Emerging 2D MXene-conjugated nanoarchitectures for nanozymes: current advances and future frontiers in biochemical sensing technologies
Ramar
Rajamanikandan
 ab,
Kandasamy
Sasikumar
ab,
Kandasamy
Sasikumar
 ab and
Heongkyu
Ju
ab and
Heongkyu
Ju
 *ab
*ab
aDepartment of Physics and Semiconductor Science, Gachon University, Seongnam-si, Republic of Korea. E-mail: batu@gachon.ac.kr
bGachon Bionano Research Institute, Gachon University, Seongnam-si, 13120, Republic of Korea
First published on 2nd October 2025
Abstract
Nanozymes, nanostructured materials with exceptional catalytic performance, are recognized for their unique benefits over natural enzymes, including notably low fabrication costs and high chemical stability. Emerging as a captivating frontier in nanozyme research, MXene-based composite material nanozymes (MXzymes) have sparked intense research interest owing to their unique compositions and structural features, which can be engineered to unlock their enzyme-mimicking catalytic prowess. This approach opens groundbreaking opportunities for detecting biologically significant, food-related, and environmentally crucial analytes. This review highlights the innovative fabrication methods of MXzymes, focusing on their diverse nanozyme activity types reported thus far, including peroxidase-like and oxidase-like functionalities, while unveiling the underlying catalytic mechanisms in detail. In addition, recent pioneering breakthroughs in MXzymes are comprehensively reviewed, including catalytic signal amplification by MXzymes, which can function as cutting-edge sensing platforms, as evidenced by colorimetric, smartphone-based, chemiluminescent, electrochemical, and surface-enhanced Raman scattering-based analyses. Finally, the promising potential applications of MXzymes and fundamental challenges associated with their scalability, stability, and biocompatibility for MXzyme-based chemical/biomedical sensors are addressed.
Environmental significanceAntibiotics, heavy metals, pesticides, organic contaminants, food contaminants, and amino acids each impact the environment. Antibiotics from agriculture and healthcare promote resistance and disrupt microbes. Heavy metals like Pb and Hg are poisonous, tenacious, and bioaccumulate. Pesticides harm non-target species and contaminate soil and water. Organic pollutants such as OPs and dioxins are toxic, carcinogenic, and degrade slowly. Food contaminants, including pesticide residues and plasticizers, enter ecosystems via waste. While amino acids are non-toxic, excess runoff can cause eutrophication. Together, these substances threaten biodiversity, disrupt ecosystems, and highlight the need for strict monitoring and regulation to protect health and the environment. This review article collects the sensitive detection of these substances via MXene-conjugated nanomaterial-based nanozyme sensors. |
1. Introduction
Natural enzymes, bio-macromolecules with extreme effectiveness and specificity for enzymatic reactions, play an essential role in biological functions.1 However, their applications have been hampered by poor stability, high production cost, and labor-intensive purification. As alternatives, artificial nanozymes were designed to overcome these limitations in recent decades.1,2 Many efforts focused on finding molecules to synthesize them, including cyclodextrins,3 fullerenes,4 polymers,5 porphyrins,6 dendrimers,7 biomolecules,8 and metal complexes.9The past few years have seen explosive growth in enzyme-like catalytic nanomaterials (nanozymes) since the discovery in 2007 that peroxidase (POD)-like catalytic features, resembling natural POD-like horseradish peroxidase (HRP), existed in magnetic nanoparticles (NPs).10 A nanozyme, a nano-sized material (1–100 nm) with enzyme-mimicking catalytic abilities, combines features of chemical and biological catalysts,1,2 thus expanding their uses.10 Hundreds of nanomaterials mimicking enzyme operations have been reported for the eco-friendly industry, healthcare, and typical catalysis.11–15 Nanozymes offer advantages over natural enzymes: inexpensive and simple fabrication, chemical stability, and customizable design.1,2,13 As nanozyme catalysis produces amplified signals for quantitative determination and allows flexible activity adjustment, nanozymes have lent themselves to chemo-bio sensing.14,15 With developments in bio-/nano-catalysis, nano(bio) technology, machine learning, and computational design, various 2D material-based nanozymes have emerged using transition metal dichalcogenide nanosheets,16 graphene,17 metal–organic frameworks (MOFs),13 and MXenes18 owing to their surface area, conductivity, and active sites.
Since their discovery in 2011, MXenes, a family of two-dimensional transition-metal carbides, nitrides, and carbonitrides, have gained interest due to their physical and chemical characteristics (including electrical conductivity, thermal conductivity, and configurable interlayer spacing).19 The term “MXene” enables this 2D material to be distinguished from graphene, encompassing both the MAX phase and derived MXenes from the MAX phase.20,21 The chemical formula of MAX phases is Mn+1AXn, where “M” denotes a transition metal, n = 1, 2, or 3, “A” is mainly a grouping of III–IV elements, and “X” refers to C and/or N (Fig. 1a). Chemical etching of the MAX phases' A-layers is the primary method exploited for fabricating MXenes (Fig. 1b).21 Thus, the attained MXenes have a 2D stacked arrangement with a formulation of Mn+1XnTx, where M represents metals (Mo, Nb, Ti, Ta, V, Hf, Cr, or Sc), X signifies N or C, and “Tx” denotes exterior functional groups (–F, –O, and –OH) generated through synthetic etching.21,22 With extensive study on optical, mechanical, thermal, and magnetic characteristics, MXenes have drawn interest from various sectors, demonstrating promise in analytical sciences, particularly in optical and electrochemical sensors.22,23 Owing to their unique physicochemical properties, Zhou L. et al. developed chitosan-2D Ti3C2 nanocomposites as an effective enzyme immobilization support for constructing high-performance biosensors.21 Similarly, Yu Chen et al. reported a Fenton-reaction-based nano-catalytic system on 2D Ti3C2 nanosheets, employing photothermal conversion nanoprobes to enhance cancer cell ablation efficiency.12 These works have inspired many subsequent studies on MXzyme-based catalysis. Beyond sensing, MXenes have been applied in catalysis, energy storage, photodetectors, environmental remediation, nanozymes, and drug delivery.24–27
MXenes' functions make them suitable supports for nanozyme-based catalytic reactions.18,21 Their physicochemical features enhance the chemical stability of supported NPs and catalytic efficiency.28 The 2D MXene Ti3C2 nanosheet-based nanozyme has unique properties: (i) water dispersibility and functionalization capability because of hydrophilic surface groups such as –OH and –O, (ii) charge-carrier transport and redox reactivity from exposed terminal Ti's valence states and metallic conductivity, (iii) eco-friendly biocompatibility, and (iv) Ti3C2 NSs exhibit inherent POD-like action.29 Due to these special characteristics, 2D MXenes are preferred over other nanozymes, including graphene, MOFs, and transition metal dichalcogenide nanosheets, in MXzyme sensors.13,14,16,17
Many reviews on MXene-based nanocomposites cover their fabrication, characteristics, and applications in energy conversion,30,31 environmental monitoring,32 photodetectors,33 electrochemical sensors,25 information technology,34etc.35 However, few papers have addressed the biomedical potential of MXzymes; future investigations should focus on thorough studies and evaluations for their great potential in biomedicine, covering therapeutic applications.36,37 While a review of MXzyme sensors for food contaminants was recently published,38 a comprehensive review of MXzyme sensors for detecting potent analytes remains unreported.
This review summarizes recent advancements in MXene nanocomposites with enzyme-like features for chemo-biosensors of biological and toxic analytes (Scheme 1). Initially, we provide an overview of MXenes' fabrication and functionalization techniques. Next, we discuss the types of nanozyme properties and catalytic mechanisms of MXenes. Subsequently, we examine the benefits of nanozyme-based sensors in various strategies, i.e., conventional colorimetry, smartphone-assisted colorimetry, fluorescence, chemiluminescence, electrochemiluminescence, and surface-enhanced Raman scattering (SERS). We explore nanozyme-mediated reactions and their application in analyte detection. Lastly, we discuss the trailblazing sensing strategies in this discipline, address technical limitations, and emphasize their significance in advanced sensors and diagnostics. We believe that this timely review with perspectives will advance our understanding of nanozyme characteristics and the development of novel nanomaterials with enzyme-mimicking activities.
2. MXene synthesis
The M–X bond in MAX phases shows metallic, covalent, and ionic characteristics. In contrast, the M–A bond is metallic and more chemically reactive, allowing it to break easily.39 Because of weaker M–A bonds compared to M–X bonds, the A layers can be selectively etched from MAX phase materials. Synthetic protocols from MAX phase materials to MXene (Fig. 1c) are described as follows.2.1. Direct hydrofluoric acid etching
Notably, 2D MXenes were first exfoliated from MAX precursors using 50% hydrofluoric acid (HF) by Naguib et al. in 2011.40 The Al layer was selectively detached from the Ti3AlC2 MAX phase at room temperature within 2 h reaction time. The prepared accordion-like MXenes (Ti3C2Tx) possessed termination groups (–F, –OH, and –O) and defects at the surface, as shown in Fig. 2a. The etching mechanism can be described by the following reactions.| Ti3AlC2 + 3HF = AlF3 + 1.5H2 + Ti3C2 | (1) |
| Ti3C2 + 2H2O = Ti3C2(OH)2 + H2 | (2) |
| Ti3C2 + 2HF = Ti3C2F2 + H2 | (3) |
 | ||
| Fig. 2 (a) Illustration of the exfoliation process for Ti3AlC2, highlighting the replacement of Al atoms by –OH groups after HF treatment [reproduced from ref. 40, with permission from John Wiley and Sons, copyright 2011]. (b) XRD patterns of samples etched using a LiF + HCl solution. The pink trace represents multilayer Ti3C2Tx, exhibiting a sharp and intense (0002) peak along with higher-order (000l) reflections corresponding to a c-lattice parameter of 28 Å and well-ordered stacking in the c direction [reproduced from ref. 47 with permission from Springer Nature, copyright 2014]. (c and d) SEM images of Ti3AlC2 and hydrothermally synthesized h-Ti3C2 (16 h reaction time) [reproduced from ref. 56 with permission from Elsevier, copyright 2018]. (e) Schematic of the HCl-assisted hydrothermal etching mechanism for fluoride-free Mo2CTx [reproduced from ref. 58 with permission from John Wiley and Sons, copyright 2021]. TEM photograph of (f) several delaminated flakes and (g) a single Ti4N3Tx flake (inset: the corresponding SAED pattern, confirming the hexagonal basal plane symmetry) [reproduced from ref. 59 with permission from the Royal Society of Chemistry, copyright 2016]. (h) Replacement reaction between the MAX phase and late transition-metal halides [reproduced from ref. 61 with permission from the American Chemical Society, copyright 2019]. (i) EDX analysis (line scan) of Ti3C2Br2 MXene [reproduced from ref. 62 with permission from the American Association for the Advancement of Science, copyright 2020]. (j) XPS full scan spectrum of the pristine Ti3AlC2 and the final Ti3C2Tx [reproduced from ref. 65 with permission from John Wiley and Sons, copyright 2018]. Anodic etching of bulk Ti3AlC2 in a binary aqueous electrolyte: (k(i–iv)), the electrochemical cell setup, XRD patterns of Ti3AlC2, Ti3C2Tx, and Ti3C2Tx MXenes, SEM images of Ti3AlC2 and Ti3C2Tx [reproduced from ref. 67 with permission from John Wiley and Sons, copyright 2018]. (l(i–vi)) Stability of MXene: AFM images of MXenes after exposure to water, SEM image, and XRD patterns of fresh and ageing MXene flakes [reproduced from ref. 68 with permission from John Wiley and Sons, copyright 2021]. | ||
According to eqn (1), the A element in the MAX phase reacts with fluoride ions from HF, initiating the removal of the M–X layer as described in reaction (2). The surface transition metal then actively interacts with water molecules or fluoride ions to develop an exfoliated graphite-like structure, i.e., MXene, terminated with –O, –OH, or –F groups (eqn (2) and (3)). This etching process was subsequently extended to many more MAX phases with diverse chemical compositions to produce MXenes by selectively removing Al or Si layers.41–44 In this context, Ti3AlC2 is used as a representative example to illustrate the general principle of direct HF etching, since it is the most extensively studied MAX phase for MXene synthesis. Different transition metals (e.g., Ti, Nb, V) provide unique electronic structures and redox properties, which influence catalytic activity. Furthermore, the interaction between the etching environment and the specific MAX phase affects the termination ratio, further customizing the catalytic properties.39,41 Therefore, although the etching principle remains consistent, the nanozyme activity of MXenes is not universal and is significantly affected by the choice of MAX precursor and the surface chemistry introduced during HF etching.
HF etching is common owing to its simplicity, versatility, and high yield, but concerns about safety and environmental impact persist because of its extreme toxicity and corrosiveness.45 The aggressive etching conditions can introduce defects, compromising the structural integrity and quality of the resulting MXene. Therefore, controlling HF concentration, reaction time, and temperature for each MAX phase composition is crucial to ensure successful transformation into MXene. HF etching typically produces multilayered MXene with an accordion-like structure, where adjacent MXene layers are connected by a combination of van der Waals forces and hydrogen bonding.46 Consequently, an extra delamination step is necessary to exfoliate these multilayered MXene structures into individual 2D nanosheets.
2.2. In situ HF etching
To ensure safety and minimize environmental impact, an alternative etching method was developed in 2014 by Ghidiu et al. This approach uses a LiF + HCl mixture to form HF in situ, enabling the selective etching of Al layers from the Ti3AlC2 MAX phase to synthesize Ti3C2Tx MXene.47 The LiF + HCl etchant is milder than HF, producing MXene flakes with larger lateral dimensions and fewer defects. Etching with metal halides results in intercalation between cations (such as Li+) and water molecules, leading to larger lattice parameters in MXene crystals. Fig. 2b shows the XRD patterns of multilayered Ti3C2Tx, with sharp (000l) peaks (pink trace). The lattice parameter was c ≈ 27–28 Å greater compared to the value (c ≈ 20 Å) obtained for HF-produced Ti3C2Tx. This increment was due to the intercalation of water molecules and metal cations between hydrophilic and negatively charged MXene sheets. The sample, after rolling into a thick film (∼40 μm), preserved the same c-direction peaks, but the major (110) peak was not seen, indicating reduced order in non-basal directions.This method has been extended to various fluoride salts, including NaF, KF, CaF2, CsF, FeF3, NH4F, NaHF2, and KHF2.47–50 Replacing HCl with milder acids like H2SO4 yields similar etching behavior. Ti3C2Tx synthesized using ammonium bifluoride (NH4HF2) etchant had intercalations of NH3 and NH4+ species, and its c-lattice parameter (∼25 Å) was 25% larger than that of HF-derived Ti3C2Tx films.51 The optimal ratio (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 M) of LiF + HCl used in the minimally intensive layer delamination (MILD) method eliminated the need for additional delamination steps required in direct HF etching. Manual shaking was sufficient to delaminate MXene into single large flakes.52 This one-pot process combines HF synthesis, etching, and intercalation into a single step. In situ HF etching offers advantages over direct HF etching, including larger lateral dimensions, lower defect concentration, improved stability, and increased interlayer spacing, due to intercalation of metal cations like K+, Na+, and NH4+ between MXene layers.53 While enabling milder exfoliation, this method is time-consuming and limited to specific MAX phase precursors for carbide and carbonitride MXenes. The etching process can be difficult to control, leading to over-etching and structural degradation. MXenes produced via in situ HF etching have surface terminations (–OH, –F, –O) dependent on etching conditions, often resulting in non-uniform surface chemistry.
9 M) of LiF + HCl used in the minimally intensive layer delamination (MILD) method eliminated the need for additional delamination steps required in direct HF etching. Manual shaking was sufficient to delaminate MXene into single large flakes.52 This one-pot process combines HF synthesis, etching, and intercalation into a single step. In situ HF etching offers advantages over direct HF etching, including larger lateral dimensions, lower defect concentration, improved stability, and increased interlayer spacing, due to intercalation of metal cations like K+, Na+, and NH4+ between MXene layers.53 While enabling milder exfoliation, this method is time-consuming and limited to specific MAX phase precursors for carbide and carbonitride MXenes. The etching process can be difficult to control, leading to over-etching and structural degradation. MXenes produced via in situ HF etching have surface terminations (–OH, –F, –O) dependent on etching conditions, often resulting in non-uniform surface chemistry.
2D MXenes can be synthesized using hydrothermal etching.54,55 Peng et al. produced Ti3C2 and Nb2C through hydrothermal etching using NaBF4 and HCl at 180 °C. Under reaction conditions, NaBF4 reacts with HCl to create HF in situ, which acts as the fluorine source for etching the parent MAX phase. As shown in Fig. 2c and d, the Ti3AlC2 precursor appears as a solid structure, but after 16 h of hydrothermal treatment, it transforms into a layered h-Ti3C2 structure, indicating broken Ti–Al bonds between layers. Compared to conventional HF-based etching, the hydrothermal approach provides more efficient Al layer removal and increases interlayer spacing in 2D Mxenes.56 Enhancing experimental safety and producing 2D MXene nanosheets with greater –O and –OH termination ratios and fewer defects are the two main benefits of the in situ HF etching method. Nevertheless, throughout the preparation process, cations (Li+, Na+, K+, etc.) will intercalate into MXenes' interlayers and are difficult to remove from MXenes, potentially impairing the final MXenes' hydrophilicity, catalytic activity, and electronic conductivity.39,41,44 Thus, it will affect the nanozyme sensors.
2.3. Fluorine-free acid etching
Fluorine-free etching has emerged as a safer synthesis approach that avoids the use of hazardous HF. Novel fluorine-free techniques are being developed. Researchers introduced thermal-assisted electrochemical etching using diluted HCl to prepare V2CTx, Cr2CTx, and Ti2CTx MXenes.39,57 A controllable HCl-assisted hydrothermal etching method has been reported for Mo2Ga2C, using 12 M HCl (40%) at 140 °C.58 The schematic process is shown in Fig. 2e. The as-prepared Mo2CTx had high purity with only –Cl and –O surface terminations and showed a layered structure with slightly increased interlayer spacing. During etching, concentrated HCl could inhibit oxidation, while lower temperature or diluted HCl would decrease efficiency and product quality.2.4. Molten salt etching
Aqueous etching methods are effective for most Al-based MAX phases at low temperatures, yielding hydrophilic MXenes, but are unsuitable for nitride-based MAX phases. To address this, a molten fluoride salt method was used by mixing Ti4AlN3 MAX with a fluoride salt mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio). The salt mixture contained KF (59 wt%), LiF (29 wt%), and NaF (12 wt%) corresponding to the ternary eutectic composition. Etching was performed at 550 °C for 30 min under an argon atmosphere, producing Ti4N3Tx MXene. The product was delaminated using tetrabutylammonium hydroxide (TBAOH) to produce multilayer- and monolayer-flakes of Ti4N3Tx (T = –F, –O, or –OH).59 During etching, Al layers were selectively removed while Ti–N bonds were preserved, maintaining the layered hexagonal Ti4N3 structure. Fig. 2f and g show TEM photographs of Ti4N3Tx flakes. The SAED of these flakes (inset in Fig. 1(g)) viewed normal to the basal (002) plane confirms their single crystalline nature and hexagonal symmetry (P63/mmc) retention from the parent MAX phase. The calculated lattice parameter (a ∼ 2.9 Å) matches that of the original MAX precursor, indicating that the in-plane structure remains unchanged after Al removal.60 Owing to ion intercalation during molten salt treatment and water molecules between layers after delamination, the out-of-plane lattice parameter (c) increases from 23 Å to ∼28 Å. This increase is smaller than that observed in previously reported MXenes, like HF-etched Nb4C3Tx (c-lattice parameter ∼31 Å), likely because of lower water content between Ti4N3Tx layers. This molten salt etching is preferred for MXenes with high formation energies and can be extended to other MAX phases for preparing MXenes.
1 ratio). The salt mixture contained KF (59 wt%), LiF (29 wt%), and NaF (12 wt%) corresponding to the ternary eutectic composition. Etching was performed at 550 °C for 30 min under an argon atmosphere, producing Ti4N3Tx MXene. The product was delaminated using tetrabutylammonium hydroxide (TBAOH) to produce multilayer- and monolayer-flakes of Ti4N3Tx (T = –F, –O, or –OH).59 During etching, Al layers were selectively removed while Ti–N bonds were preserved, maintaining the layered hexagonal Ti4N3 structure. Fig. 2f and g show TEM photographs of Ti4N3Tx flakes. The SAED of these flakes (inset in Fig. 1(g)) viewed normal to the basal (002) plane confirms their single crystalline nature and hexagonal symmetry (P63/mmc) retention from the parent MAX phase. The calculated lattice parameter (a ∼ 2.9 Å) matches that of the original MAX precursor, indicating that the in-plane structure remains unchanged after Al removal.60 Owing to ion intercalation during molten salt treatment and water molecules between layers after delamination, the out-of-plane lattice parameter (c) increases from 23 Å to ∼28 Å. This increase is smaller than that observed in previously reported MXenes, like HF-etched Nb4C3Tx (c-lattice parameter ∼31 Å), likely because of lower water content between Ti4N3Tx layers. This molten salt etching is preferred for MXenes with high formation energies and can be extended to other MAX phases for preparing MXenes.
The nonaqueous molten salt method uses Lewis acidic salts as etchants to synthesize MXenes without fluoride. In this method, MAX phase precursors are modified at high temperatures using transition metal halides in their molten state, which act as strong electron acceptors. M. Li demonstrated etching of the Ti3AlC2 MAX phase in a molten salt mixture of ZnCl2/NaCl/KCl under N2 atmosphere.61 Zn2+ cations react with Al atoms in the MAX phase, converting weakly bonded Al atoms into Al3+. The reduced Zn atoms intercalate between Ti3C2 layers and occupy Al sites, creating Ti3ZnC2, as shown in Fig. 2h. Excessive ZnCl2 etches the interlayer Zn atoms in the Ti3ZnC2 MAX phase, producing MXenes. The reaction outcome depends on the molar ratio of Al-MAX phase to ZnCl2. Because this approach is both nonaqueous and fluoride-free, the surface of the resulting ZnCl2-etched MXene is often terminated with –Cl groups instead of the typical –O, –OH, and –F groups. This approach extends to other MAX precursors like Ti2AlC, Ti2AlN, and V2AlC, resulting in MXenes with tunable surface terminations. Using Cl/Br molten salts yields MXenes with –Cl/–Br groups, shown by the EDX line scan in Fig. 2i, revealing –Br terminations on the Ti3C2Br2 surface.62 These –Cl/–Br groups can be substituted with –O, –S, or –Se groups using Li2O, Li2S, or Li2Se salts at high temperatures, while LiH eliminates surface terminations. Active ionic compounds readily replace surface terminations because of weak bonding between halogen and transition metal atoms. This enables substitution or elimination of terminations on the MXene surface, offering versatile surface chemistry control. The method is safer and suits more MAX precursors, though resulting MXenes are less hydrophilic and require complex experimental conditions.
2.5. Alkali etching
Alkali etching has become a secure and eco-friendly technique for creating MXenes with –O and –OH surface terminations, increasing their hydrophilicity. Despite advantages, alkali-based synthesis of MXenes is challenging as reactions occur spontaneously at high temperatures, making oxidation control difficult at low concentrations. MXenes produced via alkali etching show low stability because –OH terminations accelerate oxidation. Alkali compounds like KOH and NaOH were used at high temperatures to remove Al layers, particularly in Ti3AlC2, because of OH− binding with Al atoms. The alkali-assisted hydrothermal method, using NaOH and NaClO, offers a fluorine-free alternative and forms TiO2in situ, preventing Ti3C2Tx layer stacking.63 MXene nanoribbons could be synthesized via the hydrothermal method by etching Ti3AlC2 at 180 °C for 24 h in concentrated KOH solution (≈93.3 wt%).64 Li et al. achieved multilayer Ti3C2Tx (T = –O, –OH) MXene at 270 °C in 27.5 M NaOH solution with ∼92 wt% purity.65 The synthesized Ti3C2Tx shows a {002} plane spacing of 24 Å, which was larger than that of Ti3C2Tx (20 Å), produced via HF-based methods, suggesting Na+ ion intercalation from concentrated NaOH solution. XPS data confirmed reduced Al and Na contents in the final Ti3C2Tx product (Fig. 2j). The reaction temperature and alkali concentration are crucial for forming hydrophilic MXenes with high purity. However, these factors restrict large-scale MXene production. Alkali etching works best on Al-containing MAX phases because concentrated bases (NaOH and KOH) react with Al layers in a specific way to produce soluble aluminate species like [Al(OH)4]−. The Al “A-layer” may be effectively removed due to its high solubility, which yields the corresponding 2D MXene nanosheets. In contrast, the MAX phase containing other A layers of Si, Ga, and Ge generates extremely stable oxides or hydroxides (e.g., SiO2, Ga2O3) that are chemically resistant to dissolution under alkaline conditions. Under alkali circumstances, these compounds' weak solubility and inertness restrict successful 2D MXene production and full etching.39 These point out that the alkali etching technique works only for Al-containing MAX phases, limiting broader MXene family preparation.2.6. Electrochemical etching
The electrochemical etching route shows promise for selectively extracting nanolaminate materials from MAX phase precursors. Sun et al. performed electrochemical etching of the Ti2AlC MAX phase at 0.6 V (vs. Ag/AgCl) in a three-electrode setup.66 Yang et al. introduced a fluorine-free strategy using anodic corrosion of the Ti3AlC2 Max phase in a mild alkaline solution.67 The solution contains ammonium chloride (NH4Cl) and tetramethylammonium hydroxide (TMAOH) as a binary aqueous electrolyte. Ti3AlC2 served as both the anode and cathode. Cl− ions in the electrolyte caused rapid Al corrosion in the anode, breaking Ti–Al bonds. The XRD patterns showed the (002) peak of Ti3AlC2 shifting to a lower angle, corresponding to an increase in c-lattice parameter from 18.0 to 22.6 Å, because of termination groups and NH4OH molecule intercalation between Ti3C2Tx layers (Fig. 2k(i and ii)). The intercalated NH4OH molecules open the etched Ti3C2Tx anode edges, facilitating further etching. The Ti3AlC2 anode etches fully within 5 h under ambient conditions. The process yields over 90% monolayer or bilayer materials with lateral dimensions above 2 μm, larger than those for traditional HF etching methods. SEM images show that electrochemically etched Ti3C2Tx remains tightly stacked with a multilayered structure, unlike accordion-like HF-etched Ti3C2Tx structures, resembling bulk Ti3AlC2 (Fig. 2k(iii and iv)). Al layer dissolution and NH4OH intercalation produce Ti3C2Tx flakes (T = –O, –OH) up to 18.6 μm with high yield (>90%) of mono- or bilayer sheets. Electrochemical etching offers a rapid route for fluorine-free MXene synthesis. However, electrolytes require careful selection because of TMAOH safety concerns, and reaction time and etching potential significantly affect the final MXene structure.2.7. Halogen etching
Halogens have been explored as etchants for the MAX phase. Shi et al. demonstrated using iodine to etch Ti3AlC2 in anhydrous acetonitrile (CH3CN) at 100 °C, producing accordion-like Ti3C2Ix.68 The product was treated with 1 M HCl to eliminate the AlI3 byproduct from the etching reaction. The –I terminations were unstable and transformed to –OH and –O groups in HCl solution, leading to oxygen-rich Ti3C2Tx (T = –OH, –O) MXene sheets with high yield (71%), large lateral size (1.8 μm), and ultrathin structure (<5 nm). As shown in Fig. 2l(i–vi), the iodine-etched MXene sheets maintained excellent structural integrity and showed stability in water for over two weeks compared to fluoride-terminated MXenes from traditional methods. This technique needs expansion for synthesizing other MXenes, such as V2CTx, Cr2CTx, etc.2.8. Non-etching methods
The bottom-up technique can enable the fabrication of defect-free 2D monolayer MXene crystals, unlike the top-down method. Xu et al. synthesized large-area, ultrathin Mo2C crystals through chemical vapor deposition (CVD) at 1085 °C.69 Methane served as the carbon source, while Cu/Mo foil acted as the substrate. The Mo2C crystals were nanometers thick with a lateral size of ∼100 μm and good structural stability under ambient conditions. MAX and MXene thin film composites were fabricated using a two-step method involving ion beam sputtering.70 A low-energy ion facility (LEIF) using heavy ions was developed. Low-energy DC ion beam sputtering of elemental targets forms uniform layers of mixed phases or multilayers of individual phases; thermal annealing under vacuum enables diffusion and phase interactions to form desired composites. The first Ti-based MAX and MXene carbide (Ti2SnC and Ti2C) composite thin films were produced. However, MXenes prepared by bottom-up methods typically lack surface terminations and suffer from low yield and sophisticated equipment requirements. The Y2CF2 crystal, a stack of MXene-like sheets with fluorine terminations, was directly synthesized through solid-state reaction at high temperature without air and water.71 The unit cell of the 3D crystal was isostructural to MXene, but the layers are held together by ionic bonds. The yttrium carbide layers were electrically isolated by the fluorine layers, providing 2D-like electronic transport properties to this 3D crystalline material.MXenes' surface chemistry, structural integrity, and effectiveness as nanozymes are all determined by their synthesis process. For example, –O, –OH, and –F-terminated surfaces are commonly produced by standard HF and in situ HF etching; an excess of –F can impede electron transmission and lower catalytic performance. On the other hand, fluoride-free techniques, including halogen etching, electrochemical etching, alkali, or molten salt, frequently provide O-rich MXenes with better conductivity, redox activity, and environmental durability, which makes them more appropriate for catalytic and sensing applications (Table 1). Furthermore, considerations of safety, scalability, and cost vary widely across methods, with molten salt and in situ HF approaches showing the greatest promise for real-world applications.39,57 Therefore, the choice of synthetic protocol not only determines the characteristics of MXene but also plays a crucial role in determining its long-term viability and performance in nanozyme-based technologies.
