44. Adapted with permission from ref. 35. Copyright 2017, American Chemical Society. | ||
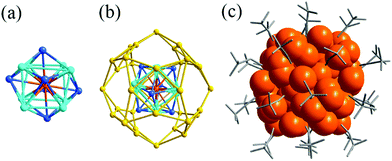 | ||
Fig. 16 (a) The Ag@Ag14 core; (b) the structure of the Ag@Ag14@Ag36 shell; and (c) overall structure of Ag51(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)32. Adapted with permission from ref. 36. Copyright 2018, Royal Society of Chemistry. C)32. Adapted with permission from ref. 36. Copyright 2018, Royal Society of Chemistry. | ||
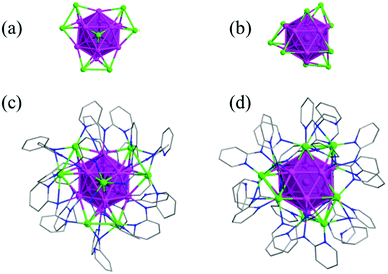
Aside from all alkynyl-protected Ag NCs, alkynyl and auxiliary ligand co-protected Ag NCs have been obtained. In 2017, Wang et al. reported two Ag NCs, Ag19(dppm)3(PhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)14+ and Ag25(dpppe)3(MeOPhC
C)14+ and Ag25(dpppe)3(MeOPhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)20 (dpppe: 1,5-bis(diphenylphosphino)pentane), which are protected by both alkynyl and phosphine ligands (Fig. 17).93 They have D3h symmetry with a centered anticuboctahedral Ag13 kernel extended by three Ag2 motifs and three tetrahedral Ag4 motifs, respectively. Later, they obtained two Ag NCs containing the protection of thiacalixarenes, Ag34(BTCA)3(tBuC
C)20 (dpppe: 1,5-bis(diphenylphosphino)pentane), which are protected by both alkynyl and phosphine ligands (Fig. 17).93 They have D3h symmetry with a centered anticuboctahedral Ag13 kernel extended by three Ag2 motifs and three tetrahedral Ag4 motifs, respectively. Later, they obtained two Ag NCs containing the protection of thiacalixarenes, Ag34(BTCA)3(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)9(tfa)4(CH3OH)3 and Ag35(H2BTCA)2(BTCA)(tBuC
C)9(tfa)4(CH3OH)3 and Ag35(H2BTCA)2(BTCA)(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)16 (H4BTCA: 4-tert-butylthiacalix[4]arene and tfa: trifluoroacetate).94,95 The Ag34 and Ag35 NCs have a centered icosahedral Ag@Ag12 kernel that is surrounded by 21 and 22 peripheral silver atoms (Fig. 18), respectively. Surrounding protection for Ag35 is provided by three thiacalixarene ligands and 16 alkynyl ligands, while Ag34 is protected by four kinds of ligands, including three BTCA, nine alkynyl ligands, four tfa, and three methanol solvent ligands.
C)16 (H4BTCA: 4-tert-butylthiacalix[4]arene and tfa: trifluoroacetate).94,95 The Ag34 and Ag35 NCs have a centered icosahedral Ag@Ag12 kernel that is surrounded by 21 and 22 peripheral silver atoms (Fig. 18), respectively. Surrounding protection for Ag35 is provided by three thiacalixarene ligands and 16 alkynyl ligands, while Ag34 is protected by four kinds of ligands, including three BTCA, nine alkynyl ligands, four tfa, and three methanol solvent ligands.
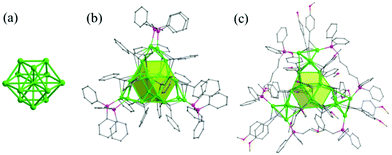 | ||
Fig. 17 (a) The Ag13 anticuboctahedron kernel; and (b and c) overall structures of Ag19(dppm)3(PhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)14 and Ag25(dpppe)3(MeOPhC C)14 and Ag25(dpppe)3(MeOPhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)20. Adapted with permission from ref. 93. Copyright 2017, Royal Society of Chemistry. C)20. Adapted with permission from ref. 93. Copyright 2017, Royal Society of Chemistry. | ||
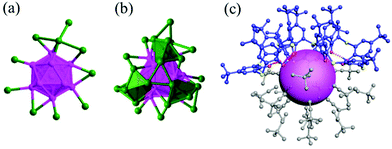 | ||
Fig. 18 X-ray structure of Ag35(H2BTCA)2(BTCA)(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)16. (a) Position of 10 peripheral Ag atoms (green) held by thiacalixarene ligands onto the Ag13 core (pink); (b) position of 12 peripheral Ag atoms (green, triangular prisms) capped by alkynyl ligands; and (c) side views of the position of surface ligands with respect to the Ag35 core. Adapted with permission from ref. 95. Copyright 2015, American Association for the Advancement of Science. C)16. (a) Position of 10 peripheral Ag atoms (green) held by thiacalixarene ligands onto the Ag13 core (pink); (b) position of 12 peripheral Ag atoms (green, triangular prisms) capped by alkynyl ligands; and (c) side views of the position of surface ligands with respect to the Ag35 core. Adapted with permission from ref. 95. Copyright 2015, American Association for the Advancement of Science. | ||
Very recently, Wang et al. solved the crystal structure of a large alkynyl and halide protected silver NC, (C7H17ClN)3[Ag112Cl6(ArC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)51].96 The cluster exhibits a four concentric core–shell structure Ag13@Ag42@Ag48@Ag9, and four types of alkynyl–Ag binding modes are observed. Chloride is found to be critical for the stabilization and formation of the Ag NC. Another interesting case is Ag48(tBuC
C)51].96 The cluster exhibits a four concentric core–shell structure Ag13@Ag42@Ag48@Ag9, and four types of alkynyl–Ag binding modes are observed. Chloride is found to be critical for the stabilization and formation of the Ag NC. Another interesting case is Ag48(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)20(CrO4)7, which is co-capped by CrO42−and tBuC
C)20(CrO4)7, which is co-capped by CrO42−and tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C− ligands.97 The pseudo-5-fold symmetric metal skeleton of Ag48 shows a core–shell structure composed of a Ag23 cylinder encircled by an outer Ag25 shell (Fig. 19). The involvement of both organic and inorganic protection is a new path for synthesizing Ag NCs and controlling the formation and structure.
C− ligands.97 The pseudo-5-fold symmetric metal skeleton of Ag48 shows a core–shell structure composed of a Ag23 cylinder encircled by an outer Ag25 shell (Fig. 19). The involvement of both organic and inorganic protection is a new path for synthesizing Ag NCs and controlling the formation and structure.
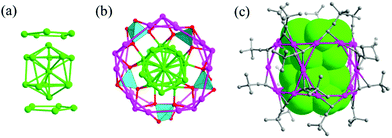 | ||
Fig. 19 X-ray structure of Ag48(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)20(CrO4)7. (a) Formation of the Ag25 shell by capping two silver pentagons on the Ag13 Ino decahedron on the top and bottom; (b) top view of the coordination of five equatorial CrO42− anions linking the Ag23 cylinder and Ag25 shell; and (c) side view of the Ag23 cylinder encircled by the outer [Ag25(tBuC C)20(CrO4)7. (a) Formation of the Ag25 shell by capping two silver pentagons on the Ag13 Ino decahedron on the top and bottom; (b) top view of the coordination of five equatorial CrO42− anions linking the Ag23 cylinder and Ag25 shell; and (c) side view of the Ag23 cylinder encircled by the outer [Ag25(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)20] shell. Adapted with permission from ref. 97. Copyright 2019, American Chemical Society. C)20] shell. Adapted with permission from ref. 97. Copyright 2019, American Chemical Society. | ||
Another interesting case is Ag48(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)20(CrO4)7, which is co-capped by CrO42−and tBuC
C)20(CrO4)7, which is co-capped by CrO42−and tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C− ligands.97 The pseudo-5-fold symmetric metal skeleton of Ag48 shows a core–shell structure composed of a Ag23 cylinder encircled by an outer Ag25 shell (Fig. 19). The involvement of both organic and inorganic protection is a new path for synthesizing Ag NCs and controlling the formation and structure.
C− ligands.97 The pseudo-5-fold symmetric metal skeleton of Ag48 shows a core–shell structure composed of a Ag23 cylinder encircled by an outer Ag25 shell (Fig. 19). The involvement of both organic and inorganic protection is a new path for synthesizing Ag NCs and controlling the formation and structure.
