Themed collection Bunsentagung 2020: Understanding Dispersion Interactions in Molecular Chemistry

Understanding dispersion interactions in molecular chemistry
This themed collection contains a selection of articles on the topic of Understanding Dispersion Interactions in Molecular Chemistry.
Phys. Chem. Chem. Phys., 2021,23, 8960-8961
https://doi.org/10.1039/D0CP90285C
Determination of the dispersion forces in the gas phase structures of ionic liquids using exclusively thermodynamic methods
Dispersion interactions in the gas phase ion pairs of ionic liquids determined using purely thermodynamic methods.
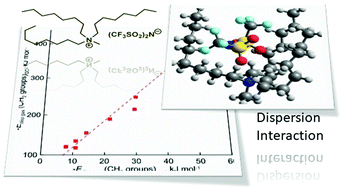
Phys. Chem. Chem. Phys., 2021,23, 7398-7406
https://doi.org/10.1039/D0CP05439A
Structures and internal dynamics of diphenylether and its aggregates with water
We report on a detailed multi-spectroscopic analysis of the structures and internal dynamics of diphenylether and its aggregates with up to three water molecules by employing molecular beam experiments.

Phys. Chem. Chem. Phys., 2020,22, 27966-27978
https://doi.org/10.1039/D0CP04104A
Understanding benzyl alcohol aggregation by chiral modification: the pairing step
Depending on how two enantiomeric benzyl alcohol molecules meet, they either get stuck in a symmetric heterochiral dimer or they synchronize their transient chirality to reach the global minimum structure.
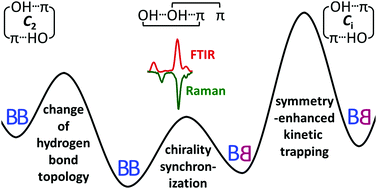
Phys. Chem. Chem. Phys., 2020,22, 25538-25551
https://doi.org/10.1039/D0CP04825A
Vibrational mode frequency correction of liquid water in density functional theory molecular dynamics simulations with van der Waals correction
We develop a frequency correction scheme for the stretch and bending modes of liquid water, which substantially improves the prediction of the vibrational spectra.
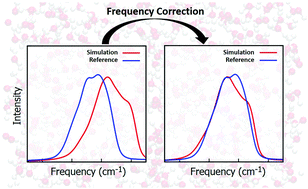
Phys. Chem. Chem. Phys., 2020,22, 12785-12793
https://doi.org/10.1039/C9CP06335H
Evaluation of bismuth-based dispersion energy donors – synthesis, structure and theoretical study of 2-biphenylbismuth(III) derivatives
Intramolecular Bi⋯π arene London dispersion interactions in (biphenyl)3−xBiXx amount to ca. 20 kJ mol−1 with distances of 3.8–4.0 Å.
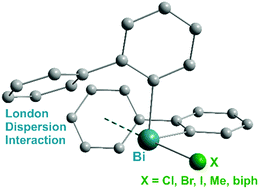
Phys. Chem. Chem. Phys., 2020,22, 10189-10211
https://doi.org/10.1039/C9CP06924K
The influence of intermolecular coupling on electron and ion transport in differently substituted phthalocyanine thin films as electrochromic materials: a chemistry application of the Goldilocks principle
Optimum molecular substitution in organic thin films is established to facilitate electron and ion transport and, thereby, fast reversible electrochromic switching.
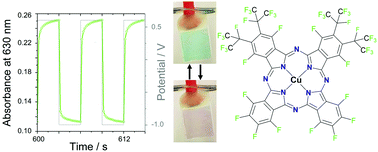
Phys. Chem. Chem. Phys., 2020,22, 7699-7709
https://doi.org/10.1039/C9CP06709D
Including dispersion in density functional theory for adsorption on flat oxide surfaces, in metal–organic frameworks and in acidic zeolites
Contrary to common believe, for eight adsorption cases, neither D3 or TS are an improvement compared to D2 nor van der Waals functionals or dDsC. Only the many body approaches are slightly better than D2(Ne) which uses Ne parameters for Mg2+ ions.
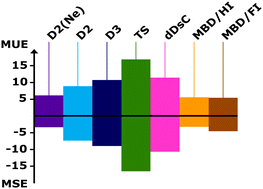
Phys. Chem. Chem. Phys., 2020,22, 7577-7585
https://doi.org/10.1039/D0CP00394H
Dissecting intermolecular interactions in the condensed phase of ibuprofen and related compounds: the specific role and quantification of hydrogen bonding and dispersion forces
Hydrogen bonding and dispersion interaction in liquid ibuprofen is analyzed by thermodynamic methods, infrared spectroscopy and quantum chemistry.
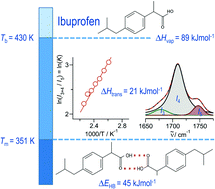
Phys. Chem. Chem. Phys., 2020,22, 4896-4904
https://doi.org/10.1039/C9CP06641A
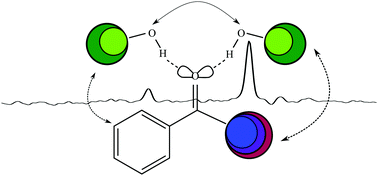
Three-dimensional docking of alcohols to ketones: an experimental benchmark based on acetophenone solvation energy balances
Jet FTIR spectroscopy of acetophenone–methanol balances reveals subtle solvation energy preferences by dispersion-tuning of the alkyl groups.
Phys. Chem. Chem. Phys., 2020,22, 2870-2877
https://doi.org/10.1039/C9CP06128B
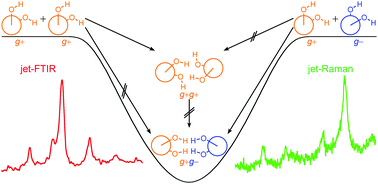
The reduced cohesion of homoconfigurational 1,2-diols
Homochiral encounters of vicinal diols are blocked from relaxing to the heterochiral global minimum dimer structure in supersonic jet expansions.
Phys. Chem. Chem. Phys., 2020,22, 1122-1136
https://doi.org/10.1039/C9CP04943F
Surface grafted agents with various molecular lengths and photochemically active benzophenone moieties
Homologues of benzophenone silane, a covalently graftable, photochemically active surface functionalizing agent, are investigated as surface functionalization agents for both small particles and planar substrates.