| Method | Etching source | Surface terminations | MXene features | Scalability and limitations |
|---|---|---|---|---|
| Direct HF etching | HF acid | –F, –OH, –O | High yield; generates stable multilayer MXenes; conductivity is decreased by F-rich surfaces | Scalable yet risky because of HF handling, safety, and sustainability problems |
| In situ HF etching | LiF/HCl, NH4F/HCl | Tunable –O/–OH/–F ratios | Improved hydrophilicity; enhanced interlayer spacing from ion intercalation | Safer than direct HF; moderately scalable; properties depend on cation type |
| F-free acid etching | HCl | –Cl dominant, some –OH, –O | Controlled layer removal | Safer than HF; termination control is limited |
| Molten salt etching | KF, and NaF | –O, some –F | F-free MXenes; enhanced conductivity, thermal stability, and oxidation resistance | Large-scale potential; requires high temperatures and careful handling |
| Alkali etching | NaOH and KOH | –OH and –O | Produces MXenes with minimal –F; improved catalytic and electronic properties | Limited to Al-containing MAX phases; harsh conditions; scalability still under evaluation |
| Electrochemical etching | Electrochemical equipment | –OH and –O rich, fewer –F | Eco-friendly; control and precise etching; good for fine-tuning surface states | Low yield is not suitable for bulk production |
| Halogen etching | I2 | –OH and –O | Unique surface with good stability | Challenging to synthesize and needs more safety precautions |
| Non-etching methods | CVD, and ion beam sputtering | Customized terminations | High purity and tunable terminations | Early processes; costly, difficult, and at present limited in scalability |
3. MXzyme classifications
There are numerous ways to classify nanozymes. In their work, M. Zandieh et al.72 divided nanozymes into two categories based on development history. The first decade was dominated by type 1 nanozymes, which are catalysts or enzymes immobilized on nanomaterials since 2004. Type 2 nanozymes, extensively studied in the past decade, are based on the self-catalytic characteristics of inorganic nanomaterial surfaces. MXzymes with catalytic activity can be divided into three types: POD, oxidase (OD), and other catalytic nanozymes like catalase (CAT) and superoxide dismutase (SOD).73,74 Hydrolases work like phosphatases, nucleases, proteases, and other enzymes in catalyzing hydrolysis. CAT-like and SOD-like nanozymes are less common in colorimetric sensing as they are mainly used in biomedical applications for removing reactive oxygen species (ROS) and protecting cell membranes.73–75 It is important to note that CAT-like nanozymes exhibit relatively lower catalytic turnover in the absence of high H2O2 concentrations, which restricts their applicability in typical biosensing environments. Similarly, SOD-like activity is mechanistically more complex and often requires precise superoxide generation and detection systems, making it less suitable for straightforward sensing platforms compared with POD- and OD-like systems.1,75 Thus, POD-like and OD-like nanozymes are our main investigation focus.76,77In biosystems, oxidative enzymes POD and OD catalyze substrate oxidation under mild circumstances by activating H2O2/O2.36,79 MXenes' large specific areas make them suitable supports for enzymatic events, with physical and chemical features enhancing supported NP stability and POD catalysis.78 O2 is the oxygen supply used by OD to catalyze oxidation.36 Jin et al.79 hydrothermally treated Ti3C2Tx MXene to create TiO2-loaded carbon quantum dots (TiO2/C-QDs), an active OD nanozyme, developing a quick colorimetric technique for GSH identification. CATs and SODs are antioxidant enzymes crucial in scavenging excess ROS and preserving redox balance. These nanozymes catalyze O2 conversion into H2O2 and O2, created by O2 and H2O breakdown and OH radical scavenging.80,81 Nb2C nanosheets modified with polyvinylpyrrolidone (PVP) were created by liquid-phase exfoliation, showing SOD-like features from surface oxidation.82 Several MXene-based nanozymes exhibit multiple enzymatic activities.83–85 V2C-based nanozymes demonstrate six enzyme-like properties (SOD, CAT, POD, GPx, TPx, and HPO), enabling biocompatibility and cytoprotection against oxidative stress.29
4. Factors affecting MXzyme-based sensors
MXzyme's enzymatic activity is influenced by its composition, oxidation state, and surface structure. The inclusion of transition metals like Nb and Ti in MXene could assist in transferring electron activities, crucial for reaction catalysts.38 The catalytic functionalities of MXene-based nanozymes can be modulated by altering MXene components. MXzymes can generate hybrids with improved catalytic activity.37,38 Additionally, MXene's layered structure offers a vast surface area and improved catalytic activity. Because thinner nanosheets provide more surface area and catalytically active sites, the lateral dimension and thickness of MXene nanosheets influence enzymatic activity. Altering electrical characteristics and active site accessibility directly influences enzyme functioning. MXzyme' catalytic activity can be impacted by surface alterations, including functional groups and surface charges.38 The synthesis conditions and chemical treatment can accommodate various functional groups on the MXene surface, including –O, –F, and –OH. The pH and temperature regulate the catalytic activity of nanozymes based on MXene.86 pH has a significant effect on catalytic properties. The catalytic capabilities can be controlled using photo-responsive technology. Under near-infrared (NIR) irradiation, the catalytic capabilities of MXzymes increase owing to high photothermal conversion efficiency in the NIR range.37 MXene's enzyme-like activity can be enhanced by considering and refining these enzymatic qualities.5. MXzyme-based sensor classification
Based on the enzyme mimic features and detection modes of MXene supported nanostructured materials, various sensing platforms such as colorimetric, smartphone assisted colorimetric, fluorescence, chemiluminescence, electrochemiluminescence, electrochemical, and SERS sensors have been explored for the detection of a wide range of analytes. The following sections will comprehensively discuss these sensing systems in detail.5.1. MXzyme colorimetric sensors
In chemical analysis, naked-eye visual analysis is a strong method that determines substance concentration by measuring solution color intensity through color absorption.87,88 Reliability, affordability, and convenience are its main advantages, driving advancements in biomolecule identification.88,89 ELISA, which uses an enzyme to activate a chromophore and produce a discernible color shift in solution, is among the most widely used methods.90 This change is visible to the naked eye or can be evaluated using a spectrometer. This method is used for quick detection in sensor technologies and is effective.91,92 However, detection sensitivity is limited by the low extinction coefficient of organic chromophores, which presents a problem.The following elements are typically included in colorimetric observing mechanisms, depending on the kinds of substances or reactions employed to fabricate colorimetric sensors:
(i) Target-induced changes in interparticle distance, size, shape, or composition cause the plasmonic NPs (such as Ag and Au NPs) to change color through surface plasmon resonance (SPR).91,92
(ii) Fluorescent on–off switching or photonic crystal color changes in the direction of targets.93,94
(iii) Catalyzed by natural enzymes, enzyme mimics, synthetic enzymes, or nanozymes, the chromogenic reaction of several substrates, including 3,3′,5,5′-tetramethylbenzidine (TMB).15
(iv) The direct or indirect production of colored products or the colorimetric alteration of chromogenic agents brought on by the targets through non-enzymatic processes.95
Enzyme mimic-based colorimetric sensors are recent developments in this field.37,96–98 A substrate is converted by nanozymes into an optically detectable product, producing a quantifiable signal. Catalytic turnover, where a single nanozyme speeds up many substrate-to-product conversion events, causes signal amplification that increases recognition sensitivity.97,98 Compared to conventional noncatalytic AuNPs, nanozymes with intrinsic optical characteristics provide increased detection sensitivity, improving the detection limit by up to 10 times.96 The optical contributions of nanozymes in colorimetric assays extend beyond catalysis. An improvement in detection limit can significantly impact assay diagnostic precision. Advancements are underway to engineer nanozymes with superior optical features by tuning their physical attributes (size, shape) and chemical composition (element distribution).15,99,100Table 2 summarizes the substrates for these enzyme mimic catalyzed reactions, which are chromogenic compounds.
 | ||
| Fig. 3 MXzyme-based colorimetric detection of heavy metal ions. (a) Pt NPs on Ti3C2Tx MXene nanoribbons (Ti3C2Tx MXene NR) were synthesized by the hydrothermal method and utilized to detect Hg2+ ions by POD activity [reproduced from ref. 101 with permission from Elsevier, copyright 2024], (b) DNA-encoded MXene–Pt nanozyme for highly sensitive colorimetric detection of Hg2+ ions by POD activity [reproduced from ref. 102 with permission from Elsevier, copyright 2022], and (c) Ti3C2Tx MXene NR hybrid decorated with self-reduced AuNPs for highly sensitive POD-like colorimetric detection of Hg2+ ions and Cys [reproduced from ref. 103 with permission from Elsevier, copyright 2023]. In (i) and (ii), the absorbance spectral profile at 652 nm and colorimetric changes (Au/Ti3C2Tx MXene NR + TMB + H2O2) with various concentrations of Hg2+ ions are represented, respectively. | ||
 | ||
| Fig. 4 MXzyme-based colorimetric detection of biothiols. (a) Mo atom nanoclusters/N–Ti3C2Tx MXzymes based POD-like sensing of GSH [reproduced from ref. 106 with permission from Elsevier, copyright 2024], (b) TiO2/C-QDs-Ti3C2Tx MXene composite MXzyme based OD-like sensing of GSH, and (i) and (ii) represent the OD-like behavior of the TiO2/C-QDs-Ti3C2Tx MXene composite and sensitive detection of GSH at increasing concentrations, respectively [reproduced from ref. 79 with permission from Royal Society of Chemistry, copyright 2020]. (c) OD-like V2C MXzyme with antibacterial qualities for colorimetric sensing application of Cys [reproduced from ref. 112 with permission from Elsevier, copyright 2023]. | ||
The need for quick, sensitive, and consistent uric acid (UA) detection has grown because of increasing cases of conditions like osteoarthritis linked to abnormal UA levels.114 Sensor materials have attracted interest owing to affordability, ease of use, and electromagnetic interference resistance. Research has focused on UA direct identification for continuous monitoring and quick diagnosis. A “naked-eye” colorimetric and ratiometric fluorescence probe using GSH-functionalized Ti3C2Tx MXene quantum dots was developed.115 The technique used uricase to oxidize UA, yielding H2O2 and allantoin, which oxidized OPD to 2,3-diaminophenazine. The UV absorption of oxOPD at 425 nm matched the fluorescence emission of GSH-Ti3C2Tx MXene QDs at 430 nm, enabling ratiometric UA measurement and FRET (Fig. 5a). The probe showed stability, selectivity, and a LOD of 125 nM. N and S co-doped Ti3C2Tx MXene nanosheets were developed for UA detection using TMB oxidation.116 Thiourea enhanced the material's POD-like activity by improving active sites and electron transport. The catalytic mechanism involved protonating TMB and H2O2 dissociation. The ZnO–Co3O4 nanofiber/MXene composite's POD-like colorimetric features detected ascorbic acid (AA).117 The MXene-supported nanocomposite showed rapid reaction kinetics and proved suitable for food analysis through orange juice testing.
 | ||
| Fig. 5 MXzyme-based colorimetric sensors for biologically important analytes. (a) Colorimetric and ratiometric fluorescence probe for detecting UA by GSH-functionalized Ti3C2Tx MXene QDs, and the inset shows colorimetric changes of nanozyme with different quantities of UA under daylight and UV light [reproduced from ref. 115 with permission from Elsevier, copyright 2020], (b) POD-like activity-based colorimetric sensing strategy for glucose using CuNPs/N-doped Ti3C2Tx MXene hybrids [reproduced from ref. 119 with permission from American Chemical Society, copyright 2022], (c) Ti3C2Tx MXene/AuNPs heterostructure nanozyme-based detection of penicillin G and the possible sensing mechanism [reproduced from ref. 125 with permission from Elsevier, copyright 2024], (d) Ti3C2Tx MXene supported-Fe3O4@PDA-aptamer capture nanoprobe for direct capture and detection of BC-CTCs [reproduced from ref. 128 with permission from Elsevier, copyright 2023], and (e) dendritic-like Ti3C2Tx MXene QDs@CuNi nanozyme-based colorimetric detection of glyphosphate, and the inset shows the colorimetric changes in the various concentrations of glyphosphate [reproduced from ref. 133 with permission from Elsevier, copyright 2024]. | ||
Glucose is the primary energetic source in our bodies and is essential to metabolic processes. Diabetes prevalence is rising in emerging nations because of economic growth and urbanization. Insulin dosages can be modified to maintain glucose levels within permitted limits and reduce complications through accurate glucose assessment.118 A POD-like activity-based colorimetric sensing strategy for glucose using CuNPs/N-doped Ti3C2Tx MXene hybrids was established.119 CuNPs were anchored onto N-doped Ti3C2Tx MXene through simple synthesis, producing a nanohybrid with stable structures and significant catalytic power (Fig. 5b). The oxidation of TMB with H2O2 resulted in a blue oxidized product with absorbance at 652 nm. The glucose sensor showed excellent sensitivity, specificity, and reliability with a LOD of 5 μM. It retained POD-like properties after a month of storage. Similarly, Pt/Ti3C2Tx MXzyme was applied for glucose and GSH detection.109 The optimized Pt/Ti3C2Tx MXzyme showed POD-like activity 7.9 times higher than that of Pt alone under NIR radiation, and six times higher in the dark. Yu He's team reported Ti3C2Tx MXene/Co nanosheet hydrogel sensors for glucose recognition via smartphone-based point-of-care testing.120 The Ti3C2Tx MXene/Co nanosheet material catalyzed TMB by H2O2, producing a blue-colored product. A sodium alginate hydrogel-embedded Ti3C2Tx MXene/Co nanosheet sensor enables real-time glucose measurement using RGB analysis and a smartphone app. G. Yang's team developed a glucose-sensing method through an immunoassay for IR-β detection using Ti3C2Tx MXene's catalytic activity.29 These studies showed MXene-based nanozymes' potential for rapid, portable glucose sensing.
Cholesterol is necessary for human survival, but can pose serious health risks when accumulated beyond certain thresholds. Keeping cholesterol intake regulated is essential for maintaining key physiological processes.121 Y. Chen et al. described an Ag/Ti3C2Tx MXzyme with boosted POD-mimicking activity for antibacterial applications and cholesterol sensing.122 The Ag/Ti3C2Tx MXzyme produced by in situ reduction indicated higher catalytic efficiency. Both Gram-positive and Gram-negative bacteria were inhibited by their conversion of H2O2 into hydroxyl radicals, which disrupted bacterial biofilms. The Ag/Ti3C2Tx MXzyme's POD-like activity was coupled with cholesterol oxidase to create a TMB-based colorimetric biosensor. This sensing technique showed accuracy when used to detect cholesterol in human blood specimens. According to TMB oxidation, Y. Li et al. reported an MXene-supported CuS nanozyme for cholesterol sensing.123
Hepatitis B virus (HBV) infection is linked to human hepatitis disorders, including chronic hepatitis, liver fibrosis, cirrhosis, and hepatoma. Researchers introduced a CRISPR-Cas12a-based colorimetric biosensor using the DNA-modulated POD-mimic activity (TMB) of Ti3C2Tx MXene–Ag/Pt nanohybrids.129 A colorimetric reaction occurred when Cas12a enzyme triggered trans-cleavage with HBV DNA, breaking down the DNA probe and inhibiting the nanohybrid's catalytic activity. The biosensor tested human serum samples with excellent sensitivity and sub-picomolar LOD. A smartphone-based platform enabled real-time, visible detection suitable for field use.88 This work demonstrated sensitive viral DNA detection by combining CRISPR technology with nanozymes. Wenhai Wang et al. developed a dual-mode aptasensor for detecting Vibrio parahaemolyticus (V.P.) using Pt, phenyl boric acid (PBA), and a ferrocene-modified Ti3C2Tx MXene nanoprobe (PBA-Fc@Pt@Ti3C2Tx MXene).130 The aptasensor collected bacteria using an aptamer-functionalized electrode, combining colorimetric and electrochemical detection. The PBA-Fc@Pt@Ti3C2Tx MXene nanohybrid formed a sandwich complex, catalyzing TMB oxidation with H2O2 to generate visible and electrochemical signals. Detection limits were 30 CFU mL−1 in colorimetric mode and 5 CFU mL−1 in electrochemical mode. The platform could detect other diseases by changing the aptamer, offering a versatile tool for pathogen screening.
According to theoretical calculations, the interaction between CuNi bimetal and MXene quantum dots, which promoted H2O2 activation, caused the high catalytic efficiency. Using this strategy, Ti3C2Tx MXene-supported nanostructured materials showed POD-like catalytic performance for detecting dipterex, astaxanthin, and guanosine 5′-monophosphate.134–136 MXzyme-based colorimetric sensors primarily use Ti-based MXenes, limiting their usefulness. Exploring Mo, V, and Nb MXenes may reveal improved catalytic activity, permanency, and discrimination, expanding detection applications. These alternate MXenes have diverse electronic structures and surface chemistries, which may improve sensitivity and detectability. Extending beyond Ti-based MXzymes can revolutionize colorimetric sensing, enabling next-gen, quick, cost-effective detection systems in environmental and biomedical areas.
5.2. MXzyme-based smartphone-integrated colorimetric sensors
Using colorimetry, which compares or measures the color depth of colored substances, one can ascertain the concentration of the targets based on the chromogenic reaction of colored compounds.137–139 Colorimetric sensors operating on smartphones can be exploited away from a lab to satisfy critical requirements, such as on-site monitoring and clinical testing. By adjusting parameters and accessories, researchers can maximize colorimetric image quality, which correlates with detection sensitivity.140 This smartphone sensor can recognize various colors, determine object hues, and assist visually impaired or color-blind people. Colors are captured by a smartphone camera after generation.141 After converting the image to color values like red, green, and blue (RGB), finding the ideal mathematical connection follows. In smartphone colorimetric technology, 0,0,0 and 255,255,255 signify absolute white and black. Smartphone technology is more useful than computer programs as it requires no data transmission and is easily accessible on phones.142,143 Commercial apps like home-grown, Color Picker, and Color Grab, built for iOS and Android, serve as examples.4 Most colorimetric sensors based on nanozymes rely on colorimetric fluctuations visible to the naked eye. However, subtle color variations cannot be seen by the naked eye.142 Smartphones have examined MXene-supported composite materials using enzyme-mimic colorimetric sensing to maintain detection accuracy. Colorimetric study depends on factors like ambient light intensity, label-to-background contrast, and human visual perception alongside NP optical properties. Portable detectors increase measurement reliability and reduce subjective interpretation.One endogenous plasma component considered as a possible cardiac biomarker is hypoxanthine (1,7-dihydro-6H-purin-6-one, Hx), which is generated after the ATP breakdown process. After 15 minutes of heart ischemia, the blood's Hx content rises, making it more responsive to acute myocardial infarction than conventional protein indicators. As a result, Hx is a perfect fit for cardiac indicators that will help detect acute myocardial infarction early and treat it quickly.144 In light of this, a Cu nanocluster (CuNC)-decorated Ti3C2Tx MXene nanosheet composite with tetra-enzyme mimic activities for detecting Hx with the assistance of a smartphone was developed and is displayed in Fig. 6a.145 Ascorbic acid oxidase (AAO), POD, (CAT), and SOD mimic activities were demonstrated by the (CuNC)-decorated Ti3C2Tx MXene nanosheets; the synergistic impact increased catalytic efficiency. This sensing study developed a quick, smartphone-supported colorimetric Hx sensing technique that achieved a LOD of 0.25 μM and a detection range of 5–200 μM. Using a smartphone app, the sensor converted discernible color changes into numerical RGB values, enabling real-time Hx monitoring in aquatic products. Enhanced electron transfer was identified as the catalytic mechanism of the POD-mimic activity instead of the production of hydroxyl radicals. According to the study's findings, CuNC-decorated Ti3C2Tx MXene nanosheets offer a portable, quick, and affordable sensing platform for applications involving food safety. The next example is the development of a Co(OH)2/Ti3C2Tx MXene nanocomposite with triple-enzyme mimic activities, including OD, POD, and CAT, for the smartphone-integrated colorimetric detection of Hx.146 A sodium alginate hydrogel was used to embed the Co(OH)2/Ti3C2Tx MXene nanocomposite, resulting in a hydrogel-assisted detecting device for on-time sensing of Hx (Fig. 6b). This investigation established that Hx was catalyzed by xanthine oxidase (XOD) to yield H2O2, which reacted with TMB in the presence of the Co(OH)2/Ti3C2Tx MXene nanocomposite, producing a visible blue color at a wavelength of 650 nm. The researchers also utilized sodium alginate hydrogel, a colorimetric signal as an indicator for Hx detection, which was further integrated with a simple smartphone. Rapid Hx quantification was made possible by the smartphone-based detection approach, which achieved a LOD of 0.2 μM and a linear range of 5–250 μM. High selectivity, stability, and repeatability were demonstrated by the sensor, and its practical usefulness was confirmed by real sample analysis in aquatic products. Using a similar TMB oxidation, Xuemin Zhou's research group developed a smartphone-aided colorimetric detection of glucose based on the Fe3O4@MXene–Au nanocomposites with high POD-like activity.147 With a high recovery rate, it was able to identify glucose in human whole blood samples. Furthermore, a portable detection technology based on agarose hydrogel was created for real-time analysis, enabling glucose quantification using a smartphone. This Fe3O4@MXene–Au nanocomposite-based nanozyme glucose biosensor demonstrated a linear range from 0 to 1.4 mM and a LOD of 0.11 mM.
 | ||
| Fig. 6 MXzyme-based smartphone colorimetric detection of potent analytes. (a) CuNC-decorated Ti3C2Tx MXene nanosheet composite with tetra enzyme mimic activities for detecting Hx with the assistance of a smartphone [reproduced from ref. 145 with permission from Elsevier, copyright 2022], (b) (i) and (ii) represent the synthesis and nanozyme activity for smartphone colorimetric sensing of Hx by using the Co(OH)2/Ti3C2Tx MXene nanocomposite and sodium alginate hydrogel embedded Co(OH)2/Ti3C2Tx MXene nanocomposite, respectively [reproduced from ref. 146 with permission from Springer Nature, copyright 2022], (c) (i) Nb2CTx@PdPt nanozyme based POD mimic activity based sensing of OPs, (ii) preparative protocol for the hydrogel, and (iii) smartphone-based sensing of OPs with the corresponding calibration plot [reproduced from ref. 149 with permission from Springer Nature, copyright 2024], (d) synthesis and POD activity based smartphone colorimetric sensing of OA [reproduced from ref. 155 with permission from Elsevier, copyright 2025]. | ||
Although the widespread use of organophosphate pesticides (OPs) has seriously harmed both human health and the environment, it is still very difficult to identify them on-site and with high effectiveness.148 In this connection, a colorimetric detection method for OPs using Nb2CTx MXene is integrated with a PdPt nanozyme and hydrogel for smartphone-assisted analysis. Using a self-reduction technique, the Nb2CTx@PdPt nanozyme was produced and demonstrated potent POD-like activity.149 According to this work's findings, the Nb2CTx@PdPt nanozyme's catalytic activity was suppressed in the presence of acetylcholinesterase (AChE) and acetylcholine chloride (ATCh), but it was restored upon exposure to OPs, enabling extremely sensitive detection (Fig. 6c(i)). The established glyphosphate distinguishing platform attained a linear range of 1.0 ng mL−1 to 1.0 μg mL−1 with a LOD of 0.419 ng mL−1. The hydrogel-based sensor provided a practical and portable detection approach by increasing stability and enabling real-time analysis through smartphone images. Nb2CTx@PdPt nanozyme, working together with a smartphone-based platform, was found to be an efficient, inexpensive, and quick on-site monitoring tool for food safety applications (Fig. 6c(ii and iii)). Furthermore, Ti3C2Tx MXene modified with platinum nanoparticles was utilized for the detection of pesticide-like chloramphenicol (CAP) in animal-derived foods.150 Based on this AChE activity, a Pd–Pt bimetallic-modified MXzyme was developed for the highly sensitive portable device (paper and smartphone) integrated colorimetric detection of AChE inhibitors, which are critical in Alzheimer's disease (AD) treatment.151 For real sample analysis, a paper-based sensor device was created, facilitating quantitative detection using a smartphone. The technique effectively identified AChE inhibitors in pharmaceutical tablets and extracts from medicinal plants, exhibiting great sensitivity, specificity, and stability. It is well known that Hg2+ ions are highly toxic. For the detection of Hg2+ ions, a smartphone-assisted fluorescence and colorimetric method was developed for on-site detection of Hg2+ and Cl− using the oxidase-like activity of Au–Hg alloys on Au/Cu/Ti3C2Tx MXene nanosheets.152 Upon adding Hg2+ ions, Hg–Au alloys catalyzed OPD oxidation, producing a fluorescence signal at 570 nm, permitting turn-on detection of Hg2+ ions. Then, the introduction of Cl− quenched the fluorescence of 2,3-diaminopenazine, allowing turn-off detection of Cl−. A smartphone-based colorimetric analysis further facilitated on-site monitoring. The Au/Cu/Ti3C2Tx MXene nanosheet-based approach enhanced environmental sensing applications. Furthermore, metal–carbon showed POD activity of TMB-based nanozymes and is also utilized for the smartphone-assisted colorimetric detection of pyrophosphate anions.153 This work illustrated the growing use of nanozymes with POD-like activity in pyrophosphate synthesis and will accelerate the development of useful sensors while ensuring environmental safety. The main toxin responsible for diarrheal shellfish poisoning is okadaic acid (OA), which is extremely dangerous to human health.154 To detect OA, a smartphone-assisted colorimetric aptasensor was developed for the ultrasensitive detection of OA using DNA-encoded porous Ti3C2Tx MXzymes (Apt-P-Ti3C2Tx MXene).155 The porous Ti3C2Tx MXene was synthesized via microwave combustion, creating unsaturated Ti center edges and residual Mn2+, which enhanced the POD-like catalytic activity of TMB. A detectable colorimetric shift resulted from the presence of OA, which decreased Apt-P-Ti3C2Tx MXene's catalytic effectiveness, which was integrated with a portable smartphone (Fig. 6d).