3.3. Other ligand-protected Ag NCs
It is an effective strategy to obtain new functional metal nanoclusters by using ligands beyond the conventional ones. For example, Liu et al. successfully synthesized and determined the structures of Ag20{E2P(OR)2}12 and Ag21{E2P(OiPr)2}12 (E = S, Se).98–100 The Ag20 and Ag21 NCs have a Ag-centered Ag13 icosahedral kernel with 7 and 8 capping Ag atoms and 12 dichalcogeno ligands.Recently, Wang et al. reported two homoleptic amido-protected Ag NCs Ag21(dpa)12 and Ag22(dpa)12 (dpa: dipyridylamido).101 The Ag21 and Ag22 NCs consist of a centered-icosahedron Ag13 core wrapped by 12 dpa ligands (Fig. 20). The flexible arrangement of the N donors in dpa facilitates the solvent-triggered reversible interconversion between Ag21 and Ag22 due to their very different solubility.
 | ||
| Fig. 20 View of the Ag21 kernel (a) and the Ag22 kernel (b); and total structures of Ag21(dpa)12 (c) and Ag22(dpa)12 (d) showing the Ag13 polyhedron. Adapted with permission from ref. 101. Copyright 2019, Nature Publishing Group. | ||
The Wang group successfully prepared and characterized a Ag NC protected by phosphine and halide, Ag15(Ntriphos)4Cl4 (N-triphos: tris((diphenylphosphino)methyl)amine).102 The Ag15 cluster has a hexacapped body-centered cubic framework which is consolidated by four tripodal N-triphos ligands, in which one Ag atom occupies the center of the Ag8 cube, while the six square faces of this Ag8 cube are capped respectively by one Ag atom (Fig. 21). Our group recently reported three oxometalate and phosphine ligand co-protected Ag NCs, Ag28(dppb)6(MoO4)4 (dppb: 1,4-bis(diphenylphosphino)butane), Ag28(dppb)6(WO4)4 and Ag32(dppb)12(MoO4)4(NO3)4.103 Each cluster comprises a double shell Ag4@Ag24 core covered by 4 oxometalates (Fig. 22). Two similar Ag28 cores of our clusters are observed in the Cu12Ag28(2,4-DCBT)24 and Cd12Ag32(SePh)36 clusters,104,105 however with vividly different metal and ligand compositions, electronic charges, and surface structures.
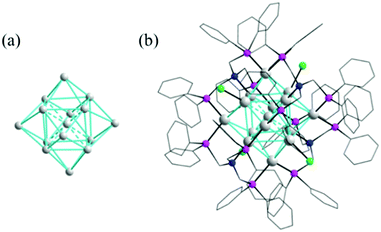 | ||
| Fig. 21 (a) The core of Ag15 in a hexacapped bcc arrangement; and (b) X-ray structure of [Ag15(Ntriphos)4(Cl4)]3+. Adapted with permission from ref. 102. Copyright 2017, Royal Society of Chemistry. | ||
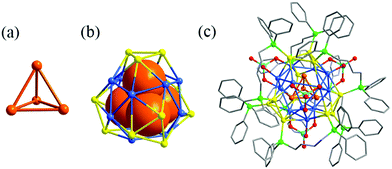 | ||
| Fig. 22 (a) View of the Ag4 inner core; (b) the structure of the two-shell Ag4@Ag24; and (c) total structure of Ag28(dppb)6(MoO4)4. Adapted with permission from ref. 103. Copyright 2020, Royal Society of Chemistry. | ||
In 2019, the Suzuki group prepared a unique ultrastable Ag NC with a C-shaped {Si2W18} building unit (Fig. 23).106 The Ag27(Si2W18O66)3 cluster was assigned to five octahedral {Ag6} clusters and three bridging Ag atoms, and it was surrounded by C-shaped {Si2W18} through direct Ag–O–W bonds. Recently, Sun et al. also reported a series of silver NC based POMs, such as Ag10@(Mo7O26)2@Ag70(MoO4)2(SiPr)36(CF3SO3)16(DMF)6, Ag10@(MoO4)7@Ag60(SPhtBu)33(mbc)18(DMF)(H2O)2, and Ag6@(MoO4)7@Ag56(MoO4)2(SiPr)28(CF3SO3)14(DMF)4.42,107–109 These large Ag NCs follow the shell-by-shell growth mode, in which a Ag6 or Ag10 innermost kernel and corresponding growth modes have been observed. For example, in the innermost region of Ag10@(Mo7O26)2@Ag70(MoO4)2(SiPr)36(CF3SO3)16(DMF)6, an unusual FCC-structured Ag10 nanocluster is locked by a pair of Mo7O2610− anions to form an inner Ag10@(Mo7O26)2 core which acts as a template to support the outer Ag70 nanocluster to form a final three-shell Ag10@(Mo7O26)2@Ag70 nanocluster (Fig. 24).
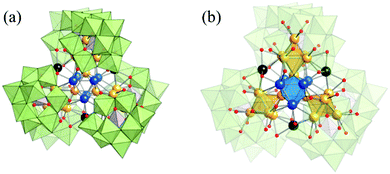 | ||
| Fig. 23 X-ray structure of Ag27(Si2W18O66)3. Adapted with permission from ref. 106. Copyright 2019, American Chemical Society. | ||
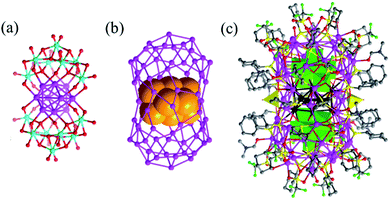 | ||
| Fig. 24 (a) The Ag10 bioctahedron locked by a pair of Mo7O2610− anions; (b) the Ag10 bioctahedron (claybank space-filling balls) residing in the Ag70 shell; and (c) overall structure of Ag10@(Mo7O26)2@Ag70(MoO4)2(SiPr)36(CF3SO3)16(DMF)6. Adapted with permission from ref. 107. Copyright 2019, Royal Society of Chemistry. | ||
In contrast to other ligand protected Ag NCs, DNA templated Ag NCs (DNA-Ag NCs) have received much interest, attributable to the photophysical properties including high quantum yield, excellent brightness, photostability, and tunable emission colors from visible to near IR.110–118 The structures and optical properties of DNA-Ag NCs are regulated by the sequences or secondary structures of DNA scaffolds that possess different binding affinities to Ag NCs. The first example of DNA-Ag NCs was discovered by Dickson and co-workers in 2004,119 where a 12-base scaffold of 5′-AGGTCGCCGCCC-3′ was employed as the template to direct the assembly of silver ions, and then reduced by NaBH4 to form Ag NCs in aqueous solution at room temperature. By choosing DNA templates with various sequences and lengths, many types of fluorescent DNA-Ag NCs were prepared.110–123 Mass spectrometry reveals that the sequence and length of DNA scaffolds could play an important role in determining the size of Ag NCs. In addition to the size, the DNA conformation and the oxidation state of Ag NCs are other factors that modulate the structures and optical properties.
The secondary structure of DNA scaffolds has also important influences on the structure and optical properties of DNA-Ag NCs. Secondary structures such as hairpin, i-motif and G-quadruplex have been made in creating DNA-Ag NCs. For example, DNA-Ag NCs can be prepared by using hairpins with a C-loop of 3 to 12 cytosines, which contained different numbers of silver atoms and showed different fluorescence.120–122
Li et al. synthesized fluorescent DNA-Ag NCs with i-motif DNA, and such NCs display an emission wavelength range over green to NIR.123 By using a G-quadruplex DNA sequence, Wang et al. synthesized dual-emissive DNA-Ag NCs possessing high thermo-stability.124 To better understand the properties and applications of DNA-Ag NCs, readers are also recommended to refer to recent reviews.125–134
4. Optical properties of Ag NCs
4.1. UV-vis absorption
Surface plasmon resonance (SPR) is the most prominent feature in the UV-vis absorption spectra of Ag NPs due to their distinct optical absorption.135–138 The SPR peak of Ag NPs is typically located at about 400 nm. In contrast, Ag NCs generally show several distinct absorption peaks in the UV-vis region. The optical absorptions of Ag NCs and Ag NPs are distinctively different, with different origins and different peak locations. Such data can be used to confirm the successful synthesis of Ag NCs and the transformation of small Ag NCs into large plasmonic Ag NPs.The surface ligands and cluster size can affect the optical absorption of Ag NCs (Table S2, ESI†). For example, thiol-protected Ag2–8 NCs showed discrete absorption peaks in their UV-vis absorption spectra.139 The UV-vis spectra of thiol-protected Ag NCs, such as Ag44(4-FTP)30, Ag55(PET)31, Ag75(PET)40, Ag114(PET)46, Ag152(PET)60, Ag202(BBS)70, Ag423(PET)105, and Ag530(PET)100, show multiple features up to Ag114 and, from Ag152 onwards, only one absorption peak at 460 nm (Fig. 25).
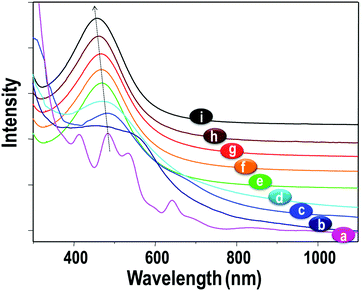 | ||
| Fig. 25 UV-vis absorption spectra of thiol-protected Ag44 [a], Ag55 [b], Ag∼75 [c], Ag∼114 [d], Ag152 [e], Ag∼202 [f], Ag∼423 [g], Ag∼530 [h] and Ag NPs [i]. Reproduced with permission from ref. 140. Copyright 2014, Royal Society of Chemistry. | ||
The change of the protecting ligands and cluster size, which affect the behavior in the excited state, results in an alteration of the electronic transition.140 Insight into the modulation of the PL properties and the relaxation from the excited state is provided in the following sections.
