Synthesis, X-ray characterization and theoretical study of all-cis 1,4:2,3:5,8:6,7-tetraepoxynaphthalenes: on the importance of the through-space α-effect†
Abstract
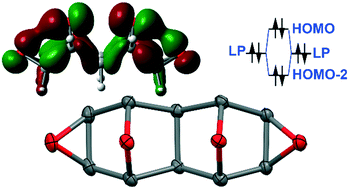
This manuscript reports the synthesis, X-ray characterization and DFT study of a series of all-cis 1,4:2,3:5,8:6,7-tetraepoxynaphthalenes (decahydro-2,6:3,5-diepoxynaphtho[2,3-b:6,7-b′]bis(oxirene) derivatives) obtained by the tandem [4 + 2] cycloaddition between dimethyl acetylenedicarboxylate (DMAD) and three difurans as bis-dienes (the IMDAF reaction). The “pincer”-type adduct formation is controlled by using kinetic conditions. For this work, we were only interested in the synthesis of “pincer”-type adducts to study the formation self-assembled dimers that are governed by quite short C–H⋯O H-bonds. The existence of the “through-space” alpha-effect in the “pincer”-type adducts increases their ability to establish bifurcated C–H⋯O2 H-bonds in the solid state. That is, the close distance between the O-bridge atoms provokes an interesting through space lone pair⋯lone pair (LP⋯LP) interaction that increases the H-bond acceptor ability of the O-atoms. It has been analyzed using DFT calculations, molecular electrostatic potential (MEP) surfaces, quantum theory of atoms-in-molecules (QTAIM) and noncovalent interaction plot (NCIplot) index computational tools. This is the second study of the literature where “alpha-effect” hydrogen bonds (AEHBs) are described and studied. The novelty of the present work resides in the exhaustive epoxidation of the double bonds in the resulting “pincer”-type adducts that increases the number of possible H-bond acceptors in the target vicinal tetra-epoxynaphthalenes. The X-ray analyses of the three synthesized tetra-epoxides reveal that the AEHBs are predominant in the solid phase.
- This article is part of the themed collection: Supramolecular & Polymorphism


 Please wait while we load your content...
Please wait while we load your content...