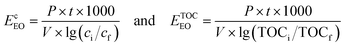Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Comparison of advanced oxidation processes in the decomposition of diuron and monuron – efficiency, intermediates, electrical energy per order and the effect of various matrices
János
Farkas
a,
Máté
Náfrádi
 bd,
Tamás
Hlogyik
b,
Bartus
Cora Pravda
c,
Krisztina
Schrantz
b,
Klára
Hernádi
ac and
Tünde
Alapi
bd,
Tamás
Hlogyik
b,
Bartus
Cora Pravda
c,
Krisztina
Schrantz
b,
Klára
Hernádi
ac and
Tünde
Alapi
 *b
*b
aResearch Group of Environmental Chemistry, University of Szeged, Rerrich Béla tér 1, H-6720, Szeged, Hungary
bDepartment of Inorganic and Analytical Chemistry, University of Szeged, Dóm tér 7, H-6720 Szeged, Hungary. E-mail: alapi@chem.u-szeged.hu
cDepartment of Applied and Environmental Chemistry, University of Szeged, Rerrich Béla tér 1, H-6720 Szeged, Hungary
dInstitute of Environmental Sciences, University of Szeged, Pf. 653, H-6701, Szeged, Hungary
First published on 3rd August 2018
Abstract
The decomposition of diuron and monuron, widely used phenylurea pesticides, via UV-induced photolysis (UV254nm), ozonation (O3), their combination (UV254nm/O3) and heterogeneous photocatalysis (TiO2/UV) were investigated and compared. The UV254nm/O3 and TiO2/UV methods proved to be effective from the aspects of both the transformation and the mineralization. The transformation rates changed in the order: O3 ≪ TiO2/UV < UV254nm < UV254nm/O3. Comparing the electric energy per order (EcEO) values calculated for the transformation, the most efficient method was UV254nm/O3. However, the lowest ETOCEO value calculated for the mineralization was obtained using TiO2/UV. Identification of the aromatic intermediates revealed that the first step in the decomposition involves transformation of the aliphatic chain, in parallel with dechlorination and hydroxylation of the aromatic ring. The amount and quality of the intermediates formed depends strongly on the method applied. Matrices such as natural waters (from the River Tisza and thermal water from Kistelek) and inorganic salts exert significant effect on the transformation rates only in the case of TiO2/UV. Humic acids behave as “light filter” and consequently decrease the rate of photoinitiated transformation using UV254nm and UV254nm/O3 methods, whereas they slightly enhance the effect of ozonation, most likely because humic acids and/or their intermediates promote the decomposition of ozone and increase the radical concentration. Regarding this parameter, the most sensitive method was heterogeneous photocatalysis, most likely because the well adsorbed humic acid strongly inhibits the formation of hydroxyl radical.
Water impactPollution of the environment by pesticide is a complex problem with widespread ecological consequences. The present study aimed to investigate and compare the UV photolysis, ozonation, their combination and heterogeneous photocatalysis for the transformation, mineralization and dechlorination of diuron and monuron. The study partly focused on the investigations of the effects of the matrices on the photooxidation of the herbicides in different types of waters, such as river and thermal waters. |
1. Introduction
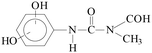
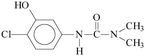
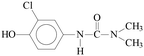
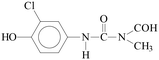
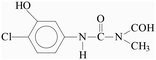
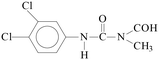
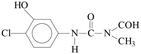
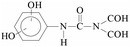
Pollution of the environment with pesticides is a complex problem with widespread ecological consequences. The contamination of surface waters and waste waters by pesticide residues, mainly resulting from agricultural activities and pesticide manufacturing plants has increased significantly during recent decades. The water supply industry is facing a difficult problem, since pesticides have been detected not only in waste waters1 and natural waters,2–5 but also in drinking water,6–8 and are probably responsible for a number of health problems.9 Although the amount detected in drinking water is generally lower than the limits set by the European Union, wastewaters from agricultural and/or industrial activities often contain excessively high levels of contamination.Phenylurea derivatives, such as diuron (3-(3,4-dichlorophenyl)-1,1-dimethylurea), and monuron (3-(4-chlorophenyl)-1,1-dimethylurea) (Fig. 1) are reported to be amongst the most widely used herbicides in agriculture. Both have a broad spectrum of uses, mainly as total herbicides in non-agricultural areas (roads and railways) for the pre- and post-emergence control of weeds, but also for plant protection purposes in the case of certain species (e.g. sugar cane and maize).10
The widely investigated advanced oxidation processes (AOPs), which are based on the generation of various reactive radicals, can offer a solution for the treatment of waters contaminated with pesticides, including phenylurea herbicides.11,12 Numerous investigations have focused on the decomposition of these target substances using conventional water treatment methods,13,14 membrane technologies15 and various AOPs,16 including direct UV photolysis,17–20 ozonation,21–24 heterogeneous photocatalysis,18,25–27 and/or combinations of these methods. Considerable effort has been devoted to research involving ozone in association with other oxidation processes,28–31 in which non-selective hydroxyl radical oxidation is believed to play a key role in the mineralization of organic substances. Other studies have focused on the identification of the main by-products, and proposals have been put forward relating to the reaction mechanisms.18,21,32–34 Investigations of the toxicity of the intermediates33,35 have revealed that this often exceeds that of the parent compounds.
AOPs based on light emission can be used successfully as post-treatment process when the aim is the decomposition of organic compounds which cannot be eliminated easily via biological processes. Post treatment is important when these persistent compounds are toxic, carcinogen or endocrine disrupting substances. The application of AOPs might be limited for the treatment of multicomponent industrial wastewater because of the negative effect of the matrix. The matrices can contain various types of components (mainly dissolved inorganic salts and natural organic matter (NOM)), which are able to affect the transformation of target substances by several possible pathways.17,20,24,36 The main components of NOM are humic substances. A fraction of humic substances, humic acids (HA) have been known to have several effects on drinking water, like aesthetic problems (colour, smell, taste). They are one of the main precursor materials of disinfection by-products, forming further harmful by-products during chlorination. Moreover they are able to increase the potential of the trihalomethanes (THM) formation after chlorination of treated waters.37 Thus, their removal requires additional chemical treatment.38 The formation of complexes with metals and organic pollutants may also have significant environmental importance.39
During AOP-treatment of humic acid containing waters, the effect of these substances depends on both the method and reaction parameters. In the case of UV light based technology, due to the wide absorption spectrum of humic acids, they can act as a light filter and reduce the light intensity absorbed by the target substances, due to competitive light absorption. They are able to increase the reactivity of various substances through the complex formation and/or partly due to the photosensitization.40 Humic acids exert an acceleration effect on the hydrolysis rate of pesticides too.41 In the case of ozonation, organic compounds may initiate the degradation of ozone resulting in increased hydroxyl radical formation and degradation performance.42 Beside this, humic acids can act as a radical scavenger and cause negative effect on the efficiency of methods based on radical formation.
In the case of heterogeneous photocatalysis the reactions take place on the surface of the photocatalyst, consequently adsorption has a crucial role.43 Moreover, the presence of various inorganic ions and the ionic strength of the matrix can change the surface potential and the formation rate of hydroxyl radical, too.44 As humic acids have amphiphil properties, they show significant adsorption on the surface of TiO2 photocatalyst,45 changing its surface properties.46 The adsorbed humic acids cause a negative surface charge, and their adsorption is highly dependent on the pH and ionic strength of the suspension. The change of the surface properties has effect on the rate of aggregation and the size of aggregates.45,47 Consequently, the complex reaction mechanisms, like the adsorption of target pollutants, the formation of reactive species and the relative contribution of the radical based reaction and direct charge transfer depend strongly on this reaction parameter.