4.2. Photoluminescence
PL is amongst the most intriguing and fascinating properties of nanomaterials due to the scope in diverse applications. Ag NCs excited from the ground state release extra energy before returning back to the ground state, which gives rise to PL. However, Ag NCs normally display low quantum yield (QY), and some fundamental issues related to the PL properties of Ag NCs are still indistinct. The PL of Ag NCs can be dictated by the cluster size, protecting ligand, and heterometal atom. Moreover, the valence electron count, oxidation state of the metal, crystal structure, temperature, and pH are crucial to regulate the PL behavior.31,32The ligand effect on the PL of a Ag62 nanocluster template has been investigated. The tetracationic silver nanocluster [Ag62S13(StBu)32]4+ (Ag-QDs) has been reported with intense red emission at 613 nm (solution) and 621 nm (solid state).65 Later, Zhu et al. reported the crystal structure of [Ag62S12(StBu)32]2+ (Ag-NCs),64 which can be regarded as [Ag62S13(StBu)32]4+ lacking the innermost S ligand. The PL intensity of the Ag-NCs was much weaker than that of the Ag-QDs due to the difference in the valence electron count. The 4 free valence electrons in the Ag-NCs cause luminescence quenching as the LMCT process (ligand-to-metal charge transfer) gets hindered, while the Ag-QDs exhibit intense PL owing to the absence of free valence electrons. Interestingly, the Ag-QDs could be electrochemically reduced into Ag-NCs, via an intermediate NC, Ag-inter.66 Though the structural integrity of the parent Ag62 remains unaltered, the PL intensity of the Ag-inter displayed a 2-fold enhancement relative to the Ag-NCs, and it was still far below the intensity of the Ag-QDs (Fig. 26).
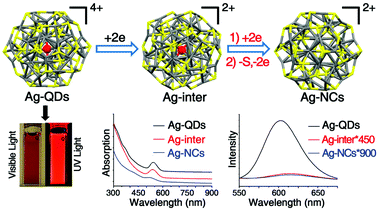 | ||
| Fig. 26 Schematic illustration of the structural transformation on the basis of a Ag62 nanocluster template from Ag-QDs to Ag-inter, and then to Ag-NCs; (bottom-left) digital photographs of Ag-QDs under visible and UV light; and (bottom-middle and -right) UV-vis and PL spectra of Ag-QDs, Ag-inter, and Ag-NCs. Reproduced with permission from ref. 31. Copyright 2019, Royal Society of Chemistry. | ||
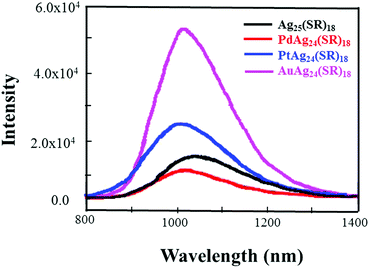 | ||
| Fig. 27 PL spectra of MAg24(SR)18 (M = Ag/Pd/Pt/Au) NCs in the crystal state. Reproduced with permission from ref. 148. Copyright 2017, American Chemical Society. | ||
The optical, electrochemical, and catalytic properties of metal NCs M1Ag24(SR)18 (M = Ag, Au, Pd, Pt) have been systematically characterized.23,144–148 For example, Bootharaju et al. demonstrated the PL property enhancement of Ag25(SPhMe2)18 doped with Pd or Au. Due to the stabilization of the charges in the LUMO of the alloy cluster akin to Au25−nAgn NCs, the luminescence of the Ag25 cluster is enhanced by a factor of 25 upon doping with gold atoms.144 Wu et al. investigated the PL property of M@Ag24(DMBT)18 (M = Ag, Pd, Pt, Au) in both crystal and solution phases.148 A blue shift of the PL with the doping of Ag25(DMBT)18 by Pd/Pt/Au heteroatoms is observed. The sequence of the PL intensity PdAg24(DMBT)18 < Ag25(DMBT)18 < PtAg24(DMBT)18 < AuAg24(DMBT)18 is exactly related to the electron affinity of the core atom (Fig. 28).
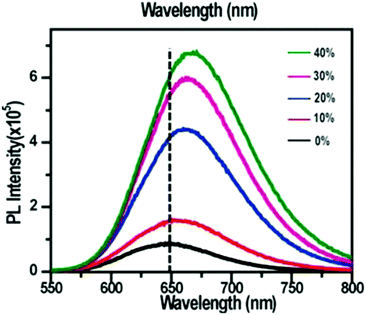 | ||
| Fig. 28 PL spectra of Ag29 and Au doped Ag29 NCs with different amounts (mmol%) of Au. The inset shows digital photographs under a UV lamp (365 nm). Adapted with permission from ref. 150. Copyright 2016, John Wiley & Sons, Inc. | ||
Ag29(S2R)12(PPh3)4 is another fluorescent Ag NC which has been studied in the context of doping. The PL characteristic of the Ag29(S2R)12(PPh3)4 NC has been tuned by doping such a NC with Au/Pt ions. The introduction of the Au or Pt heteroatoms improves the PL intensity relative to the homo-silver Ag29 NC. Bootharaju et al. reported the enhancement of the PL intensity in doped PtAg28(BDT)12(TPP)4 compared to homo-silver Ag29NCs.149 Soldan et al. prepared metal NCs Ag29−xAux(BDT)12(TPP)4 and demonstrated that the emission maxima are red shifted from 658 nm (10% Au) to 668 nm (40% Au) with an increase in the Au concentration (Fig. 28).150
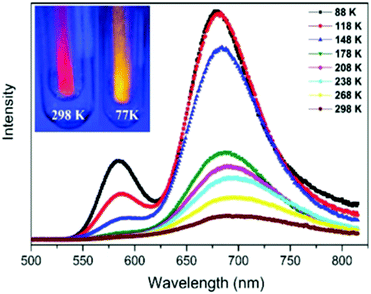 | ||
| Fig. 29 Effect of temperature on the PL of Ag18 in the solid state at 440 nm excitation. Adapted with permission from ref. 130. Copyright 2017, Royal Society of Chemistry. | ||
Solvents can also influence the PL properties of NCs, although to a lesser degree than that seen in metal complexes. Silver NCs, being composed of diverse functional groups in the surface ligand, often display solvent specific optical behavior. The nature of the solvent (polarity, protic or aprotic, coordinating or non-coordinating) controls the electronic properties, which eventually influence the emission of Ag NCs.92,101,148 Xie et al. evaluated the solvatochromism of the Ag51(tBuC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)32 NC.92 As the solvent polarity increased from less polar dichloromethane to highly polar methanol, the emission peaks of Ag51 are gradually red-shifted from 436 to 656 nm, exhibiting a remarkable bathochromic effect (Fig. 30).
C)32 NC.92 As the solvent polarity increased from less polar dichloromethane to highly polar methanol, the emission peaks of Ag51 are gradually red-shifted from 436 to 656 nm, exhibiting a remarkable bathochromic effect (Fig. 30).
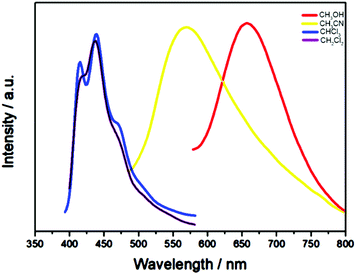 | ||
| Fig. 30 Emission bands of Ag51 in solvents of varying polarity, CH2Cl2 (purple line), CHCl3 (blue line), CH3CN (yellow line) and CH3OH (red line). Adapted with permission from ref. 92. Copyright 2018, Royal Society of Chemistry. | ||
Ag NCs feature tunable luminescence properties, photostability and a biocompatible nature, and have enthralled the scientific community for their applicability in versatile applications such as optoelectronics, catalysis, bio-sensing, and bio-imaging.6,7,12,13,29,31 More studies are required to explore new applications of Ag NCs. Further development of multi-photon excitation microscopy in Ag NCs calls for more efforts. More Ag NCs with visible range absorption still need to be developed in photovoltaic applications. Biological applications such as bio-sensing and bio-imaging demand hydrophilic Ag NCs with very high PLQY, while catalytic applications need structurally precise Ag NCs.
5. Conclusions
We have summarized significant advances in the field of Ag NCs such as synthesis, structure, and PL properties. For instance, a number of efficient synthetic strategies including direct reduction, chemical etching, and ligand exchange have been developed to produce Ag NCs. Some high-resolution analytical techniques have emerged as powerful tools to characterize Ag NCs, including UV-vis, PL, ESI-MS, SC-XRD, etc. Such techniques have been used to determine the chemical properties and crystal structures of Ag NCs. The crystal structures of Ag NCs eventually control the electronic transition and physical properties. The PL properties of Ag NCs are modulated by the valence state, electronic structure, functional groups and doping heteroatoms. Some parameters of the surroundings such as temperature and solvent also tune the PL properties of Ag NCs.However, although we have witnessed remarkable progress in the study of Ag NCs, some challenging issues still remain. For example, efficient synthetic strategies for high-purity Ag NCs in the aqueous phase are still lacking. Strategies for increasing the PL quantum yield and tuning the PL colors of nanoclusters are to be devised. Much more effort is needed to further explore promising luminescent probes for a wide spectrum of bio-imaging and bio-sensing applications.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
We gratefully acknowledge financial support from the National Natural Science Foundation of China (No. 21771071, 51672093 and 21925104).Notes and references
- G. Schmid, Large clusters and colloids. Metals in the embryonic state, Chem. Rev., 1992, 92, 1709–1727 CrossRef CAS.
- M.-C. Daniel and D. Astruc, Gold nanoparticles: assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology, Chem. Rev., 2004, 104, 293–346 CrossRef CAS PubMed.
- R. Jin, Quantum sized, thiolate-protected gold nanoclusters, Nanoscale, 2010, 2, 343–362 RSC.
- Y. Lu and W. Chen, Sub-nanometre sized metal clusters: from synthetic challenges to the unique property discoveries, Chem. Soc. Rev., 2012, 41, 3594–3623 RSC.