By achieving great sensitivity, mobility, and real-time detection, these research works did away with heavy laboratory equipment. Smartphone imaging strengthened its role in next-generation biosensors by enabling quick and affordable analysis for environmental monitoring, diagnostics, and food safety. Real-time detection will be transformed by future developments in smartphone-assisted MXene-based biosensors, which will combine multi-target sensing, IoT connectivity, and AI-driven analytics for unmatched precision in environmental monitoring, food safety, and healthcare. Enhanced nanozyme engineering and miniaturized platforms will drive ultra-sensitive, portable diagnostics, enabling rapid, cost-effective, and data-driven decision-making on a global scale.
5.3. MXzyme-based luminescence sensors
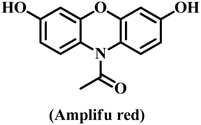
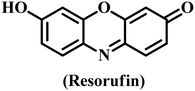
Among the different analytical approaches, the fluorescence spectral technique is a good choice for the detection of potential analytes because of its simple setup, good spectral resolution, efficiency, 100-fold higher sensitivity than colorimetric approaches, and excellent specificity.156–158 Chromogenic compounds have been utilized as substrates in the majority of nanozyme tests because they can react to modify wavelength in the visible range (350 nm to 800 nm). As a chromogenic substrate, TMB is most frequently used in colorimetric nanozyme sensors.159 Tragically, the lack of fluorescence in this molecule led scientists to hunt for alternative chemical substrates.159–161 Fluorescent and luminous signals are necessary for some applications, like bioimaging. In this regard, both OD and POD enzymes most frequently utilize Amplex red (AR) as a fluorogenic substrate.162 At neutral pH, its oxidation product, resorufin, exhibits a bright orange fluorescence peak at 585 nm; however, the fluorescence yield sharply declines at decreasing pH. The second of these fluorescence nanozyme substrates is O-phenylenediamine (OPD). It is a naturally occurring aromatic diamine. OPD is catalytically oxidized by hydroxyl radicals formed when H2O2 peroxidase is reduced, resulting in the formation of 2,3-diaminophenazine, which is highly fluorescent. OPD is excited with a wavelength of 365 nm, and its maximum emission intensity is reported at wavelength λ = 568 nm.163,164 OPD's primary drawback is associated with the nonspecific character of its oxidation. OPD is simply oxidized not only by H2O2 but also by other oxidizing agents. D-Luciferin and luminol molecules were frequently utilized in bio and chemiluminescence, respectively.159The basic process that produces an analytical signal is used to classify nanozyme-based fluorescence sensors. The first class includes fluorescence sensors in which the chemical relationship between the potential analytes and the catalytic substrate affects the emission maximum intensity. These sensors frequently support cascade catalytic events by using both natural enzymes and nanozymes. Enzymes such as glucose oxidase, uricase, and choline oxidase adjust H2O2 generation, which in turn disturbs the emission signal variation.163,164 The second group comprises fluorescence sensors where fluorescence is dictated by the direct interface between the impact analytes and nanozymes. The analytes can interact with catalytic nanostructured materials, fluctuating their enzyme-like activity.165 The interaction between the nanozyme and the analyte can boost or reduce the fluorescence of the nanozyme itself. Furthermore, the analyte could influence enzyme mimetic performance through interactions with the reaction's intermediate products.166 The third group includes nanosensors in which the analyte–catalytic product interactions modify luminescence. The analyte uses an “on–off–on” process to restore light emission after quenching agents are frequently added.167,168 Nanozymes pre-functionalized with recognition molecules (such as aptamers or antibodies) produce luminescence proportionate to analyte binding in the fourth category of bio-specific nanozyme sensors.
The previously stated chromogenic compounds serve as substrates for nanozymes that resemble OD and POD activity-based fluorescence sensors. Similarly, such substrates can be modified for luminescence-based detection by the use of different nanozymes, such as metal nanoparticles, metal oxide nanoparticles, carbon-based nanomaterials, MXene, graphene, and metal–organic frameworks.168–172 The detection of various ions, chemicals, and proteins is made possible by these nanozymes, either by themselves or in combination with recognition molecules such as aptamers and antibodies.169 Since the majority of fluorescent nanozyme systems function via oxidation reactions, they are extremely vulnerable to antioxidants, which can considerably reduce the oxidation of luminescent substrates.160 Furthermore, the surface properties of nanozymes affect their catalytic activity, which enables the selective detection of analytes by activity modulation. The creation and use of nanozymes based on composite materials developed from MXene have garnered much attention to date.160,173,174 Therefore, nanozymes are used based on composite materials obtained from MXene in chemical and biological sensing through luminescent transduction.
As a potent antioxidant that is soluble in water, ascorbic acid (AA) can lessen the harm that ROS does to DNA, lipids, and proteins. However, diarrhea, urinary tract stones, and stomach irritation can readily result from consuming excessive amounts of AA.175 Therefore, developing efficient methods for measuring AA in actual samples is essential. For the selective detection of AA, J. Huang et al. created a fluorescence-based sensor by incorporating Cu nanoparticles (CuNPs) into nitrogen-doped Ti3C2Tx (N–Ti3C2Tx) MXzymes.176 Because of the CuNPs/N–Ti3C2Tx nanocomposite's potent ascorbic acid oxidase (AAO)-like catalytic activity (Fig. 7a), AA was oxidized to dehydroascorbic acid (DHAA), which then interacted with OPD to generate a fluorescent molecule. This mechanism enabled a sensitive detection method of AA with a linear range of 5–150 μM and a LOD of 0.437 μM (Fig. 7a(i)). This research showed its excellent specific detection of AA (Fig. 7a(ii)). Furthermore, the MXzyme efficiently produced ROS, inhibiting the growth of cancer cells while posing little threat to healthy cells. While theoretical studies demonstrated how nitrogen doping can improve the material's performance, advanced characterization techniques verified the material's structural integrity and catalytic effectiveness. Through ROS-mediated cytotoxicity, this research showed the dual utility of CuNPs/N–Ti3C2Tx for biomedical applications, providing a promising strategy for both AA sensing and cancer therapy.
 | ||
| Fig. 7 MXzyme-supported fluorescence sensors for potent analytes. (a) CuNPs/N–Ti3C2Tx nanocomposite's potent AAO-like catalytic activity-based fluorescence detection of AA, and (i) and (ii) sensitive and selective detection of AA, respectively [reproduced from ref. 176 with permission from the American Chemical Society, copyright 2023]. (b) DNAzyme-ferrocene-modified Ti3C2Tx MXene/Au nano bipyramid/antimicrobial peptide-based sensing of food-borne pathogen [reproduced from ref. 178 with permission from Elsevier, copyright 2023]. | ||
As a major global public health concern, foodborne microorganisms in human food cause dangerous and deadly infectious illnesses, including fever, diarrhea, and even death.177 At this point, scientists constructed a dual-signal biosensing approach for the quick screening and quantitative identification of the common foodborne pathogen Vibrio parahaemolyticus (V.P.).178 The signal probe was a DNAzyme-ferrocene-modified Ti3C2Tx MXene/Au nano bipyramid/antimicrobial peptide (MAADF) nanocomposite, which combined fluorescence and electrochemical detection. When it interacted with V.P., a sandwich immunocomplex developed on the sensing electrode, allowing for the production of a fluorescent signal for visual screening through a catalytic click reaction and an electrochemical signal for accurate quantification from the ferrocene component (Fig. 7b). The accuracy of detection was greatly increased by the integrated cross-reference correction. The biosensor demonstrated a low detection limit of 6 CFU mL−1 and a wide detection range of 10–108 CFU mL−1. High specificity, stability, and reproducibility were displayed by the created sensor, which also showed little interference from other microorganisms. Additionally, real seafood samples were used to validate its efficacy, and good recovery rates were obtained. This strategy provided benefits like dual-mode precision, on-site applicability, and quick reaction as compared to traditional techniques. According to this study's findings, this biosensor offered a viable and effective way to identify foodborne pathogens, and by altering the capture probes, it might be further tailored to additional bacterial targets.
Aflatoxin B1 is extensively found in agronomic products (such as corn, peanuts, soybeans, etc.) and nutrition.179 The World Health Organization's cancer research department has classified AFB1 as a Class I carcinogen, making it one of the most dangerous mycotoxins with exceptionally high cancer risk, genetic mutation, fetal harm, and other toxicities, posing a serious threat to human health.180 The food industry needs to develop affordable, portable, rapid, accessible, and ultrasensitive approaches for AFB1 detection immediately. To detect AFB1 in peanuts with high sensitivity, the X. Li research group created a multimodal biosensor based on Ti3C2Tx MXene-based nanozymes.181 Fluorescence-quenching Ti3C2Tx MXzymes with POD-like activity were created and functionalized using an AFB1-specific fluorescein-labeled aptamer (Fig. 8a). By using colorimetric, fluorescence, and smartphone-based detection techniques, this sensor was able to achieve LODs of 0.09 ng mL−1, 0.61 ng mL−1, and 0.96 ng mL−1, respectively. The fluorescence detection relied on the quenching effect of Ti3C2Tx MXene on the carboxyfluorescein-labeled (ssDNA-FAM)-aptamer, which was reversed upon AFB1 binding. The POD-like activity of Ti3C2Tx MXene, which oxidized TMB and produced a color shift regulated by aptamer binding to AFB1, served as the basis for the colorimetric assay (Fig. 8a). The smartphone mode used a calibration model to translate color intensity into AFB1 quantities. The described sensor exhibited great stability over seven days and outstanding selectivity against probable interferents. Analyzing spiked peanut samples confirmed its accuracy and demonstrated its practical usefulness, producing recoveries exceeding 102%. This sensing protocol highlighted the potential of Ti3C2Tx MXene-based nanozymes for multiplexed toxin quantification and underlined the need for further improvements to enhance stability for long-term storage under ambient conditions. The same research group established the improvement of a novel biosensor based on V2C MXzyme materials (V2C MNs) for the detection of AFB1 in peanuts.182 Using 6-carboxyfluorescein-labeled aptamers (ssDNA-FAM), the researchers altered V2C MNs with exceptional suppression of fluorescence and POD-like function to produce a sensor called V2C@ssDNA-FAM. This sensor demonstrated high sensitivity with an LOD of 0.0477 ng mL−1 in fluorescence mode and 0.2789 ng mL−1 in colorimetric mode (Fig. 8b). In addition, a detection platform utilizing artificial neural networks (ANNs) was created to examine colorimetric and fluorescence signals, with detection limits of 0.6845 ng mL−1 and 0.0905 ng mL−1, respectively. In addition, a detection platform utilizing artificial neural networks (ANNs) was created to examine colorimetric and fluorescence signals, with detection limits of 0.6845 ng mL−1 and 0.0905 ng mL−1, respectively. Through automated data analysis and a decrease in false positives and negatives, the ANN-based method improved accuracy (Fig. 8b). The study emphasized the sensor's potential in food safety applications by highlighting its higher accuracy when compared to current techniques. This investigation recommended that the combination of nanomaterials and artificial intelligence (AI) could transform toxin detection in food, offering a real-time, intelligent, and highly sensitive method for ensuring food safety.
 | ||
| Fig. 8 MXene-supported fluorescence sensors for potent analytes. (a) Synthesis and Ti3C2Tx MXene-based nanozymes for the multimodal detection of AFB1 [reproduced from ref. 181 with permission from Elsevier, copyright 2023]. (b) Artificial neural networks are involved in V2C MXzyme materials (V2C MNs) for detecting AFB1 in peanuts [reproduced from ref. 182 with permission from Elsevier, copyright 2024]. (c) POD-like activity of Ti3C2 MXene/AuPt nanozymes based sensing of carbosulfan [reproduced from ref. 187 with permission from Elsevier, copyright 2025]. | ||
Metals having large atomic weights and high relative densities that can be hazardous to living things and the environment, even at trace levels, are known as heavy metals. Lead (Pb2+ ions) is one of the most pervasive and harmful environmental contaminants on the globe. One heavy metal with a reputation for harming both the environment and human health is Pb2+ ions.183,184 The industrial use of Pb2+ ions has increased dramatically in recent years, highlighting the critical need for effective measuring techniques. H. Jin and co-workers examined a dual-mode biosensor using bifunctional Ti3C2Tx MXene@PtPd NPs for sensing Pb2+ ions in environmental and food specimens.185 Researchers synthesized Ti3C2Tx MXene@PtPd NPs with luminescence quenching and POD-like activity, participating in nanozyme recognition and magnetic separation for accurate detection of Pb2+ ions. The colorimetric and fluorescence modes exhibited LOD values of 23 pM L−1 and 74 pM L−1, respectively. Real sample analysis confirmed the biosensor's dependability, demonstrating recoveries ranging from 95.4% to 111.1%. The investigation demonstrated the sensor's utility in food safety monitoring by highlighting its reliability, strength, and affordability. This investigation demonstrated the sensor's utility in food safety monitoring by highlighting its reliability, strength, and affordability. The dual-mode approach reduces ambient interferences and increases selectivity, making it a promising advancement in the detection of Pb2+ ions. The detection method depended on catalytic signal amplification and nanozyme recognition. The DNAzyme split its substrate strand in the presence of Pb2+, producing single-stranded DNA that then merged with a luminously tagged sensor to recover fluorescence. Concurrently, the POD-like activity of the Ti3C2Tx MXene@PtPd NPs catalyzed the oxidation of TMB in the colorimetric mode, resulting in a visible color change. Both sensing modes were able to function independently thanks to magnetic isolation, which decreased background interference and improved detection accuracy. Ti3C2Tx MXene played a crucial role in analyte sensing by acting as a fluorescence quencher via Förster resonance energy transfer (FRET) and a nanozyme with POD-like catalytic activity.
Because of its great effectiveness and brief residual time, carbosulfan (CBS) is one type of carbamate pesticide frequently employed in farming to control pests. The safety of food and human health is seriously threatened by CBS residues in fruits, vegetables, and water resulting from abuse or excess.186 Consequently, the protection of the environment and human health greatly depends on the precise and reliable identification of CBS. Z. Lei reported a specific and sensitive naked-eye visual approach for sensing CBS based on its hydrolysis behavior and the POD-like activity of Ti3C2 MXene/AuPt nanozymes.187 Because it breaks down into poisonous carbofuran, CBS, a common carbamate insecticide, offers a serious health concern. Rapid detection is difficult with traditional detection methods because they need costly equipment and intricate sample preparation. Using a one-step self-reduction method, this strategy produced Ti3C2 MXene/AuPt nanozymes with increased catalytic activity. In an acidic environment, CBS hydrolyzed and generated thiol linkages that coupled with Pt atoms to prevent Ti3C2 MXene/AuPt nanozymes from catalyzing (Fig. 8c). Using a technique based on color that tracked the oxidation of OPD, this inhibition was used to detect CBS. This technique proved a wide linear range (0.5 ng mL−1 to 5 μg mL−1), a LOD of 0.342 nM, and brilliant discrimination against interfering constituents. Real lake water samples were used to confirm the technique's viability, and the results showed reasonable recoveries. In contrast to traditional assays that rely on acetylcholinesterase (AChE), the advantage of the AChE-free method is enhanced specificity, decreased expenses, and streamlined detection. This study offered a new approach for creating nanozyme-based colorimetric sensors for various toxicants and demonstrated the potential of Ti3C2 MXene/AuPt nanozymes for environmental utility and pesticide residue analysis.
Cyanobacteria create a class of secondary metabolites called microcystins (MC-LR), which have cyclic heptapeptide sequences and can seriously impair both human and animal health.188 Consequently, the WHO established that 1 μg L−1 is the maximum permitted tolerable limit of MC-LR in drinking water.189 Thus, the development of trustworthy techniques for the sensitive and precise detection of MC-LR is imperative. Z. Lei's research group reported the POD-like activity of Ti3C2 MXene nanosheets (Ti3C2 NSs) and their utility in an aptasensor for sensing MC-LR, a toxic cyanobacterial metabolite.190 Through a variety of tests, the catalytic mechanism of Ti3C2 NSs was investigated, and it was discovered that the production of reactive species, such as holes and superoxide anion radicals, and electron transfer accelerated by nanozymes controlled their activity. Kinetic investigations were used to extensively describe the POD-like activity, revealing Michaelis–Menten kinetics. According to the study, ssDNA prevented substrates from reaching the nanozyme surface, thereby inhibiting the POD activity of Ti3C2 NSs. Utilizing this property, a label-free colorimetric aptasensor was designed for MC-LR detection, engaging an MC-LR-specific aptamer that adsorbed onto Ti3C2 NSs, suppressing their catalytic activity. The aptamer–nanozyme interaction caused a detectable drop in the OPD colorimetric signal upon binding to MC-LR, making quantification possible. The sensor exhibited a broad linear detection range (0.01–60 ng mL−1), a LOD value (6.5 pg mL−1), and high selectivity against potential interferents. Real water samples that had been tampered with were utilized for validating their practical use, and recoveries ranging from 97.2% to 102.1% were obtained.
Serum overexpression of thrombin (TB) is a crucial biomarker linked to many illnesses with high rates of morbidity and death.191 Current TB detection methods are laborious, necessitate complex equipment, and involve lengthy sample preparation processes, all of which prolong detection times and raise procedure costs.192 Clinical interventions begin with an early and easily accessible diagnosis at the point of care, particularly in nations with limited resources. Y. Guo and colleagues193 synthesized Ti3C2 MXene nanosheets (Ti3C2 NSs) with intrinsic POD-like activity and used them for colorimetric, label-free protein (TB) detection. Researchers discovered that Ti3C2 NSs could catalyze oxidation events by imitating POD enzymes and that the adsorption of single-stranded DNA (ssDNA) greatly increased their catalytic activity. This development was attributed to improved substrate (OPD) affinity and electrostatic interactions between ssDNA and the Ti3C2 NSs. Based on these results, this team created a biosensing method for identifying biomolecules. Aptamer-modified Ti3C2 NSs saw a decrease in catalytic activity following thrombin binding, enabling TB measurement in a colorimetric biosensor built using TB as an analyte. In real blood samples, the sensor performed satisfactorily and showed a linear detection range of 10 pM to 10 nM. From outcomes, the authors confirmed that Ti3C2 NSs are stable, reasonably priced, and highly sensitive nanozymes for biosensing applications. This study demonstrated how Ti3C2-based biosensors can integrate particular aptamers to detect a variety of biomolecules. Ti3C2/V2C MXene acted as a significant character as a nanozyme with POD-like action, aiding fluorescence and naked-eye visual approach-based detection approaches. For CBS quantification, Ti3C2 MXene/AuPt nanozymes catalytically oxidized OPD, with CBS hydrolysis impeding the chemical response, leading to a computable color variation. In AFB1 determination, Ti3C2/V2C MXene played as both a luminescence quencher and a POD mimic, allowing multimodal detection through aptamer interactions. An aptamer adsorbed onto the surface of Ti3C2 hindered its catalytic activity for MC-LR recognition, and target binding further reduced the signal. Across all fluorescence sensing studies, the aptamer-binding ability, high surface area, and catalytic activity of MXene allowed for the sensitive, economical, and selective detection of harmful substances in food and environmental samples. Table 3 summarizes various MXene-conjugated nanostructured materials used in nanozyme-based optical sensing, particularly for detecting important or biologically/chemically relevant analytes. Furthermore, Table 3 highlights the nanozyme activity, target analyte, LOD, linear range, and MXene role in this sensing approach, providing valuable insights for future research and development.
| Material | Nanozyme activity/substrate | Role of MXene | Analyte | Linear range | LOD | Ref. |
|---|---|---|---|---|---|---|
| DNA-Ti3C2Tx MXene/Pt | POD/TMB | Catalytic booster | Hg2+ | 5–50 nM | 9 nM | 102 |
| Ti3C2Tx MXene/AuNPs | POD/TMB | Reducing agent and catalyst | Hg2+ | 0–2.0 μM | 0.054 nM | 103 |
| Fe3O4/Ti3C2 QDs | POD/TMB | Catalytic booster | Cr6+ | 0–60 μM | 0.26 μM | 104 |
| Mo NCs/N–Ti3C2Tx MXene | POD/TMB | Catalytic supporter | GSH | 1–150 μM | 0.29 μM | 106 |
| Ti3C2Tx MXene/carbon dots | POD/TMB | Catalytic booster | GSH | 0.3–20 μM | 0.12 μM | 107 |
| Ti3C2Tx@Fe3O4 nanocomposites | POD/TMB | Catalytic booster | GSH | 0.5–10 μM | 0.5 μM | 108 |
| Ti3C2/Pt hybrids | POD/TMB | Synergistic catalyst | GSH | 0.4–7 μM | 8.9 nM | 109 |
| Au/Pt/Ti3C2Cl2 nanocomposite | POD/TMB | Structural and catalytic base | GSH | 50–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μM and 0.1–20 μM 000 μM and 0.1–20 μM |
10.24 μM and 0.07 μM | 110 |
| Ti3C2Tx MXene/AgNPs | POD/OPD | Catalytic supporter | GSH | 10 nM–50 μM | 7 nM | 111 |
| Ti3C2Tx MXene/AgNPs | POD/OPD | Catalytic supporter | Cys | 50 nM–50 μM | 48.5 nM | 111 |
| Ti3C2Tx MXene/AgNPs | POD/OPD | Catalytic supporter | HCy | 10 nM–250 μM | 5.5 nM | 111 |
| Ti3C2Tx MXene/AuNPs | POD/TMB | Reducing agent and catalyst | Cys | 0–100 μM | 0.03 μM | 103 |
| V2C MXene | OD/TMB | Catalyst | Cys | 0.1–15 μM | 30 nM | 112 |
| Au@Pt/Nb2C–MXene nanocomposite | POD/TMB | Conductive materials | H2O2 | 0.3–2.75 M | — | 113 |
| GSH-Ti3C2Tx MXene QDs | OD/OPD | Nanoprobe | Uric acid | 1.2–75 μM | 125 nM | 115 |
| N, S, doped-Ti3C2Tx MXene sheets | POD/TMB | Active nanozyme | Uric acid | 1–400 μM | 0.19 μM | 116 |
| CuNPs/N-doped Ti3C2Tx hybrids | POD/TMB | Catalytic booster | Glucose | 5–300 μM | 5 μM | 119 |
| Ti3C2/Pt hybrids | POD/TMB | Synergistic catalyst | Glucose | 0.04–0.5 μM | 0.85 μM | 109 |
| MXene–Ti3C2/Co nanosheets | POD/TMB | Sensing matrix and catalyst support | Glucose | 0.01–0.1 mM | 0.001 mM | 120 |
| Ti3C2Tx MXene/Ag | POD/TMB | Catalytic supporter | Cholesterol | 2–800 μM | 0.6 μM | 122 |
| MXene–Ti3C2/CuS | POD/TMB | Catalytic supporter | Cholesterol | 10–100 μM | 1.9 μM | 123 |
| Ti3C2Tx/AuNPs heterostructure | Catalase/TMB | Catalytic supporter | Penicillin G | 35.05–584 nM | 7.51 nM | 125 |
| Ti3C2Tx MXene/AuNPs | POD/TMB | Catalytic supporter | Ampicillin | 0.005–0.5 μg mL−1 | 1.1 ng mL−1 | 126 |
| Oxygen terminated Ti3C2Tx sheets | POD/TMB | Catalyst | Kanamycin | 15.28 nM–46.14 μM | 15.28 nM | 127 |
| Prussian blue–Ti3C2Tx hybrid | POD/TMB | Catalyst surface | Malathion | 1 fM–1 nM | 1 fM | 132 |
| Dendritic-like Ti3C2 QDs@CuNi | POD/TMB | Catalytic enhancer | Glyphosphate | 0–100 μM | 1.13 μM | 133 |
| Ti3C2Tx nanoribbons@AuPt | POD/TMB | Catalytic supporter | Dipterex | 1 ng mL−1–1 μg mL−1 | 0.479 ng mL−1 | 134 |
| Cu NPs@N–Ti3C2Tx nanozyme | POD/TMB | Electron mediator | Astaxanthin | 0.01–50 μM | 0.015 μM | 135 |
| CuNC-decorated Ti3C2Tx MXene nanosheets | Tetra enzyme activity/TMB | Catalytic supporter | Hypoxanthine | 5–200 μM | 0.25 μM | 145 |
| Co(OH)2/Ti3C2Tx MXene nanocomposite | Multi nanozyme activity/TMB | Catalytic supporter | Hypoxanthine | 5–250 μM | 0.2 μM | 146 |
| Fe3O4@Ti3C2Tx–Au nanocomposite | OD/TMB | Composite formation supporter | Glucose | 0–1.4 mM | 0.11 mM | 147 |
| Nb2CTx@PdPt nanozyme | POD/TMB | Reducing agent and catalyst supporter | Glyphosphate | 1 ng mL−1–1 μg mL−1 | 0.419 ng mL−1 | 149 |
| Ti3C2Tx MXene/PtNPs | POD/TMB | Catalytic carrier | Chloramphenicol | 0.0125–1.0 μg kg−1 | — | 150 |
| Au/Cu/Ti3C2 nanosheets | OD/OPD | Detection platform | Hg2+ | 8.0–200.0 nM | 0.8 nM | 152 |
| Au/Cu/Ti3C2 nanosheets | OD/OPD | Detection platform | Cl− | 5.0–350.0 μM | 27 nM | 152 |
| DNA-encoded porous Ti3C2 nanozyme | POD/TMB | Porous nature | Okadaic acid | 10–1000 ng mL−1 | 0.38 ng mL−1 | 155 |
| CuNPs/N–Ti3C2Tx nanocomposite | OD/OPD | Fluorescence enhancer | Ascorbic acid | 5–150 μM | 0.437 μM | 176 |
| DNAzyme-ferrocene-modified Ti3C2Tx MXene/Au nano bipyramid | OD/OPD | Signal mediator | Food pathogen | 10–108 CFU mL−1 | 6 CFU mL−1 | 178 |
| Ti3C2Tx MXene labelled with ssDNA-FAM | Multimode/TMB | Nanozyme support | Aflatoxin B1 | 0.10–400 ng mL−1 | 0.61 ng mL−1 | 181 |
| ssDNA-FAM labelled V2C MXene | POD/TMB | Signal amplifier | Aflatoxin B1 | 0.1–500 ng mL−1 | 0.0477 ng mL−1 | 182 |
| Ti3C2Tx MXene@PtPd NPs | POD/TMB | Fluorescence quencher | Pb2+ ions | 0.5–1000 nM | 74 pM | 185 |
| Ti3C2 MXene/AuPt nanozymes | OD/OPD | Catalytic substrate | Carbosulfan | 0.5–5000 ng mL−1 | 0.342 nM | 187 |
| Ti3C2 MXene nanosheets | POD/OPD | Catalytic activity modulator | Microcystins | 0.01–60 ng mL−1 | 6.5 pg mL−1 | 190 |
| Ti3C2 MXene nanosheets | POD/OPD | Intrinsic catalyst | Thrombin | 10–1000 pM | 10 pM | 193 |
5.4. MXzyme-based chemiluminescence sensors
Chemiluminescence (CL) is a phenomenon that occurs when a species' excited state electronically emits light during a chemical reaction process. In simple terms, when more than one chemical is combined and reacts, some of them, like reactants or intermediates, can be triggered by oxidation to generate a high-energy intermediate that can break down or transfer its energy to fluorescence probes before returning to the ground states and emitting light spontaneously.194–197 In contrast to luminescence, which is instigated via the absorption of light (photons), CL does not necessitate an external light source excitation, discontinuing interference from light scattering and background emission from the sample matrix.194 Additionally, its optical signal interpretation offers a good signal-to-noise ratio, and its ability to identify high-impact analytes is high. The CL test works by launching a computable association between the CL readout signal and the analyte concentration.198 This technique also incorporates a biomolecular recognition-based investigation that employs substantially biologically active materials, including DNA, enzymes, antibodies, or antigens, as recognition elements. CL systems act as indication transducers, qualifying the alteration of specific biological reaction identifications into quantifiable CL signals to detect the concentration of target biomolecules.195,197 Thus, the construction of CL signals is crucial for the accurate and sensitive recognition of a particular analyte.In the history of CL, initially, in 1877, a chemical reaction involving lophene and H2O2 in an alkaline medium produced luminescence that was utilized for army soldiers' lights.199 An essential factor in the development of CL as a useful tool in analytical chemistry was the synthesis of the CL reagent 3-aminophenylhydrazine (luminol) in 1902 and the discovery of its CL behavior in alkaline environments in 1928.200 In 1935, the reaction between lucigenin substance (N,N-dimethyldiacridinium nitrate) and H2O2 revealed the lucigenin CL behavior.201 However, the aforementioned CL signal is relatively weak and transient, and early CL research has advanced slowly, with few significant applications. CL signal pathways can principally be categorized into two categories: direct and indirect CL signals. In a direct CL signal, a CL compound undergoes oxidation, forming a high-energy chemiexcited intermediate. These intermediates emit light as they change to their ground state. Examples of direct CL systems include luminol and luciferin, which are readily oxidized by H2O2 or singlet oxygen to produce luminescence. In another type, energy is transferred from a chemiexcited intermediate to adjacent acceptor molecules, like fluorophores or photosensitizers, in the indirect CL signaling process. A neighboring acceptor molecule is excited to a higher energy state during this procedure by the chemiexcited intermediate, transferring its energy to it (Fig. 9a). As it returns to its ground state, the excited fluorescent molecule releases energy in the form of light. This kind of energy transfer is known as CL resonance energy transfer (CRET) (Fig. 9a).202,203
 | ||
| Fig. 9 (a) Schematic illustration of the formation of the CL mechanism. MXzyme-based CL sensors for important analytes. (b) Glassy carbon (GC) electrode modified with a 2D Ti3C2Tx MXene–Nafion composite material [reproduced from ref. 207 with permission from Elsevier, copyright 2023], (c) PB/Ti3C2Tx MXene nanocomposite-based CL detection of UA [reproduced from ref. 208 with permission from Elsevier, copyright 2024], and (d) Ag/MXene nanozymes for efficient CL sensing of UA and GSH [reproduced from ref. 211 with permission from Royal Society of Chemistry, copyright 2024]. | ||
Nanoparticles used in CL amplification systems have received immediate attention since the advent of nanozymes,204,205 which are nanomaterials having enzyme-like activity. This is because of their advantages, which include being more stable, easily obtained, inexpensive, and controlled. By utilizing the distinct features of different nano-structured materials, materials with nano sizes can be deployed as high catalyst levels for the measurement of CL signals, improving sensitivity and lowering detection limits.206 This makes it possible to use these novel nano-sensing platforms to create a high-sensitivity CL system for biological analysis. To date, the MXzyme-based CL assay has been getting more attention and utilized for the detection of potential analytes. Won-Yong Lee's research group207 examined a new electrode strategy for attractive luminol-based electrogenerated CL (Fig. 9b). The researchers developed a glassy carbon (GC) electrode modified with a 2D Ti3C2Tx MXene–Nafion composite material. A crucial development for biological and physiological applications, this Ti3C2Tx MXene–Nafion composite material showed strong electrocatalytic activity toward H2O2 oxidation and O2 reduction processes, leading to markedly enhanced cathodic CL emissions in neutral solutions. This study explored the CL-based sensing of H2O2, obtaining a detection limit of 3.1 nM and a linear range from 10 nM to 1 mM. The bare and Ti3C2Tx MXene–Nafion composite material-modified GC electrodes were outstripped by this electrocatalytic performance. Efficient H2O2 quantification in human serum samples, with recovery rates ranging from 98.8 to 104.7%, confirmed the practical applicability. Tests of long-term stability showed that the signal intensity was retained for three weeks at 75.3%. The results, as mentioned above, highlight the potential of the Ti3C2Tx MXene–Nafion composite material in the development of stable and sensitive biosensors for environmental and clinical utility.