Dissolved inorganic salts may also have an important effect on the degradation of organic pollutants, as they may act as radical transfers via certain ions, like HCO3−, NO2−, PO43−. Their effect can be especially significant in the case of heterogeneous photocatalysis,48–50 as their adsorption on the catalyst surface and the change in the ionic strength of the suspension can significantly change the reaction rates and mechanisms. It has been reported, that inorganic anions inhibit the adsorption of certain organic pollutants.51 Under visible light irradiation, humic acids were able to act as a sensitizer and inject electrons to the conduction band of TiO2, and subsequently transformed and decolorized through a series of electron transfer reactions.52
The present study focuses on the comparison of the efficiencies of four different AOPs (UV-photolysis, ozonation, their combination, and heterogeneous photocatalysis) in the transformation of two hazardous phenylurea herbicides, monuron and diuron using various matrices. Previously authors presented a publication53 about the efficiency of several combinations of various AOPs in the transformation of diuron, monuron and fenuron in distilled water. As that was mentioned previously, the application of AOPs might be limited significantly via presence of various component of the matrix. The present work focuses on the investigation of the effect inorganic salts and humic acids. The measurements are performed in distilled water spiked with NaCl, NaHCO3, NaNO3, and humic acid and in two other matrices, like river water and thermal water. Moreover, the efficiency of applied methods is compared not only based the transformation, mineralization, and dehalogenation rate, but also on the electric energy consumption (electric energy per order). The aromatic intermediates formed during the oxidative transformation are identified, too.
2. Materials and methods
2.1. Experimental parameters
For the UV photolysis, a low-pressure mercury vapour lamp (GCL307T5VH/CELL, LightTech, Hungary, 227 mm arc length) with main output at 254 nm, but with 6% of the total intensity of irradiation at 185 nm, was applied as light source, with a high-purity silica sleeve which transmits both 254 and 185 nm light. This lamp can be applied to generate ozone from oxygen in the gas phase due to the 185 nm light. The electric input of the lamp is 15 W and the effective power output in the UV range is 4.0 W. The photon flow emitted by the light source was determined by potassium ferrioxalate actinometry54 to be 8.10(±0.65) × 10−6 Einstein per s. The fluorescent UV lamp used for photocatalytic experiments had the same electric input and geometric parameters, and emits photons between 300 and 400 nm wavelength, with a maximum at 365 nm. The photon flux is 1.2(±0.06) × 10−5 Einstein per s.The UV lamps with their envelope (320 mm long and 28 mm in internal diameter) were centered in a water-cooled, tubular glass reactor (length 340 mm, inner diameter 46 mm). Oxygen or air (855 ml min−1) was led through the Teflon packing ring between the wall of the lamp and the envelope, which separates the gas phase and the aqueous solution.55
The formation of ozone from oxygen in the gas phase is due to the absorption of 185 nm light. The ozone-containing oxygen or air was bubbled through the perforated envelope into the aqueous solution. Depending on the construction of the envelope and the light source, ozonation (low-pressure mercury vapour lamp and perforated glass envelope), UV photolysis (low-pressure mercury vapour lamp and non-perforated quartz envelope) their combination (combined method, low-pressure mercury vapour lamp and perforated quartz envelope), and heterogeneous photocatalysis (fluorescent UV lamp and non-perforated quartz envelope) were investigated. Thus, the efficiency of these processes could be compared at the same energy consumption.
In the photocatalytic experiments the photocatalyst was TiO2 Aeroxide P25 commercialized by Evonik Industries (73–85% anatase, 14–17% rutile, surface area: aSBET = 35–65 m2 g−1, particle size: danatáz ∼ 25 nm, drutil ∼ 40 nm), which was suspended by sonication. The suspension contained 1.0 g dm−3 TiO2 in each case. The selection of the TiO2 concentration is based on data presented in the previous paper of the same authors.53 The transformation rate of target substances reached maximum at 0.5 g dm−3 TiO2 and slightly decreased when 1.0 g dm−3 TiO2 load was applied. However, 1.0 g dm−3 TiO2 load was chosen because at this concentration the photons must be fully absorbed by the irradiated layer of the suspension. The treated samples were centrifuged and filtered before analysis.
The thermostated (25 ± 0.5 °C) aqueous solution (500 ml) was circulated (375 ml min−1) continuously and stirred with a magnetic stirrer bar in the reservoir. Before each experiment, nitrogen, air or oxygen was bubbled through the solution for at least 10 min. The kinetic measurements were started by switching on the light source.
2.2. Methods
The concentration of diuron or monuron was measured with an Agilent 1100 type HPLC equipped with diode array detector (DAD) and a Lichrospher RP 18 column, methanol/water (v/v = 50/50) mixture serving as eluent. The flow rate was 1.0 ml min−1. Samples were analysed at 210 nm and at the absorption maximum of diuron and monuron (248 nm and 245 nm, respectively). The organic acids formed during the irradiation were determined by using a GROM-RESIN ZH column with 0.01 M sulfuric acid as eluent in the same HPLC system (λ = 206 nm). Calibrations were carried out by using standard solutions of known concentrations. The determination of monuron and diuron concentration was based on the linear regression of the calibration curve (R2 = 0.995) presenting the integrated peak areas of the chromatograms measured by HPLC-DAD. Kinetic curves show the ratio of the actual and the initial concentrations (c/c0, c0 = 1.7 × 10−4 mol dm−3). The transformation of herbicides were characterized by the initial rate (r0), which was obtained from linear regression fits of the decay curves corresponding to 20% of transformation.The intermediates were identified by HPLC-MS (Agilent 1200) after preconcentration by means of solid-phase extraction, on octadecyl cartridges (7020-01 from J. T. Baker). The solid phase extraction was achieved according to the modified method of Shankar et al.56 The cartridges were first conditioned with 5.0 ml methanol and 5.0 ml water. Typically, 200 ml of treated diuron or monuron solution were percolated at 5 ml min−1 and the cartridge was completely dried by using a Baker spe-12G apparatus (J. T. Baker) connected to a vacuum system. After rising with 1.0 ml distilled water the phase was dried for 15 min under a nitrogen flow and subsequently eluted with 2 × 2.0 ml methanol. LC-atmospheric pressure chemical ionization-MS (LC-APCI-MS) was used in the negative ion mode of operation, with a 90 V fragmentor.
The concentration of the sum of the H2O2, methyl- and ethyl-hydroperoxides was measured by a modified literature method57,58 involving the horseradish peroxidase (Sigma)-catalysed transformation of leuco crystal violet (Aldrich, Hungary) in tartrate buffer (pH = 4.0). The absorbance of the solution was measured at 592 nm with a Hewlett-Packard (HP8452A) diode array spectrophotometer. The molar absorptivity of the crystal violet formed from the leuco crystal violet was 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 cm−1.
600 M−1 cm−1.
The concentration of ozone in the gas phase was measured with the same spectrophotometer via the absorbance at 254 nm (ε254nm = 2952 M−1 s−1),59 and was found to be 1.37 × 10−5 M when oxygen was used and 4.04 × 10−6 M on the use of air. The concentration of dissolved ozone in the aqueous phase was determined by the indigo method60,61 in phosphate-buffered solution.
The absorption spectra of the treated samples were detected by spectrophotometry. The adsorption of monuron, diuron and humic acid on the TiO2 surface was determined by the same method. The suspensions were left in dark and stirred until the adsorption equilibrium.
The total organic carbon (TOC) and adsorbable organic halogens (AOX) concentration of 100 μl sample solutions were measured with Euroglass 1200 TOC apparatus.
Diuron and monuron were from Sigma (98%). NaHCO3 (Riedel-de Haën, 99.7%), NaCl (VWR, ≥99%) and NaNO3 (VWR, 99.2%) were used in 1 × 10−2 M concentration to investigate the effects of individual inorganic salts on the degradation processes. For the humic acid experiments sodium-humate was used (Aldrich, tech. grade). The effects of the matrix were investigated through comparison of the kinetic curves measured in Milli-Q bidistilled water, in filtered water from the River Tisza (sampled in Szeged, Hungary) and in thermal water (sampled in Kistelek, Hungary), containing a relatively high total mineral content (1390 mg dm−3) and NaHCO3 concentration (360 mg dm−3).
2.3. Calculation of electrical energy per order (EEO)
The electrical energy per order (EEO) is a useful concept for comparing the performance of UV-based AOPs for the degradation of organic contaminants. The parameter was introduced in 2001 by Bolton, et al. in a report published by the IUPAC Photochemistry Commission.62 When choosing the best method for wastewater treatment, the economic factor is often seen as the most relevant. Calculation is based on the amount of electric energy required to decrease the concentration of pollutant by one order of magnitude. The effectiveness of each process was evaluated based on these EEO values, reflecting the electric energy in kilowatt hours [kW h] required to treat 1 m3 of contaminated water. EEO values [kW h m−3 per order] are calculated for a batch system, using the following formula:where P is the rated power [kW] of the AOP system, V is the volume [dm3] of the treated water, t [h] is the time required to decrease the concentration of pollutant (ci and cf are the initial and final concentrations of monuron [mol dm−3], while TOCi and TOCf are the initial and final TOC content [mol dm−3]) by one order of magnitude, and lg is the symbol for the decadic logarithm. In the present work the power (P) was calculated by the electric power required for the UV lamp (15 W). Ozone was generated by 185 nm VUV light and did not required excess of electrical energy.
