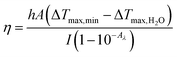Dual-responsive polydopamine-embellished Zn-MOFs enabling synergistic photothermal and antibacterial metal ion therapy for oral biofilm eradication†
Ge
Pan
ab,
Jinyao
Zheng
c,
Zongjia
Li
c,
Qian
Duan
 *a,
Miaomiao
Zhang
*b and
Dapeng
Wang
*a,
Miaomiao
Zhang
*b and
Dapeng
Wang
 *b
*b
aSchool of Materials Science and Engineering, Changchun University of Science and Technology, Changchun, 130022, P. R. China. E-mail: duanqian88@hotmail.com
bState Key Laboratory of Polymer Physics and Chemistry, Changchun Institute of Applied Chemistry Chinese Academy of Sciences, Changchun, 130022, P. R. China. E-mail: wdp@ciac.ac.cn; mmzhang@ciac.ac.cn
cState Key Laboratory of Electroanalytical Chemistry, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Changchun, 130022, P. R. China
First published on 11th February 2025
Abstract
Oral biofilms are associated with various oral diseases causing pain and discomfort, and pose a severe threat to general health. Conventional surgical debridement and antibacterial therapy often yield unsatisfactory outcomes because they either fail to fully and painlessly eliminate biofilms or increase the risk of bacterial resistance. In this study, we synthesized polydopamine-embellished Zn-MOFs (ZIF-8@PDA NPs), which can degrade under mildly acidic conditions to release Zn2+. These nanoparticles also convert near-infrared light energy into heat, thereby enabling synergistic photothermal and antibacterial metal ion therapy for oral biofilm eradication. Our findings reveal that therapy with ZIF-8@PDA NPs, when exposed to near-infrared radiation, demonstrates exceptional antibacterial efficacy and is highly effective in eradicating oral biofilms both in vitro and ex vivo. Furthermore, we used an in vivo rodent tooth biofilm model to demonstrate the suppression of dental caries. This work presents a promising solution for preventing and suppressing dental caries as well as other treating diseases linked to oral biofilm infections.
1. Introduction
The microbiomes residing within the human oral cavity are the second most abundant in the body.1,2 Insufficient oral hygiene, improper use of antibiotics, lifestyle factors (such as smoking), and alterations in saliva can precipitate dysbiosis of the oral microbiota, which is primarily composed of bacteria.3 This dysbiosis could culminate in the formation of a sophisticated and intricately organized assemblage known as the oral biofilm.4 Oral biofilms are associated with various oral diseases, including caries, periodontal disease, pulpitis, and candidiasis, which can cause pain and discomfort, and in severe cases, may lead to tooth loss or systemic health complications.1,5 Extracellular polymeric substances (EPSs) are complex mixtures of biopolymers produced by microorganisms within biofilms.5 The adhesion and protective effects of EPSs in microbial biofilms render innate immune cells and conventional antimicrobials inefficient in degrading oral biofilms and eradicating embedded microbes.6 In addition, pathogenic bacteria can ferment sugars from food sources. During this process, they metabolize sugars and produce acids as metabolic byproducts in the oral cavity.7 These acids can lower the pH levels in the immediate environment surrounding the bacteria, culminating in the onset of dental caries and other oral pathologies.5 Therefore, eradicating oral biofilms holds considerable significance for the prevention and treatment of oral biofilm-related diseases.The primary clinical treatments for oral biofilm infections currently involve mechanical debridement and antibiotic therapy.8 However, these methods have certain limitations. Surgical debridement, while effective in removing biofilms and diseased tissues, can lead to discomfort, pain, and potential complications such as bleeding or adverse reactions to anesthesia.9 Although antibiotic therapy has greatly improved treatment comfort, its overuse or inappropriate use can contribute to the development of drug resistance in oral pathogenic bacterial strains, making infections more difficult and costly to treat.10,11 Therefore, there is an urgent need to explore novel antibacterial strategies to effectively eradicate oral biofilms and prevent their recolonization. At present, novel antimicrobial strategies have been continuously developed,12 such as controlled release of antibiotics,13 polypeptides,14 nanoparticles,15–18 antibacterial metal ions,19,20 metal–organic frameworks,21,22 polymers,23 bacteriophages,24 antibacterial lysozymes,25etc. Many promising antibacterial therapy strategies based on nanoparticles have been developed,7 including photothermal therapy (PTT),26,27 photodynamic therapy (PDT),28–30 chemodynamic therapy (CDT),31–33 and so on. Every solution has its own unique advantages and limitations. For example, PTT exploits photothermal agents to convert light energy into heat, inducing local hyperthermia. This approach not only effectively kills bacteria but also achieves excellent non-mechanical removal of oral biofilms, making it a promising option for biofilm eradication.34–36 Although promising, PTT faces challenges in regulating the treatment temperature, as excessively high temperatures can damage surrounding tissues, while insufficient heat may result in incomplete bacterial ablation.37 Synergistic strategies have been proposed to tackle this issue, such as the integration of PTT with CDT or, PDT,27,34,38 immunotherapy,39,40 and so on.8,31,34,41,42 Nevertheless, applying PTT in oral biofilm therapy still faces two notable concerns: short action duration and limited permeability.43,44 These concerns could be addressed by synergistically combining bactericidal metal ions that are released on demand. Metal ions, such as zinc ions (Zn2+), exhibit broad-spectrum antibacterial activity by interfering with the metabolic process of infected bacteria.45 Zinc metal–organic frameworks (Zn-MOFs), composed of Zn2+ connected by multidentate organic linkers, can be regarded as ideal reservoirs of Zn2+ because Zn-MOFs could dissociate to release Zn2+via hydrolysis-induced decomposition when exposed to acidic conditions.46 Given their ability to release Zn2+ in response to pH changes, Zn-MOFs are expected to enhance PTT for antibacterial therapy and biofilm eradication, which are typically acidic due to bacterial metabolism.
In this work, zinc imidazolate framework-8 (ZIF-8) was chosen as the Zn2+ reservoir from various Zn-MOFs due to its simple synthesis procedure and pH-responsive characteristics, allowing it to release Zn2+ in the acidic environment of oral biofilms. The surface of ZIF-8 was coated with polydopamine (PDA) to prepare polydopamine-embellished Zn-MOFs (ZIF-8@PDA NPs), as shown in Fig. 1a. In our design, the PDA coating on the surface of ZIF-8 NPs not only facilitated the effective adhesion of ZIF-8@PDA NPs to the bacterial biofilm surface, but also endowed them with significant photothermal properties. Under 808 nm near-infrared (NIR) irradiation, ZIF-8@PDA NPs efficiently converted NIR light into heat, inducing local hyperthermia. Simultaneously, ZIF-8@PDA NPs gradually degraded in the mildly acidic microenvironment, releasing a significant amount of Zn2+ ions. These ions could penetrate the inner layers of the biofilm, effectively eradicating S. mutans and disrupting the biofilm they formed through a synergistic combination of photothermal effects and antibacterial metal ions (Fig. 1b). In addition, we established an ex vivo tooth infection model and an in vivo dental caries mouse model to study the effectiveness of ZIF-8@PDA NPs in removing oral biofilms.
