Gold-catalyzed formation of indole derivatives from 2-alkynyl arylazides and oxygen-containing heterocycles†
Xiaoxiang
Zhang
*a,
Xiaoping
Sun
a,
Hui
Fan
b,
Chang
Lyu
a,
Ping
Li
a,
Haifei
Zhang
a and
Weidong
Rao
*a
aJiangsu Key Lab of Biomass-Based Green Fuels and Chemicals, College of Chemical Engineering, Nanjing Forestry University, Nanjing 210037, China. E-mail: zhangxiaoxiang@njfu.edu.cn; weidong@njfu.edu.cn
bSchool of Petrochemical Engineering, Changzhou University, Changzhou 213164, China
First published on 7th June 2016
Abstract
A novel method for the formation of indole derivatives via gold-catalyzed tandem reactions of 2-alkynyl arylazides and oxygen-containing heterocycles has been developed. A variety of indole derivatives were prepared under mild reaction conditions.
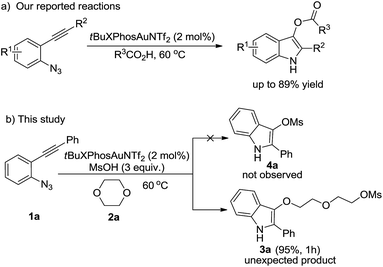
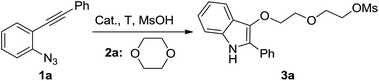
Indoles are very important core structures in organic chemistry, which can be found in many natural products and bioactive compounds.1 They are also serve as versatile building blocks for functional materials.2 For this reason, the development of a novel synthetic methodology to this class of N-containing heterocycles still remains highly active.3,4 Recently, an increasing number of studies have demonstrated the use of 2-alkynyl arylazides as efficient substrates in gold-catalyzed indole formation strategies.5 In our continuing studies on gold-catalyzed tandem reactions,6 we recently reported an efficient and convenient synthetic method to 1H-indol-3-yl esters via gold-catalyzed tandem reactions of 2-alkynyl arylazides with carboxylic acids (Scheme 1a).7 Based on these earlier studies, we reasoned 1-azido-2-(phenylethynyl)benzene 1a might also undergo a similar intermolecular trapping of putative α-imino gold carbene species5,8,9 generarated in situ by methanesulfonic acid. We anticipated the desired 2-phenyl-1H-indol-3-yl methanesulfonate 4a would be afforded under the similar gold-catalyzed reaction conditions, which could be a potential aryl mesylate cross-coupling reagent.10 In this reaction, 1,4-dioxane was used as solvent to dilute the methanesulfonic acid due to its highly activity. To our surprising, we did not detect the desired product 4a by analyzing the TLC and crud 1H NMR spectrum. However, only the unexpected ring-opening product 2-(2-((2-phenyl-1H-indol-3-yl)oxy)ethoxy)ethyl methanesulfonate 3a was obtained in 95% yield (Scheme 1b).11 Herein, we disclose the details of this efficient gold-catalyzed ring-opening reactions of 2-alkynyl arylazides with oxygen-containing heterocycles in the presence of methanesulfonic acid for the synthesis of novel indole derivatives.