Xiangjuan Zheng and co-workers reported a novel approach to biosensing through the synthesis of Prussian blue/Ti-based MXene (PB/Ti3C2Tx MXene) nanocomposites.208 The investigators employed the oxidase-like features of the PB/Ti3C2Tx MXene nanocomposite to remove the necessity for H2O2, a common oxidant in conservative CL systems (Fig. 9c). The PB/Ti3C2Tx MXene nanocomposite, acting as both a reducing agent and nanocarrier, significantly raises the CL intensity when mixed with luminol. This allowed for the sensitive detection of uric acid (UA) within a linear range of 0.2–30 μM and a detection limit of 0.19 μM. The sensor exhibited excellent specificity, reliability, and stability, successfully detecting UA in diluted human serum with recoveries ranging from 98.59% to 104.04%. This work followed an oxidase nanozyme-based pathway to produce ROS by triggering dissolved oxygen, which afterward oxidizes the substrate and finishes the whole reaction. The PB/Ti3C2Tx MXene nanocomposite catalyzed the reaction, electrocatalytically generating ROS from dissolved oxygen and reacting with just luminol to generate a powerful CL signal without needing H2O2 (Fig. 9c). Furthermore, when the concentrations of UA increase in the PB/Ti3C2Tx MXene nanocomposite–luminol combination, the added UA can react with ROS. Its CL signal drops when the ROS is used up, allowing for the successful detection of UA. The study highlighted the potential of PB/Ti3C2Tx MXene nanocomposite-based materials in the biosensing of UA, offering a greener and more efficient alternative for clinical diagnostics. This work marked a significant advancement in the development of sensitive, cost-effective, and eco-friendly biosensors.
The human body contains two common biomolecules, GSH and UA, which, depending on their levels, are essential for physiological function.209,210 Due to the biological potential of both biomolecules, early recognition of UA and GSH levels in biological fluids is crucial for public health. By integrating silver nanoparticles (AgNPs) with Ti3C2 MXene nanosheets, Xiluan Yan's research group reported an invention of Ag/MXene nanozymes for efficient CL sensing.211 The Ag/MXene composites, which were created via an in situ self-assembly technique, demonstrated potent POD-like activity by accelerating the production of ROS from H2O2. The synergistic actions of AgNPs and Ti3C2 MXene greatly increased the CL intensity, allowing for the very sensitive quantification of UA and GSH (Fig. 9d). These biosensors' linear detection ranges for GSH and UA were 50 nM–20 μM and 1 mM–35 μM, respectively, with LOD as low as 0.83 nM and 0.37 μM. The production of ROS, such as hydroxyl radicals and singlet oxygen, as important reaction intermediates, was validated by mechanistic investigations. Additionally, the sensor demonstrated exceptional stability and selectivity, successfully identifying GSH and UA in human blood serum samples. This work demonstrated the prospective use of MXzyme composites in CL applications and offered a strong biosensing platform.
5.5. MXzyme-based electrochemiluminescence sensors
In recent years, electrochemiluminescence (ECL) sensing devices have become increasingly popular, leading to notable advancements in chemo and biosensors. The utility of advanced signal amplification approaches is crucial to the progress of high-throughput ECL sensors, which will usher in an entirely new phase of sensitive analysis. ECL is the term for light emitted by excited luminophores as a result of electrochemical reactions occurring at an electrode's surface.212,213 ECL has become an eminent signal transduction system utilized broadly in various eras thanks to its distinct merits, which embrace the lack of a requirement for external light sources, exact control over the spatiotemporal nature of light, low background interference, and exceptional sensitivity.212–215 Target monitoring of potential analytes has become a significant use for ECL sensors.Research on ECL has been ongoing since the mid-1960s, when Kuwana, Hercules, Bard, and others began the first investigations on the subject. As a result, ECL (bio)sensors are now a potent analytical tool in the modern world.216–218 Next, higher detection sensitivity in ECL sensing platforms resulted from the introduction of MXene into the ECL sensing field in 2018.219 Because of its resistance to excitation light sources and dispersed light, ECL has attracted a lot of interest in clinical diagnostics. Since [Ru(bpy)3]2+ has outstanding ECL efficiency, exceptional biocompatibility, and chemical stability, it is commonly utilized in ECL-based sensing systems.220–222 Nevertheless, [Ru(bpy)3]2+ has a high-water solubility, which limits the ECL property and makes immobilization on the electrode surface difficult. Many nanocarriers have been researched to address this problem and increase the stability and [Ru(bpy)3]2+efficacy.222,223 However, because of their ability to efficiently catalyze the production of hydroxyl radicals, POD mimetic nanozymes have garnered much study interest.212,224 Under certain reaction settings, it has been established that different metal nanoparticles (MNPs), including Pt, Pd, Ag, and AuNP, may imitate a broad range of enzymes, including CAT, POD, and OD.224–226 However, the combined activity of two or more MNP components within the same nanozyme makes the hybrid nanosystem-based nanozyme a clear advantage for biosensing applications.227 More significantly, as compared to biological enzymes, nanozymes frequently have better catalytic activity. They work better because of their reduced size, which increases the surface area accessible to enzymatic action. They work better because of their reduced size, which increases the surface area accessible to enzymatic action.
Later, a variety of ECL sensors for bioanalysis were created using MXenes and MXene-based composite materials. The catalytic activity of nanozymes, which resembles an enzyme, is combined with the sensitive light-emitting mechanism of ECL to create nanozyme-based ECL.218,228 Nanozymes expand electron transport, raise fluorescence efficiency, and catalyze the construction of ROS to progress signal amplification. MXzymes' ECL combines ECL technology with the remarkable catalytic capabilities of MXene-derived nanozymes. Because of their high conductivity and adjustable surface chemistry, MXenes catalyze the production of reactive species and facilitate electron transport, which improves the amplification of ECL signals.218,228,229 This cutting-edge tactic is commonly applied for super-specific and stable quantification in environmental monitoring and biosensors.
A new dual-mode electrochemical (EC) and electrochemiluminescence (ECL) biosensor was developed by Bin Wang et al. for the ultrasensitive and real-time detection of H2O2, a crucial cancer biomarker.230 Polycrystalline manganese oxide nanoflowers (MnONFs) were self-assembled on delaminated Ti3C2 MXene nanosheet surfaces, leveraging their collaborative interface to improve catalytic efficiency, electron transfer, and stability. The hollow layered structure of Ti3C2 MXene offered a high surface area and a conductive pathway, while MnONFs supplied abundant active sites for H2O2 oxidation. This composite Ti3C2 MXene@MNOFs allowed rapid H2O2 determination, attaining an EC linear range of 0.05–650 μM and an ultrasensitive ECL limit of detection of 0.45 nM. This fabricated sensor demonstrated exceptional specificity and anti-interference capabilities in complex biological environments and effectively differentiated cancer cells from normal cells by detecting H2O2 effluxes induced by ascorbic acid stimulation. It successfully monitored as few as 20 cancer cells in 1 mL, exhibiting remarkable sensitivity, reproducibility, and stability over extended periods. The integration of EC and ECL readouts minimized false positives and negatives, improving diagnostic accuracy. The study highlighted the potential of this dual-mode sensing platform for early cancer detection, offering a reliable, non-enzymatic alternative for real-time monitoring of ROS. These advancements addressed the limitations of traditional methods, including single-readout systems and complex preparation processes, paving the way for broader applications in biomedical diagnostics and therapy.
One of the most common cancers in men is prostate cancer, and the sensitive marker for detecting prostate cancer is prostate-specific antigen (PSA). To provide a precise and sensitive detection technique for PSA ultra-sensitivity detection, Chuanping Li and colleagues discovered the fabrication of a highly efficient ZIF-67@Ti3C2 MXene-based ECL biosensor.231 Here, researchers established the use of ZIF-67@Ti3C2 MXene as a co-reaction accelerator and improved ECL efficacy by up to 830% via the specific production of hydroxyl radicals and singlet oxygen. The biosensor effectively detected PSA with an improved detection limit of 0.91 pg mL−1, leveraging a plasmonic NH2-MIL-88@Pd nanozyme for signal amplification. This study emphasized the incorporation of MXene's conductivity and ZIF-67's stability, offering a super-specific sensing platform for cancer biomarker detection, stability, and excellent sensitivity.
To the best of our knowledge, non-small cell lung cancer232 is the primary cause of lung cancer-related mortality globally. A tumor marker linked to lung cancer, cytokeratin-19 fragment (CYFRA21-1),233 is primarily detected in the cytoplasm of tumor cells originating from epithelial cells, including lung and esophageal cancer. In the normal population, it has a reference value of 0–3.3 ng mL−1. Thus, there is some clinical utility for CYFRA21-1 in the treatment, identification, and effectiveness assessment of different kinds of lung cancer. Xian Ren et al. demonstrated the development of an ECL immunosensor for CYFRA 21-1 recognition, employing Ti3C2Tx MXene@TiO2–MoS2 hybrids as co-reactive catalysts and luminol@Au@Ni–Co nanocages as probes.234 These state-of-the-art nanozyme hybrid materials helped efficient H2O2 putrefaction and boosted ECL signal production. The developed sensor established a linear range of 0.1 pg mL−1 to 100 ng mL−1, with a LOD of 0.046 pg mL−1, displaying outstanding sensitivity. The incorporation of Ti3C2Tx MXenes's high conductivity and MoS2's catalytic features aided stable, repeatable, and specific CYFRA 21-1 identification analysis, even in complex biological matrices. The strategy offered a strong foundation for the early identification of lung cancer (Fig. 10a). Additionally, this strategy offered a strong foundation for the early identification of lung cancer.
 | ||
| Fig. 10 MXene-supported material-based ECL sensors for the different analytes. (a) Ti3C2Tx MXene@TiO2–MoS2 hybrids as co-reactive catalysts and luminol@Au@Ni–Co nanocages as an ECL probe for the detection of CYFRA 21-1 [reproduced from ref. 234 with permission from American Chemical Society, copyright 2024], (b) Ti3C2-based nanozyme for ECL glucose biosensing [reproduced from ref. 235 with permission from Elsevier, copyright 2024], (c) CoS2/sulfur-doped Ti3C2-based nanozyme for the detection of CAP [reproduced from ref. 239 with permission from Elsevier, copyright 2023], and (d) synthesis and nanozyme activity of Nb2CTx-stabilized Au–Ag alloy NPs integrated into ZIF-8 frameworks for the detection of biomarkers [reproduced from ref. 224 with permission from Elsevier, copyright 2025]. | ||
In one of the more recent articles, researchers explored Ti3C2-based MXzymes for enhancing ECL glucose biosensing, employing bromophenol blue (BPB) as a co-reaction accelerator and transporter.235 Delaminated Ti3C2-based MXzymes with BPB enabled high loading of luminol and glucose oxidase (GOx), decomposing H2O2 to produce hydroxyl radicals, which discriminate ECL signals meaningfully (Fig. 10b). The device demonstrated outstanding rigidity, specificity, and consistency in human serum, with a detection range of 0.1 to 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μM and a LOD of 0.02 μM. Superior sensitivity was attained by minimizing radical breakdown through the proximity of the hydroxyl radicals and the luminophore's upshots. Furthermore, this research effort opened a new door for more sophisticated biosensor designs in clinical diagnostics by demonstrating a potential glucose detection platform.
000 μM and a LOD of 0.02 μM. Superior sensitivity was attained by minimizing radical breakdown through the proximity of the hydroxyl radicals and the luminophore's upshots. Furthermore, this research effort opened a new door for more sophisticated biosensor designs in clinical diagnostics by demonstrating a potential glucose detection platform.
The broad-spectrum antibiotic chloramphenicol (CAP) has been utilized extensively in the farming of animals because of its good antibacterial action and affordable cost.236 However, CAP residues in the environment would make their way into the human body via the food chain and cause major negative reactions and side effects, for example, aplastic anemia, gastric intolerance, and marrow suppression.237,238 Thus, it is essential to create a straightforward, affordable, highly sensitive, and selective CAP monitoring approach. To detect CAP in real water samples, Zilian Zhang et al. discovered a novel ECL aptasensor for detecting CAP, utilizing a luminol-dissolved oxygen (DO) system enhanced with cobalt sulfide (CoS2)/sulfur-doped Ti3C2-based MXzymes and micro-nano bubbles (MNBs).239 The CoS2/sulfur-doped Ti3C2-based MXzymes acted as a co-reaction accelerator, significantly improving the reduction efficacy of DO and generating ROS for enhanced ECL performance (Fig. 10c). The ECL signal was further amplified by the addition of MNBs, which raised the DO concentration and ROS generation. Notably, this tackle confirmed an 8.7-fold perfection in ECL signal intensity in contrast to orthodox luminol-DO systems, with a sensing range from 0.1 fM to 10 pM and an ultra-LOD of 105 fM. The aptasensor demonstrated outstanding selectivity, stability, and repeatability, and it was successfully used to analyze real-world samples such as milk, tap water, and pond water. However, this sensing protocol demonstrated the potential of CoS2/sulfur-doped Ti3C2-based MXzymes and MNBs to alter ECL sensing by eliminating the need for H2O2, opening the door for more practical, biocompatible, and sensitive biosensors in the monitoring of food and environmental safety.
A rather uncommon and varied class of malignancies, neuroendocrine tumors (NETs) develop from specialized cells dispersed throughout the body. Despite being somewhat uncommon, they can be difficult to diagnose and treat properly.240 The most adopted biological indicator in medical studies for NET identification is chromogranin A (CgA). Because of this, it is an asset for enhancing the knowledge and diagnosis of these intricate cancers.241 To detect CgA in human serum and saliva, F. Kareem and his colleagues described a dual-mode immunosensor that practices Nb2CTx-stabilized Au–Ag bimetallic nanoparticles (BiMNPs) integrated into ZIF-8 frameworks.13 This first study uses Nb-based MXzymes rather than Ti-based MXzymes in this part. Moreover, Nb2CTx has better electrochemical stability than Ti3C2Tx, which results in enhanced electrochemical sensing capabilities.242 Nb2CTx MXene acted as a reducing and stabilizing agent for synthesizing BiMNPs, which demonstrated excellent POD-like activity and enhanced ECL performance in the Ru(bpy)32+/TPrA system.224 The ECL signal was greatly enhanced, and colorimetric detection by TMB oxidation was made possible by the ZIF-8 framework's wide surface area and ease of biomolecule immobilization (Fig. 10d). The immunosensor exhibited a wide linear detection range (0.001–1000 pg mL−1 for ECL, 0.1–4000 ng mL−1 for colorimetry) and ultra-low detection limits (0.11 fg mL−1 for ECL, 100 pg mL−1 for colorimetry). Its remarkable recovery rates from the analysis of real-world human serum and saliva samples confirmed its great selectivity, repeatability, and stability. The work demonstrated the Nb2CTx MXene-stabilized BiMNPs@ZIF-8 nanohybrid's capacity for precise and sensitive biomarker identification, providing a viable platform for the diagnosis of neuroendocrine tumors and the monitoring of psychophysical stress (Fig. 10d). In all the systems, the catalytic generation of ROS, like hydroxyl radicals, is used to enhance ECL signals. The 2D stacked MXenes' thin nanosheets feature a lot of surface functional groups and surface area. This feature makes them extremely versatile for the quantification of a wide range of biomolecules by allowing conductivity tweaks and customized alterations aimed at biomolecules.243 When combined with other nanostructured materials, MXene's unique capabilities work in concert with the unique qualities of the combined nanomaterials to produce often better performance than when the components are used alone.217 The incorporation of MXene's high conductivity and nanostructured materials' catalytic features/electron transporters highlights the importance of catalytic efficiency and ROS generation in enhancing the detection process. These species played a pivotal role in CL or ECL reactions, amplifying the signal intensity for sensitive analyte detection.
5.6. MXzyme-based electrochemical sensors
Research on electrochemical sensors has been one of the most investigated analytical topics. Electrochemical sensors' ability to recognize specific target analytes of attention hangs deeply on the electrode's materials and/or receptors.244,245 Most potential target analytes interacting with electrode-deposited materials and/or electrode receptors in electrochemical sensors can produce a response that is possibly deciphered into computable electrochemical signals that designate the analyte dosage level. The quick reaction time, combinatorial features, and potential of portable (even wearable) measurement devices are some of the benefits of electrochemical sensors over alternative sensing techniques.246,247 They can also be made smaller and more affordable. By using biomarker-specific reactions, electrochemical biosensors use the principles of electrochemistry to convert biological processes into quantifiable electrical signals.248,249 The targeted potentially specific biomarkers (e.g., cancer biomarkers) might be detected precisely and quantitatively thanks to this electrochemical signal, which would be proportionate to their concentration.249 Lightning-fast, immediate fashion, accurate, long-lasting, and inexpensive high-impact analyte detection is made possible by the incorporation of nanozymes into electrochemical sensors. When compared to natural enzymes, nanozymes offer several advantages, such as increased robustness and durability, cost-effectiveness, adaptable catalytic characteristics, and strong electrical conductivity.73,250 Generally speaking, there are two approaches to disseminating the mechanisms of action of electrochemical sensors based on nanozymes. The first uses catalytic reactions mediated by nanozymes to generate or amplify electrochemical signals, while the second uses catalysis to produce or activate electroactive chemicals that are relevant to the target and employed as electrochemical signals.250 Examples of electrochemical methods for biomarker detection include amperometry, electrochemiluminescence (ECL), photoelectrochemistry (PEC), impedometry (electrochemical impedance spectroscopy; EIS), and various voltammetry methods, such as square wave voltammetry (SWV), linear sweep voltammetry (LSV), differential pulse voltammetry (DPV), and stripping voltammetry (SV). In general, these electrochemical sensing techniques operate as follows:(i) An antibody that resembles a tracer receptor is labeled with an electroactive species, like nanozymes.
(ii) The receptor becomes entrenched on the electrode's surface after binding with the analyte.
(iii) Applying a voltage and assessing the current, resistance, and luminescence that is induced at the electrode allows one to determine the amount of the desired analytes.
In essence, the labeled electroactive species undergo a redox reaction when the voltage is applied, producing an electrical signal that represents the amount of the receptor-bound analytes.251 Furthermore, the coating of nanostructured materials enhanced the electrochemical catalytic performance. Because of their versatility, MXenes can be effectively customized by integrating metal, metal oxide, or carbon-based nanoparticles, which makes it possible to rationally build effective MXene composites. MXenes were often designed by carefully choosing the X and M components, as well as surface terminal groups, to improve their physio-chemical performances and attain desired properties. Given that MXenes excellently trap enzymes on their surface, enabling efficient electrical communication between the electrode and the protein backbone of enzymes, they were initially utilized as an immobilization matrix of natural enzymes. Thus, MXene has been identified as a substrate that facilitates rapid electron transfer, resulting in enhanced electron kinetics and greater susceptibility of electrochemical sensors based on enzymes.250,252 It was recently found that MXenes can mimic the catalytic activity of natural enzymes such as CAT, SOD, OD, and POD, usually by effectively relating with other catalytic nanostructured materials.36,38 Based on this, the mimic features of MXene-supported composite materials have been applied in electrochemical sensors for the detection of potential analytes.
 | ||
| Fig. 11 MXzymes for electrochemical sensors for hazardous analytes. (a) Ti3C2Tx MXene nanoribbons in conjunction with Au nanozyme activity-based DPV detection of Hg2+ ions [reproduced from ref. 255 with permission from Elsevier, copyright 2023], (b) a self-assembled phosphorene/Ti3C2Tx MXene nanohybrid electrode based on nanozyme applied in the multimodal detection of NAA in agricultural/food samples [reproduced from ref. 265 with permission from Elsevier, copyright 2021], and (c) cobalt-doped 2D Ti3C2Tx MXene nanosheets with POD-like characteristics as a novel homogeneous electrochemical sensing of OPs [reproduced from ref. 269 with permission from American Chemical Society, copyright 2022]. | ||
Ochratoxins (OTs) are thermostable fungal secondary metabolites that damage kidneys, suppress immunity, and have mutagenic and carcinogenic effects. OT contamination can occur before or after harvesting, as well as during food processing, packing, distribution, and storage.258 Among the different OTs, ochratoxin A (OTA) poses a serious risk to both human and animal health.259 For the detection of OTA, a signal-on/off paper-based electrochemical aptasensor that combines Pt@NiCo-layered double hydroxide (LDH) nanozyme for signal amplification with MXene–Au as a sensing substrate was developed.259In situ reduction was used for generating the MXene–Au composite, which offers a robust and highly conductive platform for electrode fabrication. MXene–Au composite-supported Pt@NiCo-LDH nanozyme, possessing POD-like activity, catalyzed the oxidation of OPD in the presence of H2O2, producing a strong electrochemical signal in the “signal on” state. When OTA was introduced, the aptamer selectively attached itself to it, which caused the aptamer–cDNA complex to dissociate and release the Pt@NiCo-LDH nanozyme. This put the sensor in the “signal off” condition and reduced the DPV signal. The aptasensor outperformed traditional OTA detection techniques with a low detection limit of 8.9 fg mL−1 and a broad detection range of 20 fg mL−1 to 100 ng mL−1. This study demonstrated the potential of MXene-based electrochemical aptasensors for quick, accurate, and economical OTA detection, providing a viable point-of-care testing (POCT) instrument for applications including food safety. Based on the POD-like activity of AgNPs, combined N-doped Ti3C2Tx MXene was utilized for the detection of H2O2.260 This work highlighted the challenges associated with AgNPs' agglomeration, which diminished their catalytic activity, and demonstrated that nitrogen doping improved the conductivity and catalytic efficiency of the Ti3C2Tx MXene substrate. The findings of the study highlighted the importance of carrier modification for better nanozyme-based sensors by showing that the combined impacts of nitrogen-doped MXene and AgNPs increased electrochemical sensing performance.