3. Results and discussion
3.1. Transformation rates of diuron and monuron
UV photolysis proved to be efficient for the transformation of the target substances (Fig. 2a and 3a and Table 1) because of the high molar absorptivities of diuron (16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 050 M−1 cm−1) and monuron (13
050 M−1 cm−1) and monuron (13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 990 M−1 cm−1), determined at 254 nm. The data obtained are in accordance with the data reported by Sanches et al.20 and Schöler.63 In the case of photoionization, dissolved molecular oxygen generally enhances the rate of transformation because of its reaction with the hydrated electron (k(O2 + eaq− → O2˙−) = 1.9 × 1010 M−1 s−1).64 This reaction inhibits the recombination of the hydrated electron and the radical ion formed from the organic substance, and consequently dissolved oxygen causes higher rate of transformation.65 In our cases however, dissolved oxygen has no effect on the rates of transformation (Fig. 3a and d), which suggests that photoionization is insignificant in the UV-initiated transformation of both diuron and monuron. The rates of transformation of monuron (5.29 × 10−7 M s−1 (oxygen); 4.66 × 10−7 M s−1 (air); 4.83 × 10−7 mol dm−3 s−1 (nitrogen)) were approximately 2.3–3.3-fold those of diuron (1.76 × 10−7 M s−1 (oxygen); 1.77 × 10−7 M s−1 (air); 2.14 × 10−7 M s−1 (nitrogen)), in spite of the molar absorptivity of diuron exceeding that of monuron. This can be explained by the two times higher quantum yield of the direct photo-initiated transformation of monuron (0.05)63 than that of diuron (0.019).17,20
990 M−1 cm−1), determined at 254 nm. The data obtained are in accordance with the data reported by Sanches et al.20 and Schöler.63 In the case of photoionization, dissolved molecular oxygen generally enhances the rate of transformation because of its reaction with the hydrated electron (k(O2 + eaq− → O2˙−) = 1.9 × 1010 M−1 s−1).64 This reaction inhibits the recombination of the hydrated electron and the radical ion formed from the organic substance, and consequently dissolved oxygen causes higher rate of transformation.65 In our cases however, dissolved oxygen has no effect on the rates of transformation (Fig. 3a and d), which suggests that photoionization is insignificant in the UV-initiated transformation of both diuron and monuron. The rates of transformation of monuron (5.29 × 10−7 M s−1 (oxygen); 4.66 × 10−7 M s−1 (air); 4.83 × 10−7 mol dm−3 s−1 (nitrogen)) were approximately 2.3–3.3-fold those of diuron (1.76 × 10−7 M s−1 (oxygen); 1.77 × 10−7 M s−1 (air); 2.14 × 10−7 M s−1 (nitrogen)), in spite of the molar absorptivity of diuron exceeding that of monuron. This can be explained by the two times higher quantum yield of the direct photo-initiated transformation of monuron (0.05)63 than that of diuron (0.019).17,20
| UV photolysis | Ozonation | Combination of UV photolysis with ozonation | Heterogeneous photocatalysis | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N2 | Air | O2 | Air | O2 | Air | O2 | Air | O2 | ||||||||||
| r 0 | t 1/2 | r 0 | t 1/2 | r 0 | t 1/2 | r 0 | t 1/2 | r 0 | t 1/2 | r 0 | t 1/2 | r 0 | t 1/2 | r 0 | t 1/2 | r 0 | t 1/2 | |
| Monuron | 48.3 | 1.5 | 46.6 | 1.5 | 52.9 | 1.5 | 0.49 | n.d. | 0.87 | 90 | 75.5 | 1.0 | 78.9 | 1.0 | 6.76 | 15 | 7.05 | 15 |
| Diuron | 21.4 | 3.5 | 17.7 | 4.0 | 17.6 | 4.0 | 0.32 | n.d. | 0.70 | 120 | 24.9 | 3.0 | 26.7 | 3.0 | 4.33 | 15 | 4.15 | 16 |
In the case of ozonation, the concentration of ozone in the gas phase was 1.37 × 10−5 M and 4.04 × 10−6 mol dm−3, and that of dissolved ozone in pure Milli-Q bidistilled water was 2.3 × 10−6 M and 1.04 × 10−6 M when oxygen or air was used, respectively. Consequently, on the use of oxygen, the rate of degradation were about 2 times higher (7.0 × 10−9 M s−1 (diuron); 8.7 × 10−9 M s−1 (monuron)) than in the case of air (3.2 × 10−9 M s−1 (diuron); 4.9 × 10−9 M s−1 (monuron)) (Fig. 2b and 3b and Table 1). In the presence of ozone, transformation of the target substances can be initiated by reaction with molecular ozone or by reaction with radicals formed via the decomposition of ozone. During the transformation of these compounds, the pH decreased from 6.9 to 3.8 and from 8.3 to 4.1 in the case of monuron and diuron, respectively. The decomposition of ozone in aqueous solution is initiated by the hydroxide ion, but under neutral and acidic conditions a direct reaction with molecular ozone is more expected. The negligible effect of methanol (1.0 × 10−2 M) as hydroxyl radical scavenger also confirmed that, the relative contribution of radical based reaction to the transformation of these pesticides is not considerable. The phenylurea-derived pesticides react slowly or moderately fast with ozone. Moreover the Cl attached to the aromatic ring reduces its reactivity toward the electrophilic attack of O3 to the aromatic ring. The second-order rate constant for the reaction between diuron and ozone has been reported to be in the range 13.3–16.3 M s−1.23,24,66 This may be one of the reasons why the rates of transformation during ozonation were found to be more than one magnitude lower than those of direct UV photolysis. Moreover, the ozone was produced via emitted 185 nm VUV light of the lamp and its concentration was relative low. A significant difference between the initial rates of transformation of diuron and monuron was not found in this study, although the value determined for diuron, containing two Cl atoms, was lower than that of monuron containing only one Cl.
The combination of ozonation with UV photolysis resulted in a more effective method and increased the transformation rates comparing to those determined in the case of simple UV photolysis (diuron: r0(UV/O3)/r0(UV) = 1.4–1.5; monuron: r0(UV/O3)/r0(UV) = 1.5–1.6) (Fig. 2 and 3). When these methods are applied simultaneously, the 254 nm light has two functions: (i) initiating the decomposition of the aromatic target substances by direct photolysis, and (ii) enhancing the concentration of oxygen-containing radicals such as hydroxyl radical, due to the photoinitiated decomposition of ozone. A two-step process is proposed involving the light-induced homolysis of ozone and the subsequent production of hydroxyl radical by the reaction of O(1D) with water.
| O3 + hν<310nm → O2 + O(1D) | (1) |
| O(1D) + H2O → 2˙OH | (2) |
The 254 nm photolysis resulted in the concentration of dissolved ozone being one magnitude lower (3.5 × 10−7 M) than in the case of simple ozonation (2.3 × 10−6 M) because of the UV photolysis of dissolved ozone. Thus, the concentration of hydroxyl radical must be increased. The rate constants of the reactions of the hydroxyl radicals with diuron (4.8 × 109 M s−1)32,67 and monuron (7.3 × 109 M s−1)32,67 considerably exceed those with ozone. Consequently, the relative contribution of radical-based reactions to the decomposition of organic target substances is enhanced and this could be responsible for the higher rate of decomposition comparing to the simple ozonation or UV photolysis. At the same time, similarly to the UV-induced photolysis, there was no significant difference between the rates of transformation in aerated (2.49 × 10−7 M s−1 (diuron); 7.55 × 10−7 M s−1 (monuron)) and oxygen-saturated solutions (2.67 × 10−7 M s−1 (diuron); 7.89 × 10−7 M s−1 (monuron)), in part because of the high relative contribution of the direct UV photolysis to the transformation of these organic substances, even in the presence of hydroxyl radicals. This was proved by the effect of methanol. The addition of methanol (1.0 × 10−2 M) as hydroxyl radical scavenger only slightly decreased the transformation rate (rmonuron0(MeOH)/rmonuron0 = 0.73 and rdiuron0(MeOH)/rdiuron0 = 0.80) confirming the important role of direct UV photolysis, beside hydroxyl radical based reactions, even in the case of the combination of methods.