- H. Qian, M. Zhu, Z. Wu and R. Jin, Quantum sized gold nanoclusters with atomic precision, Acc. Chem. Res., 2012, 45, 1470–1479 CrossRef CAS PubMed.
- R. Jin, C. Zeng, M. Zhou and Y. Chen, Atomically precise colloidal metal nanoclusters and nanoparticles: fundamentals and opportunities, Chem. Rev., 2016, 116, 10346–10413 CrossRef CAS PubMed.
- I. Chakraborty and T. Pradeep, Atomically precise clusters of noble metals: emerging link between atoms and nanoparticles, Chem. Rev., 2017, 117, 8208–8271 CrossRef CAS PubMed.
- C. M. Aikens, Electronic structure of ligand-passivated gold and silver nanoclusters, J. Phys. Chem. Lett., 2011, 2, 99–104 CrossRef CAS PubMed.
- N. Goswami, K. Zheng and J. Xie, Bio-NCs-the marriage of ultrasmall metal nanoclusters with biomolecules, Nanoscale, 2014, 6, 13328–13347 RSC.
- A. Fernando, K. L. D. M. Weerawardene, N. V. Karimova and C. M. Aikens, Quantum mechanical studies of large metal, metal oxide, and metal chalcogenide nanoparticles and clusters, Chem. Rev., 2015, 115, 6112–6216 CrossRef CAS PubMed.
- S. A. Díaz, D. A. Hastman, I. L. Medintz and E. Oh, Understanding energy transfer with luminescent gold nanoclusters: a promising new transduction modality for biorelated applications, J. Mater. Chem. B, 2017, 5, 7907–7926 RSC.
- S. Maity, D. Bain and A. Patra, An overview on the current understanding of the photophysical properties of metal nanoclusters and their potential applications, Nanoscale, 2019, 11, 22685–22723 RSC.
- D. Bain, S. Maity and A. Patra, Opportunities and challenges in energy and electron transfer of nanocluster based hybrid materials and their sensing applications, Phys. Chem. Chem. Phys., 2019, 21, 5863–5881 RSC.
- Y. Du, H. Sheng, D. Astruc and M. Zhu, Atomically precise noble metal nanoclusters as efficient catalysts: a bridge between structure and properties, Chem. Rev., 2020, 120, 526–622 CrossRef CAS PubMed.
- R. Jin, Atomically precise metal nanoclusters: stable sizes and optical properties, Nanoscale, 2015, 7, 1549–1565 RSC.
- M. W. Heaven, A. Dass, P. S. White, K. M. Holt and R. W. Murray, Crystal structure of the gold nanoparticle [N(C8H17)4][Au25(SCH2CH2Ph)18], J. Am. Chem. Soc., 2008, 130, 3754–3755 CrossRef CAS PubMed.
- M. Zhu, C. M. Aikens, F. J. Hollander, G. C. Schatz and R. Jin, Correlating the crystal structure of a thiol-protected Au25 cluster and optical properties, J. Am. Chem. Soc., 2008, 130, 5883–5885 CrossRef CAS PubMed.
- M. Zhu, W. T. Eckenhoff, T. Pintauer and R. Jin, Conversion of anionic [Au25(SCH2CH2Ph)18]− cluster to charge neutral cluster via air oxidation, J. Phys. Chem. C, 2008, 112, 14221–14224 CrossRef CAS.
- N. K. Chaki, Y. Negishi, H. Tsunoyama, Y. Shichibu and T. Tsukuda, Ubiquitous 8 and 29 kDa gold: alkanethiolate cluster compounds: mass-spectrometric determination of molecular formulas and structural implications, J. Am. Chem. Soc., 2008, 130, 8608–8610 CrossRef CAS PubMed.
- H. Qian, Y. Zhu and R. Jin, Size-socusing synthesis, optical and electrochemical properties of monodisperse Au38(SC2H4Ph)24 nanoclusters, ACS Nano, 2009, 3, 3795–3803 CrossRef CAS PubMed.
- H. Qian, W. T. Eckenhoff, Y. Zhu, T. Pintauer and R. Jin, Total structure determination of thiolate-protected Au38 nanoparticles, J. Am. Chem. Soc., 2010, 132, 8280–8281 CrossRef CAS PubMed.
- P. D. Jadzinsky, G. Calero, C. J. Ackerson, D. A. Bushnell and R. D. Kornberg, Structure of a thiol monolayer-protected gold nanoparticle at 1.1 Å resolution, Science, 2007, 318, 430–433 CrossRef CAS PubMed.
- C. P. Joshi, M. S. Bootharaju, M. J. Alhilaly and O. M. Bakr, [Ag25(SR)18]−: The “golden” silver nanoparticle, J. Am. Chem. Soc., 2015, 137, 11578–11581 CrossRef CAS PubMed.
- L. G. AbdulHalim, M. S. Bootharaju, Q. Tang, S. Del Gobbo, R. G. AbdulHalim, M. Eddaoudi, D.-E. Jiang and O. M. Bakr, Ag29(BDT)12(TPP)4: a tetravalent nanocluster, J. Am. Chem. Soc., 2015, 137, 11970–11975 CrossRef CAS PubMed.
- A. Desireddy, B. E. Conn, J. Guo, B. Yoon, R. N. Barnett, B. M. Monahan, K. Kirschbaum, W. P. Griffith, R. L. Whetten, U. Landman and T. P. Bigioni, Ultrastable silver nanoparticles, Nature, 2013, 501, 399–402 CrossRef CAS PubMed.
- H. Yang, Y. Wang, H. Huang, L. Gell, L. Lehtovaara, S. Malola, H. Häkkinen and N. Zheng, All-thiol-stabilized Ag44 and Au12Ag32 nanoparticles with single-crystal structures, Nat. Commun., 2013, 4, 2422–2429 CrossRef PubMed.
- J. Yan, B. K. Teo and N. Zheng, Surface chemistry of atomically precise coinage-metal nanoclusters: from structural control to surface reactivity and catalysis, Acc. Chem. Res., 2018, 51, 3084–3093 CrossRef CAS PubMed.
- T. Udayabhaskararao and T. Pradeep, New protocols for the synthesis of stable Ag and Au nanocluster molecules, J. Phys. Chem. Lett., 2013, 4, 1553–1564 CrossRef CAS PubMed.
- K. Zheng, X. Yuan, N. Goswami, Q. Zhang and J. Xie, Recent advances in the synthesis, characterization, and biomedical applications of ultrasmall thiolated silver nanoclusters, RSC Adv., 2014, 4, 60581–60596 RSC.
- J. Yang and R. Jin, New advances in atomically precise silver nanoclusters, ACS Mater. Lett., 2019, 1, 482–489 CrossRef CAS.
- X. Kang and M. Zhu, Tailoring the photoluminescence of atomically precise nanoclusters, Chem. Soc. Rev., 2019, 48, 2422–2457 RSC.
- H. Yu, B. Rao, W. Jiang, S. Yang and M. Zhu, The photoluminescent metal nanoclusters with atomic precision, Coord. Chem. Rev., 2019, 378, 595–617 CrossRef CAS.
- Q. Yao, T. Chen, X. Yuan and J. Xie, Toward total synthesis of thiolate-protected metal nanoclusters, Acc. Chem. Res., 2018, 51, 1338–1348 CrossRef CAS PubMed.
- N. Goswami, Q. Yao, T. Chen and J. Xie, Mechanistic exploration and controlled synthesis of precise thiolate-gold nanoclusters, Coord. Chem. Rev., 2016, 329, 1–15 CrossRef CAS.
- M. Qu, H. Li, L.-H. Xie, S.-T. Yan, J.-R. Li, J.-H. Wang, C.-Y. Wei, Y.-W. Wu and X.-M. Zhang, Bidentate phosphine-assisted synthesis of an all-alkynyl-protected Ag74 nanocluster, J. Am. Chem. Soc., 2017, 139, 12346 CrossRef CAS PubMed.
- G.-X. Duan, L. Tian, J.-B. Wen, L.-Y. Li, Y.-P. Xie and X. Lu, An atomically precise all-tert-butylethynide-protected Ag51 superatom nanocluster with color tenability, Nanoscale, 2018, 10, 18915–18919 RSC.
- Y. Chen, C. Zeng, D. R. Kauffman and R. Jin, Tuning the magic size of atomically precise gold nanoclusters via isomeric methylbenzenethiols, Nano Lett., 2015, 15, 3603–3609 CrossRef CAS PubMed.
- M. A. Muhammed, F. Aldeek, G. Palui, L. Trapiella-Alfonso and H. Mattoussi, Growth of in situ functionalized luminescent silver nanoclusters by direct reduction and size focusing, ACS Nano, 2012, 6, 8950–8961 CrossRef CAS PubMed.
- B. Adhikari and A. Banerjee, Facile synthesis of water-soluble fluorescent silver nanoclusters and HgII sensing, Chem. Mater., 2010, 22, 4364–4371 CrossRef CAS.
- Z. Wu, E. Lanni, W. Chen, M. E. Bier, D. Ly and R. Jin, High yield, large scale synthesis of thiolate-protected Ag7 clusters, J. Am. Chem. Soc., 2009, 131, 16672–16674 CrossRef CAS PubMed.
- I. Chakraborty, T. Udayabhaskararao and T. Pradeep, High temperature nucleation and growth of glutathione protected∼ Ag75 clusters, Chem. Commun., 2012, 48, 6788–6790 RSC.
