Nalmefene non-enantioselectively targets myeloid differentiation protein 2 and inhibits toll-like receptor 4 signaling: wet-lab techniques and in silico simulations†
Abstract
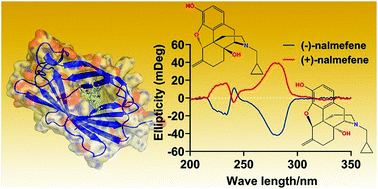
Nalmefene is an opiate derivative having a similar structure to naltrexone. Recent evidence suggests that nalmefene, acting as the innate immune protein toll-like receptor 4 (TLR4) antagonist, effectively reduces the injury of lung ischemia–reperfusion and prevents neuroinflammation. However, the molecular recognition mechanism, especially the enantioselectivity, of nalmefene by the innate immune receptor is not well understood. Herein in vitro assays and in silico simulations were performed to dissect the innate immune recognition of nalmefene at the atomic, molecular, and cellular levels. Biophysical binding experiments and molecular dynamic simulations provide direct evidence that (−)-nalmefene and (+)-nalmefene bind to the hydrophobic cavity of myeloid differentiation protein 2 (MD-2) and behave similarly, which is primarily driven by hydrophobic interactions. The inhibition activity and the calculated binding free energies show that no enantioselectivity was observed during the interaction of nalmefene with MD-2 as well as the inhibition of TLR4 signaling. Interestingly, nalmefene showed ∼6 times better TLR4 antagonisic activity than naltrexone, indicating that the bioisosteric replacement with the methylene group is critical for the molecular recognition of nalmefene by MD-2. In all, this study provides molecular insight into the innate immune recognition of nalmefene, which demonstrates that nalmefene is non-enantioselectively sensed by MD-2.


 Please wait while we load your content...
Please wait while we load your content...