Using heterogeneous photocatalysis the transformation of organic substances (S) can be initiated by direct charge transfer via photogenerated charges (hole (hVB+))
| hVB+ + S → S˙+ | (3) |
| hVB+ + H2O → HO˙ + H+ | (4) |
| hVB+ + OH− → HO˙ | (5) |
Another way is the further transformation of superoxide radical ion (O2˙−) which is generated via direct charge transfer from adsorbed oxygen molecule and photogenerated electron (eCB−)
| O2 + eCB− → O2˙− | (6) |
| HO2˙ ⇌ H+ + O2˙− | (7) |
| HO2˙ + O2˙− + H+ → H2O2 + O2 | (8) |
The adsorbed H2O2 can transform via direct charge transfer and result partly in hydroxyl radical
| H2O2(ads) + eCB− → HO˙ + OH− | (9) |
| H2O2(aq) + eCB− → HO˙ + OH− | (10) |
| H2O2(ads) + hVB+ → O2˙− + 2H+ | (11) |
Both herbicides are only negligibly adsorbed.68,69 At the given initial concentration in 1.0 g dm−3 TiO2 suspension less than 4% of diuron or monuron was adsorbed in the conditions used. The initial rate of transformation of monuron (air: 6.76 × 10−8 M s−1; oxygen: 7.05 × 10−8 M s−1) was higher than diuron (air: 4.33 × 10−8 M s−1; oxygen: 4.15 × 10−8 M s−1), similarly to the rate constants of their reactions with hydroxyl radical. The values reported by Oturan et al. are 7.3 × 109 and 4.8 × 109 M s−1 for monuron and diuron, respectively.32 The ratio of the initial rates of transformation (rmonuron0/rdiuron0 = 1.6–1.7) is similar to the ratio of the reaction rate constants (kmonuron/kdiuron = 1.5), which confirms that their degradation was initiated by photogenerated hydroxyl radicals. Krýsa et al.69 reported similar consequence in the case of photocatalytic transformation of monuron. The significant negative effect of 1.0 × 10−2 M methanol, as hydroxyl radical scavenger, (rmonuron0(MeOH)/rmonuron0 = 0.29 and rdiuron0(MeOH)/rdiuron0 = 0.35) confirmed the essential role of hydroxyl radical.
Sharp, nearly linear decrease in the monuron and diuron concentration was observed during UV photolysis, ozonation and their combination until ∼70% elimination. In the photocatalytic systems there is exponential-like decrease of the concentration with time during the whole treatment, probably because of the strong competition between diuron/monuron and the formed intermediates, which are adsorb better than the parent compounds and successfully compete for the reactive species.
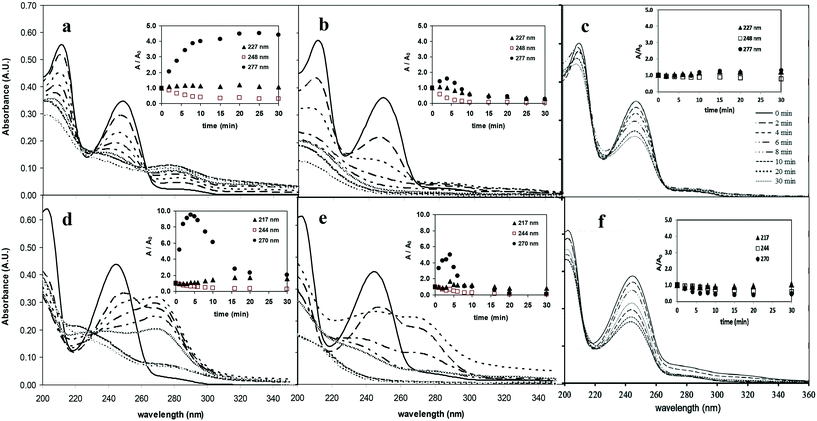
3.2. Aromatic intermediates
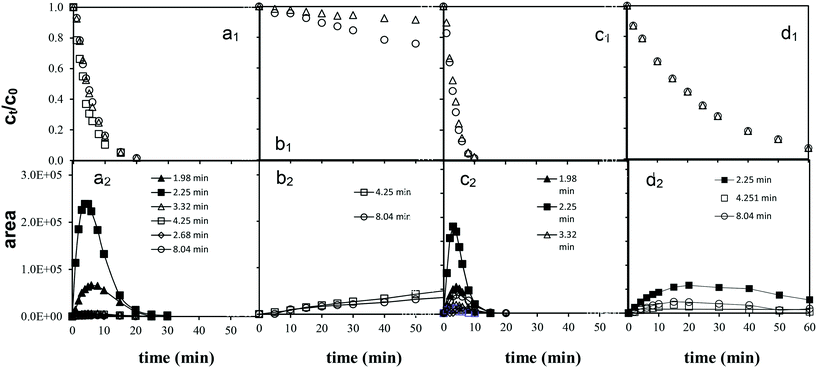
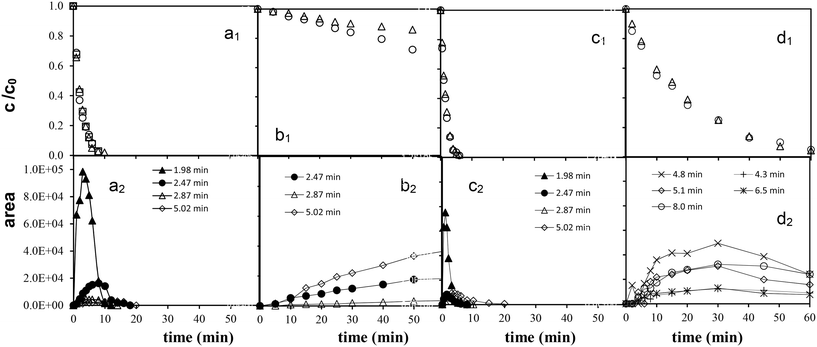
In UV irradiated solutions, the absorbance at 248 and 244 nm (the absorbance maximum of diuron and monuron, respectively) decreased in parallel with the concentration of phenylurea pesticides. The time dependence of the relative absorption at 277 and 270 nm suggested the occurrence not only of the formation, but also the decomposition of these aromatic intermediates, in parallel with the decomposition of the parent compounds. After the decomposition of 60–70% of pesticides the concentration of these intermediates decreased sharply. The transformation of both chlorinated substances by ozonation is a very slow process. The spectra do not change significantly during 30 min of treatment therefore they are not presented here.
The colourless aqueous solution of diuron became yellowish-pink when UV photolysis was applied, while that of monuron became pink. When the combination of UV photolysis with ozonation was employed, the intensity of the colour change was weaker. Each solution became colourless shortly after the decomposition of the target substances. On ozonation and heterogeneous photocatalysis no change in colour was observed, and there was no increase in the absorbance at 277 and 270 nm. During ozonation two main pathways of degradation were suggested for both target compounds: N-demethylation and OH-substitution of a Cl atom on the phenyl ring.21 In addition, the main transformation ways of monuron using heterogeneous photocatalysis include the hydroxylation of the aromatic ring too.18,25 In the cases of reaction with molecular ozone and hydroxyl radical based transformation probably the formation of ring-opened products are more favourable than the formation of aromatic intermediates.
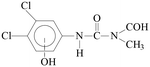
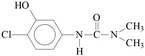
The MS results (Table 2) indicated that the transformations of diuron and monuron commenced with oxidation of the N-terminus group, in parallel with hydroxylation and dehalogenation of the aromatic ring. The time dependence of the areas of the peaks in the chromatograms confirmed the occurrence of the formation and transformation of the aromatic intermediates together with the decomposition of the parent compounds. The addition of ozone to the UV-irradiated solution strongly enhanced the rates of transformation and decomposition of the aromatic substances. These observations are in agreement with the spectrophotometric measurements: relative absorbance determined at 277 nm (diuron) and 270 nm (monuron) increased much sharply in UV-irradiated solution and reached higher value than in the case of combination with ozonation (Fig. 4a, b, d and e).
Quantification of the intermediates demonstrated that, demethylation and oxidation of the N-terminus groups on ozonation was more favorable than dehalogenation of the aromatic ring. Rather than dechlorination, the formation of ring-opened products appeared to be preferred.