Due to their harmful effects, phenolic compounds (PCs), which are abundant in both the natural environment and human life, represent a serious risk to human health.261 Consequently, the development of robust, efficient, and trustworthy PC detection techniques is required. For example, strong adsorption between the hydroxyl group of PCs and oxygen atoms on the MXene surface allowed the researchers to establish the catalytic properties of Ti3C2Tx MXzymes, proving their effectiveness in the oxidation of phenolic substances.262 Through the integration of MXene with alkaline phosphatase for electrochemical signal improvement, a cascade catalytic amplification technique (1-naphthyl phosphate substrate) was created, allowing for the detection of the BCR/ABL fusion gene, a crucial biomarker for leukemia diagnosis, using an ultrasensitive biosensor. This sensor demonstrated excellent specificity, stability, and reproducibility, with a detection limit of 0.05 fM. The biosensor's clinical application was demonstrated when it successfully identified the fusion gene in human serum. Next, the development of a robust biosensing platform using Ti3C2Tx MXzymes was applied for the ultrasensitive detection of phenol.263 To build a mediator-free electrochemical biosensor, this sensing investigation used MXene as a matrix to immobilize tyrosinase, an enzyme that can catalyze phenol oxidation. The biosensor exhibited a broad linear detection range (0.05–15.5 μM), a low detection limit of 12 nM, and high sensitivity (414.4 mA M−1). This sensor maintained high enzymatic activity throughout time and showed outstanding repeatability, reproducibility, and stability. Overall, these studies underscored the potential of Ti3C2Tx MXene in electrochemical biosensing, organic synthesis, and environmental applications, paving the way for advanced nanozyme-based analytical tools.
A common phyto-regulator (plant growth regulator) made from naphthalene, α-naphthalene acetic acid (NAA), can be found in harvests to increase their protein and fat content. Nevertheless, NAA poses a risk to human health since it can remain in agricultural products by absorbing environmental contaminants; dishonest traders even use it to produce rootless soybean sprouting. Thus, it is essential to create a new, sensitive, quick, easy, and affordable technique to identify the trace level of NAA in agricultural land settings and agricultural commodities utilizing wireless intelligent portable devices for the fortification of nutriment and agro-product protection, as well as ecological contamination monitoring.264 In this regard, a new method employing a portable wireless nanozyme sensor was developed to detect ultra-trace quantities of NAA, a plant growth regulator.265 A self-assembled phosphorene/Ti3C2Tx MXene nanohybrid electrode was designed in this sensing experiment on a flexible laser-induced porous graphene substrate, guaranteeing excellent ambient stability, effective electrochemical, colorimetric, and wireless responsiveness in real samples (Fig. 11b). This sensor achieved a low detection limit of 1.6 nM and a broad linear range (0.02–40 μM) through the use of LSV for NAA detection. Furthermore, a machine learning approach based on artificial neural networks was used for intelligent data interpretation, increasing dynamic range and accuracy. According to the results of the suggested sensing investigation, this highly sensitive, quick, and affordable sensor offered a viable platform for identifying hazardous materials in agriculture.
The main purpose of organophosphorus pesticides (OPs) is to keep insects away from farms. Since the utilization of OPs in many farming processes has increased significantly, it is now crucial to precisely track their concentration levels to safeguard ecosystems and the supply of food. Nonetheless, OPs are categorized as extremely harmful chemical substances by the WHO.266–268 Given their ubiquitous use and toxicity, simple, fast, and highly sensitive procedures must be designed, for example, using cobalt-doped 2D Ti3C2Tx MXene nanosheets with POD-like characteristics as a novel homogeneous electrochemical sensing technique for the detection of organophosphate pesticides (OPs).269 A considerable reduction current is produced by stimulating the oxidation of OPD into an electrically active product, describing exactly how this sensor operates. By blocking acetylcholinesterase (AChE) activity, the system illustrated a distinct reaction to thiol chemicals, enabling selective OP detection, preventing thiol production, and reestablishing the POD-like action of cobalt-doped 2D Ti3C2Tx MXene nanosheets (Fig. 11c). Electrochemical measurements confirmed the sensor's sensitivity, yielding a detection limit of 0.02 ng mL−1 with a broad linear range. This sensing protocol emphasized the potential of cobalt-doped 2D Ti3C2Tx MXene nanosheet-based homogeneous electrochemical sensors for more precise, rapid, and cost-effective pesticide residue monitoring. Based on the same AChE activity, chitosan functionalized Ti3C2Tx MXene nanocomposites were applied for detecting OPs, particularly malathion.21 The researchers established that biosensors exhibited a linear detection range between 10 fM and 10 nM, with a LOD of 3 fM. The combination of chitosan boosted biocompatibility and enzyme stability, while the high conductivity and large surface area of Ti3C2Tx MXene nanosheets enhanced electron transfer. Furthermore, using a similar concept like AChE action, self-reduced Au–Pd bimetallic nanoparticles on ultrathin Ti3C2Tx MXene nanosheets were fabricated for the sensing of paraoxan.270 Effective AChE immobilization on a screen-printed electrode (SPE) was completely attainable by the excellent conductivity, catalytic activity, and stability of the Au–Pd bimetallic nanoparticles. This sensor worked by identifying the paraoxon-induced suppression of AChE activity, which was measured using DPV. Under optimized experimental settings, the DPV sensor unveiled an extensive linear detection range (0.1–1000 μg L−1) and an ultra-LOD of 1.75 ng L−1. The promise of MXene-based nanocomposites as affordable, sensitive, and environmentally benign substrates for pesticide tracking in food and environmental samples was highlighted by these OP sensing investigations, opening up opportunities for more widespread uses in environmental analysis and biosensing.
Gliotoxin (GTX), the most famous example of an epidithiodioxopiperazine mycotoxin, has drawn a lot of attention during the past 20 years. The health of humans and animals is seriously threatened by it, as it has a wide variety of proapoptotic effects on different types of cells in diverse species, including humans, primates, rats, pigs, fish, and insects.274 Consequently, a practical method for sensing GTX must be established to monitor safety and aid in the early recognition of fungal infections, particularly invasive aspergillosis. For instance, a label-free electrochemical biosensor for the specific detection of GTX by DNA nanostructure/Ti3C2Tx MXene nano complexes was developed.275 It explained how phosphate–titanium coordination was used to immobilize tetrahedral DNA nanostructures (TDNs) onto Ti3C2Tx MXene nanosheets, obviating the requirement for expensive DNA alterations. The high conductivity and huge surface area of the Ti3C2Tx MXene nanosheets improved signal amplification and electron transport. This biosensor was successfully fabricated with the help of EIS and CV techniques, which exhibited a linear range from 5 pM to 10 nM, with a LOD of 5 pM. Reliable detection was ensured by the great selectivity of this GTX sensing technology against potential interferents, such as proteins and mycotoxins. Furthermore, this study found that combining MXene and TDNs allowed for a selective, ultrasensitive, and economical method of gliotoxin detection, underscoring its potential for wider uses in clinical diagnostics and mycotoxin monitoring. In addition, a summary of electrochemical MXene conjugated material based potential sensors for important analytes is given in Table 4. MXene-based nanohybrids boost nanozyme sensor performance in chemiluminescence, electrochemiluminescence, electrochemistry, and SERS, offering high sensitivity and a dynamic linear range for target analytes.
| Material | Nanozyme activity/substrate | Role of MXene | Analyte | Linear range | LOD | Ref. |
|---|---|---|---|---|---|---|
| Ti3C2Tx MXene–Nafion composite | OD/Luminol | Catalytic enhancer | H2O2 | 10 nM–1 mM | 3.1 nM | 207 |
| PB/Ti3C2Tx MXene nanocomposite | OD/Luminol | Reducing agent and carrier | Uric acid | 0.2–30 μM | 0.19 μM | 208 |
| Ag/MXene nanozymes | POD/Luminol | Catalytic supporter | GSH | 50 nM–20 μM | 0.83 nM | 211 |
| Ag/MXene nanozymes | POD/Luminol | Catalytic supporter | Uric acid | 1–35 μM | 0.37 μM | 211 |
| Nb2CTx-stabilized Au–Ag bimetallic nanoparticles | POD/Ru(bpy)32+/POD/TMB | Catalytic supporter | Chromogranin A | 0.001–1000 pg mL−1 | 0.11 fg mL−1 | 224 |
| Ti3C2 MXene@MNOFs | POD/Luminol | Signal amplifier | H2O2 from cancer cells | 0.05–650 μM | 0.45 nM | 230 |
| ZIF-67@Ti3C2 MXene | POD/Luminol | Catalytic enhancer | Prostate-specific antigen | — | 0.91 pg mL−1 | 231 |
| Ti3C2Tx MXene@TiO2–MoS2 hybrids | POD/Luminol | Electron mediator | Cytokeratin-19 fragment | 0.1 pg mL−1–100 ng mL−1 | 0.046 pg mL−1 | 234 |
| Bromophenol blue with delaminated Ti3C2 | POD/Luminol | Catalytic accelerator | Glucose | 0.1–40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μM 000 μM |
0.02 μM | 235 |
| CoS2/sulfur-doped Ti3C2 MXene | POD/Luminol | Conductive material | Chloramphenicol | 0.1 fM–1. pM | 105 fM | 239 |
| Ti3C2Tx MXene@Au nanohybrid | OD/OPD | Catalytic enhancer | Hg2+ | 0.4–2000 nM | 17 pM | 255 |
| MXene–au composite-supported Pt@NiCo-LDH | POD/OPD | Catalytic supporter | Ochratoxin A | 20 fg mL−1–100 ngmL−1 | 8.9 fg mL−1 | 259 |
| N-Doped Ti3C2Tx MXene combined AgNPs | POD | Catalytic promoter | H2O2 | 260 | ||
| Ti3C2Tx MXzymes | POD/1-naphthyl phosphate | Catalyst | BCR/ABL fusion gene | 0.2 fM–20 nM | 0.05 fM | 262 |
| Ti3C2Tx MXzymes | Catalase/tyrosinase | Electron carrier | Phenol | 0.05–15.5 μM | 12 nM | 263 |
| Phosphorene/Ti3C2Tx MXene nanohybrid | OD | Catalyst stability enhancer | α-Naphthalene acetic acid | 0.02–40 μM | 1.6 nM | 265 |
| Cobalt-doped 2D Ti3C2Tx MXene nanosheets | POD/OPD | Nanozyme catalyst | Paraoxon | 0.05–50 ng mL−1 | 0.02 ng mL−1 | 268 |
| Chitosan functionalized Ti3C2Tx MXene nanocomposites | POD/acetylcholine | Catalytic supporter | Malathion | 10 fM–10 nM | 3 fM | 21 |
| Ultrathin Ti3C2Tx nanosheets/Au–Pd BMNPs | POD/acetylcholine | Catalytic supporter | Paraoxon | 0.1–1000 μg L−1 | 1.75 ng L−1 | 270 |
| AuNP self-assembled Ti3C2Tx MXene@hemin composite | POD/TMB | Catalytic supporter | Tuberculosis | 100 fg mL−1–50 ng mL−1 | 2.36 fg mL−1 | 273 |
| DNA nanostructure/Ti3C2Tx MXene nano complexes | OD | Catalyst booster | Gliotoxin | 5–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 pM 000 pM |
5 pM | 275 |
| Ti3C2Tx MXene nanosheets | POD/horseradish | Nanozyme catalyst | Prostate-specific antigen | 0.1–50 ng mL−1 | 0.031 ng mL−1 | 276 |
| Ti3C2Tx MXene/Ag2S combined with CDs decorated with Au@Ag core–shell nanozymes | POD | Catalytic supporter | MicroRNA-155, a cancer biomarker | 1–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fM 000 fM |
0.83 fM | 277 |
| Ti3C2Tx MXene@AuNPs | OD/thionine | Catalytic supporter | Creatinine | 0.4–5000 μg mL−1 | 0.03 μg mL−1 | 279 |
| Ti3C2Tx incorporated Fe atom-based single-atom nanozymes | POD/TMB | Catalytic support | Ascorbic acid | 10−8 to 10−3 M | — | 288 |
| Uric acid | ||||||
| Melatonin | ||||||
| GSH | ||||||
| Tea polyphenols |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 fM. F. Zhang's research team reported a competitive electrochemical aptasensor based on a cDNA-ferrocene/Ti3C2Tx MXene probe for detecting the breast cancer biomarker mucin1 (MUC1).278 Notable for their excellent electrical conductivity and extensive surface area, Ti3C2Tx MXene nanosheets were employed as transporters for the cDNA-ferrocene (cDNA-Fc) probe. The aptasensor was fabricated by immobilizing an MUC1 aptamer on an AuNP-modified glassy carbon electrode (Au/GCE) via Au–S bonding, forming Apt/Au/GCE. The complementary cDNA-Fc probe was then attached to the Ti3C2Tx MXene surface, creating the cDNA-Fc/Ti3C2Tx MXene probe, which hybridized with the aptamer to form the final cDNA-Fc/Ti3C2Tx MXene/Apt/Au/GCE aptasensor. This reaction scheme is displayed in Fig. 12b. The cDNA-Fc/Ti3C2Tx MXene probe was removed from the electrode via a competitive recognition process upon the introduction of MUC1, lowering the electrical signal that was linked with the concentration of MUC1. This aptasensor was monitored by SWV and EIS. This sensor exhibited a wide linear detection range (1 pM to 10 mM) and a low detection limit (0.33 pM), demonstrating high sensitivity. The findings demonstrated the MXene-based aptasensor's potential for clinical diagnostic applications by showing that it offered an early breast cancer detection method that was sensitive, selective, and economical. In addition, molecularly imprinted polymer (MIP) and a bio-mimic nanozyme using Ti3C2Tx MXene@AuNPs have been applied for the detection of creatinine.279 To refine its synthesis and surface functions, clarify the catalytic mechanisms, and realize its applications, more research studies are needed. The development of MXene and its composite-based nanozymes is still in its early stages. Superactive MXzymes would need to be carefully designed and developed, and their successful integration with simple detection methods would be necessary for their broad use. MXzyme may develop into a strong contender for electrochemical biosensing of cancer biomarkers as a result of these initiatives.
000 fM. F. Zhang's research team reported a competitive electrochemical aptasensor based on a cDNA-ferrocene/Ti3C2Tx MXene probe for detecting the breast cancer biomarker mucin1 (MUC1).278 Notable for their excellent electrical conductivity and extensive surface area, Ti3C2Tx MXene nanosheets were employed as transporters for the cDNA-ferrocene (cDNA-Fc) probe. The aptasensor was fabricated by immobilizing an MUC1 aptamer on an AuNP-modified glassy carbon electrode (Au/GCE) via Au–S bonding, forming Apt/Au/GCE. The complementary cDNA-Fc probe was then attached to the Ti3C2Tx MXene surface, creating the cDNA-Fc/Ti3C2Tx MXene probe, which hybridized with the aptamer to form the final cDNA-Fc/Ti3C2Tx MXene/Apt/Au/GCE aptasensor. This reaction scheme is displayed in Fig. 12b. The cDNA-Fc/Ti3C2Tx MXene probe was removed from the electrode via a competitive recognition process upon the introduction of MUC1, lowering the electrical signal that was linked with the concentration of MUC1. This aptasensor was monitored by SWV and EIS. This sensor exhibited a wide linear detection range (1 pM to 10 mM) and a low detection limit (0.33 pM), demonstrating high sensitivity. The findings demonstrated the MXene-based aptasensor's potential for clinical diagnostic applications by showing that it offered an early breast cancer detection method that was sensitive, selective, and economical. In addition, molecularly imprinted polymer (MIP) and a bio-mimic nanozyme using Ti3C2Tx MXene@AuNPs have been applied for the detection of creatinine.279 To refine its synthesis and surface functions, clarify the catalytic mechanisms, and realize its applications, more research studies are needed. The development of MXene and its composite-based nanozymes is still in its early stages. Superactive MXzymes would need to be carefully designed and developed, and their successful integration with simple detection methods would be necessary for their broad use. MXzyme may develop into a strong contender for electrochemical biosensing of cancer biomarkers as a result of these initiatives.
 | ||
| Fig. 12 MXene composite material-based nanozyme sensors for cancer biomarkers. (a) PEC-based sensors for microRNA. In (i) and (ii), preparation methods for hairpin probe (S1)-conjugated Au@Ag@CDs and multifunctional signal amplification strategies, based on 2D Ti3C2Tx MXene/Ag2S composites, Ag@Ag@CDs, and target microRNA-induced formation of triple helix molecules on the electrode, were exemplified, respectively [reproduced from ref. 277 with permission from Elsevier, copyright 2022]. (b) Synthetic protocol of the cDNA-Fc/Ti3C2Tx MXene probe and the formation of the cDNA-Fc/Ti3C2Tx MXene/Apt/Au/GCE aptasensor for MUC1 detection [reproduced from ref. 278 with permission from Elsevier, copyright 2020]. | ||
5.7. MXzyme-based SERS sensors
The ongoing development of sensor technology is necessary to meet the growing requirement for a secure and dependable approach in metabolic sensing. Surface-enhanced Raman spectroscopy (SERS) has become one of its most anticipated tools for sensitivity and identification or detection in biomedical and agri-food applications in the past 20 years. SERS is distinct and adaptable among spectroscopy methods because it circumvents the intrinsic sensitivity limit of Raman spectroscopy, which produces vibrational “fingerprint” spectra of molecules.280–283 The pyridine Raman spectra on polished Ag were originally observed in 1974, although the investigators were unaware that these spectra were the result of any novel, increased, or unexpected events at the time.284 SERS is, at its most basic, a method of greatly boosting the signal from the weak but substantially enriched Raman scattering technique.282 Although scientists have used several techniques, such as electronic resonance intensification and triggered Raman processes, to improve the effectiveness of Raman scattering, SERS provides the most notable amplification of the Raman signal.285,286 Because of the significant improvement, single molecules are now frequently detected at their most complicated level.282 Furthermore, SERS is a superb method for characterizing a limited number of molecules that are attached to or close to nanostructured material surfaces.280Biologically important compounds, such as melatonin (ME), AA, UA, GSH, and tea polyphenols (TP), are five antioxidants that are directly linked to human disorders. Free radicals can be captured and neutralized by these antioxidants in the human body. An excess of these antioxidants will result in oxidative stress and many diseases.287 Thus, it is very desirable to develop appropriate sensing techniques for these antioxidants. P. Yin's research group investigated the development of Ti3C2Tx incorporated Fe atom-based single-atom nanozymes (Fe-SA/Ti3C2Tx) as efficient alternatives to natural enzymes for the detection of five different antioxidants by the SERS method.288 By stimulating the oxidation of TMB by colorimetric reactions, these materials showed inherent POD-like activity. By stimulating the oxidation of TMB by colorimetric reactions, the Fe-SA/Ti3C2Tx material showed inherent POD-like activity. Density functional theory (DFT) simulations verified the spontaneous dissociation of H2O2 and the generation of hydroxyl radicals, whereas X-ray absorption fine structure (XAFS) studies verified electron transport between Fe and Ti3C2Txvia Fe–O–Ti ligands. Furthermore, Fe-SA/Ti3C2Tx-based nanozyme was utilized as a SERS substrate for sensing oxidized TMB (TMB+), accomplishing noteworthy indicator magnification. This method made it easier to distinguish between five antioxidants, including AA, UA, GSH, ME, and TP, depending on the degree to which they inhibit free radical reactions. Furthermore, all five antioxidants were found to have a broad detection range of 10−8 to 10−3 M. This sensing analysis opened the door for the use of Fe-SA/Ti3C2Tx in biochemical sensing by demonstrating its potential as an efficient and discriminating platform for antioxidant detection (Table 4). In the Fe-SA/Ti3C2Tx-based nanozyme, Ti3C2Tx MXene played a crucial role as a support material for Fe single-atom nanozymes. Through the use of Fe–O–Ti ligands, electron transport between Fe atoms and the substrate was made easier by the highly conductive and chemically robust platform that the MXene matrix offered. Additionally, Ti3C2Tx subsidized the POD-like catalytic activity of Fe-SA/Ti3C2Tx-based nanozyme by boosting the impulsive dissociation of H2O2 and helping the production of hydroxyl radicals. Furthermore, Ti3C2Tx enhanced the SERS signal response, permitting the specific sensing of antioxidants by magnifying the signals of oxidized TMB (TMB+). As a result, Ti3C2Tx MXene greatly improved the activity of Fe-SA nanozymes in antioxidant sensing applications by acting as a structural, electrical, and catalytic booster.
6. Conclusions
Undoubtedly, nanozymes have been increasingly popular in recent years due to their enzyme-mimicking ability, chemical stability, and low fabrication cost. Particularly, MXzymes have been recognized as one of the representative nanozymes that have yet to be developed from a material point of view. In this review, we first briefly summarize the synthesis of MXenes and their unique characteristics. The enzyme-like capabilities of MXene materials with their flexible physicochemical features have found applications that include environmental engineering and catalytic synthesis. In particular, it is indisputable that MXzymes can lend themselves to sensing technologies. In addition, MXzymes manifested as POD, ODs, CAT, and SOD agents, and function in multi-enzyme mimic reactions. Different types of nanozymes incorporated into 2D MXene nanocomposites, the sensing mechanisms of MXzyme-based sensors, and the factors affecting nanozyme activity were discussed. Multiple investigations have shown that MXzymes can serve as core building blocks for biosensors that detect high-impact analytes due to their superior enzyme-mimicking ability, high electrical conductivity, high surface area, and customizable surface chemistry. MXzymes have been exploited for the analysis of a wide range of chemical and biological analytes in various sensing platforms such as colorimetric, smartphone-assisted, fluorescent, chemiluminescent, electrochemiluminescent, electrochemical, and SERS. Dealing with comprehensive details of the MXzymes, this review aims to provide insights into a strong basis for future research and pathways to solutions to challenges encountered in sensing applications.Future perspectives
Although research into design, synthesis, and performance has significantly advanced over the past decade, the use of MXzymes for biochemical sensing and monitoring is still at a relatively early-stage. Their future development in this area depends on several key factors. Creating MXene-based nanomaterials with precise sizes, uniform thickness, and high durability remains difficult. Additionally, it is crucial to develop effective techniques for scalable production using environmentally friendly chemicals. Future research on MXene-based catalysts should focus on improving their catalytic efficiency and addressing current deployment challenges. Potential strategies include designing hybrid MXene composites with metals, carbon materials, or polymers for better stability and conductivity, as well as surface modifications through heteroatom doping or defect engineering to enhance activity and selectivity. Progress in their practical application will also require more cost-effective and scalable manufacturing methods, improved oxidation resistance, and deeper mechanistic understanding via in situ characterization. They must also possess excellent dispersibility for various uses.The effects of MXenes on the environment and human health must be carefully considered as they advance toward practical biosensing applications. By reducing negative effects, surface functionalization with polymers or biocompatible ligands like antioxidants may enhance safety profiles. It is also crucial to comprehend their ecological fate, which includes persistence, bioaccumulation, and environmental modification. The implementation of MXene-based biosensors will be safer and more sustainable with the development of green synthesis techniques and regulatory norms.
Future studies will focus on elucidating the environmental stability, biodegradation, and biocompatibility of MXene-based nanomaterials, as these characteristics are currently mostly unknown. According to recent research, harmful effects like oxidative stress and inflammatory reactions may be brought on by the surface chemistry and possible metal ion release of V-, Mo-, and Nb-based MXenes. These results highlight how crucial it is to conduct thorough toxicological analyses to guarantee safe use.289,290 However, the great catalytic potential of V-, Mo-, and Nb-based MXenes beyond the comparatively safer Ti3C2 MXene could be utilized for novel applications if these toxicity issues are properly addressed. The availability of diverse surface chemistries and electrical properties of these alternative MXenes may benefit high sensitivity and binding flexibility for sensing applications. Beyond Ti-based MXzymes, this exploration may transform sensing strategies and open the way for next-generation, rapid, economical, and extremely effective detection systems in the biological and environmental domains.
While the majority of the current MXene-based nanozymes catalyze redox reactions, it is worthwhile to discover new types of catalytic activities due to their enzyme-mimicking ability and structural versatility. Further developments may enable MXenes to replicate their structures as well as functions towards multifunctional designs with the aid of advanced molecular engineering and nanotechnology. These developments would increase the potential of MXzymes in a variety of disciplines, including biochemical detection.
The inherent chemical stability of MXzymes that allows sensing techniques to be realized in a portable scheme, even in challenging circumstances, can be further optimized for smartphone-assisted MXene-based biosensors. This smartphone-assisted platform may revolutionize the real-time monitoring format that enables multi-target sensing, IoT integration, and artificial intelligence-powered analyses as the next-generation sensors for health care and environmental monitoring.
The commercialization of nanozyme-based sensors for potential analytes remains slow due to challenges in translating lab research into practical technologies. Achieving high sensitivity and specificity in complex biological and environmental matrices is crucial for quantifiable relevance. Nanozymes have promising real-world applications due to their remarkable stability, affordability, and adjustable features. Still, large-scale manufacture and severe clinical validation are vital for biomarkers. Cost-effective manufacturing and close multidisciplinary cooperation between scientists, business executives, and legislators will be necessary for future developments. By integrating advanced approaches, these sensors can revolutionize health care and environmental monitoring. As the investigation intensifies, nanozyme-based sensing technology will become a vital tool for early disease detection, enhancing healthcare, and precision environmental monitoring. Breakthrough developments in healthcare and other fields will be made possible by minimizing the gap between proof-of-concept and commercial reality. We strongly believe that this comprehensive review will be a good starting point for future research into MXzyme sensors for high-impact analytes.
Conflicts of interest
There are no conflicts to declare.Data availability
No new primary data were generated for this review. The analysis of the methodologies presented herein is based entirely on data extracted from previously published studies, all of which are appropriately cited within the manuscript.Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grants funded by the Korean government (MSIT) (No. NRF-2021M3H4A3A02086939 and No. RS-2023-00279149).References
- Y. Huang, J. Ren and X. Qu, Nanozymes: classification, catalytic mechanisms, activity regulation, and applications, Chem. Rev., 2019, 119(6), 4357–4412 CrossRef CAS PubMed
.
- S. Ahmadi, K. Rahimizadeh, A. Shafiee, N. Rabiee and S. Iravani, Nanozymes and their emerging applications in biomedicine, Process Biochem., 2023, 131, 154–174 CrossRef CAS
.
- H. L. Anderson and J. K. Sanders, Enzyme mimics based on cyclic porphyrin oligomers: strategy, design, and exploratory synthesis. Journal of the Chemical Society, Perkin Trans. 1, 1995, 18, 2223–2229 RSC
.
- S. J. Hong, H. Chun, M. Hong and B. Han, N-and B-doped fullerene as peroxidase-and catalase-like metal-free nanozymes with pH-switchable catalytic activity: a first-principles approach, Appl. Surf. Sci., 2022, 598, 153715 CrossRef CAS
.