2. Results and discussion
2.1. Preparation and characterization of ZIF-8@PDA NPs
ZIF-8@PDA NPs can be easily synthesized under mild conditions, as shown in Fig. 1a. Firstly, zinc acetate dihydrate (Zn(OAc)2·2H2O) and 2-methylimidazole (HmIm) were used to produce ZIF-8 NPs. Then, a PDA coating was naturally deposited onto the surface of ZIF-8 to form ZIF-8@PDA NPs. Our findings demonstrate a progressive enhancement in the photothermal effect of ZIF-8@PDA NPs with increasing PDA content (Fig. S1a, ESI†), however, this enhancement is accompanied by the potential for elevated temperature-induced thermal damage to tissues. Concurrently, the increase in the PDA proportion markedly decelerates the degradation rate of the material, resulting in a significant reduction in Zn2+ release (Fig. S1b, ESI†). To achieve an optimal balance between photothermal performance and Zn2+ release efficiency, an optimal ZIF-8 to PDA ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 was ultimately established.
2 was ultimately established.
The morphology of ZIF-8 NPs is characterized as a polyhedral structure with an average diameter of 440 nm, as illustrated in Fig. 2a–c. Zeta potential measurements indicated that the potential of ZIF-8 decreased from 9.6 mV to −0.3 mV after coating with PDA (Fig. 2d), attributable to the negative charge of PDA. The PDA coating has scarcely altered the morphology of ZIF-8 (Fig. 2e and f). Upon PDA coating, the diameter of resulting ZIF-8@PDA NPs is increased to 460 nm, suggesting that the thickness of the PDA coating is approximately 10 nm (Fig. 2h). The energy spectrum showed uniform distributions of the four elements: carbon (C), nitrogen (N), zinc (Zn), and oxygen (O) in ZIF-8@PDA NPs (Fig. 2g). Fig. 2i shows the XPS patterns of ZIF-8 NPs and ZIF-8@PDA NPs. Comparatively, we observed that two binding energy peaks appeared at 1021 and 1045 eV for ZIF-8 NPs, which can be attributed to the Zn 2p3/2 and Zn 2p1/2 orbitals (Fig. S2a, ESI†).47,48 In contrast, these peaks are absent for the ZIF-8@PDA NPs. This phenomenon has been previously observed and can be ascribed to the chelation of the catechol moiety of DA with the Zn2+ metal nodes.49 This is because the Zn 2p spectra are associated with the oxidation state of zinc, leading to the disruption of the Zn–Mim coordination bonds in the parent ZIF-8 NPs.49 The peaks emerged at 399.1 eV, 284.8 eV, and 533.0 eV in the XPS spectra of ZIF-8@PDA NPs correspond to N 1s, C 1s, and O 1s, respectively.50 Note that the X-ray diffraction pattern of ZIF-8@PDA NPs is identical to that of ZIF-8 NPs (Fig. S2b, ESI†), indicating that the PDA coating almost has not altered the crystal structure of ZIF-8 NPs.51 Moreover, the ultraviolet-visible (UV-vis) spectra provide evidence for the successful coating of polydopamine (PDA) on ZIF-8 NPs, as illustrated in Fig. S2c (ESI†). The FTIR spectrum of ZIF-8@PDA NPs reveals peaks at 1495 and 1600 cm−1 that were assigned to the benzene skeleton vibrations of PDA (Fig. 2j).52 Combined with the peak at 421 cm−1 corresponding to the Zn–N bond and the peaks at 1145 and 997 cm−1 corresponding to the C–N bond, FTIR results further confirmed the successful coating of PDA on ZIF-8 NPs. Taken together, all results confirmed the successful synthesis of ZIF-8@PDA NPs. Simultaneously, as the control group of the photothermal experiment, we synthesized PDA NPs, and the results presented in Fig. S3a and b (ESI†) indicate that their diameter is 250 nm.
2.2. pH responsiveness and photothermal effect of ZIF-8@PDA NPs
As mentioned in the introduction, pathogenic bacteria are capable of metabolizing sugars, thereby lowering pH in the oral microenvironment by producing acidic metabolic by-products. This acidic environment can lead to the demineralization of the tooth's hard tissue, contributing to the formation of caries' lesions. Conversely, the acidic conditions can also be leveraged as a stimulus to perform controlled antibacterial actions. Note that the framework structure of ZIF-8 could be disrupted in an acidic environment, leading to the release of the Zn2+ ions contained within.21 To evaluate the pH responsiveness of ZIF-8@PDA NPs, they were suspended in PBS solutions of varying pH for 2 h, and their morphology was examined via SEM (Fig. 3a). As the pH declined, the three-dimensional framework of ZIF-8@PDA NPs gradually disintegrated. When suspended in a solution at a pH of 5.5 for 2 h, some ZIF-8@PDA NPs were nearly entirely dissociated. To quantitatively evaluate the dissociation rate of ZIF-8@PDA NPs, the NPs were suspended in PBS solutions with different pH levels for durations of 2, 4, 6, and 8 h. We employed centrifugation to isolate undegraded ZIF-8@PDA NPs, with the residual quantity ascertained through precise weighing. Fig. 3b illustrates the hydrolysis rate of ZIF-8@PDA NPs under different pH conditions and durations. Generally, extending the duration can enhance the hydrolysis rate. When the solution pH reaches 5.5, an 8-hour immersion results in a hydrolysis rate of 48 ± 3.5%. These results corroborated that in an acidic environment, the ZIF-8@PDA NPs can gradually disassociate, with a faster hydrolysis rate observed as the pH value decreases.Next, we investigated the photothermal conversion performance of ZIF-8@PDA NPs under NIR irradiation. For the NIR source, we employed an 808 nm laser operating at a power density of 1.5 W cm−2. The ZIF-8 NPs, PDA NPs, and ZIF-8@PDA NPs were suspended in PBS solutions. As a control, we observed that the NIR irradiation caused only a marginal increase in temperature for either the PBS solution or ZIF-8 NP solution (Fig. 3c and d). In contrast, the temperature of ZIF-8@PDA NP solutions showed a noticeable increase after 10 min laser irradiation, highlighting their noticeable photothermal conversion performance (Fig. 3c and d). The temperature increase depended on the concentration of ZIF-8@PDA NPs (Fig. 3e). Furthermore, we studied the photothermal stability of ZIF-8@PDA NPs by alternately activating and deactivating the NIR laser at ten-minute intervals. After six alternating cycles, the photothermal effect of the ZIF-8@PDA NP solution did not change. It remained consistently at 55 ± 2 °C (Fig. 3f) and the photothermal conversion efficiency of ZIF-8@PDA NPs was 37%, indicating the good photothermal stability of the ZIF-8@PDA NPs. The photothermal effect is known to kill bacteria via local hyperthermia and oxidative stress induction in some cases.53 The maximum temperature of 55 °C is considered safe for the oral mucosa, making it suitable for application in the oral environment.34 The measurements of photothermal conversion and Zn2+ release of ZIF-8@PDA NPs could serve as a foundation for evaluating their antibacterial properties and effectiveness in eradicating oral biofilms in vitro, ex vivo, and in vivo.