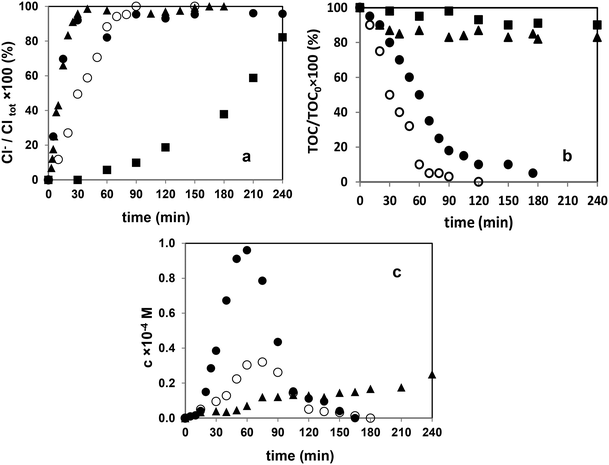
3.3. Mineralization and dehalogenation
As a result of the dehalogenation of diuron and monuron, as well as their intermediates, chloride ion was formed (Fig. 5a). There was no significant difference between the rate of accumulation of chloride ion and hence, the rate of dehalogenation between UV photolysis alone and its combination with ozonation. In solutions of diuron treated with these two methods, the concentration of chloride ion reached a maximum after 20 min. During this period, more than 90% of diuron decomposed in both cases. The ozonation alone was less effective in the decomposition of diuron, but dehalogenation also proceeded during the period needed for the transformation of diuron (about 240 min). Similar observations were made in the case of monuron. In all cases the transformation of Cl containing intermediates takes place simultaneously with diuron/monuron and after the transformation of the parent compounds there are no measurable chlorinated organic substances in the treated solution/suspension.Concerning mineralization, only the combination of UV photolysis with ozonation and heterogeneous photocatalysis were effective methods. In the case of ozonation and UV photolysis, the TOC concentration decreased only slightly (Fig. 5b). Radical based reactions and mineralization of organic pollutants in the presence of dissolved oxygen generally occurs through the formation of peroxyl radicals.65,70 Further fragmentation and transformation of peroxyl radicals result in HO2˙/O2˙− having low reactivity towards organic substances. The H2O2 mainly forms and accumulates due to the recombination of these species.
In this work, using the method based on catalysed transformation of leuco crystal violet, the sum of the concentration of H2O2 and methyl- and ethyl-hydroperoxides was measured. The obtained value was much higher using the combination of methods, than using UV-irradiation, and it was not measurable during ozonation (Fig. 5c). Using heterogeneous photocatalysis the reactions can take place on the TiO2 surface via direct charge transfer. In this case the decomposition of H2O2 is highly favored due to its reactions with photogenerated charges (both hVB+ and eCB−), which strongly inhibits its accumulation (9–11).
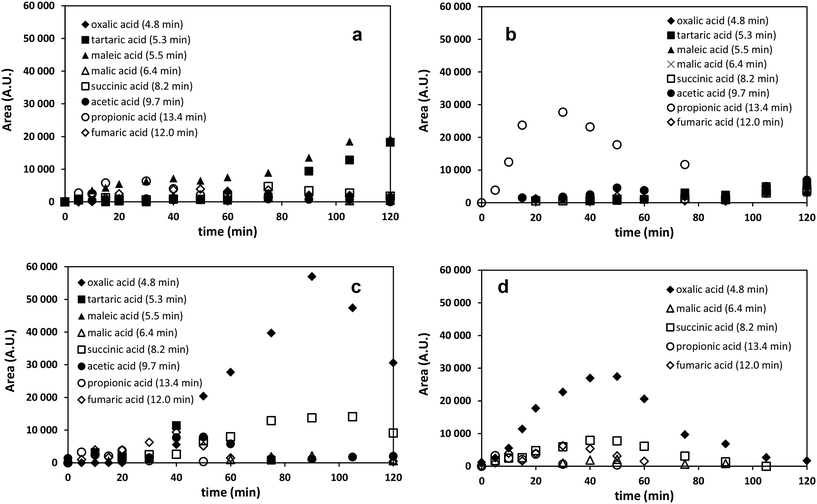
Aliphatic intermediates such as oxalic, fumaric, malic, maleic, acetic, succinic, propionic and tartaric acids were formed during all treatments, confirming the opening of the aromatic ring and further fragmentation (Fig. 6). The time dependence of the oxalic acid concentration is the most characteristic. Relative to UV photolysis or ozonation alone, the concentration of this compound was one magnitude higher when the combination of UV photolysis with ozonation was applied. In the case of single UV photolysis and ozonation only the slow accumulation was observed, while using their combination the time dependence of the oxalic acid concentration showed maximum curve (Fig. 6). Using heterogeneous photocatalysis the transformation of well adsorbed organic acids is quite fast, preventing their accumulation and enhancing the rate of mineralization. Thus, the maximum concentration of oxalic acid was lower comparing to the maximum concentration measured in the case of combination of UV photolysis with ozonation. For a typical reaction system containing photocatalyst, the photocatalytic oxidation of carboxylic acids follows a photo-Kolbe reaction mechanism: the decomposition results in the formation of CO2 as the primary reaction product and a carbon-centered radical as the primary reaction intermediate.
3.4. Economic efficiency – electrical energy per order (EEO)
To compare the economic efficiency of the applied AOPs the values of electrical energy per order (EEO) were calculated for both transformation (EcEO) and mineralization (ETOCEO) of monuron and diuron. The EcEO value was not calculated for ozonation, since its efficiency was negligible. For the same reason, ETOCEO values were calculated only for the combination of UV photolysis with ozonation and heterogeneous photocatalysis.As the values presented on Fig. 7 show, the energy requirement of UV photolysis and its combination with ozonation is about one magnitude lower than that of heterogeneous photocatalysis. In the respect of mineralization the heterogeneous photocatalysis is definitely the most feasible method, and needs about the half of the energy requirement of the combination of UV photolysis with ozonation. Using heterogeneous photocatalysis the EcEO and ETOCEO values are close to each other. Opposite to this, the ETOCEO value of the combined method is more than one magnitude higher than EcEO value, most probably because UV photolysis is quite inefficient in the transformation of intermediates which do not absorb 254 nm light, while heterogeneous photocatalysis is an unselective method for the transformation of various type of organic substances.
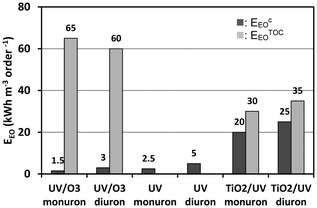 | ||
| Fig. 7 Values of electrical energy per order (EEO) calculated for transformation (EcEO) and mineralization (ETOCEO) of monuron and diuron. | ||
4. The matrix effect
4.1. Influence of inorganic salts and two natural matrices
Addition of inorganic salts has no significant effect in the case of ozonation, UV photolysis and their combination. Even addition of HCO3−, which can behave as hydroxyl radical scavenger (k = 8.5 × 106 M s−1)73 inhibited only slightly the transformation using the combined method (Fig. 8). The heterogeneous photocatalysis was found to be much more sensitive to the ion concentration. The addition of various salts, even HCO3−, increased the transformation rates. Surface properties can be changed strongly by the presence of inorganic ions and ionic strength in the suspension. These parameters affect not only the adsorption properties of the catalyst, but also the formation rate of hydroxyl radicals. Surprisingly, the effect was more significantly manifested in the case of diuron, than monuron.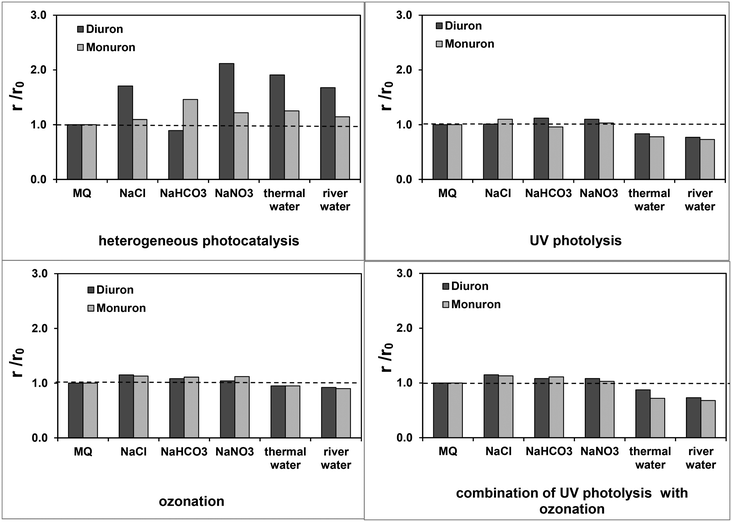 | ||
| Fig. 8 Relative rate of transformations (r: transformation rate determined in the presence of added salt or in thermal or river water; r0: transformation rate determined in MilliQ water). | ||
In this study, treatments were carried out in natural waters such as the water of the River Tisza and in thermal water. The rates of transformation of both target substances obtained in the mentioned matrices were compared with those measured in Milli-Q bidistilled water. Based on the results, no significant effect of these matrices, expect in the case of heterogeneous photocatalysis (Fig. 8) could be observed. This effect is most probably due to the high ionic strength of the thermal water, having relatively high HCO3− concentration. It is manifested in river water too, in spite of the presence of several components (mainly small amount of organic substances), that can behave as radical scavengers.