- D. Zhu, M. Zhang, L. Pu, P. Gai and F. Li, Nitrogen-enriched conjugated polymer enabled metal-free carbon nanozymes with efficient oxidase-like activity, Small, 2022, 18(3), 2104993 CrossRef CAS PubMed
.
- E. Cheng, B. Li and Q. Zou, Porphyrin-based nanozymes for biomedical applications, Adv. Ther., 2024, 7(4), 2300329 CrossRef CAS
.
- L. J. Zhi and A. L. Sun, Platinum nanozyme-encapsulated poly (amidoamine) dendrimer for voltammetric immunoassay of pro-gastrin-releasing peptide, Anal. Chim. Acta, 2020, 1134, 106–114 CrossRef CAS PubMed
.
- R. Yu, R. Wang, Z. Wang, Q. Zhu and Z. Dai, Applications of DNA-nanozyme-based sensors, Analyst, 2021, 146(4), 1127–1141 RSC
.
- D. Wang, D. Jana and Y. Zhao, Metal–organic framework derived nanozymes in biomedicine, Acc. Chem. Res., 2020, 53(7), 1389–1400 CrossRef CAS PubMed
.
- L. Gao, J. Zhuang, L. Nie, J. Zhang, Y. Zhang, N. Gu and X. Yan, Intrinsic peroxidase-like activity of ferromagnetic nanoparticles, Nat. Nanotechnol., 2007, 2(9), 577–583 CrossRef CAS PubMed
.
- M. K. Sarangi, G. Rath and D. K. Yi, Metal-organic framework-based nanozymes explored with their environmental pollution control strategies, Inorg. Chem. Commun., 2024, 113334 CrossRef CAS
.
- R. Liang, Y. Li, M. Huo, H. Lin and Y. Chen, Triggering sequential catalytic fenton reaction on 2D MXenes for hyperthermia-augmented synergistic nanocatalytic cancer therapy, ACS Appl. Mater. Interfaces, 2019, 11(46), 42917–42931 CrossRef CAS PubMed
.
- L. Zhang, X. Bi, X. Liu, Y. He, L. Li and T. You, Advances in the application of metal–organic framework nanozymes in colorimetric sensing of heavy metal ions, Nanoscale, 2023, 15(31), 12853–12867 RSC
.
- X. Wang, H. Wang and S. Zhou, Progress and perspective on carbon-based nanozymes for peroxidase-like applications, J. Phys. Chem. Lett., 2021, 12(48), 11751–11760 CrossRef CAS PubMed
.
- B. Unnikrishnan, C. W. Lien, H. W. Chu and C. C. Huang, A review on metal nanozyme-based sensing of heavy metal ions: Challenges and future perspectives, J. Hazard. Mater., 2021, 401, 123397 CrossRef CAS PubMed
.
- M. Xu, S. Zhao, W. Zhang, D. Li, Y. Peng, M. Tanemura and Y. Yang, Wheatgrass-inspired MoSe2@ Pt heterojunctions with enhanced catalytic and photothermal functionalities for highly sensitive pathogen diagnosis based on machine learning, Sens. Actuators, B, 2025, 426, 137020 CrossRef CAS
.
- J. Zhao, Z. Shi, M. Chen and F. Xi, Highly active nanozyme based on nitrogen-doped graphene quantum dots and iron ion nanocomposite for selective colorimetric detection of hydroquinone, Talanta, 2025, 281, 126817 CrossRef CAS PubMed
.
- X. Kang, Y. Li, Z. Duan, X. Shen, R. Fu and D. Fan, A Mxene@ TA/Fe dual-nanozyme composited antifouling hydrogel for burn wound repair, Chem. Eng. J., 2023, 476, 146420 CrossRef CAS
.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, Two-dimensional nanocrystals produced by exfoliation of Ti3AlC2, Adv. Mater., 2011, 23, 4248e4253 Search PubMed
.
- A. M. Patil, A. A. Jadhav, N. R. Chodankar, A. T. Avatare, J. Hong, S. D. Dhas and S. C. Jun, Recent progress of MXene synthesis, properties, microelectrode fabrication techniques for micro supercapacitors and microbatteries energy storage devices and integration: A comprehensive review, Coord. Chem. Rev., 2024, 517, 216020 CrossRef CAS
.
- L. Zhou, X. Zhang, L. Ma, J. Gao and Y. Jiang, Acetylcholinesterase/chitosan-transition metal carbides nanocomposites-based biosensor for the organophosphate pesticides detection, Biochem. Eng. J., 2017, 128, 243–249 CrossRef CAS
.
- R. Rajamanikandan, K. Sasikumar and H. Ju, Ti3C2 MXene quantum dots as an efficient fluorescent probe for bioflavonoid quercetin quantification in food samples, Anal. Chim. Acta, 2024, 1322, 343069 CrossRef CAS PubMed
.
- P. K. Kalambate, N. S. Gadhari, X. Li, Z. Rao, S. T. Navale, Y. Shen and Y. Huang, Recent advances in MXene–based electrochemical sensors and biosensors, TrAC, Trends Anal. Chem., 2019, 120, 115643 CrossRef CAS
.
- A. Rhouati, M. Berkani, Y. Vasseghian and N. Golzadeh, MXene-based electrochemical sensors for detection of environmental pollutants: A comprehensive review, Chemosphere, 2022, 291, 132921 CrossRef CAS PubMed
.
- B. Muthukutty, P. S. Kumar, A. K. Vivekanandan, M. Sivakumar, S. Lee and D. Lee, Progress and Perspective in harnessing MXene–carbon-based composites (0–3D): synthesis, performance, and applications, Chemosphere, 2024, 141838 CrossRef CAS PubMed
.
- R. Mangiri, T. Ramachandran, Y. A. Kumar, A. Ghosh, A. G. Al-Sehemi, A. K. Yadav and D. Mani, Surface engineering of M5X4 MXenes for next-gen energy solutions, Mater. Today Chem., 2025, 48, 102864 CrossRef CAS
.
- B. Lu, Z. Zhu, B. Ma, W. Wang, R. Zhu and J. Zhang, 2D MXene nanomaterials for versatile biomedical applications: current trends and future prospects, Small, 2021, 17(46), 2100946 CrossRef CAS PubMed
.
- K. Enoch, A. Sundaram, S. S. Ponraj, S. Palaniyappan, S. D. B. George and R. K. Manavalan, Enhancement of MXene optical properties towards medical applications via metal oxide incorporation, Nanoscale, 2023, 15(42), 16874–16889 RSC
.
- X. Wu, T. Chen, Y. Chen and G. Yang, Modified Ti3C2 nanosheets as peroxidase mimetics for use in colorimetric detection and immunoassays, J. Mater. Chem. B, 2020, 8(13), 2650–2659 RSC
.
- A. Sreedhar, M. R. Pallavolu and J. S. Noh, Progress in surface and interlayer distance modulated 2D Ti3C2 MXenes for potential flexible supercapacitors: a review, Appl. Mater. Today, 2023, 35, 101942 CrossRef
.
- S. Vallem, S. Venkateswarlu, S. Song, J. Kim, Y. Li and J. Bae, Flexible MXene-conjugated polymer nanoarchitectures: Current developments and future frontiers in battery technology, Coord. Chem. Rev., 2024, 510, 215778 CrossRef CAS
.
- A. Sreedhar, Q. T. H. Ta and J. S. Noh, Advancements in the photocatalytic activity of various bismuth-based semiconductor/Ti3C2 MXene interfaces for sustainable environmental management: A review, J. Ind. Eng. Chem., 2022, 115, 26–47 CrossRef CAS
.
- A. Sreedhar, Q. T. H. Ta and J. S. Noh, Versatile role of 2D Ti3C2 MXenes for advancements in the photodetector performance: A review, J. Ind. Eng. Chem., 2023, 127, 1–17 CrossRef CAS
.
- S. Y. Pang, W. F. Io, F. Guo, Y. Zhao and J. Hao, Two-dimensional MXene-based devices for information technology, Mater. Sci. Eng., R, 2025, 163, 100894 CrossRef
.
- S. Wang, H. Ma, S. Ge, M. Rezakazemi and J. Han, Advanced design strategies and multifunctional applications of Nanocellulose/MXene composites: A comprehensive review, Mater. Sci. Eng., R, 2025, 163, 100925 CrossRef
.
- R. Yang, S. Wen, S. Cai, W. Zhang, T. Wu and Y. Xiong, MXene-based nanomaterials with enzyme-like properties for biomedical applications, Nanoscale Horiz., 2023, 8(10), 1333–1344 RSC
.
- S. Iravani and R. S. Varma, MXene-based composites as nanozymes in biomedicine: a perspective, Nano-Micro Lett., 2022, 14(1), 213 CrossRef CAS PubMed
.
- C. Y. Hsu, N. M. Alshik, I. Ahmad, S. Uthirapathy, S. Ballal, A. Singh and K. K. Joshi, Recent advances in MXene nanozymes-based optical and electrochemical biosensors for food safety analysis, Nanoscale, 2025, 17, 7697–7712 RSC
.
- P. F. Huang and W. Q. Han, Recent advances and perspectives of lewis acidic etching route: an emerging preparation strategy for MXenes, Nano-Micro Lett., 2023, 15, 68 CrossRef CAS PubMed
.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, Two-dimensional nanocrystals produced by exfoliation of Ti3AlC2, Adv. Mater., 2011, 23(37), 4248–4253 CrossRef CAS PubMed
.
- V. Natu and M. W. Barsoum, MXene surface terminations: a perspective, J. Phys. Chem. C, 2023, 127(41), 20197–20206 CrossRef CAS
.
- M. Alhabeb, K. Maleski, T. S. Mathis, A. Sarycheva, C. B. Hatter, S. Uzun, A. Levitt and Y. Gogotsi, Selective Etching of Silicon from Ti3SiC2 (MAX) to Obtain 2D Titanium Carbide (MXene), Angew. Chem., Int. Ed., 2018, 57(19), 5444–5448 CrossRef CAS PubMed
.
- G. Deysher, C. E. Shuck, K. Hantanasirisakul, N. C. Frey, A. C. Foucher, K. Maleski, A. Sarycheva, V. B. Shenoy, E. A. Stach, B. Anasori and Y. Gogotsi, Synthesis of Mo4VAlC4 MAX phase and two-dimensional Mo4VC4 MXene with five atomic layers of transition metals, ACS Nano, 2019, 14(1), 204–217 CrossRef PubMed
.
- A. VahidMohammadi, A. Hadjikhani, S. Shahbazmohamadi and M. Beidaghi, Two-dimensional vanadium carbide (MXene) as a high-capacity cathode material for rechargeable aluminum batteries, ACS Nano, 2017, 11(11), 11135–11144 CrossRef CAS PubMed
.
- P. Lakhe, E. M. Prehn, T. Habib, J. L. Lutkenhaus, M. Radovic, M. S. Mannan and M. J. Green, Process safety analysis for Ti3C2Tx MXene synthesis and processing, Ind. Eng. Chem. Res., 2019, 58(4), 1570–1579 CrossRef CAS
.
- Y. An, Y. Tian, H. Shen, Q. Man, S. Xiong and J. Feng, Two-dimensional MXenes for flexible energy storage devices, Energy Environ. Sci., 2023, 16(10), 4191–4250 RSC
.
- M. Ghidiu, M. R. Lukatskaya, M. Q. Zhao, Y. Gogotsi and M. W. Barsoum, Conductive two-dimensional titanium carbide ‘clay’ with high volumetric capacitance, Nature, 2014, 516(7529), 78–81 CrossRef CAS PubMed
.
- X. Wang, C. Garnero, G. Rochard, D. Magne, S. Morisset, S. Hurand, P. Chartier, J. Rousseau, T. Cabioc'h, C. Coutanceau and V. Mauchamp, A new etching environment (FeF3/HCl) for the synthesis of two-dimensional titanium carbide MXenes: a route towards selective reactivity vs. water, J. Mater. Chem. A, 2017, 5(41), 22012–22023 RSC
.
- F. Liu, A. Zhou, J. Chen, J. Jia, W. Zhou, L. Wang and Q. Hu, Preparation of Ti3C2 and Ti2C MXenes by fluoride salts etching and methane adsorptive properties, Appl. Surf. Sci., 2017, 416, 781–789 CrossRef CAS
.
- A. Feng, Y. Yu, Y. Wang, F. Jiang, Y. Yu, L. Mi and L. Song, Two-dimensional MXene Ti3C2 produced by exfoliation of Ti3AlC2, Mater. Des., 2017, 114, 161–166 CrossRef CAS
.
- J. Halim, M. R. Lukatskaya, K. M. Cook, J. Lu, C. R. Smith, L. Å. Näslund, S. J. May, L. Hultman, Y. Gogotsi, P. Eklund and M. W. Barsoum, Transparent conductive two-dimensional titanium carbide epitaxial thin films, Chem. Mater., 2014, 26(7), 2374–2381 CrossRef CAS PubMed
.
- M. Alhabeb, K. Maleski, B. Anasori, P. Lelyukh, L. Clark, S. Sin and Y. Gogotsi, Guidelines for synthesis and processing of two-dimensional titanium carbide (Ti3C2Tx MXene), Chem. Mater., 2017, 29(18), 7633–7644 CrossRef CAS
.
- S. Zhou, Y. Guan, L. Tan, X. Li, H. Zhu, Q. Zhang, Z. Dong, N. Yang and Y. Cong, Recent advances in multiple transition metal MXenes: Synthesis, properties, and applications in energy storage, J. Energy Storage, 2025, 120, 116419 CrossRef
.
- L. Wang, H. Zhang, B. Wang, C. Shen, C. Zhang, Q. Hu, A. Zhou and B. Liu, Synthesis and electrochemical performance of Ti3C2Tx with hydrothermal process, Electron. Mater. Lett., 2016, 12, 702–710 CrossRef CAS
.
- Y. Guo, S. Jin, L. Wang, P. He, Q. Hu, L. Z. Fan and A. Zhou, Synthesis of two-dimensional carbide Mo2CTx MXene by hydrothermal etching with fluorides and its thermal stability, Ceram. Int., 2020, 46(11), 19550–19556 CrossRef CAS
.
- C. Peng, P. Wei, X. Chen, Y. Zhang, F. Zhu, Y. Cao, H. Wang, H. Yu and F. Peng, A hydrothermal etching route to synthesis of 2D MXene (Ti3C2, Nb2C): Enhanced exfoliation and improved adsorption performance, Ceram. Int., 2018, 44(15), 18886–18893 CrossRef CAS
.
- S. Siddique, A. Waheed, M. Iftikhar, M. T. Mehran, M. Z. Zarif, H. A. Arafat and F. Shahzad, Fluorine-free MXenes via molten salt Lewis acidic etching: applications, challenges, and future outlook, Prog. Mater. Sci., 2021, 139, 101183 CrossRef
.
- C. Wang, H. Shou, S. Chen, S. Wei, Y. Lin, P. Zhang, Z. Liu, K. Zhu, X. Guo, X. Wu and P. M. Ajayan, HCl-based hydrothermal etching strategy toward fluoride-free MXenes, Adv. Mater., 2021, 33(27), 2101015 CrossRef CAS PubMed
.
- P. Urbankowski, B. Anasori, T. Makaryan, D. Er, S. Kota, P. L. Walsh, M. Zhao, V. B. Shenoy, M. W. Barsoum and Y. Gogotsi, Synthesis of two-dimensional titanium nitride Ti4N3 (MXene), Nanoscale, 2016, 8(22), 11385–11391 RSC
.
- I. R. Shein and A. L. Ivanovskii, Graphene-like titanium carbides and nitrides Tin+1Cn, Tin+ 1Nn (n= 1, 2, and 3) from de-intercalated MAX phases: First-principles probing of their structural, electronic properties and relative stability, Comput. Mater. Sci., 2012, 65, 104–114 CrossRef CAS
.
- M. Li, J. Lu, K. Luo, Y. Li, K. Chang, K. Chen, J. Zhou, J. Rosen, L. Hultman, P. Eklund and P. O. Persson, Element replacement approach by reaction with Lewis acidic molten salts to synthesize nanolaminated MAX phases and MXenes, J. Am. Chem. Soc., 2019, 141(11), 4730–4737 CrossRef CAS PubMed
.
- V. Kamysbayev, A. S. Filatov, H. Hu, X. Rui, F. Lagunas, D. Wang, R. F. Klie and D. V. Talapin, Covalent surface modifications and superconductivity of two-dimensional metal carbide MXenes, Science, 2020, 369(6506), 979–983 CrossRef CAS PubMed
.
- K. Xie, J. Wang, K. Xu, Z. Wei, M. Zhang and J. Zhang, In-situ synthesis of fluorine-free MXene/TiO2 composite for high-performance supercapacitor, Arabian J. Chem., 2024, 17(2), 105551 Search PubMed
.
- B. Zhang, J. Zhu, P. Shi, W. Wu and F. Wang, Fluoride-free synthesis and microstructure evolution of novel two-dimensional Ti3C2(OH)2 nanoribbons as high-performance anode materials for lithium-ion batteries, Ceram. Int., 2019, 45(7), 8395–8405 CrossRef CAS
.
- T. Li, L. Yao, Q. Liu, J. Gu, R. Luo, J. Li, X. Yan, W. Wang, P. Liu, B. Chen and W. Zhang, Fluorine-free synthesis of high-purity Ti3C2Tx (T= OH, O) via alkali treatment, Angew. Chem., Int. Ed., 2018, 57(21), 6115–6119 CrossRef CAS PubMed
.
- W. Sun, S. A. Shah, Y. Chen, Z. Tan, H. Gao, T. Habib, M. Radovic and M. J. Green, Electrochemical etching of Ti2AlC to Ti2CTx (MXene) in low-concentration hydrochloric acid solution, J. Mater. Chem. A, 2017, 5(41), 21663–21668 RSC
.
- S. Yang, P. Zhang, F. Wang, A. G. Ricciardulli, M. R. Lohe, P. W. Blom and X. Feng, Fluoride-free synthesis of two-dimensional titanium carbide (MXene) using a binary aqueous system, Angew. Chem., Int. Ed., 2018, 57(47), 15491–15495 CrossRef CAS PubMed
.
- H. Shi, P. Zhang, Z. Liu, S. Park, M. R. Lohe, Y. Wu, A. Shaygan Nia, S. Yang and X. Feng, Ambient-stable two-dimensional titanium carbide (MXene) enabled by iodine etching, Angew. Chem., Int. Ed., 2021, 60(16), 8689–8693 CrossRef CAS PubMed
.
- C. Xu, L. Wang, Z. Liu, L. Chen, J. Guo, N. Kang, X. L. Ma, H. M. Cheng and W. Ren, Large-area high-quality 2D ultrathin Mo2C superconducting crystals, Nat. Mater., 2015, 14(11), 1135–1141 CrossRef CAS PubMed
.
- J. Vacík, P. Horák, S. Bakardjieva, V. Bejsovec, G. Ceccio, A. Cannavo, A. Torrisi, V. Lavrentiev and R. Klie, Ion sputtering for preparation of thin MAX and MXene phases, Radiat. Eff. Defects Solids, 2020, 175(1–2), 177–189 CrossRef
.
- D. L. Druffel, M. G. Lanetti, J. D. Sundberg, J. T. Pawlik, M. S. Stark, C. L. Donley, L. M. McRae, K. M. Scott and S. C. Warren, Synthesis and electronic structure of a 3D crystalline stack of MXene-like sheets, Chem. Mater., 2019, 31(23), 9788–9796 CrossRef CAS
.
- M. Zandieh and J. Liu, Nanozymes: definition, activity, and mechanisms, Adv. Mater., 2024, 36(10), 2211041 Search PubMed
.
- X. Zhang, X. Chen and Y. Zhao, Nanozymes: versatile platforms for cancer diagnosis and therapy, Nano-Micro Lett., 2022, 14(1), 95 CrossRef CAS PubMed
.
- A. Deisseroth and A. L. Dounce, Catalase: Physical and chemical properties, mechanism of catalysis, and physiological role, Physiol. Rev., 1970, 50(3), 319–375 CrossRef CAS PubMed
.
- Y. Chen, B. Li, K. Li and Y. Lin, Superoxide dismutase nanozymes: current status and future perspectives on brain disease treatment and diagnosis, Chem. Commun., 2024, 60(31), 4140–4147 RSC
.
- C. Ding, J. Liang, Z. Zhou, Y. Li, W. Peng, G. Zhang and X. Fan, Photothermal enhanced enzymatic activity of lipase covalently immobilized on functionalized Ti3C2TX nanosheets, Chem. Eng. J., 2019, 378, 122205 CrossRef CAS
.
- J. Wang, X. Wei, X. Wang, W. Song, W. Zhong, M. Wang and Y. Tang, Plasmonic Au nanoparticle@ Ti3C2Tx heterostructures for improved oxygen evolution performance, Inorg. Chem., 2021, 60(8), 5890–5897 CrossRef CAS PubMed
.
- S. Wen, Y. Xiong, S. Cai, H. Li, X. Zhang, Q. Sun and R. Yang, Plasmon-enhanced photothermal properties of Au@ Ti3C2Tx nanosheets for antibacterial applications, Nanoscale, 2022, 14(44), 16572–16580 Search PubMed
.
- Z. Jin, G. Xu, Y. Niu, X. Ding, Y. Han, W. Kong and Y. Xu, Ti3C2Tx MXene-derived TiO2/C-QDs as oxidase mimics for the efficient diagnosis of glutathione in human serum, J. Mater. Chem. B, 2020, 8(16), 3513–3518 RSC
.
- H. Hu, H. Huang, L. Xia, X. Qian, W. Feng, Y. Chen and Y. Li, Engineering vanadium carbide MXene as multienzyme mimetics for efficient in vivo ischemic stroke treatment, Chem. Eng. J., 2022, 440, 135810 CrossRef CAS
.
- D. Zhang, L. Jiang, L. Li, X. Li, W. Zheng, L. Gui and M. Gong, Integrated metabolomics revealed the fibromyalgia-alleviation effect of Mo2C nanozyme through regulated homeostasis of oxidative stress and energy metabolism, Biomaterials, 2022, 287, 121678 CrossRef CAS PubMed
.
- X. Ren, M. Huo, M. Wang, H. Lin, X. Zhang, J. Yin and H. Chen, Highly catalytic niobium carbide (MXene) promotes hematopoietic recovery after radiation by free radical scavenging, ACS Nano, 2019, 13(6), 6438–6454 CrossRef CAS PubMed
.
- X. Zhao, Y. Chen, R. Niu, Y. Tang, Y. Chen, H. Su and L. Meng, NIR plasmonic nanozymes: synergistic enhancement mechanism and multi-modal anti-infection applications of MXene/MOFs, Adv. Mater., 2024, 36(8), 2307839 CrossRef CAS PubMed
.
- X. Kang, Y. Li, Z. Duan, X. Shen, R. Fu and D. Fan, A Mxene@ TA/Fe dual-nanozyme composited antifouling hydrogel for burn wound repair, Chem. Eng. J., 2023, 476, 146420 CrossRef CAS
.
- H. Wang, C. Liu, Y. Zhao, X. Luo, P. Yin, F. Du and G. Zeng, Facile preparation of V2C@VOx nanosheets with excellent multi-enzyme activity and their colorimetric sensing application, New J. Chem., 2024, 48(11), 4886–4895 RSC
.
- W. Feng, X. Han, H. Hu, M. Chang, L. Ding, H. Xiang and Y. Li, 2D vanadium carbide MXenzyme to alleviate ROS-mediated inflammatory and neurodegenerative diseases, Nat. Commun., 2021, 12(1), 2203 CrossRef CAS PubMed
.
- T. Sahare, N. Singh, B. N. Sahoo and A. Joshi, Smartphone-enhanced nanozyme sensors: Colorimetric and fluorescence sensing techniques, Biosens. Bioelectron.:X, 2024, 100544 CAS
.
- N. N. Phuong Truong, R. Rajamanikandan, K. Sasikumar and H. Ju, Smartphone-Assisted Plasmonic Nanosensor for Visual and Specific Sensing of Toxic Cyanide Ions by β– Cyclodextrin Templated Gold-Rich/Silver Bimetallic Alloy Nanoparticles, Materials, 2025, 18(7), 1604 CrossRef CAS PubMed
.
- B. Liu, J. Zhuang and G. Wei, Recent advances in the design of colorimetric sensors for environmental monitoring, Environ. Sci.: Nano, 2020, 7(8), 2195–2213 RSC
.
- R. Rajamanikandan, K. Sasikumar and H. Ju, β-Cyclodextrin Functionalized Au@Ag Core-Shell Nanoparticles: Plasmonic Sensors for Cysteamine and Efficient Nanocatalysts for Nitrobenzene-to-Aniline Conversion, Biosensors, 2024, 14(11), 544 CrossRef CAS PubMed
.
- R. Rajamanikandan and M. Ilanchelian, Simple smartphone-merged rapid colorimetric platform for the environmental monitoring of toxic sulfide ions by cysteine functionalized silver nanoparticles, Microchem. J., 2022, 174, 107071 CrossRef CAS
.
-
(a) S. Kaushal, S. S. Nanda, D. K. Yi and H. Ju, Effects of aspect ratio heterogeneity of an assembly of gold nanorods on localized surface plasmon resonance, J. Phys. Chem. Lett., 2020, 11(15), 5972–5979 CrossRef CAS PubMed
; (b) N. T. T. Phuong, V. Q. Dang, T. N. Bach, B. X. Khuyen, H. K. T. Ta, H. Ju and N. H. T. Tran, Functionalized silver nanoparticles for SERS amplification with enhanced reproducibility and for ultrasensitive optical fiber sensing in environmental and biochemical assays, RSC Adv., 2022, 12(48), 31352–31362 RSC
.
- K. Sasikumar, R. Rajamanikandan and H. Ju, Fluorescent carbon dots for highly sensitive bilirubin sensing with excellent selectivity, J. Sci.:Adv. Mater. Devices, 2023, 8(3), 100599 Search PubMed
.
- A. S. Sharma and N. Y. Lee, Advancements in visualizing loop-mediated isothermal amplification (LAMP) reactions: a comprehensive review of colorimetric and fluorometric detection strategies for precise diagnosis of infectious diseases, Coord. Chem. Rev., 2024, 509, 215769 Search PubMed
.
- L. Yang, X. Xu, Y. Song, J. Huang and H. Xu, Research progress of nanozymes in colorimetric biosensing: Classification, activity, and application, Chem. Eng. J., 2024, 150612 CrossRef CAS
.
- V. G. Panferov and J. Liu, Optical and catalytic properties of nanozymes for colorimetric biosensors: Advantages, limitations, and perspectives, Adv. Opt. Mater., 2024, 12(30), 2401318 CrossRef CAS
.
- Z. Lyu, J. Zhou, S. Ding, D. Du, J. Wang, Y. Liu and Y. Lin, Recent advances in single-atom nanozymes for colorimetric biosensing, TrAC, Trends Anal. Chem., 2023, 168, 117280 CrossRef CAS
.
- S. Mansouri, Detection of pathogenic bacteria with nanozymes-based colorimetric biosensors: Advances, challenges and future prospects, Microchem. J., 2024, 111392 CrossRef CAS
.