2.3. Antibacterial efficacy in vitro
The antibacterial efficacy of ZIF-8@PDA NPs was assessed using the representative oral pathogenic bacteria, Streptococcus mutans (S. mutans). Fig. 4a shows S. mutans (1 × 106 CFU mL−1) that were treated with (I) PBS solution (control), (II) Zn(OAc)2·2H2O solution at 20 μg mL−1, (III) ZIF-8 NPs solution at 200 μg mL−1, (IV) PDA NP solution at 200 μg mL−1 under an 808 nm laser (1.5 W cm−2, 10 min), (V) ZIF-8@PDA NP solution at 200 μg mL−1 and (VI) ZIF-8@PDA NPs solution at 200 μg mL−1 under an 808 nm laser (1.5 W cm−2, 10 min), respectively for 6 h at 37 °C. Fig. 4b shows representative photographs of the colonies formed on agar plates after various treatments. We found that Zn2+ from Zn(OAc)2·2H2O and ZIF-8 NPs exhibited certain antibacterial properties. The photothermal effect can also produce a significant antibactericidal effect as evidenced by the results obtained using PDA NPs with NIR light. Notably, after being irradiated with NIR light, the CFU of the bacteria incubated with ZIF-8@PDA NPs showed a 5-log reduction (Fig. 4d). A comparative analysis of the results with and without NIR irradiation highlighted the synergistic action of photothermal and Zn2+ in eliminating S. mutans. Furthermore, bacterial morphology was examined using SEM, revealing the most significant membrane contraction in the ZIF-8@PDA + NIR group (Fig. 4c). Additionally, the residual shell of ZIF-8@PDA NPs adhering to the surface of bacteria was also observed. This suggested that the ZIF-8@PDA NPs function by initially adhering to the bacterial surface and subsequently inducing a photothermal effect combined with Zn2+ to kill the bacteria.Pathogenic bacteria can ferment sugars from food sources to produce acids, leading to a decrease in pH below 5.5.54 To study whether the presence of pathogenic bacteria can lower the environmental pH, we examined the pH values of the bacterial solution. As shown in Fig. 4e, the pH values of S. mutans suspensions steadily decreased and ultimately reached approximately 5.0 after 6 h in the presence of 1% sucrose. These measurements demonstrated the acid-producing ability of S. mutans. Interestingly, the pH values of the bacterial suspensions remain approximately constant when co-cultured with ZIF-8@PDA NPs and subjected to NIR irradiation (Fig. 4e). In addition, the bacteria were co-cultured with ZIF-8@PDA NPs at different pH values, and the OD values at 600 nm (OD600nm) were recorded, as this measurement is commonly used to monitor bacterial viability.55Fig. 4f shows that after coculturing with ZIF-8@PDA NPs, the OD values of the oral bacteria changed; lower pH values under the culture conditions resulted in greater changes in OD600nm. This indicated that the antibacterial capability of ZIF-8@PDA NPs increases with decreasing pH value. This could be attributed to the release of Zn2+.46 Taken together, these experiments demonstrated that ZIF-8@PDA NPs can respond to the weakly acidic environment of oral bacterial biofilms, making them more effective at killing bacteria at lower pH values.
2.4. Efficacy of ZIF-8@PDA NPs for oral biofilm eradication in vitro
We further evaluated whether ZIF-8@PDA NPs could effectively eradicate oral biofilms, which were formed by S. mutans and 1% sucrose in BHI at 37 °C for 24 h. As illustrated in Fig. 5a, the established biofilms underwent five various treatments. The live/dead BacLight bacterial viability kit was used to evaluate and assess bacterial viability, where dead or dying bacteria cells with damaged membranes were stained red, while live bacteria cells with intact membranes were stained green.56 The 3D reconstructed confocal images revealed that in the control group, where no NPs were added, green fluorescence was evenly distributed, and virtually no red fluorescence was observed (Fig. 5b). This signified a high concentration of live bacteria within the biofilm. After treatment with Zn2+ and ZIF-8 NPs, a significant amount of green fluorescence was still observed, indicating the presence of abundant viable bacteria within the biofilm. Notably, a significant reduction in green fluorescence and a marked decrease in biofilm thickness were observed when ZIF-8@PDA NPs were used in conjunction with NIR (Fig. 5b and c), indicating that ZIF-8@PDA NPs could effectively eradicate the biofilm. Furthermore, we assessed the number of regrown bacteria from the biofilms after various treatments (Fig. 5d and e). The bacteria incubated with ZIF-8@PDA NPs showed a 3-log CFU reduction (Fig. 5e), indicating that the combination of ZIF-8@PDA NPs and NIR effectively killed the bacteria within the biofilm, thereby preventing biofilm recolonization to a significant extent. Crystal violet (CV) staining was used to further quantify the residual amount of the biofilm after different treatments. We observed that the residual amount of the biofilm was minimal in the presence of ZIF-8@PDA NPs under NIR irradiation (Fig. 5f and g). These results confirmed that ZIF-8@PDA NPs could effectively eradicate the biofilm composed of S. mutans under NIR irradiation.Furthermore, we studied the effect of ZIF-8@PDA NPs on the pH variation within the biofilm. To achieve this, we used enamel slices to simulate the tooth surface. Enamel slices covered with oral biofilms were immersed in ZIF-8@PDA NP suspension, PBS solution, and saliva solution for 30 min, and the pH values of the enamel slice surfaces were subsequently measured. Fig. S4 (ESI†) shows that the pH on the tooth surface consistently remained above 7 after immersion in the ZIF-8@PDA NP suspension, whereas the pH after immersion in PBS and artificial saliva was about 5, indicating a weakly acidic condition. These experiments demonstrated that ZIF-8@PDA NPs could effectively respond to and neutralize the acidic microenvironment of the pathological biofilm.