Initially, we chose to focus our attention on ring-opening reaction of 1-azido-2-(phenylethynyl)benzene 1a and 1,4-dioxane in the presence of 3 equivalents of methanesulfonic acid by a variety of gold catalysts to establish the reaction conditions (Table 1). This revealed treating a solution of reaction containing 1a (1 equiv.) and 2a (0.5 mL) with 2 mol% tBuXPhosAuNTf2 in the presence of 3 equivalents of MsOH at 60 °C for 1 h gave the best result (entry 1). Under these conditions, 2-(2-((2-phenyl-1H-indol-3-yl)oxy)ethoxy)ethyl methanesulfonate 3a was obtained in 95% yield. On the other hand, a slightly lower product yields were observed when the gold catalyst was changed from tBuXPhosAuNTf2 to PPh3AuNTf2, XPhosAuNTf2 and IPrAuNTf2 (entries 2–4). In contrast, the use of AuCl3 as the catalyst led to a mixture of side products that could not be identified by 1H NMR analysis (entry 5). Notably, only the starting material was recovered when JohnPhosAuCl or AgNTf2 used as catalyst (entries 6–7). Similar result was obtained when this reaction was repeated in the absence of any gold catalyst (entry 8). The reaction temperature was also checked. High or low temperature led to a markedly lower yields of 59–71% (entries 9–10). Additional control experiment without MsOH led to a mixture of unknown products that could not be identified by TLC and 1H NMR analysis (entry 11). In contrast, other acid additives were also examined. The corresponding product 2-(2-((2-phenyl-1H-indol-3-yl)oxy)ethoxy)ethyl 4-methylbenzenesulfonate 5 was obtained in 61% yield where the MsOH was replaced with p-TsOH·H2O as the acid additive (entry 12). However, either no reaction or a mixture of unknown compounds were observed when TFA or TfOH was used as acid additive (entries 13–14).
| Entry | Catalyst | T (°C) | t (h) | Yieldb (%) |
|---|---|---|---|---|
a Unless stated otherwise, all reactions were performed in 0.5 mL 1,4-dioxane with a catalyst/1a/MsOH ratio of 0.02![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.
b Isolated yield.
c Mixture of unknown side products afforded based on 1H NMR analysis of the crude mixture.
d No reaction was observed. Only starting material left.
e Reaction conducted without catalyst.
f Reaction conducted without MsOH.
g
p-TsOH·H2O used as acid additive.
h TFA used as acid additive.
i TfOH used as acid additive. 3.
b Isolated yield.
c Mixture of unknown side products afforded based on 1H NMR analysis of the crude mixture.
d No reaction was observed. Only starting material left.
e Reaction conducted without catalyst.
f Reaction conducted without MsOH.
g
p-TsOH·H2O used as acid additive.
h TFA used as acid additive.
i TfOH used as acid additive.
|
||||
| 1 | tBuXPhosAuNTf2 | 60 | 1 | 95 |
| 2 | PPh3AuNTf2 | 60 | 1 | 92 |
| 3 | XPhosAuNTf2 | 60 | 1 | 93 |
| 4 | IPrAuNTf2 | 60 | 1 | 86 |
| 5 | AuCl3 | 60 | 1 | —c |
| 6 | JohnPhosAuCl | 60 | 1 | n.r.d |
