Conformational preferences in a series of α-hydroxy ketone derivatives: interplay of conformational energies and lattice cohesive energies†
Abstract
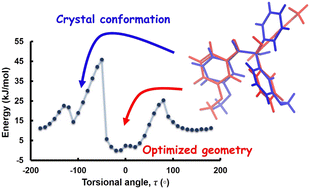
Crystal structures of a series of six α-hydroxy ketone derivatives have been analysed in the context of their molecular conformational preferences. The crystal structures obtained by single crystal X-ray diffraction have been investigated in terms of their inter and intramolecular interactions as well as molecular conformations. A detailed conformational energy analysis of the molecules shows their torsional preferences around the hydroxy and keto functional groups in the isolated state as compared to the crystalline state. A quantitative analysis of the intermolecular interactions in the series of crystal structures reveals that weak C–H⋯π, C–H⋯O and the rare O–H⋯π interactions play significant roles in the crystal packing. An analysis of the interaction topology using the energy framework tool demonstrates the dominance of the dispersion component along with the electrostatic interactions in stabilizing these structures. The estimation of lattice cohesive energies in these structures provides a means to understand the interplay between intramolecular conformational preferences and intermolecular interaction energies and a hierarchy in lattice cohesive energy that is correlated to the observed conformational trends in these structures.
- This article is part of the themed collection: New Talent 2022


 Please wait while we load your content...
Please wait while we load your content...