Decellularized organ biomatrices facilitate quantifiable in vitro 3D cancer metastasis models†
Abstract
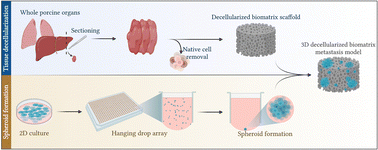
Metastatic cancers are chemoresistant, involving complex interplay between disseminated cancer cell aggregates and the distant organ microenvironment (extracellular matrix and stromal cells). Conventional metastasis surrogates (scratch/wound healing, Transwell migration assays) lack 3D architecture and ECM presence. Metastasis studies can therefore significantly benefit from biomimetic 3D in vitro models recapitulating the complex cascade of distant organ invasion and colonization by collective clusters of cells. We aimed to engineer reproducible and quantifiable 3D models of highly therapy-resistant cancer processes: (i) colorectal cancer liver metastasis; and (ii) breast cancer lung metastasis. Metastatic seeds are engineered using 3D tumor spheroids to recapitulate the 3D aggregation of cancer cells both in the tumor and in circulation throughout the metastatic cascade of many cancers. Metastatic soil was engineered by decellularizing porcine livers and lungs to generate biomatrix scaffolds, followed by extensive materials characterization. HCT116 colorectal and MDA-MB-231 breast cancer spheroids were generated on hanging drop arrays to initiate clustered metastatic seeding into liver and lung biomatrix scaffolds, respectively. Between days 3–7, biomatrix cellular colonization was apparent with increased metabolic activity and the presence of cellular nests evaluated via multiphoton microscopy. HCT116 and MDA-MB-231 cells colonized liver and lung biomatrices, and at least 15% of the cells invaded more than 20 μm from the surface. Engineered metastases also expressed increased signatures of genes associated with the metastatic epithelial to mesenchymal transition (EMT). Importantly, inhibition of matrix metalloproteinase-9 inhibited metastatic invasion into the biomatrix. Furthermore, metastatic nests were significantly more chemoresistant (>3 times) to the anti-cancer drug oxaliplatin, compared to 3D spheroids. Together, our data indicated that HCT116 and MDA-MB-231 spheroids invade, colonize, and proliferate in livers and lungs establishing metastatic nests in 3D settings in vitro. The metastatic nature of these cells was confirmed with functional readouts regarding EMT and chemoresistance. Modeling the dynamic metastatic cascade in vitro has potential to identify therapeutic targets to treat or prevent metastatic progression in chemoresistant metastatic cancers.
- This article is part of the themed collection: Soft matter aspects of cancer


 Please wait while we load your content...
Please wait while we load your content...