| 7 | AgNTf2 | 60 | 1 | n.r.d |
| 8 | —e | 60 | 1 | n.r.d |
| 9 | PPh3AuNTf2 | r.t. | 24 | 59 |
| 10 | PPh3AuNTf2 | 80 | 1 | 71 |
| 11 | tBuXPhosAuNTf2 | 60 | 1 | —c,f |
| 12 | tBuXPhosAuNTf2 | 60 | 1 | 61g |
| 13 | tBuXPhosAuNTf2 | 60 | 4 | n.r.d,h |
| 14 | tBuXPhosAuNTf2 | 60 | 1 | —c,i |
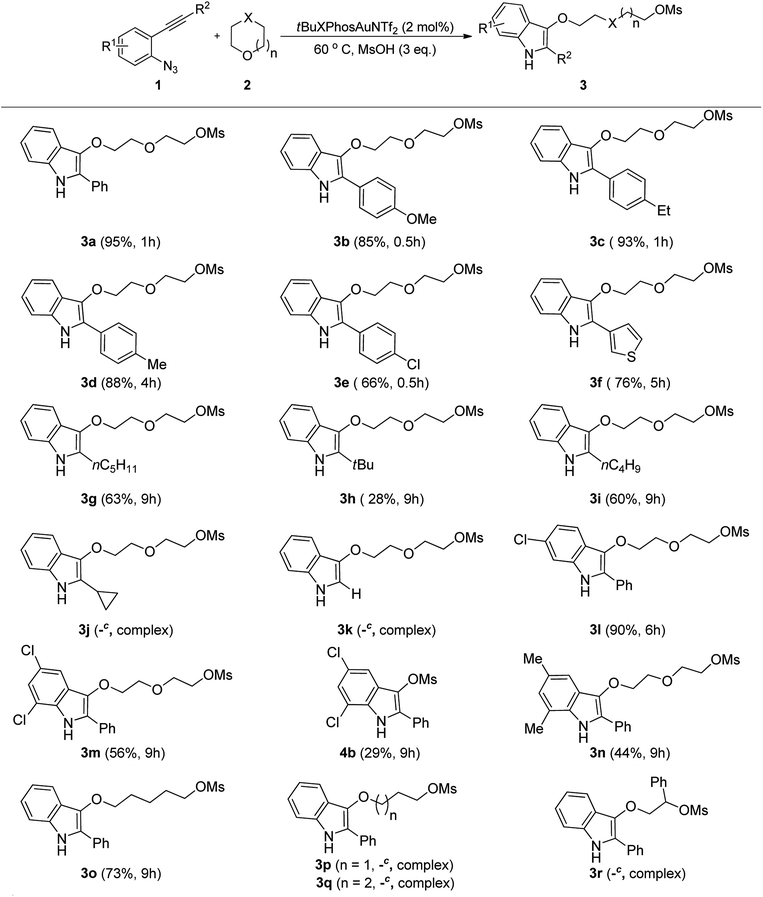
To define the scope of the present procedure, we next turned our attentions to the reactions of a variety of 2-alkynyl arylazides 1 with oxygen-containing heterocycles 2 (Table 2). These experiments showed that with tBuXPhosAuNTf2, 2-alkynyl arylazides bearing para-substituted electron-donating or electron-withdrawing substituents on the phenyl ring or thienyl group 1b–f efficiently underwent the tandem ring-opening process and gave the corresponding products 3b–f in good to excellent yields. We also tested the effect of an aliphatic substituent on the alkyne carbon and found that, for substrates where R2 = nC5H11 or nC4H9, lower yields 63% and 60% were obtained for 3g and 3i, respectively. Under the standard reaction conditions, the desired ring-opening indole 3 h was also provided with the sterically hindered substrate 1 h However, the substrate with cyclopropyl group 1j or terminal alkyne moiety 1k only gave a mixture of unknown side products that could not be identified by TLC and 1H NMR analysis. Next, arylazides with Cl or Me group on the phenyl ring 3l–n also can provide the desired indole derivatives in moderate to excellent yields. Notably, the 5,7-dichloro-2-phenyl-1H-indol-3-yl methanesulfonate 4b was obtained in 29% yield as a side product when the substrate with disubstituted group on the aromatic ring was employed. Next, other oxygen-containing heterocycles were also examined under the optimal reaction conditions. When dioxane was changed to tetrahydro-2H-pyran, we found that the corresponding ring-opening indole product 3o was afforded in 73% yield. Very interesting, the reaction of 1a with oxetane 2c, tetrahydrofuran 2d, or 2-phenyloxirane 2e only gave a mixture of unknown side products that could not be identified by TLC and 1H NMR analysis.
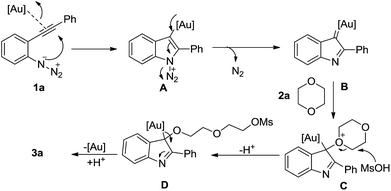
We tentatively propose the present tBuXPhosAuNTf2 catalyzed ring-opening indole-forming reaction to proceed by the mechanism outlined in Scheme 2, although it is highly speculative. This could involve activation of 1-azido-2-(phenylethynyl)benzene 1a through coordination of the gold catalyst with the alkyne, which would be attacked by the azide to deliver intermediate A. Subsequently, the α-imino gold carbene B was formed after the expulsion of N2 with the assist of gold catalyst, which was further attacked by 1,4-dioxane to release the intermediate C. Ring opening of C with methanesulfonic acid would provide the complex D, which undergo a final protodemetalation step to deliver the 2-(2-((2-phenyl-1H-indol-3-yl)oxy)ethoxy)ethyl methanesulfonate 3a.