Based on the above results, we proposed a mechanism for the eradication of oral biofilms using ZIF-8@PDA NPs under NIR irradiation. It is well-established that the PDA coating exhibits exceptional interfacial adhesion properties, enabling it to readily bond to the biofilm surface upon initial contact.57 Subsequently, the ZIF-8@PDA NPs respond to the acidic microenvironment of the biofilm (Fig. 4c), leading to the dissociation of the internal ZIF-8 NP structure and the release of Zn2+ ions. Zn2+ ions are known to have antimicrobial properties by disrupting bacterial cell membranes and interfering with essential enzymatic activities.57 Meanwhile, under NIR irradiation, the ZIF-8@PDA NPs induce a photothermal effect, which causes local hyperthermia. PDA exhibits excellent adhesion, multifunctionality, and antibacterial properties.57 It can generate nitro phenolic compounds through oxidative reactions, which subsequently react with amino groups on bacterial surfaces to form adhesive structures, thereby achieving antibacterial effects.57 This not only induces the denaturation of bacterial DNA and proteins but also enhances the permeability of Zn2+ within the biofilm due to the increased diffusion coefficient.58,59 In addition, hyperthermia has the potential to damage the biofilm, further accelerating the permeation of Zn2+ ions. Moreover, PDA has been shown to have excellent adhesion, multifunctionality, and antibacterial properties.60,61 It can generate nitro phenolic compounds through oxidative reactions, which subsequently react with amino groups on bacterial surfaces, causing bacterial death. Thus, by synergizing the photothermal and antibacterial properties of Zn2+ ions, the antibacterial efficacy of each can be significantly enhanced. As treatment progresses, the destruction of bacteria and the eradication of the biofilm prevent the production of acidic substances from bacterial metabolism, thereby maintaining a stable pH level (Fig. 4e).
2.5. Biofilm eradication on the extracted teeth surface
To further explore the efficacy of ZIF-8@PDA NPs combined with NIR irradiation in biofilm removal on the surface of the extracted tooth, an ex vivo biofilm model was constructed using human teeth. The teeth were subjected to different treatments (Fig. 6a). Note that all treatments described in this section were exposed to an 808 nm laser (1.5 W cm−2, 10 min). The results indicated that the temperature of teeth coated with ZIF-8@PDA NPs rapidly increased to 50 °C, respectively (Fig. 6b and c). Subsequently, the viable bacteria count in the biofilm was quantified using the plate count method (Fig. 6d). The results revealed that treatments involving Zn2+ + NIR had little effect on reducing the counts of S. mutans within the biofilm. Conversely, treatments with ZIF-8 NPs + NIR and PDA NPs + NIR were effective in decreasing bacterial counts. Notably, the ZIF-8@PDA NPs + NIR treatment showed the most promising results, decreasing bacterial counts by 3-log CFU units (Fig. 6e). This result was consistent with the in vitro biofilm eradication experiments (Fig. 5e).The morphology of biofilms on tooth surfaces after different treatments was examined using SEM. As shown in Fig. 6f, the biofilm of S. mutans completely covered the tooth surface when treated with PBS or Zn2+, indicating that Zn2+ alone cannot eradicate the biofilm. After treatment with ZIF-8 NPs, the number of bacteria observed within the biofilm was notably decreased. Interestingly, treating with PDA NPs led to the disruption of the biofilm, confirming that photo-induced hyperthermia could damage the biofilm.62 The ZIF-8@PDA NPs + NIR treatments almost eliminated the biofilm and the bacteria it harbored. We did not observe any well-formed dead bacteria, only some suspected debris (Fig. 6f). This result demonstrated the superior efficacy of the ZIF-8@PDA NPs + NIR in biofilm eradication.
2.6. Efficacy of ZIF-8@PDA NPs for oral biofilm eradication in vivo
To validate the efficacy of the ZIF-8@PDA NPs combined with NIR in removing oral biofilms in vivo, we established a rodent model infected with S. mutans (Fig. 7a). This is due to the progression of dental caries in rats is similar to that observed in humans, advancing from shallow caries to moderate caries and ultimately to deep caries with extensive lesions.54 Sprague–Dawley (SD) rats were treated with PBS, ZIF-8 NPs + NIR, PDA NPs + NIR, ZIF-8@PDA NPs, and ZIF-8@PDA NPs + NIR. In vivo NIR imaging of SD rats showed that the temperature of teeth coated with ZIF-8 NPs, PDA NPs, and ZIF-8@PDA NPs rapidly increased, particularly in the ZIF-8@PDA NPs group, which reached up to 55 °C (Fig. 7b and c), which was high enough to eliminate oral bacteria. Throughout the treatment period, all groups of rats maintained good health status, with no significant difference in the body weight gain (Fig. 7d). Optical photographs of rat teeth observed after 7 days of treatment revealed that the bacterial biofilm on the rat teeth surfaces was significantly reduced following the ZIF-8@PDA NPs + NIR treatment. The degree of tooth whiteness was markedly superior compared to that of other treatment modalities (Fig. 7e).After 7 days of treatment, sterile swabs were used to collect oral microbiota for culturing bacteria on BHI agar plates. We defined the antibacterial rate as one minus the ratio of the cultured bacterial count on rats’ teeth after different treatments to that in the control group (Fig. 7f and g). Treatments involving ZIF-8 NPs combined with NIR and PDA NPs combined with NIR did not achieve the desired ideal caries prevention effect. In contrast, treatment with ZIF-8@PDA NPs notably reduced the bacterial count; however, a significant number of viable bacteria persisted. Notably, when ZIF-8@PDA NPs were combined with NIR irradiation, the antibacterial rate reached 93 ± 5%, indicating superior antibacterial efficacy. Photographs of rat teeth were further examined to evaluate the therapeutic effects of different treatments (Fig. 7h). In the control group, ZIF-8 NPs + NIR group and PDA NPs + NIR group, lesions and caries were readily observable. The ZIF-8@PDA NP treatment showed a certain degree of therapeutic effect; however, localized remnants of the biofilm persisted. The ZIF-8@PDA NPs + NIR group exhibited the most remarkable therapeutic effect, significantly diminishing dental lesions and caries from the tooth surface. The treatment of ZIF-8@PDA NPs combined with NIR enhances the eradication of bacterial biofilms through the synergistic effect of Zn2+ release and photothermal therapy. Compared to traditional 1% sodium hypochlorite (NaOCl) irrigation and the single use of photothermal therapy, photodynamic therapy, Zn-MOF (Table S1, ESI†), the ZIF-8@PDA NPs combined with NIR method exhibits superior biocompatibility and a controlled release mechanism, thereby minimizing damage to healthy tissues and enhancing treatment safety. Furthermore, the gentle photothermal effect obviates the need for invasive procedures such as sandblasting and rubber cup polishing, markedly improving patient comfort and broadening its clinical applicability.