- C. P. Kurup and M. U. Ahmed, Nanozymes towards personalized diagnostics: a recent progress in biosensing, Biosensors, 2023, 13(4), 461 CrossRef CAS PubMed
.
- X. Niu, X. Li, Z. Lyu, J. Pan, S. Ding, X. Ruan and Y. Lin, Metal–organic framework based nanozymes: promising materials for biochemical analysis, Chem. Commun., 2020, 56(77), 11338–11353 RSC
.
- G. Zhu, J. Hou, J. Xu, J. Li, C. Wang and Y. Yi, Enhanced peroxidase-like activity based on electron transfer between platinum nanoparticles and Ti3C2TX MXene nanoribbons coupled smartphone-assisted hydrogel platform for detecting mercury ions, Anal. Chim. Acta, 2024, 1329, 343250 CrossRef CAS PubMed
.
- Y. Shi, Z. Liu, R. Liu, R. Wu and J. Zhang, DNA-encoded MXene-Pt nanozyme for enhanced colorimetric sensing of mercury ions, Chem. Eng. J., 2022, 442, 136072 CrossRef CAS
.
- X. Zhou, J. Zhang, D. Liao, K. Wu, H. Liu, G. Zhu and Y. Yi, Self-reduce gold nanoparticles on Ti3C2Tx MXene nanoribbons to highly sensitive colorimetric determination of mercury ion and cysteine based on the mercury-motivated peroxidase mimetic activity, Sens. Actuators, B, 2023, 379, 133271 CrossRef CAS
.
- Y. Cheng, P. Shen, X. Li, X. Li, K. Chu and Y. Guo, Synergistically enhanced peroxidase-like activity of Fe3O4/Ti3C2 MXene quantum dots and its application in colorimetric determination of Cr (VI), Sens. Actuators, B, 2023, 376, 132979 CrossRef CAS
.
- Z. G. Khan and P. O. Patil, A comprehensive review on carbon dots and graphene quantum dots based fluorescent sensor for biothiols, Microchem. J., 2020, 157, 105011 CrossRef CAS
.
- J. Huang, P. Wu, Y. Qin, J. Zhang, W. Wang, X. Yi and Z. Chen, Tailoring the peroxidase-like properties of Mo atom nanoclusters/N-MXene nanozymes for sensitive colorimetric detection of glutathione, Talanta, 2024, 278, 126485 CrossRef CAS PubMed
.
- X. Lu, J. Jia, Z. Wang and W. Wang, MXene/Carbon Dots Nanozyme Composites for Glutathione Detection and Tumor Therapy, Nanomaterials, 2024, 14(13), 1090 CrossRef CAS PubMed
.
- J. Wang, W. Xu, L. Zhou, T. Zhang, N. Yang, M. Wang and W. Ge, Sensitive colorimetric sensing of glutathione and H2O2 based on enhanced peroxidase mimetic activity of MXene@ Fe3O4, Microchim. Acta, 2022, 189(12), 452 CrossRef CAS PubMed
.
- H. Geng, Z. Li, Q. Liu, Q. Yang, H. Jia, Q. Chen and W. He, Boosting the peroxidase-like activity of Pt nanozymes by a synergistic effect of Ti3C2 nanosheets for dual mechanism detection, Dalton Trans., 2022, 51(31), 11693–11702 RSC
.
- X. Xi, J. Wang, Y. Wang, H. Xiong, M. Chen, Z. Wu and W. Wen, Preparation of Au/Pt/Ti3C2Cl2 nanoflakes with self-reducing method for colorimetric detection of glutathione and intracellular sensing of hydrogen peroxide, Carbon, 2022, 197, 476–484 Search PubMed
.
- Z. Lei, J. Guo, J. Zou and Z. Wang, Colorimetric determination of biothiols based on peroxidase-mimicking Ag nanoparticles decorated Ti3C2 nanosheets, Microchim. Acta, 2022, 189(10), 369 CrossRef CAS PubMed
.
- M. Lian, Y. Zhao, J. Zhao, W. Zhang, H. Zhang and D. Chen, Oxidase-like V2C MXene nanozyme with inherent antibacterial properties for colorimetric sensing, Talanta, 2023, 265, 124872 Search PubMed
.
- S. Siva, G. A. Bodkhe, C. Cong, S. H. Kim and M. Kim, Heterostructured Au@ Pt/Nb2C-MXene nanocomposite as a colorimetric sensor with peroxidase-like activity for reactive oxygen species of H2O2, J. Ind. Eng. Chem., 2023, 124, 523–531 CrossRef CAS
.
- C. Ma, N. Jiang, X. Sun, L. Kong, T. Liang, X. Wei and P. Wang, Progress in optical sensors-based uric acid detection, Biosens. Bioelectron., 2023, 237, 115495 CrossRef CAS PubMed
.
- M. Liu, Y. He, J. Zhou, Y. Ge, J. Zhou and G. Song, A “naked-eye”colorimetric and ratiometric fluorescence probe for uric acid based on Ti3C2 MXene quantum dots, Anal. Chim. Acta, 2020, 1103, 134–142 CrossRef CAS PubMed
.
- D. Chen, S. Shao, W. Zhang, J. Zhao and M. Lian, Nitrogen and sulfur co-doping strategy to trigger the peroxidase-like and electrochemical activity of Ti3C2 nanosheets for sensitive uric acid detection, Anal. Chim. Acta, 2022, 1197, 339520 CrossRef CAS PubMed
.
- M. H. Facure, L. A. Mercante, Y. Gogotsi and D. S. Correa, ZnO–Co3O4 Nanofibers/MXene Composite with Peroxidase-Like Activity for Ascorbic Acid Detection, ACS Appl. Nano Mater., 2025, 8, 4291–4299 CrossRef CAS
.
- Q. Huang, J. Chen, Y. Zhao, J. Huang and H. Liu, Advancements in electrochemical glucose sensors, Talanta, 2025, 281, 126897 Search PubMed
.
- J. Huang, H. Gu, X. Feng, G. Wang and Z. Chen, Copper Nanoparticle/N-Doped Ti3C2Tx MXene Hybrids with Enhanced Peroxidase-like Activity for Colorimetric Glucose Sensing, ACS Appl. Nano Mater., 2022, 5(10), 15531–15538 CrossRef CAS
.
- Z. Li, T. Lei, T. Pei, K. Chen, Z. Zhao, M. Wang and Y. He, Facile synthesis of MXene-Ti3C2/Co nanosheet hydrogel sensor with the assistance of a smartphone for on-site monitoring of glucose in beverages, Molecules, 2023, 28(13), 5075 Search PubMed
.
- M. A. Sha, P. C. Meenu, H. Haspel and Z. Kónya, Metal-based non-enzymatic systems for cholesterol detection: mechanisms, features, and performance, RSC Adv., 2024, 14(34), 24561–24573 Search PubMed
.
- Y. Chen, C. Rong, W. Gao, S. Luo, Y. Guo, Y. Gu and L. L. Qu, Ag-MXene as peroxidase-mimicking nanozyme for enhanced bacteriocide and cholesterol sensing, J. Colloid Interface Sci., 2024, 653, 540–550 CrossRef CAS PubMed
.
- Y. Li, Z. Kang, L. Kong, H. Shi, Y. Zhang, M. Cui and D. P. Yang, MXene-Ti3C2/CuS nanocomposites: Enhanced peroxidase-like activity and sensitive colorimetric cholesterol detection, Mater. Sci. Eng., C, 2019, 104, 110000 Search PubMed
.
- V. K. Sharma, N. Johnson, L. Cizmas, T. J. McDonald and H. Kim, A review of the influence of treatment strategies on antibiotic resistant bacteria and antibiotic resistance genes, Chemosphere, 2016, 150, 702–714 CrossRef CAS PubMed
.
- W. Wang and S. Gunasekaran, MXene/Gold nanoparticles heterostructure as catalase mimic for colorimetric detection of penicillin G, Chem. Eng. J., 2024, 482, 148693 CrossRef CAS
.
- S. Zhang, Q. Peng, N. Jiang, C. Qiao, S. Li and W. Yue, Peroxidase-like activity and mechanism of gold nanoparticle-modified Ti3C2 MXenes for the construction of H2O2 and ampicillin colorimetric sensors, Microchim. Acta, 2024, 191(4), 195 CrossRef CAS PubMed
.
- W. Wang, Y. Yin and S. Gunasekaran, Oxygen-terminated few-layered Ti3C2Tx MXene nanosheets as peroxidase-mimic nanozyme for colorimetric detection of kanamycin, Biosens. Bioelectron., 2022, 218, 114774 CrossRef CAS PubMed
.
- L. Yang, H. Guo, T. Hou, J. Zhang and F. Li, Metal-mediated Fe3O4@ polydopamine-aptamer capture nanoprobe coupling multifunctional MXene@ Au@ Pt nanozyme for direct and portable photothermal analysis of circulating breast cancer cells, Biosens. Bioelectron., 2023, 234, 115346 CrossRef CAS PubMed
.
- Y. Tao, K. Yi, H. Wang, H. W. Kim, K. Li, X. Zhu and M. Li, CRISPR-Cas12a-regulated DNA adsorption and metallization on MXenes as enhanced enzyme mimics for sensitive colorimetric detection of hepatitis B virus DNA, J. Colloid Interface Sci., 2022, 613, 406–414 CrossRef CAS PubMed
.
- W. Wang, S. Xiao, Z. Jia, H. Xie, T. Li, Q. Wang and N. Gan, A dual-mode aptasensor for foodborne pathogens detection using Pt, phenylboric acid and ferrocene modified Ti3C2 MXenes nanoprobe, Sens. Actuators, B, 2022, 351, 130839 CrossRef CAS
.
- X. Xu, W. Zhang, J. Huang and H. Xu, Colorimetric sensors for detection of organophosphorus pesticides in food: From sensing strategies to chemometrics-driven discrimination, Trends Food Sci. Technol., 2024, 104683 CrossRef CAS
.
- Y. He, X. Zhou, L. Zhou, X. Zhang, L. Ma, Y. Jiang and J. Gao, Self-Reducing Prussian Blue on Ti3C2Tx MXene Nanosheets as a Dual-Functional Nanohybrid for Hydrogen Peroxide and Pesticide Sensing, Ind. Eng. Chem. Res., 2020, 59(35), 15556–15564 CrossRef CAS
.
- Y. Guo, X. Li, P. Shen, X. Li, Y. Cheng and K. Chu, Dendritic-like MXene quantum dots@ CuNi as an efficient peroxidase candidate for colorimetric determination of glyphosate, J. Colloid Interface Sci., 2024, 661, 533–543 CrossRef CAS PubMed
.
- Y. Yi, J. Li, X. Bi, L. Zhang, Y. Ren, L. Li and T. You, Highly active Ti3C2Tx MXene nanoribbons@ AuPt bimetallic nanozyme constructed in a “two birds with one stone” manner for colorimetric sensing of dipterex, Talanta, 2025, 281, 126881 CrossRef CAS PubMed
.
- H. Gu, H. Li, G. Wang, J. Huang, B. Peng, J. Pei and H. Zhai, Electronic Metal–Support Interactions between Copper Nanoparticles and Nitrogen-Doped
Ti3C2Tx MXene to Boost Peroxidase-like Activity for Detecting Astaxanthin, ACS Appl. Mater. Interfaces, 2023, 15(22), 26363–26372 CrossRef CAS PubMed
.
- Y. Shi, Z. Niu, S. Chen, S. Wang, L. Yang and Y. Wang, Sensitive Detection of Umami Substances in Meat with Synthesized Electrochemical Sensor Enhanced by MXene Surface-Doped Peroxidase-Mimic Nanoenzymes (Fe3O4), Food Bioprocess Technol., 2025, 18(1), 807–817 CrossRef CAS
.
- R. Rajamanikandan and M. Ilanchelian, β-Cyclodextrin protected gold nanoparticle based cotton swabs as an effective candidate for specific sensing of trace levels of cyanide, Anal. Methods, 2019, 11(1), 97–104 RSC
.
- B. Kaur, N. Kaur and S. Kumar, Colorimetric metal ion sensors–A comprehensive review of the years 2011–2016, Coord. Chem. Rev., 2018, 358, 13–69 CrossRef CAS
.
- K. Kaviya, R. Rajamanikandan, M. Santhamoorthy, M. A. Farah and K. S. Mani, A rhodamine-conjugated fluorescent and colorimetric receptor for the detection of Cu2+ ions: environmental utility and smartphone integration, Anal. Methods, 2025, 17(6), 1389–1400 RSC
.
- D. S. Ali, R. O. Hassan, H. O. Othman, H. T. Taha, A. M. Khaneghah and S. Smaoui, Revolutionizing detection: Smartphone-powered colorimetry for the drugs and food analysis, Microchem. J., 2024, 205, 111228 CrossRef CAS
.
- S. Kanchi, M. I. Sabela, P. S. Mdluli and K. Bisetty, Smartphone based bioanalytical and diagnosis applications: A review, Biosens. Bioelectron., 2018, 102, 136–149 CrossRef CAS PubMed
.
- R. Rajamanikandan, M. Ilanchelian and H. Ju, Smartphone-enabled colorimetric visual quantification of highly hazardous trivalent chromium ions in environmental waters and catalytic reduction of p-nitroaniline by thiol-functionalized gold nanoparticles, Chemosphere, 2023, 340, 139838 CrossRef CAS PubMed
.
- R. Rajamanikandan, K. Shanmugaraj, M. Ilanchelian and H. Ju, Cysteamine-decorated gold nanoparticles for plasmon-based colorimetric on-site sensors for detecting cyanide ions using the smart-phone color ratio and for catalytic reduction of 4-nitrophenol, Chemosphere, 2023, 316, 137836 CrossRef CAS PubMed
.
- A. Zhou, J. Chen, X. Dong, P. Bai, Q. Zhu, H. M. Meng and Z. Li, Bimetallic CuRu nanozymes for colorimetric and smartphone-assisted rapid visual hypoxanthine biosensing in serum samples, Microchim. Acta, 2025, 192(3), 159 CrossRef CAS PubMed
.
- Q. Yang, X. Cui, Y. Qin, T. Lei, Y. He and G. Song, Cu nanoclusters decorated Ti3C2 nanosheets composite with tetraenzyme mimic activities and the application for smartphone-assisted detection of hypoxanthine, Anal. Chim. Acta, 2022, 1232, 340494 CrossRef CAS PubMed
.
- Q. Yang, Y. Qin, T. Lei, Y. Zhong, X. Cui, Y. He and G. Song, Co(OH)2/MXene-Ti3C2 nanocomposites with triple-enzyme mimic activities as hydrogel sensing platform for on-site detection of hypoxanthine, Microchim. Acta, 2022, 189(12), 481 CrossRef CAS PubMed
.
- J. Fei, W. Yang, Y. Dai, W. Xu, H. Fan, Y. Zheng and X. Zhou, A biosensor based on Fe3O4@ MXene-Au nanocomposites with high peroxidase-like activity for colorimetric and smartphone-based detection of glucose, Microchim. Acta, 2023, 190(8), 336 CrossRef CAS PubMed
.
- S. E. Ekerim, N. Taşci and M. F. Demirkan, Determination of glyphosate with a novel optic membrane sensor, Food Chem., 2025, 143361 CrossRef CAS PubMed
.
- D. Liao, Y. Zhao, Y. Zhou, Y. Yi, W. Weng and G. Zhu, Colorimetric detection of organophosphorus pesticides based on Nb2CTx MXene self-reducing PdPt nanozyme integrated with hydrogel and smartphone, J. Food Meas. Charact., 2024, 18(11), 9223–9232 CrossRef
.
- M. Lin, Z. Gao, Z. Qian, Y. Deng, Y. Chen, Y. Wang and X. Li, Ultrasensitive Ti3C2Tx@ Pt-Based Immunochromatography with Catalytic Amplification and a Dual Signal for the Detection of Chloramphenicol in Animal-Derived Foods, Foods, 2024, 13(9), 1416 CrossRef CAS PubMed
.
- L. Zhang, A. Li, M. Shen, Z. Zhang, R. Wufuer and D. Wang, Palladium–platinum bimetallic modified MXene nanozyme for highly sensitive detection of active substances with acetylcholinesterase inhibitory effect, Talanta, 2025, 282, 127020 CrossRef CAS PubMed
.
- K. Chen, S. Fu, C. Jin, F. Guo, Y. He, Q. Ren and X. Wang, Smartphone-Enabled Fluorescence and Colorimetric Platform for the On-Site Detection of Hg2+ and Cl− Based on the Au/Cu/Ti3C2 Nanosheets, Molecules, 2023, 28(14), 5355 CrossRef CAS PubMed
.
- H. Li, S. Zhao, Z. Wang and F. Li, Engineering a two-dimensional metal-carbon nanozyme-based portable paper-based colorimetric chip for onsite and visual analysis of pyrophosphate, Talanta, 2024, 278, 126490 CrossRef CAS PubMed
.
- T. Jiang, P. Ju, F. Bi, J. Chi, S. Wen, F. Jiang and Z. Chi, Target-induced enzymatic cleavage cycle amplification reaction-gated organic photoelectrochemical transistor biosensor for rapid detection of okadaic acid, Biosens. Bioelectron., 2025, 267, 116745 CrossRef CAS PubMed
.
- J. Chi, P. Ju, F. Bi, S. Wen, Z. Xiang, J. Chen and M. Qiu, A smartphone-assisted ultrasensitive colorimetric aptasensor based on DNA-encoded porous MXene nanozyme for visual detection of okadaic acid, Food Chem., 2025, 464, 141776 CrossRef CAS PubMed
.
- D. K. Mal, H. Pal and G. Chakraborty, A comprehensive review on recent advances in fluorescence-based bio-analytes sensing, TrAC, Trends Anal. Chem., 2024, 171, 117493 CrossRef CAS
.
- D. Wu, A. C. Sedgwick, T. Gunnlaugsson, E. U. Akkaya, J. Yoon and T. D. James, Fluorescent chemosensors: the past, present and future, Chem. Soc. Rev., 2017, 46(23), 7105–7123 RSC
.
- R. Rajamanikandan and M. Ilanchelian, Highly selective and sensitive biosensing of dopamine based on glutathione coated silver nanoclusters enhanced fluorescence, New J. Chem., 2017, 41(24), 15244–15250 Search PubMed
.
- D. S. Chumakov, S. S. Evstigneeva and O. I. Guliy, Luminescent Nanozyme-Based Sensors for Bacteria Detection, Appl. Biochem. Microbiol., 2024, 60(6), 1022–1034 CrossRef CAS
.
- J. Zhang and J. Liu, Nanozyme-based luminescence detection, Luminescence, 2020, 35(8), 1185–1194 CrossRef CAS PubMed
.
- M. Zhou, Z. Diwu, N. Panchuk-Voloshina and R. P. Haugland, A stable nonfluorescent derivative of resorufin for the fluorometric determination of trace hydrogen peroxide: applications in detecting the activity of phagocyte NADPH oxidase and other oxidases, Anal. Biochem., 1997, 253(2), 162–168 Search PubMed
.
- X. Yang and E. Wang, A nanoparticle autocatalytic sensor for Ag+ and Cu2+ ions in aqueous solution with high sensitivity and selectivity and its application in test paper, Anal. Chem., 2011, 83(12), 5005–5011 Search PubMed
.
- J. Sun, B. Wang, X. Zhao, Z. J. Li and X. Yang, Fluorescent and colorimetric dual-readout assay for inorganic pyrophosphatase with Cu2+-triggered oxidation of o-phenylenediamine, Anal. Chem., 2016, 88(2), 1355–1361 CrossRef CAS PubMed
.
- T. Lin, Y. Qin, Y. Huang, R. Yang, L. Hou, F. Ye and S. Zhao, A label-free fluorescence assay for hydrogen peroxide and glucose based on the bifunctional MIL-53 (Fe) nanozyme, Chem. Commun., 2018, 54(14), 1762–1765 RSC
.
- S. Chen, Z. Li, Z. Huang and Q. Jia, Investigation of efficient synergistic and protective effects of chitosan on copper nanoclusters: Construction of highly active and stable nanozyme for colorimetric and fluorometric dual-signal biosensing, Sens. Actuators, B, 2021, 332, 129522 CrossRef CAS
.
- D. Li, S. L. Garisto, P. J. J. Huang, J. Yang, B. Liu and J. Liu, Fluorescent detection of fluoride by CeO2 nanozyme oxidation of Amplex red, Inorg. Chem. Commun., 2019, 106, 38–42 CrossRef CAS
.
- C. Song, W. Zhao, H. Liu, W. Ding, L. Zhang, J. Wang and C. Yao, Two-dimensional FeP@ C nanosheets as a robust oxidase mimic for fluorescence detection of cysteine and Cu 2+, J. Mater. Chem. B, 2020, 8(33), 7494–7500 Search PubMed
.
- X. Xu, X. Zou, S. Wu, L. Wang, X. Niu, X. Li and M. Lan, In situ formation of fluorescent polydopamine catalyzed by peroxidase-mimicking FeCo-LDH for pyrophosphate ion and pyrophosphatase activity detection, Anal. Chim. Acta, 2019, 1053, 89–97 Search PubMed
.
- R. Govindaraju and J. Kim, MXene-enabled fluorescent sensing systems: recent advances in biomolecule detection, Microchem. J., 2025, 218, 115082 CrossRef CAS
.
- W. Song, B. Zhao, C. Wang, Y. Ozaki and X. Lu, Functional nanomaterials with unique enzyme-like characteristics for sensing applications, J. Mater. Chem. B, 2019, 7(6), 850–875 RSC
.
- L. Han, H. Zhang, D. Chen and F. Li, Protein-directed metal oxide nanoflakes with tandem enzyme-like characteristics: colorimetric glucose sensing based on one-pot enzyme-free cascade catalysis, Adv. Funct. Mater., 2018, 28(17), 1800018 Search PubMed
.
- D. Wang, D. Jana and Y. Zhao, Metal-organic framework derived nanozymes in biomedicine, Acc. Chem. Res., 2020, 53(7), 1389–1400 CrossRef CAS PubMed
.
- J. Chi, P. Ju, F. Bi, S. Wang, T. Jiang, S. Wen and M. Qiu, MXene@ MnIn2S4-Gated Organic Photoelectrochemical Transistors with Nanozyme-Mediated Multiple Quenching Effects for Ultrasensitive Detection of Okadaic Acid, Adv. Funct. Mater., 2024, 34(45), 2407201 Search PubMed
.
- U. Amara, I. Hussain, M. Ahmad, K. Mahmood and K. Zhang, 2D MXene-based biosensing: a review, Small, 2023, 19(2), 2205249 CrossRef CAS PubMed
.
- K. Dhara and R. M. Debiprosad, Review on nanomaterials-enabled electrochemical sensors for ascorbic acid detection, Anal. Biochem., 2019, 586, 113415 CrossRef CAS PubMed
.
- J. Huang, C. Shen, H. Gu, G. Wang, P. Zhou, X. Liu and Z. Chen, Cu Nanoparticles/N-Doped Ti3C2Tx-Based Fluorescence Sensor for Detecting Ascorbic Acid, ACS Sustainable Chem. Eng., 2023, 11(49), 17472–17481 Search PubMed
.
- A. Rohde, J. A. Hammerl, I. Boone, W. Jansen, S. Fohler, G. Klein and S. Al Dahouk, Overview of validated alternative methods for the detection of foodborne bacterial pathogens, Trends Food Sci. Technol., 2017, 62, 113–118 CrossRef CAS
.
- S. Wang, J. Hu, S. Xiao, M. Wang, J. Yu, Z. Jia and N. Gan, Fluorescent/electrochemical dual-signal response biosensing strategy mediated by DNAzyme-ferrocene-triggered click chemistry for simultaneous rapid screening and quantitative detection of Vibrio parahaemolyticus, Sens. Actuators, B, 2023, 380, 133393 CrossRef CAS
.
- B. A. Al-Jaal, M. Jaganjac, A. Barcaru, P. Horvatovich and A. Latiff, Aflatoxin, fumonisin, ochratoxin, zearalenone and deoxynivalenol biomarkers in human biological fluids: A systematic literature review, 2001–2018, Food Chem. Toxicol., 2019, 129, 211–228 CrossRef CAS PubMed
.
- H. F. Hassan, R. Kordahi, H. Dimassi, A. El Khoury, R. Daou, N. Alwan and L. Karam, Aflatoxin B1 in Rice: Effects of storage duration, grain type and size, production site, and season, J. Food Prot., 2022, 85(6), 938–944 CrossRef CAS PubMed
.
- Y. Kong, Y. Zhu, J. Song, Q. Liu, L. Song, X. Fei and X. Li, A novel multimode biosensor for sensitive detection of AFB1 in food based on Mxenes nano enzymes, Food Chem., 2023, 426, 136645 Search PubMed
.
- Y. Kong, Z. Li, Q. Liu, J. Song, Y. Zhu, J. Lin and X. Li, Artificial neural network-facilitated V2C MNs-based colorimetric/fluorescence dual-channel biosensor for highly sensitive detection of AFB1 in peanut, Talanta, 2024, 266, 125056 CrossRef CAS PubMed
.
- N. Wang, L. Xu, S. Liu, M. He and L. Wang, An interferometric optical fiber sensor based on ion-imprinted sodium alginate for Pb(II) detection, Opt. Fiber Technol., 2025, 90, 104053 Search PubMed
.
- F. Farahmandzadeh, K. Kermanshahian, E. Molahosseini, M. Molaei and M. Karimipour, Highly fluorescent CdTe/ZnSe quantum dot-based “turn-off” sensor for the on-site rapid detection of Lead ions in aqueous medium, Spectrochim. Acta, Part A, 2025, 324, 124914 CrossRef CAS PubMed
.
- Z. Suo, L. Zhang, Z. Zhang, R. Liang, H. Shen, X. Chen and H. Jin, A bifunctional MXene@ PtPd NPs cascade DNAzyme-mediated fluorescence/colorimetric dual-mode biosensor for Pb2+ determination, Food Chem., 2025, 464, 141845 Search PubMed
.
- D. Liu, W. Chen, J. Wei, X. Li, Z. Wang and X. Jiang, A highly sensitive, dual-readout assay based on gold nanoparticles for organophosphorus and carbamate pesticides, Anal. Chem., 2012, 84(9), 4185–4191 CrossRef CAS PubMed
.
- S. Dai, M. Hu, W. Zhang and Z. Lei, Selective colorimetric detection of carbosulfan based on its hydrolysis behavior and Ti3C2/AuPt nanozyme, Anal. Chim. Acta, 2025, 1336, 343519 Search PubMed
.
- Z. Lei, P. Lei, J. Guo and Z. Wang, Recent advances in nanomaterials-based optical and electrochemical aptasensors for detection of cyanotoxins, Talanta, 2022, 248, 123607 CrossRef CAS PubMed
.
- O. D. Hendrickson, E. A. Zvereva, A. V. Zherdev and B. B. Dzantiev, Ultrasensitive lateral flow immunoassay of phycotoxin microcystin-LR in seafood based on magnetic particles and peroxidase signal amplification, Food Control, 2022, 133, 108655 CrossRef CAS
.