- Z. Wang, R. K. Gupta, G.-G. Luo and D. Sun, Design and development of highly efficient light-emitting layers in OLEDs with dimesitylboranes: an updated review, Chem. Rec., 2019, 19, 1–15 CrossRef.
- I. Chakraborty, T. Udayabhaskararao, G. K. Deepesh and T. Pradeep, Sunlight mediated synthesis and antibacterial properties of monolayer protected silver clusters, J. Mater. Chem. B, 2013, 1, 4059–4064 RSC.
- T. Zhou, M. Rong, Z. Cai, C. J. Yang and X. Chen, Sonochemical synthesis of highly fluorescent glutathione-stabilized Ag nanoclusters and S2− sensing, Nanoscale, 2012, 4, 4103–4106 RSC.
- B. S. González, M. Blanco and M. A. López-Quintela, Single step electrochemical synthesis of hydrophilic/hydrophobic Ag5 and Ag6 blue luminescent clusters, Nanoscale, 2012, 4, 7632–7635 RSC.
- X. Le Guével, C. Spies, N. Daum, G. Jung and M. Schneider, Highly fluorescent silver nanoclusters stabilized by glutathione: a promising fluorescent label for bioimaging, Nano Res., 2012, 5, 379–387 CrossRef.
- X. Yuan, M. I. Setyawati, A. S. Tan, C. N. Ong, D. T. Leong and J. Xie, Highly luminescent silver nanoclusters with tunable emissions: cyclic reduction-decomposition synthesis and antimicrobial properties, NPG Asia Mater., 2013, 5, e39 CrossRef CAS.
- K. V. Mrudula, T. Udayabhaskararao and T. Pradeep, Interfacial synthesis of luminescent 7 kDa silver clusters, J. Mater. Chem., 2009, 19, 4335–4342 RSC.
- T. Udayabhaskararao and T. Pradeep, Luminescent Ag7 and Ag8 clusters by interfacial synthesis, Angew. Chem., Int. Ed., 2010, 49, 3925–3929 CrossRef CAS PubMed.
- L. Dhanalakshmi, T. Udayabhaskararao and T. Pradeep, Conversion of double layer charge-stabilized Ag@citrate colloids to thiol passivated luminescent quantum clusters, Chem. Commun., 2012, 48, 859–861 RSC.
- L. G. AbdulHalim, N. Kothalawala, L. Sinatra, A. Dass and O. M. Bakr, Neat and complete: Thiolate-ligand exchange on a silver molecular nanoparticle, J. Am. Chem. Soc., 2014, 136, 15865–15868 CrossRef CAS PubMed.
- M. S. Bootharaju, V. M. Burlakov, T. M. D. Besong, C. P. Joshi, L. G. AbdulHalim, D. M. Black, R. L. Whetten, A. Goriely and O. M. Bakr, Reversible size control of silver nanoclusters via ligand-exchange, Chem. Mater., 2015, 27, 4289–4297 CrossRef CAS.
- M. S. Bootharaju, C. P. Joshi, M. J. Alhilaly and O. M. Bakr, Switching a nanocluster core from hollow to nonhollow, Chem. Mater., 2016, 28, 3292–3297 CrossRef CAS.
- E. Khatun, A. Ghosh, D. Ghosh, P. Chakraborty, A. Nag, B. Mondal, S. Chennu and T. Pradeep, [Ag59(2,5-DCBT)32]3−: a new cluster and a precursor for three well-known clusters, Nanoscale, 2017, 9, 8240–8248 RSC.
- T. Udayabhaskararao, B. Nataraju and T. Pradeep, Ag9 quantum cluster through a solid-state route, J. Am. Chem. Soc., 2010, 132, 16304–16307 CrossRef PubMed.
- T. Udayabhaskararao, M. Bootharaju and T. Pradeep, Thiolate-protected Ag32 clusters: mass spectral studies of composition and insights into the Ag-thiolate structure from NMR, Nanoscale, 2013, 5, 9404–9411 RSC.
- I. Chakraborty, A. Govindarajan, J. Erusappan, A. Ghosh, T. Pradeep, B. Yoon, R. L. Whetten and U. Landman, The superstable 25 kDa monolayer protected silver nanoparticle: measurements and interpretation as an icosahedral Ag152(SCH2CH2Ph)60 cluster, Nano Lett., 2012, 12, 5861–5866 CrossRef CAS PubMed.
- I. Chakraborty, W. Kurashige, K. Kanehira, L. Gell, H. Häkkinen, Y. Negishi and T. Pradeep, Ag44(SeR)30: a hollow cage silver cluster with selenolate protection, J. Phys. Chem. Lett., 2013, 4, 3351–3355 CrossRef CAS PubMed.
- I. Chakraborty, T. Udayabhaskararao and T. Pradeep, Luminescent sub-nanometer clusters for metal ion sensing: A new direction in nanosensors, J. Hazard. Mater., 2012, 211–212, 396–403 CrossRef CAS PubMed.
- W. Du, S. Jin, L. Xiong, M. Chen, J. Zhang, X. Zou, Y. Pei, S. Wang and M. Zhu, Ag50(Dppm)6(SR)30 and its homologue AuxAg50–x(Dppm)6(SR)30 alloy nanocluster: seeded growth, structure determination, and differences in properties, J. Am. Chem. Soc., 2017, 139, 1618–1624 CrossRef CAS PubMed.
- X. Kang and M. Zhu, Intra-cluster growth meets inter-cluster assembly: the molecular and supramolecular chemistry of atomically precise nanoclusters, Coord. Chem. Rev., 2019, 394, 1–38 CrossRef CAS.
- X. Du, J. Chai, S. Yang, Y. Li, T. Higaki, S. Li and R. Jin, Fusion growth patterns in atomically precise metal nanoclusters, Nanoscale, 2019, 11, 19158–19165 RSC.
- T. Higaki, C. Zeng, Y. Chen, E. Hussain and R. Jin, Controlling the crystalline phases (FCC, HCP and BCC) of thiolate-protected gold nanoclusters by ligand-based strategies, CrystEngComm, 2016, 18, 6979–6986 RSC.
- S. Jin, S. Wang, Y. Song, M. Zhou, J. Zhong, J. Zhang, A. Xia, Y. Pei, M. Chen, P. Li and M. Zhu, Crystal structure and optical properties of the [Ag62S12(SBut)32]2+ nanocluster with a complete face-centered cubic kernel, J. Am. Chem. Soc., 2014, 136, 15559–15565 CrossRef CAS PubMed.
- G. Li, Z. Lei and Q.-M. Wang, Luminescent molecular Ag–S nanocluster [Ag62S13(SBut)32](BF4)4, J. Am. Chem. Soc., 2010, 132, 17678–17679 CrossRef CAS PubMed.
- S. Jin, S. Wang, L. Xiong, M. Zhou, S. Chen, W. Du, A. Xia, Y. Pei and M. Zhu, Two electron reduction: From quantum dots to metal nanoclusters, Chem. Mater., 2016, 28, 7905–7911 CrossRef CAS.
- X. Liu, J. Chen, J. Yuan, Y. Li, J. Li, S. Zhou, C. Yao, L. Liao, S. Zhuang, Y. Zhao, H. Deng, J. Yang and Z. Wu, A silver nanocluster containing interstitial sulfur and unprecedented chemical bonds, Angew. Chem., Int. Ed., 2018, 57, 11273–11277 CrossRef CAS PubMed.
- H. Yang, J. Lei, B. Wu, Y. Wang, M. Zhou, A. Xia, L. Zheng and N. Zheng, Crystal structure of a luminescent thiolated Ag nanocluster with an octahedral Ag64+ core, Chem. Commun., 2013, 49, 300–302 RSC.
- C. Liu, T. Li, H. Abroshan, Z. Li, C. Zhang, H. Kim, G. Li and R. Jin, Chiral Ag23 nanocluster with open shell electronic structure and helical face-centered cubic framework, Nat. Commun., 2018, 9, 744–749 CrossRef PubMed.
- B. K. Teo, H. Yang, J. Yan and N. Zheng, Supercubes, supersquares, and superrods of face-centered cubes (FCC): atomic and electronic requirements of [Mm(SR)l(PR′3)8]q nanoclusters (M = Coinage Metals) and their implications with respect to nucleation and growth of FCC metals, Inorg. Chem., 2017, 56, 11470 CrossRef CAS PubMed.
- H. Yang, J. Yan, Y. Wang, H. Su, L. Gell, X. Zhao, C. Xu, B. K. Teo, H. Häkkinen and N. Zheng, Embryonic growth of face-center-cubic silver nanoclusters shaped in nearly perfect half-cubes and cubes, J. Am. Chem. Soc., 2017, 139, 31–34 CrossRef CAS PubMed.
- Z.-Y. Wang, M.-Q. Wang, Y.-L. Li, P. Luo, T.-T. Jia, R.-W. Huang, S.-Q. Zang and T. C. W. Mak, Atomically precise site-specific tailoring and directional assembly of superatomic silver nanoclusters, J. Am. Chem. Soc., 2018, 140, 1069–1076 CrossRef CAS PubMed.
- J. Chai, S. Yang, Y. Lv, T. Chen, S. Wang, H. Yu and M. Zhu, A unique pair: Ag40 and Ag46 nanoclusters with the same surface but different cores for structure-property correlation, J. Am. Chem. Soc., 2018, 140, 15582–15585 CrossRef CAS PubMed.