2.7. Biocompatibility assessment
The findings above demonstrated the effective eradication of oral bacteria and the efficient eradication of oral biofilms using the ZIF-8@PDA NPs + NIR treatments. Finally, we assessed the biocompatibility of ZIF-8@PDA NPs + NIR treatments through hemolysis and cell toxicity experiments. First, L929 cells were chosen to assess cytotoxicity. The live/dead cell staining revealed that ZIF-8@PDA NPs did not noticeably affect the morphology of L929 cells (Fig. 8a). Cell viability was further evaluated using the CCK-8 assay (Fig. 8b). The data revealed showed that, when the NP concentrations were below 200 μg mL−1, cell viability remained above 90% in all groups: ZIF-8 NPs, ZIF-8@PDA NPs, and ZIF-8@PDA NPs + NIR. However, as the NP concentration increased, a decline in cell viability was observed in both the ZIF-8 NPs and ZIF-8@PDA NPs + NIR groups. Consequently, within a concentration of 200 μg mL−1, the NPs demonstrated superior biocompatibility, and thus, the concentration of 200 μg mL−1 was employed for subsequent measurements. Furthermore, F-actin staining was performed to analyze the impact of ZIF-8@PDA NPs and ZIF-8@PDA NPs + NIR treatments on cell morphology and cytoskeleton (Fig. 8d). Both the ZIF-8@PDA NPs and the ZIF-8@PDA NPs + NIR treatments had a minimal impact on the structural organization of F-actin. It is noteworthy that the structural organization of F-actin was preserved even after NIR irradiation (Fig. 8d). In addition, a hemolysis assay was performed to assess the hemolytic potential of ZIF-8@PDA NPs on erythrocytes (Fig. 8c). When erythrocytes derived from fresh mouse blood cells were incubated in ultrapure water, they burst and released hemoglobin due to the low osmotic pressure of water. In contrast, the hemolysis rates for the pure PBS solution, as well as for the solutions containing the ZIF-8@PDA NPs (with or without NIR irradiation) were all below 5%, indicating excellent hemocompatibility.To further investigate the in vivo biocompatibility and safety of the ZIF-8@PDA NPs and the ZIF-8@PDA NPs + NIR treatments, histological analysis using H&E staining was conducted on key organs, including the lip, tongue, and gingival tissue (Fig. S5 and S6, ESI†). Pathological image analysis revealed that following ZIF-8@PDA NPs and ZIF-8@PDA NPs + NIR treatments, no significant signs of adverse reactions were observed in any treatment group, including inflammatory responses, proliferative changes, tissue damage, or necrosis. Taken together, these results showed the excellent biocompatibility of ZIF-8@PDA NPs and the biosafety of therapy using them.
3. Conclusions
In conclusion, we have developed ZIF-8@PDA NPs that degrade under mildly acidic conditions to release Zn2+ and convert light energy into heat, inducing localized hyperthermia. Using ZIF-8@PDA NPs enables a synergistic combination of photothermal and antibacterial metal ion therapy for effective oral biofilm eradication. This is because local hyperthermia could damage the biofilm, thereby accelerating the permeation of Zn2+ through it. Thus, by synergizing the photothermal and antibacterial properties of Zn2+ ions, the antibacterial efficacy of each can be significantly enhanced. Our results demonstrated that the therapy employing ZIF-8@PDA NPs, when subjected to NIR, exhibits remarkable antibacterial properties and proves effective in eradicating oral biofilms both in vitro and ex vivo. Furthermore, we used an in vivo rodent tooth biofilm model to demonstrate the suppression of dental caries. We also conducted a thorough study and confirmed that the NPs and the therapy using them are biocompatible. Beyond their application in clinical biofilm treatment, we anticipate that these NPs may be incorporated into toothpaste and utilized with near-infrared light-emitting toothbrushes. Their ease of synthesis, biocompatibility, affordability, and efficacy in eradicating biofilms could elevate conventional oral care practices.4. Experimental section
4.1. Materials
Zinc acetate dihydrate (Zn(OAc)2·2H2O), 2-methylimidazole (HmIm), glutaraldehyde, and dopamine hydrochloride (DA) were purchased from Aladdin Industrial Co., Ltd (China). Tris powder was acquired from Bio Basic Inc. (Canada). Polyglutamic acid was acquired from Xuankai Biological Technology (Nanjing) Co., Ltd (China). The CCK-8 assay kit was obtained from Beijing Labgic Technology Co., Ltd (China). The Live/Dead BacLight Bacterial Viability Kit (including SYTO9 and propidium iodide) and Calcein-AM/PI Cell Viability/Cytotoxicity Assay Kit were purchased from Beijing Beyotime Technology Co., Ltd (China). RhB-phalloidin (phalloidin labeled with RhB) and DAPI were obtained from Solarbio Life Sciences (China). Streptococcus mutans (S. mutans) and the murine gingival fibroblast cell (L929) were purchased from the American Type Culture Collection (USA). Dulbecco's modified Eagle's medium (DMEM) was supplied by VivaCell (China). The glass-bottom Petri dishes used for confocal fluorescence imaging were acquired from NEST Biotechnology (China).4.2. Synthesis of ZIF-8 NPs and ZIF-8@PDA NPs
First, Zn(OAc)2·2H2O (0.219 g) and HmIm (1.6440 g) were dissolved in 4.5 mL ultrapure water, stirred thoroughly, and left for 24 h. Next, the white precipitate particles were washed with ultrapure water and dried at 50 °C to obtain ZIF-8 NPs. Then, the prepared ZIF-8 NPs (100 mg) were dispersed in Tris–HCl buffer solution (120 mL, pH = 8.5), followed by the slow addition of a certain amount of DA, and the proportions between ZIF-8 and PDA were 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 1
2, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 2
1, and 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, respectively. The progress of the reaction is monitored by observing the color change from light white to dark brown. Afterward, ZIF-8@PDA NPs were collected through centrifugation (10
5, respectively. The progress of the reaction is monitored by observing the color change from light white to dark brown. Afterward, ZIF-8@PDA NPs were collected through centrifugation (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm), washing, and lyophilization. DA (40 mg) was dispersed in Tris–HCl buffer solution (120 mL, pH = 8.5) and stirred for 2 h to obtain PDA NPs.
000 rpm), washing, and lyophilization. DA (40 mg) was dispersed in Tris–HCl buffer solution (120 mL, pH = 8.5) and stirred for 2 h to obtain PDA NPs.
4.3. The photothermal effect and pH sensitivity of ZIF-8@PDA NPs
The PBS, the ZIF-8 suspensions (200 μg mL−1), and the ZIF-8@PDA NP suspensions with different concentrations (50, 100, 200, and 400 μg mL−1) were placed in a centrifuge tube. Each sample was then irradiated with an 808 nm laser at a density of 1.5 W cm−2 for 10 min, respectively, and their real-time temperature changes were assessed using an infrared thermal imager (TiS40, Fluke, USA). The photothermal stability of ZIF-8@PDA NPs was evaluated by measuring the temperature changes over six cycles of laser on/off. The following equation can be used to calculate the photothermal conversion efficiency of ZIF-8@PDA NPs:where h represents the heat transfer coefficient, A is the surface area of the container, I means the laser power, Aλ represents the absorbance of ZIF-8@PDA NPs at 808
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) nm, and ΔTmax,min and ΔTmax,H2O stand for the temperature change of the ZIF-8@PDA NP dispersion and water at the maximum steady-state temperature, respectively.
nm, and ΔTmax,min and ΔTmax,H2O stand for the temperature change of the ZIF-8@PDA NP dispersion and water at the maximum steady-state temperature, respectively.