4.2. Effect of humic acids
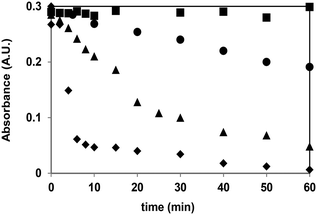
Humic acids can impact by several pathways the decomposition and transformation of organic molecules and are able to decrease or increase the rates of transformation of the target substances.36,71,72 At first, the efficiency of UV radiation, ozonation, their combination and heterogeneous photocatalysis on the elimination of humic acid was investigated. UV irradiation had no effect, in spite of the fact, that humic acid is able to absorb 254 nm UV light. There was no change of the absorbance during 60 min irradiation. In contrast, during ozonation the measured absorbance significantly decreased, proving that the transformation of humic acids (Fig. 9) can be initiated by the direct reaction with ozone, even at the very low ozone concentration applied in this work. The combination of these two methods is more efficient than simple ozonation, because of the well-known formation of the highly reactive hydroxyl radical.Humic acid is well adsorbed on the TiO2 surface. Less than 5 ppm humic acid is completely adsorbed in 1.0 g L−1 TiO2 suspension and was not measurable by spectrophotometric method after filtration. Using 20 ppm initial concentration, 13% of humic acid was adsorbed. Heterogeneous photocatalysis was proved to be widely the most effective method in the elimination of humic acid, comparing to other methods investigated in this work (rhumicacid0(TiO2/UV300–400nm ≫ rhumicacid0(O3/UV254nm > rhumicacid0(UV254nm) > rhumicacid0(O3)) (Fig. 8).
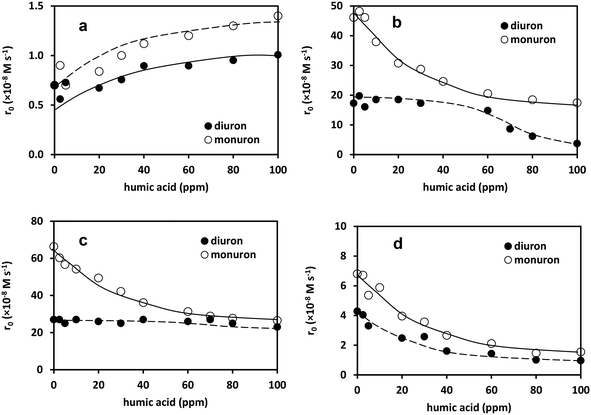
On ozonation, the presence of humic acids slightly increased the rate of transformation (Fig. 10a). The decomposition of ozone was probably positively affected by the humic acid. This effect could be attributed to the reactions of intermediates originated from the ozonation of humic acid, that behaves as a strong promoter of ozone decomposition. Consequently, the relative contribution of radical-based reactions to the transformation of diuron and monuron increased in the presence of humic acid and results in higher transformation rate of pesticides.
In UV-irradiated solutions, the humic acid caused a decrease in the rate of transformation of the pesticides, most likely because of acting as a “UV filter”. The resulting competitive light absorption is responsible for the decrease (Fig. 10b). In the case of monuron this negative effect is much more pronounced, most likely because of the higher quantum yield of the UV light irradiated transformation. In diuron solution the negative effect of humic acid was observed only above 60 ppm concentration.
When the combination of ozonation and UV photolysis was applied, the dependence of the initial rate of transformation on the concentration of humic acids was similar to that in the case of UV photolysis (Fig. 10c). With regards to the high molar absorbance of both diuron and monuron, the relative contribution of direct photolysis to the transformation remained dominant in the case of the UV-irradiated ozone-containing solutions. In this case humic acids can behave both as a “UV filter” and hydroxyl radical scavenger. Surprisingly, the negative effect of humic acid was observed only in the case of monuron, when the relative contribution of direct photolysis must be higher than in the case of diuron. Probably any kind of photosensitization is able to compensate the “UV filter” effect of humic acid in the latter case.
The negative effect of humic acid was well manifested in the case of heterogeneous photocatalysis, due to the favored adsorption of humic acid on the TiO2 surface and its competition with diuron/monuron for the formed hydroxyl radical. The behavior of monuron and diuron in the presence of humic acid is very similar in this case.
5. Conclusions
Decomposition of diuron and monuron by UV-induced photolysis, ozonation, their combination, and heterogeneous photocatalysis was investigated and compared at the same energy input. The UV photolysis was very effective in the transformation of the target pesticides. The addition of ozone enhanced the transformation rates due to the hydroxyl radical formation. However, the direct photolysis remained the dominant pathway for the initialization of transformation in the latter case. Mineralization was significant only using the combined method and heterogeneous photocatalysis. Definitely, heterogeneous photocatalysis was the most efficient and economically most feasible method from the mineralization point of view.Several aromatic intermediates were detected and identified. Demethylation and oxidation of the aliphatic chain occurred in parallel with the dechlorination and hydroxylation of the aromatic ring. The identified aromatic intermediates suggest that ring opening is more favourable in case of ozonation and heterogeneous photocatalysis than in case of UV photolysis and its combination with ozonation.
Humic acids behaved as “UV filter” and had a negative effect on the transformation rates in UV-irradiated solutions, but a positive effect in the case of ozonation, due to their behaviour as ozone transformation promoter, and consequently due to the increased radical concentration. The effects of natural waters (water of River Tisza and thermal water) as matrix, similarly to the effect of added inorganic salts, were negligible, except in case of heterogeneous photocatalysis, most likely due to the change of surface properties and the formation rate of hydroxyl radical.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
The financial support of the Swiss Contribution (SH7/2/20) is acknowledged and greatly appreciated. The authors acknowledge the bilateral financial support of the Tempus Public Foundation and the German Academic Exchange Service (TKA-DAAD 151955).References
- U. E. Bollmann, C. Tang, E. Eriksson, K. Jonsson, J. Vollertsen and K. Bester, Biocides in urban wastewater treatment plant influent at dry and wet weather: Concentrations, mass flows and possible sources, Water Res., 2014, 60, 64–74 CrossRef PubMed.
- D. B. Donald, A. J. Cessna, E. Sverko and N. E. Glozier, Pesticides in surface drinking-water supplies of the northern Great Plains, Environ. Health Perspect., 2007, 115, 1183–1191 CrossRef PubMed.
- M. Kuster, J. Lopez, M. de Alda, M. D. Dolores Hernando, M. Petrovic, J. Martín-Alonso and D. Barceló, Analysis and occurrence of pharmaceuticals, estrogens, progestogens and polar pesticides in sewage treatment plant effluents, river water and drinking water in the Llobregat river basin (Barcelona, Spain), J. Hydrol., 2008, 358, 112–123 CrossRef.
- C. Hao, B. Nguyen, X. Zhiao, E. Chen and P. Yang, Determination of Residual Carbamate, Organophosphate, and Phenyl Urea Pesticides in Drinking and Surface Water by High-Performance Liquid Chromatography/Tandem Mass Spectrometry, J. AOAC Int., 2010, 93, 400–410 Search PubMed.
- K. Nödler, D. Voutsa and T. Licha, Polar organic micropollutants in the coastal environment of differentmarine systems, Mar. Pollut. Bull., 2014, 85, 50–59 CrossRef PubMed.
- S. Rodriguez-Mozaz, M. J. L. de Alda and D. Barcelo, Monitoring of estrogens, pesticides and bisphenol A in natural waters and drinking water treatment plants by solid-phase extraction-liquid chromatography-mass spectrometry, J. Chromatogr. A, 2004, 1045, 85–92 CrossRef PubMed.
- O. Kolychalow, B. Schmalz, A. Matthiessen, G. Ostendorp, M. Hippelein and N. Fohrer, Pesticides and their metabolites in private drinking water wells in Schleswig-Holstein, Hydrol. Wasserbewirtsch., 2012, 56, 193–202 Search PubMed.
