The decisive role of 4f-covalency in the structural direction and oxidation state of XPrO compounds (X: group 13 to 17 elements)†
Abstract
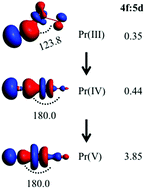
Lanthanide oxo compounds are of vital importance in lanthanide chemistry, as well as in environmental and materials sciences. Praseodymium, as an exceptional element in lanthanides which can form a +V formal oxidation state (OSf) besides the dominant +III among the 4f-block element, displays the significant participation of the Pr 4f orbitals in bonding interactions which is commonly crucial in stabilizing the high oxidation state of Pr in PrO2+ and NPrO species. Here, we report a systematic theoretical study on the structures and stabilities of a series of XPrO (X: B, Al, C, Si, N, P, As, O, S, F, Cl) compounds along with [XPrO]+ cation (X: O, S) and [X3PrO] complexes (X: F and Cl). This work reveals that Pr is able to achieve the lowest and highest OSf and the OSf exhibits periodic variation from +I in BOPr and AlOPr to +II in SiOPr to +III in CPrO, FPrO, ClPrO and AsPrO to +IV in OPrO and SPrO and even to +V in NPrO, [OPrO]+, [SPrO]+, F3PrO and Cl3PrO. We found that the molecular structures are correlated to the Pr oxidation state due to the highly important 4f orbital in the chemical bonding of the high oxidation state compounds. Thus, not only the electronegativity of the ligand but also the quasi-degenerate Pr valence 4f orbitals, namely energetic covalency, control the oxidation state and play a fundamental role in affecting the electronic structural stability of Pr(V) compounds as well. This work demonstrates the structurally directing role of the f-orbital in the formation of the linear structure and is constructive for achieving the higher oxidation state of a given element by tuning the ligand.
- This article is part of the themed collection: Quantum Theory: The Challenge of Transition Metal Complexes


 Please wait while we load your content...
Please wait while we load your content...