In summary, an unexpected gold-catalyzed synthetic method to indole derivatives based on the ring-opening reaction of 2-alkynyl arylazides with oxygen-containing heterocycles in the presence of methanesulfonic acid has been reported. These results show that the reaction tolerates a structurally diverse set of 2-alkynyl arylazides substrates that can be prepared easily in two steps from commercially available materials. Moreover, the gold-catalyzed tandem reactions were also shown to be practical since inert and moisture-free conditions were not required. Further studies on the applications of this synthetic method are currently underway in our laboratory.
Acknowledgements
This project was supported by the National Natural Science Foundation of China Grant (21302096, 21502093), Natural Science Foundation of Jiangsu Province Grant (BK20130962, BK20150871) and the Project Fund from the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).Notes and references
- (a) X.-H. Cai, Z.-Z. Du and X.-D. Luo, Org. Lett., 2007, 9, 1817 CrossRef CAS; (b) X.-H. Cai, Q.-G. Tan, Y.-P. Liu, T. Feng, Z.-Z. Du, W.-Q. Li and X.-D. Luo, Org. Lett., 2008, 10, 577 CrossRef CAS; (c) T. Feng, X.-H. Cai, P.-J. Zhao, Z.-Z. Du, W.-Q. Li and X.-D. Luo, Planta Med., 2009, 75, 1537 CrossRef CAS; (d) X.-H. Cai, Y.-P. Liu, T. Feng and X.-D. Luo, Chin. J. Nat. Med., 2008, 6, 20 CAS; (e) X.-H. Cai, J.-H. Shang, T. Feng, X.-D. Z. Naturforsch and B. J. Luo, Chem. Sci., 2010, 65, 1164 CAS; (f) X.-J. Qin, Y.-L. Zhao, P.-K. Lunga, X.-W. Yang, C.-W. Song, G.-G. Cheng, L. Liu, Y.-Y. Chen, Y.-P. Liu and X.-D. Luo, Tetrahedron, 2015, 71, 4372 CrossRef CAS; (g) L. Liu, Y.-Y. Chen, X.-J. Qin, B. Wang, Q. Jin, Y.-P. Liu and X.-D. Luo, Fitoterapia, 2015, 105, 160 CrossRef CAS; (h) X.-J. Qin, Y.-L. Zhao, C.-W. Song, B. Wang, Y.-Y. Chen, L. Liu, Q. Li, D. Li, Y.-P. Liu and X.-D. Luo, Nat. Prod. Bioprospect., 2015, 5, 185 CrossRef CAS; (i) H. Zhou, H.-P. He, X.-D. Luo, Y.-H. Wang, X.-W. Yang, Y.-T. Di and X.-J. Hao, Helv. Chim. Acta, 2005, 88, 2508 CrossRef CAS; (j) L. Zhang, Acc. Chem. Res., 2014, 47, 877 CrossRef CAS; (k) A. S. K. Hashmi, Acc. Chem. Res., 2014, 47, 864 CrossRef CAS; (l) C. Obradors and A. M. Echavarren, Chem. Commun., 2014, 50, 16 RSC; (m) J. Xiao and X. Li, Angew. Chem., Int. Ed., 2011, 50, 7226 CrossRef CAS; (n) A. S. K. Hashmi, Angew. Chem., Int. Ed., 2010, 49, 5232 CrossRef CAS.
- G. Nie, Z. Bai, J. Chen and W. Yu, ACS Macro Lett., 2012, 1, 1304 CrossRef CAS.