- M. Bodiuzzaman, A. Ghosh, K. S. Sugi, A. Nag, E. Khatun, B. Varghese, G. Paramasivam, S. Antharjanam, G. Natarajan and T. Pradeep, Camouflaging structural diversity: Co-crystallization of two different nanoparticles having different cores but the same shell, Angew. Chem., Int. Ed., 2019, 58, 189–194 CrossRef CAS PubMed.
- M. Alhilaly, M. Bootharaju, C. Joshi, T. Besong, A. Emwas, R. Juarez-Mosqueda, S. Kaappa, S. Malola, K. Adil, A. Shkurenko, H. Häkkinen, M. Eddaoudi and O. Bakr, [Ag67(SPhMe2)32(PPh3)8]3+: Synthesis, total structure, and optical properties of a large box-shaped silver nanocluster, J. Am. Chem. Soc., 2016, 138, 14727 CrossRef CAS PubMed.
- X. Yuan, C. Sun, X. Li, S. Malola, B. K. Teo, H. Häkkinen, L. Zheng and N. Zheng, Combinatorial identification of hydrides in a ligated Ag40 nanocluster with noncompact metal core, J. Am. Chem. Soc., 2019, 141, 11905–11911 CrossRef CAS PubMed.
- C. Zeng, C. Liu, Y. Chen, N. L. Rosi and R. Jin, Gold-thiolate ring as a protecting motif in the Au20(SR)16 nanocluster and implications, J. Am. Chem. Soc., 2014, 136, 11922–11925 CrossRef CAS PubMed.
- C. Zeng, T. Li, A. Das, N. L. Rosi and R. Jin, Chiral structure of thiolate-protected 28-gold-atom nanocluster determined by X-ray crystallography, J. Am. Chem. Soc., 2013, 135, 10011–10013 CrossRef CAS PubMed.
- C. Zeng, Y. Chen, K. Kirschbaum, K. Appavoo, M. Y. Sfeir and R. Jin, Structural patterns at all scales in a nonmetallic chiral Au133(SR)52 nanoparticle, Sci. Adv., 2015, 1, e1500045 CrossRef PubMed.
- H. Yang, Y. Wang and N. Zheng, Stabilizing subnanometer Ag(0) nanoclusters by thiolate and diphosphine ligands and their crystal structures, Nanoscale, 2013, 5, 2674–2677 RSC.
- X. Zou, S. Jin, W. Du, Y. Li, P. Li, S. Wang and M. Zhu, Multi-ligand-directed synthesis of chiral silver nanoclusters, Nanoscale, 2017, 9, 16800–16805 RSC.
- H. Yang, J. Yan, Y. Wang, G. Deng, H. Su, X. Zhao, C. Xu, B. K. Teo and N. Zheng, From racemic metal nanoparticles to optically pure enantiomers in one pot, J. Am. Chem. Soc., 2017, 139, 16113–16116 CrossRef CAS PubMed.
- F. Tian and R. Chen, Pd-mediated synthesis of Ag33 chiral nanocluster with core-shell structure in T point group, J. Am. Chem. Soc., 2019, 141, 7107–7114 CrossRef CAS PubMed.
- H. Y. Yang, Y. Wang, X. Chen, X. J. Zhao, L. Gu, H. Q. Huang, J. Z. Yan, C. F. Xu, G. Li, J. C. Wu, A. J. Edwards, B. Dittrich, Z. C. Tang, D. D. Wang, L. Lehtovaara, H. Häkkinen and N. Zheng, Plasmonic twinned silver nanoparticles with molecular precision, Nat. Commun., 2016, 7, 12809–12816 CrossRef CAS PubMed.
- L. Ren, P. Yuan, H. Su, S. Malola, S. Lin, Z. Tang, B. K. Teo, H. Häkkinen, L. Zheng and N. Zheng, Bulky surface ligands promote surface reactivities of [Ag141X12(S-Adm)40]3+ (X = Cl, Br, I) nanoclusters: models for multiple-twinned nanoparticles, J. Am. Chem. Soc., 2017, 139, 13288–13291 CrossRef CAS PubMed.
- Y. B. Song, K. Lambright, M. Zhou, K. Kirschbaum, J. Xiang, A. D. Xia, M.-Z. Zhu and R. C. Jin, Large-scale synthesis, crystal structure, and optical properties of the Ag146Br2(SR)80 nanocluster, ACS Nano, 2018, 12, 9318–9325 CrossRef CAS PubMed.
- J. Z. Yan, J. Zhang, X. M. Chen, S. Malola, B. Zhou, E. Selenius, X. M. Zhang, P. Yuan, G. C. Deng, K. L. Liu, H. F. Su, B. K. Teo, H. Häkkinen, L. S. Zheng and N. Zheng, Thiol-stabilized atomically precise, superatomic silver nanoparticles for catalysing cycloisomerization of alkynyl amines, Natl. Sci. Rev., 2018, 5, 694–702 CrossRef CAS.
- J. Y. Liu, F. Alkan, Z. Wang, Z. Y. Zhang, M. Kurmoo, Z. Yan, Q. Q. Zhao, C. M. Aikens, C. H. Tung and D. Sun, Different silver nanoparticles in one crystal: Ag210(iPrPhS)71(Ph3P)5Cl and Ag211(iPrPhS)71(Ph3P)6Cl, Angew. Chem., Int. Ed., 2019, 58, 195–199 CrossRef CAS PubMed.
- E. Khatun, M. Bodiuzzaman, K. S. Sugi, P. Chakraborty, G. Paramasivam, W. A. Dar, T. Ahuja, S. Antharjanam and T. Pradeep, Confining an Ag10 core in an Ag12 shell: a four-electron superatom with enhanced photoluminescence upon crystallization, ACS Nano, 2019, 13, 5753–5759 CrossRef CAS PubMed.
- Z. Lei, X. K. Wan, S. F. Yuan, Z. J. Guan and Q. M. Wang, Alkynyl approach toward the protection of metal nanoclusters, Acc. Chem. Res., 2018, 51, 2465–2474 CrossRef CAS PubMed.
- Q.-M. Wang, Y.-M. Lin and K.-G. Liu, Role of anions associated with the formation and properties of silver clusters, Acc. Chem. Res., 2015, 48, 1570–1579 CrossRef CAS PubMed.
- Z. Lei, X.-K. Wan, S.-F. Yuan, J.-Q. Wang and Q.-M. Wang, Alkynyl-protected gold and gold-silver nanoclusters, Dalton Trans., 2017, 46, 3427–3434 RSC.
- S.-F. Yuan, P. Li, Q. Tang, X.-K. Wan, Z.-A. Nan, D.-E. Jiang and Q.-M. Wang, Alkynyl-protected silver nanoclusters featuring an anticuboctahedral kernel, Nanoscale, 2017, 9, 11405–11409 RSC.
- Z.-J. Guan, F. Hu, S.-F. Yuan, Z.-A. Nan, Y.-M. Lin and Q.-M. Wang, The stability enhancement factor beyond eight-electron shell closure in thiacalix[4] arene-protected silver clusters, Chem. Sci., 2019, 10, 3360–3365 RSC.
- Z.-J. Guan, J.-L. Zeng, Z.-A. Nan, X.-K. Wan, Y.-M. Lin and Q.-M. Wang, Thiacalix[4] arene: new protection for metal nanoclusters, Sci. Adv., 2016, 2, e1600323 CrossRef PubMed.
- F. Hu, J.-J. Li, Z.-J. Guan, S.-F. Yuan and Q.-M. Wang, Formation of an alkynyl-protected silver nanocluster Ag112 promoted by in situ released chlorides from CH2Cl2, Angew. Chem., Int. Ed., 2020, 59, 5312–5315 CrossRef CAS PubMed.
- S.-S. Zhang, F. Alkan, H.-F. Su, C. M. Aikens, C.-H. Tung and D. Sun, [Ag48(C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CtBu)20(CrO4)7]: An atomically precise silver nanocluster co-protected by inorganic and organic ligands, J. Am. Chem. Soc., 2019, 141, 4460–4467 CrossRef CAS PubMed.
CtBu)20(CrO4)7]: An atomically precise silver nanocluster co-protected by inorganic and organic ligands, J. Am. Chem. Soc., 2019, 141, 4460–4467 CrossRef CAS PubMed. - R. S. Dhayal, J.-H. Liao, Y.-C. Liu, M.-H. Chiang, S. Kahlal, J.-Y. Saillard and C. W. Liu, [Ag21{S2P(OiPr)2}12]+: an eight-electron superatom, Angew. Chem., Int. Ed., 2015, 54, 3702 CrossRef CAS PubMed.
- R. S. Dhayal, Y.-R. Lin, J.-H. Liao, Y.-J. Chen, Y.-C. Liu, M.-H. Chiang, S. Kahlal, J.-Y. Saillard and C. W. Liu, [Ag20{S2P(OR)2}12]: a superatom complex with a chiral metallic core and high potential for isomerism, Chem. – Eur. J., 2016, 22, 9943 CrossRef CAS PubMed.
- W.-T. Chang, P.-Y. Lee, J.-H. Liao, K. K. Chakrahari, S. Kahlal, Y.-C. Liu, M.-H. Chiang, J.-Y. Saillard and C. W. Liu, Eight-electron silver and mixed gold/silver nanoclusters stabilized by selenium donor ligands, Angew. Chem., Int. Ed., 2017, 56, 10178–10182 CrossRef CAS PubMed.
