Recent advances in β-l-rhamnosylation
Abstract
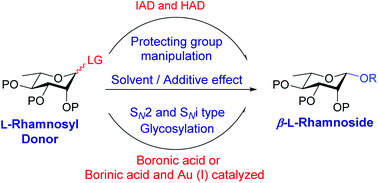
L-Rhamnose forms the key components of important antigenic oligo- and polysaccharides of a variety of pathogens. Obtaining 1,2-cis stereoselectivity in the glycosylation of L-rhamnoside is quite challenging due to the unavailability of neighboring group participation and disfavoring of the anomeric effect and stereoelectronic effect of the substituents on the C-2 axial position. Nevertheless, various methodologies have been developed exploiting diverse pathways for obtaining β-stereoselectivity in the glycosylation of L-rhamnose. This review describes the recent advances in β-L-rhamnosylation and its applications in the total synthesis of β-L-rhamnose-containing biologically important oligosaccharides.
- This article is part of the themed collections: Chemical Biology in OBC and Glycosylation: New methodologies and applications


 Please wait while we load your content...
Please wait while we load your content...