- For recent reviews on the synthesis of indoles, see: (a) S. Cacchi and G. Fabrizi, Chem. Rev., 2011, 111, 215 CrossRef; (b) R. Vicente, Org. Biomol. Chem., 2011, 9, 6469 RSC; (c) S. Cacchi, G. Fabrizi and A. Goggiamani, Org. Biomol. Chem., 2011, 9, 641 RSC; (d) M. Bandini and A. Eichholzer, Angew. Chem., Int. Ed., 2009, 48, 9608 CrossRef CAS; (e) I. V. Seregin and V. Gevorgyan, Chem. Soc. Rev., 2007, 36, 1173 RSC; (f) G. R. Humphrey and J. T. Kuethe, Chem. Rev., 2006, 106, 2875 CrossRef CAS; (g) S. Cacchi and G. Fabrizi, Chem. Rev., 2005, 105, 2873 CrossRef CAS; (h) G. Zeni and R. C. Larock, Chem. Rev., 2004, 104, 2285 CrossRef CAS; (i) G. Balme, D. Bouyssi, T. Lomberget and N. Monteiro, Synthesis, 2003, 2115 CrossRef CAS.
- For recent selected examples on the synthesis of indoles, see: (a) J. Barluenga, F. Rodríguez and F. J. Fańanás, Chem.–Asian J., 2009, 4, 1036 CrossRef CAS; (b) O. Miyata, N. Takeda and T. Naito, Heterocycles, 2009, 78, 843 CrossRef CAS; (c) K. Krüger, A. Tillack and M. Beller, Adv. Synth. Catal., 2008, 350, 2153 CrossRef.
- (a) N. Li, T.-Y. Wang, L.-Z. Gong and L. Zhang, Chem.–Eur. J., 2015, 21, 1 CrossRef; (b) B. Lu, Y. Luo, L. Liu, L. Ye, Y. Wang and L. Zhang, Angew. Chem., Int. Ed., 2011, 50, 8358 CrossRef CAS.
- For our recent studies on the gold-catalyzed tandem reactions, see: (a) W. Rao, D. Susanti, B. J. Ayers and P. W. H. Chan, J. Am. Chem. Soc., 2015, 137, 6350 CrossRef CAS; (b) W. Rao, M. J. Koh, D. Li, H. Hirao and P. W. H. Chan, J. Am. Chem. Soc., 2013, 135, 7926 CrossRef CAS; (c) W. Rao, Sally, M. J. Koh and P. W. H. Chan, J. Org. Chem., 2013, 78, 3183 CrossRef CAS; (d) W. Rao, M. J. Koh, P. Kothandaraman and P. W. H. Chan, J. Am. Chem. Soc., 2012, 134, 10811 CrossRef CAS; (e) W. Rao, D. Susanti and P. W. H. Chan, J. Am. Chem. Soc., 2011, 133, 15248 CrossRef CAS.
- X. Zhang, X. Sun, H. Zhang, X. Cui and M. Ma, Chin. J. Inorg. Chem., 2015, 35, 1469 CAS.
- For recent selected reviews on the generation of gold carbenes, see: (a) R. Dorel and A. M. Echavarren, Chem. Rev., 2015, 115, 9028 CrossRef CAS; (b) D. Qian and J. Zhang, Chem. Soc. Rev., 2015, 44, 677 RSC; (c) Y. Wang, M. E. Muratore and A. M. Echavarren, Chem.–Eur. J., 2015, 21, 7332 CrossRef CAS; (d) F. Wei, C. Song, Y. Ma, L. Zhou, C.-H. Tung and Z. Xu, Sci. Bull., 2015, 60, 1479 CrossRef CAS; (e) H.-S. Yeom and S. Shin, Acc. Chem. Res., 2014, 47, 966 CrossRef CAS; (f) L. Fensterbank and M. Malacria, Acc. Chem. Res., 2014, 47, 953 CrossRef CAS PubMed; (g) C. Obradors and A. M. Echavarren, Acc. Chem. Res., 2014, 47, 902 CrossRef CAS PubMed.