- S.-F. Yuan, Z.-J. Guan, W.-D. Liu and Q.-M. Wang, Solvent-triggered reversible interconversion of all-nitrogen-donor-protected silver nanoclusters and their responsive optical properties, Nat. Commun., 2019, 10, 4032 CrossRef PubMed.
- X.-T. Shen, X.-Li. Ma, Q.-L. Ni, M.-X. Ma, L.-C. Gui, C. Hou, R.-B. Hou and X.-J. Wang, [Ag15(N-triphos)4(Cl4)](NO3)3: a stable Ag-P superatom with eight electrons (N-triphos = tris ((diphenylphosphino) methyl) amine), Nanoscale, 2018, 10, 515–519 RSC.
- G.-X. Duan, J. Han, B.-Z. Yang, Y.-P. Xie and X. Lu, Oxometalate and phosphine ligand co-protected silver nanoclusters: Ag28(dppb)6(MO4)4 and Ag32(dppb)12(MO4)4 (NO3)4, Nanoscale, 2020, 12, 1617–1622 RSC.
- J. Yan, H. Su, H. Yang, C. Hu, S. Malola, S. Lin, B.-K. Teo, H. Häkkinen and N. Zheng, Asymmetric synthesis of chiral bimetallic [Ag28Cu12(SR)24]4− nanoclusters via ion pairing, J. Am. Chem. Soc., 2016, 138, 12751 CrossRef CAS PubMed.
- M. S. Bootharaju, H. Chang, G. Deng, S. Malola, W. Baek, H. Häkkinen, N. Zheng and T. Hyeon, Cd12Ag32(SePh)36: non-noble metal doped silver nanoclusters, J. Am. Chem. Soc., 2019, 141, 8422 CrossRef CAS PubMed.
- K. Yonesato, H. Ito, H. Itakura, D. Yokogawa, T. Kikuchi, N. Mizuno, K. Yamaguchi and K. Suzuki, Controlled assembly synthesis of atomically precise ultrastable silver nanoclusters with polyoxometalates, J. Am. Chem. Soc., 2019, 141, 19550–19554 CrossRef CAS PubMed.
- Y.-M. Su, Z. Wang, G.-L. Zhuang, Q.-Q. Zhao, X.-P. Wang, C.-H. Tung and D. Sun, Unusual fcc-structured Ag10 kernels trapped in Ag70 nanoclusters, Chem. Sci., 2019, 10, 564–568 RSC.
- J.-W. Liu, Z. Wang, Y.-M. Chai, M. Kurmoo, Q.-Q. Zhao, X.-P. Wang, C.-H. Tung and D. Sun, Core modulation of 70-nuclei core-shell silver nanoclusters, Angew. Chem., Int. Ed., 2019, 58, 6276–6279 CrossRef CAS PubMed.
- Z. Wang, H.-T. Sun, M. Kurmoo, Q.-Y. Liu, G.-L. Zhuang, Q.-Q. Zhao, X.-P. Wang, C.-H. Tung and D. Sun, Carboxylic acid stimulated silver shell isomerism in a triple core–shell Ag84 nanocluster, Chem. Sci., 2019, 10, 4862–4867 RSC.
- T. Vosch, Y. Antoku, J.-C. Hsiang, C. I. Richards, J. I. Gonzalez and R. M. Dickson, Strongly emissive individual DNA-encapsulated Ag nanoclusters as single-molecule fluorophores, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 12616–12621 CrossRef CAS PubMed.
- D. Schultz, K. Gardner, S. S. R. Oemrawsingh, N. Markesevic, K. Olsson, M. Debord, D. Bouwmeester and E. Gwinn, Evidence for rod-shaped DNA-stabilized silver nanocluster emitters, Adv. Mater., 2013, 25, 2797–2803 CrossRef CAS PubMed.
- Z. Yuan, Y.-C. Chen, H.-W. Li and H.-T. Chang, Fluorescent silver nanoclusters stabilized by DNA scaffolds, Chem. Commun., 2014, 50, 9800–9815 RSC.
- J. T. Petty, O. O. Sergev, M. Ganguly, I. J. Rankine, D. M. Chevrier and P. Zhang, A segregated, partially oxidized, and compact Ag10 cluster within an encapsulating DNA host, J. Am. Chem. Soc., 2016, 138, 3469–3477 CrossRef CAS PubMed.
- E. Thyrhaug, S. A. Bogh, M. R. Carro-Temboury, C. S. Madsen, T. Vosch and D. Zigmantas, Ultrafast coherence transfer in DNA-templated silver nanoclusters, Nat. Commun., 2017, 8, 15577 CrossRef CAS PubMed.
- J. M. Obliosca, C. Liu and H.-C. Yeh, Fluorescent silver nanoclusters as DNA probes, Nanoscale, 2013, 5, 8443–8461 RSC.
- W. Guo, J. Yuan, Q. Dong and E. Wang, Highly sequence-dependent formation of fluorescent silver nanoclusters in hybridized DNA duplexes for single nucleotide mutation identification, J. Am. Chem. Soc., 2010, 132, 932–934 CrossRef CAS PubMed.
- H.-C. Yeh, J. Sharma, J. J. Han, J. S. Martinez and J. H. Werner, A DNA−silver nanocluster probe that fluoresces upon hybridization, Nano Lett., 2010, 10, 3106–3110 CrossRef CAS PubMed.
- X. Liu, F. Wang, R. Aizen, O. Yehezkeli and I. Willner, Graphene oxide/nucleic-acid-stabilized silver nanoclusters: functional hybrid materials for optical aptamer sensing and multiplexed analysis of pathogenic DNAs, J. Am. Chem. Soc., 2013, 135, 11832–11839 CrossRef CAS PubMed.
- J. T. Petty, J. Zheng, N. V. Hud and R. M. Dickson, DNA-templated Ag nanocluster formation, J. Am. Chem. Soc., 2004, 126, 5207–5212 CrossRef CAS PubMed.
- E. G. Gwinn, P. O’Neill, A. J. Guerrero, D. Bouwmeester and D. K. Fygenson, Sequence-dependent fluorescence of DNA hosted silver nanoclusters, Adv. Mater., 2008, 20, 279–283 CrossRef CAS.
- P. R. O’Neill, K. Young, D. Schiffels and D. K. Fygenson, Few-atom fluorescent silver clusters assemble at programmed sites on DNA nanotubes, Nano Lett., 2012, 12, 5464–5469 CrossRef PubMed.
- J. Li, X. Jia, D. Li, J. Ren, Y. Han, Y. Xia and E. Wang, Stem-directed growth of highly fluorescent silver nanoclusters for versatile logic devices, Nanoscale, 2013, 5, 6131–6138 RSC.
- W. Li, L. Liu, Y. Fu, Y. Sun, J. Zhang and R. Zhang, Effects of polymorphic DNA on the fluorescent properties of silver nanoclusters, Photochem. Photobiol. Sci., 2013, 12, 1864–1872 RSC.
- J. Ai, W. Guo, B. Li, T. Li, D. Li and E. Wang, DNA G-quadruplex-templated formation of the fluorescent silver nanocluster and its application to bioimaging, Talanta, 2012, 88, 450–455 CrossRef CAS PubMed.
- H. X. Xu and K. S. Suslick, Water-soluble fluorescent silver nanoclusters, Adv. Mater., 2010, 22, 1078–1082 CrossRef CAS PubMed.
- I. Diez and R. H. Ras, Fluorescent silver nanoclusters, Nanoscale, 2011, 3, 1963–1970 RSC.
- L. Shang, S. Dong and G. U. Nienhaus, Ultra-small fluorescent metal nanoclusters: synthesis and biological applications, Nano Today, 2011, 6, 401–418 CrossRef CAS.
- S. J. Guo and E. K. Wang, Noble metal nanomaterials: controllable synthesis and application in fuel cells and analytical sensors, Nano Today, 2011, 6, 240–264 CrossRef CAS.
- S. Choi, R. M. Dickson and J. Yu, Developing luminescent silver nanodots for biological applications, Chem. Soc. Rev., 2012, 41, 1867–1891 RSC.
- B. Han and E. Wang, DNA-templated fluorescent silver nanoclusters, Anal. Bioanal. Chem., 2012, 402, 129–138 CrossRef CAS PubMed.
- J. T. Petty, S. P. Story, J.-C. Hsiang and R. M. Dickson, DNA-templated molecular silver fluorophores, J. Phys. Chem. Lett., 2013, 4, 1148–1155 CrossRef CAS PubMed.
- Y. Tao, M. Li, J. Ren and X. Qu, Metal nanoclusters: novel probes for diagnostic and therapeutic applications, Chem. Soc. Rev., 2015, 44, 8636–8663 RSC.
- A. Latorre and A. Somoza, DNA-mediated silver nanoclusters: synthesis, properties and applications, ChemBioChem, 2012, 13, 951–958 CrossRef CAS PubMed.
- Y. Chen, M. L. Phipps, J. H. Werner, S. Chakraborty and J. S. Martinez, DNA-templated metal nanoclusters: from emergent properties to unique applications, Acc. Chem. Res., 2018, 51, 2756–2763 CrossRef CAS PubMed.
- R. Jiang, B. Li, C. Fang and J. Wang, Unraveling the evolution and nature of the plasmons in (Au core)-(Ag shell) nanorods, Adv. Mater., 2014, 26, 5274–5309 CrossRef CAS PubMed.
- L. M. Liz-Marźan, C. J. Murphy and J. Wang, Nanoplasmonics, Chem. Soc. Rev., 2014, 43, 3820–3822 RSC.
