Soluble and stable symmetric tetrazines as anolytes in redox flow batteries†
Abstract
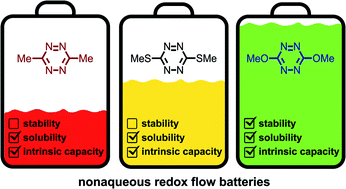
Nonaqueous redox flow batteries are a promising technology for grid-scale energy storage, however, their commercial success relies on identifying redox-active materials that exhibit extreme potentials, high solubilities in all states of charge, and long cycling stabilities. Meeting these requirements has been particularly challenging for molecules capable of storing negative charge. Within this context, tetrazines remain unexplored despite their unique structural properties that enable them to meet some of these challenges. Herein, we prepared symmetric s-tetrazines substituted with methyl, methoxy, and thiomethyl substituents and evaluated their electrochemical properties, solubility, and cycling stability. These studies revealed that highly soluble 3,6-dimethoxy-s-tetrazine undergoes a reversible one-electron reduction to generate a stable (t1/2 > 1240 h) radical anion. When implemented in a lab-scale symmetric flow cell (0.125 M), it exhibited a relatively slow capacity fade of 8% over 50 cycles (17 h). Given their high solubility and cycling stability, we believe that s-tetrazine derivatives should be further explored for nonaqueous redox flow batteries.


 Please wait while we load your content...
Please wait while we load your content...