- For recent selected a review on the generation of α-imino gold carbenes, see: P. W. Davies and M. Garzûn, Asian J. Org. Chem., 2015, 4, 694; for recent selected examples, see: 5a, 5b and (a) Y. Pan, G.-W. Chen, C.-H. Shen, W. He and L.-W. Ye, Org. Chem. Front., 2016, 3, 491 RSC; (b) H. Jin, L. Huang, J. Xie, M. Rudolph, F. Rominger and A. S. K. Hashmi, Angew. Chem., Int. Ed., 2016, 55, 794 CrossRef CAS; (c) A.-H. Zhou, Q. He, C. Shu, Y.-F. Yu, S. Liu, T. Zhao, W. Zhang, X. Lu and L.-W. Ye, Chem. Sci., 2015, 6, 1265 RSC; (d) C. Shu, Y.-H. Wang, B. Zhou, X.-L. Li, Y.-F. Ping, X. Lu and L.-W. Ye, J. Am. Chem. Soc., 2015, 137, 9567 CrossRef CAS; (e) L. Zhu, Y. Yu, Z. Mao and X. Huang, Org. Lett., 2015, 17, 30 CrossRef CAS; (f) S. K. Pawar, R. L. Sahani and R.-S. Liu, Chem.–Eur. J., 2015, 21, 10843 CrossRef CAS; (g) A. Prechter, G. Henrion, P. F. dit Bel and F. Gagosz, Angew. Chem., Int. Ed., 2014, 53, 4959 CrossRef CAS; (h) M. Garzón and P. W. Davies, Org. Lett., 2014, 16, 4850 CrossRef; (i) Y. Tokimizu, S. Oishi, N. Fujii and H. Ohno, Org. Lett., 2014, 16, 3138 CrossRef CAS; (j) E. Chatzopoulou and P. W. Davies, Chem. Commun., 2013, 49, 8617 RSC; (k) Z.-Y. Yan, Y. Xiao and L. Zhang, Angew. Chem., Int. Ed., 2012, 51, 8624 CrossRef CAS; (l) Y. Xiao and L. Zhang, Org. Lett., 2012, 14, 4662 CrossRef CAS; (m) P. W. Davies, A. Cremonesand and L. Dumitrescu, Angew. Chem., Int. Ed., 2011, 50, 8931 CrossRef CAS; (n) A. Wetzel and F. Gagosz, Angew. Chem., Int. Ed., 2011, 50, 7354 CrossRef CAS; (o) C. Li and L. Zhang, Org. Lett., 2011, 13, 1738 CrossRef CAS; (p) D. J. Gorin, N. R. Davis and F. D. Toste, J. Am. Chem. Soc., 2005, 127, 11260 CrossRef CAS.
- (a) C. F. Wai, M. S. Chau and Y. K. Fuk, Org. Lett., 2015, 17, 5906 CrossRef; (b) C. M. So and F. Y. Kwong, Chem. Soc. Rev., 2011, 40, 4963 RSC; (c) S. I. Kozhushkov, H. K. Potukuchi and L. Ackermann, Catal. Sci. Technol., 2013, 3, 562 RSC; (d) M. C. Wilkinson, Org. Lett., 2011, 13, 2232 CrossRef CAS; (e) G. A. Molander and I. Shin, Org. Lett., 2012, 14, 3138 CrossRef CAS.
- For a gold-catalyzed ring-opening reaction of tetrahydrofuran, see: W. Huang, J. Xiang and W. He, Chem. Lett., 2014, 43, 893 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental procedures and analysis data for new compounds. See DOI: 10.1039/c6ra09761h |
| This journal is © The Royal Society of Chemistry 2016 |