To verify the photothermal effect of ZIF-8@PDA NPs on the tooth surface, PBS, the ZIF-8 NP suspension (200 μg mL−1), and the ZIF-8@PDA NP suspension (200 μg mL−1) were each applied to the extracted human teeth using an applicator. The teeth were then exposed to NIR for 10 min. The real-time temperature of the teeth was recorded using an infrared thermal imager. ZIF-8@PDA NPs were added into PBS solutions with different pH values (pH = 5.5, 6.0, 6.5, and 7.0), and incubated at 37 °C for 2, 4, 6, and 8 h. Following incubation, the samples underwent centrifugation at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, enabling the collection of solid residues. These residues were subsequently weighed and used for quantitative calculations of the hydrolysis rate of ZIF-8@PDA NPs. Additionally, the morphological alterations of the ZIF-8@PDA NPs in solutions with different pH values and incubated at 37 °C for 2 h were examined by SEM.
000 rpm, enabling the collection of solid residues. These residues were subsequently weighed and used for quantitative calculations of the hydrolysis rate of ZIF-8@PDA NPs. Additionally, the morphological alterations of the ZIF-8@PDA NPs in solutions with different pH values and incubated at 37 °C for 2 h were examined by SEM.
4.4. Antibacterial activity and biofilm eradication in vitro
To investigate the oral antibacterial efficacy of ZIF-8@PDA NPs with NIR laser, Streptococcus mutans (S. mutans) was chosen for the antibacterial assays. To assess the biofilm eradication efficacy of synergistic photothermal and antibacterial metal ion therapy, a mature oral biofilm was established using S. mutans in vitro. Further details about these experimental methods are detailed in the ESI.†4.5. Establishment and treatment of a mature biofilm model on extracted teeth ex vivo
To further investigate the biofilm removal efficacy of ZIF-8@PDA NPs plus NIR irradiation, we established an oral biofilm model ex vivo to assess whether various treatments effectively eliminated mature biofilms on extracted tooth surfaces. The human teeth were provided by the Stomatological Hospital of Jilin University. The extracted human teeth enamels were washed several times with sterile PBS which was cut into slices (4 mm × 4 mm) and immersed in the S. mutans suspension (1 × 108 CFU mL−1) for 24 h. Then, the extracted slices were washed 3 times with PBS and added BHI medium containing: (I) the control (PBS solution), (II) 20 μg mL−1 Zn(OAc)2·2H2O, (III) 200 μg mL−1 ZIF-8 NPs, (IV) 200 μg mL−1 PDA NPs and (V) 200 μg mL−1 ZIF-8@PDA NPs. All the solutions contained 1% polyglutamic acid to increase the adhesion of the NPs to the extract teeth. After application, all slices were exposed to an 808 nm laser (1.5 W cm−2, 10 min). Then, the extracted slices were co-cultured with various BHI media for 6 h in a 37 °C incubator. Subsequently, biofilms on the enamel surface were collected into PBS through vigorous agitation and then diluted. The bacterial suspensions were spread on an agar plate and incubated at 37 °C for 24 h to count the number of CFUs. Additionally, the biofilms on the enamel surface were observed using a SEM. Finally, the biofilms on the enamel surface were immersed in PBS, artificial saliva, or ZIF-8@PDA NP solutions for 30 min, respectively, and the pH of the enamel slices was measured.4.6. Biosafety experiment
To assess the cytotoxicity of ZIF-8@PDA NPs, the survival rates of L929 mouse gingival fibroblast cells treated with ZIF-8 NPs, ZIF-8@PDA NPs and ZIF-8@PDA NPs + NIR were measured at 24 h using the CCK-8 assay. The NP concentrations used were 50, 100, 200, 300, and 400 μg mL−1, respectively, and the NIR light was obtained from an 808 nm laser at a density of 1.5 W cm−2 for 10 min. Then, the cells treated with PBS, ZIF-8@PDA NPs (200 μg mL−1) and ZIF-8@PDA NPs + NIR were stained with a Calcein-AM/PI assay kit for both living and dead cells and observed by CLSM fluorescence imaging.63 After that, the cell nuclei and cytoskeleton were stained using RhB-phalloidin and DAPI and observed by confocal laser scanning microscopy (CLSM) fluorescence imaging.64,65 Additionally, the hemolysis rates were determined through a hemolysis assay of different treatments. Finally, histological evaluation was performed to assess the toxicity of the nanomaterials in vivo. Further details about these experimental methods are detailed in the ESI.†4.7. In vivo therapeutic effect
All animal experiments conformed to the guidelines established by the Animal Care and Ethics Committee of the Changchun Institute of Applied Chemistry (CIAC), Chinese Academy of Sciences. The occurrence of lesions on rat teeth’ smooth surface and fissure surface was measured using a stereomicroscope (NSZ-405, China) to assess the therapeutic effects of PBS, ZIF-8 NPs + NIR, PDA NPs + NIR, ZIF-8@PDA NPs, and ZIF-8@PDA NPs + NIR treatments for the oral biofilm in vivo. The coating of nanoparticles was performed by dipping a cotton swab into the solution and then applying it to the surface of the teeth. NIR was applied twice a day at a density of 1.5 W cm−2 for 10 minutes each time, and the real-time temperature changes were monitored using an infrared thermal imager. A standard plate counting assay was conducted to evaluate the antibacterial effect. The hearts, livers, spleens, lungs, kidneys, tongue mucosa, lips, and gingival tissues of rats were collected for H&E staining Further details about these experimental methods are detailed in the ESI.†Author contributions
Ge Pan: conceptualization, methodology, investigation, validation, and writing – original draft preparation. Miaomiao Zhang: data curation, formal analysis, investigation, and writing – review. Jinyao Zheng and Zongjia Li: methodology. Qian Duan: conceptualization, writing – review and editing, and funding acquisition. Dapeng Wang: conceptualization, funding acquisition, investigation, methodology, project administration, resources, supervision, validation, and writing – review and editing.Data availability
All relevant data are within the manuscript and its additional files.Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
This work was supported by the National Key Research and Development Program of China (Grant No. 2021YFC2101700), the Natural Science Foundation of Jilin Province (Grant No. 20240101179JC), the Key Research Program of Frontier Sciences, Chinese Academy of Sciences (Grant No. ZDBS-LY-SLH033) and the Special Project for High-tech Industrialization of Science and Technology Cooperation between Jilin Province and Chinese Academy of Sciences (Grant No. 2023SYHZ0030).References
- L. Sedghi, V. DiMassa, A. Harrington, S. V. Lynch and Y. L. Kapila, Periodontol. 2000, 2021, 87, 107–131 CrossRef PubMed.
- H.-C. Flemming, J. Wingender, U. Szewzyk, P. Steinberg, S. A. Rice and S. Kjelleberg, Nat. Rev. Microbiol., 2016, 14, 563–575 CrossRef CAS PubMed.
- L. Gao, T. Xu, G. Huang, S. Jiang, Y. Gu and F. Chen, Protein Cell, 2018, 9, 488–500 CrossRef CAS PubMed.