- C. Rodríguez, P. Taylor, B. Devine, P. Van Buynder, P. Weinstein and A. Cook, Assessing Health Risks from Pesticides in Recycled Water: A Case Study of Augmentation of Drinking Water Supplies in Perth Western Australia, Hum. Ecol. Risk Assess., 2012, 18, 1216–1236 CrossRef.
- S. Mostafalou and M. Abdollahi, Pesticides and human chronic diseases: Evidences, mechanisms, and perspectives, Toxicol. Appl. Pharmacol., 2013, 268, 157–177 CrossRef PubMed.
- S. Giacomazzi and N. Cochet, Environmental impact of diuron transformation: a review, Chemosphere, 2004, 56, 1021–1032 CrossRef PubMed.
- S. Chiron, A. Fernandez-Alba, A. Rodriguez and E. Garcia-Calvo, Pesticide chemical oxidation: state-of-the-art, Water Res., 2000, 34, 366–377 CrossRef.
- I. Oller, S. Malato and J. A. Sánchez-Pérez, Combination of Advanced Oxidation Processes and biological treatments for wastewater decontamination: A review, Sci. Total Environ., 2011, 409, 4141–4166 CrossRef PubMed.
- B. Weinberg and C. Teodisiu, Monitoring and assessment of herbicides removal by industrial wastewater treatment, Environ. Eng. Manage. J., 2012, 11, 215–224 CrossRef.
- S. Chusaksri, S. Sutthivaiyakit, D. L. Sedlak and P. Sutthivaiyakit, Reactions of phenylurea compounds with aqueous chlorine: Implications for herbicide transformation during drinking water disinfection, J. Hazard. Mater., 2012, 209, 484–491 CrossRef PubMed.
- F. J. Benitez, F. Acero, L. Juan and J. Francisco, Combination of chemical oxidation-membrane filtration processes for the elimination of phenyl-ureas in water matrices, J. Chem. Technol. Biotechnol., 2009, 84, 1883–1893 CrossRef.
- A. R. Ribeiro, O. C. Nunes, M. F. R. Pereira and A. M. T. Silva, An overview on the advanced oxidation processes applied for the treatment of water pollutants defined in the recently launched Directive 2013/39/EU, Environ. Int., 2014, 75, 33–51 CrossRef PubMed.
- F. J. Benitez, F. J. Real, J. L. Acero and C. Garcia, Photochemical oxidation processes for the elimination of phenyl-urea herbicides in waters, J. Hazard. Mater., 2006, 138, 278–287 CrossRef PubMed.
- M. Bobu, S. Wilson, T. Greibrokk, E. Lundanes and I. Siminiceanu, Comparison of advanced oxidation processes and identification of monuron photodegradation products in aqueous solution, Chemosphere, 2006, 63, 1718–1727 CrossRef PubMed.
- K. Djebbar, P. Sehili Mazellier and J. De Laat, Phototransformation of diuron in aqueous solution by UV irradiation in the absence and in the presence of H2O2, Environ. Technol., 2003, 24, 479–489 CrossRef PubMed.
- S. Sanches, M. T. Barreto Crespo and V. J. Pereira, Drinking water treatment of priority pesticides using low pressure UV photolysis and advanced oxidation processes, Water Res., 2010, 44, 1809–1818 CrossRef PubMed.
- L. A. Tahmasseb, S. Nélieu, L. Kerhoas and J. Einhorn, Ozonation of chlorophenylurea pesticides in water: reaction monitoring and degradation pathways, Sci. Total Environ., 2002, 291, 33–44 CrossRef PubMed.
- M. I. Maldonado, S. Malato, L. A. Pérez-Estrada, W. Gernjak, I. Oller, X. X. Doménech and J. Peral, Partial degradation of five pesticides and an industrial pollutant by ozonation in a pilot-plant scale reactor, J. Hazard. Mater., 2006, 138, 363–369 CrossRef PubMed.
- W. R. Chen, C. Wu, M. S. Elovitz, K. G. Linden and I. H. Suffet, Reactions of thiocarbamate, triazine and urea herbicides, RDX and benzenes on EPA Contaminant Candidate List with ozone and with hydroxyl radicals, Water Res., 2008, 42, 137–144 CrossRef PubMed.
- F. J. Benitez, F. J. Real, J. L. Acero and C. Garcia, Kinetics of the transformation of phenyl-urea herbicides during ozonation of natural waters: Rate constants and model predictions, Water Res., 2007, 41, 4073–4084 CrossRef PubMed.
- J. Fenoll, P. Sabater, G. Navarro, G. Pérez-Lucas and S. Navarro, Photocatalytic transformation of sixteen substituted phenylurea herbicides in aqueous semiconductor suspensions: Intermediates and degradation pathways, J. Hazard. Mater., 2013, 244–245, 370–379 CrossRef PubMed.
- Zs. Pap, V. Danciu and Zs. Cegled, The influence of rapid heat treatment in still air on the photocatalytic activity of titania photocatalysts for phenol and monuron degradation, Appl. Catal., B, 2011, 101, 461–470 CrossRef.
- H. Mestankova, G. Mailhot and J. Jirkovsky, Effect of iron speciation on the photodegradation of Monuron in combined photocatalytic systems with immobilized or suspended TiO2, Environ. Chem. Lett., 2009, 7, 127–132 CrossRef.
- M. P. Ormad, N. Miguel, M. Lanao, R. Mosteo and J. L. Ovelleiro, Effect of Application of Ozone and Ozone Combined with Hydrogen Peroxide and Titanium Dioxide in the Removal of Pesticides From Water, Ozone: Sci. Eng., 2010, 32, 25–32 CrossRef.
- R. Rajeswari and S. Kanmani, Degradation of Pesticide by Photocatalytic Ozonation Process and Study of Synergistic Effect by Comparison with Photocatalysis and UV/Ozonation Processes, J. Adv. Oxid. Technol., 2009, 12, 208–214 Search PubMed.
- R. Rajeswari and S. Kanmani, Comparative study on photocatalytic oxidation and photolytic ozonation for the degradation of pesticide wastewaters, Desalination, 2010, 19, 301–306 CrossRef.
- G. Simon, T. Gyulavári, K. Hernádi, M. Molnár, Z. Pap, G. Veréb, K. Schrantz, M. Náfrádi and T. Alapi, Photocatalytic ozonation of monuron over suspended and immobilized TiO2–study of transformation, mineralization and economic feasibility, J. Photochem. Photobiol., A, 2018, 356, 512–520 CrossRef.
- M. A. Oturan, M. C. Edelahi, N. Oturan, K. El Kacemi and J.-J. Aaron, Kinetics of oxidative degradation/mineralization pathways of the phenylurea herbicides diuron, monuron and fenuron in water during application of the electro-Fenton process, Appl. Catal., B, 2010, 97, 82–89 CrossRef.
- H. Mestankova, B. Escher, K. Schirmer, U. von Gunten and S. Canonica, Evolution of algal toxicity during (photo)oxidative degradation of diuron, Aquat. Toxicol., 2011, 101, 466–473 CrossRef PubMed.
- M. A. Oturan, N. Oturan, M. C. Edelahi, F. I. Podvorica and K. El Kacemib, Oxidative degradation of herbicide diuron in aqueous medium by Fenton's reaction based advanced oxidation processes, J. Chem. Eng., 2011, 171, 127–135 CrossRef.
- S. Pesce, S. Lissalde, D. Lavieille, C. Margoum, N. Mazzella, V. Roubeix and B. Montuelle, Evaluation of single and joint toxic effects of diuron and its main metabolites on natural phototrophic biofilms using a pollution-induced community tolerance (PICT) approach, Aquat. Toxicol., 2010, 99, 492–499 CrossRef PubMed.
- A. C. Gerecke, S. Canonica, S. R. Müller, M. Schärer and R. P. Schwarzenbach, Quantification of Dissolved Natural Organic Matter (DOM) Mediated Phototransformation of Phenylurea Herbicides in Lakes, Environ. Sci. Technol., 2001, 35, 3915–3923 CrossRef PubMed.
- S. Liu, M. Lim, R. Fabris, C. Chow, M. Drikas and R. Amal, Comparison of photocatalytic degradation of natural organic matter in two Australian surface waters using multiple analytical techniques, Org. Geochem., 2010, 41, 124–129 CrossRef.
- S. S. Kavurmaci and M. Bekbolet, Photocatalytic degradation of humic acid in the presence of montmorillonite, Appl. Clay Sci., 2013, 75–76, 60–66 CrossRef.