- Q. B. Zhang, Y. N. Tan, J. P. Xie and J. Y. Lee, Colloidal synthesis of plasmonic metallic nanoparticles, Plasmonics, 2009, 4, 9–22 CrossRef CAS.
- J. Huang, Y. Zhu, M. Lin, Q. Wang, L. Zhao, Y. Yang, K. X. Yao and Y. Han, Site-specific growth of Au-Pd alloy horns on Au nanorods: a platform for highly sensitive monitoring of catalytic reactions by surface enhancement Raman spectroscopy, J. Am. Chem. Soc., 2013, 135, 8552–8561 CrossRef CAS PubMed.
- T. H. Lee and R. M. Dickson, Discrete two-terminal single nanocluster quantum optoelectronic logic operations at room temperature, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 3043–3046 CrossRef CAS.
- I. Chakraborty, J. Erusappan, A. Govindarajan, K. S. Sugi, T. Udayabhaskararao, A. Ghosh and T. Pradeep, Emergence of metallicity in silver clusters in the 150 atom regime: a study of differently sized silver clusters, Nanoscale, 2014, 6, 8024–8031 RSC.
- L. A. Peyser, A. E. Vinson, A. P. Bartko and R. M. Dickson, Photoactivated fluorescence from individual silver nanoclusters, Science, 2001, 291, 103–106 CrossRef CAS PubMed.
- S. Li, X.-S. Du, B. Li, J.-Y. Wang, G.-P. Li, G.-G. Gao and S.-Q. Zang, Atom-precise modification of silver(I) thiolate cluster by shell ligand substitution: a new approach to generation of cluster functionality and chirality, J. Am. Chem. Soc., 2018, 140, 594–597 CrossRef CAS PubMed.
- E. Khatun, A. Ghosh, P. Chakraborty, P. Singh, M. Bodiuzzaman, P. Ganesan, G. Nataranjan, J. Ghosh, S. K. Pal and T. Pradeep, A thirty-fold photoluminescence enhancement induced by secondary ligands in monolayer protected silver clusters, Nanoscale, 2018, 10, 20033–20042 RSC.
- M. S. Bootharaju, C. P. Joshi, M. R. Parida, O. F. Mohammed and O. M. Bakr, Templated atom-precise calvanic synthesis and structure elucidation of a [Ag24Au(SR)18]− nanocluster, Angew. Chem., Int. Ed., 2016, 55, 922–926 CrossRef CAS PubMed.
- J. Yan, H. Su, H. Yang, S. Malola, S. Lin, H. Häkkinen and N. Zheng, Total structure and electronic structure analysis of doped thiolated silver [MAg24(SR)18]2− (M = Pd, Pt) clusters, J. Am. Chem. Soc., 2015, 137, 11880–11883 CrossRef CAS PubMed.
- X. Kang, S. Chen, S. Jin, Y. Song, Y. Xu, H. Yu, H. Sheng and M. Zhu, Heteroatom effects on the optical and electrochemical properties of Ag25(SR)18 and its dopants, ChemElectroChem, 2016, 3, 1261–1265 CrossRef CAS.
- Y. Liu, X. Chai, X. Cai, M. Chen, R. Jin, W. Ding and Y. Zhu, Central doping of a foreign atom into the silver cluster for catalytic conversion of CO2 toward C−C bond formation, Angew. Chem., Int. Ed., 2018, 57, 9775–9779 CrossRef CAS PubMed.
- X. Liu, J. Yuan, C. Yao, J. Chen, L. Li, X. Bao, J. Yang and Z. Wu, Crystal and solution photoluminescence of MAg24(SR)18 (M = Ag/Pd/Pt/Au) nanoclusters and some implications for the photoluminescence mechanisms, J. Phys. Chem. C, 2017, 121, 13848–13853 CrossRef CAS.
- M. S. Bootharaju, S. M. Kozlov, Z. Cao, A. Shkurenko, A. M. El-Zohry, O. F. Mohammed, M. Eddaoudi, O. M. Bakr, L. Cavallo and J.-M. Basset, Tailoring the crystal structure of nanoclusters unveiled high photoluminescence via ion pairing, Chem. Mater., 2018, 30, 2719–2725 CrossRef CAS.
- G. Soldan, M. A. Aljuhani, M. S. Bootharaju, L. G. AbdulHalim, M. R. Parida, A.-H. Emwas, O. F. Mohammed and O. M. Bakr, Gold doping of silver nanoclusters: a 26-Fold enhancement in the luminescence quantum yield, Angew. Chem., Int. Ed., 2016, 55, 5749–5753 CrossRef CAS PubMed.
- S.-S. Zhang, L. Feng, R. D. Senanayake, C. M. Aikens, X.-P. Wang, Q.-Q. Zhao, C.-H. Tung and D. Sun, Diphosphine-protected ultrasmall gold nanoclusters: opened icosahedral Au13 and heart-shaped Au8 clusters, Chem. Sci., 2018, 9, 1251–1258 RSC.
- Y.-M. Su, H.-F. Su, Z. Wang, Y.-A. Li, S. Schein, Q.-Q. Zhao, X.-P. Wang, C.-H. Tung, D. Sun and L.-S. Zheng, Three silver nests capped by thiolate/phenylphosphonate, Chem. – Eur. J., 2018, 24, 15096–15103 CrossRef CAS.
- J.-W. Liu, L. Feng, H.-F. Su, Z. Wang, Q.-Q. Zhao, X.-P. Wang, C.-H. Tung, D. Sun and L.-S. Zheng, Anisotropic assembly of Ag52 and Ag76 nanoclusters, J. Am. Chem. Soc., 2018, 140, 1600–1603 CrossRef CAS PubMed.
- J.-W. Liu, H.-F. Su, Z. Wang, Y.-A. Li, Q.-Q. Zhao, X.-P. Wang, C.-H. Tung, D. Sun and L.-S. Zheng, A giant 90-nucleus silver cluster templated by hetero-anions, Chem. Commun., 2018, 54, 4461–4464 RSC.
- X.-Y. Li, Z. Wang, H.-F. Su, S. Feng, M. Kurmoo, C.-H. Tung, D. Sun and L.-S. Zheng, Anion-templated nanosized silver clusters protected by mixed thiolate and diphosphine, Nanoscale, 2017, 9, 3601–3608 RSC.
- Z. Wang, H.-F. Su, Y.-Z. Tan, S. Schein, S.-C. Lin, W. Liu, S.-A. Wang, W.-G. Wang, C.-H. Tung, D. Sun and L.-S. Zheng, Assembly of silver trigons into a buckyball-like Ag180 nanocage, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, E10505 CrossRef PubMed.
- S.-S. Zhang, H.-F. Su, G.-L. Zhuang, X.-P. Wang, C.-H. Tung, D. Sun and L.-S. Zheng, A hexadecanuclear silver alkynyl cluster based NbO framework with triple emissions from the visible to near-infrared II region, Chem. Commun., 2018, 54, 11905–11908 RSC.
- Z. Wang, H.-F. Su, M. Kurmoo, C.-H. Tung, D. Sun and L.-S. Zheng, Trapping an octahedral Ag6 kernel in a seven-fold symmetric Ag56 nanowheel, Nat. Commun., 2018, 9, 2094 CrossRef PubMed.
- Z. Wang, H.-F. Su, C.-H. Tung, D. Sun and L.-S. Zheng, Deciphering synergetic core-shell transformation from [Mo6O22@Ag44] to [Mo8O28@Ag50], Nat. Commun., 2018, 9, 4407 CrossRef PubMed.
- R.-W. Huang, Y.-S. Wei, X.-Y. Dong, X.-H. Wu, C.-X. Du, S.-Q. Zang and T. C. W. Mak, Hypersensitive dual-function luminescence switching of a silver-chalcogenolate cluster-based metal-organic framework, Nat. Chem., 2017, 9, 689–697 CrossRef CAS.
- R.-W. Huang, X.-Y. Dong, B.-J. Yan, X.-S. Du, D.-H. Wei, S.-Q. Zang and T. C. W. Mak, Tandem silver cluster isomerism and mixed linkers to modulate the photoluminescence of cluster-assembled materials, Angew. Chem., Int. Ed., 2018, 57, 8560–8566 CrossRef CAS PubMed.
- X.-Y. Dong, H.-L. Huang, J.-Y. Wang, H.-Y. Li and S.-Q. Zang, A flexible fluorescent SCC-MOF for switchable molecule identification and temperature display, Chem. Mater., 2018, 30, 2160–2167 CrossRef CAS.
- Q.-Q. Xu, X.-Y. Dong, R.-W. Huang, B. Li, S.-Q. Zang and T. C. W. Mak, A thermochromic silver nanocluster exhibiting dual emission character, Nanoscale, 2015, 7, 1650–1654 RSC.
- B. Li, R.-W. Huang, J.-H. Qin, S.-Q. Zang, G.-G. Gao, H.-W. Hou and T. C. W. Mak, Thermochromic luminescent nest-like silver thiolate cluster, Chem. – Eur. J., 2014, 20, 12416–12420 CrossRef CAS PubMed.
- X.-J. Xi, J.-S. Yang, J.-Y. Wang, X.-Y. Dong and S.-Q. Zang, New stable isomorphous Ag34 and Ag33 Au nanoclusters with an open shell electronic structure, Nanoscale, 2018, 10, 21013–21018 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/d0qm00117a |
| This journal is © the Partner Organisations 2020 |