- H. C. Flemming and J. Wingender, Nat. Rev. Microbiol., 2010, 8, 623–633 CrossRef CAS PubMed.
- J. L. Baker, J. L. M. Welch, K. M. Kauffman, J. S. McLean and X. S. He, Nat. Rev. Microbiol., 2024, 22, 89–104 CrossRef CAS PubMed.
- D. Davies, Nat. Rev. Drug Discovery, 2003, 2, 114–122 CrossRef CAS PubMed.
- D. S. W. Benoit, K. R. Sims, Jr. and D. Fraser, ACS Nano, 2019, 13, 4869–4875 CrossRef CAS PubMed.
- Z. Yin, Y. Liu, A. Anniwaer, Y. You, J. Guo, Y. Tang, L. Fu, L. Yi and C. Huang, Adv. Mater., 2023, 2305633 CrossRef PubMed.
- Y. Yu, Y. Cheng, L. Tan, X. Liu, Z. Li, Y. Zheng, T. Wu, Y. Liang, Z. Cui, S. Zhu and S. Wu, Chem. Eng. J., 2022, 431, 133279 CrossRef CAS.
- P. S. Stewart and J. William Costerton, Lancet, 2001, 358, 135–138 CrossRef CAS PubMed.
- Y. Tang, Q.-X. Huang, D.-W. Zheng, Y. Chen, L. Ma, C. Huang and X.-Z. Zhang, Mater. Today, 2022, 53, 71–83 CrossRef CAS.
- H. Koo, R. N. Allan, R. P. Howlin, P. Stoodley and L. Hall-Stoodley, Nat. Rev. Microbiol., 2017, 15, 740–755 CrossRef CAS PubMed.
- S. E. Birk, J. A. J. Haagensen, H. K. Johansen, S. Molin, L. H. Nielsen and A. Boisen, Adv. Healthcare Mater., 2020, 9, 1901779 CrossRef CAS PubMed.
- Y. Yang, Y. Qian, M. Zhang, S. Hao, H. Wang, Y. Fan, R. Liu, D. Xu and F. Wang, J. Mater. Sci. Technol., 2023, 133, 77–88 CrossRef CAS.
- Y. Liu, L. Shi, L. Su, H. C. van der Mei, P. C. Jutte, Y. Ren and H. J. Busscher, Chem. Soc. Rev., 2019, 48, 428–446 RSC.
- S. Wu, C. Xu, Y. Zhu, L. Zheng, L. Zhang, Y. Hu, B. Yu, Y. Wang and F.-J. Xu, Adv. Funct. Mater., 2021, 31, 2103591 CrossRef CAS.
- Y. Li, Y. Liu, Y. Ren, L. Su, A. Li, Y. An, V. Rotello, Z. Zhang, Y. Wang, Y. Liu, S. Liu, J. Liu, J. D. Laman, L. Shi, H. C. van der Mei and H. J. Busscher, Adv. Funct. Mater., 2020, 30, 2004942 CrossRef CAS PubMed.
- Y. Yang, M. Li, G. Pan, J. Chen and B. Guo, Adv. Funct. Mater., 2023, 33, 2214089 CrossRef CAS.
- Y. Yang, X. Wu, C. He, J. Huang, S. Yin, M. Zhou, L. Ma, W. Zhao, L. Qiu, C. Cheng and C. Zhao, ACS Appl. Mater. Interfaces, 2020, 12, 13698–13708 CrossRef CAS PubMed.
- X. Liu, X. Sun, Y. Peng, Y. Wang, D. Xu, W. Chen, W. Wang, X. Yan and X. Ma, ACS Nano, 2022, 16, 14666–14678 CrossRef CAS PubMed.
- J. Wang, S. P. Teong, S. N. Riduan, A. Armugam, H. Lu, S. Gao, Y. K. Yean, J. Y. Ying and Y. Zhang, J. Am. Chem. Soc., 2024, 146, 599–608 CrossRef CAS PubMed.
- R. Li, T. Chen and X. Pan, ACS Nano, 2021, 15, 3808–3848 CrossRef CAS PubMed.
- A. Wang, G. Fan, H. Qi, H. Li, C. Pang, Z. Zhu, S. Ji, H. Liang, B. Jiang and X. Shen, Biomaterials, 2022, 289, 121798 CrossRef CAS PubMed.
- F. Kunisch, C. Campobasso, J. Wagemans, S. Yildirim, B. K. Chan, C. Schaudinn, R. Lavigne, P. E. Turner, M. J. Raschke, A. Trampuz and M. Gonzalez Moreno, Nat. Commun., 2024, 15, 8572 CrossRef CAS PubMed.
- A. Wang, L. Li, L. Zheng, B. P. Jiang, Y. Liu, R. Huang, H. Qiu, S. Ji, H. Liang and X. C. Shen, Aggregate, 2024, e662 Search PubMed.
- G. M. Neelgund, A. Oki, S. Bandara and L. Carson, J. Mater. Chem. B, 2021, 9, 1792–1803 RSC.
- Z. Yuan, C. Lin, Y. He, B. Tao, M. Chen, J. Zhang, P. Liu and K. Cai, ACS Nano, 2020, 14, 3546–3562 CrossRef CAS PubMed.
- W. Xiong, L. Wang, X. Chen, H. Tang, D. Cao, G. Zhang and W. Chen, J. Mater. Chem. B, 2020, 8, 5234–5244 RSC.
- T. C. Pagonis, J. Chen, C. R. Fontana, H. Devalapally, K. Ruggiero, X. Song, F. Foschi, J. Dunham, Z. Skobe, H. Yamazaki, R. Kent, A. C. R. Tanner, M. M. Amiji and N. S. Soukos, J. Endod., 2010, 36, 322–328 CrossRef PubMed.
- M. Chen, J. Zhang, J. Qi, R. Dong, H. Liu, D. Wu, H. Shao and X. Jiang, ACS Nano, 2022, 16, 7732–7744 CrossRef CAS PubMed.
- P. C. Naha, Y. Liu, G. Hwang, Y. Huang, S. Gubara, V. Jonnakuti, A. Simon-Soro, D. Kim, L. Gao, H. Koo and D. P. Cormode, ACS Nano, 2019, 13, 4960–4971 CrossRef CAS PubMed.
- J. Xiao, L. Hai, Y. Li, H. Li, M. Gong, Z. Wang, Z. Tang, L. Deng and D. He, Small, 2022, 18, 2105465 CrossRef CAS PubMed.
- Y. Mu, H.-H. Tran, Z. Xiang, A. Majumder, E. Hsu, E. Steager, H. Koo and D. Lee, Small, 2024, 2402292 CrossRef CAS PubMed.
- L. Chen, M. Peng, H. Li, J. Zhou, W. He, R. Hu, F. Ye, Y. Li, L. Shi and Y. Liu, Adv. Mater., 2024, 36, 2306376 CrossRef CAS PubMed.
- L. Xiao, M. Feng, C. Chen, Q. Xiao, Y. Cui and Y. Zhang, Adv. Mater., 2023, 2304982 CrossRef PubMed.
