Photoredox-catalyzed intramolecular cyclopropanation of alkenes with α-bromo-β-keto esters†
Abstract
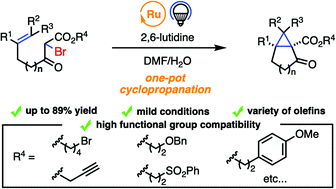
A mild photoredox-catalyzed intramolecular cyclopropanation of alkenes with α-bromo-β-keto esters in an aqueous medium was developed. The sequential reaction process comprising the intramolecular radical addition of α-bromo-β-keto esters to olefins under photoredox catalysis, and subsequent cyclization to form cyclopropane proceeds in one-pot under exceptionally mild conditions at room temperature in the presence of 2,6-lutidine. A broad range of substrates consisting of various alkenes and both base- and acid-sensitive functionalized esters were feasible under the reaction conditions, resulting in a wide range of functionalized bicyclic cyclopropanes.
- This article is part of the themed collections: Synthetic methodology in OBC and Catalysis & biocatalysis in OBC


 Please wait while we load your content...
Please wait while we load your content...