Phys. Chem. Chem. Phys., 2020,22, 1774-1783
https://doi.org/10.1039/C9CP05722F
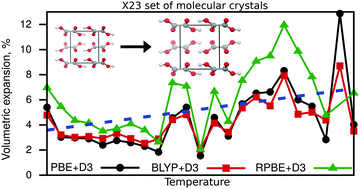
Revised values for the X23 benchmark set of molecular crystals
A revised reference value set for molecular crystals: X23b; new cell volumes and lattice energies including volumetric expansion due to zero-point energy and thermal effects.
Phys. Chem. Chem. Phys., 2019,21, 24333-24344
https://doi.org/10.1039/C9CP04488D
About this collection
Physical Chemistry Chemical Physics (PCCP) is proud to publish the official themed issue of the annual Bunsentagung meeting, organised by the Deutsche Bunsen-Gesellschaft, this year held 1 – 3 April 2020 in Gießen, Germany.
Bunsentagung 2020 focuses on dispersion interactions and their multifold manifestations in chemistry. Dispersion is the driving force for molecular aggregation that plays a key role in the thermodynamic stability of (meta)stable structures, molecular recognition, chemical selectivity through transition-state stabilization, protein folding, enzyme catalysis, and many more. Despite the pioneering work of London and others in the 1930s, our understanding of dispersion interactions only recently has become much better.
The aim of the Bunsentagung and the devoted PCCP themed collection is therefore to highlight the progress in a rapidly developing field encompassing both experiment and theory.