- J. Guo, G. Wang, J. Zou and Z. Lei, DNA controllable peroxidase-like activity of Ti3C2 nanosheets for colorimetric detection of microcystin-LR, Anal. Bioanal. Chem., 2023, 415(17), 3559–3569 CrossRef CAS PubMed
.
- R. Eivazzadeh-Keihan, Z. Saadatidizaji, A. Maleki, M. D. L. Guardia, M. Mahdavi, S. Barzegar and S. Ahadian, Recent progresses in development of biosensors for thrombin detection, Biosensors, 2022, 12(9), 767 CrossRef CAS PubMed
.
- A. Bini, M. Minunni, S. Tombelli, S. Centi and M. Mascini, Analytical performances of aptamer-based sensing for thrombin detection, Anal. Chem., 2007, 79(7), 3016–3019 CrossRef CAS PubMed
.
- M. Li, X. Peng, Y. Han, L. Fan, Z. Liu and Y. Guo, Ti3C2 MXenes with intrinsic peroxidase-like activity for label-free and colorimetric sensing of proteins, Microchem. J., 2021, 166, 106238 CrossRef CAS
.
- L. Zhao, J. Xu, L. Xiong, S. Wang, C. Yu, J. Lv and J. M. Lin, Recent development of chemiluminescence for bioanalysis, TrAC, Trends Anal. Chem., 2023, 117213 CrossRef CAS
.
- H. Zhu, X. Huang, Y. Deng, H. Chen, M. Fan and Z. Gong, Applications of nanomaterial-based chemiluminescence sensors in environmental analysis, TrAC, Trends Anal. Chem., 2023, 158, 116879 CrossRef CAS
.
- Z. Zhang, S. Zhang and X. Zhang, Recent developments and applications of chemiluminescence sensors, Anal. Chim. Acta, 2005, 541(1–2), 37–46 Search PubMed
.
- K. Deshpande and L. Kanungo, Chemiluminescence and Fluorescence Biosensors for food application: A Review, Sens Actuators Rep., 2023, 5, 100137 CrossRef
.
- H. Y. Li, S. Q. Zhang, M. L. Chen and J. H. Wang, Lumi-HOF@ Tb as probes for multiple ratiometric fluorescence and chemiluminescence sensing of α-glucosidase, Anal. Chem., 2022, 94(44), 15448–15455 CrossRef CAS PubMed
.
- B. R. Radziszewski, Untersuchungen über hydrobenzamid, amarin und lophin, Ber. Dtsch. Chem. Ges., 1877, 10(1), 70–75 Search PubMed
.
- H. O. Albrecht, Über die chemiluminescenz des aminophthalsäurehydrazids, Z. Phys. Chem., 1928, 136(1), 321–330 CrossRef
.
- K. Gleu and W. Petsch, Die chemiluminescent der dimethyl-diacridyliumsalze, Angew. Chem., Int. Ed., 1935, 48(3), 57–59 CrossRef CAS
.
- W. Adam, D. V. Kazakov and V. P. Kazakov, Singlet-oxygen chemiluminescence in peroxide reactions, Chem. Rev., 2005, 105(9), 3371–3387 CrossRef CAS PubMed
.
- M. Yang, J. Huang, J. Fan, J. Du, K. Pu and X. Peng, Chemiluminescence for bioimaging and therapeutics: recent advances and challenges, Chem. Soc. Rev., 2020, 49(19), 6800–6815 RSC
.
- F. Manea, F. B. Houillon, L. Pasquato and P. Scrimin, Nanozymes: gold-nanoparticle-based transphosphorylation catalysts, Angew. Chem., Int. Ed., 2004, 43, 6165–6169 CrossRef CAS PubMed
.
- L. Gao, J. Zhuang, L. Nie, J. Zhang, Y. Zhang, N. Gu and X. Yan, The intrinsic peroxidase-like activity of ferromagnetic nanoparticles, Nat. Nanotechnol., 2007, 2(9), 577–583 Search PubMed
.
- S. N. A. Shah, A. H. Shah, X. Dou, M. Khan, L. Lin and J. M. Lin, Radical-triggered chemiluminescence of phenanthroline derivatives: an insight into radical–aromatic interaction, ACS Omega, 2019, 4(12), 15004–15011 Search PubMed
.
- J. H. Choi, D. H. Lee and W. Y. Lee, Enhanced cathodic electrogenerated chemiluminescence of luminol at a MXene–Nafion composite-modified electrode in neutral aqueous solution, J. Electroanal. Chem., 2023, 936, 117389 CrossRef
.
- Z. Shi, T. Wu, W. Feng, B. Hu, X. Yan and X. Zheng, Enhanced luminol chemiluminescence with oxidase-like properties of prussian blue/MXene nanocomposite without H2O2 for the sensitive detection of uric acid, Microchem. J., 2024, 206, 111455 Search PubMed
.
- R. Rajamanikandan, M. Ilanchelian and H. Ju, Highly selective uricase-based quantification of uric acid using hydrogen peroxide sensitive poly-(vinylpyrrolidone) templated copper nanoclusters as a fluorescence probe, Chem, 2023, 11(5), 268 CAS
.
- K. S. Mani, R. Rajamanikandan, M. Ilanchelian, N. Muralidharan, M. Jothi and S. P. Rajendran, Smartphone-assisted quinoline-hemicyanine-based fluorescent probe for the selective detection of glutathione and the application in living cells, Spectrochim. Acta, Part A, 2020, 243, 118809 Search PubMed
.
- X. Zheng, Z. Shi, C. Han, H. Mu, S. Cheng and X. Yan, Convenient in situ self-assembled formation of dual-functional Ag/MXene nanozymes for efficient chemiluminescence sensing, Anal. Methods, 2024, 16(48), 8324–8332 RSC
.
- Y. Fu, X. Teng and C. Lu, Iridium-based electrochemiluminescence systems and their application in bioanalysis, TrAC, Trends Anal. Chem., 2023, 167, 117273 CrossRef CAS
.
- M. M. Richter, Electrochemiluminescence (ecl), Chem. Rev., 2004, 104(6), 3003–3036 CrossRef CAS PubMed
.
- L. Li, Y. Chen and J. J. Zhu, Recent advances in electrochemiluminescence analysis, Anal. Chem., 2017, 89(1), 358–371 CrossRef CAS PubMed
.
- Z. Liu, W. Qi and G. Xu, Recent advances in electrochemiluminescence, Chem. Soc. Rev., 2015, 44(10), 3117–3142 Search PubMed
.
-
A. J. Bard, Electrogenerated Chemiluminescence, Marcel Dekker, New York, 2004 Search PubMed
.
- N. Arab, M. Hosseini and G. Xu, Emerging trends and recent advances in MXene/MXene-based nanocomposites toward electrochemiluminescence sensing and biosensing, Biosens. Bioelectron., 2024, 116623 CrossRef CAS PubMed
.
- L. Gao and C. He, Advances in MXene-based luminescence sensing strategies, Anal. Methods, 2024, 16, 1718–1735 RSC
.
- Y. Fang, X. Yang, T. Chen, G. Xu, M. Liu, J. Liu and Y. Xu, Two-dimensional titanium carbide (MXene)-based solid-state electrochemiluminescent sensor for label-free single-nucleotide mismatch discrimination in human urine, Sens. Actuators, B, 2018, 263, 400–407 Search PubMed
.
- S. P. Hong, N. A. Keasberry and M. U. Ahmed, Development of a gliadin immunosensor incorporating gold nanourchin, molybdenum disulfide, titanium dioxide, and Nafion for enhanced electrochemiluminescence, Microchem. J., 2023, 193, 109059 CrossRef CAS
.
- J. Adhikari, M. Rizwan and M. U. Ahmed, Development of a label-free electrochemiluminescence biosensor for the sensitive detection of porcine gelatin using carbon nanostructured materials, Sens. Diagn., 2022, 1(5), 968–976 RSC
.
- I. Tomac, M. Šeruga and J. Labuda, Evaluation of antioxidant activity of chlorogenic acids and coffee extracts by an electrochemical DNA-based biosensor, Food Chem., 2020, 325, 126787 CrossRef CAS PubMed
.
- T. Zhan, Y. Su, W. Lai, Z. Chen and C. Zhang, A dry chemistry-based ultrasensitive electrochemiluminescence immunosensor for sample-to-answer detection of Cardiac Troponin I, Biosens. Bioelectron., 2022, 214, 114494 CrossRef CAS PubMed
.
- F. Kareem, Y. F. C. Chau and M. U. Ahmed, Nb2CTx-supported bimetallic NPs@ ZIF-8 nanohybrid as ECL signal amplifier and peroxidase mimics for chromogranin a immunosensing in human serum and saliva, Int. J. Biol. Macromol., 2025, 287, 138476 CrossRef CAS PubMed
.
- M. Pietrzak and P. Ivanova, Bimetallic and multimetallic nanoparticles as nanozymes, Sens. Actuators, B, 2021, 336, 129736 Search PubMed
.
- Y. Lu, X. Li, S. Shi, X. Liu, L. Jia, L. Shang and H. Wang, Tungsten-based polyoxometalate nanoclusters with remarkable reactive oxygen species-scavenging activity efficiently quenched luminol-based electrochemiluminescence for sensitive detection of Her-2, Microchim. Acta, 2024, 191(1), 21 Search PubMed
.
- F. Arshad, N. F. Mohd-Naim, R. Chandrawati, D. Cozzolino and M. U. Ahmed, Nanozyme-based sensors for detection of food biomarkers: a review, RSC Adv., 2022, 12(40), 26160–26175 RSC
.
- H. Riazi, G. Taghizadeh and M. Soroush, MXene-based nanocomposite sensors, ACS Omega, 2021, 6(17), 11103–11112 CrossRef CAS PubMed
.
- N. H. Solangi, S. A. Mazari, N. M. Mubarak, R. R. Karri, N. Rajamohan and D. V. N. Vo, Recent trends in MXene-based material for biomedical applications, Environ. Res., 2023, 222, 115337 CrossRef CAS PubMed
.
- B. Wang, S. M. Khoshfetrat and H. Mohamadimanesh, Peroxidase-like manganese oxide nanoflowers-delaminated Ti3C2 MXene for ultrasensitive dual-mode and real-time detection of H2O2 released from cancer cells, Microchem. J., 2024, 207, 111796 Search PubMed
.
- J. Wang, R. Hong, Z. Yang, X. Meng, R. Wu, Z. Liu and C. Li, Ultrasensitive Electrochemiluminescence Biosensor with ZIF-67@ MXene as an Efficient Co-Reaction Accelerator and Plasmonic Nanozyme as a Smart Signal Amplification Probe, Small, 2024, 20(50), 2404330 CrossRef CAS PubMed
.
-
J. Liang, X. Guan, G. Bao, Y. Yao and X. Zhong, Molecular subtyping of small cell lung cancer, In Seminars in Cancer Biology, Academic Press, 2022, vol. 86, pp. 450–462 Search PubMed
.
- M. Chen, Y. Wang, H. Su, L. Mao, X. Jiang, T. Zhang and X. Dai, Three-dimensional electrochemical DNA biosensor based on 3D graphene-Ag nanoparticles for sensitive detection of CYFRA21-1 in non-small cell lung cancer, Sens. Actuators, B, 2018, 255, 2910–2918 Search PubMed
.
- X. Ren, M. Shao, Z. Xie, X. Li, H. Ma, D. Fan and Q. Wei, A Co-Reactive Immunosensor Based on Ti3C2Tx MXene@ TiO2–MoS2 Hybrids Promoting luminol@ Au@ Ni–Co NCs Electrochemiluminescence for CYFRA 21–1 Detection, ACS Sens., 2024, 9(4), 1992–1999 Search PubMed
.
- F. Momeni, S. M. Khoshfetrat, H. Bagheri and K. Zarei, Ti3C2 MXene-based nanozyme as a coreaction accelerator for enhancing electrochemiluminescence of glucose biosensing, Biosens. Bioelectron., 2024, 250, 116078 CrossRef CAS PubMed
.
- J. Chen, J. Zheng, K. Zhao, A. Deng and J. Li, Electrochemiluminescence resonance energy transfer system between non-toxic SnS2 quantum dots and ultrathin Ag@ Au nanosheets for chloramphenicol detection, Chem. Eng. J., 2020, 392, 123670 Search PubMed
.
- D. Jiang, M. Wei, X. Du, M. Qin, X. Shan and Z. Chen, One-pot synthesis of ZnO quantum dots/N-doped Ti3C2 MXene: Tunable nitrogen-doping properties and efficient electrochemiluminescence sensing, Chem. Eng. J., 2022, 430, 132771 Search PubMed
.
- R. Joseph, S. K. Rani and N. Vasimalai, A novel molecular imprinted polymer grafted on N, S co-doped carbon quantum dots-based fluorescence sensor for chloramphenicol in food and clinical samples, Microchem. J., 2024, 207, 111675 CrossRef
.
- Z. Zhang, D. Jiang, H. Ding, X. Shan, W. Wang, H. Shiigi and Z. Chen, Cobaltous sulfide/S-doped Ti3C2 MXene nanocomposites with co-rection acceleration effect for high-performance luminol electrochemiluminescence sensing by coupling micro-nano bubbles-mediated amplification strategy, Chem. Eng. J., 2023, 475, 146182 CrossRef CAS
.
- X. Liu, Y. Li, L. He, Y. Feng, H. Tan, X. Chen and W. Yang, Simultaneous detection of multiple neuroendocrine tumor markers in patient serum with an ultrasensitive and antifouling electrochemical immunosensor, Biosens. Bioelectron., 2021, 194, 113603 CrossRef CAS PubMed
.
- D. R. Iyer, J. Venkatraman, E. Tanguy, N. Vitale and N. R. Mahapatra, Chromogranin A and its derived peptides: potential regulators of cholesterol homeostasis, Cell. Mol. Life Sci., 2023, 80(9), 271 CrossRef CAS PubMed
.
- A. M. Arjun, N. Shabana, M. Ankitha and P. A. Rasheed, Electrochemical deposition of Prussian blue on Nb2CTx MXene modified carbon cloth for the non-enzymatic electrochemical detection of hydrogen peroxide, Microchem. J., 2023, 185, 108301 CrossRef
.
- Z. Otgonbayar and W. C. Oh, Comprehensive and multi-functional MXene based sensors: An updated review, FlatChem, 2023, 100524 CrossRef CAS
.
- R. Singh, R. Gupta, D. Bansal, R. Bhateria and M. Sharma, A review on recent trends and future developments in electrochemical sensing, ACS Omega, 2024, 9(7), 7336–7356 CAS
.
- A. M. Mohamed, F. H. Fouad, G. R. Fayek, K. M. El Sayed, M. N. Ahmed, R. Z. Mahmoud and R. M. El Nashar, Recent advances in electrochemical sensors based on nanomaterials for detection of red dyes in food products: A review, Food Chem., 2024, 435, 137656 Search PubMed
.
- E. Bakker and M. Telting-Diaz, Electrochemical sensors, Anal. Chem., 2002, 74(12), 2781–2800 CrossRef CAS PubMed
.
- N. Jadon, R. Jain, S. Sharma and K. Singh, Recent trends in electrochemical sensors for multianalyte detection–A review, Talanta, 2016, 161, 894–916 CrossRef CAS PubMed
.
- M. Hasanzadeh, N. Shadjou and M. de la Guardia, Early stage screening of breast cancer using electrochemical biomarker detection, TrAC, Trends Anal. Chem., 2017, 91, 67–76 CrossRef CAS
.
- B. Mohan, S. Kumar, H. Xi, S. Ma, Z. Tao, T. Xing and P. Ren, Fabricated Metal-Organic Frameworks (MOFs) as luminescent and electrochemical biosensors for cancer biomarkers detection, Biosens. Bioelectron., 2022, 197, 113738 CrossRef CAS PubMed
.
- A. Thamilselvan and M. I. Kim, Recent advances on nanozyme-based electrochemical biosensors for cancer biomarker detection, TrAC, Trends Anal. Chem., 2024, 117815 CrossRef CAS
.
- B. S. Munge, T. Stracensky, K. Gamez, D. DiBiase and J. F. Rusling, Multiplex immunosensor arrays for electrochemical detection of cancer biomarker proteins, Electroanalysis, 2016, 28(11), 2644–2658 CrossRef CAS PubMed
.
- H. L. Chia, C. C. Mayorga-Martinez, N. Antonatos, Z. Sofer, J. J. Gonzalez-Julian, R. D. Webster and M. Pumera, MXene titanium carbide-based biosensor: strong dependence of exfoliation method on performance, Anal. Chem., 2020, 92(3), 2452–2459 CrossRef CAS PubMed
.
- C. V. Raju, C. H. Cho, G. M. Rani, V. Manju, R. Umapathi, Y. S. Huh and J. P. Park, Emerging insights into the use of carbon-based nanomaterials for the electrochemical detection of heavy metal ions, Coord. Chem. Rev., 2023, 476, 214920 CrossRef
.
- R. Rajamanikandan and M. Ilanchelian, β-cyclodextrin functionalised silver nanoparticles as a dual colorimetric probe for ultrasensitive detection of Hg2+ and S2− ions in environmental water samples, Mater. Today Commun., 2018, 15, 61–69 CrossRef CAS
.
- T. Liu, R. Zhou, K. Wu and G. Zhu, Colorimetric method transforms into highly sensitive homogeneous voltammetric sensing strategy for mercury ion based on mercury-stimulated Ti3C2Tx MXene nanoribbons@ gold nanozyme activity, Anal. Chim. Acta, 2023, 1250, 340975 CrossRef CAS PubMed
.
- A. Mukhopadhyay, S. Duttagupta and A. Mukherjee, Emerging organic contaminants in global community drinking water sources and supply: A review of occurrence, processes and remediation, J. Environ. Chem. Eng., 2023, 10(3), 107560 CrossRef
.
- K. Sasikumar, R. Rajamanikandan and H. Ju, A high-performance NiMoO4/g-C3N4 direct Z-scheme heterojunction photocatalyst for the degradation of organic pollutants, Surf. Interfaces, 2023, 42, 103389 CrossRef CAS
.
- M. S. Samuel, K. Jeyaram, S. Datta, N. Chandrasekar, R. Balaji and E. Selvarajan, Detection, contamination, toxicity, and prevention methods of ochratoxins: An update review, J. Agric. Food Chem., 2021, 69(46), 13974–13989 CrossRef CAS PubMed
.
- X. Zhang, F. Wang, H. Zhi, J. Zhao, P. Wan and L. Feng, Electrochemical “signal on/off” paper-based aptasensor for ochratoxin A detection based on MXene-Au and Pt@ NiCo-LDH-catalyzed signal amplification, Sens. Actuators, B, 2022, 368, 132161 CrossRef CAS
.
- B. Zhu, D. An, Z. Bi, W. Liu, W. Shan, Y. Li and M. Qiu, Two-dimensional nitrogen-doped Ti3C2 promoted catalysis performance of silver nanozyme for ultrasensitive detection of hydrogen peroxide, ChemElectroChem, 2022, 9(10), e202200050 CrossRef CAS
.
- S. Fu, Y. Zhu, Y. Zhang, M. Zhang, Y. Zhang, L. Qiao and D. Wang, Recent advances in carbon nanomaterials-based electrochemical sensors for phenolic compounds detection, Microchem. J., 2021, 171, 106776 CrossRef CAS
.
- R. Yu, J. Xue, Y. Wang, J. Qiu, X. Huang, A. Chen and J. Xue, Novel Ti3C2Tx MXene nanozyme with manageable catalytic activity and application to electrochemical biosensor, J. Nanobiotechnol., 2022, 20(1), 119 CrossRef CAS PubMed
.
- L. Wu, X. Lu, Z. S. Wu, Y. Dong, X. Wang, S. Zheng and J. Chen, 2D transition metal carbide MXene as a robust biosensing platform for enzyme immobilization and ultrasensitive detection of phenol, Biosens. Bioelectron., 2018, 107, 69–75 CrossRef CAS PubMed
.
- H. Karimi-Maleh, F. Karimi, M. Alizadeh and A. L. Sanati, Electrochemical sensors, a bright future in the fabrication of portable kits in analytical systems, Chem. Rec., 2020, 20(7), 682–692 CrossRef CAS PubMed
.
- X. Zhu, L. Lin, R. Wu, Y. Zhu, Y. Sheng, P. Nie and Y. Wen, Portable wireless intelligent sensing of ultra-trace phytoregulator α-naphthalene acetic acid using self-assembled phosphorene/Ti3C2-MXene nanohybrid with high ambient stability on laser-induced porous graphene as nanozyme flexible electrode, Biosens. Bioelectron., 2021, 179, 113062 CrossRef CAS PubMed
.
- M. Bhattu, M. Verma and D. Kathuria, Recent advancements in the detection of organophosphate pesticides: a review, Anal. Methods, 2021, 13(38), 4390–4428 RSC
.
- T. O. Ajiboye, P. O. Oladoye, C. A. Olanrewaju and G. O. Akinsola, Organophosphorus pesticides: Impacts, detection and removal strategies, Environ. Nanotechnol., Monit. Manage., 2022, 17, 100655 CAS
.
- L. Wang, N. Wu, L. Wang, Y. Song and G. Ma, Accurate detection of organophosphorus pesticides based on covalent organic framework nanofiber with a turn-on strategy, Sens. Actuators, B, 2022, 372, 132608 CrossRef CAS
.
- L. Yu, J. Chang, X. Zhuang, H. Li, T. Hou and F. Li, Two-dimensional cobalt-doped Ti3C2 MXene nanozyme-mediated homogeneous electrochemical strategy for pesticides assay based on in situ generation of electroactive substances, Anal. Chem., 2022, 94(8), 3669–3676 CrossRef CAS PubMed
.
- F. Zhao, Y. Yao, C. Jiang, Y. Shao, D. Barceló, Y. Ying and J. Ping, Self-reduction bimetallic nanoparticles on ultrathin MXene nanosheets as functional platform for pesticide sensing, J. Hazard. Mater., 2020, 384, 121358 CrossRef CAS PubMed
.
- S. Kumar and R. Singh, Recent optical sensing technologies for the detection of various biomolecules, Opt. Laser Technol., 2021, 134, 106620 CrossRef CAS
.
- J. Zhang, Y. Li, S. Duan and F. He, Highly electrically conductive two-dimensional Ti3C2 Mxenes-based 16S rDNA electrochemical sensor for detecting Mycobacterium tuberculosis, Anal. Chim. Acta, 2020, 1123, 9–17 CrossRef CAS PubMed
.
- Y. Chen, Y. Gu, X. Yi, H. Huang, Y. Li, B. Yang and L. Bai, Conductive nano-gold self-assembled MXene@ hemin with high catalytic activity achieved by strong metal-support interactions: A powerful nanozyme for development of electrochemical aptasensor in tuberculosis diagnosis, Chem. Eng. J., 2023, 466, 143112 CrossRef CAS
.
- X. Zheng, Z. Hu, S. Gao, Z. Li, J. Chen, G. Zhang and W. Liu, One-pot assay using a target-driven split aptamer recognition and assembly strategy for convenient and rapid detection of gliotoxin, Food Chem., 2024, 454, 139738 CrossRef CAS PubMed
.
- H. Wang, H. Li, Y. Huang, M. Xiong, F. Wang and C. Li, A label-free electrochemical biosensor for highly sensitive detection of gliotoxin based on DNA nanostructure/MXene nanocomplexes, Biosens. Bioelectron., 2019, 142, 111531 CrossRef CAS PubMed
.
- J. Chen, P. Tong, L. Huang, Z. Yu and D. Tang, Ti3C2 MXene nanosheet-based capacitance immunoassay with tyramine-enzyme repeats to detect prostate-specific antigen on interdigitated micro-comb electrode, Electrochim. Acta, 2019, 319, 375–381 CrossRef CAS
.
- G. Jiang, H. Liu, J. Liu, L. E. Liu, Y. Li, L. Xue and R. Yang, Engineering of multifunctional carbon nanodots-decorated plasmonic Au@ Ag nanoenzymes for photoelectrochemical biosensing of microRNA-155, Sens. Actuators, B, 2022, 360, 131653 CrossRef CAS
.
- H. Wang, J. Sun, L. Lu, X. Yang, J. Xia, F. Zhang and Z. Wang, Competitive electrochemical aptasensor based on a cDNA-ferrocene/MXene probe for detection of breast cancer marker Mucin1, Anal. Chim. Acta, 2020, 1094, 18–25 CrossRef CAS PubMed
.
- R. Manikandan, M. J. Kim, H. G. Jang, A. Mugunthan, C. S. Kim, J. H. Yoon and S. C. Chang, Fabrication of bio-mimic nanozyme based on Mxene@ AuNPs and molecular imprinted poly (thionine) films for creatinine detection, Biosens. Bioelectron., 2025, 271, 117075 CrossRef CAS PubMed
.
- J. Perumal, Y. Wang, A. B. E. Attia, U. S. Dinish and M. Olivo, Towards a point-of-care SERS sensor for biomedical and agri-food analysis applications: A review of recent advancements, Nanoscale, 2021, 13(2), 553–580 RSC
.
- D. Graham, M. Moskovits and Z. Q. Tian, SERS–facts, figures and the future, Chem. Soc. Rev., 2017, 46(13), 3864–3865 RSC
.
- C. Liu, D. Xu, X. Dong and Q. Huang, A review: Research progress of SERS-based sensors for agricultural applications, Trends Food Sci. Technol., 2022, 128, 90–101 CrossRef CAS
.
- B. Sharma, R. R. Frontiera, A. I. Henry, E. Ringe and R. P. Van Duyne, SERS: Materials, applications, and the future, Mater. Today, 2012, 15(1–2), 16–25 CrossRef CAS
.
- M. Fleischmann, P. J. Hendra and A. J. McQuillan, Raman spectra of pyridine adsorbed at a silver electrode, Chem. Phys. Lett., 1974, 26(2), 163–166 CrossRef CAS
.
- A. Campion and P. Kambhampati, Surface-enhanced Raman scattering, Chem. Soc. Rev., 1998, 27(4), 241–250 RSC
.
- M. Shameer, K. V. Anand, J. B. Parambath, S. Columbus and H. Alawadhi, Direct detection of melamine in milk via surface-enhanced Raman scattering using gold-silver anisotropic nanostructures, Spectrochim. Acta, Part A, 2025, 327, 125412 CrossRef CAS PubMed
.
- A. M. Pisoschi and A. Pop, The role of antioxidants in the chemistry of oxidative stress: A review, Eur. J. Med. Chem., 2015, 97, 55–74 CrossRef CAS PubMed
.
- H. Xi, H. Gu, Y. Han, T. You, P. Wu, Q. Liu and P. Yin, Peroxidase-like single Fe atoms anchored on Ti3C2Tx MXene as surface enhanced Raman scattering substrate for the simultaneous discrimination of multiple antioxidants, Nano Res., 2023, 16(7), 10053–10060 CrossRef CAS
.
- V. G. Gayathri, R. Bartholomew, J. T. Chacko, J. Bayry and P. A. Rasheed, Non-Ti MXenes: new biocompatible and biodegradable candidates for biomedical applications, J. Mater. Chem. B, 2025, 13, 1212–1228 RSC
.
- E. Pargoletti and Y. Gogotsi, MXene-Based Nanozymes: Current Challenges and Future Prospects, ChemCatChem, 2025, e00730 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2025 |