- B. R. Eggins, F. L. Palmer and J. A. Bryne, Photocatalytic treatment of humic substances in drinking water, Water Res., 1997, 31, 1223–1226 CrossRef.
- M. Kamiya and K. Kameyama, Photochemical effects of humic substances on the degradation of organophosphorous pesticides, Chemosphere, 1998, 36, 2337–2344 CrossRef.
- H. Prosen and L. Zupancic-Kralj, Evaluation of photolysis and hydrolysis of atrazine and its first degradation products in the presence of humic acids, Environ. Pollut., 2005, 133, 517–529 CrossRef PubMed.
- P. S. Bailey, Ozonation in organic chemistry, Vol. II. Nonolefininc Compounds, Academic Press, Elsevier, 1982 Search PubMed.
- C. Minero, F. Catozzo and E. Pelizetti, Role of adsorption in photocatalyzed reactions of organic molecules in aquesus TiO2 suspensions, Langmuir, 1992, 8, 481–486 CrossRef.
- E. Kudlek, M. Dudziak and J. Bohdziewicz, Influence of inorganic ions and organic substances on the degradation of pharmaceutical compound in water matrix, Water, 2016, 8, 532 CrossRef.
- D. D. Sun and P. F. Lee, TiO2 microsphere for the removal of humic acid from water: Complex surface adsorption mechanisms, Sep. Purif. Technol., 2012, 91, 30–37 CrossRef.
- M. Zhu, H. Wang, A. A. Keller, T. Wang and F. Li, The effect of humic acid on the aggregation of titanium dioxide nanoparticles under differ pH and ionic strengths, Sci. Total Environ., 2014, 487, 375–380 CrossRef PubMed.
- E. Tombácz, Talajreleváns határfelületi és kolloid kölcsönhatások, Glove Edit, Mauritius, 2017 Search PubMed.
- S. Tsuneda, Y. Ishihara, M. Hamachi and A. Hirata, Inhibition effect of chlorine ion on hydroxyl radical generation in UV-H2O2 process, Water Sci. Technol., 2002, 46, 33–38 CrossRef PubMed.
- H. Barndõk, D. Hermosilla, L. Cortijo, C. Negro and Á. Blanco, Assessing the effect of inorganic anions 1 on TiO2-photocatalysis and ozone oxidation treatment efficiencies, J. Adv. Oxid. Technol., 2016, 15, 125–132 Search PubMed.
- T. Jasemizad, M. T. Ghaneian, M. H. Ehrampoush, I. Rezapour, A. Jebali and F. Sahlabadi, Effects of radical scavengers on humic acid removal by electron beam irradiation, Desalin. Water Treat., 2016, 57, 29036–29043 CrossRef.
- H. Y. Chen, H. Y. Zahraa and M. Bouchy, Inhibition of the adsorption and photocatalytic degradation of an organic contaminant in an aqueous suspension of TiO2 by inorganic ions, J. Photochem. Photobiol., A, 1997, 108, 37–44 CrossRef.
- Y. Cho and W. Choi, Visible light-induced reactions of humic acids on TiO2, J. Photochem. Photobiol., A, 2002, 148, 129–135 CrossRef.
- K. Kovács, J. Farkas, G. Veréb, E. Arany, G. Simon, K. Schrantz, A. Dombi, K. Hernádi and T. Alapi, Comparison of various advanced oxidation processes for the degradation of phenylurea herbicides, J. Environ. Sci. Health, Part B, 2016, 1–10 Search PubMed.
- C. G. Hatchard and C. A. Parker, A New Sensitive Chemical Actinometer. II. Potassium Ferrioxalate as a Standard Chemical Actinometer, Proc. R. Soc. London, Ser. A, 1956, 235, 518–536 CrossRef.
- T. Alapi, L. Berecz, E. Arany and A. Dombi, Comparison of the UV-Induced Photolysis, Ozonation, and Their Combination at the Same Energy Input Using a Self-Devised Experimental Apparatus, Ozone: Sci. Eng., 2013, 35, 350–358 CrossRef.
- M. V. Shankar, S. Néieu, L. Kerhoas and J. Einhorn, Photo-induced degradation of diuron in aqueous solution by nitrites and nitrates: Kinetics and pathways, Chemosphere, 2007, 66, 767–774 CrossRef PubMed.
- E. Gere, B. Bérczi, P. Simándi, Gy. Wittmann and A. Dombi, Simultaneous Determination of Hydrogen Peroxide and Organic Hydroperoxides in Water, Int. J. Environ. Anal. Chem., 2002, 82, 443–448 CrossRef.
- L.-S. Zhang and G. T. F. Wong, Spectrophotometric determination of H2O2 in marine waters with leuco crystal violet, Talanta, 1994, 41, 2137–2145 CrossRef PubMed.
- R. Atkinson, D. L. Baulch, R. A. Cox, R. F. Hampson, J. A. Kerr, M. J. Rossi and J. Troe, Evaluated Kinetic, Photochemical and Heterogeneous Data for Atmospheric Chemistry: Supplement V. IUPAC Subcommittee on Gas Kinetic Data Evaluation for Atmospheric Chemistry, J. Phys. Chem. Ref. Data, 1997, 26, 521–547 CrossRef.
- H. Bader and J. Hoigne, Determination of ozone in water by the indigo method, Water Res., 1981, 15, 449–456 CrossRef.
- H. Bader and J. Hoigne, Determination of Ozone In Water By The Indigo Method: A Submitted Standard Method, Ozone: Sci. Eng., 1982, 4, 169–176 CrossRef.
- J. R. Bolton, K. G. Bircher, W. Tuman and C. A. Tolman, Figure-of-merit for the technical development and application of advanced oxidation technologies for both electric- and solar-driven systems, Pure Appl. Chem., 2001, 73, 627–637 Search PubMed.
- N. A. Schöler, Photoabbau von Herbiziden in Wasser durch UV-Strahlung aus Hg-Niederdruck-Strahlern, Vom Wasser, 1996, 86, 57–92 Search PubMed.
- A. J. Elliot, A pulse radiolysis study of the temperature dependence of reactions involving H, OH and e-aq in aqueous solutions, Radiat. Phys. Chem., 1989, 34, 753–758 CrossRef.
- T. Alapi and A. Dombi, Comparative study of the UV and UV/VUV-induced photolysis of phenol in aqueous solution, J. Photochem. Photobiol., A, 2007, 188, 409–418 CrossRef.
- J. De Laat, P. Maouala-Makata and M. Dore, Rate constants for reactions of ozone and hydroxyl radicals with several phenyl-ureas and acetamides, Environ. Technol., 1996, 17, 707–716 CrossRef.
- L. Wojnárovits and E. Takács, Rate coefficients of hydroxyl radical reactions with pesticide molecules and related compounds: A review, Radiat. Phys. Chem., 2014, 96, 120–134 CrossRef.
- K. Macounová, H. Krýsová, J. Ludvík and J. Jirkovský, Kinetics of photocatalytic degradation of diuron in aqueous colloidal solutions of Q-TiO2, J. Photochem. Photobiol., A, 2003, 156, 273 CrossRef.
- J. Krýsa, G. Waldner, H. Měštánková, J. Jirkovský and G. Grabnerd, Photocatalytic degradation of model organic pollutants on an immobilized particulate TiO2 layer: Roles of adsorption processes and mechanistic complexity, Appl. Catal., B, 2006, 64, 290–301 CrossRef.
- C. von Sonntag and H.-P. Schuchmann, Peroxyl radicals in aqueous solutions. in Alfassi, The Chemistry of Free Radicals: Peroxyl Radicals, John Wiley and sons, New York, 1997, pp. 173–234 Search PubMed.
- K. H. Chan and W. Chu, Effect of humic acid on the photolysis of the pesticide atrazine in a surfactant-aided soil-washing system in acidic condition, Water Res., 2005, 39, 2154–2166 CrossRef PubMed.
- J. R. Garbin, D. Miloria, L. M. Simões, T. L. W. da Silva and L. M. Neto, Influence of humic substances on the photolysis of aqueous pesticide residues, Chemosphere, 2007, 66, 1692–1698 CrossRef PubMed.
- G. V. Buxton, C. L. Greenstock, W. P. Helman and A. B. Ross, Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (˙OH/˙O-) in aqueous solution, J. Phys. Chem. Ref. Data, 1988, 17, 513–886 CrossRef.
| This journal is © The Royal Society of Chemistry 2018 |