- Y. Yu, Y. Zhang, Y. Cheng, Y. Wang, Z. Chen, H. Sun, X. Wei, Z. Ma, J. Li, Y. Bai, Z. Wu and X. Zhang, Bioact. Mater., 2022, 13, 269–285 CAS.
- J. Wang, B. Hao, K. Xue, H. Fu, M. Xiao, Y. Zhang, L. Shi and C. Zhu, Adv. Mater., 2022, 34, 2205653 CrossRef CAS PubMed.
- L. He, J. Dragavon, S. Cho, C. Mao, A. Yildirim, K. Ma, R. Chattaraj, A. P. Goodwin, W. Park and J. N. Cha, J. Mater. Chem. B, 2016, 4, 4455–4461 RSC.
- Y. Li, X. Liu, B. Li, Y. Zheng, Y. Han, D.-f Chen, K. W. K. Yeung, Z. Cui, Y. Liang, Z. Li, S. Zhu, X. Wang and S. Wu, ACS Nano, 2020, 14, 8157–8170 CrossRef CAS PubMed.
- L. Wang, C. Zhang, J. Zhao, Z. Zhu, J. Wang, W. Fan and W. Jia, Adv. Mater., 2024, 36, 2308110 CrossRef CAS PubMed.
- W. Guo, Y. Wang, K. Zhang, X. Dai, Z. Qiao, Z. Liu, B. Yu, N. Zhao and F. Xu, Chem. Mater., 2023, 35, 6853–6864 CrossRef CAS.
- S. Gopalakrishnan, A. Gupta, J. M. V. Makabenta, J. Park, J. J. Amante, A. N. Chattopadhyay, D. Matuwana, C. J. Kearney and V. M. Rotello, Adv. Healthcare Mater., 2022, 11, 2201060 CrossRef CAS PubMed.
- C. Montoya, L. Roldan, M. Yu, S. Valliani, C. Ta, M. Yang and S. Orrego, Bioact. Mater., 2023, 24, 1–19 CAS.
- S. Goel, C. A. Ferreira, F. Chen, P. A. Ellison, C. M. Siamof, T. E. Barnhart and W. Cai, Adv. Mater., 2018, 30, 1704367 CrossRef PubMed.
- C. R. Mendes, G. Dilarri, C. F. Forsan, V. d M. R. Sapata, P. R. M. Lopes, P. B. de Moraes, R. N. Montagnolli, H. Ferreira and E. D. Bidoia, Sci. Rep., 2022, 12, 2658 CrossRef CAS PubMed.
- M. Taheri, D. Ashok, T. Sen, T. G. Enge, N. K. Verma, A. Tricoli, A. Lowe, D. R. Nisbet and T. Tsuzuki, Chem. Eng. J., 2021, 413, 127511 CrossRef CAS.
- Z. Lv, X. Zhang, Q. Gao, C. Wen, Y. He, H. Tan, L. Qian, W. Qi, X. Chen and Z. Li, Adv. Funct. Mater., 2024, 34, 2409274 CrossRef CAS.
- Y. Lu, D. Fan, Z. Shen, H. Zhang, H. Xu and X. Yang, Nano Energy, 2022, 95, 107016 CrossRef CAS.
- Z. Wang, Y. Zou, Y. Li and Y. Cheng, Small, 2020, 16, 1907042 CrossRef CAS PubMed.
- T. Wu, L. Huang, J. Sun, J. Sun, Q. Yan, B. Duan, L. Zhang and B. Shi, Carbohydr. Polym., 2021, 269, 118276 CrossRef CAS PubMed.
- M. Song, J. Xing, H. Cai, X. Gao, C. Li, C. Liu, X. Li, X. Fu, S. Ding, W. Cheng and R. Chen, ACS Nano, 2023, 17, 10748–10759 CrossRef CAS PubMed.
- J. Ruan, H. Liu, B. Chen, F. Wang, W. Wang, Z. Zha, H. Qian, Z. Miao, J. Sun, T. Tian, Y. He and H. Wang, ACS Nano, 2021, 15, 11428–11440 CrossRef CAS PubMed.
- L. Chen, M. Peng, J. Zhou, X. Hu, Y. Piao, H. Li, R. Hu, Y. Li, L. Shi and Y. Liu, Adv. Mater., 2023, 35, 2301664 CrossRef CAS PubMed.
- N. B. Pitts, D. T. Zero, P. D. Marsh, K. Ekstrand, J. A. Weintraub, F. Ramos-Gomez, J. Tagami, S. Twetman, G. Tsakos and A. Ismail, Nat. Rev. Dis. Primers, 2017, 3, 17030 CrossRef PubMed.
- G. Pan, H. Wang, Z. Li, J. Zheng, B. Peng, Q. Duan and M. Zhang, Colloids Surf., B, 2024, 243, 114133 CrossRef CAS PubMed.
- Z. Li, W. Wei, M. Zhang, X. Guo, B. Zhang, D. Wang, X. Jiang, F. Liu and J. Tang, Adv. Healthcare Mater., 2023, 12, 2301060 CrossRef CAS PubMed.
- P. Yang, F. Zhu, Z. Zhang, Y. Cheng, Z. Wang and Y. Li, Chem. Soc. Rev., 2021, 50, 8319–8343 RSC.
- K. Lyu, H. Chen, J. Gao, J. Jin, H. Shi, D. K. Schwartz and D. Wang, Biomacromolecules, 2022, 23, 4709–4717 CrossRef CAS PubMed.
- K. Lyu, Y. Zhao, M. Zhang, J. Tang, J. Zhang, Y. Liu, X. Bian, X. Chen, H. Chen and D. Wang, Langmuir, 2023, 39, 13534–13545 CrossRef CAS PubMed.
- D. Wang, H. Wu, L. Liu, J. Chen and D. K. Schwartz, Phys. Rev. Lett., 2019, 123, 118002 CrossRef CAS PubMed.
- M. Hu, H. Chen, H. Wang, S. Burov, E. Barkai and D. Wang, ACS Nano, 2023, 17, 21708–21718 CrossRef PubMed.
- C. Shuai, J. Zan, F. Deng, Y. Yang, S. Peng and Z. Zhao, ACS Sustainable Chem. Eng., 2021, 9, 1814–1825 CrossRef CAS.
- Z. Zhang, H. Chen, M. Hu and D. Wang, J. Am. Chem. Soc., 2023, 145, 10512–10521 CrossRef CAS PubMed.
- Y. Zhao, H. Chen, S. Geng, S. Liu and D. Wang, Chin. J. Chem., 2023, 41, 3245–3252 CrossRef CAS.
- H. Chen, M. Hu, Y. Zhao, K. Lyu, Y. Xu, Y. Sun, Z. Xie, J. Huang and D. Wang, Aggregate, 2024, 6, e649 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4tb02427c |
| This journal is © The Royal Society of Chemistry 2025 |